Intervention Strategies to Control Campylobacter at Different Stages of the Food Chain
Abstract
1. Introduction
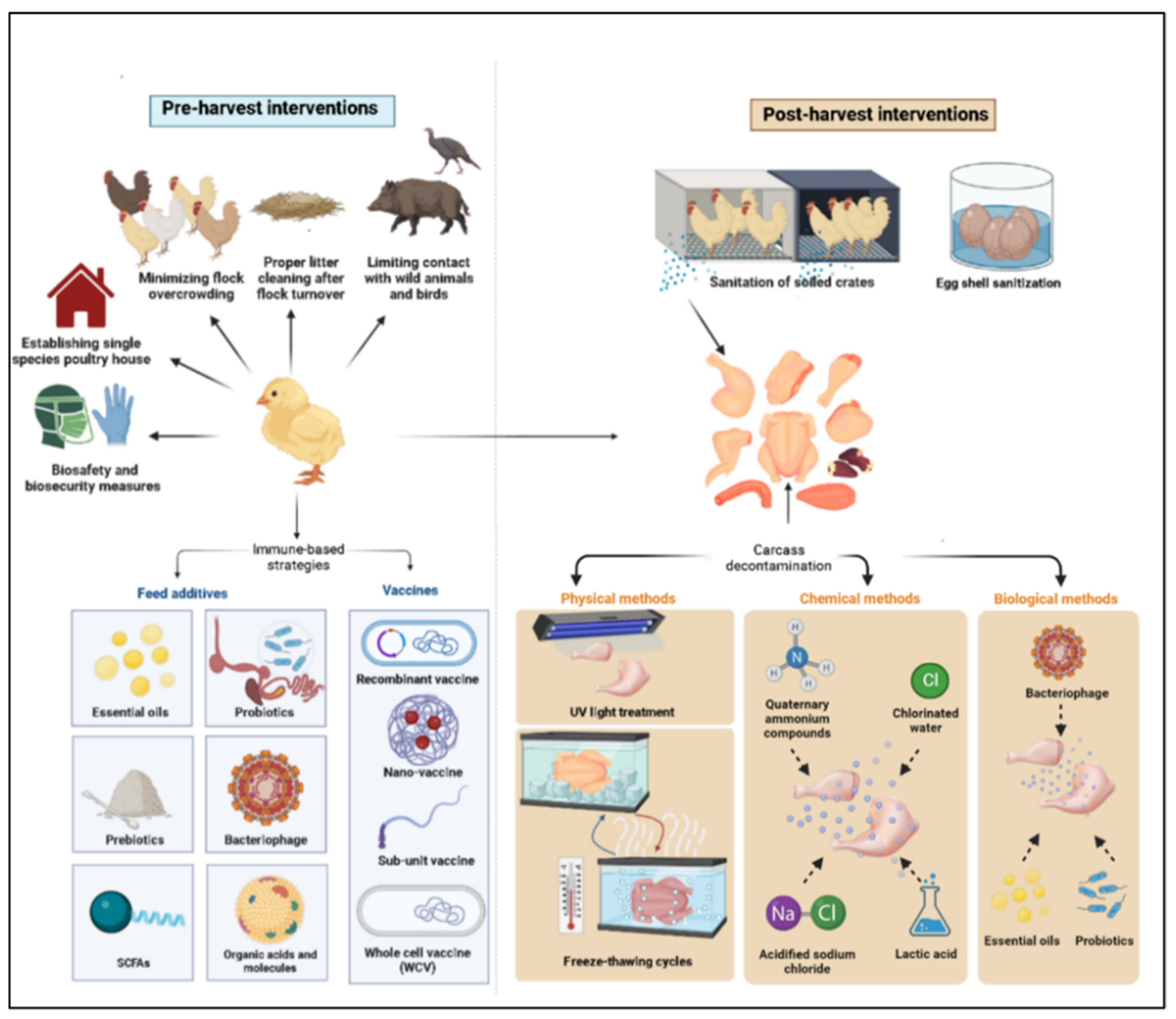
2. Pre-Harvest Control Measures (On-Farm Control)
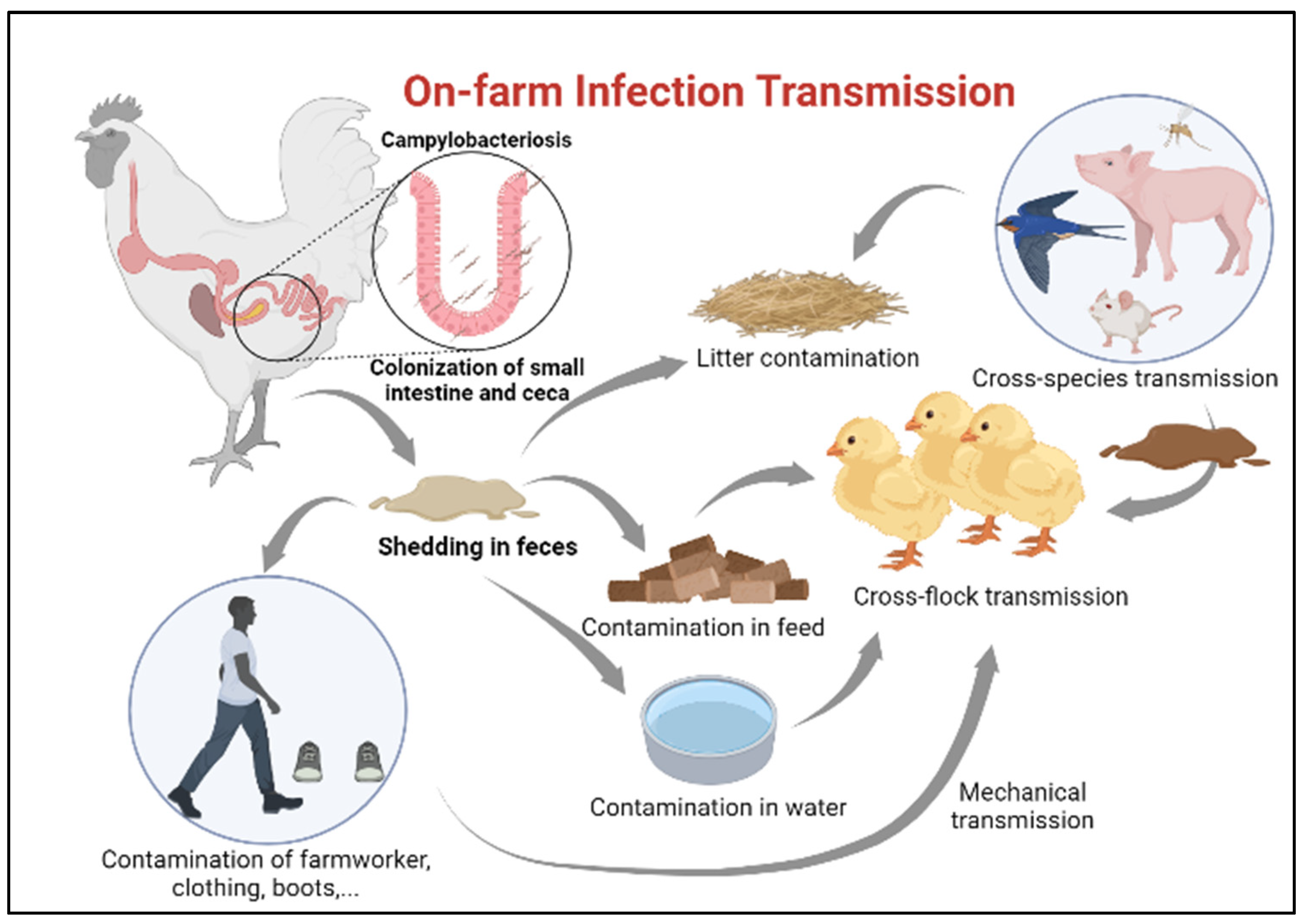
2.1. On-Farm Biosecurity Measures
2.2. Immune-Based Strategies to Control Campylobacter Colonization in Poultry Flocks
2.2.1. Vaccines
Whole Cell Vaccines (WCV)
Subunit Vaccines
Recombinant Vaccines
Nanoparticles-Based Vaccines
2.2.2. Feed Additives
Prebiotics
Probiotics and Their Products
Synbiotics
Essential Oils
Organic Acids
Small Molecule Inhibitors
Short Chain Fatty Acids
Bacteriophages
2.2.3. Fecal Microbial Transplant and Microbial Consortia
3. Post-Harvest Control Measures (Production Chain Interventions)
3.1. Slaughter Plants Cleaning and Sanitation
3.2. Carcass Decontamination
3.2.1. Physical and Chemical Methods
3.2.2. Biological Methods
3.3. Eggshell Sanitation
4. Conclusions and Future Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Silva, J.; Leite, D.; Fernandes, M.; Mena, C.; Gibbs, P.A.; Teixeira, P. Campylobacter spp. as a foodborne pathogen: A review. Front. Microbiol. 2011, 2, 200. [Google Scholar] [CrossRef] [PubMed]
- Fernández, H.; Lobos, M.; Concha, M. Inducing enterotoxigenic properties in Campylobacter jejuni and Campylobacter coli by serial intraperitoneal passage in mice. Memórias Do Inst. Oswaldo Cruz 1999, 94, 101–102. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Facciolà, A.; Riso, R.; Avventuroso, E.; Visalli, G.; Delia, S.; Laganà, P. Campylobacter: From microbiology to prevention. J. Prev. Med. Hyg. 2017, 58, E79. [Google Scholar]
- Acheson, D.; Allos, B.M. Campylobacter jejuni infections: Update on emerging issues and trends. Clin. Infect. Dis. 2001, 32, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Terefe, Y.; Deblais, L.; Ghanem, M.; Helmy, Y.A.; Mummed, B.; Chen, D.; Singh, N.; Ahyong, V.; Kalantar, K.; Yimer, G.; et al. Co-occurrence of Campylobacter Species in Children From Eastern Ethiopia, and Their Association With Environmental Enteric Dysfunction, Diarrhea, and Host Microbiome. Front. Public Health 2020, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.S.; Blaser, M.J. Fatalities associated with Campylobacter jejuni infections. JAMA 1985, 253, 2873–2875. [Google Scholar] [CrossRef]
- Peters, S.; Pascoe, B.; Wu, Z.; Bayliss, S.C.; Zeng, X.; Edwinson, A.; Veerabadhran-Gurunathan, S.; Jawahir, S.; Calland, J.K.; Mourkas, E. Campylobacter jejuni genotypes are associated with post-infection irritable bowel syndrome in humans. Commun. Biol. 2021, 4, 1015. [Google Scholar] [CrossRef]
- Malik, A.; Brudvig, J.M.; Gadsden, B.J.; Ethridge, A.D.; Mansfield, L.S. Campylobacter jejuni induces autoimmune peripheral neuropathy via Sialoadhesin and Interleukin-4 axes. Gut Microbes 2022, 14, 2064706. [Google Scholar] [CrossRef]
- Kassem, I.; Helmy, Y.A.; Kashoma, I.P.; Rajashekara, G. The emergence of antibiotic resistance on poultry farms. In AchievingSustainable Production of Poultry Meat: Safety, Quality and Sustainability; Ricke, S., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2016; Volume 1, ISBN 978-1-78676-064-7. [Google Scholar]
- Nachamkin, I.; Allos, B.M.; Ho, T. Campylobacter species and Guillain-Barre syndrome. Clin. Microbiol. Rev. 1998, 11, 555–567. [Google Scholar] [CrossRef]
- Epps, S.V.; Harvey, R.B.; Hume, M.E.; Phillips, T.D.; Anderson, R.C.; Nisbet, D.J. Foodborne Campylobacter: Infections, metabolism, pathogenesis and reservoirs. Int. J. Environ. Res. Public Health 2013, 10, 6292–6304. [Google Scholar] [CrossRef]
- Kassem, I.I.; Kehinde, O.O.; Helmy, Y.A.; Kumar, A.; Chandrashekhar, K.; Pina-Mimbela, R.; Rajashekara, G. Campylobacter in poultry: The conundrums of highly adaptable and ubiquitous foodborne pathogens. In Foodborne Diseases: Case Studies of Outbreaks in the Agri-Food Industries; Mei Soon, J., Manning, L., Wallace, C.A., Eds.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Buzby, J.C.; Allos, B.M.; Roberts, T. The economic burden of Campylobacter-associated Guillain-Barre syndrome. J. Infect. Dis. 1997, 176, S192–S197. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, M.; Chatham-Stephens, K.; Geissler, A. Chapter 4: Travel-Related Infectious Disease: Campylobacteriosis. In Centers for Disease Control and Prevention. CDC Yellow Book 2020: Health Information for International Travel; Oxford University Press: Oxford, UK, 2017; pp. 180–181. [Google Scholar]
- Wilson, D.J.; Gabriel, E.; Leatherbarrow, A.J.; Cheesbrough, J.; Gee, S.; Bolton, E.; Fox, A.; Fearnhead, P.; Hart, C.A.; Diggle, P.J. Tracing the source of campylobacteriosis. PLoS Genet. 2008, 4, e1000203. [Google Scholar] [CrossRef]
- van Gerwe, T. Poultry meat as a source of human campylobacteriosis. Tijdschr. Voor Diergeneeskd. 2012, 137, 172–176. [Google Scholar]
- Hailu, W.; Helmy, Y.A.; Carney-Knisely, G.; Kauffman, M.; Fraga, D.; Rajashekara, G. Prevalence and Antimicrobial Resistance Profiles of Foodborne Pathogens Isolated from Dairy Cattle and Poultry Manure Amended Farms in Northeastern Ohio, the United States. Antibiotics 2021, 10, 1450. [Google Scholar] [CrossRef] [PubMed]
- Hermans, D.; Van Deun, K.; Martel, A.; Van Immerseel, F.; Messens, W.; Heyndrickx, M.; Haesebrouck, F.; Pasmans, F. Colonization factors of Campylobacter jejuni in the chicken gut. Vet. Res. 2011, 42, 82. [Google Scholar] [CrossRef] [PubMed]
- Awad, W.A.; Molnár, A.; Aschenbach, J.R.; Ghareeb, K.; Khayal, B.; Hess, C.; Liebhart, D.; Dublecz, K.; Hess, M. Campylobacter infection in chickens modulates the intestinal epithelial barrier function. Innate Immun. 2015, 21, 151–160. [Google Scholar] [CrossRef]
- Newell, D.; Fearnley, C. Sources of Campylobacter colonization in broiler chickens. Appl. Environ. Microbiol. 2003, 69, 4343–4351. [Google Scholar] [CrossRef] [PubMed]
- Connerton, P.L.; Richards, P.J.; Lafontaine, G.M.; O’Kane, P.M.; Ghaffar, N.; Cummings, N.J.; Smith, D.L.; Fish, N.M.; Connerton, I.F. The effect of the timing of exposure to Campylobacter jejuni on the gut microbiome and inflammatory responses of broiler chickens. Microbiome 2018, 6, 88. [Google Scholar] [CrossRef] [PubMed]
- Newell, D.; Elvers, K.; Dopfer, D.; Hansson, I.; Jones, P.; James, S.; Gittins, J.; Stern, N.; Davies, R.; Connerton, I. Biosecurity-based interventions and strategies to reduce Campylobacter spp. on poultry farms. Appl. Environ. Microbiol. 2011, 77, 8605–8614. [Google Scholar] [CrossRef]
- Meunier, M.; Guyard-Nicodème, M.; Dory, D.; Chemaly, M. Control strategies against C ampylobacter at the poultry production level: Biosecurity measures, feed additives and vaccination. J. Appl. Microbiol. 2016, 120, 1139–1173. [Google Scholar] [CrossRef]
- Dai, L.; Sahin, O.; Grover, M.; Zhang, Q. New and alternative strategies for the prevention, control, and treatment of antibiotic-resistant Campylobacter. Transl. Res. 2020, 223, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Marmion, M.; Ferone, M.; Wall, P.; Scannell, A. On farm interventions to minimise Campylobacter spp. contamination in chicken. Br. Poult. Sci. 2021, 62, 53–67. [Google Scholar] [CrossRef]
- Hakeem, M.J.; Lu, X. Survival and control of Campylobacter in poultry production environment. Front. Cell. Infect. Microbiol. 2021, 10, 615049. [Google Scholar] [CrossRef]
- Barrios, P.R.; Reiersen, J.; Lowman, R.; Bisaillon, J.-R.; Michel, P.; Fridriksdóttir, V.; Gunnarsson, E.; Stern, N.; Berke, O.; McEwen, S. Risk factors for Campylobacter spp. colonization in broiler flocks in Iceland. Prev. Vet. Med. 2006, 74, 264–278. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.G.; Bueschkens, D.H. Horizontal spread of human and poultry-derived strains of Campylobacter jejuni among broiler chicks held in incubators and shipping boxes. J. Food Prot. 1988, 51, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, A.S.; Shivaprasad, H.; Schaberg, D.; Wier, F.; Weber, S.; Bandli, D. Campylobacter jejuni infection in broiler chickens. Avian Dis. 2006, 50, 55–58. [Google Scholar] [CrossRef]
- Guerin, M.T.; Martin, W.; Reiersen, J.; Berke, O.; McEwen, S.A.; Bisaillon, J.-R.; Lowman, R. A farm-level study of risk factors associated with the colonization of broiler flocks with Campylobacter spp. in Iceland, 2001–2004. Acta Vet. Scand. 2007, 49, 18. [Google Scholar] [CrossRef]
