Milk Transmission of Mammalian Retroviruses
Abstract
1. Introduction into Breast Milk Transmission of Retroviruses
2. How Is Breast Milk Produced?
3. How Is Breast Milk Composed in Different Species?
3.1. Proteins
3.2. Fat
3.3. Lactose
3.4. Oligosaccharides
3.5. Cells
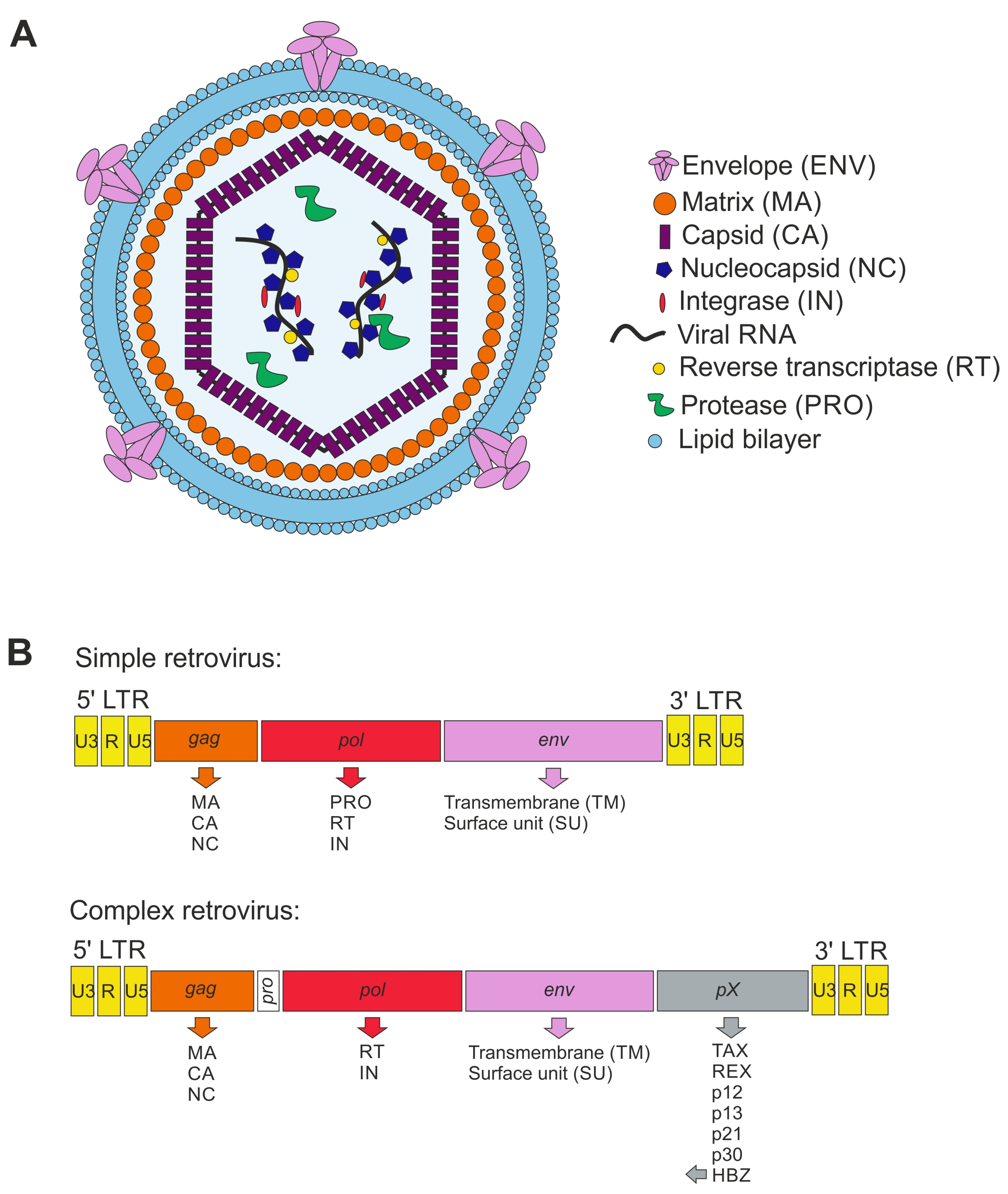
4. Brief Overview of Retroviruses
4.1. Taxonomy
4.2. Retroviral Particles
4.3. Genome Organization
5. Which Animal Retroviruses from the Family Orthoretrovirinae Are Transmitted by Breast Milk?
5.1. Betaretroviruses (Non-Human)
5.1.1. Jaagsiekte Sheep Retrovirus (JSRV)
5.1.2. Mason–Pfizer Monkey Virus (MPMV)
| Virus | Natural Host | Transmission Routes | Site of Infection | Refs. |
|---|---|---|---|---|
| Jaagsiekte sheep retrovirus (JSRV) | Sheep, rarely goats | Inhalation of the virus;maternal route (milk) | Type II pneumocytes; somatic cells in colostrum and milk; white blood cells, lymph nodes and other lymphoid organs; bronchioalveolar epithelial cells; respiratory tract | [72,76,77,86,87,88,89] |
| Mason–Pfizer monkey virus (MPMV) | Rhesus macaques | Horizontal transmission | Infectious virus in saliva and milk | [80,83,84,85,90] |
| Mouse mammary tumor virus (MMTV) | Mice | Milk through intestinal epithelium | Dendritic cells, B lymphocytes in Peyer’s patches; T lymphocytes; epithelial tissues, including salivary gland, kidney, seminal vesicle, epididymis, and testis | [91,92,93,94,95,96,97,98,99,100] |
| Enzootic nasal tumor virus (ENTV) | Sheep and goats | Horizontal transmission; airborne route; nasal secretions; transuterine and oral transmission cannot be excluded | Nasal fluids; nasal epithelial cells; Bowman glands | [101,102,103,104,105,106] |
5.1.3. Mouse Mammary Tumor Virus (MMTV)
5.1.4. Enzootic Nasal Tumor Virus (ENTV)
5.2. Gammaretroviruses (Non-Human)
5.2.1. Feline Leukemia Virus (FeLV)
| Virus | Natural Host | Transmission Routes | Site of Infection | Refs. |
|---|---|---|---|---|
| Feline leukemia virus (FeLV) | Domestic cats, cats | Horizontal transmission; friendly contacts, biting, mutual grooming, shared use of litter boxes and feeding dishes; vertical transmission; milk; from queen to kitten | Lymphocytes and monocytes in lymphoid tissue; neutrophils; saliva, feces, urine, and milk; mucosa of oropharynx; tonsils and lymph nodes; bone marrow | [115,117,118,119,120,121,122,124,125] |
| Gibbon ape leukemia virus (GaLV) | Captive gibbons | Transmission not fully understood | Neoplastic lymphocytic infiltration in lymph nodes, heart, liver, salivary glands, mesentery, kidney, ureters, pituitary, and choroid of eye; bone marrow | [126,127] |
| Koala retrovirus (KoRV) | Captive and free-living koalas | Horizontal and vertical transmission; dam–joey interactions with ingested milk, pap and/or infected fluids during perinatal period and parturition; sexual transmission not known | Unknown; no active virus recovered in milk | [126,128,129,130] |
| Murine leukemia virus (MLV) | Mice | Vertical and horizontal transmission; breast milk; between fighting mice | Macrophages (virus capture); T and B cells; external secretions, saliva, semen, uterine secretions; Peyer’s patch (small intestine) | [131,132,133,134] |
5.2.2. Gibbon Ape Leukemia Virus (GaLV)
5.2.3. Koala Retrovirus (KoRV)
5.2.4. Murine Leukemia Virus (MLV)
5.3. Deltaretroviruses (Human and Non-Human)
5.3.1. Bovine Leukemia Virus (BLV)
5.3.2. Human T Cell Lymphotropic Virus (HTLV)
5.3.3. Simian T-Lymphotropic Virus (STLV)
| Virus | Natural Host | Transmission Routes | Site of Infection | Ref. |
|---|---|---|---|---|
| Bovine leukemia virus (BLV) | Cattle, water buffalo, capybaras; experimental: sheep | Horizontal and vertical transmission; nasal secretions and saliva; blood; tissue; fluids around birth; colostrum and milk; transplacental; direct contact; possible transmission via insects | B lymphocytes; various immune cell populations such as CD5+ IgM+ and CD5− IgM+ B cells; CD2+, CD3+, CD4+, and CD8+; monocytes and granulocytes in peripheral blood and lymphoid tissues | [141,143,144,147,148,149,150,151,152,153,154,155,157,158,201] |
| Human T-lymphotropic virus type 1 (HTLV-1) | Human | Vertical and horizontal transmission; sexual contact; contaminated blood products; breast milk | CD4+ and CD8+ T cells; dendritic cells; monocytes | [177,202,203,204,205,206,207,208,209,210] |
| Human T-lymphotropic virus type 2 (HTLV-2) | Human | Sexual contact; blood transfusion; breastfeeding; contaminated needles | CD8+ T cells | [181,186] |
| Human T-lymphotropic virus type 3 (HTLV-3) | Human | Unknown | PBMCs, detailed tropism unknown | [187,189] |
| Human T-lymphotropic virus type 4 (HTLV-4) | Human | Probably through hunting of wild gorillas | PBMCs, detailed tropism unknown | [188,189,190] |
| Simian T-lymphotropic virus type 1–4 (STLV-1) | Several non-human primate species, e.g., Papio, Cerocebus, and others; gorillas for STLV-4 | Horizontal and maternal route; sexual contact; aggressive behavior; breastfeeding | CD4+ T cells; CD8+ T cells | [190,191,198,211,212,213,214,215,216,217,218] |
5.4. Lentiviruses
5.4.1. Bovine Immunodeficiency Virus (BIV)
5.4.2. Equine Infectious Anemia Virus (EIAV)
5.4.3. Feline Immunodeficiency Virus (FIV)
5.4.4. Caprine Arthritis Encephalitis Virus (CAEV)
5.4.5. Visna-Maedi Virus (VMV)
5.4.6. Human Immunodeficiency Virus (HIV)
5.4.7. Simian Immunodeficiency Virus (SIV)
| Virus | Natural Host | Transmission Routes | Site of Infection | Refs. |
|---|---|---|---|---|
| Bovine immunodeficiency virus (BIV) | Cattle and buffalo | Vertical transmission; in utero and/or transplacental; blood; exact mode unknown | Macrophages; monocytes; lymphocytes; CD3+, CD4+, and CD8+ T cells; B cells; milk-derived leukocytes; liver; lung; spleen; brain | [156,162,219,220,221,248] |
| Equine infectious anemia virus (EIAV) | Horses; donkeys susceptible | Mechanically on mouth parts of biting flies; whole blood; transplacental, colostrum, milk, semen | Monocytes; macrophages | [222,223,224,225,249,250,251] |
| Feline immunodeficiency virus (FIV) | Domestic cats, lions, leopards, tigers, pumas, snow leopards, jaguars, Pallas’s cats, flat-headed cats, cheetahs, and bobcats | Biting; during mating; semen; vertical transmission; milk; in utero; transmission in the wild possible through sexual routes | CD4+ T cells; monocytes; CD8+ T cells; B cells; saliva, blood, serum, plasma, genital secretions; PBMCs, brain, thymus, bone marrow, mesenteric lymph node, spleen, liver | [116,226,227,228,229,230,252,253,254] |
| Caprine arthritis encephalitis virus (CAEV) | Goats | Vertical transmission; colostrum; direct contact; placentas may represent transmission route; rarely sexual contact | Monocytes; macrophages; semen; genital tract; mammary gland; brain; spinal cord; lung; joints; liver; spleen; lymph node; thyroid follicle; intestinal enterocytes | [233,235,255] |
| Visna-maedi virus (VMV) | Sheep | Colostrum and milk; aerosol transmission; contacts between ewe and lamb, oral transmission via small intestine | Monocytes and macrophages; semen; blood; milk; colostrum; mammary gland; lung; brain | [235,236,237] |
| Human immunodeficiency virus 1 (HIV-1) | Human | Horizontal and vertical transmission; semen; blood and blood products; cervicovaginal and rectal secretions; maternal blood; breast milk | CD4+ T cells; cells of monocyte and macrophage lineage; mucosa; submucosa; draining lymphatics; gut-associated lymphoid tissue; system lymphatic tissue; vagina, ecto- and endocervix; inner foreskin; penile urethra; rectum; upper gastrointestinal tract; bloodstream | [238,256,257,258] |
| Human immunodeficiency virus 2 (HIV-2) | Human | Transmission similar to HIV-1; lower transmission rates | CD4+ T cells | [244] |
| Simian immunodeficiency virus (SIV) | African non-human primates | Sexual routes and aggression are suggested; rare mother-to-infant transmission | Short-lived, activated CD4+ T cells; monocytes; macrophages; dendritic cells | [245,246,247,259,260,261] |
