Duration of Immunity Induced after Vaccination of Cattle with a Live Attenuated or Inactivated Lumpy Skin Disease Virus Vaccine
Abstract
1. Introduction
2. Materials and Methods
2.1. Challenge Virus and Cell Line
2.2. Serological Analysis
2.3. Virus Isolation and Titration
2.4. Animal Trial
2.4.1. Vaccines
2.4.2. Experimental Animal Study Design
2.4.3. Clinical Evaluation and Scoring
2.4.4. Sampling
2.5. Pan Capripox RT-PCR
2.6. PCR Assay for Differentiating Infected from Vaccinated Animals (DIVA)
2.7. IFNγ Release Assay
3. Results
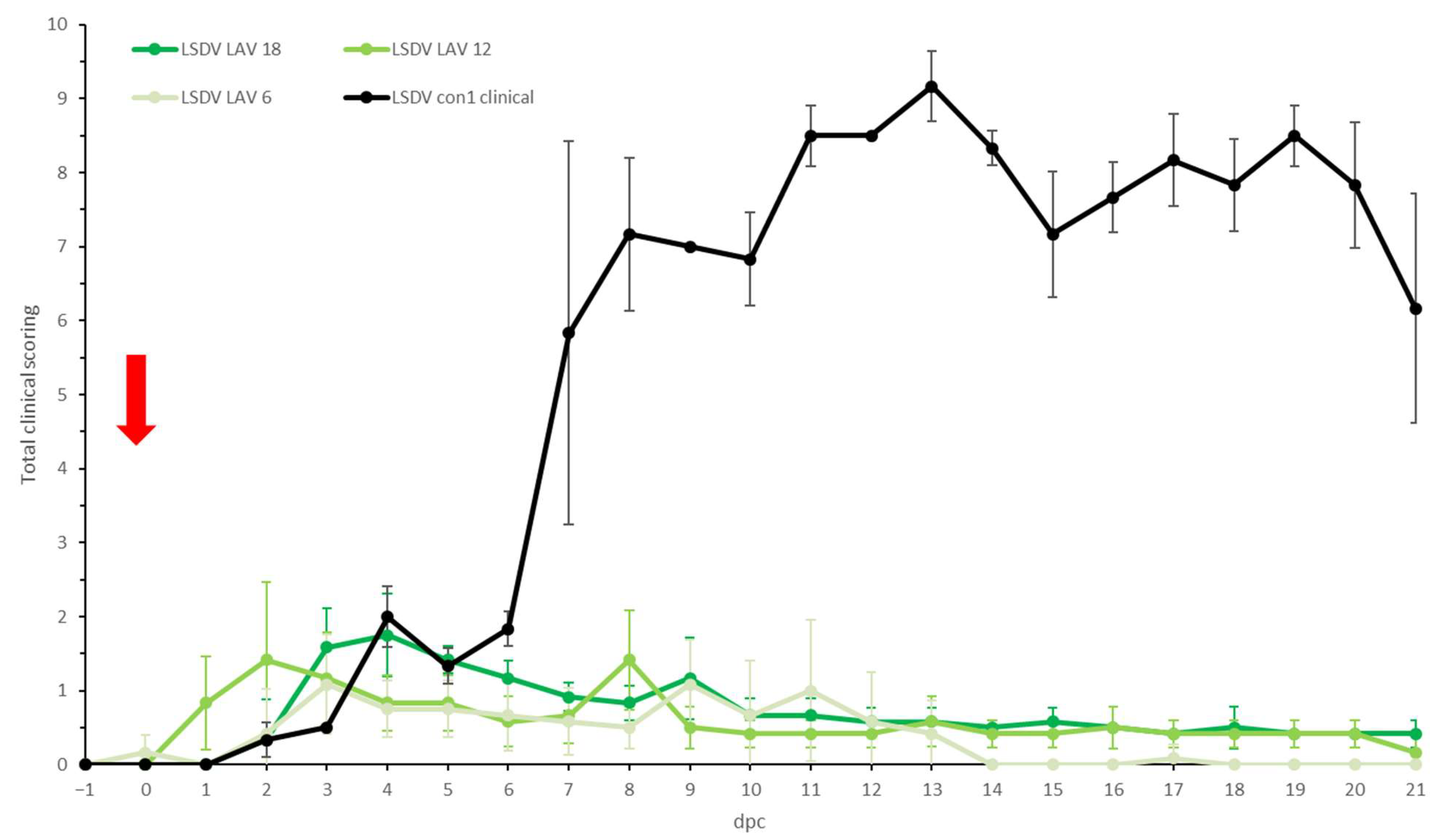
3.1. Unvaccinated Control Groups
3.1.1. Clinical Observations and Scoring
3.1.2. Virology and Serology
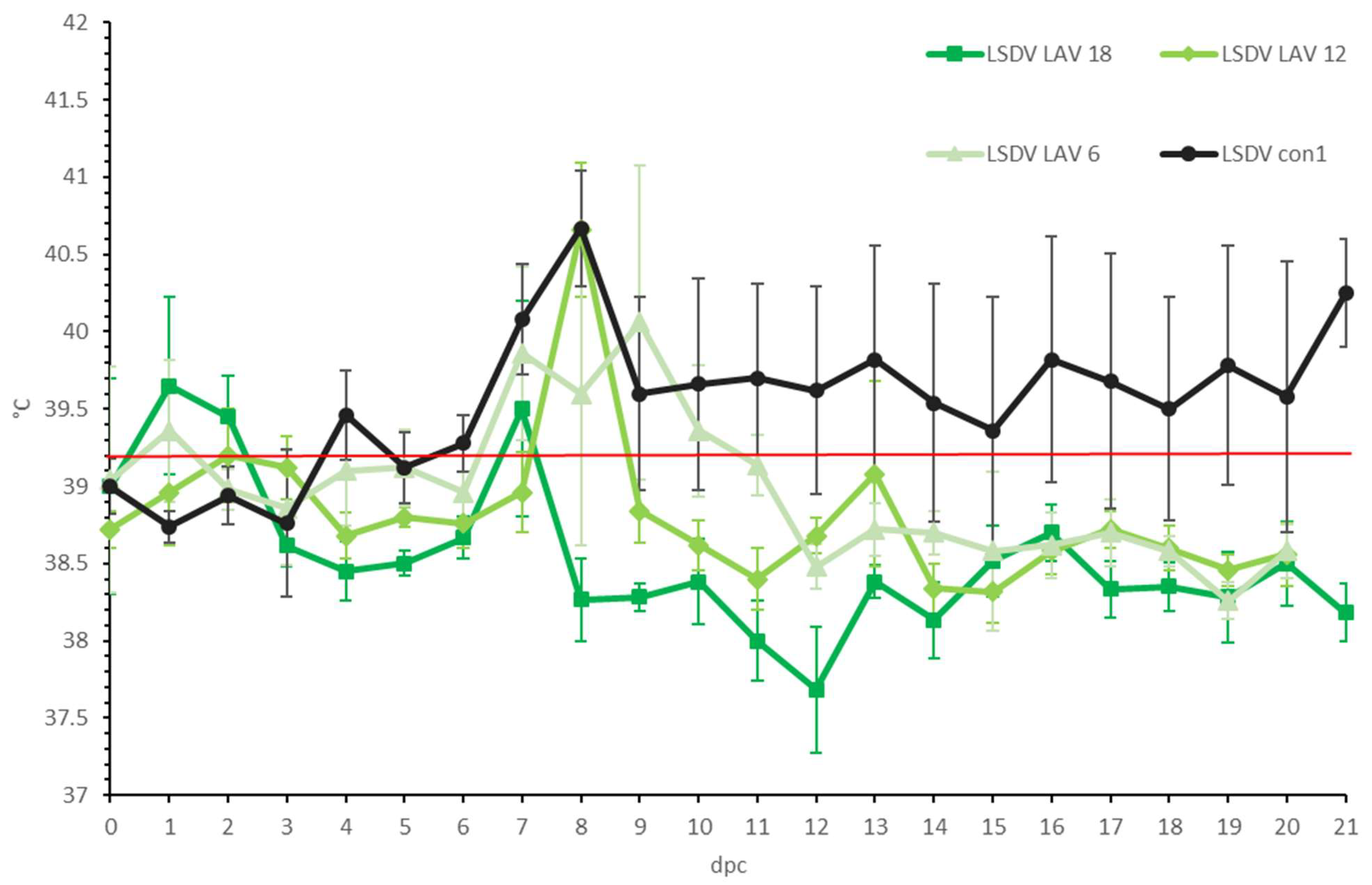
3.2. LSDV LAV-Vaccinated Groups
3.2.1. Clinical Observations and Scoring
3.2.2. Virology
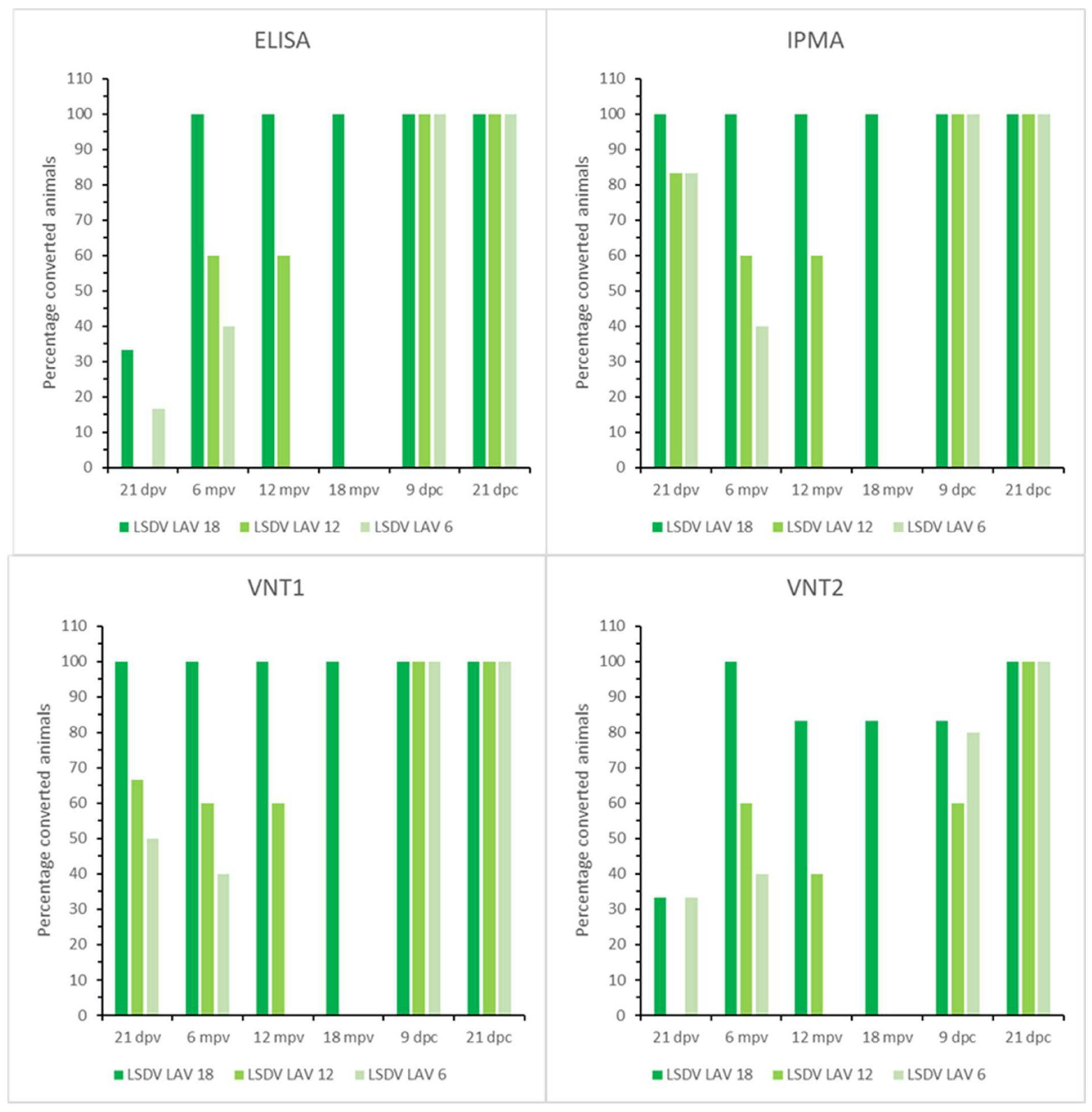
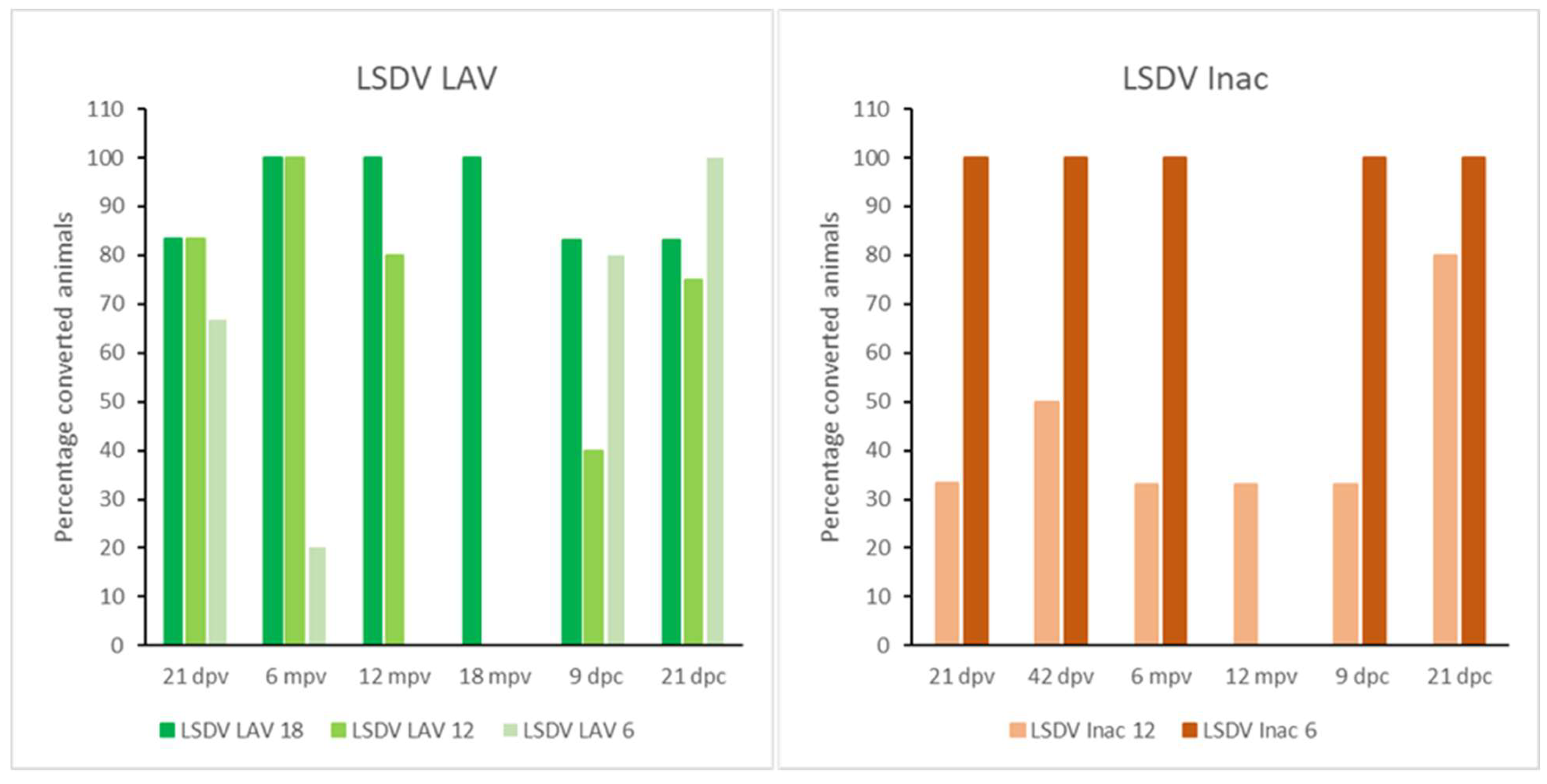
3.2.3. Serology
3.2.4. Cellular Immunity (IFNγ-Assay)
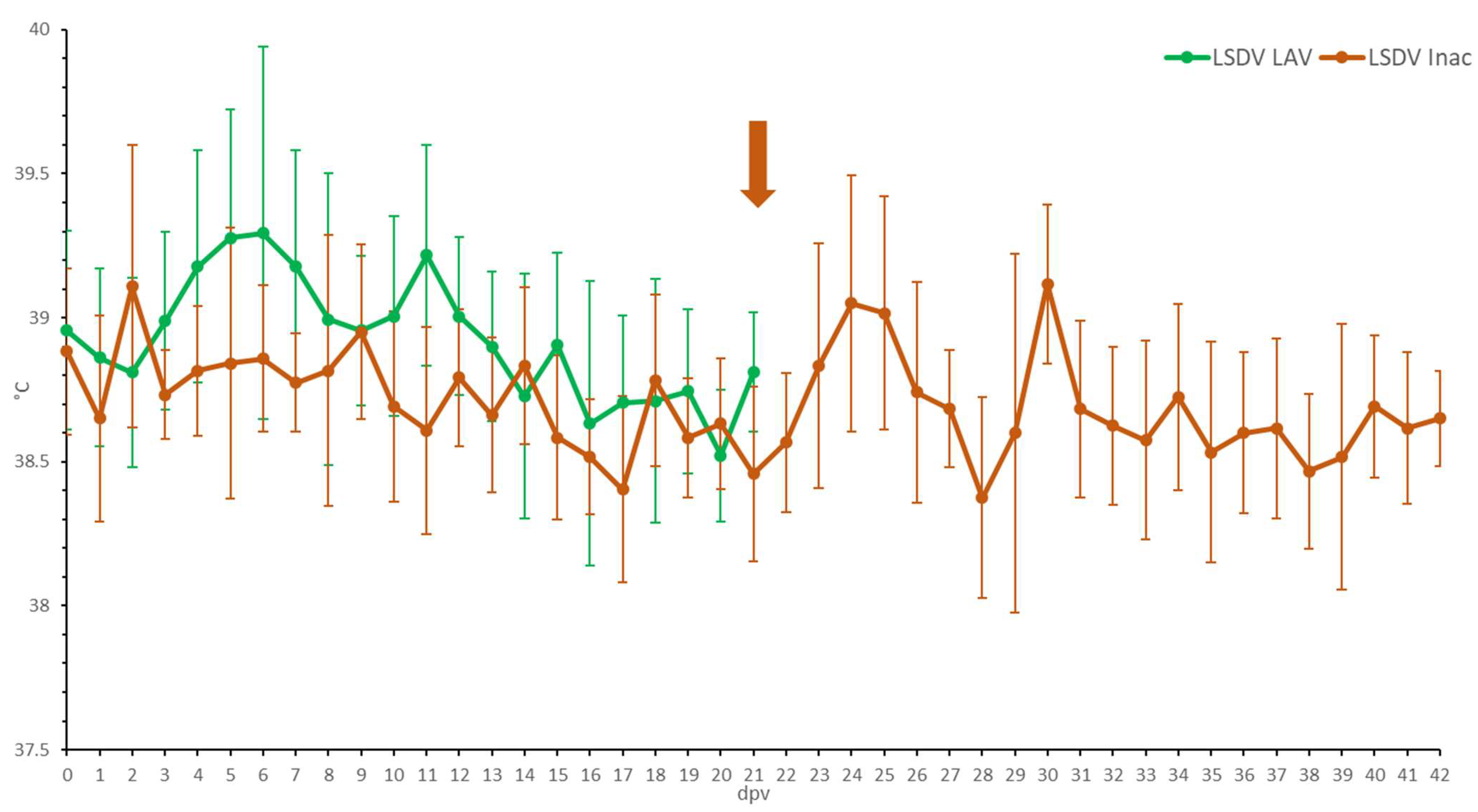
3.3. LSDV Inac Vaccinated Groups
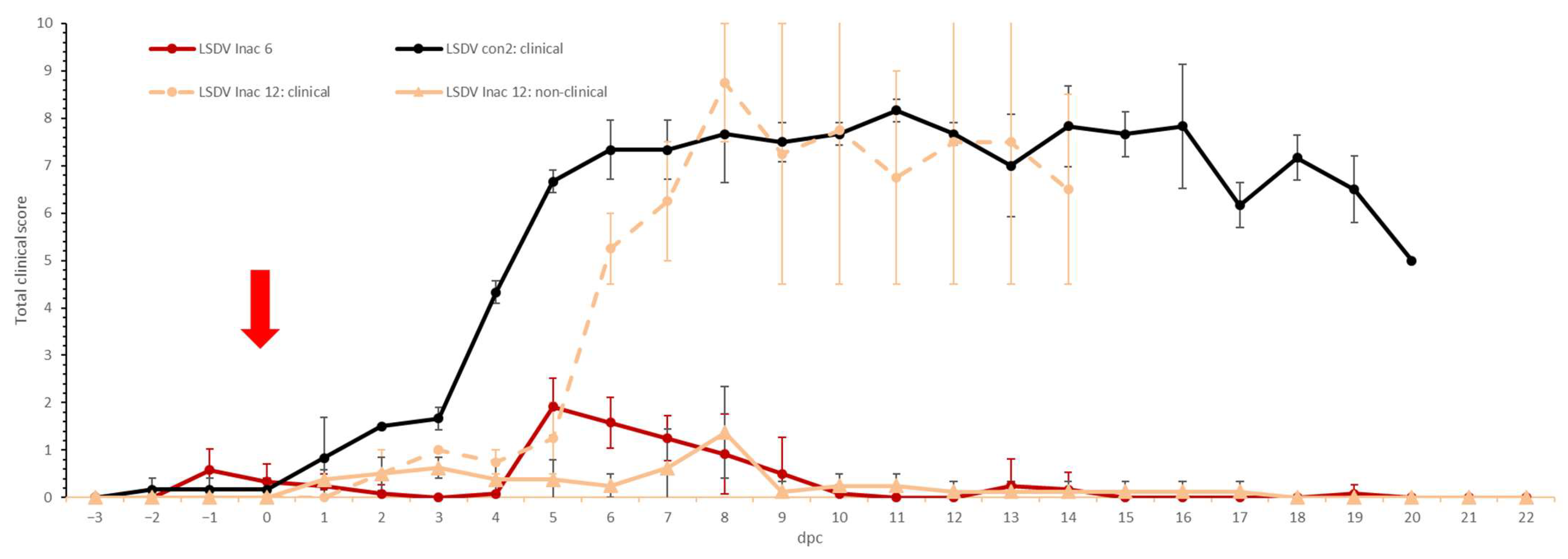
3.3.1. Clinical Observations and Scoring
3.3.2. Virology
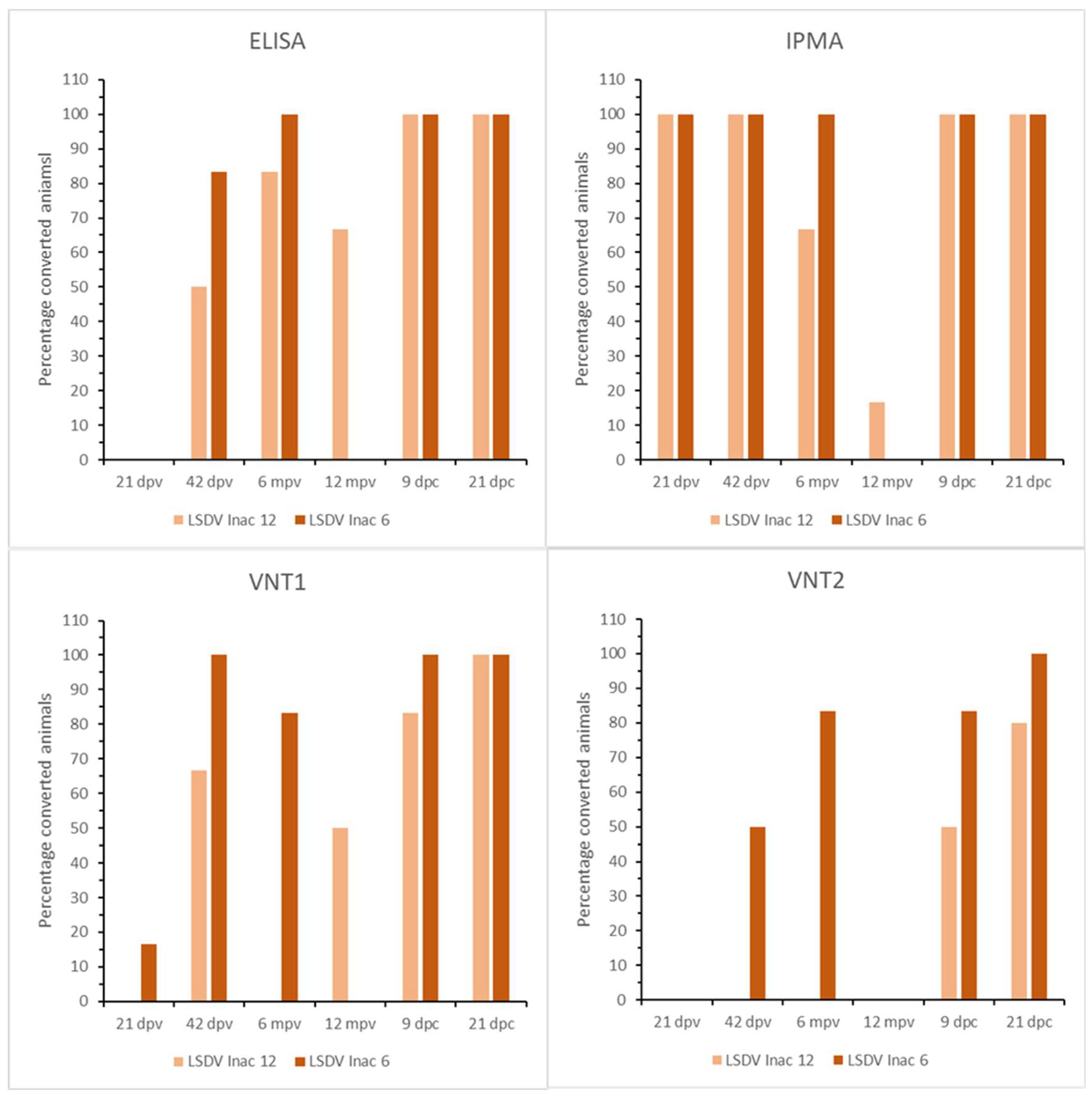
3.3.3. Serology
3.3.4. Cellular Immunity (IFNγ-Assay)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tulman, E.R.; Afonso, C.L.; Lu, Z.; Zsak, L.; Sur, J.-H.; Sandybaev, N.T.; Kerembekova, U.Z.; Zaitsev, V.L.; Kutish, G.F.; Rock, D.L. The Genomes of Sheeppox and Goatpox Viruses. J. Virol. 2002, 76, 6054–6061. [Google Scholar] [CrossRef]