- Ellis-Iversen, J.; Ridley, A.; Morris, V.; Sowa, A.; Harris, J.; Atterbury, R.; Sparks, N.; Allen, V. Persistent environmental reservoirs on farms as risk factors for Campylobacter in commercial poultry. Epidemiol. Infect. 2012, 140, 916–924. [Google Scholar] [CrossRef]
- Hansson, I.; Sandberg, M.; Habib, I.; Lowman, R.; Engvall, E.O. Knowledge gaps in control of Campylobacter for prevention of campylobacteriosis. Transbound. Emerg. Dis. 2018, 65, 30–48. [Google Scholar] [CrossRef]
- Sibanda, N.; McKenna, A.; Richmond, A.; Ricke, S.C.; Callaway, T.; Stratakos, A.C.; Gundogdu, O.; Corcionivoschi, N. A review of the effect of management practices on Campylobacter prevalence in poultry farms. Front. Microbiol. 2018, 9, 2002. [Google Scholar] [CrossRef]
- Battersby, T.; Whyte, P.; Bolton, D. The pattern of Campylobacter contamination on broiler farms; external and internal sources. J. Appl. Microbiol. 2016, 120, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Kassem, I.I.; Helmy, Y.A.; Kathayat, D.; Candelero-Rueda, R.A.; Kumar, A.; Deblais, L.; Huang, H.C.; Sahin, O.; Zhang, Q.; Rajashekara, G. Nonculturability Might Underestimate the Occurrence of Campylobacter in Broiler Litter. Foodborne Pathog. Dis. 2017, 14, 472–477. [Google Scholar] [CrossRef]
- Bailey, M.A.; Bourassa, D.V.; Krehling, J.T.; Munoz, L.; Chasteen, K.S.; Escobar, C.; Macklin, K.S. Effects of Common Litter Management Practices on the Prevalence of Campylobacter jejuni in Broilers. Animals 2022, 12, 858. [Google Scholar] [CrossRef]
- Callicott, K.A.; Friðriksdóttir, V.; Reiersen, J.; Lowman, R.; Bisaillon, J.-R.; Gunnarsson, E.; Berndtson, E.; Hiett, K.L.; Needleman, D.S.; Stern, N.J. Lack of evidence for vertical transmission of Campylobacter spp. in chickens. Appl. Environ. Microbiol. 2006, 72, 5794–5798. [Google Scholar] [CrossRef] [PubMed]
- Gharbi, M.; Béjaoui, A.; Ben Hamda, C.; Alaya, N.; Hamrouni, S.; Bessoussa, G.; Ghram, A.; Maaroufi, A. Campylobacter spp. in Eggs and Laying Hens in the North-East of Tunisia: High Prevalence and Multidrug-Resistance Phenotypes. Vet. Sci. 2022, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Rossi, D.A.; Fonseca, B.B.; Melo, R.T.d.; Felipe, G.d.S.; Silva, P.L.d.; Mendonça, E.P.; Filgueiras, A.L.L.; Beletti, M.E. Transmission of Campylobacter coli in chicken embryos. Braz. J. Microbiol. 2012, 43, 535–543. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fonseca, B.; Beletti, M.; Melo, R.; Mendonca, E.; Vieira, C.; Levenhagen, M.; Rossi, D. Transfer, viability and colonisation of Campylobacter jejuni in the chicken vitellus and in embryos. Br. Poult. Sci. 2011, 52, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Royden, A.; Christley, R.; Prendiville, A.; Williams, N.J. The Role of Biosecurity in the Control of Campylobacter: A Qualitative Study of the Attitudes and Perceptions of UK Broiler Farm Workers. Front. Vet. Sci. 2021, 8, 1552. [Google Scholar] [CrossRef]
- Hertogs, K.; Heyndrickx, M.; Gelaude, P.; De Zutter, L.; Dewulf, J.; Rasschaert, G. The effect of partial depopulation on Campylobacter introduction in broiler houses. Poult. Sci. 2021, 100, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Lynch, H.; Franklin-Hayes, P.; Koolman, L.; Egan, J.; Gutierrez, M.; Byrne, W.; Golden, O.; Bolton, D.; Reid, P.; Coffey, A. Prevalence and levels of Campylobacter in broiler chicken batches and carcasses in Ireland in 2017–2018. Int. J. Food Microbiol. 2022, 372, 109693. [Google Scholar] [CrossRef]
- Greening, S.S.; Zhang, J.; Midwinter, A.C.; Wilkinson, D.A.; Fayaz, A.; Williamson, D.A.; Anderson, M.J.; Gates, M.C.; French, N.P. Transmission dynamics of an antimicrobial resistant Campylobacter jejuni lineage in New Zealand’s commercial poultry network. Epidemics 2021, 37, 100521. [Google Scholar] [CrossRef] [PubMed]
- Widders, P.; Thomas, L.; Long, K.; Tokhi, M.; Panaccio, M.; Apos, E. The specificity of antibody in chickens immunised to reduce intestinal colonisation with Campylobacter jejuni. Vet. Microbiol. 1998, 64, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Rice, B.E.; Rollins, D.M.; Mallinson, E.T.; Carr, L.; Joseph, S.W. Campylobacter jejuni in broiler chickens: Colonization and humoral immunity following oral vaccination and experimental infection. Vaccine 1997, 15, 1922–1932. [Google Scholar] [CrossRef] [PubMed]
- Meunier, M.; Guyard-Nicodeme, M.; Vigouroux, E.; Poezevara, T.; Beven, V.; Quesne, S.; Bigault, L.; Amelot, M.; Dory, D.; Chemaly, M. Promising new vaccine candidates against Campylobacter in broilers. PLoS ONE 2017, 12, e0188472. [Google Scholar] [CrossRef] [PubMed]
- Cawthraw, S.; Newell, D. Investigation of the presence and protective effects of maternal antibodies against Campylobacter jejuni in chickens. Avian Dis. 2010, 54, 86–93. [Google Scholar] [CrossRef]
- Kashoma, I.P.; Srivastava, V.; Rajashekara, G. Advances in vaccines for controlling Campylobacter in poultry. In Food Safety in Poultry Meat Production; Springer: Berlin/Heidelberg, Germany, 2019; pp. 191–210. [Google Scholar]
- Ziprin, R.L.; Young, C.R.; Byrd, J.A.; Stanker, L.H.; Hume, M.E.; Gray, S.A.; Kim, B.J.; Konkel, M.E. Role of Campylobacter jejuni potential virulence genes in cecal colonization. Avian Dis. 2001, 45, 549–557. [Google Scholar] [CrossRef]
- Jeon, B.; Saisom, T.; Sasipreeyajan, J.; Luangtongkum, T. Live-Attenuated Oral Vaccines to Reduce Campylobacter Colonization in Poultry. Vaccines 2022, 10, 685. [Google Scholar] [CrossRef]
- Nothaft, H.; Davis, B.; Lock, Y.Y.; Perez-Munoz, M.E.; Vinogradov, E.; Walter, J.; Coros, C.; Szymanski, C.M. Engineering the Campylobacter jejuni N-glycan to create an effective chicken vaccine. Sci. Rep. 2016, 6, 26511. [Google Scholar] [CrossRef]
- Neal-McKinney, J.M.; Samuelson, D.R.; Eucker, T.P.; Nissen, M.S.; Crespo, R.; Konkel, M.E. Reducing Campylobacter jejuni colonization of poultry via vaccination. PLoS ONE 2014, 9, e114254. [Google Scholar] [CrossRef]
- Hodgins, D.C.; Barjesteh, N.; St Paul, M.; Ma, Z.; Monteiro, M.A.; Sharif, S. Evaluation of a polysaccharide conjugate vaccine to reduce colonization by Campylobacter jejuni in broiler chickens. BMC Res. Notes 2015, 8, 204. [Google Scholar] [CrossRef]
- Annamalai, T.; Pina-Mimbela, R.; Kumar, A.; Binjawadagi, B.; Liu, Z.; Renukaradhya, G.; Rajashekara, G. Evaluation of nanoparticle-encapsulated outer membrane proteins for the control of Campylobacter jejuni colonization in chickens. Poult. Sci. 2013, 92, 2201–2211. [Google Scholar] [CrossRef]
- Cui, Y.; Guo, F.; Guo, J.; Cao, X.; Wang, H.; Yang, B.; Zhou, H.; Su, X.; Zeng, X.; Lin, J.; et al. Immunization of Chickens with the Enterobactin Conjugate Vaccine Reduced Campylobacter jejuni Colonization in the Intestine. Vaccines 2020, 8, 747. [Google Scholar] [CrossRef]
- Nothaft, H.; Perez-Muñoz, M.; Gouveia, G.; Duar, R.; Wanford, J.; Lango-Scholey, L.; Panagos, C.; Srithayakumar, V.; Plastow, G.; Coros, C.J.A.; et al. Coadministration of the Campylobacter jejuni N-glycan-based vaccine with probiotics improves vaccine performance in broiler chickens. Appl. Environ. Microbiol. 2017, 83, e01523-17. [Google Scholar] [CrossRef] [PubMed]
- Buckley, A.M.; Wang, J.; Hudson, D.L.; Grant, A.J.; Jones, M.A.; Maskell, D.J.; Stevens, M.P. Evaluation of live-attenuated Salmonella vaccines expressing Campylobacter antigens for control of C. jejuni in poultry. Vaccine 2010, 28, 1094–1105. [Google Scholar] [CrossRef] [PubMed]
- Wyszyńska, A.; Raczko, A.; Lis, M.; Jagusztyn-Krynicka, E.K. Oral immunization of chickens with avirulent Salmonella vaccine strain carrying C. jejuni 72Dz/92 cjaA gene elicits specific humoral immune response associated with protection against challenge with wild-type Campylobacter. Vaccine 2004, 22, 1379–1389. [Google Scholar] [CrossRef]
- Huang, J.-l.; Yin, Y.-X.; Pan, Z.-m.; Zhang, G.; Zhu, A.-p.; Liu, X.-f.; Jiao, X.-a. Intranasal immunization with chitosan/pCAGGS-flaA nanoparticles inhibits Campylobacter jejuni in a White Leghorn model. J. Biomed. Biotechnol. 2010, 2010, 589476. [Google Scholar] [CrossRef] [PubMed]
- Kobierecka, P.A.; Wyszyńska, A.K.; Gubernator, J.; Kuczkowski, M.; Wiśniewski, O.; Maruszewska, M.; Wojtania, A.; Derlatka, K.E.; Adamska, I.; Godlewska, R. Chicken anti-Campylobacter vaccine–comparison of various carriers and routes of immunization. Front. Microbiol. 2016, 7, 740. [Google Scholar] [CrossRef]
- Taha-Abdelaziz, K.; Hodgins, D.C.; Alkie, T.N.; Quinteiro-Filho, W.; Yitbarek, A.; Astill, J.; Sharif, S. Oral administration of PLGA-encapsulated CpG ODN and Campylobacter jejuni lysate reduces cecal colonization by Campylobacter jejuni in chickens. Vaccine 2018, 36, 388–394. [Google Scholar] [CrossRef]
