Alum Addition Triggers Hypoxia in an Engineered Pit Lake
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Sampling Strategy
2.3. Geochemical Analyses
2.4. DNA Extraction and Quantification
2.5. Amplicon Sequencing
2.6. Statistical Analyses
3. Results and Discussion
3.1. Pre- and Post-Alum BML Water Cap Physico-Chemistry and Geochemistry
3.2. FFT Porewater Chemistry
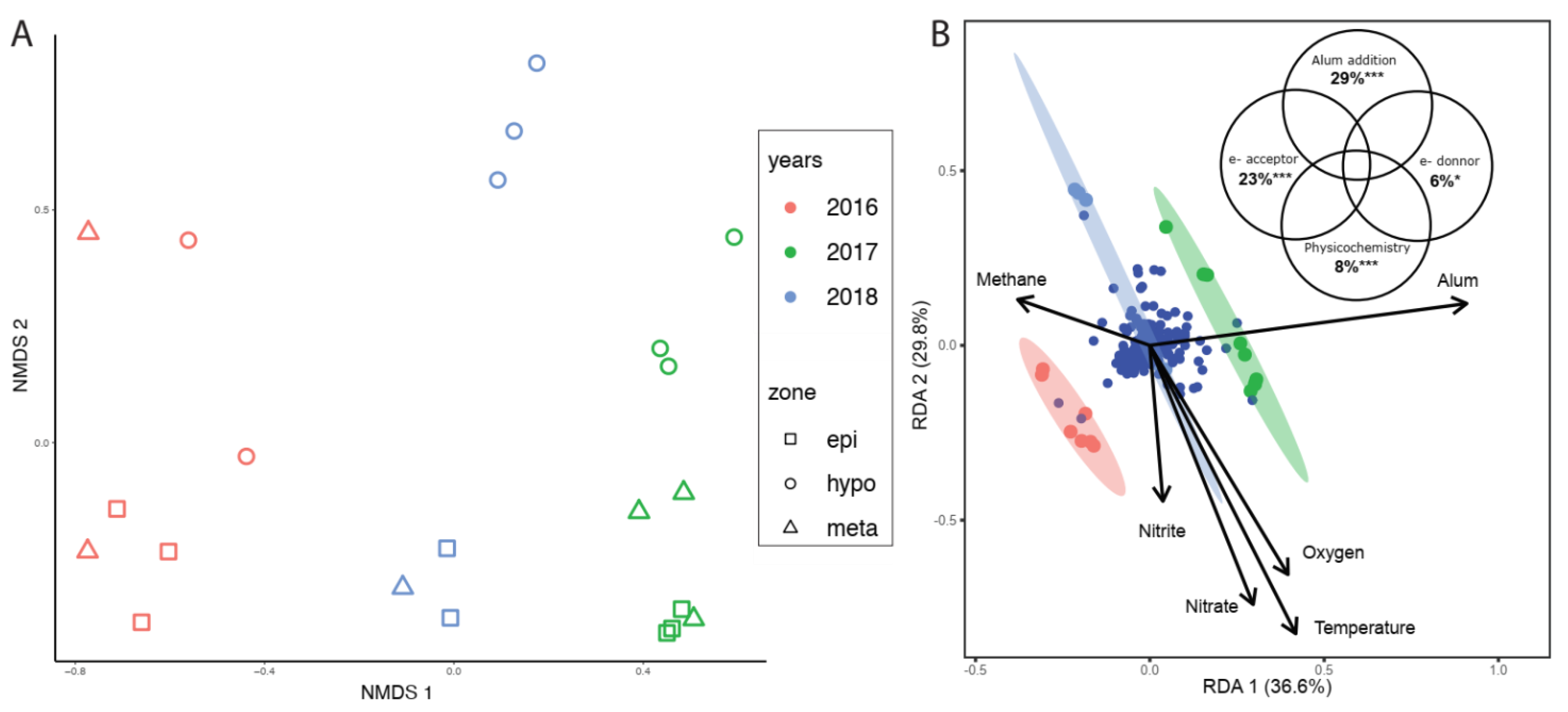
3.3. Shifts in Microbial Community Structure through Time
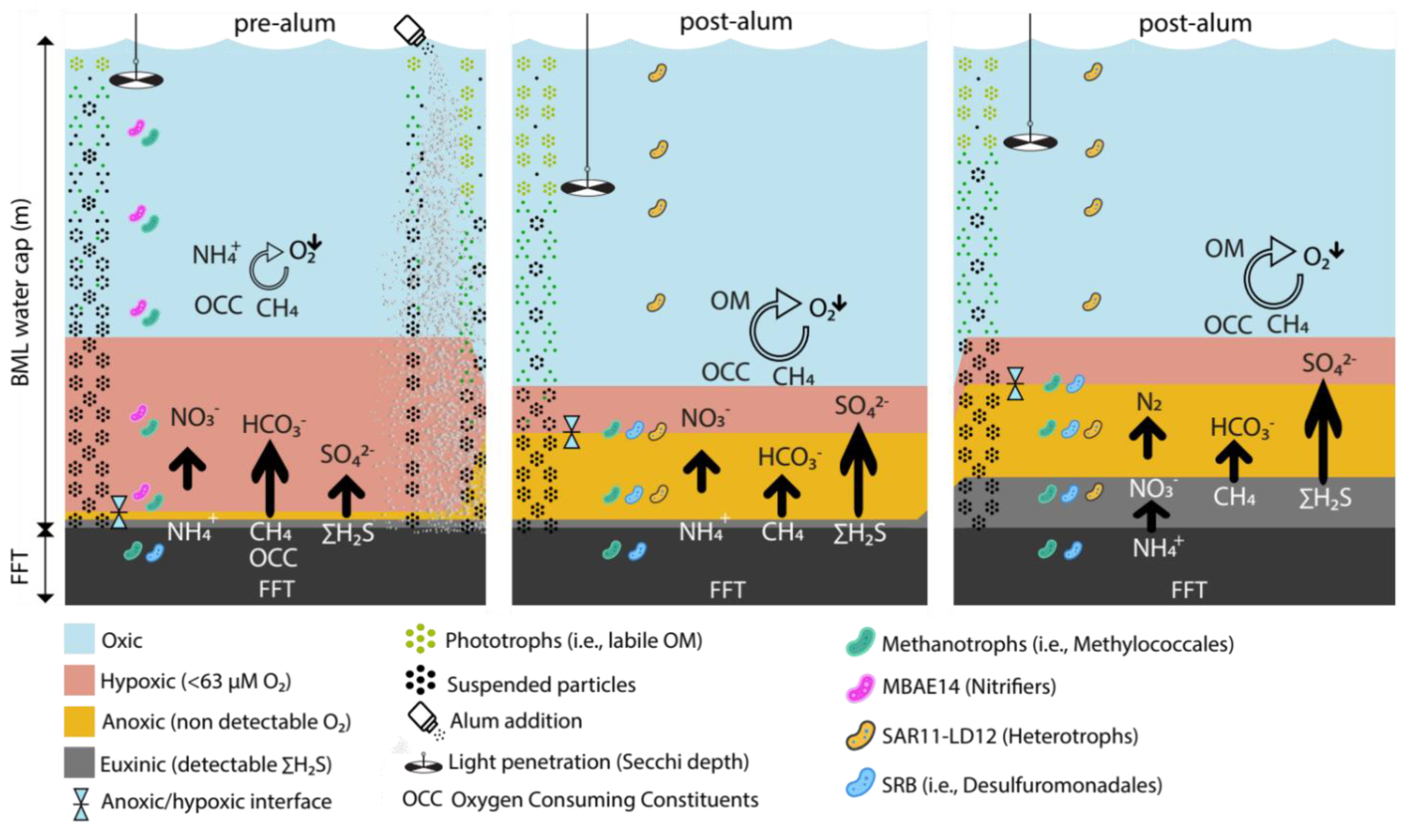
3.4. Improved Clarity and Biogeochemical Consequences in BML
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dompierre, K.A.; Lindsay, M.B.J.; Cruz-Hernández, P.; Halferdahl, G.M. Initial geochemical characteristics of fluid fine tailings in an oil sands end pit lake. Sci. Total Environ. 2016, 556, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Arriaga, D.; Nelson, T.C.; Risacher, F.F.; Morris, P.K.; Goad, C.; Slater, G.F.; Warren, L.A. The co-importance of physical mixing and biogeochemical consumption in controlling water cap oxygen levels in Base Mine Lake. Appl. Geochem. 2019, 111, 104442. [Google Scholar] [CrossRef]
- Foght, J.M.; Gieg, L.M.; Siddique, T. The microbiology of oil sands tailings: Past, present, future. FEMS Microbiol. Ecol. 2017, 93, fix034. [Google Scholar] [CrossRef]
- Kong, J.D.; Wang, H.; Siddique, T.; Foght, J.; Semple, K.; Burkus, Z.; Lewis, M.A. Second-generation stoichiometric mathematical model to predict methane emissions from oil sands tailings. Sci. Total Environ. 2019, 694, 133645. [Google Scholar] [CrossRef] [PubMed]
- Siddique, T.; Kuznetsova, A. Linking hydrocarbon biodegradation to greenhouse gas emissions from oil sands tailings and its impact on tailings management. Can. J. Soil Sci. 2020, 100, 537–545. [Google Scholar] [CrossRef]
- Alberta Energy Regulator. State of Fluid Tailings Management for Mineable Oil Sands, 2019; Alberta Energy Regulator: Calgary, AB, Canada, 2020.
