Proximate Drivers of Population-Level Lizard Gut Microbial Diversity: Impacts of Diet, Insularity, and Local Environment
Abstract
1. Introduction
2. Material & Methods
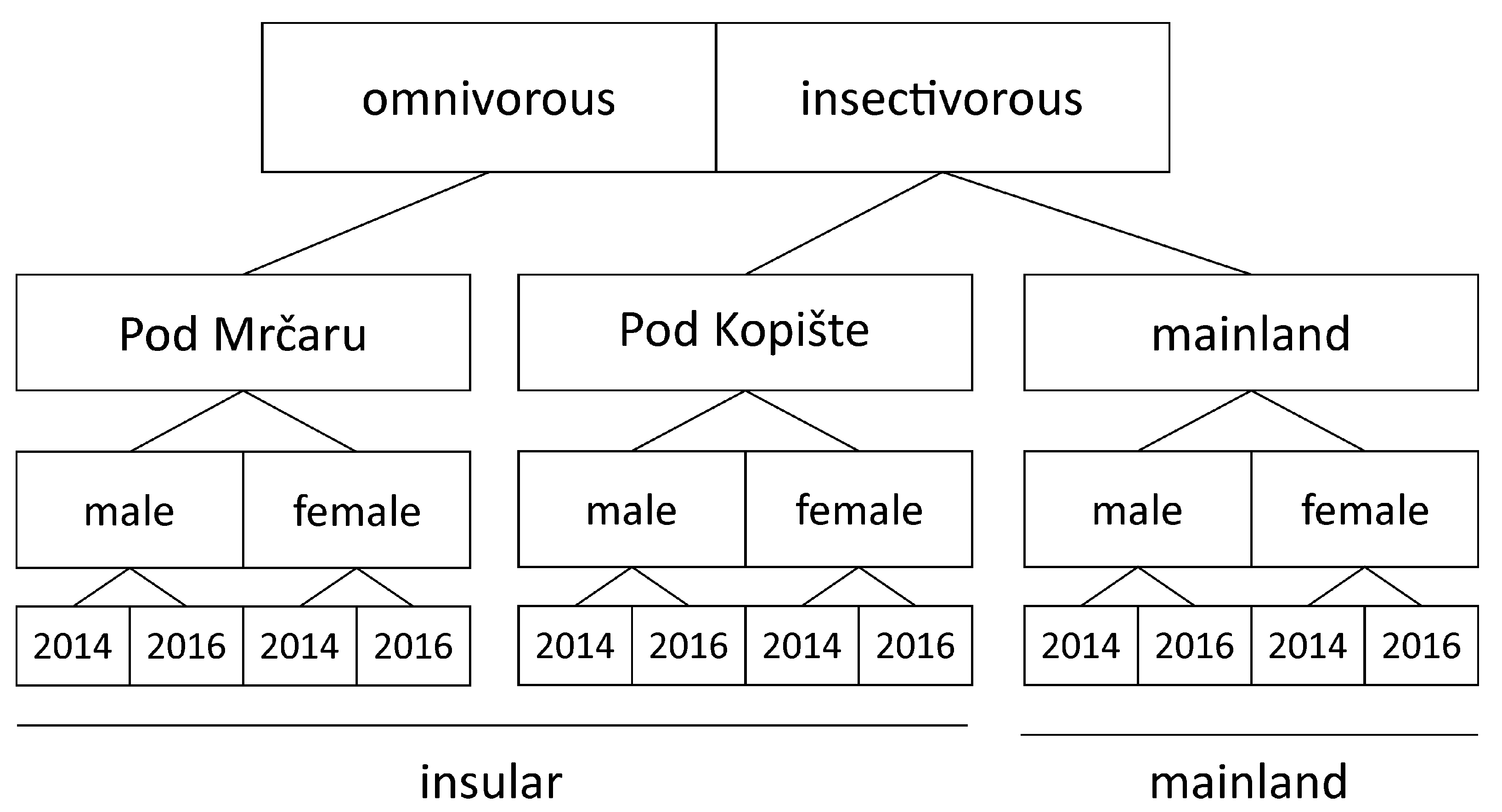
2.1. Sample Collection
2.2. 16S rRNA Gene Sequencing and Processing
2.3. Statistical Analyses
2.4. ANCOM Analysis
2.5. Alpha Diversity
2.6. Beta Diversity Index and Ordination
3. Results
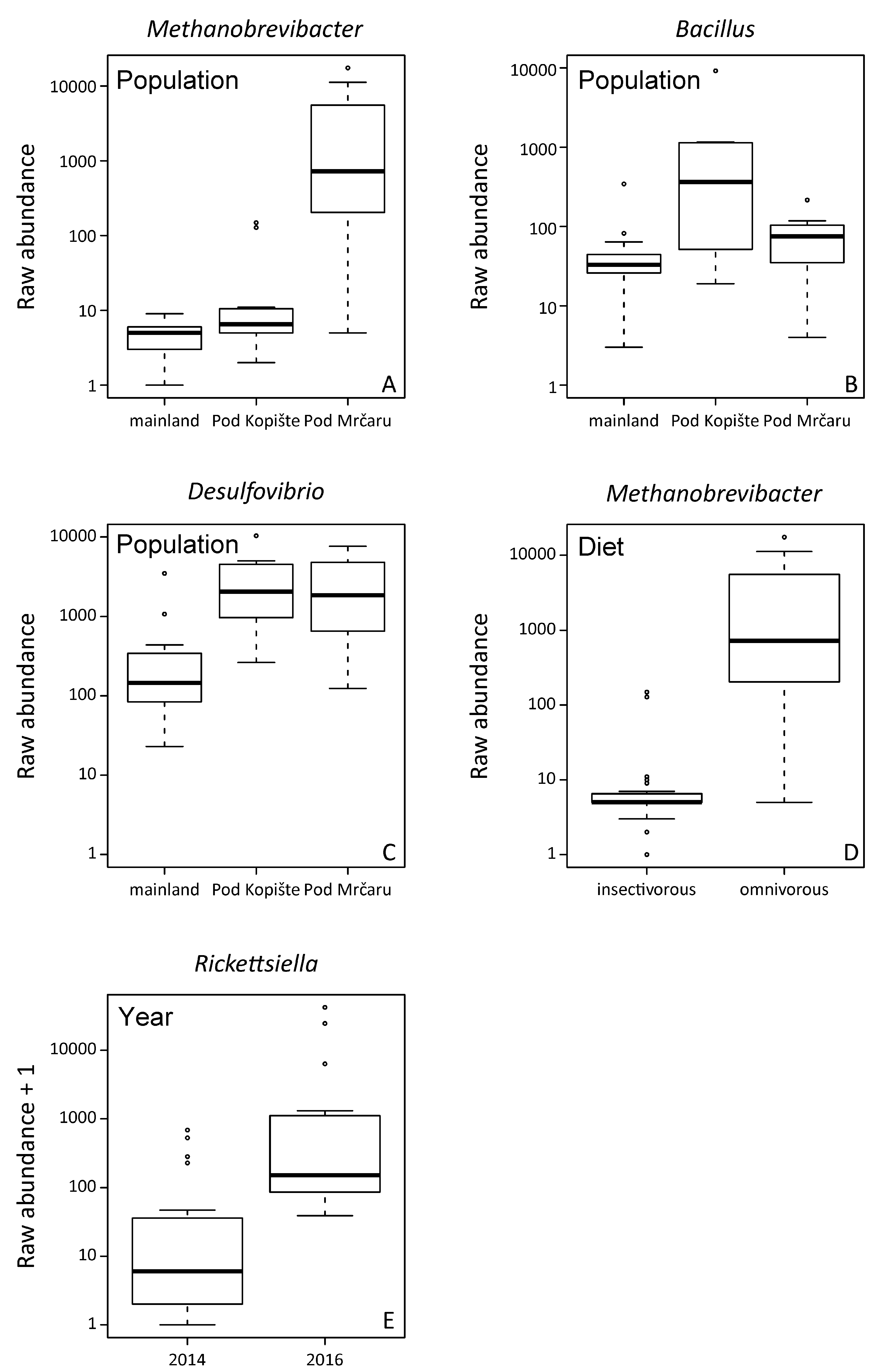
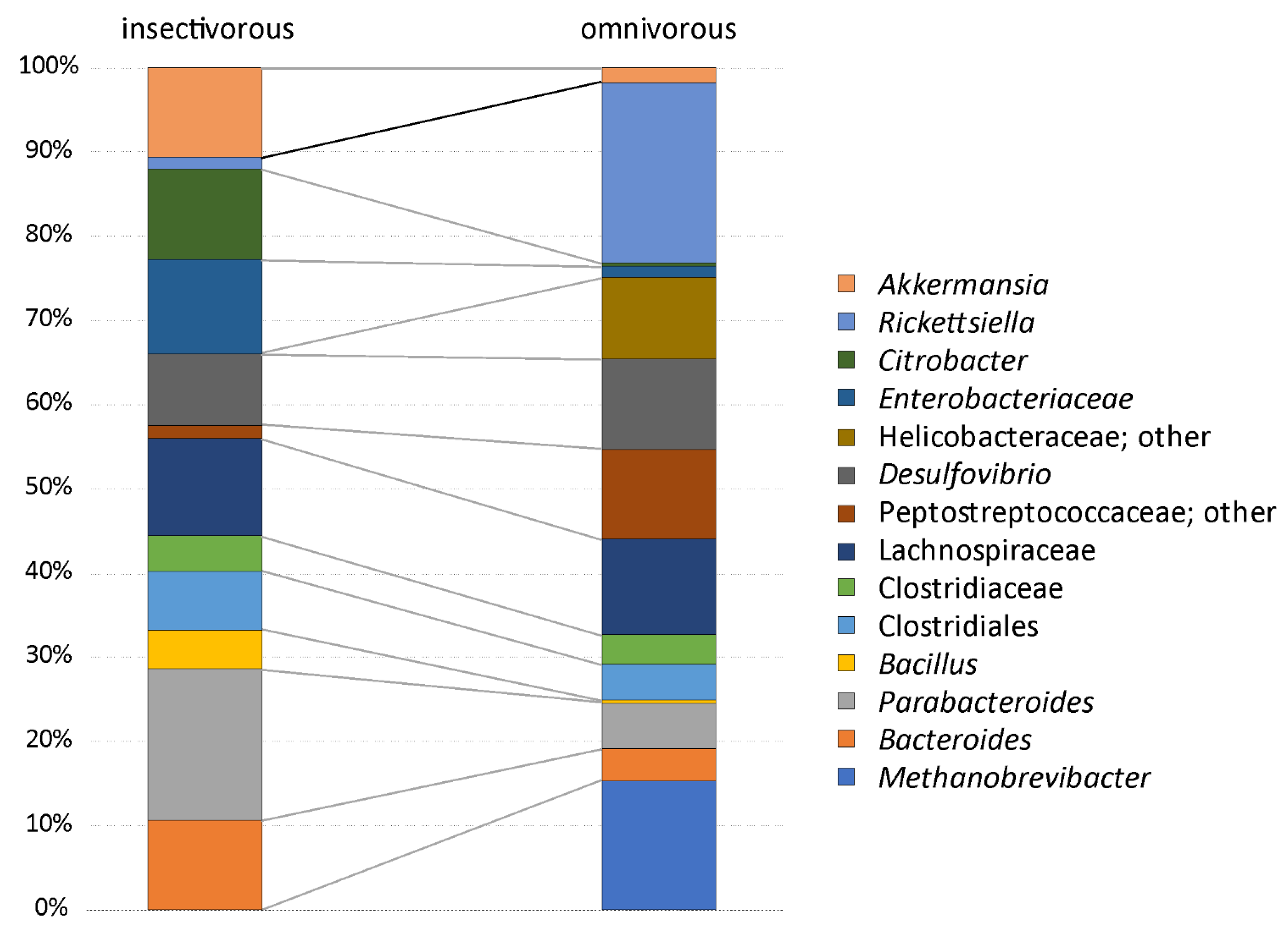
3.1. Analysis of Gut Microbiota Composition
3.2. Alpha Diversity Analyses
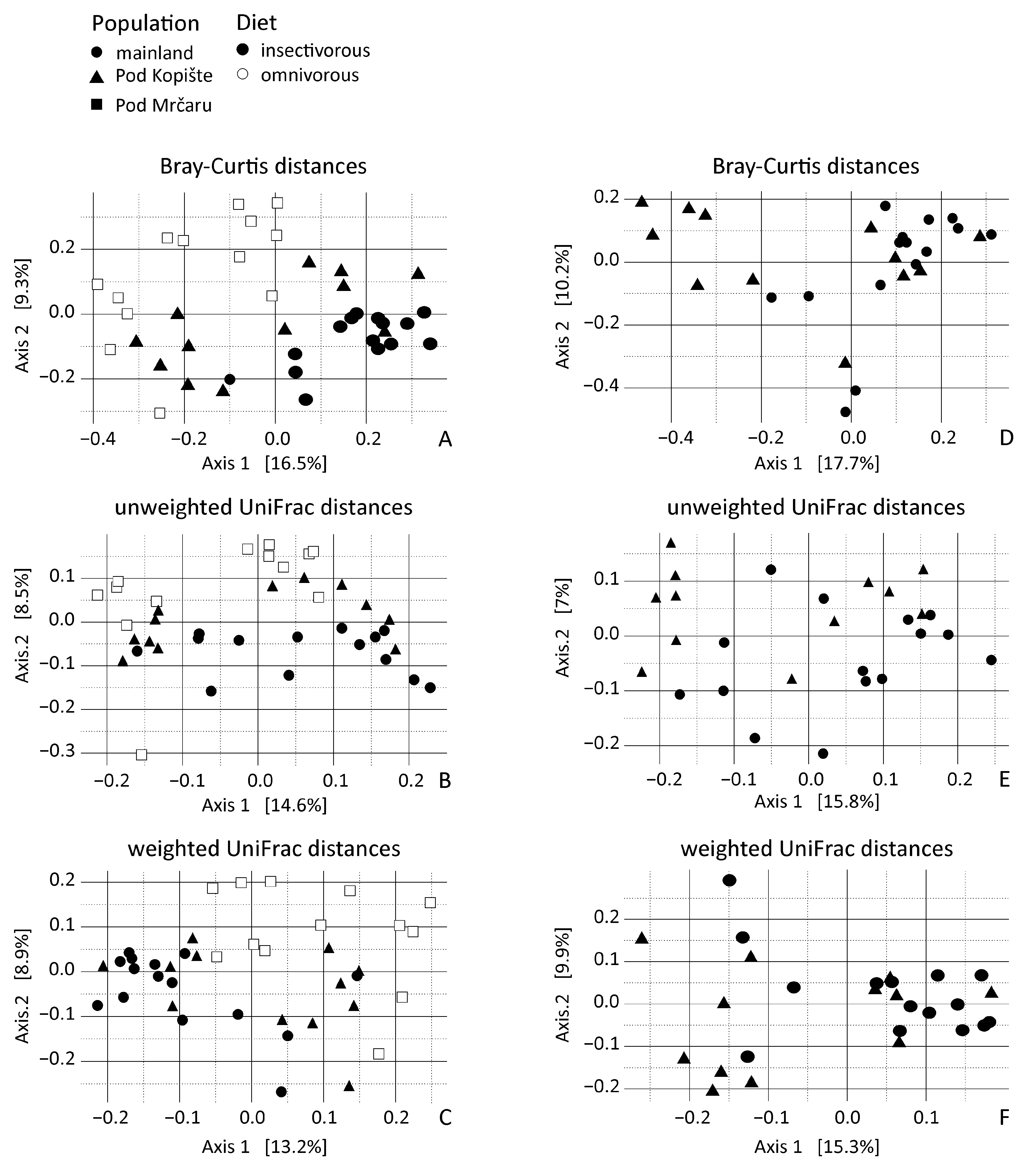
3.3. Beta Diversity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Karasov, W.H.; Martinez del Rio, C. Physiological Ecology: How Animals Process Energy, Nutrients, and Toxins; Princeton University Press: Princeton, NJ, USA, 2007; 744p. [Google Scholar]
- Ley, R.E.; Hamady, M.; Lozupone, C.; Turnbaugh, P.J.; Ramey, R.R.; Bircher, J.S.; Schlegel, M.L.; Tucker, T.A.; Schrenzel, M.D.; Knight, R.; et al. Evolution of mammals and their gut microbes. Science 2008, 320, 1647–1651. [Google Scholar] [CrossRef] [PubMed]
- McFall-Ngai, M.; Hadfield, M.G.; Bosch, T.C.G.; Carey, H.V.; Domazet-Lošo, T.; Douglas, A.E.; Wernegreen, J.J. Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl. Acad. Sci. USA 2013, 110, 3229–3236. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.A.; Ochman, H.; Hammer, T.J. Evolutionary and Ecological Consequences of Gut Microbial Communities. Annu. Rev. Ecol. Evol. Syst. 2019, 50, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, J.R.; Ravel, J. The vocabulary of microbiome research: A proposal. Microbiome 2015, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D.; Brun, A.; Magallanes, M.; Brinkerhoff, J.; Laspiur, A.; Acosta, J.C.; Caviedes-Vidal, E.; Bordenstein, S.R. Gut