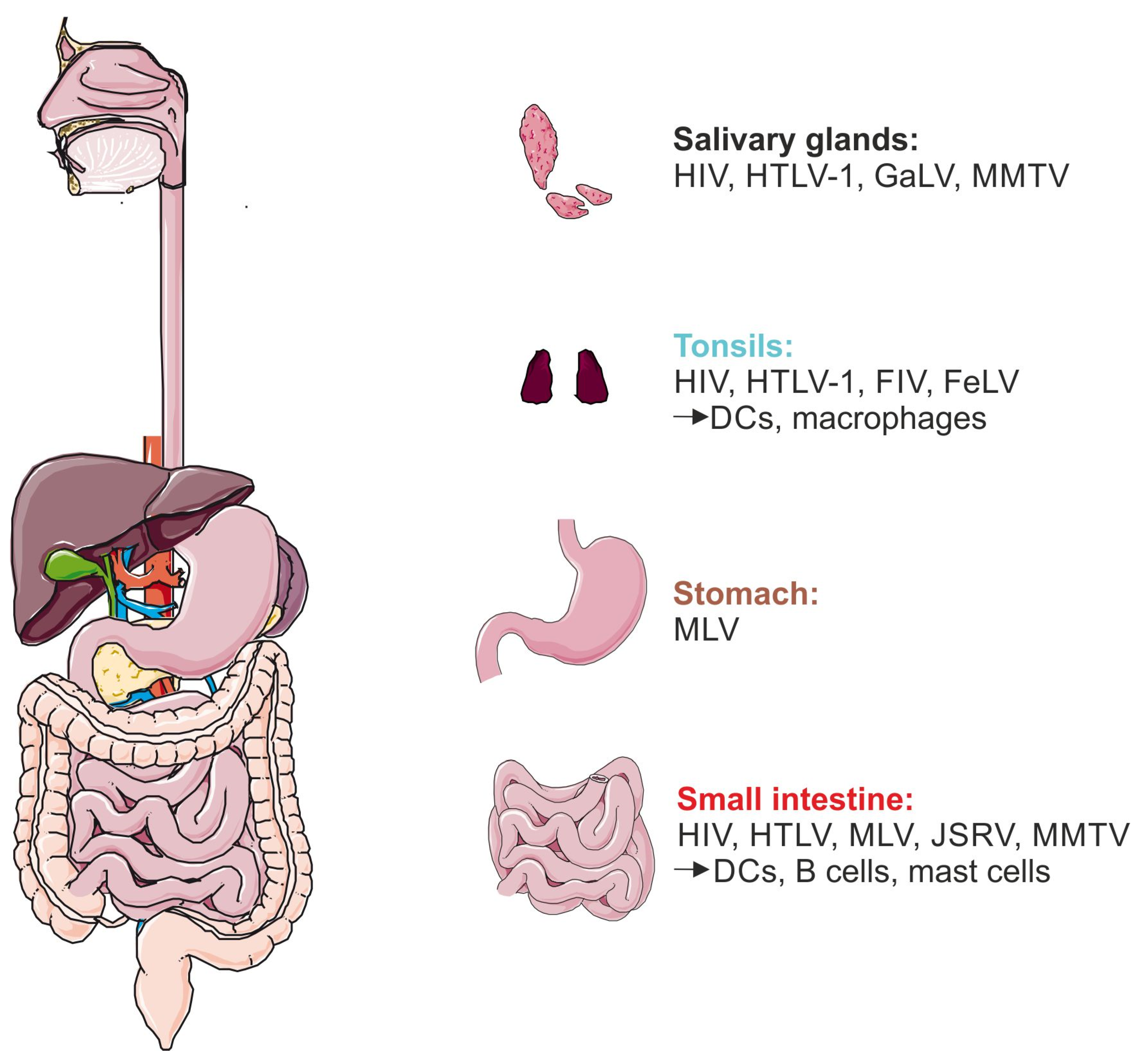
6. How Could Mammalian Retroviruses Enter Infants and Suckling Animals?
6.1. Salivary Glands
6.2. Tonsils
6.3. Pharynx
6.4. Gastrointestinal Tract: Stomach and Small Intestine
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- WHO. Health Topics Breastfeeding. Available online: https://www.who.int/health-topics/breastfeeding#tab=tab_1 (accessed on 24 May 2023).
- Van de Perre, P.; Molès, J.P.; Nagot, N.; Tuaillon, E.; Ceccaldi, P.E.; Goga, A.; Prendergast, A.J.; Rollins, N. Revisiting Koch’s postulate to determine the plausibility of viral transmission by human milk. Pediatr. Allergy Immunol. 2021, 32, 835–842. [Google Scholar] [CrossRef]
- Colt, S.; Garcia-Casal, M.N.; Peña-Rosas, J.P.; Finkelstein, J.L.; Rayco-Solon, P.; Weise Prinzo, Z.C.; Mehta, S. Transmission of Zika virus through breast milk and other breastfeeding-related bodily-fluids: A systematic review. PLoS Neglected Trop. Dis. 2017, 11, e0005528. [Google Scholar] [CrossRef] [PubMed]
- Desgraupes, S.; Hubert, M.; Gessain, A.; Ceccaldi, P.-E.; Vidy, A. Mother-to-Child Transmission of Arboviruses during Breastfeeding: From Epidemiology to Cellular Mechanisms. Viruses 2021, 13, 1312. [Google Scholar] [CrossRef] [PubMed]
- Prendergast, A.J.; Goga, A.E.; Waitt, C.; Gessain, A.; Taylor, G.P.; Rollins, N.; Abrams, E.J.; Lyall, E.H.; Van de Perre, P. Transmission of CMV, HTLV-1, and HIV through breastmilk. Lancet Child Adolesc. Health 2019, 3, 264–273. [Google Scholar] [CrossRef]
- Betts, J.G.; Desaix, P.; Johnson, E.; Johnson, J.; Korol, O.; Kruse, D.; Poe, B.; Wise, J.; Womble, M.; Young, K. Anatomy and Physiology; OpenStax; Rice University: Houston, TX, USA, 2013. [Google Scholar]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. 2013, 60, 49–74. [Google Scholar]
- Nguyen, D.; Parlow, A.; Neville, M. Hormonal regulation of tight junction closure in the mouse mammary epithelium during the transition from pregnancy to lactation. J. Endocrinol. 2001, 170, 347–356. [Google Scholar] [CrossRef]
- Zhu, J.; Dingess, K.A. The functional power of the human milk proteome. Nutrients 2019, 11, 1834. [Google Scholar] [CrossRef] [PubMed]
- Neville, M.C. Anatomy and physiology of lactation. Pediatr. Clin. N. Am. 2001, 48, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Bravi, F.; Wiens, F.; Decarli, A.; Dal Pont, A.; Agostoni, C.; Ferraroni, M. Impact of maternal nutrition on breast-milk composition: A systematic review. Am. J. Clin. Nutr. 2016, 104, 646–662. [Google Scholar] [CrossRef] [PubMed]
- Bryan, D.-L.; Hart, P.H.; Forsyth, K.D.; Gibson, R.A. Immunomodulatory constituents of human milk change in response to infant bronchiolitis. Pediatr. Allergy Immunol. 2007, 18, 495–502. [Google Scholar] [CrossRef]
- Castellote, C.; Casillas, R.; Ramírez-Santana, C.; Pérez-Cano, F.J.; Castell, M.; Moretones, M.G.; López-Sabater, M.C.; Franch, À. Premature delivery influences the immunological composition of colostrum and transitional and mature human milk. J. Nutr. 2011, 141, 1181–1187. [Google Scholar] [CrossRef]
- Dawarkadas, A.; Saha, K.; Mathur, N. A Comparative study of cells and anti-microbial proteins in colostrum of mothers delivering pre- and full-term babies. J. Trop. Pediatr. 1991, 37, 214–219. [Google Scholar] [CrossRef]
- Golan, Y.; Assaraf, Y.G. Genetic and physiological factors affecting human milk production and composition. Nutrients 2020, 12, 1500. [Google Scholar] [CrossRef]
- Hassiotou, F.; Hepworth, A.R.; Williams, T.M.; Twigger, A.-J.; Perrella, S.; Lai, C.T.; Filgueira, L.; Geddes, D.T.; Hartmann, P.E. Breastmilk cell and fat contents respond similarly to removal of breastmilk by the infant. PLoS ONE 2013, 8, e78232. [Google Scholar] [CrossRef]
- Khan, S.; Hepworth, A.R.; Prime, D.K.; Lai, C.T.; Trengove, N.J.; Hartmann, P.E. Variation in fat, lactose, and protein composition in breast milk over 24 hours: Associations with infant feeding patterns. J. Hum. Lact. 2013, 29, 81–89. [Google Scholar] [CrossRef]
- Lazar, K.; Kussmann, T.; Pawelec, G.; Pöschel, S.; Goelz, R.; Hamprecht, K.; Wistuba-Hamprecht, K. Immunomonitoring of human breast milk cells during HCMV-reactivation. Front. Immunol. 2021, 12, 723010. [Google Scholar] [CrossRef]
- Roy, D.; Ye, A.; Moughan, P.J.; Singh, H. Composition, structure, and digestive dynamics of milk from different species—A review. Front. Nutr. 2020, 7, 577759. [Google Scholar] [CrossRef]
- Sharp, J.A.; Lefèvre, C.; Watt, A.; Nicholas, K.R. Analysis of human breast milk cells: Gene expression profiles during pregnancy, lactation, involution, and mastitic infection. Funct. Integr. Genom. 2016, 16, 297–321. [Google Scholar] [CrossRef] [PubMed]
- Witkowska-Zimny, M.; Kaminska-El-Hassan, E. Cells of human breast milk. Cell. Mol. Biol. Lett. 2017, 22, 1–11. [Google Scholar] [CrossRef]
- Hamprecht, K.; Witzel, S.; Maschmann, J.; Dietz, K.; Baumeister, A.; Mikeler, E.; Goelz, R.; Speer, C.P.; Jahn, G. Rapid detection and quantification of cell free cytomegalovirus by a high-speed centrifugation-based microculture assay: Comparison to longitudinally analyzed viral DNA load and pp67 late transcript during lactation. J. Clin. Virol. 2003, 28, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.W.; Haenlein, G.F. Other minor species milk (reindeer, caribou, musk ox, llama, alpaca, moose, elk, and others). In Milk and Dairy Products in Human Nutrition: Production, Composition and Health; John Wiley& Sons: West Sussex, UK, 2013; pp. 644–658. [Google Scholar]
- Park, Y.W.; Haenlein, G.F.; Wendorff, W. Overview of milk of non-bovine mammals. In Handbook of Milk of Non-Bovine Mammals; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 1–9. [Google Scholar]
- Creamer, L.K.; Plowman, J.E.; Liddell, M.J.; Smith, M.H.; Hill, J.P. Micelle stability: κ-casein structure and function. J. Dairy Sci. 1998, 81, 3004–3012. [Google Scholar] [CrossRef] [PubMed]
- El-Hatmi, H.; Jrad, Z.; Salhi, I.; Aguibi, A.; Nadri, A.; Khorchani, T. Comparison of composition and whey protein fractions of human, camel, donkey, goat and cow milk. Mljekarstvo 2015, 65, 159–167. [Google Scholar] [CrossRef]
- Andreas, N.J.; Kampmann, B.; Le-Doare, K.M. Human breast milk: A review on its composition and bioactivity. Early Hum. Dev. 2015, 91, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Atyeo, C.; Alter, G. The multifaceted roles of breast milk antibodies. Cell 2021, 184, 1486–1499. [Google Scholar] [CrossRef]
- Palmeira, P.; Carneiro-Sampaio, M. Immunology of breast milk. Rev. Assoc. Méd. Bras. 2016, 62, 584–593. [Google Scholar] [CrossRef]
- Barrington, G.M.; Parish, S.M. Bovine neonatal immunology. Vet. Clin. N. Am. Food Anim. Pract. 2001, 17, 463–476. [Google Scholar] [CrossRef]
- Hurley, W.L.; Theil, P.K. Perspectives on immunoglobulins in colostrum and milk. Nutrients 2011, 3, 442–474. [Google Scholar] [CrossRef]
- Thum, C.; Wall, C.; Day, L.; Szeto, I.M.; Li, F.; Yan, Y.; Barnett, M.P. Changes in human milk fat globule composition throughout lactation: A review. Front. Nutr. 2022, 9, 835856. [Google Scholar] [CrossRef]
- Saarela, T.; Kokkonen, J.; Koivisto, M. Macronutrient and energy contents of human milk fractions during the first six months of lactation. Acta Paediatr. 2005, 94, 1176–1181. [Google Scholar] [CrossRef]
- Schaafsma, G. Lactose and lactose derivatives as bioactive ingredients in human nutrition. Int. Dairy J. 2008, 18, 458–465. [Google Scholar] [CrossRef]
- Lewinsky, R.H.; Jensen, T.G.; Møller, J.; Stensballe, A.; Olsen, J.; Troelsen, J.T. T −13910 DNA variant associated with lactase persistence interacts with Oct-1 and stimulates lactase promoter activity in vitro. Hum. Mol. Genet. 2005, 14, 3945–3953. [Google Scholar] [CrossRef]
