Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula)
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Collection of Blood Samples and Haematology and Blood Chemistry Analysis
2.3. Detection of Trypanosomes and Babesias in Blood Samples
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Calisher, C.H.; Childs, J.E.; Field, H.E.; Holmes, K.V.; Schountz, T. Bats: Important reservoir hosts of emerging viruses. Clin. Microbiol. Rev. 2006, 19, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Kosoy, M.; Bai, Y.; Lynch, T.; Kuzmin, I.V.; Niezgoda, M.; Franka, R.; Agwanda, B.; Breiman, R.F.; Rupprecht, C.E. Bartonella spp. in Bats, Kenya. Emerg. Infect. Dis. 2010, 16, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Evans, N.J.; Brown, K.; Timofte, D.; Simpson, V.R.; Birtles, R.J. Fatal Borreliosis in Bat Caused by Relapsing Fever Spirochete, United Kingdom. Emerg. Infect. Dis. 2009, 15, 1331–1333. [Google Scholar] [CrossRef] [PubMed]
- García-Fraile, P.; Chudíčková, M.; Benada, O.; Pikula, J.; Kolařík, M. Serratia myotis sp. nov. and Serratia vespertilionis sp. nov. isolated from bats hibernating in caves in the Czech Republic. Int. J. Syst. Evol. Microbiol. 2015, 65, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Blehert, D.S.; Maluping, R.P.; Green, D.E.; Berlowski-Zier, B.M.; Ballman, A.E.; Langenberg, J.A. Acute pasteurellosis in wild big brown bats (Eptesicus fuscus). J. Wildl. Dis. 2014, 50, 136–139. [Google Scholar] [CrossRef]
- Mühldorfer, K. Bats and Bacterial Pathogens: A Review. Zoonoses Public Health 2013, 60, 93–103. [Google Scholar] [CrossRef]
- Sebghati, T.S.; Engle, J.T.; Goldman, W.E. Intracellular parasitism by Histoplasma capsulatum: Fungal virulence and calcium dependence. Science 2000, 290, 1368–1372. [Google Scholar] [CrossRef]
- Gardner, R.A.; Molyneux, D.H. Schizotrypanum in British bats. Parasitology 1988, 97, 43–50. [Google Scholar] [CrossRef]
- Steindel, M.; Grisard, E.C.; de Carvalho Pinto, C.J.; Cordeiro, F.D.; Ribeiro-Rodriques, R.; Romanha, A.J. Characterization of trypanosomes from the subgenus Schizotrypanum isolated from bats, Eptesicus sp. (Chiroptera: Vespertilionidae), captured in Florianópolis, Santa Catarina State, Brazil. J. Parasitol. 1998, 84, 601–607. [Google Scholar] [CrossRef]
- Lisboa, C.V.; Pinho, A.P.; Herrera, H.M.; Gerhardt, M.; Cupolillo, E.; Jansen, A.M. Trypanosoma cruzi (Kinetoplastida, Trypanosomatidae) genotypes in neotropical bats in Brazil. Vet. Parasitol. 2008, 156, 314–318. [Google Scholar] [CrossRef]
- Hamilton, P.B.; Teixeira, M.M.G.; Stevens, J.R. The evolution of Trypanosoma cruzi: The ‘bat seeding’ hypothesis. Trends Parasitol. 2012, 28, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, P.B.; Cruickshank, C.; Stevens, J.R.; Teixeira, M.M.G.; Mathews, F. Parasites reveal movement of bats between the New and Old Worlds. Mol. Phylogenet. Evol. 2012, 63, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Lima, L.; Espinosa-Álvarez, O.; Hamilton, P.B.; Neves, L.; Takata, C.S.A.; Campaner, M.; Attias, M.; de Souza, W.; Camargo, E.P.; Teixeira, M.M.G. Trypanosoma livingstonei: A new species from African bats supports the bat seeding hypothesis for the Trypanosoma cruzi clade. Parasites Vectors 2013, 6, 221. [Google Scholar] [CrossRef] [PubMed]