microbial ecology of lizards: Insights into diversity in the wild, effects of captivity, variation across gut regions and transmission. Mol. Ecol. 2017, 26, 1175–1189. [Google Scholar] [CrossRef]
- Frese, S.A.; Parker, K.; Calvert, C.C.; Mills, D.A. Diet shapes the gut microbiome of pigs during nursing and weaning. Microbiome 2015, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Gorvitovskaia, A.; Holmes, S.P.; Huse, S.M. Interpreting Prevotella and Bacteroides as biomarkers of diet and lifestyle. Microbiome 2016, 4, 15. [Google Scholar] [CrossRef]
- Ravel, J.; Blaser, M.J.; Braun, J.; Brown, E.; Bushman, F.D.; Chang, E.B.; Davies, J.; Dewey, K.G.; Dinan, T.; Dominguez-Bello, M.; et al. Human microbiome science: Vision for the future. Microbiome 2014, 2, 16. [Google Scholar] [CrossRef]
- Umu Ö, C.; Frank, J.A.; Fangel, J.U.; Oostindjer, M.; da Silva, C.S.; Bolhuis, E.J.; Bosch, G.; Willats, W.G.; Pope, P.B.; Diep, D.B. Resistant starch diet induces change in the swine microbiome and a predominance of beneficial bacterial populations. Microbiome 2015, 3, 16. [Google Scholar] [CrossRef]
- Brooks, A.W.; Kohl, K.D.; Brucker, R.M.; van Opstal, E.J.; Bordenstein, S.R. Phylosymbiosis: Relationships and Functional Effects of Microbial Communities across Host Evolutionary History. PLoS Biol. 2016, 14, e2000225. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D. Ecological and evolutionary mechanisms underlying patterns of phylosymbiosis in host-associated microbial communities. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190251. [Google Scholar] [CrossRef] [PubMed]