- Ségurel, L.; Bon, C. On the Evolution of Lactase Persistence in Humans. Annu. Rev. Genom. Hum. Genet. 2017, 18, 297–319. [Google Scholar] [CrossRef]
- Tishkoff, S.A.; Reed, F.A.; Ranciaro, A.; Voight, B.F.; Babbitt, C.C.; Silverman, J.S.; Powell, K.; Mortensen, H.M.; Hirbo, J.B.; Osman, M.; et al. Convergent adaptation of human lactase persistence in Africa and Europe. Nat. Genet. 2007, 39, 31–40. [Google Scholar] [CrossRef]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef]
- Soyyılmaz, B.; Mikš, M.H.; Röhrig, C.H.; Matwiejuk, M.; Meszaros-Matwiejuk, A.; Vigsnæs, L.K. The mean of milk: A review of human milk oligosaccharide concentrations throughout lactation. Nutrients 2021, 13, 2737. [Google Scholar] [CrossRef]
- Coppa, G.V.; Pierani, P.; Zampini, L.; Bruni, S.; Carloni, I.; Gabrielli, O. Characterization of oligosaccharides in milk and feces of breast-fed infants by high-performance anion-exchange chromatography. Bioact. Compon. Hum. Milk 2001, 501, 307–314. [Google Scholar] [CrossRef]
- Engfer, M.B.; Stahl, B.; Finke, B.; Sawatzki, G.; Daniel, H. Human milk oligosaccharides are resistant to enzymatic hydrolysis in the upper gastrointestinal tract. Am. J. Clin. Nutr. 2000, 71, 1589–1596. [Google Scholar] [CrossRef]
- Gnoth, M.J.; Kunz, C.; Kinne-Saffran, E.; Rudloff, S. Human milk oligosaccharides are minimally digested In Vitro. J. Nutr. 2000, 130, 3014–3020. [Google Scholar] [CrossRef] [PubMed]
- Bode, L. The functional biology of human milk oligosaccharides. Early Hum. Dev. 2015, 91, 619–622. [Google Scholar] [CrossRef]
- Sánchez, C.; Franco, L.; Regal, P.; Lamas, A.; Cepeda, A.; Fente, C. Breast Milk: A source of functional compounds with potential application in nutrition and therapy. Nutrients 2021, 13, 1026. [Google Scholar] [CrossRef]
- Moore, R.E.; Xu, L.L.; Townsend, S.D. Prospecting human milk oligosaccharides as a defense against viral infections. ACS Infect. Dis. 2021, 7, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Coppa, G.; Pierani, P.; Zampini, L.; Carloni, I.; Carlucci, A.; Gabrielli, O. Oligosaccharides in human milk during different phases of lactation. Acta Paediatr. 1999, 88, 89–94. [Google Scholar] [CrossRef]
- Gabrielli, O.; Zampini, L.; Galeazzi, T.; Padella, L.; Santoro, L.; Peila, C.; Giuliani, F.; Bertino, E.; Fabris, C.; Coppa, G.V. Preterm milk oligosaccharides during the first month of lactation. Pediatrics 2011, 128, e1520–e1531. [Google Scholar] [CrossRef] [PubMed]
- Thurl, S.; Munzert, M.; Henker, J.; Boehm, G.; Müller-Werner, B.; Jelinek, J.; Stahl, B. Variation of human milk oligosaccharides in relation to milk groups and lactational periods. Br. J. Nutr. 2010, 104, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Gopal, P.K.; Gill, H. Oligosaccharides and glycoconjugates in bovine milk and colostrum. Br. J. Nutr. 2000, 84, 69–74. [Google Scholar] [CrossRef]
- Goto, K.; Fukuda, K.; Senda, A.; Saito, T.; Kimura, K.; Glander, K.E.; Hinde, K.; Dittus, W.; Milligan, L.A.; Power, M.L.; et al. Chemical characterization of oligosaccharides in the milk of six species of New and Old world monkeys. Glycoconj. J. 2010, 27, 703–715. [Google Scholar] [CrossRef]
- Kunz, C.; Rudloff, S.; Schad, W.; Braun, D. Lactose-derived oligosaccharides in the milk of elephants: Comparison with human milk. Br. J. Nutr. 1999, 82, 391–399. [Google Scholar] [CrossRef]
- Mudd, A.T.; Salcedo, J.; Alexander, L.S.; Johnson, S.K.; Getty, C.M.; Chichlowski, M.; Berg, B.M.; Barile, D.; Dilger, R.N. Porcine Milk Oligosaccharides and Sialic Acid Concentrations Vary Throughout Lactation. Front. Nutr. 2016, 3, 39. [Google Scholar] [CrossRef]
- Tao, N.; Ochonicky, K.L.; German, J.B.; Donovan, S.M.; Lebrilla, C.B. Structural determination and daily variations of porcine milk oligosaccharides. J. Agric. Food Chem. 2010, 58, 4653–4659. [Google Scholar] [CrossRef]
- Tao, N.; Wu, S.; Kim, J.; An, H.J.; Hinde, K.; Power, M.L.; Gagneux, P.; German, J.B.; Lebrilla, C.B. Evolutionary glycomics: Characterization of milk oligosaccharides in primates. J. Proteome Res. 2011, 10, 1548–1557. [Google Scholar] [CrossRef]
- van Leeuwen, S.S.; Te Poele, E.M.; Chatziioannou, A.C.; Benjamins, E.; Haandrikman, A.; Dijkhuizen, L. Goat milk oligosaccharides: Their diversity, quantity, and functional properties in comparison to human milk oligosaccharides. J. Agric. Food Chem. 2020, 68, 13469–13485. [Google Scholar] [CrossRef]
- Paxson, C.L., Jr.; Cress, C.C. Survival of human milk leukocytes. J. Pediatr. 1979, 94, 61–64. [Google Scholar] [CrossRef]
- Smith, C.; Goldman, A. The cells of human colostrum. I. In Vitro studies of morphology and functions. Pediatr. Res. 1968, 2, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Martin Carli, J.F.; Trahan, G.D.; Jones, K.L.; Hirsch, N.; Rolloff, K.P.; Dunn, E.Z.; Friedman, J.E.; Barbour, L.A.; Hernandez, T.L.; MacLean, P.S.; et al. single cell rna sequencing of human milk-derived cells reveals sub-populations of mammary epithelial cells with molecular signatures of progenitor and mature states: A novel, non-invasive framework for investigating human lactation physiology. J. Mammary Gland. Biol. Neoplasia 2020, 25, 367–387. [Google Scholar] [CrossRef]
- Nyquist, S.K.; Gao, P.; Haining, T.K.; Retchin, M.R.; Golan, Y.; Drake, R.S.; Kolb, K.; Mead, B.E.; Ahituv, N.; Martinez, M.E.; et al. Cellular and transcriptional diversity over the course of human lactation. Proc. Natl. Acad. Sci. USA 2022, 119, 2121720119. [Google Scholar] [CrossRef]
- Trend, S.; de Jong, E.; Lloyd, M.L.; Kok, C.H.; Richmond, P.; Doherty, D.A.; Simmer, K.; Kakulas, F.; Strunk, T.; Currie, A. leukocyte populations in human preterm and term breast milk identified by multicolour flow cytometry. PLoS ONE 2015, 10, e0135580. [Google Scholar] [CrossRef] [PubMed]
- Twigger, A.-J.; Engelbrecht, L.K.; Bach, K.; Schultz-Pernice, I.; Pensa, S.; Stenning, J.; Petricca, S.; Scheel, C.H.; Khaled, W.T. Transcriptional changes in the mammary gland during lactation revealed by single cell sequencing of cells from human milk. Nat. Commun. 2022, 13, 562. [Google Scholar] [CrossRef] [PubMed]
- Cregan, M.D.; Fan, Y.; Appelbee, A.; Brown, M.L.; Klopcic, B.; Koppen, J.; Mitoulas, L.R.; Piper, K.M.; Choolani, M.A.; Chong, Y.-S.; et al. Identification of nestin-positive putative mammary stem cells in human breastmilk. Cell Tissue Res. 2007, 329, 129–136. [Google Scholar] [CrossRef]
- Hassiotou, F.; Beltran, A.; Chetwynd, E.; Stuebe, A.M.; Twigger, A.-J.; Metzger, P.; Trengove, N.; Lai, C.T.; Filgueira, L.; Blancafort, P.; et al. Breastmilk is a novel source of stem cells with multilineage differentiation potential. Stem Cells 2012, 30, 2164–2174. [Google Scholar] [CrossRef]
- Indumathi, S.; Dhanasekaran, M.; Rajkumar, J.; Sudarsanam, D. Exploring the stem cell and non-stem cell constituents of human breast milk. Cytotechnology 2012, 65, 385–393. [Google Scholar] [CrossRef]
- Hassiotou, F.; Geddes, D.T.; Hartmann, P.E. Cells in human milk: State of the science. J. Hum. Lact. 2013, 29, 171–182. [Google Scholar] [CrossRef]
- Sordillo, L.; Shafer-Weaver, K.; DeRosa, D. Immunobiology of the mammary gland. J. Dairy Sci. 1997, 80, 1851–1865. [Google Scholar] [CrossRef]
- Schukken, Y.; Wilson, D.; Welcome, F.; Garrison-Tikofsky, L.; Gonzalez, R. Monitoring udder health and milk quality using somatic cell counts. Vet.-Res. 2003, 34, 579–596. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; García, M.B.M. A 100-Year Review: Advances in goat milk research. J. Dairy Sci. 2017, 100, 10026–10044. [Google Scholar] [CrossRef] [PubMed]
- ICTV. Current ICTV Taxonomy Release. Available online: https://ictv.global/taxonomy (accessed on 24 May 2023).