- Cottontail, V.M.; Kalko, E.K.; Cottontail, I.; Wellinghausen, N.; Tschapka, M.; Perkins, S.L.; Pinto, C.M. High local diversity of Trypanosoma in a common bat species, and implications for the biogeography and taxonomy of the T. cruzi clade. PLoS ONE 2014, 9, e108603. [Google Scholar] [CrossRef] [PubMed]
- Hodo, C.L.; Goodwin, C.C.; Mayes, B.C.; Mariscal, J.A.; Waldrup, K.A.; Hamer, S.A. Trypanosome species, including Trypanosoma cruzi, in sylvatic and peridomestic bats of Texas, USA. Acta Trop. 2016, 164, 259–266. [Google Scholar] [CrossRef]
- Wang, L.J.; Han, H.J.; Zhao, M.; Liu, J.W.; Luo, L.M.; Wen, H.L.; Qin, X.R.; Zhou, C.M.; Qi, H.; Yu, H.; et al. Trypanosoma dionisii in insectivorous bats from northern China. Acta Trop. 2019, 193, 124–128. [Google Scholar] [CrossRef]
- Concannon, R.; Wynn-Owen, K.; Simpson, V.R.; Birtles, R.J. Molecular characterization of haemoparasites infecting bats (Microchiroptera) in Cornwall, UK. Parasitology 2005, 131, 489–496. [Google Scholar] [CrossRef]
- Gardner, R.A.; Molyneux, D.H. Polychromophilus murinus: A malarial parasite of bats: Life-history and ultrastructural studies. Parasitology 1988, 96, 591–605. [Google Scholar] [CrossRef]
- Corduneanu, A.; Hrazdilová, K.; Sándor, A.D.; Matei, I.A.; Ionică, A.M.; Barti, L.; Ciocănău, M.A.; Măntoiu, D.S.; Coroiu, I.; Hornok, S.; et al. Babesia vesperuginis, a neglected piroplasmid: New host and geographical records, and phylogenetic relations. Parasites Vectors 2017, 10, 598. [Google Scholar] [CrossRef]
- Ranaivoson, H.C.; Héraud, J.M.; Goethert, H.K.; Telford, S.R., III; Rabetafika, L.; Brook, C.E. Babesial infection in the Madagascan flying fox, Pteropus rufus É. Geoffroy, 1803. Parasites Vectors 2019, 12, 51. [Google Scholar] [CrossRef]
- Schaer, J.; Perkins, S.L.; Decher, J.; Leendertz, F.H.; Fahr, J.; Weber, N.; Matuschewski, K. High diversity of West African bat malaria parasites and a tight link with rodent Plasmodium taxa. Proc. Natl. Acad. Sci. USA 2013, 110, 17415–17419. [Google Scholar] [CrossRef] [PubMed]
- Minozzo, G.A.; da Silva Mathias, B.; Riediger, I.N.; de Oliveira Guimarães, L.; dos Anjos, C.C.; Monteiro, E.F.; dos Santos, A.P.; Biondo, A.W.; Kirchgatter, K. First Molecular Detection of Polychromophilus Parasites in Brazilian Bat Species. Microorganisms 2021, 9, 1240. [Google Scholar] [CrossRef] [PubMed]
- Munshi-South, J.; Wilkinson, G.S. Bats and birds: Exceptional longevity despite high metabolic rates. Ageing Res. Rev. 2010, 9, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Cowled, C.; Shi, Z.; Huang, Z.; Bishop-Lilly, K.A.; Fang, X.; Wynne, J.W.; Xiong, Z.; Baker, M.L.; Zhao, W.; et al. Comparative Analysis of Bat Genomes Provides Insight into the Evolution of Flight and Immunity. Science 2013, 339, 456–460. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, T.J.; Cryan, P.; Cunningham, A.A.; Fooks, A.R.; Hayman, D.T.S.; Luis, A.D.; Peel, A.J.; Plowright, R.K.; Wood, J.L.N. Bat flight and zoonotic viruses. Emerg. Infect. Dis. 2014, 20, 741–745. [Google Scholar] [CrossRef]