- Tuppurainen, E.S.; Venter, E.H.; Coetzer, J.A. The detection of lumpy skin disease virus in samples of experimentally infected cattle using different diagnostic techniques. Onderstepoort J. Veter. Res. 2005, 72, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Babiuk, S.; Bowden, T.R.; Boyle, D.B.; Wallace, D.B.; Kitching, R.P. Capripoxviruses: An Emerging Worldwide Threat to Sheep, Goats and Cattle. Transbound. Emerg. Dis. 2008, 55, 263–272. [Google Scholar] [CrossRef]
- Gupta, T.; Patial, V.; Bali, D.; Angaria, S.; Sharma, M.; Chahota, R. A review: Lumpy skin disease and its emergence in India. Veter.-Res. Commun. 2020, 44, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Green, H.F. Lumpy Skin Disease: Its Effect on Hides and Leather and a Comparison on this Respect with some other Skin Diseases. Bull. Epizoot. Dis. Afr. 1959, 7, 63–74. [Google Scholar]
- Gari, G.; Bonnet, P.; Roger, F.; Waret-Szkuta, A. Epidemiological aspects and financial impact of lumpy skin disease in Ethiopia. Prev. Veter. Med. 2011, 102, 274–283. [Google Scholar] [CrossRef]
- Molla, W.; de Jong, M.C.; Gari, G.; Frankena, K. Economic impact of lumpy skin disease and cost effectiveness of vaccination for the control of outbreaks in Ethiopia. Prev. Veter.-Med. 2017, 147, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Tuppurainen, E.S.M.; Antoniou, S.-E.; Tsiamadis, E.; Topkaridou, M.; Labus, T.; Debeljak, Z.; Plavšić, B.; Miteva, A.; Alexandrov, T.; Pite, L.; et al. Field observations and experiences gained from the implementation of control measures against lumpy skin disease in South-East Europe between 2015 and 2017. Prev. Veter. Med. 2020, 181, 104600. [Google Scholar] [CrossRef]