- Hanson, H.M.; Willkomm, N.A.; Yang, H.; Mansky, L.M. Human Retrovirus Genomic RNA Packaging. Viruses 2022, 14, 1094. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.L.; Maldonado, J.O.; Mueller, J.D.; Zhang, W.; Mansky, L.M. Molecular studies of HTLV-1 replication: An Update. Viruses 2016, 8, 31. [Google Scholar] [CrossRef]
- Hofacre, A.; Fan, H. Jaagsiekte sheep retrovirus biology and oncogenesis. Viruses 2010, 2, 2618–2648. [Google Scholar] [CrossRef]
- DeMartini, J.C.; York, D.F. Retrovirus-associated neoplasms of the respiratory system of sheep and goats: Ovine pulmonary carcinoma and enzootic nasal tumor. Vet.-Clin. N. Am. Food Anim. Pract. 1997, 13, 55–70. [Google Scholar] [CrossRef]
- Maeda, N.; Palmarini, M.; Murgia, C.; Fan, H. Direct transformation of rodent fibroblasts by jaagsiekte sheep retrovirus DNA. Proc. Natl. Acad. Sci. USA 2001, 98, 4449–4454. [Google Scholar] [CrossRef]
- Dungal, N. Experiments with jaagsiekte. Am. J. Pathol. 1946, 22, 737–759. [Google Scholar]
- Borobia, M.; De las Heras, M.; Godino, J.; Ferrer, L.M.; Lacasta, D.; Loste, A.; Ramos, J.J.; Ortín, A. Jaagsiekte sheep retrovirus found in milk macrophages but not in milk lymphocytes or mammary gland epithelia of naturally infected sheep. J. Vet.-Diagn. Investig. 2022, 34, 112–115. [Google Scholar] [CrossRef]
- Borobia, M.; De las Heras, M.; Ramos, J.J.; Ferrer, L.M.; Lacasta, D.; De Martino, A.; Fernández, A.; Loste, A.; Marteles, D.; Ortín, A. Jaagsiekte sheep retrovirus can reach peyer’s patches and mesenteric lymph nodes of lambs nursed by infected mothers. Vet.-Pathol. 2016, 53, 1172–1179. [Google Scholar] [CrossRef]
- Shi, W.; Jia, S.; Guan, X.; Yao, X.; Pan, R.; Huang, X.; Ma, Y.; Wei, J.; Xu, Y. A survey of jaagsiekte sheep retrovirus (JSRV) infection in sheep in the three northeastern provinces of China. Arch. Virol. 2021, 166, 831–840. [Google Scholar] [CrossRef]
- Hemida, M.G.; Alnaeem, A.A. Betaretrovirus infections in dromedary camels in Saudi Arabia. Vet.-Med. Sci. 2022, 8, 1330–1336. [Google Scholar] [CrossRef]
- Chopra, H.C.; Mason, M.M. A new virus in a spontaneous mammary tumor of a rhesus monkey. Cancer Res. 1970, 30, 2081–2086. [Google Scholar]
- Henrickson, R.V.; Maul, D.H.; Osborn, K.G.; Sever, J.L.; Madden, D.L.; Ellingsworth, L.R.; Anderson, J.H.; Lowenstine, L.J.; Gardner, M.B. Epidemic of acquired immunodeficiency in rhesus monkeys. Lancet 1983, 321, 388–390. [Google Scholar] [CrossRef]
- Marx, P.A.; Maul, D.H.; Osborn, K.G.; Lerche, N.W.; Moody, P.; Lowenstine, L.J.; Henrickson, R.V.; Arthur, L.O.; Gilden, R.V.; Gravell, M.; et al. Simian AIDS: Isolation of a Type D Retrovirus and Transmission of the Disease. Science 1984, 223, 1083–1086. [Google Scholar] [CrossRef]
- Fine, D. Mason-pfizer monkey virus and simian aids. Lancet 1984, 323, 335. [Google Scholar] [CrossRef]
- Fine, D.L.; Clarke, G.C.; Arthur, L.O. Characterization of infection and replication of mason-pfizer monkey virus in human cell cultures. J. Gen. Virol. 1979, 44, 457–469. [Google Scholar] [CrossRef]
- Bogden, A.E.; Esber, H.J.; Jensen, E.M. Evidence for the horizontal transmission of infectious Mason-Pfizer monkey virus in the rhesus monkey. Proc. Am. Soc. Microbiol. 1978, 78. [Google Scholar]
- Grego, E.; De Meneghi, D.; Álvarez, V.; Benito, A.A.; Minguijón, E.; Ortín, A.; Mattoni, M.; Moreno, B.; Pérez de Villarreal, M.; Alberti, A.; et al. Colostrum and milk can transmit jaagsiekte retrovirus to lambs. Vet.-Microbiol. 2008, 130, 247–257. [Google Scholar] [CrossRef]
- Caporale, M.; Centorame, P.; Giovannini, A.; Sacchini, F.; Di Ventura, M.; De las Heras, M.; Palmarini, M. Infection of lung epithelial cells and induction of pulmonary adenocarcinoma is not the most common outcome of naturally occurring JSRV infection during the commercial lifespan of sheep. Virology 2005, 338, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Borobia, M.; Ortín, A.; Ferrer, L.M.; Ramos, J.J.; Lacasta, D.; De Las Heras, M. Cells infected with Jaagsiekte sheep retrovirus are detected in the bone marrow of asymptomatic sheep. Can. J. Vet.-Res. 2014, 78, 237–240. [Google Scholar]
- Holland, M.J.; Palmarini, M.; Garcia-Goti, M.; Gonzalez, L.; McKendrick, I.; de las Heras, M.; Sharp, J.M. Jaagsiekte retrovirus is widely distributed both in T and B lymphocytes and in mononuclear phagocytes of sheep with naturally and experimentally acquired pulmonary adenomatosis. J. Virol. 1999, 73, 4004–4008. [Google Scholar] [CrossRef] [PubMed]
- Colcher, D.; Schochetman, G.; Schlom, J. Mason-pfizer monkey virus (MPMV)-horizontally transmitted oncornavirus of rhesus-monkeys. Proc. Am. Assoc. Cancer Res. 1975, 16, 22. [Google Scholar]
- Ross, S.R. Mouse Mammary Tumor Virus Molecular Biology and Oncogenesis. Viruses 2010, 2, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Parisi, F.; Freer, G.; Mazzanti, C.M.; Pistello, M.; Poli, A. Mouse mammary tumor virus (MMTV) and MMTV-LIKE viruses: An in-depth look at a controversial issue. Viruses 2022, 14, 977. [Google Scholar] [CrossRef]
- Dudley, J.P.; Golovkina, T.V.; Ross, S.R. Lessons learned from mouse mammary tumor virus in animal models. ILAR J. 2016, 57, 12–23. [Google Scholar] [CrossRef]
- Golovkina, T.V.; Shlomchik, M.; Hannum, L.; Chervonsky, A. Organogenic role of B lymphocytes in mucosal immunity. Science 1999, 286, 1965–1968. [Google Scholar] [CrossRef]
- Muhlbock, O. Mammary tumor-agent in the sperm of high-cancer-strain male mice. J. Natl. Cancer Inst. 1950, 10, 861–864. [Google Scholar] [CrossRef]
- Smith, G.H. Role of the Milk Agent in disappearance of mammary cancer in C3H/StWi mice. J. Natl. Cancer Inst. 1966, 36, 685–701. [Google Scholar] [CrossRef] [PubMed]
- Tsubura, Y.; Imai, S.; Morimoto, J.; Hilgers, J. Strain difference in the expression of mammary tumor virus antigen in the male genital organs of mice during aging. Gan 1981, 72, 424–429. [Google Scholar] [PubMed]
- Imai, S.; Okumoto, M.; Iwai, M.; Haga, S.; Mori, N.; Miyashita, N.; Moriwaki, K.; Hilgers, J.; Sarkar, N.H. Distribution of mouse mammary tumor virus in Asian wild mice. J. Virol. 1994, 68, 3437–3442. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, A.; Nakano, H.; Yoshimoto, T.; Sayama, K. Biology of mouse mammary tumor virus (MMTV). Cancer Lett. 1995, 90, 3–11. [Google Scholar] [CrossRef]
- Bittner, J.J. Some Possible Effects of nursing on the mammary gland tumor incidence in mice. Science 1936, 84, 162. [Google Scholar] [CrossRef] [PubMed]
- Dirks, C.; Duh, F.-M.; Rai, S.K.; Lerman, M.I.; Miller, A.D. Mechanism of cell entry and transformation by enzootic nasal tumor virus. J. Virol. 2002, 76, 2141–2149. [Google Scholar] [CrossRef]
- De las Heras, M.; Sharp, J.M.; Ferrer, L.M.; García de Jalon, J.A.; Cebrian, L.M. Evidence for a type D-like retrovirus in enzootic nasal tumour of sheep. Vet.-Rec. 1993, 132, 441. [Google Scholar] [CrossRef]
- Cousens, C.; Minguijón, E.; Dalziel, R.G.; Ortin, A.; Garcia, M.; Park, J.; Gonzalez, L.; Sharp, J.M.; de las Heras, M. Complete sequence of enzootic nasal tumor virus, a retrovirus associated with transmissible intranasal tumors of sheep. J. Virol. 1999, 73, 3986–3993. [Google Scholar] [CrossRef]
- De las Heras, M.; Sharp, J.M.; Garcia de Jalon, J.; Dewar, P. Enzootic nasal tumour of goats: Demonstration of a type D-related retrovirus in nasal fluids and tumours. J. Gen. Virol. 1991, 72, 10. [Google Scholar] [CrossRef]
- Rosales Gerpe, M.C.; van Lieshout, L.P.; Domm, J.M.; Ingrao, J.C.; Datu, J.; Walsh, S.R.; Yu, D.L.; Jong, J.; Krell, P.J.; Wootton, S.K. The U3 and Env Proteins of Jaagsiekte Sheep Retrovirus and Enzootic Nasal Tumor Virus Both Contribute to Tissue Tropism. Viruses 2019, 11, 1061. [Google Scholar] [CrossRef]
- de Cecco, B.S.; Lorenzett, M.P.; Henker, L.C.; Weber, M.N.; Moséna, A.C.S.; Baumbach, L.; Canal, C.W.; Driemeier, D.; Pavarini, S.P.; Sonne, L. Detection of enzootic nasal tumor virus (ENTV) in a sheep flock in southern Brazil. Trop. Anim. Health Prod. 2019, 51, 2095–2098. [Google Scholar] [CrossRef]
- Graff, S.; Moore, D.H.; Stanley, W.M.; Randall, H.T.; Haagensen, C.D. Isolation of mouse mammary carcinoma virus. Cancer 1949, 2, 755–762. [Google Scholar] [CrossRef]
- Axel, R.; Schlom, J.; Spiegelman, S. Presence in human breast cancer of RNA homologous to mouse mammary tumour virus RNA. Nature 1972, 235, 32–36. [Google Scholar] [CrossRef]
- Holt, M.; Shevach, E.; Punkosdy, G. Endogenous mouse mammary tumor viruses (Mtv): New roles for an old virus in cancer, infection, and immunity. Front. Oncol. 2013, 3, 287. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lawson, J.S.; Glenn, W.K. Mouse Mammary tumour virus (MMTV) in human breast cancer—The value of bradford hill criteria. Viruses 2022, 14, 721. [Google Scholar] [CrossRef]
- Amarante, M.K.; de Sousa Pereira, N.; Vitiello, G.A.F.; Watanabe, M.A.E. Involvement of a mouse mammary tumor virus (MMTV) homologue in human breast cancer: Evidence for, against and possible causes of controversies. Microb. Pathog. 2019, 130, 283–294. [Google Scholar] [CrossRef]
- De las Heras, M.; Ortín, A.; Cousens, C.; Minguijón, E.; Sharp, J.M. Enzootic nasal adenocarcinoma of sheep and goats. Jaagsiekte Sheep Retrovir. Lung Cancer 2003, 275, 201–223. [Google Scholar] [CrossRef]
- Walsh, S.R.; Stinson, K.J.; Wootton, S.K. Seroconversion of sheep experimentally infected with enzootic nasal tumor virus. BMC Res. Notes 2016, 9, 15. [Google Scholar] [CrossRef]
- Leroux, C.; Mornex, J.F. Retroviral infections in sheep and the associated diseases. Small Rumin. Res. 2008, 76, 68–76. [Google Scholar] [CrossRef]
- Hardy, W.D., Jr.; Hess, P.W.; Essex, M.; Cotter, S.; McClelland, A.J.; MacEwen, G. Horizontal transmission of feline leukemia virus in cats. Bibl. Haematol. 2015, 40, 67–74. [Google Scholar] [CrossRef]
- Hartmann, K. Feline immunodeficiency virus infection: An overview. Vet.-J. 1998, 155, 123–137. [Google Scholar] [CrossRef]
- Hofmann-Lehmann, R.; Hartmann, K. Feline leukaemia virus infection: A practical approach to diagnosis. J. Feline Med. Surg. 2020, 22, 831–846. [Google Scholar] [CrossRef] [PubMed]
- Lutz, H.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Hosie, M.J.; Lloret, A.; et al. Feline leukaemia. ABCD guidelines on prevention and management. J. Feline Med. Surg. 2009, 11, 565–574. [Google Scholar] [CrossRef]
- Pacitti, A.M.; Jarrett, O.; Hay, D. Transmission of feline leukaemia virus in the milk of a non-viraemic cat. Vet. Rec. 1986, 118, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Rojko, J.L.; Hoover, E.A.; Mathes, L.E.; Olsen, R.G.; Schaller, J.P. Pathogenesis of experimental feline leukemia virus infection2. Gynecol. Oncol. 1979, 63, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Rojko, J.L.; Kociba, G.J. Pathogenesis of infection by the feline leukemia virus. J. Am. Vet.-Med. Assoc. 1991, 199, 1305–1310. [Google Scholar]
- Grant, C.K.; Essex, M.; Gardner, M.B.; Hardy, W.D., Jr. Natural feline leukemia virus infection and the immune response of cats of different ages. Cancer Res. 1980, 40, 823–829. [Google Scholar]
- Patel, M.; Carritt, K.; Lane, J.; Jayappa, H.; Stahl, M.; Bourgeois, M. Comparative efficacy of feline leukemia virus (FeLV) inactivated whole-virus vaccine and canarypox virus-vectored vaccine during virulent FeLV challenge and immunosuppression. Clin. Vaccine Immunol. 2015, 22, 798–805. [Google Scholar] [CrossRef]
- Gomeskeller, M.; Gönczi, E.; Grenacher, B.; Tandon, R.; Hofmanlehmann, R.; Lutz, H. Fecal shedding of infectious feline leukemia virus and its nucleic acids: A transmission potential. Vet. Microbiol. 2009, 134, 208–217. [Google Scholar] [CrossRef]
- Gomes-Keller, M.; Gonczi, E.; Tandon, R.; Riondato, F.; Hofmann-Lehmann, R.; Meli, M.L.; Lutz, H. Detection of feline leukemia virus RNA in saliva from naturally infected cats and correlation of PCR results with those of current diagnostic methods. J. Clin. Microbiol. 2006, 44, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Denner, J. Transspecies transmission of gammaretroviruses and the origin of the gibbon ape leukaemia virus (GaLV) and the koala retrovirus (KoRV). Viruses 2016, 8, 336. [Google Scholar] [CrossRef] [PubMed]
- Gallo, R.C.; Gallagher, R.E.; Wong-Staal, F.; Aoki, T.; Markham, P.; Schetters, H.; Ruscetti, F.D.; Valerio, M.; Walling, M.J.; O’Keeffe, R.; et al. Isolation and tissue distribution of type-C virus and viral components from a gibbon ape (Hylobates lar) with lymphocytic leukemia. Virology 1978, 84, 359–373. [Google Scholar] [CrossRef] [PubMed]