- Flieger, M.; Bandouchova, H.; Cerny, J.; Chudickova, M.; Kolarik, M.; Kovacova, V.; Martinkova, N.; Novak, P.; Sebesta, O.; Stodulkova, E.; et al. Vitamin B-2 as a virulence factor in Pseudogymnoascus destructans skin infection. Sci. Rep. 2016, 6, 33200. [Google Scholar] [CrossRef]
- Carey, H.V.; Andrews, M.T.; Martin, S.L. Mammalian hibernation: Cellular and molecular responses to depressed metabolism and low temperature. Physiol. Rev. 2003, 83, 1153–1181. [Google Scholar] [CrossRef]
- Storey, K.B. Out Cold: Biochemical Regulation of Mammalian Hibernation—A Mini-Review. Gerontology 2010, 56, 220–230. [Google Scholar] [CrossRef]
- Drew, K.L.; Osborne, P.G.; Frerichs, K.U.; Hu, Y.; Koren, R.E.; Hallenbeck, J.M.; Rice, M.E. Ascorbate and glutathione regulation in hibernating ground squirrels. Brain Res. 1999, 851, 1–8. [Google Scholar] [CrossRef]
- Morin, P., Jr.; Ni, Z.; McMullen, D.C.; Storey, K.B. Expression of Nrf2 and its downstream gene targets in hibernating 13-lined ground squirrels, Spermophilus tridecemlineatus. Mol. Cell. Biochem. 2008, 312, 121–129. [Google Scholar] [CrossRef]
- Bouma, H.R.; Strijkstra, A.M.; Boerema, A.S.; Deelman, L.E.; Epema, A.H.; Hut, R.A.; Kroese, F.G.; Henning, R.H. Blood cell dynamics during hibernation in the European Ground Squirrel. Vet. Immunol. Immunopathol. 2010, 136, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Bouma, H.R.; Carey, H.V.; Kroese, F.G.M. Hibernation: The immune system at rest? J. Leukoc. Biol. 2010, 88, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Haitlinger, R.; Lupicki, D. Arthropods (Acari, Siphonaptera, Heteroptera, Psocoptera) associated with Nyctalus noctula (Schreber, 1774) (Chiroptera: Vespertilionidae) in Southern Poland. Wiad. Parazytol. 2008, 54, 124–130. [Google Scholar]
- Hornok, S. Ixodes vespertilionis Koch, 1844 (Figs. 29–31). In Ticks of Europe and North Africa, 1st ed.; Estrada-Peña, A., Mihalcs, A.D., Petney, T.N., Eds.; Springer: Berlin, Germany, 2017; pp. 97–107. [Google Scholar]
- Lučan, R.K.; Bandouchova, H.; Bartonička, T.; Pikula, J.; Zahradníková, A., Jr.; Zukal, J.; Martínková, N. Ectoparasites may serve as vectors for the white-nose syndrome fungus. Parasites Vectors 2016, 9, 16. [Google Scholar] [CrossRef]
- George, D.B.; Webb, C.T.; Farnsworth, M.L.; O’Shea, T.J.; Bowen, R.A.; Smith, D.L.; Stanley, T.R.; Ellison, L.E.; Rupprecht, C.E. Host and viral ecology determine bat rabies seasonality and maintenance. Proc. Natl. Acad. Sci. USA 2011, 108, 10208–10213. [Google Scholar] [CrossRef]
- Callait, M.P.; Gauthier, D. Parasite adaptation to hibernation in Alpine Marmots (Marmota marmota). In Life in the Cold: Eleventh International Hibernation Symposium, 1st ed.; Heldmaier, G., Klingenspor, M., Eds.; Springer: Berlin, Germany, 2000; pp. 139–146. [Google Scholar]
- Coggins, J.R.; Tedesco, J.L.; Rupprecht, C.E. Seasonal changes and overwintering of parasites in the bat, Myotis lucifugus (Le Conte) in a Wisconsin hibernaculum. Am. Midl. Nat. 1982, 107, 305–315. [Google Scholar] [CrossRef]
- Ramírez, J.D.; Tapia-Calle, G.; Muñoz-Cruz, G.; Poveda, C.; Rendón, L.M.; Hincapié, E.; Guhl, F. Trypanosome species in neo-tropical bats: Biological, evolutionary and epidemiological implications. Infect. Genet. Evol. 2014, 22, 250–256. [Google Scholar] [CrossRef]