- Thomas, A.D.; Mare, C.V.E. Knopvelsiekte. J. S. Afr. Veter. Assoc. 1945, 16, 36–43. [Google Scholar]
- Coetzer, J.A.W.; Tustin, R.C. Infectious Diseases of Livestock, 2nd ed.; Oxford University Press: Cape Town, South Africa, 2004; Volume 3, pp. 1268–1276. [Google Scholar]
- Tageldin, M.H.; Wallace, D.B.; Gerdes, G.H.; Putterill, J.F.; Greyling, R.R.; Phosiwa, M.N.; Al Busaidy, R.M.; Al Ismaaily, S.I. Lumpy skin disease of cattle: An emerging problem in the Sultanate of Oman. Trop. Anim. Health Prod. 2014, 46, 241–246. [Google Scholar] [CrossRef]
- MacDonald, R.A.S. Pseudo-urticaria of cattle. In Annual Report for 1930–1931; Department of Animal Health, Government of Northern Rhodesia: Lusaka, Zambia, 1931; pp. 20–21. [Google Scholar]
- Tasioudi, K.E.; Antoniou, S.E.; Iliadou, P.; Sachpatzidis, A.; Plevraki, E.; Agianniotaki, E.I.; Fouki, C.; Mangana-Vougiouka, O.; Chondrokouki, E.; Dile, C. Emergence of Lumpy Skin Disease in Greece, 2015. Transbound. Emerg. Dis. 2016, 63, 260–265. [Google Scholar] [CrossRef] [PubMed]
- FAO. Sustainable Prevention, Control and Elimination of Lumpy Skin Disease—Eastern Europe and the Balkans. Position Paper. No. 2; FAO Animal Production and Health: Rome, Italy, 2017; p. 26. ISBN 978-92-5-109937-7. [Google Scholar]