- Noor, S.M.; Husband, A.; Widders, P. In ovo oral vaccination with cmpylobacter jejuni establishes early development of intestinal immunity in chickens. Br. Poult. Sci. 1995, 36, 563–573. [Google Scholar] [CrossRef]
- Glünder, G.; Spiering, N.; Hinz, K. Investigations on parenteral immunization of chickens with a Campylobacter mineral oil vaccine. In Proceedings of the International Congress of the World Veterinary Poultry Association; Nagy, B., Mulder, R., Eds.; European Commission: Budapest, Hungary, 1997; pp. 247–253. [Google Scholar]
- Guerry, P. Campylobacter flagella: Not just for motility. Trends Microbiol. 2007, 15, 456–461. [Google Scholar] [CrossRef]
- Radomska, K.A.; Vaezirad, M.M.; Verstappen, K.M.; Wösten, M.M.; Wagenaar, J.A.; van Putten, J.P. Chicken immune response after in ovo immunization with chimeric TLR5 activating flagellin of Campylobacter jejuni. PLoS ONE 2016, 11, e0164837. [Google Scholar] [CrossRef] [PubMed]
- Layton, S.; Morgan, M.; Cole, K.; Kwon, Y.; Donoghue, D.; Hargis, B.; Pumford, N. Evaluation of Salmonella-vectored Campylobacter peptide epitopes for reduction of Campylobacter jejuni in broiler chickens. Clin. Vaccine Immunol. 2011, 18, 449–454. [Google Scholar] [CrossRef] [PubMed]
- de Zoete, M.R.; van Putten, J.P.; Wagenaar, J.A. Vaccination of chickens against Campylobacter. Vaccine 2007, 25, 5548–5557. [Google Scholar] [CrossRef] [PubMed]
- Hanuszkiewicz, A.; Pittock, P.; Humphries, F.; Moll, H.; Rosales, A.R.; Molinaro, A.; Moynagh, P.N.; Lajoie, G.A.; Valvano, M.A. Identification of the flagellin glycosylation system in Burkholderia cenocepacia and the contribution of glycosylated flagellin to evasion of human innate immune responses. J. Biol. Chem. 2014, 289, 19231–19244. [Google Scholar] [CrossRef]
- Monteiro, M.A.; Baqar, S.; Hall, E.R.; Chen, Y.-H.; Porter, C.K.; Bentzel, D.E.; Applebee, L.; Guerry, P. Capsule polysaccharide conjugate vaccine against diarrheal disease caused by Campylobacter jejuni. Infect. Immun. 2009, 77, 1128–1136. [Google Scholar] [CrossRef]
- Würfel, S.; Da Silva, W.; de Oliveira, M.; Kleinubing, N.; Lopes, G.; Gandra, E.; Dellagostin, O. Genetic diversity of Campylobacter jejuni and Campylobacter coli isolated from poultry meat products sold on the retail market in Southern Brazil. Poult. Sci. 2019, 98, 932–939. [Google Scholar] [CrossRef]
- Vohra, P.; Chintoan-Uta, C.; Terra, V.S.; Bremner, A.; Cuccui, J.; Wren, B.W.; Vervelde, L.; Stevens, M.P. Evaluation of glycosylated FlpA and SodB as subunit vaccines against Campylobacter jejuni colonisation in chickens. Vaccines 2020, 8, 520. [Google Scholar] [CrossRef]
- Nascimento, I.; Leite, L. Recombinant vaccines and the development of new vaccine strategies. Braz. J. Med. Biol. Res. 2012, 45, 1102–1111. [Google Scholar] [CrossRef]
- Theoret, J.R.; Cooper, K.K.; Zekarias, B.; Roland, K.L.; Law, B.F.; Curtiss III, R.; Joens, L.A. The Campylobacter jejuni Dps homologue is important for in vitro biofilm formation and cecal colonization of poultry and may serve as a protective antigen for vaccination. Clin. Vaccine Immunol. 2012, 19, 1426–1431. [Google Scholar] [CrossRef] [PubMed]
- Pei, Y.; Parreira, V.R.; Roland, K.L.; Curtiss, R.; Prescott, J.F. Assessment of attenuated Salmonella vaccine strains in controlling experimental Salmonella Typhimurium infection in chickens. Can. J. Vet. Res. 2014, 78, 23–30. [Google Scholar]
- Taha-Abdelaziz, K.; Yitbarek, A.; Alkie, T.N.; Hodgins, D.C.; Read, L.R.; Weese, J.S.; Sharif, S. PLGA-encapsulated CpG ODN and Campylobacter jejuni lysate modulate cecal microbiota composition in broiler chickens experimentally challenged with C. jejuni. Sci. Rep. 2018, 8, 12076. [Google Scholar] [CrossRef]
- Pumtang-On, P.; Mahony, T.J.; Hill, R.A.; Vanniasinkam, T. A systematic review of Campylobacter jejuni vaccine candidates for chickens. Microorganisms 2021, 9, 397. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Rappuoli, R. Reverse vaccinology: Developing vaccines in the era of genomics. Immunity 2010, 33, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.; Naik, S.; Vakil, B. Probiotics, prebiotics and synbiotics-a review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef] [PubMed]
- Teng, P.-Y.; Kim, W.K. Roles of prebiotics in intestinal ecosystem of broilers. Front. Vet. Sci. 2018, 5, 245. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.A.; Jang, M.J.; Kim, S.Y.; Yang, Y.; Pavlidis, H.O.; Ricke, S.C. Potential for prebiotics as feed additives to limit foodborne Campylobacter establishment in the poultry gastrointestinal tract. Front. Microbiol. 2019, 10, 91. [Google Scholar] [CrossRef] [PubMed]
- Van den Abbeele, P.; Duysburgh, C.; Rakebrandt, M.; Marzorati, M. Dried yeast cell walls high in beta-glucan and mannan-oligosaccharides positively affect microbial composition and activity in the canine gastrointestinal tract in vitro. J. Anim. Sci. 2020, 98, skaa173. [Google Scholar] [CrossRef]
- Froebel, L.; Jalukar, S.; Lavergne, T.; Lee, J.; Duong, T. Administration of dietary prebiotics improves growth performance and reduces pathogen colonization in broiler chickens. Poult. Sci. 2019, 98, 6668–6676. [Google Scholar] [CrossRef]
- Baurhoo, B.; Phillip, L.; Ruiz-Feria, C. Effects of purified lignin and mannan oligosaccharides on intestinal integrity and microbial populations in the ceca and litter of broiler chickens. Poult. Sci. 2007, 86, 1070–1078. [Google Scholar] [CrossRef]
- Solis-Cruz, B.; Hernandez-Patlan, D.; Hargis, B.M.; Tellez, G. Use of prebiotics as an alternative to antibiotic growth promoters in the poultry industry. In Prebiotics and Probiotics-Potential Benefits in Nutrition and Health; IntechOpen: London, UK, 2019. [Google Scholar]
- Yusrizal, Y.; Chen, T. Effect of adding chicory fructans in feed on fecal and intestinal microflora and excreta volatile ammonia. Int. J. Poult. Sci. 2003, 2, 188–194. [Google Scholar]
- Park, S.H.; Lee, S.I.; Kim, S.A.; Christensen, K.; Ricke, S.C. Comparison of antibiotic supplementation versus a yeast-based prebiotic on the cecal microbiome of commercial broilers. PLoS ONE 2017, 12, e0182805. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.R.; Gaghan, C.; Gorrell, K.; Sharif, S.; Taha-Abdelaziz, K. Probiotics as Alternatives to Antibiotics for the Prevention and Control of Necrotic Enteritis in Chickens. Pathogens 2022, 11, 692. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, M.; Shojadoost, B.; Boodhoo, N.; Astill, J.; Taha-Abdelaziz, K.; Hodgins, D.C.; Kulkarni, R.R.; Sharif, S. Necrotic enteritis in chickens: A review of pathogenesis, immune responses and prevention, focusing on probiotics and vaccination. Anim. Health Res. Rev. 2021, 22, 147–162. [Google Scholar] [CrossRef]
- Kulkarni, R.R.; Taha-Abdelaziz, K.; Shojadoost, B.; Astill, J.; Sharif, S. Gastrointestinal diseases of poultry: Causes and nutritional strategies for prevention and control. In Improving Gut Health in Poultry; Burleigh Dodds Science Publishing: Cambridge, UK, 2019; pp. 205–236. [Google Scholar]
- Alizadeh, M.; Shojadoost, B.; Astill, J.; Taha-Abdelaziz, K.; Karimi, S.H.; Bavananthasivam, J.; Kulkarni, R.R.; Sharif, S. Effects of in ovo inoculation of multi-Strain Lactobacilli on cytokine gene expression and antibody-mediated Immune responses in chickens. Front. Vet. Sci. 2020, 7, 105. [Google Scholar] [CrossRef]
- Taha-Abdelaziz, K.; Hodgins, D.C.; Lammers, A.; Alkie, T.N.; Sharif, S. Effects of early feeding and dietary interventions on development of lymphoid organs and immune competence in neonatal chickens: A review. Vet. Immunol. Immunopathol. 2018, 201, 1–11. [Google Scholar] [CrossRef]
- Alizadeh, M.; Bavananthasivam, J.; Shojadoost, B.; Astill, J.; Taha-Abdelaziz, K.; Alqazlan, N.; Boodhoo, N.; Shoja Doost, J.; Sharif, S. In ovo and oral administration of probiotic lactobacilli modulate cell-and antibody-mediated immune responses in newly hatched chicks. Front. Immunol. 2021, 12, 664387. [Google Scholar] [CrossRef]
- Kathayat, D.; Closs, G., Jr.; Helmy, Y.A.; Deblais, L.; Srivastava, V.; Rajashekara, G. In Vitro and In Vivo Evaluation of Lacticaseibacillus rhamnosus GG and Bifidobacterium lactis Bb12 Against Avian Pathogenic Escherichia coli and Identification of Novel Probiotic-Derived Bioactive Peptides. Probiotics Antimicrob. Proteins 2021. [Google Scholar] [CrossRef] [PubMed]