- Bestion, E.; Jacob, S.; Zinger, L.; Di Gesu, L.; Richard, M.; White, J.; Cote, J. Global warming reduces gut microbiota diversity in a vertebrate ectotherm. Nat. Ecol. Evol. 2017, 1, 161. [Google Scholar] [CrossRef] [PubMed]
- Indest, K.J.; Everman, S.J.; Lindsay, J.H.; Jung, C.M.; Smith, J.C.; Newell, S.B. Effects of acute exposures of 2,4,6-trinitrotoluene and inorganic lead on the fecal microbiome of the green anole (Anolis carolinensis). PLoS ONE 2018, 13, e0208281. [Google Scholar] [CrossRef] [PubMed]
- Trevelline, B.K.; McLeod, K.J.; Langkilde, T.; Kohl, K.D. Gestation alters the gut microbiome of an oviparous lizard. FEMS Microb. Ecol. 2019, 95, fiz086. [Google Scholar] [CrossRef]
- Nevo, E.; Gorman, G.; Soulé, M.; Yang, S.Y.; Clover, R.; Jovanović, V. Competitive exclusion between insular Lacerta species (Sauria, Lacertidae). Oecologia 1972, 10, 183–190. [Google Scholar] [CrossRef]
- Herrel, A.; Huyghe, K.; Vanhooydonck, B.; Backeljau, T.; Breugelmans, K.; Grbac, I.; Van Damme, R.; Irschick, D.J. Rapid large-scale evolutionary divergence in morphology and performance associated with exploitation of a different dietary resource. Proc. Natl. Acad. Sci. USA 2008, 105, 4792–4795. [Google Scholar] [CrossRef]
- Herrel, A. Herbivory and foraging mode in lizards. In Lizard Ecology: The Evolutionary Consequences of Foraging Mode; Reilly, S.M., McBrayer, L.D., Miles, D.B., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 209–236. [Google Scholar]
- Wehrle, B.A.; Herrel, A.; Nguyen-Phuc, B.Q.; Maldonado, S.; Jr Dang, R.K.; Agnihotri, R.; Tadić, Z.; German, D.P. Rapid dietary shift in Podarcis siculus resulted in localized changes in gut function. Physiol. Biochem. Zool. 2020, 93, 396–415. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D.; Brun, A.; Magallanes, M.; Brinkerhoff, J.; Laspiur, A.; Acosta, J.C.; Bordenstein, S.R.; Caviedes-Vidal, E. Physiological and microbial adjustments to diet quality permit facultative herbivory in an omnivorous lizard. J. Exp. Biol. 2016, 219, 1903–1912. [Google Scholar] [CrossRef] [PubMed]
- Hong, P.Y.; Wheeler, E.; Cann, I.K.O.; Mackie, R.I. Phylogenetic analysis of the fecal microbial community in herbivorous land and marine iguanas of the Galápagos Islands using 16S rRNA-based pyrosequencing. ISME J. 2011, 5, 1461–1470. [Google Scholar] [CrossRef]
- Hong, P.Y.; Mao, Y.; Ortiz-Kofoed, S.; Shah, R.; Cann, I.; Mackie, R.I. Metagenomic-based study of the phylogenetic and functional gene diversity in Galápagos land and marine iguanas. Microb. Ecol. 2015, 69, 444–456. [Google Scholar] [CrossRef]
- MacLeod, A.; Rodríguez, A.; Vences, M.; Orozco-terWengel, P.; García, C.; Trillmich, F.; Gentile, G.; Caccone, A.; Quezada, G.; Steinfartz, S. Hybridization masks speciation in the evolutionary history of the Galápagos marine iguana. Proc. R. Soc. B Biol. Sci. 2015, 282, 20150425. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.Y.; Ma, J.E.; Li, J.; Zhang, X.J.; Li, L.M.; He, N.; Liu, H.Y.; Luo, S.Y.; Wu, Z.J.; Han, R.C.; et al. Diets alter the gut microbiome of crocodile lizards. Front. Microbiol. 2017, 8, 2073. [Google Scholar] [CrossRef] [PubMed]
- Baldo, L.; Riera, J.L.; Mitsi, K.; Pretus, J.L. Processes shaping gut microbiota diversity in allopatric populations of the endemic Podarcis lilfordi from Menorcan Islets. FEMS Microbiol. Ecol. 2018, 94, fix186. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Kahrl, A.F.; Wu, M.; Cox, R.M. Does adaptive radiation of a host lineage promote ecological diversity of its bacterial communities? A test using gut microbiota of Anolis lizards. Mol. Ecol. 2016, 25, 4793–4804. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, N.; Tang, X.; Liu, N.; Zhao, W. Changes in intestinal microbiota across an altitudinal gradient in the lizard Phrynocephalus vlangalii. Ecol. Evol. 2018, 8, 4695–4703. [Google Scholar] [CrossRef] [PubMed]
- Taverne, M.; Fabre, A.C.; King-Gillies, N.; Krajnovic, M.; Lisicic, D.; Martin, L.; Michal, L.; Petricioli, D.; Štambuk, A.; Tadić, A.; et al. Diet variability among insular populations of Podarcis lizards reveals diverse strategies to face resource-limited environments. Ecol. Evol. 2019, 9, 12408–12420. [Google Scholar] [CrossRef]
- Vervust, B.; Van Dongen, S.; Grbac, I.; Van Damme, R. The mystery of the missing toes: Extreme levels of natural mutilation in island lizard populations. Funct. Ecol. 2009, 23, 996–1003. [Google Scholar] [CrossRef]
- Maura, M.; Vignoli, L.; Bologna, M.A.; Rugiero, L.; Luiselli, L. Population density of syntopic, differently sized lizards in three fragmented woodlands from Mediterranean Central Italy. Community Ecol. 2011, 12, 249–258. [Google Scholar] [CrossRef]
- Jelić, D. Checklist of Croatian amphibians and reptiles with bibliography of 250 years of research. Nat. Slov. 2014, 16, 17–72. [Google Scholar]
- Vervust, B.; Grbac, I.; Brecko, J.; Tvrtković, N.; Van Damme, R. Distribution of reptiles and amphibians in the nature park Lastovo Archipelago: Possible underlying biotic and abiotic causes. Nat. Croat. 2009, 18, 113–127. [Google Scholar]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Scweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucl. Acid Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 25 November 2017).