- Barnabe, C.; Brisse, S.; Tibayrenc, M. Phylogenetic diversity of bat trypanosomes of subgenus Schizotrypanum based on multilocus enzyme electrophoresis, random amplified polymorphic DNA, and cytochrome b nucleotide sequence analyses. Infect. Genet. Evol. 2003, 2, 201–208. [Google Scholar] [CrossRef]
- Lima, L.; da Silva, F.M.; Neves, L.; Attias, M.; Takata, C.S.A.; Campaner, M.; de Souza, W.; Hamilton, P.B.; Teixeira, M.M.G. Evolutionary insights from bat trypanosomes: Morphological, developmental and phylogenetic evidence of a new species, Trypanosoma (Schizotrypanum) erneyi sp. nov., in African bats closely related to Trypanosoma (Schizotrypanum) cruzi and allied species. Protist 2012, 163, 856–872. [Google Scholar] [CrossRef]
- Gardner, R.A.; Molyneux, D.H.; Stebbings, R.E. Studies on the prevalence of haematozoa of British bats. Mammal. Rev. 1987, 17, 75–80. [Google Scholar] [CrossRef]
- Gardner, R.A.; Molyneux, D.H. Babesia vesperuginis: Natural and experimental infections in British bats (Microchiroptera). Parasitology 1987, 95, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Bower, S.M.; Woo, P.T.K. The development of Trypanosoma (Schizotrypanum) hedricki in Cimex brevis (Hemiptera: Cimicidae). Can. J. Zool. 1981, 59, 546–554. [Google Scholar] [CrossRef]
- Hornok, S.; Szőke, K.; Kováts, D.; Estók, P.; Görföl, T.; Boldogh, S.A.; Takács, N.; Kontschán, J.; Földvári, G.; Barti, L.; et al. DNA of Piroplasms of ruminants and dogs in Ixodid bat ticks. PLoS ONE 2016, 11, e0167735. [Google Scholar] [CrossRef]
- Simpson, V.R. Veterinary advances in the investigation of wildlife diseases in Britain. Res. Vet. Sci 2000, 69, 11–16. [Google Scholar] [CrossRef]
- Oliveira, M.P.C.; Cortes, M.; Maeda, F.Y.; Fernandes, M.C.; Haapalainen, E.F.; Yoshida, N.; Mortara, R.A. Unique behavior of Trypanosoma dionisii interacting with mammalian cells: Invasion, intracellular growth, and nuclear localization. Acta Trop. 2009, 110, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Pikula, J.; Bandouchova, H.; Kovacova, V.; Linhart, P.; Piacek, V.; Zukal, J. Reproduction of Rescued Vespertilionid Bats (Nyctalus noctula) in Captivity: Veterinary and Physiologic Aspects. Vet. Clin. N. Am. Exot. Anim. Pract. 2017, 20, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Bandouchova, H.; Bartonička, T.; Berkova, H.; Brichta, J.; Kokurewicz, T.; Kovacova, V.; Linhart, P.; Piacek, V.; Pikula, J.; Zahradníková, A.; et al. Alterations in the health of hibernating bats under pathogen pressure. Sci. Rep. 2018, 8, 6067. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Seward, E.A.; Votýpka, J.; Kment, P.; Lukeš, J.; Kelly, S. Description of Phytomonas oxycareni n. sp. from the salivary glands of Oxycarenus lavaterae. Protist 2017, 168, 71–79. [Google Scholar] [CrossRef]
- Zintl, A.; Finnerty, E.J.; Murphy, T.M.; de Waal, T.; Gray, J.S. Babesias of red deer (Cervus elaphus) in Ireland. Vet. Res. 2011, 42, 7. [Google Scholar] [CrossRef]
- Basic Local Alignment Search Tool (BLAST). Available online: https://blast.ncbi.nlm.nih.gov/Blast.cgi, (accessed on 23 April 2019).