- Kathayat, D.; Closs, G., Jr.; Helmy, Y.A.; Lokesh, D.; Ranjit, S.; Rajashekara, G. Peptides Affecting the Outer Membrane Lipid Asymmetry System (MlaA-OmpC/F) Reduce Avian Pathogenic Escherichia coli (APEC) Colonization in Chickens. Appl. Env. Microbiol. 2021, 87, e0056721. [Google Scholar] [CrossRef]
- Taha-Abdelaziz, K.; Astill, J.; Kulkarni, R.R.; Read, L.R.; Najarian, A.; Farber, J.M.; Sharif, S. In vitro assessment of immunomodulatory and anti-Campylobacter activities of probiotic lactobacilli. Sci. Rep. 2019, 9, 17903. [Google Scholar] [CrossRef]
- Helmy, Y.A.; Kassem, I.I.; Rajashekara, G. Immuno-modulatory effect of probiotic E. coli Nissle 1917 in polarized human colonic cells against Campylobacter jejuni infection. Gut Microbes 2021, 13, 1857514. [Google Scholar] [CrossRef]
- Helmy, Y.A.; Kassem, I.; Kumar, A.; Rajashekara, G. In Vitro Evaluation of the Impact of the Probiotic E. coli Nissle 1917 on Campylobacter jejuni’s Invasion and Intracellular Survival in Human Colonic Cells. Front. Microbiol. 2017, 8, 1588. [Google Scholar] [CrossRef] [PubMed]
- Mawad, A.; Helmy, Y.A.; Shalkami, A.G.; Kathayat, D.; Rajashekara, G.E. coli Nissle microencapsulation in alginate-chitosan nanoparticles and its effect on Campylobacter jejuni in vitro. Appl Microbiol. Biotechnol. 2018, 102, 10675–10690. [Google Scholar] [CrossRef] [PubMed]
- Saint-Cyr, M.J.; Guyard-Nicodème, M.; Messaoudi, S.; Chemaly, M.; Cappelier, J.-M.; Dousset, X.; Haddad, N.J.F.i.m. Recent advances in screening of anti-Campylobacter activity in probiotics for use in poultry. Front. Microbiol. 2016, 7, 553. [Google Scholar] [CrossRef]
- Helmy, Y.A.; Closs, J.G.; Jung, k.; Kathayat, D.; Vlasova, A.; Rajashekara, G. Effect of probiotic E. coli Nissle 1917 supplementation on the growth performance, immune responses, intestinal morphology, gut microbes, of Campylobacter jejuni infected chickens. Infect. Immun. 2022, 90, e0033722. [Google Scholar] [CrossRef] [PubMed]
- Saint-Cyr, M.J.; Haddad, N.; Taminiau, B.; Poezevara, T.; Quesne, S.; Amelot, M.; Daube, G.; Chemaly, M.; Dousset, X.; Guyard-Nicodème, M. Use of the potential probiotic strain Lactobacillus salivarius SMXD51 to control Campylobacter jejuni in broilers. Int. J. Food Microbiol. 2017, 247, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kobierecka, P.A.; Wyszyńska, A.K.; Aleksandrzak-Piekarczyk, T.; Kuczkowski, M.; Tuzimek, A.; Piotrowska, W.; Górecki, A.; Adamska, I.; Wieliczko, A.; Bardowski, J. In vitro characteristics of Lactobacillus spp. strains isolated from the chicken digestive tract and their role in the inhibition of Campylobacter colonization. MicrobiologyOpen 2017, 6, e00512. [Google Scholar] [CrossRef]
- Nishiyama, K.; Nakazato, A.; Ueno, S.; Seto, Y.; Kakuda, T.; Takai, S.; Yamamoto, Y.; Mukai, T. Cell surface-associated aggregation-promoting factor from L actobacillus gasseri SBT 2055 facilitates host colonization and competitive exclusion of C ampylobacter jejuni. Mol. Microbiol. 2015, 98, 712–726. [Google Scholar] [CrossRef]
- Nishiyama, K.; Seto, Y.; Yoshioka, K.; Kakuda, T.; Takai, S.; Yamamoto, Y.; Mukai, T. Lactobacillus gasseri SBT2055 reduces infection by and colonization of Campylobacter jejuni. PLoS ONE 2014, 9, e108827. [Google Scholar] [CrossRef]
- Neal-McKinney, J.M.; Lu, X.; Duong, T.; Larson, C.L.; Call, D.R.; Shah, D.H.; Konkel, M.E. Production of organic acids by probiotic lactobacilli can be used to reduce pathogen load in poultry. PLoS ONE 2012, 7, e43928. [Google Scholar] [CrossRef]
- Guyard-Nicodeme, M.; Keita, A.; Quesne, S.; Amelot, M.; Poezevara, T.; Le Berre, B.; Sánchez, J.; Vesseur, P.; Martín, Á.; Medel, P. Efficacy of feed additives against Campylobacter in live broilers during the entire rearing period. Poult. Sci. 2016, 95, 298–305. [Google Scholar] [CrossRef]
- Gracia, M.; Millán, C.; Sanchez, J.; Guyard-Nicodeme, M.; Mayot, J.; Carre, Y.; Csorbai, A.; Chemaly, M.; Medel, P. Efficacy of feed additives against Campylobacter in live broilers during the entire rearing period: Part B. Poult. Sci. 2016, 95, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, V.F.; Donoghue, A.M.; Arsi, K.; Reyes-Herrera, I.; Metcalf, J.H.; de los Santos, F.S.; Blore, P.J.; Donoghue, D.J. Targeting motility properties of bacteria in the development of probiotic cultures against Campylobacter jejuni in broiler chickens. Foodborne Pathog. Dis. 2013, 10, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Fritts, C.; Kersey, J.; Motl, M.; Kroger, E.; Yan, F.; Si, J.; Jiang, Q.; Campos, M.; Waldroup, A.; Waldroup, P. Bacillus subtilis C-3102 (Calsporin) improves live performance and microbiological status of broiler chickens. J. Appl. Poult. Res. 2000, 9, 149–155. [Google Scholar] [CrossRef]
- Robyn, J.; Rasschaert, G.; Hermans, D.; Pasmans, F.; Heyndrickx, M. In vivo broiler experiments to assess anti-Campylobacter jejuni activity of a live Enterococcus faecalis strain. Poult. Sci. 2013, 92, 265–271. [Google Scholar] [CrossRef]
- Netherwood, T.; Gilbert, H.; Parker, D.; O’donnell, A. Probiotics shown to change bacterial community structure in the avian gastrointestinal tract. Appl. Environ. Microbiol. 1999, 65, 5134–5138. [Google Scholar] [CrossRef] [PubMed]
- Baffoni, L.; Gaggìa, F.; Di Gioia, D.; Santini, C.; Mogna, L.; Biavati, B. A Bifidobacterium-based synbiotic product to reduce the transmission of C. jejuni along the poultry food chain. Int. J. Food Microbiol. 2012, 157, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Santini, C.; Baffoni, L.; Gaggia, F.; Granata, M.; Gasbarri, R.; Di Gioia, D.; Biavati, B. Characterization of probiotic strains: An application as feed additives in poultry against Campylobacter jejuni. Int. J. Food Microbiol. 2010, 141, S98–S108. [Google Scholar] [CrossRef]
- Morishita, T.Y.; Aye, P.P.; Harr, B.S.; Cobb, C.W.; Clifford, J.R. Evaluation of an avian-specific probiotic to reduce the colonization and shedding of Campylobacter jejuni in broilers. Avian Dis. 1997, 41, 850–855. [Google Scholar] [CrossRef]
- Arsi, K.; Donoghue, A.; Woo-Ming, A.; Blore, P.; Donoghue, D. The efficacy of selected probiotic and prebiotic combinations in reducing Campylobacter colonization in broiler chickens. J. Appl. Poult. Res. 2015, 24, 327–334. [Google Scholar] [CrossRef]
- Willis, W.L.; Reid, L. Investigating the effects of dietary probiotic feeding regimens on broiler chicken production and Campylobacter jejuni presence. Poult. Sci. 2008, 87, 606–611. [Google Scholar] [CrossRef]
- Aho, M.; Nuotio, L.; Nurmi, E.; Kiiskinen, T. Competitive exclusion of campylobacters from poultry with K-bacteria and Broilact®. Int. J. Food Microbiol. 1992, 15, 265–275. [Google Scholar] [CrossRef]
- Ghareeb, K.; Awad, W.; Mohnl, M.; Porta, R.; Biarnes, M.; Böhm, J.; Schatzmayr, G. Evaluating the efficacy of an avian-specific probiotic to reduce the colonization of Campylobacter jejuni in broiler chickens. Poult. Sci. 2012, 91, 1825–1832. [Google Scholar] [CrossRef] [PubMed]
- Cean, A.; Stef, L.; Simiz, E.; Julean, C.; Dumitrescu, G.; Vasile, A.; Pet, E.; Drinceanu, D.; Corcionivoschi, N. Effect of human isolated probiotic bacteria on preventing Campylobacter jejuni colonization of poultry. Foodborne Pathog. Dis. 2015, 12, 122–130. [Google Scholar] [CrossRef]
- Smialek, M.; Burchardt, S.; Koncicki, A. The influence of probiotic supplementation in broiler chickens on population and carcass contamination with Campylobacter spp.-Field study. Res. Vet. Sci. 2018, 118, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Schoeni, J.L.; Wong, A. Inhibition of Campylobacter jejuni colonization in chicks by defined competitive exclusion bacteria. Appl. Environ. Microbiol. 1994, 60, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Lin, C.-H.; Sung, C.T.; Fang, J.-Y. Antibacterial activities of bacteriocins: Application in foods and pharmaceuticals. Front. Microbiol. 2014, 5, 241. [Google Scholar] [PubMed]
- Zhang, T.; Zhang, Y.; Li, L.; Jiang, X.; Chen, Z.; Zhao, F.; Yi, Y. Biosynthesis and Production of Class II Bacteriocins of Food-Associated Lactic Acid Bacteria. Fermentation 2022, 8, 217. [Google Scholar] [CrossRef]
- Messaoudi, S.; Kergourlay, G.; Dalgalarrondo, M.; Choiset, Y.; Ferchichi, M.; Prévost, H.; Pilet, M.-F.; Chobert, J.-M.; Manai, M.; Dousset, X. Purification and characterization of a new bacteriocin active against Campylobacter produced by Lactobacillus salivarius SMXD51. Food Microbiol. 2012, 32, 129–134. [Google Scholar] [CrossRef]