- Mandal, S.; Van Treuren, W.; White, R.A.; Eggsbo, M.; Knight, R.; Peddada, S.D. Analysis of composition of microbiomes: A novel method for studying microbial composition. Microb. Ecol. Health Dis. 2015, 26, 27663. [Google Scholar] [CrossRef]
- Gloor, G.B.; Macklaim, J.M.; Pawlowsky-Glahn, V.; Egozcue, J.J. Microbiome datasets are compositional: And this is not optional. Front. Microbiol. 2017, 8, 2224. [Google Scholar] [CrossRef] [PubMed]
- Shannon, C.E. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Su, L.; Yang, L.; Huang, S.; Su, X.; Li, Y.; Wang, F.; Wang, E.; Kang, N.; Xu, J.; Song, A. Comparative gut microbiomes of four species representing the higher and the lower termites. J. Insect Sci. 2016, 16, 97. [Google Scholar] [CrossRef] [PubMed]
- Herlemann, D.P.R.; Geissinger, O.; Brune, A. The termite group I phylum is highly diverse and widespread in the environment. Appl. Environ. Microbiol. 2007, 73, 6682–6685. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.Q.; Long, R.J.; Yang, H.; Yang, H.J.; Shen, X.H.; Shi, R.F.; Wang, Z.Y.; Du, J.G.; Qi, X.J.; Ye, Q.H. Fiber degradation potential of natural co-cultures of Neocallimastix frontalis and Methanobrevibacter ruminantium isolated from yaks (Bos grunniens) grazing on the Qinghai Tibetan Plateau. Anaerobe 2016, 39, 158–164. [Google Scholar] [CrossRef]
- Mountfort, D.O.; Asher, R.A.; Bauchop, T. Fermentation of cellulose to methane and carbon dioxide by a rumen anaerobic fungus in a triculture with Methanobrevibacter sp. strain RA1 and Methanosarcina barkeri. Appl. Environ. Microbiol. 1982, 44, 128–134. [Google Scholar] [CrossRef]
- Piknová, M.; Javorský, P.; Guczynska, W.; Kasperowicz, A.; Michalowski, T.; Pristas, P. New species of rumen treponemes. Folia Microbiol. 2006, 51, 303–305. [Google Scholar] [CrossRef]
- Santana, R.H.; Catão, E.C.P.; Lopes, F.A.C.; Constantino, R.; Barreto, C.C.; Krüger, R.H. The gut microbiota of workers of the litter-feeding termite Syntermes wheeleri (Termitidae: Syntermitinae): Archaeal, bacterial, and fungal communities. Microb. Ecol. 2015, 70, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Köhler, T.; Dietrich, C.; Scheffrahn, R.H.; Brune, A. High-resolution analysis of gut environment and bacterial microbiota reveals functional compartmentation of the gut in wood-feeding higher termites (Nasutitermes spp.). Appl. Environ. Microbiol. 2012, 78, 4691–4701. [Google Scholar] [CrossRef] [PubMed]
- Schauer, C.; Thompson, C.; Brune, A. Pyrotag sequencing of the gut microbiota of the cockroach Shelfordella lateralis reveals a highly dynamic core but only limited effects of diet on community structure. PLoS ONE 2014, 9, e85861. [Google Scholar] [CrossRef] [PubMed]
- Doolittle, W.F.; Zhaxybayeva, O. Metagenomics and the units of biological organization. Bioscience 2010, 60, 102–112. [Google Scholar] [CrossRef]
- Langille, M.G.I.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Thurber, R.L.V.; Knight, R.; et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef]
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.K.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced extinctions in the gut microbiota compound over generations. Nature 2016, 529, 212–215. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009, 137, 1712–1716. [Google Scholar] [CrossRef]
- Robertson, O.J.; McAlpine, C.; House, A.; Maron, M. Influence of interspecific competition and landscape structure on spatial homogenization of avian assemblages. PLoS ONE 2013, 8, e65299. [Google Scholar] [CrossRef]
- Anderson, M.J.; Gorley, R.N.; Clark, K.L. PRIMER 6 & PERMANOVA+; PRIMER-E Ltd: Plymouth, UK, 2009. [Google Scholar]
- Preheim, S.P.; Perrotta, A.R.; Friedman, J.; Smilie, C.; Brito, I.; Smith, M.B.; Alm, E. Computational methods for high-throughput comparative analyses of natural microbial communities. Methods Enzymol. 2013, 531, 353–370. [Google Scholar]
- Hill, M.O. Diversity and Evenness: A Unifying Notation and Its Consequences. Ecology 1973, 54, 427–432. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, P.R.; O’Hara, R.B.; Solymos, P.; Stevens, M.H.H.; Szoecs, E.; et al. Package ‘Vegan’. 2019. Available online: https://cran.r-project.org/web/packages/vegan/vegan.pdf (accessed on 25 November 2017).
- Jost, L. Entropy and diversity. Oikos 2006, 113, 363–375. [Google Scholar] [CrossRef]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. {nlme}: Linear and Nonlinear Mixed Effects Models. 2018. Available online: https://cran.r-project.org/package=nlme (accessed on 27 February 2018).