- Kayesh, M.E.H.; Hashem, M.A.; Tsukiyama-Kohara, K. Koala retrovirus epidemiology, transmission mode, pathogenesis, and host immune response in koalas (Phascolarctos cinereus): A review. Arch. Virol. 2020, 165, 2409–2417. [Google Scholar] [CrossRef]
- Joyce, B.A.; Blyton, M.D.J.; Johnston, S.D.; Young, P.R.; Chappell, K.J. Koala retrovirus genetic diversity and transmission dynamics within captive koala populations. Proc. Natl. Acad. Sci. USA 2021, 118, e2024021118. [Google Scholar] [CrossRef] [PubMed]
- Kalman, B.; Levigne, H. The Life Cycle of a Koala; Crabtree Publishing Company: New York, NY, USA, 2001. [Google Scholar]
- Portis, J.L.; McAtee, F.J.; Hayes, S.F. Horizontal transmission of murine retroviruses. J. Virol. 1987, 61, 1037–1044. [Google Scholar] [CrossRef]
- Buffett, R.F.; Grace, J.T., Jr.; DiBerardino, L.A.; Mirand, E.A. Vertical transmission of murine leukemia virus. Cancer Res 1969, 29, 588–595. [Google Scholar]
- Pi, R.; Iwasaki, A.; Sewald, X.; Mothes, W.; Uchil, P.D. Murine leukemia virus exploits innate sensing by toll-like receptor 7 in B-1 cells to establish infection and locally spread in mice. J. Virol. 2019, 93, e00930-19. [Google Scholar] [CrossRef]
- Li, F.; Sewald, X.; Jin, J.; Sherer, N.M.; Mothes, W. Murine leukemia virus gag localizes to the uropod of migrating primary lymphocytes. J. Virol. 2014, 88, 10541–10555. [Google Scholar] [CrossRef]
- McKee, J.; Clark, N.; Shapter, F.; Simmons, G. A new look at the origins of gibbon ape leukemia virus. Virus Genes 2017, 53, 165–172. [Google Scholar] [CrossRef]
- Waugh, C.A.; Hanger, J.; Loader, J.; King, A.; Hobbs, M.; Johnson, R.; Timms, P. Infection with koala retrovirus subgroup B (KoRV-B), but not KoRV-A, is associated with chlamydial disease in free-ranging koalas (Phascolarctos cinereus). Sci. Rep. 2017, 7, 134. [Google Scholar] [CrossRef]
- Xu, W.; Stadler, C.K.; Gorman, K.; Jensen, N.; Kim, D.; Zheng, H.; Tang, S.; Switzer, W.M.; Pye, G.W.; Eiden, M.V. An exogenous retrovirus isolated from koalas with malignant neoplasias in a US zoo. Proc. Natl. Acad. Sci. USA 2013, 110, 11547–11552. [Google Scholar] [CrossRef] [PubMed]
- Hashem, M.A.; Maetani, F.; Kayesh, M.E.H.; Eiei, T.; Mochizuki, K.; Ito, A.; Sakurai, H.; Asai, T.; Tsukiyama-Kohara, K. Transmission of koala retrovirus from parent koalas to a joey in a Japanese zoo. J. Virol. 2020, 94, e00019-20. [Google Scholar] [CrossRef] [PubMed]
- Rein, A. Murine leukemia virus: Objects and organisms. Adv. Virol. 2011, 2011, 403419. [Google Scholar] [CrossRef]
- Bartlett, P.C.; Sordillo, L.M.; Byrem, T.M.; Norby, B.; Grooms, D.L.; Swenson, C.L.; Zalucha, J.; Erskine, R.J. Options for the control of bovine leukemia virus in dairy cattle. J. Am. Vet. Med. Assoc. 2014, 244, 914–922. [Google Scholar] [CrossRef]
- Ruiz, V.; Porta, N.G.; Lomónaco, M.; Trono, K.; Alvarez, I. Bovine leukemia virus infection in neonatal calves. risk factors and control measures. Front. Vet.-Sci. 2018, 5, 267. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Animal Health and Welfare (AHAW). Enzootic bovine leukosis. EFSA J. 2015, 13, 4188. [Google Scholar] [CrossRef]
- Kuczewski, A.; Orsel, K.; Barkema, H.W.; Mason, S.; Erskine, R.; van der Meer, F. Invited review: Bovine leukemia virus—Transmission, control, and eradication. J. Dairy Sci. 2021, 104, 6358–6375. [Google Scholar] [CrossRef]
- Gillet, N.; Florins, A.; Boxus, M.; Burteau, C.; Nigro, A.; Vandermeers, F.; Balon, H.; Bouzar, A.-B.; Defoiche, J.; Burny, A.; et al. Mechanisms of leukemogenesis induced by bovine leukemia virus: Prospects for novel anti-retroviral therapies in human. Retrovirology 2007, 4, 18. [Google Scholar] [CrossRef]
- Aida, Y.; Murakami, H.; Takahashi, M.; Takeshima, S.-N. Mechanisms of pathogenesis induced by bovine leukemia virus as a model for human T-cell leukemia virus. Front. Microbiol. 2013, 4, 328. [Google Scholar] [CrossRef]
- Forlani, G.; Shallak, M.; Accolla, R.S.; Romanelli, M.G. HTLV-1 Infection and pathogenesis: New Insights from cellular and animal models. Int. J. Mol. Sci. 2021, 22, 8001. [Google Scholar] [CrossRef]
- Evermann, J.F.; Digiacomo, R.F.; Hopkins, S.G. Bovine leukosis virus: Understanding viral transmission and the methods of control. Vet. Med. 1987, 27, 1051–1058. [Google Scholar]
- Hopkins, S.G.; DiGiacomo, R.F. Natural transmission of bovine leukemia virus in dairy and beef cattle. Vet.- Clin. North Am. Food Anim. Pract. 1997, 13, 107–128. [Google Scholar] [CrossRef] [PubMed]
- Kohara, J.; Takeuchi, M.; Hirano, Y.; Sakurai, Y.; Takahashi, T. Vector control efficacy of fly nets on preventing bovine leukemia virus transmission. J. Vet.-Med. Sci. 2018, 80, 1524–1527. [Google Scholar] [CrossRef]
- Kono, Y.; Sentsui, H.; Arai, K.; Ishida, H.; Irishio, W. Contact transmission of bovine leukemia virus under insect-free conditions. Nihon Juigaku Zasshi 1983, 45, 799–802. [Google Scholar] [CrossRef] [PubMed]
- Panei, C.J.; Larsen, A.E.; Fuentealba, N.A.; Metz, G.E.; Echeverría, M.G.; Galosi, C.M.; Valera, A.R. Study of horn flies as vectors of bovine leukemia virus. Open Vet.-J. 2019, 9, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Sajiki, Y.; Konnai, S.; Nishimori, A.; Okagawa, T.; Maekawa, N.; Goto, S.; Nagano, M.; Kohara, J.; Kitano, N.; Takahashi, T.; et al. Intrauterine infection with bovine leukemia virus in pregnant dam with high viral load. J. Vet.-Med. Sci. 2017, 79, 2036–2039. [Google Scholar] [CrossRef][Green Version]
- Yuan, Y.; Kitamura-Muramatsu, Y.; Saito, S.; Ishizaki, H.; Nakano, M.; Haga, S.; Matoba, K.; Ohno, A.; Murakami, H.; Takeshima, S.-N.; et al. Detection of the BLV provirus from nasal secretion and saliva samples using BLV-CoCoMo-qPCR-2: Comparison with blood samples from the same cattle. Virus Res. 2015, 210, 248–254. [Google Scholar] [CrossRef]
- Stott, M.L.; Thurmond, M.C.; Dunn, S.J.; Osburn, B.I.; Stott, J.L.; Lembo, D.; Angeretti, A.; Foresta, P.; Gribaudo, G.; Gariglio, M.; et al. Integrated bovine leukosis proviral DNA in T helper and T cytotoxic/suppressor lymphocytes. J. Gen. Virol. 1991, 72, 307–315. [Google Scholar] [CrossRef]
- Williams, D.L.; Barta, O.; Amborski, G.F. Molecular studies of T-lymphocytes from cattle infected with bovine leukemia virus. Vet.-Immunol. Immunopathol. 1988, 19, 307–323. [Google Scholar] [CrossRef]
- Wu, D.; Murakami, K.; Morooka, A.; Jin, H.; Inoshima, Y.; Sentsui, H. In vivo transcription of bovine leukemia virus and bovine immunodeficiency-like virus. Virus Res. 2003, 97, 81–87. [Google Scholar] [CrossRef]
- Wu, D.; Takahashi, K.; Murakami, K.; Tani, K.; Koguchi, A.; Asahina, M.; Goryo, M.; Aida, Y.; Okada, K. B-1a, B-1b and conventional B cell lymphoma from enzootic bovine leukosis. Vet.-Immunol. Immunopathol. 1996, 55, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Panei, C.J.; Takeshima, S.-N.; Omori, T.; Nunoya, T.; Davis, W.C.; Ishizaki, H.; Matoba, K.; Aida, Y. Estimation of bovine leukemia virus (BLV) proviral load harbored by lymphocyte subpopulations in BLV-infected cattle at the subclinical stage of enzootic bovine leucosis using BLV-CoCoMo-qPCR. BMC Vet.-Res. 2013, 9, 95. [Google Scholar] [CrossRef]
- Burny, A.; Cleuter, Y.; Kettmann, R.; Mammerickx, M.; Marbaix, G.; Portetelle, D.; Van den Broeke, A.; Willems, L.; Thomas, R. Bovine leukaemia: Facts and hypotheses derived from the study of an infectious cancer. Cancer Surv. 1987, 6, 139–159. [Google Scholar] [PubMed]
- Olson, C.; Miller, J. Enzootic Bovine Leukosis and Bovine Leukemia Virus; Martinus Nijhoff Publishing: Leiden, The Netherlands, 1987. [Google Scholar]
- Willems, L.; Burny, A.; Dangoisse, O.; Collete, D.; Dequiedt, F.; Gatot, J.-S.; Kerkhofs, P.; Lefebvre, L.; Merezak, C.; Portetelle, D. Bovine leukemia virus as a model for human T-cell leukemia virus. Curr. Top. Virol. 1999, 73, 8160–8166. [Google Scholar]
- Meas, S.; Usui, T.; Ohashi, K.; Sugimoto, C.; Onuma, M. Vertical transmission of bovine leukemia virus and bovine immunodeficiency virus in dairy cattle herds. Vet.-Microbiol. 2002, 84, 275–282. [Google Scholar] [CrossRef]
- Rodríguez, S.M.; Florins, A.; Gillet, N.; De Brogniez, A.; Sánchez-Alcaraz, M.T.; Boxus, M.; Boulanger, F.; Gutiérrez, G.; Trono, K.; Alvarez, I.; et al. Preventive and Therapeutic Strategies for Bovine Leukemia Virus: Lessons for HTLV. Viruses 2011, 3, 1210–1248. [Google Scholar] [CrossRef]
- Suarez Archilla, G.; Gutiérrez, G.; Camussone, C.; Calvinho, L.; Abdala, A.; Alvarez, I.; Petersen, M.; Franco, L.; Destefano, G.; Monti, G.; et al. A safe and effective vaccine against bovine leukemia virus. Front. Immunol. 2022, 13, 980514. [Google Scholar] [CrossRef]
- Buehring, G.C.; Sans, H.M. Breast Cancer Gone Viral? Review of Possible Role of Bovine Leukemia Virus in Breast Cancer, and Related Opportunities for Cancer Prevention. Int. J. Environ. Res. Public Health 2019, 17, 209. [Google Scholar] [CrossRef]
- Gillet, N.A.; Willems, L. Whole genome sequencing of 51 breast cancers reveals that tumors are devoid of bovine leukemia virus DNA. Retrovirology 2016, 13, 15. [Google Scholar] [CrossRef]
- Khatami, A.; Pormohammad, A.; Farzi, R.; Saadati, H.; Mehrabi, M.; Kiani, S.J.; Ghorbani, S. Bovine leukemia virus (BLV) and risk of breast cancer: A systematic review and meta-analysis of case-control studies. Infect. Agents Cancer 2020, 15, 41. [Google Scholar] [CrossRef]
- Yamanaka, M.P.; Saito, S.; Hara, Y.; Matsuura, R.; Takeshima, S.-N.; Hosomichi, K.; Matsumoto, Y.; Furuta, R.A.; Takei, M.; Aida, Y. No evidence of bovine leukemia virus proviral DNA and antibodies in human specimens from Japan. Retrovirology 2022, 19, 7. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.; Tagaya, Y.; Gallo, R. Time to eradicate HTLV-1: An open letter to WHO. Lancet 2018, 391, 1893–1894. [Google Scholar] [CrossRef] [PubMed]
- Gessain, A.; Cassar, O. Epidemiological Aspects and World Distribution of HTLV-1 Infection. Front. Microbiol. 2012, 3, 388. [Google Scholar] [CrossRef] [PubMed]
- Poiesz, B.J.; Ruscetti, F.W.; Reitz, M.S.; Kalyanaraman, V.S.; Gallo, R.C. Isolation of a new type C retrovirus (HTLV) in primary uncultured cells of a patient with Sézary T-cell leukaemia. Nature 1981, 294, 268–271. [Google Scholar] [CrossRef] [PubMed]
- Schierhout, G.; McGregor, S.; Gessain, A.; Einsiedel, L.; Martinello, M.; Kaldor, J. Association between HTLV-1 infection and adverse health outcomes: A systematic review and meta-analysis of epidemiological studies. Lancet Infect. Dis. 2020, 20, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.; Thoma-Kress, A.K. Molecular Mechanisms of HTLV-1 Cell-to-Cell Transmission. Viruses 2016, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Itabashi, K.; Miyazawa, T. Mother-to-Child Transmission of Human T-Cell Leukemia virus type 1: Mechanisms and nutritional strategies for prevention. Cancers 2021, 13, 4100. [Google Scholar] [CrossRef]
- Itabashi, K.; Miyazawa, T.; Uchimaru, K. How Can We Prevent Mother-to-Child Transmission of HTLV-1? Int. J. Mol. Sci. 2023, 24, 6961. [Google Scholar] [CrossRef]
- Paiva, A.M.; Assone, T.; Haziot, M.E.J.; Smid, J.; Fonseca, L.A.M.; Luiz, O.D.C.; de Oliveira, A.C.P.; Casseb, J. Risk factors associated with HTLV-1 vertical transmission in Brazil: Longer breastfeeding, higher maternal proviral load and previous HTLV-1-infected offspring. Sci. Rep. 2018, 8, 7742. [Google Scholar] [CrossRef]
- Rosadas, C.; Taylor, G.P. Mother-to-child HTLV-1 transmission: Unmet research needs. Front. Microbiol. 2019, 10, 999. [Google Scholar] [CrossRef]
- Millen, S.; Thoma-Kress, A.K. Milk Transmission of HTLV-1 and the need for innovative prevention strategies. Front. Med. 2022, 9, 867147. [Google Scholar] [CrossRef] [PubMed]
- Barr, R.S.; Drysdale, S.B.; Boullier, M.; Lyall, H.; Cook, L.; Collins, G.P.; Kelly, D.F.; Phelan, L.; Taylor, G.P. A Review of the prevention of mother-to-child transmission of human T-Cell lymphotrophic virus type 1 (HTLV-1) with a proposed management algorithm. Front. Med. 2022, 9, 941647. [Google Scholar] [CrossRef] [PubMed]
- WHO. Human T-Lymphotropic Virus Type 1: Technical Report. [Internet]. 2021. Available online: https://www.who.int/publications/i/item/9789240020221 (accessed on 26 January 2022).