- FAO. Introduction and Spread of Lumpy Skin Disease in South, East and Southeast Asia—Qualitative Risk Assessment and Management; FAO Animal Production and Health: Rome, Italy, 2020; p. 183. [Google Scholar]
- Ben-Gera, J.; Klement, E.; Khinich, E.; Stram, Y.; Shpigel, N. Comparison of the efficacy of Neethling lumpy skin disease virus and x10RM65 sheep-pox live attenuated vaccines for the prevention of lumpy skin disease—The results of a randomized controlled field study. Vaccine 2015, 33, 4837–4842. [Google Scholar] [CrossRef] [PubMed]
- Tuppurainen, E.S.M.; Venter, E.H.; Shisler, J.L.; Gari, G.; Mekonnen, G.A.; Juleff, N.; Lyons, N.A.; De Clercq, K.; Upton, C.; Bowden, T.R.; et al. Review: Capripoxvirus Diseases: Current Status and Opportunities for Control. Transbound. Emerg. Dis. 2017, 64, 729–745. [Google Scholar] [CrossRef] [PubMed]
- Klement, E.; Broglia, A.; Antoniou, S.-E.; Tsiamadis, V.; Plevraki, E.; Petrović, T.; Polaček, V.; Debeljak, Z.; Miteva, A.; Alexandrov, T.; et al. Neethling vaccine proved highly effective in controlling lumpy skin disease epidemics in the Balkans. Prev. Veter.-Med. 2020, 181, 104595. [Google Scholar] [CrossRef] [PubMed]
- Haegeman, A.; De Leeuw, I.; Mostin, L.; Van Campe, W.; Aerts, L.; Venter, E.; Tuppurainen, E.; Saegerman, C.; De Clercq, K. Comparative Evaluation of Lumpy Skin Disease Virus-Based Live Attenuated Vaccines. Vaccines 2021, 9, 473. [Google Scholar] [CrossRef]
- Gethmann, J.; Zilow, V.; Probst, C.; Elbers, A.; Conraths, F. Why German farmers have their animals vaccinated against Bluetongue virus serotype 8: Results of a questionnaire survey. Vaccine 2015, 33, 214–221. [Google Scholar] [CrossRef]