- Mathur, R.; Kim, G.; Morales, W.; Sung, J.; Rooks, E.; Pokkunuri, V.; Weitsman, S.; Barlow, G.M.; Chang, C.; Pimentel, M. Intestinal Methanobrevibacter smithii but not total bacteria is related to diet-induced weight gain in rats. Obesity 2013, 21, 748–754. [Google Scholar] [CrossRef] [PubMed]
- Samuel, B.S.; Gordon, J.I. A humanized gnotobiotic mouse model of host–archaeal–bacterial mutualism. Proc. Natl. Acad. Sci. USA 2006, 103, 10011–10016. [Google Scholar] [CrossRef]
- Fan, P.; Liu, P.; Song, P.; Chen, X.; Ma, X. Moderate dietary protein restriction alters the composition of gut microbiota and improves ileal barrier function in adult pig model. Sci. Rep. 2017, 7, 43412. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.K.; Gordon, J.I.; Secor, S.M.; Knight, R. Postprandial remodeling of the gut microbiota in Burmese pythons. ISME J. 2010, 4, 1375–1385. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.G.; das Graças Pimenta Sanna, M.; Rocha, G.A.; Rocha, A.M.; Santos, A.; Dani, R.; Marinho, F.P.; Moreira, L.S.; de Lourdes Abreu Ferrari, M.; Moura, S.B.; et al. Helicobacter Species in the Intestinal Mucosa of Patients with Ulcerative Colitis. J. Clin. Microbiol. 2004, 42, 384–386. [Google Scholar] [CrossRef]
- Leclerque, A.; Kleespies, R.G. Type IV secretion system components as phylogenetic markers of entomopathogenic bacteria of the genus Rickettsiella. FEMS Microbiol. Lett. 2008, 279, 167–173. [Google Scholar] [CrossRef][Green Version]
- Antonio, D.; Andree, K.B.; Moore, J.D.; Friedman, C.S.; Hedrick, R.P. Detection of rickettsiales-like prokaryotes by in situ hybridization in Black Abalone, Haliotis cracherodii, with withering syndrome. J. Invertebr. Pathol. 2000, 75, 180–182. [Google Scholar] [CrossRef]
- Amabebe, E.; Robert, F.O.; Agbalalah, T.; Orubu, E.S.F. Microbial dysbiosis-induced obesity: Role of gut microbiota in homoeostasis of energy metabolism. Br. J. Nutr. 2020, 123, 1127–1137. [Google Scholar] [CrossRef]
- Riddle, M.R.; Aspiras, A.C.; Gaudenz, K.; Peuß, R.; Sung, J.Y.; Martineau, B.; Peavey, M.; Box, A.C.; Tabin, J.A.; McGaugh, S.; et al. Insulin resistance in cavefish as an adaptation to a nutrient-limited environment. Nature 2018, 555, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Ilinskaya, O.N.; Ulyanova, V.V.; Yarullina, D.R.; Gataullin, I.G. Secretome of intestinal bacilli: A natural guard against pathologies. Front. Microbiol. 2017, 8, 1666. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wang, Y.; Zhang, J.; Chen, Y.; Wu, M. Isolation and identification of Citrobacter spp. from the intestine of Procambarus clarkii. J. Fish. Res. 2018, 2, 1–6. [Google Scholar] [CrossRef]
- Derrien, M.; Vaughan, E.E.; Plugge, C.M.; de Vos, W.M. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int. J. Syst. Evol. Microbiol. 2004, 54, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Sonoyama, K.; Fujiwara, R.; Takemura, N.; Ogasawara, T.; Watanabe, J.; Ito, H.; Morita, T. Response of gut microbiota to fasting and hibernation in Syrian Hamsters. Appl. Environ. Microbiol. 2009, 75, 6451–6456. [Google Scholar] [CrossRef] [PubMed]
- Rey, F.E.; Gonzalez, M.D.; Cheng, J.; Wu, M.C.; Ahern, P.P.; Gordon, J.I. Metabolic niche of a prominent sulfate-reducing human gut bacterium. Proc. Natl. Acad. Sci. USA 2013, 110, 13582–13587. [Google Scholar] [CrossRef]
- Painter, T.J. Algal Polysaccharides. In The Polysaccharides; Aspinall, G.O., Ed.; Academic Press Inc.: New York, NY, USA, 1983; Volume 2, pp. 196–285. [Google Scholar]
- Martin, M.O.; Gilman, F.R.; Weiss, S.L. Sex-specific asymmetry within the cloacal microbiota of the striped plateau lizard, Sceloporus virgatus. Symbiosis 2010, 51, 97–105. [Google Scholar] [CrossRef]
| W-Statistic | ||||
|---|---|---|---|---|
| Diet | Location | Sex | Year | |