- Ciminale, V.; Rende, F.; Bertazzoni, U.; Romanelli, M.G. HTLV-1 and HTLV-2: Highly similar viruses with distinct oncogenic properties. Front. Microbiol. 2014, 5, 398. [Google Scholar] [CrossRef]
- Heneine, W.; Woods, T.; Green, D.; Fukuda, K.; Giusti, R.; Castillo, L.; Armien, B.; Gracia, F.; Kaplan, J.E. Detection of HTLV-II in breastmilk of HTLV-II infected mothers. Lancet 1992, 340, 1157–1158. [Google Scholar] [CrossRef]
- Nyambi, P.N.; Ville, Y.; Louwagie, J.; Bedjabaga, I.; Glowaczower, E.; Peeters, M.; Kerouedan, D.; Dazza, M.; Larouzé, B.; van der Groen, G.; et al. Mother-to-Child Transmission of human T-Cell Lymphotropic virus types I and II (HTLV-I/II) in Gabon: A prospective follow-up of 4 years. J. Acquir. Immune. Defic. Syndr. Hum. Retrovirol. 1996, 12, 187–192. [Google Scholar] [CrossRef]
- Roucoux, D.F.; Murphy, E.L. The epidemiology and disease outcomes of human T-lymphotropic virus type II. Aids Rev. 2004, 6, 144–154. [Google Scholar]
- Biswas, H.H.; Kaidarova, Z.; Garratty, G.; Gibble, J.W.; Newman, B.H.; Smith, J.W.; Ziman, A.; Fridey, J.L.; Sacher, R.A.; Murphy, E.L. Increased All-Cause and Cancer Mortality in HTLV-II Infection. J. Acquir. Immune. Defic. Syndr. 2010, 54, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Khabbaz, R.F.; Onorato, I.M.; Cannon, R.O.; Hartley, T.M.; Roberts, B.; Hosein, B.; Kaplan, J.E. Seroprevalence of HTLV-I and HTLV-II among intravenous drug users and persons in clinics for sexually transmitted diseases. N. Engl. J. Med. 1992, 326, 375–380. [Google Scholar] [CrossRef]
- Calattini, S.; Chevalier, S.A.; Duprez, R.; Bassot, S.; Froment, A.; Mahieux, R.; Gessain, A. Discovery of a new human T-cell lymphotropic virus (HTLV-3) in Central Africa. Retrovirology 2005, 2, 30. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, N.D.; Heneine, W.; Carr, J.K.; Garcia, A.D.; Shanmugam, V.; Tamoufe, U.; Torimiro, J.N.; Prosser, A.T.; LeBreton, M.; Mpoudi-Ngole, E.; et al. Emergence of unique primate T-lymphotropic viruses among central African bushmeat hunters. Proc. Natl. Acad. Sci. USA 2005, 102, 7994–7999. [Google Scholar] [CrossRef]
- Mahieux, R.; Gessain, A. HTLV-3/STLV-3 and HTLV-4 viruses: Discovery, epidemiology, serology and molecular aspects. Viruses 2011, 3, 1074–1090. [Google Scholar] [CrossRef] [PubMed]
- LeBreton, M.; Switzer, W.M.; Djoko, C.F.; Gillis, A.; Jia, H.; Sturgeon, M.M.; Shankar, A.; Zheng, H.; Nkeunen, G.; Tamoufe, U.; et al. A gorilla reservoir for human T-lymphotropic virus type 4. Emerg. Microbes Infect. 2014, 3, e7. [Google Scholar] [CrossRef] [PubMed]
- Hayami, M.; Komuro, A.; Nozawa, K.; Shotake, T.; Ishikawa, K.; Yamamoto, K.; Ishida, T.; Honjo, S.; Hinuma, Y. Prevalence of antibody to adult T-cell leukemia virus-associated antigens (ATLA) in Japanese monkeys and other non-human primates. Int. J. Cancer 1984, 33, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, I.; Yoshimoto, S.; Fujishita, M.; Taguchi, H.; Kubonishi, I.; Niiya, K.; Minezawa, M. Natural adult t-cell Leukaemia virus infection in Japanese monkeys. Lancet 1982, 320, 658. [Google Scholar] [CrossRef]
- Enose-Akahata, Y.; Caruso, B.; Haner, B.; Charlip, E.; Nair, G.; Massoud, R.; Billioux, B.J.; Ohayon, J.; Switzer, W.M.; Jacobson, S. Development of neurologic diseases in a patient with primate T lymphotropic virus type 1 (PTLV-1). Retrovirology 2016, 13, 56. [Google Scholar] [CrossRef]
- Mossoun, A.; Calvignac-Spencer, S.; Anoh, A.E.; Pauly, M.S.; Driscoll, D.A.; Michel, A.O.; Nazaire, L.G.; Pfister, S.; Sabwe, P.; Thiesen, U.; et al. Bushmeat Hunting and zoonotic transmission of simian t-lymphotropic virus 1 in tropical west and central Africa. J. Virol. 2017, 91, e02479-16. [Google Scholar] [CrossRef]
- Jégado, B.; Kashanchi, F.; Dutartre, H.; Mahieux, R. STLV-1 as a model for studying HTLV-1 infection. Retrovirology 2019, 16, 41. [Google Scholar] [CrossRef]
- Calattini, S.; Chevalier, S.A.; Duprez, R.; Afonso, P.; Froment, A.; Gessain, A.; Mahieux, R. Human T-Cell Lymphotropic virus type 3: Complete nucleotide sequence and characterization of the human tax3 protein. J. Virol. 2006, 80, 9876–9888. [Google Scholar] [CrossRef]
- Ahuka-Mundeke, S.; Lunguya-Metila, O.; Mbenzo-Abokome, V.; Butel, C.; Inogwabini, B.-I.; Omasombo, V.; Muyembe-Tamfum, J.-J.; Georgiev, A.V.; Muller, M.N.; Ndjango, J.-B.N.; et al. Genetic diversity of STLV-2 and interspecies transmission of STLV-3 in wild-living bonobos. Virus Evol. 2016, 2, vew011. [Google Scholar] [CrossRef]
- Murata, M.; Yasunaga, J.-I.; Washizaki, A.; Seki, Y.; Kuramitsu, M.; Tan, W.K.; Hu, A.; Okuma, K.; Hamaguchi, I.; Mizukami, T.; et al. Frequent horizontal and mother-to-child transmission may contribute to high prevalence of STLV-1 infection in Japanese macaques. Retrovirology 2020, 17, 15. [Google Scholar] [CrossRef]
- Alais, S.; Pasquier, A.; Jegado, B.; Journo, C.; Rua, R.; Gessain, A.; Tobaly-Tapiero, J.; Lacoste, R.; Turpin, J.; Mahieux, R. STLV-1 co-infection is correlated with an increased SFV proviral load in the peripheral blood of SFV/STLV-1 naturally infected non-human primates. PLoS Negl. Trop. Dis. 2018, 12, e0006812. [Google Scholar] [CrossRef]
- Rocamonde, B.; Alais, S.; Pelissier, R.; Moulin, V.; Rimbaud, B.; Lacoste, R.; Aurine, N.; Baquerre, C.; Pain, B.; Tanaka, Y.; et al. STLV-1 Commonly targets neurons in the brain of asymptomatic non-human primates. mBio 2023, 14, e0352622. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, I.; Bensaid, A.; Polack, B.; Perrin, B.; Berthelemy, M.; Levy, D. In vivo leukocyte tropism of bovine leukemia virus in sheep and cattle. J. Virol. 1994, 68, 4589–4596. [Google Scholar] [CrossRef] [PubMed]
- Hino, S.; Katamine, S.; Kawase, K.; Miyamoto, T.; Doi, H.; Tsuji, Y.; Yamabe, T. Intervention of maternal transmission of HTLV-1 in Nagasaki, Japan. Leukemia 1994, 8 (Suppl. S1), S68–S70. [Google Scholar] [PubMed]
- Sibon, D.; Cassar, O.; Duga, I.; Brouzes, C.; Ghez, D.; Pasquier, C.; Sibon, C.; Desrames, A.; Mortreux, F.; Wattel, E.; et al. Adult T-cell leukemia/lymphoma in a caucasian patient after sexual transmission of human T-cell lymphotropic virus type 1. Open Forum Infect. Dis. 2015, 2, ofv032. [Google Scholar] [CrossRef] [PubMed]
- Macatonia, S.E.; Cruickshank, J.; Rudge, P.; Knight, S.C. Dendritic cells from patients with tropical spastic paraparesis are infected with HTLV-1 and stimulate autologous lymphocyte proliferation. AIDS Res. Hum. Retroviruses 1992, 8, 1699–1706. [Google Scholar] [CrossRef]
- Koyanagi, Y.; Itoyama, Y.; Nakamura, N.; Takamatsu, K.; Kira, J.; Iwamasa, T.; Goto, I.; Yamamoto, N. In Vivo infection of human T-Cell leukemia virus type I in non-T cells. Virology 1993, 196, 25–33. [Google Scholar] [CrossRef]
- Grant, C.; Barmak, K.; Alefantis, T.; Yao, J.; Jacobson, S.; Wigdahl, B. Human T cell leukemia virus type I and neurologic disease: Events in bone marrow, peripheral blood, and central nervous system during normal immune surveillance and neuroinflammation. J. Cell. Physiol. 2002, 190, 133–159. [Google Scholar] [CrossRef]
- Kitamura, K.; Rudolph, D.L.; Goldsmith, C.; Folks, T.M.; Lal, R.B. Isolation, characterization, and transmission of human T-lymphotropic virus types I and II in culture. Curr. Microbiol. 1993, 27, 355–360. [Google Scholar] [CrossRef]
- Maguer-Satta, V.; Gazzolo, L.; Dodon, M. Human immature thymocytes as target cells of the leukemogenic activity of human T-cell leukemia virus type I. Blood 1995, 86, 1444–1452. [Google Scholar] [CrossRef]
- Manca, N.; Perandin, F.; De Simone, N.; Giannini, F.; Bonifati, D.; Angelini, C. Detection of HTLV-I tax-rex and pol gene sequences of thymus gland in a large group of patients with myasthenia gravis. J. Acquir. Immune. Defic. Syndr. 2002, 29, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Furuta, R.; Yasunaga, J.-I.; Miura, M.; Sugata, K.; Saito, A.; Akari, H.; Ueno, T.; Takenouchi, N.; Fujisawa, J.-I.; Koh, K.-R.; et al. Human T-cell leukemia virus type 1 infects multiple lineage hematopoietic cells in vivo. PLoS Pathog. 2017, 13, e1006722. [Google Scholar] [CrossRef] [PubMed]
- Turpin, J.; Alais, S.; Marçais, A.; Bruneau, J.; Melamed, A.; Gadot, N.; Tanaka, Y.; Hermine, O.; Melot, S.; Lacoste, R.; et al. Whole body clonality analysis in an aggressive STLV-1 associated leukemia (ATLL) reveals an unexpected clonal complexity. Cancer Lett. 2017, 389, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Fultz, P.N.; Gordon, T.P.; Anderson, D.C.; McClure, H.M. Prevalence of natural infection with simian immunodeficiency virus and simian T-cell leukemia virus type I in a breeding colony of sooty mangabey monkeys. Aids 1990, 4, 619–625. [Google Scholar] [CrossRef]
- Georges-Courbot, M.C.; Moisson, P.; Leroy, E.; Pingard, A.M.; Nerrienet, E.; Dubreuil, G.; Wickings, E.J.; Debels, F.; Bedjabaga, I.; Poaty-Mavoungou, V.; et al. Occurrence and frequency of transmission of naturally occurring simian retroviral infections (SIV, STLV, and SRV) at the CIRMF Primate Center, Gabon. J. Med. Primatol. 1996, 25, 313–326. [Google Scholar] [CrossRef]
- Locatelli, S.; Peeters, M. Cross-species transmission of simian retroviruses: How and why they could lead to the emergence of new diseases in the human population. Aids 2012, 26, 659–673. [Google Scholar] [CrossRef]
- Souquière, S.; Mouinga-Ondeme, A.; Makuwa, M.; Beggio, P.; Radaelli, A.; De Giuli Morghen, C.; Mortreux, F.; Kazanji, M. T-Cell tropism of simian T-cell leukaemia virus type 1 and cytokine profiles in relation to proviral load and immunological changes during chronic infection of naturally infected mandrills (Mandrillus sphinx). J. Med. Primatol. 2009, 38, 279–289. [Google Scholar] [CrossRef]