- Chimelli, L.; Scaravilli, F. Trypanosomiasis. Brain Pathol. 1997, 7, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Köster, L.S.; Lobetti, R.G.; Kelly, P. Canine babesiosis: A perspective on clinical complications, biomarkers, and treatment. Vet. Med. 2015, 6, 119–128. [Google Scholar] [CrossRef]
- Jacobson, L.S.; Lobetti, R. Glucose, lactate, and pyruvate concentrations in dogs with babesiosis. Am. J. Vet. Res. 2005, 66, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Leisewitz, A.L.; Jacobson, L.S.; de Morais, H.S.A.; Reyers, F. The Mixed Acid-Base Disturbances of Severe Canine Babesiosis. J. Vet. Intern. Med. 2001, 15, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, S.L.; Abou-Madi, N.; Messick, J.B.; Birkenheuer, A.; Kollias, G.V. Diagnosis and Treatment of Babesia odocoilei in Captive Reindeer (Rangifer tarandus tarandus) and Recognition of Three Novel Host Species. J. Zoo Wildl. Med. 2009, 40, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Usinger, R.L. Monograph of Cimicidae (Hemiptera—Heteroptera), 1st ed.; Entomological Society of America: New York, NY, USA, 1966; pp. 10–33. [Google Scholar]
- Manzano-Román, R.; Díaz-Martín, V.; de la Fuente, J.; Pérez-Sánchez, R. Soft ticks as pathogen vectors: Distribution, surveillance and control. In Parasitology, 1st ed.; Shah, M.M., Ed.; InTechOpen: London, UK, 2012; pp. 125–162. [Google Scholar]
- Balvín, O.; Ševčík, M.; Jahelková, H.; Bartonička, T.; Orlova, M.; Vilímková, J. Transport of bugs of the genus Cimex (Heteroptera: Cimicidae) by bats in western Palaearctic. Vespertilio 2012, 16, 43–54. [Google Scholar]
- Cardoso, M.S.; Reis-Cunha, J.L.; Bartholomeu, D.C. Evasion of the Immune Response by Trypanosoma cruzi during Acute Infection. Front. Immunol. 2016, 6, 659. [Google Scholar] [CrossRef]
- Chauvin, A.; Moreau, E.; Bonnet, S.; Plantard, O.; Malandrin, L. Babesia and its hosts: Adaptation to long-lasting interactions as a way to achieve efficient transmission. Vet. Res. 2009, 40, 37. [Google Scholar] [CrossRef]
- Molyneux, D.H. Trypanosomes of Bats. In Parasitic Protozoa, 1st ed.; Kreier, J.P., Baker, J.R., Eds.; Academic Press: New York, NY, USA, 1991; pp. 195–223. [Google Scholar]
- Ponte-Sucre, A. An Overview of Trypanosoma brucei Infections: An Intense Host–Parasite Interaction. Front. Microbiol. 2016, 7, 2126. [Google Scholar] [CrossRef]