- Alberta Energy Regulator. Directive 085: Fluid Tailings Management for Oil Sands Mining Projects; Alberta Energy Regulator: Calgary, AB, Canada, 2016.
- Simieritsch, T.; Obad, J.; Dyer, S. Tailings Performance Criteria and Requirements for Oil Sands Mining Schemes; Pembina Institute: Drayton Valley, AB, Canada, 2010. [Google Scholar]
- Clearwater Environmental Consultants. End Pit Lakes Technical Guidance Document; Clearwater Environmental Consultants: Fort McMurray, AB, Canada, 2013; Volume 53. [Google Scholar]
- Kabwe, L.K.; Scott, J.D.; Beier, N.A.; Wilson, G.W.; Jeeravipoolvarn, S. Environmental implications of end pit lakes at oil sand mines in Alberta, Canada. Environ. Geotech. 2019, 6, 67–74. [Google Scholar] [CrossRef]
- Canada’s Oil Sands Innovation Alliance. Pit Lakes: A Surface Mining Perspective: Tailings Environmental Priority Area (EPA); Canada’s Oil Sands Innovation Alliance: Calgary, AB, Canada, 2021. [Google Scholar]
- Risacher, F.F.; Morris, P.K.; Arriaga, D.; Goad, C.; Nelson, T.C.; Slater, G.F.; Warren, L.A. The interplay of methane and ammonia as key oxygen consuming constituents in early stage development of Base Mine Lake, the first demonstration oil sands pit lake. Appl. Geochem. 2018, 93, 49–59. [Google Scholar] [CrossRef]
- Fisseha, B.; Wilson, G.W.; Simms, P. Assessment of Self-Weight Consolidation of Flocculated Fluid Fine Tailings under Various Environmental Conditions. Proceedings of the Paste 2018: 21st International Seminar on Paste and Thickened Tailings, 2018; Australian Centre for Geomechanics: Crawley, Australia, 2018; pp. 291–304. [Google Scholar]
- Mori, J.F.; Chen, L.-X.; Jessen, G.L.; Rudderham, S.B.; McBeth, J.M.; Lindsay, M.B.J.; Slater, G.F.; Banfield, J.F.; Warren, L.A. Putative Mixotrophic Nitrifying-Denitrifying Gammaproteobacteria Implicated in Nitrogen Cycling Within the Ammonia/Oxygen Transition Zone of an Oil Sands Pit Lake. Front. Microbiol. 2019, 10, 2435. [Google Scholar] [CrossRef]
- Tedford, E.; Halferdahl, G.; Pieters, R.; Lawrence, G.A. Temporal variations in turbidity in an oil sands pit lake. Environ. Fluid Mech. 2019, 19, 457–473. [Google Scholar] [CrossRef]
- Clark, M.G.; Drewitt, G.B.; Carey, S.K. Energy and carbon fluxes from an oil sands pit lake. Sci. Total Environ. 2021, 752, 141966. [Google Scholar] [CrossRef]
- Syncrude. Base Mine Lake Monitoring and Research Summary Report: Results from 2013–2018; Syncrude: Fort McMurray, AB, Canada, 2019. [Google Scholar]
- Huser, B.J.; Egemose, S.; Harper, H.; Hupfer, M.; Jensen, H.; Pilgrim, K.M.; Reitzel, K.; Rydin, E.; Futter, M. Longevity and effectiveness of aluminum addition to reduce sediment phosphorus release and restore lake water quality. Water Res. 2016, 97, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Özkundakci, D.; Hamilton, D.; Scholes, P. Effect of Intensive Catchment and in—Lake Restoration Procedures on Phosphorus Concentrations in a Eutrophic Lake. Ecol. Eng. 2010, 36, 396–405. [Google Scholar] [CrossRef]