- Wachtman, L.; Mansfield, K. Chapter 1—Viral Diseases of Nonhuman Primates. In Nonhuman Primates in Biomedical Research, 2nd ed.; Abee, C.R., Mansfield, K., Tardif, S., Morris, T., Eds.; Academic Press: Boston, MA, USA, 2012; pp. 1–104. [Google Scholar]
- D’Offay, J.M.; Eberle, R.; Sucol, Y.; Schoelkopf, L.; White, M.A.; Valentine, B.D.; White, G.L.; Lerche, N.W. Transmission dynamics of simian T-lymphotropic virus type 1 (STLV1) in a baboon breeding colony: Predominance of female-to-female transmission. Comp. Med. 2007, 57, 105–114. [Google Scholar]
- Auffray, C.; Fogg, D.; Garfa, M.; Elain, G.; Join-Lambert, O.; Kayal, S.; Sarnacki, S.; Cumano, A.; Lauvau, G.; Geissmann, F. Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior. Science 2007, 317, 666–670. [Google Scholar] [CrossRef]
- Moody, C.A.; Pharr, G.T.; Murphey, J.; Hughlett, M.B.; Weaver, C.C.; Nelson, P.D.; Coats, K.S. Confirmation of Vertical Transmission of bovine immunodeficiency virus in naturally infected dairy cattle using the polymerase chain reaction. J. Vet.-Diagn. Investig. 2002, 14, 113–119. [Google Scholar] [CrossRef]
- Bhatia, S.; Patil, S.S.; Sood, R. Bovine immunodeficiency virus: A lentiviral infection. Indian J. Virol. 2013, 24, 332–341. [Google Scholar] [CrossRef]
- Nash, J.W.; Hanson, L.A.; St Cyr Coats, K. Detection of bovine immunodeficiency virus in blood and milk-derived leukocytes by use of polymerase chain reaction. Am. J. Vet.-Res. 1995, 56, 445–449. [Google Scholar]
- Payne, S.L.; Fuller, F.J. Virulence determinants of equine infectious anemia virus. Curr. HIV Res. 2010, 8, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Craigo, J.K.; Montelaro, R.C. Equine Infectious Anemia Virus. In Encyclopedia of Virology, 3rd ed.; Mahy, B.W.J., Van Regenmortel, M.H.V., Eds.; Academic Press: Oxford, UK, 2008; pp. 167–174. [Google Scholar]
- Leroux, C.; Cadoré, J.-L.; Montelaro, R.C. Equine Infectious Anemia Virus (EIAV): What has HIV’s country cousin got to tell us? Vet. Res. 2004, 35, 485–512. [Google Scholar] [CrossRef] [PubMed]
- Foil, L.D.; Issel, C.J. Transmission of retroviruses by arthropods. Annu. Rev. Èntomol. 1991, 36, 355–381. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, L.L.; Burkhard, M.J.; Diehl, L.J.; Hoover, E.A. Vertical transmission of feline immunodeficiency virus. Semin. Vet.-Med. Surg. Small Anim. 1995, 10, 266–278. [Google Scholar] [CrossRef]
- Kanzaki, L.I.B.; Looney, D.J. Feline immunodeficiency virus: A concise review. Front. Biosci. 2004, 9, 370–377. [Google Scholar] [CrossRef]
- Jordan, H.L.; Howard, J.G.; Bucci, J.G.; Butterworth, J.L.; English, R.; Kennedy-Stoskopf, S.; Tompkins, M.B.; Tompkins, W.A. Horizontal transmission of feline immunodeficiency virus with semen from seropositive cats. J. Reprod. Immunol. 1998, 41, 341–357. [Google Scholar] [CrossRef]
- Shimojima, M. Feline immunodeficiency virus tropism. Uirusu 2007, 57, 75–82. [Google Scholar] [CrossRef][Green Version]
- Sellon, R.K.; Jordan, H.L.; Kennedy-Stoskopf, S.; Tompkins, M.B.; Tompkins, W.A. Feline immunodeficiency virus can be experimentally transmitted via milk during acute maternal infection. J. Virol. 1994, 68, 3380–3385. [Google Scholar] [CrossRef]
- Allison, R.W.; Hoover, E.A. Feline Immunodeficiency virus is concentrated in milk early in lactation. AIDS Res. Hum. Retroviruses 2003, 19, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Hoover, E.A.; Mullins, J.I.; Chu, H.J.; Wasmoen, T.L. Development and testing of an inactivated feline leukemia virus vaccine. Semin. Vet.-Med. Surg. Small Anim. 1995, 10, 238–243. [Google Scholar] [PubMed]
- Narayan, O.; Clements, J.E.; Strandberg, J.D.; Cork, L.C.; Griffin, D.E. Biological characterization of the virus causing leukoencephalitis and arthritis in goats. J. Gen. Virol. 1980, 50, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Kaba, J.; Czopowicz, M.; Witkowski, L.; Szaluś-Jordanow, O.; Mickiewicz, M.; Markowska-Daniel, I.; Puchała, R.; Bagnicka, E. Longitudinal study on seroreactivity of goats exposed to colostrum and milk of small ruminant lentivirus–infected dams. J. Vet.-Res. 2022, 66, 511–521. [Google Scholar] [CrossRef]
- Peterhans, E.; Greenland, T.; Badiola, J.; Harkiss, G.; Bertoni, G.; Amorena, B.; Eliaszewicz, M.; Juste, R.A.; Krassnig, R.; Lafont, J.-P.; et al. Routes of transmission and consequences of small ruminant lentiviruses (SRLVs) infection and eradication schemes. Vet. Res. 2004, 35, 257–274. [Google Scholar] [CrossRef]
- Larruskain, A.; Jugo, B.M. Retroviral infections in sheep and goats: Small ruminant lentiviruses and host interaction. Viruses 2013, 5, 2043–2061. [Google Scholar] [CrossRef]
- McNeilly, T.N.; Baker, A.; Brown, J.K.; Collie, D.; MacLachlan, G.; Rhind, S.M.; Harkiss, G.D. Role of alveolar macrophages in respiratory transmission of visna/maedi virus. J. Virol. 2008, 82, 1526–1536. [Google Scholar] [CrossRef]
- Shaw, G.M.; Hunter, E. HIV Transmission. Cold Spring Harb. Perspect. Med. 2012, 2, a006965. [Google Scholar] [CrossRef]
- Van de Perre, P.; Rubbo, P.-A.; Viljoen, J.; Nagot, N.; Tylleskär, T.; Lepage, P.; Vendrell, J.-P.; Tuaillon, E. HIV-1 Reservoirs in breast milk and challenges to elimination of breast-feeding transmission of HIV-1. Sci. Transl. Med. 2012, 4, 143sr3. [Google Scholar] [CrossRef]
- WHO. HIV/AIDS: Infant Feeding and Nutrition. Available online: https://www.who.int/news-room/questions-and-answers/item/hiv-aids-infant-feeding-and-nutrition# (accessed on 29 May 2023).
- Cardenas, M.C.; Farnan, S.; Hamel, B.L.; Mejia Plazas, M.C.; Sintim-Aboagye, E.; Littlefield, D.R.; Behl, S.; Punia, S.; Enninga, E.A.L.; Johnson, E.; et al. Prevention of the vertical transmission of HIV.; A recap of the journey so far. Viruses 2023, 15, 849. [Google Scholar] [CrossRef]
- Van De Perre, P.; Kankasa, C.; Nagot, N.; Meda, N.; Tumwine, J.K.; Coutsoudis, A.; Tylleskär, T.; Coovadia, H. Pre-exposure prophylaxis for infants exposed to HIV through breast feeding. BMJ 2017, 356, j1053. [Google Scholar] [CrossRef] [PubMed]
- Van de Perre, P.; Goga, A.; Ngandu, N.; Nagot, N.; Moodley, D.; King, R.; Molès, J.-P.; Mosqueira, B.; Chirinda, W.; Scarlatti, G.; et al. Eliminating postnatal HIV transmission in high incidence areas: Need for complementary biomedical interventions. Lancet 2021, 397, 1316–1324. [Google Scholar] [CrossRef]
- Ceccarelli, G.; Giovanetti, M.; Sagnelli, C.; Ciccozzi, A.; D’ettorre, G.; Angeletti, S.; Borsetti, A.; Ciccozzi, M. Human Immunodeficiency Virus Type 2: The Neglected Threat. Pathogens 2021, 10, 1377. [Google Scholar] [CrossRef]
- Klatt, N.R.; Silvestri, G.; Hirsch, V. Nonpathogenic Simian Immunodeficiency Virus Infections. Cold Spring Harb. Perspect. Med. 2012, 2, a007153. [Google Scholar] [CrossRef] [PubMed]
- Chahroudi, A.; Meeker, T.; Lawson, B.; Ratcliffe, S.; Else, J.; Silvestri, G. Mother-to-infant transmission of simian immunodeficiency virus is rare in sooty mangabeys and is associated with low viremia. J. Virol. 2011, 85, 5757–5763. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Swanstrom, A.E.; Del Prete, G.Q.; Deleage, C.; Elser, S.E.; Lackner, A.A.; Hoxie, J.A. The SIV envelope glycoprotein, viral tropism, and pathogenesis: Novel insights from nonhuman primate models of AIDS. Curr. HIV Res. 2018, 16, 29–40. [Google Scholar] [CrossRef]
- Matthew, A.G.; Gene, L.D.; Steven, E.F.; Gregory, J.T. Bovine immunodeficiency virus: Molecular biology and virus-host interactions. Virus Res. 1994, 32, 155–181. [Google Scholar] [CrossRef]
- Cook, S.J.; Cook, R.F.; Montelaro, R.C.; Issel, C.J. Differential responses of Equus caballus and Equus asinus to infection with two pathogenic strains of equine infectious anemia virus. Vet. Microbiol. 2001, 79, 93–109. [Google Scholar] [CrossRef]
- Kemen, M.J.; Coggins, L. Equine infectious anemia: Transmission from infected mares to foals. J. Am. Vet.-Med. Assoc. 1972, 161, 496–499. [Google Scholar]
- More, S.J.; Aznar, I.; Myers, T.; Leadon, D.P.; Clegg, T.A. An outbreak of equine infectious anaemia in Ireland during 2006: The modes of transmission and spread in the Kildare cluster. Equine Vet.-J. 2008, 40, 709–711. [Google Scholar] [CrossRef]
- Pedersen, N.C.; Ho, E.W.; Brown, M.L.; Yamamoto, J.K. Isolation of a T-Lymphotropic virus from domestic cats with an immunodeficiency-like syndrome. Science 1987, 235, 790–793. [Google Scholar] [CrossRef]
- Courchamp, F.; Pontier, D. Feline immunodeficiency virus: An epidemiological review. C. R. Acad. Sci. III 1994, 317, 1123–1134. [Google Scholar]
- Rogers, A.B.; Hoover, E.A. Maternal-fetal feline immunodeficiency virus transmission: Timing and tissue tropisms. J. Infect. Dis. 1998, 178, 960–967. [Google Scholar] [CrossRef]
- Murphy, B.G.; Castillo, D.; Mete, A.; Vogel, H.; Goldsmith, D.; Barro, M.; Gonzales-Viera, O. Caprine arthritis encephalitis virus is associated with renal lesions. Viruses 2021, 13, 1051. [Google Scholar] [CrossRef]
- Wilen, C.B.; Tilton, J.C.; Doms, R.W. HIV: Cell binding and entry. Cold Spring Harb. Perspect. Med. 2012, 2, a006866. [Google Scholar] [CrossRef]
- Haase, A.T. Targeting early infection to prevent HIV-1 mucosal transmission. Nature 2010, 464, 217–223. [Google Scholar] [CrossRef]
- Klatzmann, D.; Champagne, E.; Chamaret, S.; Gruest, J.; Guetard, D.; Hercend, T.; Gluckman, J.-C.; Montagnier, L. T-lymphocyte T4 molecule behaves as the receptor for human retrovirus LAV. Nature 1984, 312, 767–768. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.C.; Burdo, T.H. HIV and SIV infection: The role of cellular restriction and immune responses in viral replication and pathogenesis. Apmis 2009, 117, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, K.G.; Lerche, N.W.; Gardner, M.B.; Lackner, A.A. Origins of simian immunodeficiency virus infection in macaques at The New England Regional Primate Research Center. J. Med. Primatol. 1995, 24, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.; Brown, C.R.; Ourmanov, I.; Pandrea, I.; Buckler-White, A.; Erb, C.; Nandi, J.S.; Foster, G.J.; Autissier, P.; Schmitz, J.E.; et al. Comparison of simian immunodeficiency virus sivagmver replication and CD4 + T-cell dynamics in vervet and Sabaeus African green monkeys. J. Virol. 2006, 80, 4868–4877. [Google Scholar] [CrossRef] [PubMed][Green Version]