- Stern, N.; Svetoch, E.; Eruslanov, B.; Perelygin, V.; Mitsevich, E.; Mitsevich, I.; Pokhilenko, V.; Levchuk, V.; Svetoch, O.; Seal, B. Isolation of a Lactobacillus salivarius strain and purification of its bacteriocin, which is inhibitory to Campylobacter jejuni in the chicken gastrointestinal system. Antimicrob. Agents Chemother. 2006, 50, 3111–3116. [Google Scholar] [CrossRef]
- Cole, K.; Farnell, M.; Donoghue, A.; Stern, N.; Svetoch, E.; Eruslanov, B.; Volodina, L.; Kovalev, Y.; Perelygin, V.; Mitsevich, E. Bacteriocins reduce Campylobacter colonization and alter gut morphology in turkey poults. Poult. Sci. 2006, 85, 1570–1575. [Google Scholar] [CrossRef] [PubMed]
- Stern, N.J.; Svetoch, E.A.; Eruslanov, B.V.; Kovalev, Y.N.; Volodina, L.I.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Levchuk, V.P. Paenibacillus polymyxa purified bacteriocin to control Campylobacter jejuni in chickens. J. Food Prot. 2005, 68, 1450–1453. [Google Scholar] [CrossRef]
- Svetoch, E.A.; Eruslanov, B.V.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Borzenkov, V.N.; Levchuk, V.P.; Svetoch, O.E.; Kovalev, Y.N.; Stepanshin, Y.G. Diverse antimicrobial killing by Enterococcus faecium E 50-52 bacteriocin. J. Agric. Food Chem. 2008, 56, 1942–1948. [Google Scholar] [CrossRef] [PubMed]
- Line, J.; Svetoch, E.; Eruslanov, B.; Perelygin, V.; Mitsevich, E.; Mitsevich, I.; Levchuk, V.; Svetoch, O.; Seal, B.; Siragusa, G. Isolation and purification of enterocin E-760 with broad antimicrobial activity against gram-positive and gram-negative bacteria. Antimicrob. Agents Chemother. 2008, 52, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- Stern, N.J.; Eruslanov, B.V.; Pokhilenko, V.D.; Kovalev, Y.N.; Volodina, L.L.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Borzenkov, V.N.; Levchuk, V.P.J.M.E.i.H.; et al. Bacteriocins reduce Campylobacter jejuni colonization while bacteria producing bacteriocins are ineffective. Microb. Ecol. Health Dis. 2008, 20, 74–79. [Google Scholar]
- Hoang, K.; Stern, N.J.; Lin, J. Development and stability of bacteriocin resistance in Campylobacter spp. J. Appl. Microbiol. 2011, 111, 1544–1550. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. The role of probiotics, prebiotics and synbiotics in animal nutrition. Gut Pathog. 2018, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M. Prebiotics and synbiotics: Concepts and nutritional properties. Br. J. Nutr. 1998, 80, S197–S202. [Google Scholar] [CrossRef]
- Baffoni, L.; Gaggìa, F.; Garofolo, G.; Di Serafino, G.; Buglione, E.; Di Giannatale, E.; Di Gioia, D. Evidence of Campylobacter jejuni reduction in broilers with early synbiotic administration. Int. J. Food Microbiol. 2017, 251, 41–47. [Google Scholar] [CrossRef]
- Micciche, A.; Rothrock Jr, M.J.; Yang, Y.; Ricke, S.C. Essential oils as an intervention strategy to reduce Campylobacter in poultry production: A review. Front. Microbiol. 2019, 10, 1058. [Google Scholar] [CrossRef]
- Irshad, M.; Subhani, M.A.; Ali, S.; Hussain, A. Biological importance of essential oils. Essent. Oils-Oils Nat. 2020, 22, 70. [Google Scholar]
- de Los Santos, F.S.; Donoghue, A.; Venkitanarayanan, K.; Dirain, M.; Reyes-Herrera, I.; Blore, P.; Donoghue, D. Caprylic acid supplemented in feed reduces enteric Campylobacter jejuni colonization in ten-day-old broiler chickens. Poult. Sci. 2008, 87, 800–804. [Google Scholar] [CrossRef]
- De Los Santos, F.S.; Donoghue, A.; Venkitanarayanan, K.; Metcalf, J.; Reyes-Herrera, I.; Dirain, M.; Aguiar, V.; Blore, P.; Donoghue, D. The natural feed additive caprylic acid decreases Campylobacter jejuni colonization in market-aged broiler chickens. Poult. Sci. 2009, 88, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Arsi, K.; Donoghue, A.; Venkitanarayanan, K.; Kollanoor-Johny, A.; Fanatico, A.; Blore, P.; Donoghue, D. The efficacy of the natural plant extracts, thymol and carvacrol against Campylobacter colonization in broiler chickens. J. Food Saf. 2014, 34, 321–325. [Google Scholar] [CrossRef]
- Szott, V.; Reichelt, B.; Alter, T.; Friese, A.; Roesler, U. In vivo efficacy of carvacrol on Campylobacter jejuni prevalence in broiler chickens during an entire fattening period. Eur. J. Microbiol. Immunol. 2020, 10, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Hermans, D.; Martel, A.; Van Deun, K.; Van Immerseel, F.; Heyndrickx, M.; Haesebrouck, F.; Pasmans, F. The cinnamon-oil ingredient trans-cinnamaldehyde fails to target Campylobacter jejuni strain KC 40 in the broiler chicken cecum despite marked in vitro activity. J. Food Prot. 2011, 74, 1729–1734. [Google Scholar] [CrossRef] [PubMed]
- Johny, A.K.; Darre, M.; Donoghue, A.; Donoghue, D.; Venkitanarayanan, K. Antibacterial effect of trans-cinnamaldehyde, eugenol, carvacrol, and thymol on Salmonella Enteritidis and Campylobacter jejuni in chicken cecal contents in vitro. J. Appl. Poult. Res. 2010, 19, 237–244. [Google Scholar] [CrossRef]
- Kurekci, C.; Padmanabha, J.; Bishop-Hurley, S.L.; Hassan, E.; Al Jassim, R.A.; McSweeney, C.S. Antimicrobial activity of essential oils and five terpenoid compounds against Campylobacter jejuni in pure and mixed culture experiments. Int. J. Food Microbiol. 2013, 166, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Broom, L. Organic acids for improving intestinal health of poultry. World’s Poult. Sci. J. 2015, 71, 630–642. [Google Scholar] [CrossRef]
- Khan, R.U.; Chand, N.; Akbar, A. Effect of organic acids on the performance of Japanese quails. Pak. J. Zool. 2016, 48, 1799–1803. [Google Scholar]
- Giannenas, I.; Papaneophytou, C.; Tsalie, E.; Pappas, I.; Triantafillou, E.; Tontis, D.; Kontopidis, G. Dietary supplementation of benzoic acid and essential oil compounds affects buffering capacity of the feeds, performance of turkey poults and their antioxidant status, pH in the digestive tract, intestinal microbiota and morphology. Asian-Australas. J. Anim. Sci. 2014, 27, 225–236. [Google Scholar] [CrossRef]
- Dittoe, D.K.; Ricke, S.C.; Kiess, A.S. Organic acids and potential for modifying the avian gastrointestinal tract and reducing pathogens and disease. Front. Vet. Sci. 2018, 5, 216. [Google Scholar] [CrossRef] [PubMed]
- Beier, R.C.; Byrd, J.A.; Caldwell, D.; Andrews, K.; Crippen, T.L.; Anderson, R.C.; Nisbet, D.J. Inhibition and interactions of Campylobacter jejuni from broiler chicken houses with organic acids. Microorganisms 2019, 7, 223. [Google Scholar] [CrossRef] [PubMed]
- Peh, E.; Kittler, S.; Reich, F.; Kehrenberg, C. Antimicrobial activity of organic acids against Campylobacter spp. and development of combinations—A synergistic effect? PLoS ONE 2020, 15, e0239312. [Google Scholar] [CrossRef]
- Hermans, D.; Martel, A.; Van Deun, K.; Verlinden, M.; Van Immerseel, F.; Garmyn, A.; Messens, W.; Heyndrickx, M.; Haesebrouck, F.; Pasmans, F. Intestinal mucus protects Campylobacter jejuni in the ceca of colonized broiler chickens against the bactericidal effects of medium-chain fatty acids. Poult. Sci. 2010, 89, 1144–1155. [Google Scholar] [CrossRef]
- Skånseng, B.; Kaldhusdal, M.; Moen, B.; Gjevre, A.G.; Johannessen, G.; Sekelja, M.; Trosvik, P.; Rudi, K. Prevention of intestinal Campylobacter jejuni colonization in broilers by combinations of in-feed organic acids. J. Appl. Microbiol. 2010, 109, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Grilli, E.; Vitari, F.; Domeneghini, C.; Palmonari, A.; Tosi, G.; Fantinati, P.; Massi, P.; Piva, A. Development of a feed additive to reduce caecal C ampylobacter jejuni in broilers at slaughter age: From in vitro to in vivo, a proof of concept. J. Appl. Microbiol. 2013, 114, 308–317. [Google Scholar] [CrossRef]
- Ramana, J.; Reddy, R.; Rao, D.; Shakila, S.; Suresh, J. Effect of organic acid supplementation on performance of poultry. J. Anim. Feed Sci. Technol 2017, 5, 15–23. [Google Scholar]
- Li, Q.; Kang, C. Mechanisms of action for small molecules revealed by structural biology in drug discovery. Int. J. Mol. Sci. 2020, 21, 5262. [Google Scholar] [CrossRef]
- Johnson, T.J.; Shank, J.M.; Johnson, J.G. Current and potential treatments for reducing Campylobacter colonization in animal hosts and disease in humans. Front. Microbiol. 2017, 8, 487. [Google Scholar] [CrossRef]
- Lewis, R.J.; Vetter, I.; Cardoso, F.C.; Inserra, M.; King, G. Does Nature Do Ion Channel Drug Discovery Better than Us; Royal Society of Chemistry: London, UK, 2015. [Google Scholar]