| Methanobrevibacter | 13 * | 13 * | 0 | 3 |
| Bacteroides | 4 | 7 | 0 | 7 |
| Parabacteroides | 6 | 8 | 0 | 9 |
| Bacillus | 6 | 11 * | 0 | 6 |
| Clostridiales | 8 | 8 | 0 | 8 |
| Clostridiaceae | 7 | 8 | 0 | 6 |
| Lachnospiraceae | 7 | 7 | 0 | 5 |
| Peptostreptococcaceae; Other | 8 | 10 | 0 | 6 |
| Desulfovibrio | 7 | 11 * | 0 | 6 |
| Helicobacteraceae; Other | 8 | 10 | 0 | 5 |
| Enterobacteriaceae | 7 | 8 | 0 | 3 |
| Citrobacter | 7 | 8 | 0 | 5 |
| Rickettsiella | 4 | 10 | 0 | 13 * |
| Akkermansia | 8 | 9 | 0 | 4 |
| Fixed Effect | Random Effect | Likelihood Ratio | p |
|---|---|---|---|
| Diet | Sex, Year, Insularity, Location | 5.18 | 0.023 |
| Location | Sex, Year, Insularity, Diet | 4.25 | 0.12 |
| Insularity | Sex, Year, Location, Diet | 0.24 | 0.62 |
| Sex | Year, Insularity, Location, Diet | 0.45 | 0.50 |
| Year | Sex, Insularity, Location, Diet | 2.38 | 0.12 |
| Distance Measure | Model | Db-RDA | ||
|---|---|---|---|---|
| F | R2 Adjusted | p | ||
| Bray–Curtis | Location | 2.02 | 0.050 | <0.001 |
| Unweighted UniFrac | Location | 2.86 | 0.087 | <0.001 |
| Weighted UniFrac | Location | 2.33 | 0.066 | 0.001 |
| Factor | F | p | |
|---|---|---|---|
| Bray–Curtis | Diet | 5.49 | 0.024 |
| Location | 1.12 | 0.35 | |
| Insularity | 0.79 | 0.38 | |
| Year | 4.43 | 0.041 | |
| Sex | 2.83 | 0.099 | |
| Unweighted UniFrac | Diet | 4.32 | 0.032 |
| Location | 1.21 | 0.34 | |
| Insularity | 0.39 | 0.59 | |
| Year | 0.37 | 0.57 | |
| Sex | 3.07 | 0.09 | |
| Weighted UniFrac | Diet | 0.11 | 0.74 |
| Location | 0.02 | 0.98 | |
| Insularity | 0.53 | 0.47 | |
| Year | 11.11 | 0.0014 | |
| Sex | 1.37 | 0.24 |
| Distance Measure | Model | Controlled Factors | F | R2 Adjusted | p |
|---|---|---|---|---|---|
| Bray–Curtis | Location | NA | 1.96 | 0.034 | 0.006 |
| Unweighted UniFrac | Location + Sex + Year | NA | 1.72 | 0.077 | <0.001 |
| Location | Sex + Year | 2.15 | 0.058 | 0.0036 | |
| Sex | Location + Year | 1.61 | 0.023 | 0.0086 | |
| Year | Location + Sex | 1.38 | 0.015 | 0.028 | |
| Weighted UniFrac | Location | NA | 1.96 | 0.036 | 0.0079 |
| Factor | F | p | |
|---|---|---|---|
| Bray–Curtis | Location | 3.09 | 0.10 |
| Year | 3.09 | 0.088 | |
| Sex | 4.93 | 0.034 | |
| Unweighted UniFrac | Location | 0.89 | 0.35 |
| Year | 0.61 | 0.44 | |
| Sex | 6.76 | 0.014 | |
| Weighted UniFrac | Location | 0.78 | 0.39 |
| Year | 0.389 | 0.55 | |
| Sex | 0.82 | 0.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemieux-Labonté, V.; Vigliotti, C.; Tadic, Z.; Wehrle, B.; Lopez, P.; Bapteste, E.; Lapointe, F.-J.; German, D.P.; Herrel, A. Proximate Drivers of Population-Level Lizard Gut Microbial Diversity: Impacts of Diet, Insularity, and Local Environment. Microorganisms 2022, 10, 1550. https://doi.org/10.3390/microorganisms10081550
Lemieux-Labonté V, Vigliotti C, Tadic Z, Wehrle B, Lopez P, Bapteste E, Lapointe F-J, German DP, Herrel A. Proximate Drivers of Population-Level Lizard Gut Microbial Diversity: Impacts of Diet, Insularity, and Local Environment. Microorganisms. 2022; 10(8):1550. https://doi.org/10.3390/microorganisms10081550
Chicago/Turabian StyleLemieux-Labonté, Virginie, Chloé Vigliotti, Zoran Tadic, Beck Wehrle, Philippe Lopez, Eric Bapteste, François-Joseph Lapointe, Donovan P. German, and Anthony Herrel. 2022. "Proximate Drivers of Population-Level Lizard Gut Microbial Diversity: Impacts of Diet, Insularity, and Local Environment" Microorganisms 10, no. 8: 1550. https://doi.org/10.3390/microorganisms10081550
APA StyleLemieux-Labonté, V., Vigliotti, C., Tadic, Z., Wehrle, B., Lopez, P., Bapteste, E., Lapointe, F.-J., German, D. P., & Herrel, A. (2022). Proximate Drivers of Population-Level Lizard Gut Microbial Diversity: Impacts of Diet, Insularity, and Local Environment. Microorganisms, 10(8), 1550. https://doi.org/10.3390/microorganisms10081550