- Wane, A.; Dione, M.; Wieland, B.; Rich, K.M.; Yena, A.S.; Fall, A. Willingness to Vaccinate (WTV) and Willingness to Pay (WTP) for Vaccination Against Peste des Petits Ruminants (PPR) in Mali. Front. Veter.-Sci. 2020, 6, 488. [Google Scholar] [CrossRef]
- Hamdi, J.; Boumart, Z.; Daouam, S.; El Arkam, A.; Bamouh, Z.; Jazouli, M.; Tadlaoui, K.O.; Fihri, O.F.; Gavrilov, B.; El Harrak, M. Development and Evaluation of an Inactivated Lumpy Skin Disease Vaccine for Cattle. Veter.-Microbiol. 2020, 245, 108689. [Google Scholar] [CrossRef]
- Wolff, J.; Moritz, T.; Schlottau, K.; Hoffmann, D.; Beer, M.; Hoffmann, B. Development of a Safe and Highly Efficient Inactivated Vaccine Candidate against Lumpy Skin Disease Virus. Vaccines 2020, 9, 4. [Google Scholar] [CrossRef]
- Mutua, E.; de Haan, N.; Tumusiime, D.; Jost, C.; Bett, B. A Qualitative Study on Gendered Barriers to Livestock Vaccine Uptake in Kenya and Uganda and Their Implications on Rift Valley Fever Control. Vaccines 2019, 7, 86. [Google Scholar] [CrossRef]
- Sadio, Y.A. Etude des Facteurs Affectant la Vaccination des Ruminants Domestiques dans les Régions de Sikasso et Mopti, Mali. Master’s Thesis, Cheikh Anta Diop University, Dakar, Senegal, 2018. Animal Production and Sustainable Development (Animal Production Engineering). Available online: https://cgspace.cgiar.org/handle/10568/97909 (accessed on 14 November 2022).