- Johnson, J.G.; Yuhas, C.; McQuade, T.J.; Larsen, M.J.; DiRita, V.J. Narrow-spectrum inhibitors of Campylobacter jejuni flagellar expression and growth. Antimicrob. Agents Chemother. 2015, 59, 3880–3886. [Google Scholar] [CrossRef] [PubMed]
- Deblais, L.; Helmy, Y.A.; Kumar, A.; Antwi, J.; Kathayat, D.; Acuna, U.M.; Huang, H.-c.; de Blanco, E.C.; Fuchs, J.R.; Rajashekara, G. Novel narrow spectrum benzyl thiophene sulfonamide derivatives to control Campylobacter. J. Antibiot. 2019, 72, 555–565. [Google Scholar] [CrossRef]
- Kumar, A.; Drozd, M.; Pina-Mimbela, R.; Xu, X.; Helmy, Y.A.; Antwi, J.; Fuchs, J.R.; Nislow, C.; Templeton, J.; Blackall, P.J. Novel anti-Campylobacter compounds identified using high throughput screening of a pre-selected enriched small molecules library. Front. Microbiol. 2016, 7, 405. [Google Scholar] [CrossRef] [PubMed]
- Klančnik, A.; Možina, S.S.; Zhang, Q. Anti-Campylobacter activities and resistance mechanisms of natural phenolic compounds in Campylobacter. PLoS ONE 2012, 7, e51800. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Chen, H.; Gao, Y.; An, N.; Li, X.; Pan, X.; Yang, X.; Tian, L.; Sun, J.; Xiong, X. Gut microbiota-derived short-chain fatty acids and hypertension: Mechanism and treatment. Biomed. Pharmacother. 2020, 130, 110503. [Google Scholar] [CrossRef] [PubMed]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.; Faber, K.N.; Hermoso, M.A. Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef]
- Lassen, B.; Helwigh, B.; Kahl Petersen, C.; Ellis-Iversen, J. Systematic review of products with potential application for use in the control of Campylobacter spp. in organic and free-range broilers. Acta Vet. Scand. 2022, 64, 24. [Google Scholar] [CrossRef]
- van der Wielen, P.W.; Biesterveld, S.; Notermans, S.; Hofstra, H.; Urlings, B.A.; van Knapen, F. Role of volatile fatty acids in development of the cecal microflora in broiler chickens during growth. Appl. Environ. Microbiol. 2000, 66, 2536–2540. [Google Scholar] [CrossRef]
- Kasman, L.M.; Porter, L.D. Bacteriophages. In StatPearls [Internet]; StatPearls Publishing: Tampa, FL, USA, 2021. [Google Scholar]
- Wagenaar, J.A.; Van Bergen, M.A.; Mueller, M.A.; Wassenaar, T.M.; Carlton, R.M. Phage therapy reduces Campylobacter jejuni colonization in broilers. Vet. Microbiol. 2005, 109, 275–283. [Google Scholar] [CrossRef]
- D’angelantonio, D.; Scattolini, S.; Boni, A.; Neri, D.; Di Serafino, G.; Connerton, P.; Connerton, I.; Pomilio, F.; Di Giannatale, E.; Migliorati, G. Bacteriophage therapy to reduce colonization of campylobacter jejuni in broiler chickens before slaughter. Viruses 2021, 13, 1428. [Google Scholar] [CrossRef]
- Fischer, S.; Kittler, S.; Klein, G.; Glünder, G. Impact of a single phage and a phage cocktail application in broilers on reduction of Campylobacter jejuni and development of resistance. PLoS ONE 2013, 8, e78543. [Google Scholar] [CrossRef]
- Kittler, S.; Fischer, S.; Abdulmawjood, A.; Glünder, G.; Klein, G. Effect of bacteriophage application on Campylobacter jejuni loads in commercial broiler flocks. Appl. Environ. Microbiol. 2013, 79, 7525–7533. [Google Scholar] [CrossRef]
- Tang, Y.; Li, J.; Wang, Y.; Song, Z.; Ying, H.; Kong, L.; Jiao, X.A.; Huang, J. Campylobacter jejuni Developed the Resistance to Bacteriophage CP39 by Phase Variable Expression of 06875 Encoding the CGPTase. Viruses 2022, 14, 485. [Google Scholar] [CrossRef]
- Loc Carrillo, C.; Atterbury, R.J.; El-Shibiny, A.; Connerton, P.L.; Dillon, E.; Scott, A.; Connerton, I.F. Bacteriophage therapy to reduce Campylobacter jejuni colonization of broiler chickens. Appl. Environ. Microbiol. 2005, 71, 6554–6563. [Google Scholar] [CrossRef] [PubMed]
- Wernicki, A.; Nowaczek, A.; Urban-Chmiel, R. Bacteriophage therapy to combat bacterial infections in poultry. Virol. J. 2017, 14, 179. [Google Scholar] [CrossRef] [PubMed]
- Hammerl, J.A.; Jäckel, C.; Alter, T.; Janzcyk, P.; Stingl, K.; Knüver, M.T.; Hertwig, S. Reduction of Campylobacter jejuni in broiler chicken by successive application of group II and group III phages. PLoS ONE 2014, 9, e114785. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.S.; Rahman, S.R. Use of Phages to Treat Antimicrobial-Resistant Salmonella Infections in Poultry. Vet. Sci. 2022, 9, 438. [Google Scholar] [CrossRef]
- Han, Z.; Willer, T.; Li, L.; Pielsticker, C.; Rychlik, I.; Velge, P.; Kaspers, B.; Rautenschlein, S. Influence of the gut microbiota composition on Campylobacter jejuni colonization in chickens. Infect. Immun. 2017, 85, e00380-17. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, S.; Chaloner, G.; Kemmett, K.; Davidson, N.; Williams, N.; Kipar, A.; Humphrey, T.; Wigley, P. Campylobacter jejuni is not merely a commensal in commercial broiler chickens and affects bird welfare. MBio 2014, 5, e01364-14. [Google Scholar] [CrossRef]
- Yitbarek, A.; Taha-Abdelaziz, K.; Hodgins, D.C.; Read, L.; Nagy, É.; Weese, J.S.; Caswell, J.L.; Parkinson, J.; Sharif, S. Gut microbiota-mediated protection against influenza virus subtype H9N2 in chickens is associated with modulation of the innate responses. Sci. Rep. 2018, 8, 13189. [Google Scholar]
- Bavananthasivam, J.; Astill, J.; Matsuyama-Kato, A.; Taha-Abdelaziz, K.; Shojadoost, B.; Sharif, S. Gut microbiota is associated with protection against Marek’s disease virus infection in chickens. Virology 2021, 553, 122–130. [Google Scholar] [CrossRef]
- Gilroy, R.; Chaloner, G.; Wedley, A.; Lacharme-Lora, L.; Jopson, S.; Wigley, P. Campylobacter jejuni transmission and colonisation in broiler chickens is inhibited by faecal microbiota transplantation. BioRxiv 2018, 476119. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Siegerstetter, S.-C.; Magowan, E.; Lawlor, P.G.; O′ Connell, N.E.; Zebeli, Q. Fecal microbiota transplant from highly feed efficient donors affects cecal physiology and microbiota in low-and high-feed efficient chickens. Front. Microbiol. 2019, 10, 1576. [Google Scholar] [CrossRef]
- Ty, M.; Taha-Abdelaziz, K.; Demey, V.; Castex, M.; Sharif, S.; Parkinson, J. Performance of distinct microbial based solutions in a Campylobacter infection challenge model in poultry. Anim. Microbiome 2022, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Davies, R.; De Cesare, A.; Herman, L.; Hilbert, F. Update and review of control options for Campylobacter in broilers at primary production. EFSA J. 2020, 18, e06090. [Google Scholar] [PubMed]
- Seliwiorstow, T.; Baré, J.; Van Damme, I.; Uyttendaele, M.; De Zutter, L. Campylobacter carcass contamination throughout the slaughter process of Campylobacter-positive broiler batches. Int. J. Food Microbiol. 2015, 194, 25–31. [Google Scholar] [CrossRef]
- Zhao, G.; Huang, X.; Zhao, J.; Liu, N.; Li, Y.; Wang, L.; Gao, Y.; Wang, J.; Qu, Z.; Liu, J. Risk Prevention and Control Points Through Quantitative Evaluation of Campylobacter in a Large Broiler Slaughterhouse. Front. Vet. Sci. 2020, 7, 172. [Google Scholar] [CrossRef]
- Lázaro, B.; Cárcamo, J.; Audícana, A.; Perales, I.; Fernández-Astorga, A. Viability and DNA maintenance in nonculturable spiral Campylobacter jejuni cells after long-term exposure to low temperatures. Appl. Environ. Microbiol. 1999, 65, 4677–4681. [Google Scholar] [CrossRef]
- Habib, I.; Mohamed, M.-Y.I.; Lakshmi, G.B.; Khan, M.; Li, D. Quantification of Campylobacter contamination on chicken carcasses sold in retail markets in the United Arab Emirates. Int. J. Food Contam. 2022, 9, 9. [Google Scholar] [CrossRef]
- Abu-Madi, M.; Behnke, J.M.; Sharma, A.; Bearden, R.; Al-Banna, N. Prevalence of virulence/stress genes in Campylobacter jejuni from chicken meat sold in Qatari retail outlets. PLoS ONE 2016, 11, e0156938. [Google Scholar] [CrossRef]
- Alarjani, K.M.; Elkhadragy, M.F.; Al-Masoud, A.H.; Yehia, H.M. Detection of Campylobacter jejuni and Salmonella typhimurium in chicken using PCR for virulence factor hipO and invA genes (Saudi Arabia). Biosci. Rep. 2021, 41, BSR20211790. [Google Scholar] [CrossRef]
- Royden, A.; Christley, R.; Jones, T.; Williams, A.; Awad, F.; Haldenby, S.; Wigley, P.; Rushton, S.P.; Williams, N.J. Campylobacter contamination at retail of Halal Chicken produced in the United Kingdom. J. Food Prot. 2021, 84, 1433–1445. [Google Scholar] [CrossRef]
- Deckert, A.; Valdivieso-Garcia, A.; Reid-Smith, R.; Tamblyn, S.; Seliske, P.; Irwin, R.; Dewey, C.; Boerlin, P.; McEwen, S.A. Prevalence and antimicrobial resistance in Campylobacter spp. isolated from retail chicken in two health units in Ontario. J. Food Prot. 2010, 73, 1317–1324. [Google Scholar] [CrossRef]