- Lozano, I.M.D.; De Pablos, L.M.; Longhi, S.A.; Zago, M.P.; Schijman, A.G.; Osuna, A. Immune complexes in chronic Chagas disease patients are formed by exovesicles from Trypanosoma cruzi carrying the conserved MASP N-terminal region. Sci. Rep. 2017, 7, 44451. [Google Scholar] [CrossRef]
- Linhart, P.; Bandouchova, H.; Zukal, J.; Votypka, J.; Kokurewicz, T.; Dundarova, H.; Apoznanski, G.; Heger, T.; Kubickova, A.; Nemcova, M.; et al. Trypanosomes in Eastern and Central European bats. Acta Vet. Brno 2020, 89, 69–78. [Google Scholar] [CrossRef]
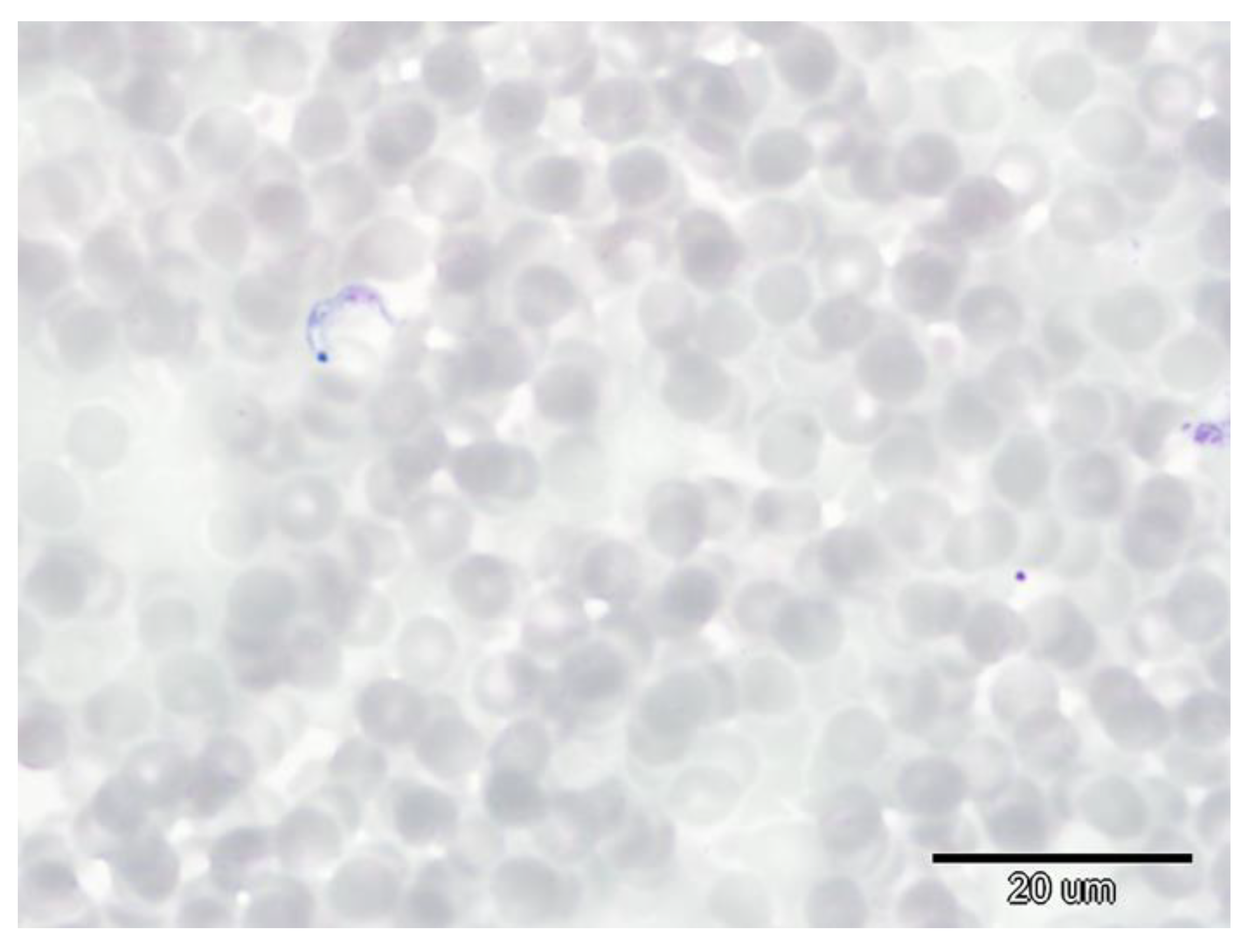
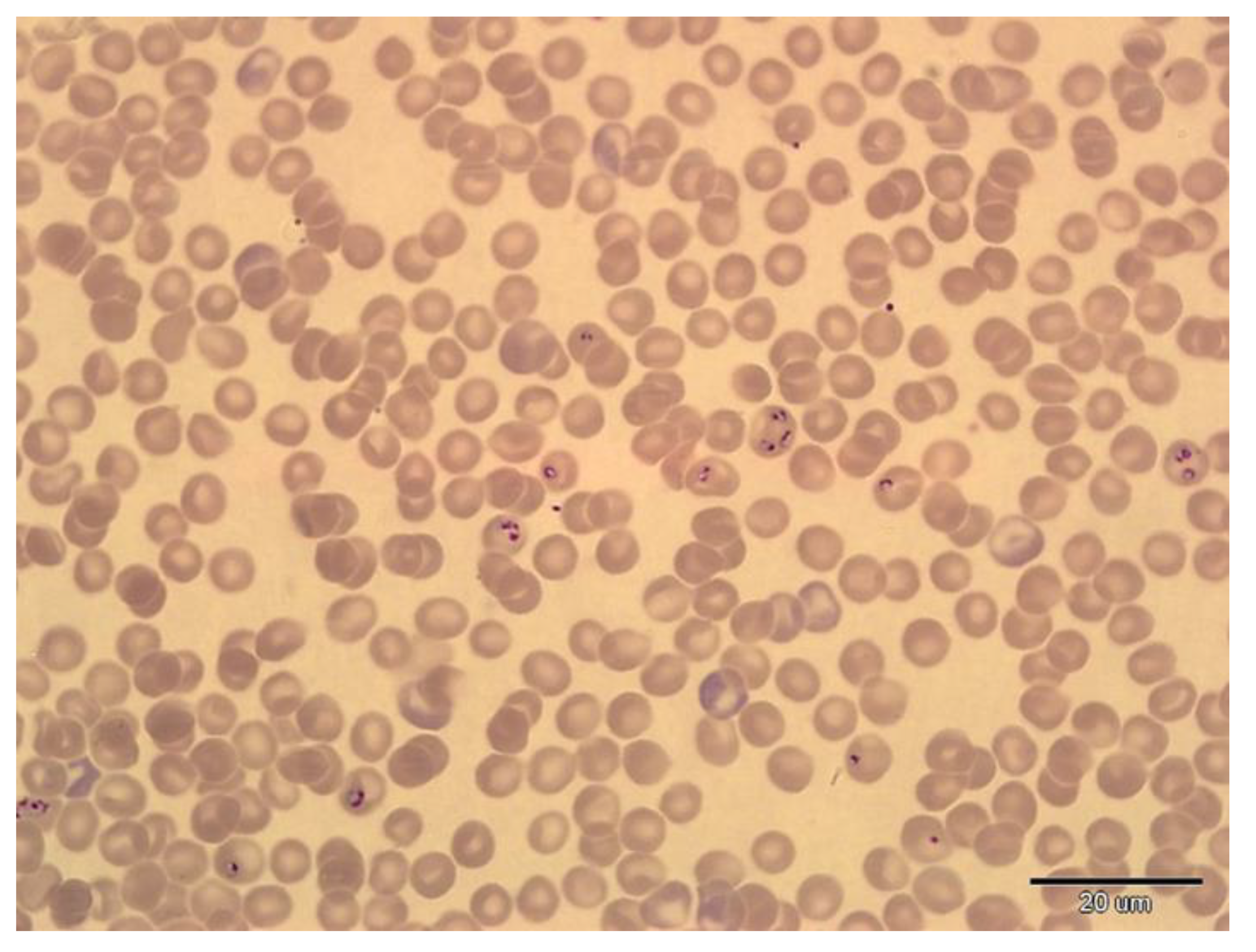
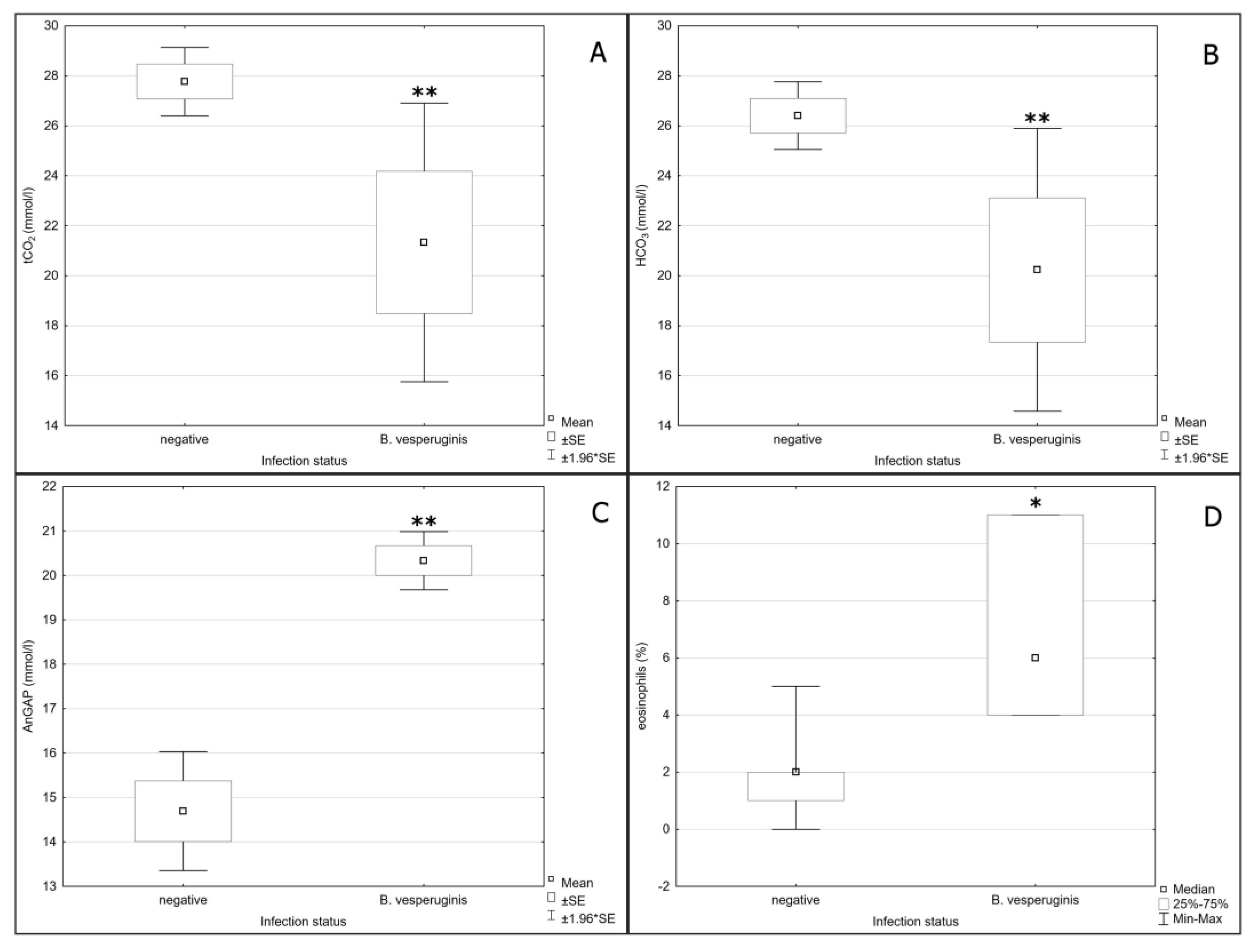
| Sex | No. | B. vesperuginis | T. dionisii | T. vespertilionis |
|---|---|---|---|---|
| Female | 35 | 8.6% | 40.0% | 8.6% |
| Male | 15 | 6.7% | 26.7% | 6.7% |
| Total | 50 | 8.0% | 36.0% | 8.0% |
| Sex | No. | B. vesperuginis | T. dionisii | T. vespertilionis |
|---|---|---|---|---|
| Female | 35 | 5.7% | 28.6% | 0% |
| Male | 15 | 6.7% | 20.0% | 0% |
| Total | 50 | 6.0% | 26.0% | 0% |
| Parameter | Infection Status | Measures | Sex | Change |
|---|---|---|---|---|
| tCO2 | F = 6.209/p = 0.011 | F = 4.945/p = 0.042 | - | ↓ |