- Donadeu, M.; Nwankpa, N.; Abela-Ridder, B.; Dungu, B. Strategies to increase adoption of animal vaccines by smallholder farmers with focus on neglected diseases and marginalized populations. PLoS Negl. Trop. Dis. 2019, 13, e0006989, Erratum in: PLoS Negl. Trop. Dis. 2019, 13, e0007279. [Google Scholar] [CrossRef]
- Dione, M.M.; Traoré, I.; Kassambara, H.; Sow, A.N.; Touré, C.O.; Sidibé, C.A.K.; Séry, A.; Yena, A.S.; Wieland, B.; Dakouo, M.; et al. Integrated Approach to Facilitate Stakeholder Participation in the Control of Endemic Diseases of Livestock: The Case of Peste Des Petits Ruminants in Mali. Front. Veter.-Sci. 2019, 6, 392. [Google Scholar] [CrossRef]
- Kitching, R.P. Vaccines for lumpy skin disease, sheep pox and goat pox. Dev. Biol. 2003, 114, 161–167. [Google Scholar]
- Amanova, Z.; Zhugunissov, K.; Barakbayev, K.; Kondybaeva, Z.; Sametova, Z.; Shayakhmetov, Y.; Kaissenov, D.; Dzhekebekov, K.; Zhunushov, A.; Abduraimov, Y.; et al. Duration of Protective Immunity in Sheep Vaccinated with a Combined Vaccine against Peste des Petits Ruminants and Sheep Pox. Vaccines 2021, 9, 912. [Google Scholar] [CrossRef]
- Achour, H.A.; Bouguedour, R.; Bouhbal, A.; Guechtouli, A.; Aouissat, M. Étude comparative du pouvoir immunisant de quelques souches atténuées de virus de la clavelée et d’un vaccin sensibiliser. Rev. Sci. Tech. Oie 2000, 19, 773–783. [Google Scholar] [CrossRef]
- Ramyar, H.; Hessami, M. Studies on the Duration of Immunity conferred by a Live-Modified Sheep Pox Tissue Culture Virus Vaccine. Zent. Veter. B. 1970, 17, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Bhanuprakash, V.; Hosamani, M.; Singh, R. Prospects of control and eradication of capripox from the Indian subcontinent: A perspective. Antivir. Res. 2011, 91, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Ngichabe, C.K.; Wamwayi, H.M.; Ndungu, E.K.; Mirangi, P.K.; Bostock, C.J.; Black, D.N.; Barrett, T. Long term immunity in African cattle vaccinated with a recombinant capripox-rinderpest virus vaccine. Epidemiol. Infect. 2002, 128, 343–349. [Google Scholar] [CrossRef]
- Sohier, C.; Haegeman, A.; Mostin, L.; De Leeuw, I.; Van Campe, W.; De Vleeschauwer, A.; Tuppurainen, E.S.M.; van den Berg, T.; De Regge, N.; De Clercq, K. Experimental evidence of mechanical Lumpy Skin Disease virus transmission by Stomoxys calcitrans biting flies and Haematopota spp. horseflies. Sci. Rep. 2019, 9, 20076. [Google Scholar] [CrossRef]
- Sanz-Bernardo, B.; Haga, I.R.; Wijesiriwardana, N.; Basu, S.; Larner, W.; Diaz, A.V.; Langlands, Z.; Denison, E.; Stoner, J.; White, M.; et al. Quantifying and Modeling the Acquisition and Retention of Lumpy Skin Disease Virus by Hematophagus Insects Reveals Clinically but Not Subclinically Affected Cattle Are Promoters of Viral Transmission and Key Targets for Control of Disease Outbreaks. J. Virol. 2021, 95, e02239-20. [Google Scholar] [CrossRef]
- Davies, F.G. Lumpy skin disease of cattle: A growing problem in Africa and the Near East. World Anim. Rev. 1991, 68, 37–42. [Google Scholar]
- Haegeman, A.; De Leeuw, I.; Mostin, L.; Van Campe, W.; Aerts, L.; Vastag, M.; De Clercq, K. An Immunoperoxidase Monolayer Assay (IPMA) for the detection of lumpy skin disease antibodies. J. Virol. Methods 2020, 277, 113800. [Google Scholar] [CrossRef] [PubMed]
- Spearman, C. The method of “right and wrong cases” (constant stimuli) without Gauss’s formula. Br. J. Psychol. 1908, II, 227–242. [Google Scholar] [CrossRef]
- Kärber, G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Archiv. Experiment. Pathol. Pharmakol. 1931, 162, 480–483. [Google Scholar] [CrossRef]
- Haegeman, A.; Zro, K.; Vandenbussche, F.; Demeestere, L.; Van Campe, W.; Ennaji, M.; De Clercq, K. Development and validation of three Capripoxvirus real-time PCRs for parallel testing. J. Virol. Methods 2013, 193, 446–451. [Google Scholar] [CrossRef]
- Agianniotaki, E.I.; Chaintoutis, S.C.; Haegeman, A.; Tasioudi, K.E.; De Leeuw, I.; Katsoulos, P.-D.; Sachpatzidis, A.; De Clercq, K.; Alexandropoulos, T.; Polizopoulou, Z.S.; et al. Development and validation of a TaqMan probe-based real-time PCR method for the differentiation of wild type lumpy skin disease virus from vaccine virus strains. J. Virol. Methods 2017, 249, 48–57. [Google Scholar] [CrossRef]
- Parida, S.; Oh, Y.; Reid, S.; Cox, S.; Statham, R.; Mahapatra, M.; Anderson, J.; Barnett, P.; Charleston, B.; Paton, D. Interferon-γ production in vitro from whole blood of foot-and-mouth disease virus (FMDV) vaccinated and infected cattle after incubation with inactivated FMDV. Vaccine 2006, 24, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Regulation (EU) 2019/6. Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on veterinary medicinal products and repealing Directive 2001/82/EC. Off. J. Eur. Union. 2018, L4, 43–167. [Google Scholar]
- EMEA/CVMP/682/99-Final. Committee for Veterinary Medicinal Products, Note for Guidance: Duration of Protection Achieved by Veterinary Vaccines; European Agency for the Evaluation of Medicinal Products: Amsterdam, The Netherlands, 2000. [Google Scholar]