- The European Food Safety Authority. Scientific Opinion on Campylobacter in broiler meat production: Control options and performance objectives and/or targets at different stages of the food chain. EFSA J. 2011, 9, 2105. [Google Scholar] [CrossRef]
- Peyrat, M.; Soumet, C.; Maris, P.; Sanders, P. Recovery of Campylobacter jejuni from surfaces of poultry slaughterhouses after cleaning and disinfection procedures: Analysis of a potential source of carcass contamination. Int. J. Food Microbiol. 2008, 124, 188–194. [Google Scholar] [CrossRef]
- EFSA; ECDC. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA J. 2018, 16, e05500. [Google Scholar]
- Bai, Y.; Ding, X.; Zhao, Q.; Sun, H.; Li, T.; Li, Z.; Wang, H.; Zhang, L.; Zhang, C.; Xu, S. Development of an organic acid compound disinfectant to control food-borne pathogens and its application in chicken slaughterhouses. Poult. Sci. 2022, 101, 101842. [Google Scholar] [CrossRef]
- Sohaib, M.; Anjum, F.M.; Arshad, M.S.; Rahman, U.U. Postharvest intervention technologies for safety enhancement of meat and meat based products; a critical review. J. Food Sci. Technol. 2016, 53, 19–30. [Google Scholar] [CrossRef]
- Rahman, M.H.; Hossain, M.M.; Rahman, S.M.E.; Hashem, M.A.; Oh, D.-H. Effect of repeated freeze-thaw cycles on beef quality and safety. Korean J. Food Sci. Anim. Resour. 2014, 34, 482. [Google Scholar] [CrossRef]
- Georgsson, F.; Þorkelsson, Á.E.; Geirsdóttir, M.; Reiersen, J.; Stern, N.J. The influence of freezing and duration of storage on Campylobacter and indicator bacteria in broiler carcasses. Food Microbiol. 2006, 23, 677–683. [Google Scholar] [CrossRef]
- Bhaduri, S.; Cottrell, B. Survival of cold-stressed Campylobacter jejuni on ground chicken and chicken skin during frozen storage. Appl. Environ. Microbiol. 2004, 70, 7103–7109. [Google Scholar] [CrossRef]
- Park, S.F. The physiology of Campylobacter species and its relevance to their role as foodborne pathogens. Int. J. Food Microbiol. 2002, 74, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Sampers, I.; Habib, I.; De Zutter, L.; Dumoulin, A.; Uyttendaele, M. Survival of Campylobacter spp. in poultry meat preparations subjected to freezing, refrigeration, minor salt concentration, and heat treatment. Int. J. Food Microbiol. 2010, 137, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Albert, T.; Braun, P.G.; Saffaf, J.; Wiacek, C. Physical methods for the decontamination of meat surfaces. Curr. Clin. Microbiol. Rep. 2021, 8, 9–20. [Google Scholar] [CrossRef]
- Maragkoudakis, P.A.; Mountzouris, K.C.; Psyrras, D.; Cremonese, S.; Fischer, J.; Cantor, M.D.; Tsakalidou, E. Functional properties of novel protective lactic acid bacteria and application in raw chicken meat against Listeria monocytogenes and Salmonella enteritidis. Int. J. Food Microbiol. 2009, 130, 219–226. [Google Scholar] [CrossRef]
- Sakaridis, I.; Soultos, N.; Batzios, C.; Ambrosiadis, I.; Koidis, P. Lactic acid bacteria isolated from chicken carcasses with inhibitory activity against Salmonella spp. and Listeria monocytogenes. Czech J. Food Sci. 2014, 32, 61–68. [Google Scholar] [CrossRef]
- Woo-Ming, A.N. Reduction of Campylobacter jejuni on Chicken Wingettes by Treatment with Caprylic Acid, Chitosan or Protective Cultures of Lactobacillus spp.; University of Arkansas: Fayetteville, AR, USA, 2015. [Google Scholar]
- Melero, B.; Vinuesa, R.; Diez, A.; Jaime, I.; Rovira, J. Application of protective cultures against Listeria monocytogenes and Campylobacter jejuni in chicken products packaged under modified atmosphere. Poult. Sci. 2013, 92, 1108–1116. [Google Scholar] [CrossRef]
- Long, C.; Phillips, C.A. The effect of sodium citrate, sodium lactate and nisin on the survival of Arcobacter butzleri NCTC 12481 on chicken. Food Microbiol. 2003, 20, 495–502. [Google Scholar] [CrossRef]
- Atterbury, R.J.; Connerton, P.L.; Dodd, C.E.; Rees, C.E.; Connerton, I.F. Application of host-specific bacteriophages to the surface of chicken skin leads to a reduction in recovery of Campylobacter jejuni. Appl. Environ. Microbiol. 2003, 69, 6302–6306. [Google Scholar] [CrossRef]
- Zampara, A.; Sørensen, M.C.H.; Elsser-Gravesen, A.; Brøndsted, L. Significance of phage-host interactions for biocontrol of Campylobacter jejuni in food. Food Control 2017, 73, 1169–1175. [Google Scholar] [CrossRef]
- Zampara, A.; Sørensen, M.C.H.; Gencay, Y.E.; Grimon, D.; Kristiansen, S.H.; Jørgensen, L.S.; Kristensen, J.R.; Briers, Y.; Elsser-Gravesen, A.; Brøndsted, L. Developing innolysins against campylobacter jejuni using a novel prophage receptor-binding protein. Front. Microbiol. 2021, 12, 619028. [Google Scholar] [CrossRef]
- Djenane, D.; Yangueela, J.; Gomez, D.; Roncales, P. perspectives on the use of essential oils as antimicrobials against Campylobacter jejuni CECT 7572 in retail chicken meats packaged in microaerobic atmosphere. J. Food Saf. 2012, 32, 37–47. [Google Scholar] [CrossRef]
- Shrestha, S.; Wagle, B.; Upadhyay, A.; Arsi, K.; Donoghue, D.; Donoghue, A. Carvacrol antimicrobial wash treatments reduce Campylobacter jejuni and aerobic bacteria on broiler chicken skin. Poult. Sci. 2019, 98, 4073–4083. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.; Luís, Â.; Oleastro, M.; Domingues, F.C. Antioxidant properties of coriander essential oil and linalool and their potential to control Campylobacter spp. Food Control 2016, 61, 115–122. [Google Scholar] [CrossRef]
- Shanker, S.; Lee, A.; Sorrell, T. Campylobacter jejuni in broilers: The role of vertical transmission. Epidemiol. Infect. 1986, 96, 153–159. [Google Scholar]
- Neill, S.; Campbell, J.; O’brien, J. Egg penetration by Campylobacter jejuni. Avian Pathol. 1985, 14, 313–320. [Google Scholar] [CrossRef]
- Sahin, O.; Kobalka, P.; Zhang, Q. Detection and survival of Campylobacter in chicken eggs. J. Appl. Microbiol. 2003, 95, 1070–1079. [Google Scholar] [CrossRef]
- Alkaya, G.B.; Erdogdu, F.; Halkman, A.K.; Ekiz, H.I. Surface decontamination of whole-shell eggs using far-infrared radiation. Food Bioprod. Process. 2016, 98, 275–282. [Google Scholar] [CrossRef]
- Turtoi, M.; Borda, D. Decontamination of egg shells using ultraviolet light treatment. World’s Poult. Sci. J. 2014, 70, 265–278. [Google Scholar] [CrossRef]
| Vaccine Type | Vaccine Active Components | Route of Administration | C. jejuni Challenge Strain/Dose per Bird | Effect on Campylobacter Colonization | Reference |
|---|---|---|---|---|---|
| Whole-cell vaccines | Whole cell (WC) and flagellin (Fla) or purified Fla | Intra-peritoneally (IP) followed by an IP or oral booster doses | C. jejuni, isolate #V2 (by mixing with seeder chickens) | WC+Fla (twice IP): Up to 2 log10 CFU/g WC+Fla (IP and orally) or Fla alone: No reduction | [45] |
| Formalin inactivated WC with or without LT adjuvant | Esophageal gavage | C. jejuni, F1BCB (by mixing with seeder chickens) | WC alone: Up to 0.4 log10 on day 7 WC+LT: Up to 1.9 log10 on day 46 | [46] | |
| Wild-type parental strain or the mutated strains (CadF9, CiaB5, PldA23, and DnaJA) | Intraperitoneal | C. jejuni F38011 (104 CFU) | Up to 0.9 log10 CFU/g in CiaB5 group only | [50] | |
| C. jejuni NCTC 11168 isogenic knockout mutants of AhpC, KatA, and SodB | Oral | C. jejuni NCTC 11168 (1 × 108 CFU) | ΔahpC mutant: Up to 3 log10 ΔkatA: Up to 2 log10 ΔsodB: No effect on day 42 of age | [51] | |
| Subunit vaccines | C. jejuni N-glycan with GlycoTag, or fused to the E. coli lipopolysaccharide-core | Oral | C. jejuni, 81–176 (102 or 106 CFU) | 10 log10 reduction on day 35 | [52] |
| FlaA/CadF/FlpA/CmeC protein or CadF-FlaA-FlpA fusion protein | Intramuscular | C. jejuni F38011 (2 × 108 CFU) | ≥3 log10 on day 35 | [53] | |
| Capsular polysaccharide conjugated to diphtheria toxoid CRM | Subcutaneous | C. jejuni 81–176 (2 × 107 CFU) | 0.64 log10 CFU/g on day 38 of age | [54] | |
| C. jejuni outer membrane proteins | Subcutaneous/Oral | C. jejuni, 81–176 (1 × 108 CFU) | SC: Below the detection limit (<10 CFU) Oral: No protection on day 42 of age | [55] | |
| C. jejuni Enterobactin-KLH conjugate | Intramuscular | C. jejuni, NCTC 11168 (1 × 104 CFU) | >4 log10 on day 58 of age | [56] | |
| Recombinant vaccines | E. coli expressing N-glycan protein with probiotics (A. mobilis DSM 15930 or L. reuteri CSF8) | Oral | C. jejuni, 81–176 (106 CFU) | Up to 6 log10 on day 35 | [57] |