- White, A.B.; Mirjahangir, J.F.; Horvath, H.; Anglemyer, A.; Read, J.S. Antiretroviral interventions for preventing breast milk transmission of HIV. Cochrane Database Syst. Rev. 2014, 4, CD011323. [Google Scholar] [CrossRef]
- Dressman, J.B.; Berardi, R.R.; Dermentzoglou, L.C.; Russell, T.L.; Schmaltz, S.P.; Barnett, J.L.; Jarvenpaa, K.M. Upper Gastrointestinal (GI) pH in Young, Healthy Men and Women. Pharm. Res. 1990, 7, 756–761. [Google Scholar] [CrossRef]
- Beasley, D.E.; Koltz, A.M.; Lambert, J.E.; Fierer, N.; Dunn, R.R. The evolution of stomach acidity and its relevance to the human microbiome. PLoS ONE 2015, 10, e0134116. [Google Scholar] [CrossRef]
- Jeffers, L.; Webster-Cyriaque, J.Y. Viruses and salivary gland disease (SGD): Lessons from HIV SGD. Adv. Dent. Res. 2011, 23, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, G.M.; Pringle, S.; Bootsma, H.; Kroese, F.G.M. Epithelial–immune cell interplay in primary Sjögren syndrome salivary gland pathogenesis. Nat. Rev. Rheumatol. 2021, 17, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Sumida, T.; Yonaha, F.; Maeda, T.; Kita, Y.; Iwamoto, I.; Koike, T.; Yoshida, S. Expression of sequences homologous to htlv-itax gene in the labial salivary glands of japanese patients with sjögren’s syndrome. Arthritis Rheum. 1994, 37, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Shimizu, T.; Takatani, A.; Suematsu, T.; Nakamura, T.; Kawakami, A. Initial human T-cell leukemia virus type 1 infection of the salivary gland epithelial cells requires a biofilm-like structure. Virus Res. 2019, 269, 197643. [Google Scholar] [CrossRef]
- Mazzanti, C.M.; Lessi, F.; Armogida, I.; Zavaglia, K.; Franceschi, S.; AI Hamad, M.; Roncella, M.; Ghilli, M.; Boldrini, A.; Aretini, P.; et al. Human saliva as route of inter-human infection for mouse mammary tumor virus. Oncotarget 2015, 6, 18355–18363. [Google Scholar] [CrossRef]
- Lessi, F.; Grandi, N.; Mazzanti, C.M.; Civita, P.; Scatena, C.; Aretini, P.; Bandiera, P.; Fornaciari, A.; Giuffra, V.; Fornaciari, G.; et al. A human MMTV-like betaretrovirus linked to breast cancer has been present in humans at least since the copper age. Aging 2020, 12, 15978–15994. [Google Scholar] [CrossRef]
- Kariuki, S.M.; Selhorst, P.; Ariën, K.K.; Dorfman, J.R. The HIV-1 transmission bottleneck. Retrovirology 2017, 14, 22. [Google Scholar] [CrossRef]
- Simonon, A.; Lepage, P.; Karita, E.; Hitimana, D.G.; Dabis, F.; Msellati, P.; Van Goethem, C.; Nsengumuremyi, F.; Bazubagira, A.; Van de Perre, P. An assessment of the timing of mother-to-child transmission of human immunodeficiency virus type 1 by means of polymerase chain reaction. JAIDS J. Acquir. Immune Defic. Syndr. 1994, 7, 952–957. [Google Scholar]
- Sufiawati, I.; Herrera, R.; Mayer, W.; Cai, X.; Borkakoti, J.; Lin, V.; Rosbe, K.; Tugizov, S.M. Human immunodeficiency virus (HIV) and human cytomegalovirus (HCMV) Coinfection of infant tonsil epithelium may synergistically promote both HIV-1 and HCMV spread and infection. J. Virol. 2021, 95, e00921. [Google Scholar] [CrossRef]
- Tugizov, S.M.; Herrera, R.; Chin-Hong, P.; Veluppillai, P.; Greenspan, D.; Michael Berry, J.; Pilcher, C.D.; Shiboski, C.H.; Jay, N.; Rubin, M.; et al. HIV-associated disruption of mucosal epithelium facilitates paracellular penetration by human papillomavirus. Virology 2013, 446, 378–388. [Google Scholar] [CrossRef]
- Martin-Latil, S.; Gnädig, N.F.; Mallet, A.; Desdouits, M.; Guivel-Benhassine, F.; Jeannin, P.; Prevost, M.-C.; Schwartz, O.; Gessain, A.; Ozden, S.; et al. Transcytosis of HTLV-1 across a tight human epithelial barrier and infection of subepithelial dendritic cells. Blood 2012, 120, 572–580. [Google Scholar] [CrossRef]
- Langlois, M.; Bounou, S.; Tremblay, M.J.; Barbeau, B. Infection of the Ex Vivo tonsil model by HTLV-1 Envelope-pseudotyped viruses. Pathogens 2023, 12, 182. [Google Scholar] [CrossRef] [PubMed]
- Takenouchi, N.; Matsuoka, E.; Moritoyo, T.; Nagai, M.; Katsuta, K.; Hasui, K.; Ueno, K.; Eizuru, Y.; Usuku, K.; Osame, M.; et al. Molecular pathologic analysis of the tonsil in HTLV-I–infected individuals. Am. J. Ther. 1999, 22, 200–207. [Google Scholar] [CrossRef]
- Miller, C.; Boegler, K.; Carver, S.; MacMillan, M.; Bielefeldt-Ohmann, H.; VandeWoude, S. Pathogenesis of oral FIV infection. PLoS ONE 2017, 12, e0185138. [Google Scholar] [CrossRef]
- Massoni-Badosa, R.; Soler-Vila, P.; Aguilar-Fernández, S.; Nieto, J.C.; Elosua-Bayes, M.; Marchese, D.; Kulis, M.; Vilas-Zornoza, A.; Bühler, M.M.; Rashmi, S.; et al. An Atlas of Cells in the Human Tonsil. bioRxiv 2022. [Google Scholar] [CrossRef]
- Jain, P.; Manuel, S.L.; Khan, Z.K.; Ahuja, J.; Quann, K.; Wigdahl, B. DC-SIGN Mediates cell-free infection and transmission of human T-cell lymphotropic virus type 1 by dendritic cells. J. Virol. 2009, 83, 10908–10921. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.S.; Petrow-Sadowski, C.; Huang, Y.K.; Bertolette, D.C.; Ruscetti, F.W. Cell-free HTLV-1 infects dendritic cells leading to transmission and transformation of CD4+ T cells. Nat. Med. 2008, 14, 429–436. [Google Scholar] [CrossRef]
- Shimauchi, T.; Caucheteux, S.; Finsterbusch, K.; Turpin, J.; Blanchet, F.; Ladell, K.; Triantafilou, K.; Czubala, M.; Tatsuno, K.; Easter, T.; et al. Dendritic cells promote the spread of human T-cell leukemia virus type 1 via bidirectional interactions with CD4+ T cells. J. Investig. Dermatol. 2019, 139, 157–166. [Google Scholar] [CrossRef]
- Burleigh, L.; Lozach, P.-Y.; Schiffer, C.; Staropoli, I.; Pezo, V.; Porrot, F.; Canque, B.; Virelizier, J.-L.; Arenzana-Seisdedos, F.; Amara, A. Infection of dendritic cells (DCs), Not DC-SIGN-mediated internalization of human immunodeficiency virus, is required for long-term transfer of virus to T cells. J. Virol. 2006, 80, 2949–2957. [Google Scholar] [CrossRef]
- Sewald, X.; Ladinsky, M.S.; Uchil, P.D.; Beloor, J.; Pi, R.; Herrmann, C.; Motamedi, N.; Murooka, T.T.; Brehm, M.A.; Greiner, D.L.; et al. Retroviruses use CD169-mediated trans-infection of permissive lymphocytes to establish infection. Science 2015, 350, 563–567. [Google Scholar] [CrossRef]
- Ogobuiro, I.; Gonzales, J.; Tuma, F. Physiology, Gastrointestinal. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2023. [Google Scholar]
- Haugh, K.A.; Ladinsky, M.S.; Ullah, I.; Stone, H.M.; Pi, R.; Gilardet, A.; Grunst, M.W.; Kumar, P.; Bjorkman, P.J.; Mothes, W.; et al. In vivo imaging of retrovirus infection reveals a role for Siglec-1/CD169 in multiple routes of transmission. Elife 2021, 10, e64179. [Google Scholar] [CrossRef] [PubMed]
- Uchil, P.D.; Haugh, K.A.; Pi, R.; Mothes, W. In Vivo imaging-driven approaches to study virus dissemination and pathogenesis. Annu. Rev. Virol. 2019, 6, 501–524. [Google Scholar] [CrossRef]
- Ye, K.; Dhiman, H.K.; Suhan, J.; Schultz, J.S. Effect of pH on infectivity and morphology of ecotropic moloney murine leukemia virus. Biotechnol. Prog. 2003, 19, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Mason, S. Some Aspects of gastric function in the newborn. Arch. Dis. Child. 1962, 37, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Neal-Kluever, A.; Fisher, J.; Grylack, L.; Kakiuchi-Kiyota, S.; Halpern, W. Physiology of the neonatal gastrointestinal system relevant to the disposition of orally administered medications. Drug Metab. Dispos. 2019, 47, 296–313. [Google Scholar] [CrossRef]
- Liu, R.; Huang, L.; Li, J.; Zhou, X.; Zhang, H.; Zhang, T.; Lei, Y.; Wang, K.; Xie, N.; Zheng, Y.; et al. HIV infection in gastric epithelial cells. J. Infect. Dis. 2013, 208, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Haynes, R.A., 2nd; Zimmerman, B.; Millward, L.; Ware, E.; Premanandan, C.; Yu, L.; Phipps, A.J.; Lairmore, M.D. Early spatial and temporal events of human T-lymphotropic virus type 1 spread following blood-borne transmission in a rabbit model of infection. J. Virol. 2010, 84, 5124–5130. [Google Scholar] [CrossRef] [PubMed]
- Jiang, A.-P.; Jiang, J.-F.; Wei, J.-F.; Guo, M.-G.; Qin, Y.; Guo, Q.-Q.; Ma, L.; Liu, B.-C.; Wang, X.; Veazey, R.S.; et al. Human mucosal mast cells capture HIV-1 and mediate viral trans-infection of CD4+ T Cells. J. Virol. 2015, 90, 2928–2937. [Google Scholar] [CrossRef] [PubMed]
- Song, S.-T.; Wu, M.-L.; Zhang, H.-J.; Su, X.; Wang, J.-H. Mast cell activation triggered by retrovirus promotes acute viral infection. Front. Microbiol. 2022, 13, 798660. [Google Scholar] [CrossRef] [PubMed]
- Shen, R.; Smythies, L.E.; Clements, R.H.; Novak, L.; Smith, P.D. Dendritic cells transmit HIV-1 through human small intestinal mucosa. J. Leukoc. Biol. 2010, 87, 663–670. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kemeter, L.M.; Birzer, A.; Heym, S.; Thoma-Kress, A.K. Milk Transmission of Mammalian Retroviruses. Microorganisms 2023, 11, 1777. https://doi.org/10.3390/microorganisms11071777
Kemeter LM, Birzer A, Heym S, Thoma-Kress AK. Milk Transmission of Mammalian Retroviruses. Microorganisms. 2023; 11(7):1777. https://doi.org/10.3390/microorganisms11071777
Chicago/Turabian StyleKemeter, Laura M., Alexandra Birzer, Stefanie Heym, and Andrea K. Thoma-Kress. 2023. "Milk Transmission of Mammalian Retroviruses" Microorganisms 11, no. 7: 1777. https://doi.org/10.3390/microorganisms11071777
APA StyleKemeter, L. M., Birzer, A., Heym, S., & Thoma-Kress, A. K. (2023). Milk Transmission of Mammalian Retroviruses. Microorganisms, 11(7), 1777. https://doi.org/10.3390/microorganisms11071777