- Calistri, P.; Declercq, K.; De Vleeschauwer, A.; Gubbins, S.; Klement, E.; Stegeman, A.; Abrahantes, J.C.; Antoniou, S.-E.; Broglia, A.; Gogin, A. Lumpy skin disease: Scientific and technical assistance on control and surveillance activities. EFSA J. 2018, 16, e05452. [Google Scholar] [CrossRef]
- Abera, Z.; Degefu, H.; Gari, G.; Ayana, Z. Review on Epidemiology and Economic Importance of Lumpy Skin Disease. Int. J. Basic Appl. Virol. 2015, 4, 8–21. [Google Scholar] [CrossRef]
- Elhaig, M.M.; Selim, A.; Mahmoud, M. Lumpy skin disease in cattle: Frequency of occurrence in a dairy farm and a preliminary assessment of its possible impact on Egyptian buffaloes. Onderstepoort J. Veter.-Res. 2017, 84, e1–e6. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, P.S.; Mardani, K.; Dalir-Naghadeh, B.; Jalilzadeh-Amin, G. Epidemiological Study of Lumpy Skin Disease Outbreaks in North-western Iran. Transbound. Emerg. Dis. 2017, 64, 1782–1789. [Google Scholar] [CrossRef] [PubMed]
- Molla, W.; Frankena, K.; Gari, G.; Kidane, M.; Shegu, D.; de Jong, M.C. Seroprevalence and risk factors of lumpy skin disease in Ethiopia. Prev. Veter.-Med. 2018, 160, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Şevik, M.; Doğan, M. Epidemiological and Molecular Studies on Lumpy Skin Disease Outbreaks in Turkey during 2014–2015. Transbound. Emerg. Dis. 2017, 64, 1268–1279. [Google Scholar] [CrossRef]
- Sudhakar, S.B.; Mishra, N.; Kalaiyarasu, S.; Jhade, S.K.; Hemadri, D.; Sood, R.; Bal, G.C.; Nayak, M.K.; Pradhan, S.K.; Singh, V.P. Lumpy skin disease (LSD) outbreaks in cattle in Odisha state, India in August 2019: Epidemiological features and molecular studies. Transbound. Emerg. Dis. 2020, 67, 2408–2422. [Google Scholar] [CrossRef]
- Wolff, J.; Tuppurainen, E.; Adedeji, A.; Meseko, C.; Asala, O.; Adole, J.; Atai, R.; Dogonyaro, B.; Globig, A.; Hoffmann, D.; et al. Characterization of a Nigerian Lumpy Skin Disease Virus Isolate after Experimental Infection of Cattle. Pathogens 2021, 11, 16. [Google Scholar] [CrossRef]
- Bernardo, B.S.; Haga, I.R.; Wijesiriwardana, N.; Hawes, P.C.; Simpson, J.; Morrison, L.R.; MacIntyre, N.; Brocchi, E.; Atkinson, J.; Haegeman, A.; et al. Lumpy Skin Disease Is Characterized by Severe Multifocal Dermatitis With Necrotizing Fibrinoid Vasculitis Following Experimental Infection. Veter.-Pathol. 2020, 57, 388–396. [Google Scholar] [CrossRef]
- Möller, J.; Moritz, T.; Schlottau, K.; Krstevski, K.; Hoffmann, D.; Beer, M.; Hoffmann, B. Experimental lumpy skin disease virus infection of cattle: Comparison of a field strain and a vaccine strain. Arch. Virol. 2019, 164, 2931–2941. [Google Scholar] [CrossRef]
- Babiuk, S.; Bowden, T.R.; Parkyn, G.; Dalman, B.; Manning, L.; Neufeld, J.; Embury-Hyatt, C.; Copps, J.; Boyle, D.B. Quantification of Lumpy Skin Disease Virus Following Experimental Infection in Cattle. Transbound. Emerg. Dis. 2008, 55, 299–307. [Google Scholar] [CrossRef]
- Kononov, A.; Prutnikov, P.; Shumilova, I.; Kononova, S.; Nesterov, A.; Byadovskaya, O.; Pestova, Y.; Diev, V.; Sprygin, A. Determination of lumpy skin disease virus in bovine meat and offal products following experimental infection. Transbound. Emerg. Dis. 2019, 66, 1332–1340. [Google Scholar] [CrossRef]
- Katsoulos, P.-D.; Chaintoutis, S.C.; Dovas, C.I.; Polizopoulou, Z.S.; Brellou, G.D.; Agianniotaki, E.I.; Tasioudi, K.E.; Chondrokouki, E.; Papadopoulos, O.; Karatzias, H.; et al. Investigation on the incidence of adverse reactions, viraemia and haematological changes following field immunization of cattle using a live attenuated vaccine against lumpy skin disease. Transbound. Emerg. Dis. 2018, 65, 174–185. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Lumpy skin disease: I. Data collection and analysis. EFSA J. 2017, 15, e04773. [Google Scholar] [CrossRef]
- Bamouh, Z.; Hamdi, J.; Fellahi, S.; Khayi, S.; Jazouli, M.; Tadlaoui, K.; Fihri, O.; Tuppurainen, E.; Elharrak, M. Investigation of Post Vaccination Reactions of Two Live Attenuated Vaccines against Lumpy Skin Disease of Cattle. Vaccines 2021, 9, 621. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, I.; Garigliany, M.; Bertels, G.; Willems, T.; Desmecht, D.; De Clercq, K. Bluetongue Virus RNA Detection by Real-Time RT-PCR in Post-Vaccination Samples from Cattle. Transbound. Emerg. Dis. 2015, 62, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Carn, V.M. Control of capripoxvirus infections. Vaccine 1993, 11, 1275–1279. [Google Scholar] [CrossRef] [PubMed]
- Costa-Pereira, C.; Moreira, M.L.; Soares, R.P.; Marteleto, B.H.; Ribeiro, V.M.; França-Dias, M.H.; Cardoso, L.M.; Viana, K.F.; Giunchetti, R.C.; Martins-Filho, A.O.; et al. One-year timeline kinetics of cytokine-mediated cellular immunity in dogs vaccinated against visceral leishmaniasis. BMC Veter.-Res. 2015, 11, 92. [Google Scholar] [CrossRef]