| S. Typhimurium ΔaroA mutant expressing CjaA as a plasmid-encoded fusion to fragment C of tetanus toxin | Subcutaneous /Oral | C. jejuni, MI (1 × 107 CFU) | Oral: 1.4 log10 CFU/g Subcutaneous: 3.78 log10 | [58] | |
| Salmonella strain carrying C. jejuni 72Dz/92 CjaA gene | Oral | C. jejuni, pUOA18 (2 × 108 CFU) | 6 log10 at 12 days post challenge | [59] | |
| Nanoparticle-based vaccines | Chitosan/pCAGGS-flaA nanoparticles | Intranasal | C. jejuni ALM-80 (5 × 107 CFU) | 2 log10 CFU/g on day 35 of age | [60] |
| PLGA-encapsulated C. jejuni outer membrane proteins | Subcutaneous /Oral | C. jejuni 81–176 (1 × 108 CFU) | SC: Below the limit of detection Oral: No protection on day 42 of age | [55] | |
| Liposome encapsulated proteins (CjaALysM and CjaDLysM) | In ovo | C. jejuni 12/2 (106 CFU) | 2 log10 CFU/g on day 28 of age | [61] | |
| PLGA-encapsulated CpG ODN and C. jejuni lysate | Oral | C. jejuni, (81–176/ 107 CFU) | Up to 2.4 log10 on day 37 of age | [62] |
| Probiotic Strain | Type of Probiotics | Administration | Campylobacter Strain/Dose | Effect on Campylobacter Colonization | Reference |
|---|---|---|---|---|---|
| Single strain | Escherichia coli Nissle 1917 | Two-weeks pre-harvest | Cocktail of six C. jejuni strains | Up to 2.6 log10 reduction | [101] |
| Lactobacillus salivarius SMXD51 | Administered at day 1 then every 2–3 days until 35 days orally | C. jejuni C97ANSES640 (1 × 104 CFU) | 0.8 log10 at 14 days and 2.81 log10 at 35 days | [102] | |
| Lactobacillus plantarum PA18A | Day 1 and 4 orally | C. jejuni strain 12/2 (1 × 104 CFU) | 1 log10 reduction | [103] | |
| Lactobacillus gasseri SBT2055 LG2055 WTCM, Δapf1 and Δapf2 mutant strains | Day 2–14 orally | C. jejuni 81–176 (1 × 106 CFU) | WTCM and Δapf2: Up to 270-fold reduction Δapf1: No reduction | [104] | |
| Lactobacillus gasseri SBT2055 | Day 2–14 orally | C. jejuni 81–176 (1 × 106 CFU) | 250-fold reduction | [105] | |
| Lactobacillusacidophilus NCFM or Lactobacillus crispatus JCM5810 or Lactobacillus gallinarum ATCC or Lactobacillus helveticus CNRZ32 | Day 1 and 4 orally | C. jejuni F38011 (1 × 108 CFU) | Around 2 log10 reduction | [106] | |
| Calsporin® (Bacillus subtilis C-3102) Ecobiol® (Bacillus amyloliquefaciens CECT 5940) | Day 1 and 42 in feed | C. jejuni C97ANSES640 (1 × 104 CFU) | Calsporin®: 0.25 log10 reduction on day 14 and 1.7 log10 on day 42 Ecobiol®: 1.12 log10 on day 35 and 1.2 log10 on day 42 | [107] | |
| Bacillus subtilis DSM 17299 or Saccharomyces cerevisiae boulardii | Day 21–42 in feed | C. jejuni ST45 (1 × 104 CFU) | B. subtilis: No reduction S. cerevisiae: Up to 0.3 log10 reduction | [108] | |
| Bacillus spp. (10 isolates individually tested) | Day 1 orally or intracloacally | C. jejuni cocktail of 4 strains (2.5 × 106 CFU) | Intracloacally: 1–3 log10 Orally: 1 log10 for only 1 isolate | [109] | |
| Calsporin® (Bacillus subtilis C-3102) | Day 1–42 in feed | Fecal contamination during processing | 0.2 log10 reduction on chicken carcasses | [110] | |
| Enterococcus faecalis MB 5259 | Day 1–21 orally | C. jejuni MB 4185 (KC 40) (2 × 104 CFU) | 0.4 log10 in only one of the groups received 104 CFU E. faecalis No reduction in the chickens received 108 CFU E. faecalis | [111] | |
| Enterococcus faecium NCIMB 11508 | Day 1 and 28 orally | Naturally infected | No reduction in the relative abundance of Campylobacter | [112] | |
| Microencapsulated Bifidobacterium longum PCB133 + oligosaccharides | Day 1–14 in feed | Naturally infected | Up to 1.4 log10 | [113] | |
| Bifidobacterium longum PCB 133 | Day 1–15 intraesophageally | Naturally infected | 1 log10 reduction | [114] | |
| Multi-strain | Avian Pac Soluble (Lactobacillus acidophilus + Streptococcus faecium) | Day 1–3 in drinking water | C. jejuni C101 (2.7 × 104 CFU) | Two-thirds reduction in C. jejuni shedding | [115] |
| Bacillus spp.+ Lactobacillus salivarius subsp. salivarius + L. salivarius sub sp. salicinius | Day 1 orally | C. jejuni cocktail of 4 strains (2.5 × 106 CFU) | 1–2 log10 in only one of 3 trials | [116] | |
| PrimaLac (Lactobacillus acidophilus + Lactobacillus casei + Bifidobacterium thermophilus + Enterococcus faecium) | Day 1–42 in feed | Naturally infected | 12% reduction of C. jejuni presence | [117] | |
| K-bacteria + competitive exclusion Broilact ® | Day 1–38 in drinking water | C. jejuni T23/42 (1.3 × 104 CFU) | Up to 2 log10 | [118] | |
| PoultryStar sol®(Enterococcus faecium + Pediococcus acidilactici + Bifidobacterium animalis + Lactobacillus salivarius + Lactobacillus reuteri) | Day 1–15 in drinking water | C. jejuni 3015/2010 (1 × 104 CFU) | ≥ 6 log10 | [119] | |
| Lactobacillus paracasei J.R + Lactobacillus rhamnosus 15b + Lactococcus lactis Y + Lactococcus lactis FOa | Day 1–42 in drinking water | Naturally infected | Up to 5 log10 | [120] | |
| Lavipan (multispecies probiotic): Lactococcus lactis IBB 500, Carnobacterium divergens S-1, Lactobacillus casei OCK 0915, L0915, L. plantarum OCK 0862, and Saccharomyces cerevisiae OCK 0141 | Day 1–37 in feed | Naturally infected | <1 log10 | [121] | |
| Citrobacter diversus 22 + Klebsiella pneumonia 23 + Escherichia coli 25 + mannose | Day 1 and 3 orally | C. jejuni 108 (1 × 108 CFU) | Up to 70 % reduction | [122] |
| Bacteriocins Source and Name | Dose and Duration of Administration | C. jejuni Strain and Dose | Effect on Campylobacter Colonization | References |
|---|---|---|---|---|
| Enterococcus faecium (E 50–52) | 31.2 mg/kg of feed Day 4–7 of age | 106 CFU C. jejuni isolates B1 and L4 on day of hatch | <102 CFU/g reduction on day 15 of age | [129] |
| 12.5 mg/liter of drinking water Day 35–41 of age | Environmentally infected | Below the limit of detection on days 40 and 41 of age | ||
| Enterococcus durans/faecium/hirae (E-760) | 31.2 mg/kg of feed Day 4–7 of age | 106 CFU C. jejuni isolates B1 and L4 on day of hatch | Below the limit of detection on day 7 of age | [130] |
| 125 mg/kg of feed | Naturally colonized | Below the limit of detection on day day 43 of age | ||
| Lactobacillus Salivarius (OR-7) | 250 mg/kg of feed Day 7–9 of age | 108 CFU C. jejuni strain AL-22 or BH-6 or CL-11 on day 1 of age | Below the limit of detection on day 10 of age | [126] |
| Paenibacillus polymyxa (B602), or Lactobacillus salivarius (OR7) | 250 mg/kg of feed on day 10–12 of age (turkey poults) | 106 CFU of a mixture of 3 C. coli isolates on day 3 of age | Below the limit of detection on day on day 12 of age | [127] |
| Microencapsulated Paenibacillus polymyxa (B-30509), or Lactobacillus. salivarius (B-30514) | 250 mg/kg of feed | 6 × 106 CFU on day 1–4 of age | P. polymyxa B-30509: complete elimination L. salivarius B-30514: <1 log10 on day 7 of age | [131] |
| Paenibacillus polymyxa microencapsulated (SRCAM 602) | 250 mg/kg of feed on day 7–9 of age | 108 CFU C. jejuni strain AL-22 or BH-6 or CL-11 on day 1 of age | Below the limit of detection on day 10 of age | [128] |
| Enterococcus faecium (E-760) or (E-760 E- resistant mutants (JL341, K58, or JL106)) | 5 mg/kg body weight/day orally on day 9 for 3 consecutive days | 107 CFU C. jejuni NCTC 11168 on day 2 of age | Slightly reduced on days 24 and 44 of age | [132] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taha-Abdelaziz, K.; Singh, M.; Sharif, S.; Sharma, S.; Kulkarni, R.R.; Alizadeh, M.; Yitbarek, A.; Helmy, Y.A. Intervention Strategies to Control Campylobacter at Different Stages of the Food Chain. Microorganisms 2023, 11, 113. https://doi.org/10.3390/microorganisms11010113
Taha-Abdelaziz K, Singh M, Sharif S, Sharma S, Kulkarni RR, Alizadeh M, Yitbarek A, Helmy YA. Intervention Strategies to Control Campylobacter at Different Stages of the Food Chain. Microorganisms. 2023; 11(1):113. https://doi.org/10.3390/microorganisms11010113
Chicago/Turabian StyleTaha-Abdelaziz, Khaled, Mankerat Singh, Shayan Sharif, Shreeya Sharma, Raveendra R. Kulkarni, Mohammadali Alizadeh, Alexander Yitbarek, and Yosra A. Helmy. 2023. "Intervention Strategies to Control Campylobacter at Different Stages of the Food Chain" Microorganisms 11, no. 1: 113. https://doi.org/10.3390/microorganisms11010113
APA StyleTaha-Abdelaziz, K., Singh, M., Sharif, S., Sharma, S., Kulkarni, R. R., Alizadeh, M., Yitbarek, A., & Helmy, Y. A. (2023). Intervention Strategies to Control Campylobacter at Different Stages of the Food Chain. Microorganisms, 11(1), 113. https://doi.org/10.3390/microorganisms11010113