| HCO3 | F = 6.052/p = 0.018 | F = 5.863/p = 0.028 | - | ↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Linhart, P.; Bandouchova, H.; Zukal, J.; Votýpka, J.; Baláž, V.; Heger, T.; Kalocsanyiova, V.; Kubickova, A.; Nemcova, M.; Sedlackova, J.; et al. Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula). Microorganisms 2022, 10, 1028. https://doi.org/10.3390/microorganisms10051028
Linhart P, Bandouchova H, Zukal J, Votýpka J, Baláž V, Heger T, Kalocsanyiova V, Kubickova A, Nemcova M, Sedlackova J, et al. Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula). Microorganisms. 2022; 10(5):1028. https://doi.org/10.3390/microorganisms10051028
Chicago/Turabian StyleLinhart, Petr, Hana Bandouchova, Jan Zukal, Jan Votýpka, Vojtech Baláž, Tomas Heger, Vendula Kalocsanyiova, Aneta Kubickova, Monika Nemcova, Jana Sedlackova, and et al. 2022. "Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula)" Microorganisms 10, no. 5: 1028. https://doi.org/10.3390/microorganisms10051028
APA StyleLinhart, P., Bandouchova, H., Zukal, J., Votýpka, J., Baláž, V., Heger, T., Kalocsanyiova, V., Kubickova, A., Nemcova, M., Sedlackova, J., Seidlova, V., Veitova, L., Vlaschenko, A., Divinova, R., & Pikula, J. (2022). Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula). Microorganisms, 10(5), 1028. https://doi.org/10.3390/microorganisms10051028