- Mohn, K.G.-I.; Bredholt, G.; Brokstad, K.A.; Pathirana, R.D.; Aarstad, H.J.; Tøndel, C.; Cox, R.J. Longevity of B-Cell and T-Cell Responses After Live Attenuated Influenza Vaccination in Children. J. Infect. Dis. 2015, 211, 1541–1549. [Google Scholar] [CrossRef]
- Eneslätt, K.; Rietz, C.; Rydén, P.; Stöven, S.; House, R.V.; Wolfraim, L.A.; Tärnvik, A.; Sjöstedt, A. Persistence of cell-mediated immunity three decades after vaccination with the live vaccine strain of Francisella tularensis. Eur. J. Immunol. 2011, 41, 974–980. [Google Scholar] [CrossRef]
- Abdelwahab, M.G.; Khafagy, H.A.; Moustafa, A.M.; Saad, M.A. Evaluation of humoral and cell-mediated immunity of lumpy skin disease vaccine prepared from local strain in calves and its related to maternal immunity. J. Am. Sci. 2016, 12, 38–45. [Google Scholar] [CrossRef]
- Milovanović, M.; Dietze, K.; Milićević, V.; Radojičić, S.; Valčić, M.; Moritz, T.; Hoffmann, B. Humoral immune response to repeated lumpy skin disease virus vaccination and performance of serological tests. BMC Veter.-Res. 2019, 15, 80. [Google Scholar] [CrossRef]
- Sólyom, F.; Perenlei, L.; Roith, J. Sheep-pox vaccine prepared from formaldehyde inactivated virus adsorbed to aluminium hydroxide gel. Acta Microbiol. Acad. Sci. Hung. 1982, 29, 69–75. [Google Scholar]
- Awad, M.; Michael, A.; Soliman, S.M.; Samir, S.S.; Daoud, A.M. Trials for preparation of inactivated sheep pox vaccine using binary ethyleneimine. Egypt. J. Immunol. 2003, 10, 67–72. [Google Scholar] [PubMed]
- Boumart, Z.; Daouam, S.; Belkourati, I.; Rafi, L.; Tuppurainen, E.; Tadlaoui, K.O.; El Harrak, M. Comparative innocuity and efficacy of live and inactivated sheeppox vaccines. BMC Veter. Res. 2016, 12, 133. [Google Scholar] [CrossRef] [PubMed]
- Es-Sadeqy, Y.; Bamouh, Z.; Ennahli, A.; Safini, N.; El Mejdoub, S.; Tadlaoui, K.O.; Gavrilov, B.; El Harrak, M. Development of an inactivated combined vaccine for protection of cattle against lumpy skin disease and bluetongue viruses. Veter.-Microbiol. 2021, 256, 109046. [Google Scholar] [CrossRef]
- Belsham, G.J. Towards improvements in foot-and-mouth disease vaccine performance. Acta Veter.-Scand. 2020, 62, 20. [Google Scholar] [CrossRef]
- Maradei, E.; La Torre, J.; Robiolo, B.; Esteves, J.; Seki, C.; Pedemonte, A.; Iglesias, M.; D’Aloia, R.; Mattion, N. Updating of the correlation between lpELISA titers and protection from virus challenge for the assessment of the potency of polyvalent aphtovirus vaccines in Argentina. Vaccine 2008, 26, 6577–6586. [Google Scholar] [CrossRef]
- Kitching, P.; Hammond, J.; Jeggo, M.; Charleston, B.; Paton, D.; Rodriguez, L.; Heckert, R. Global FMD control—Is it an option? Vaccine 2007, 25, 5660–5664. [Google Scholar] [CrossRef] [PubMed]
- Aziz-Boaron, O.; Leibovitz, K.; Gelman, B.; Kedmi, M.; Klement, E. Safety, Immunogenicity and Duration of Immunity Elicited by an Inactivated Bovine Ephemeral Fever Vaccine. PLoS ONE 2013, 8, e82217. [Google Scholar] [CrossRef]
- WOAH. Frequently Asked Questions (FAQ) on Lumpy skin disease (LSD) Vaccination. Available online: https://rr-asia.woah.org/wp-content/uploads/2021/09/faq_on_lsd_vaccination.pdf (accessed on 14 November 2022).

| Type of Vaccine | Duration of Immunity Examined | Trial | Group Acronym | Animal Id | Nr. of Animals |
|---|---|---|---|---|---|
| Live attenuated vaccine | 18 months | 1 | LSDV LAV 18 | R1 to R6 | 6 |
| 12 months | 1 | LSDV LAV 12 | R7 to R12 | 6 | |
| 6 months | 2 | LSDV LAV 6 | R13 to R18 | 6 | |
| Inactivated vaccine | 12 months | 2 | LSDV Inac 12 | R1 to R6 | 6 |
| 6 months | 2 | LSDV Inac 6 | R7 to R12 | 6 | |
| Controls | |||||
| Unvaccinated group | 1 | LSDV con1 | R1 to R5 | 5 | |
| 2 | LSDV con2 | R1 to R5 | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haegeman, A.; De Leeuw, I.; Mostin, L.; Van Campe, W.; Philips, W.; Elharrak, M.; De Regge, N.; De Clercq, K. Duration of Immunity Induced after Vaccination of Cattle with a Live Attenuated or Inactivated Lumpy Skin Disease Virus Vaccine. Microorganisms 2023, 11, 210. https://doi.org/10.3390/microorganisms11010210
Haegeman A, De Leeuw I, Mostin L, Van Campe W, Philips W, Elharrak M, De Regge N, De Clercq K. Duration of Immunity Induced after Vaccination of Cattle with a Live Attenuated or Inactivated Lumpy Skin Disease Virus Vaccine. Microorganisms. 2023; 11(1):210. https://doi.org/10.3390/microorganisms11010210
Chicago/Turabian StyleHaegeman, Andy, Ilse De Leeuw, Laurent Mostin, Willem Van Campe, Wannes Philips, Mehdi Elharrak, Nick De Regge, and Kris De Clercq. 2023. "Duration of Immunity Induced after Vaccination of Cattle with a Live Attenuated or Inactivated Lumpy Skin Disease Virus Vaccine" Microorganisms 11, no. 1: 210. https://doi.org/10.3390/microorganisms11010210
APA StyleHaegeman, A., De Leeuw, I., Mostin, L., Van Campe, W., Philips, W., Elharrak, M., De Regge, N., & De Clercq, K. (2023). Duration of Immunity Induced after Vaccination of Cattle with a Live Attenuated or Inactivated Lumpy Skin Disease Virus Vaccine. Microorganisms, 11(1), 210. https://doi.org/10.3390/microorganisms11010210