- Galvez-Cloutier, R.; Saminathan, S.K.M.; Boillot, C.; Triffaut-Bouchet, G.; Bourget, A.; Soumis-Dugas, G. An evaluation of several in-lake restoration techniques to improve the water quality problem (eutrophication) of Saint-Augustin Lake, Quebec, Canada. Environ. Manag. 2012, 49, 1037–1053. [Google Scholar] [CrossRef] [PubMed]
- Gelda, R.K.; Effler, S.W.; Peng, F. Modeling Turbidity and the Effects of Alum Application for a Water Supply Reservoir. J. Environ. Eng. 2012, 138, 38–47. [Google Scholar] [CrossRef]
- Diemert, S.; Andrews, R. The impact of alum coagulation on pharmaceutically active compounds, endocrine disrupting compounds and natural organic matter. Water Sci. Technol. Water Supply 2013, 13, 1348. [Google Scholar] [CrossRef]
- Nogaro, G.; Burgin, A.J.; Schoepfer, V.A.; Konkler, M.J.; Bowman, K.L.; Hammerschmidt, C.R. Aluminum sulfate (alum) application interactions with coupled metal and nutrient cycling in a hypereutrophic lake ecosystem. Environ. Pollut. 2013, 176, 267–274. [Google Scholar] [CrossRef]
- Huser, B.J.; Futter, M.; Lee, J.T.; Perniel, M. In-lake measures for phosphorus control: The most feasible and cost-effective solution for long-term management of water quality in urban lakes. Water Res. 2016, 97, 142–152. [Google Scholar] [CrossRef]
- Steinman, A.D.; Ogdahl, M.E. Macroinvertebrate response and internal phosphorus loading in a Michigan Lake after alum treatment. J. Environ. Qual. 2012, 41, 1540–1548. [Google Scholar] [CrossRef][Green Version]
- Rönicke, H.; Frassl, M.A.; Rinke, K.; Tittel, J.; Beyer, M.; Kormann, B.; Gohr, F.; Schultze, M. Suppression of bloom-forming colonial cyanobacteria by phosphate precipitation: A 30 years case study in Lake Barleber (Germany). Ecol. Eng. 2021, 162, 106171. [Google Scholar] [CrossRef]
- Wang, X.; Li, W.; Xiao, Y.; Cheng, A.; Shen, T.; Zhu, M.; Yu, L. Abundance and diversity of carbon-fixing bacterial communities in karst wetland soil ecosystems. CATENA 2021, 204, 105418. [Google Scholar] [CrossRef]
- Zhang, B.; Wei, Z.; Schaeffer, S.M.; Liang, A.; Ding, X. Recovery of bacterial communities and functions of soils under ridge tillage and no-tillage after different intensities and frequencies of drying-wetting disturbances in agroecosystems of northeastern China. CATENA 2021, 203, 105367. [Google Scholar] [CrossRef]
- Allison, S.D.; Martiny, J.B.H. Colloquium paper: Resistance, resilience, and redundancy in microbial communities. Proc. Natl. Acad. Sci. USA 2008, 105, 11512–11519. [Google Scholar] [CrossRef] [PubMed]
- Shade, A.; Peter, H.; Allison, S.; Baho, D.; Berga, M.; Buergmann, H.; Huber, D.; Langenheder, S.; Lennon, J.; Martiny, J.; et al. Fundamentals of Microbial Community Resistance and Resilience. Front. Microbiol. 2012, 3, 417. [Google Scholar] [CrossRef] [PubMed]
- Francis, D.J.; Barbour, S.L.; Lindsay, M.B.J. Ebullition enhances chemical mass transport across the tailings-water interface of oil sands pit lakes. J. Contam. Hydrol. 2022, 245, 103938. [Google Scholar] [CrossRef] [PubMed]
- Dompierre, K.A.; Barbour, S.L. Thermal properties of oil sands fluid fine tailings: Laboratory and in situ testing methods. Can. Geotech. J. 2017, 54, 428–440. [Google Scholar] [CrossRef]
- Whaley-Martin, K.J.; Chen, L.-X.; Nelson, T.C.; Gordon, J.; Kantor, R.; Twible, L.E.; Marshall, S.; Rossi, L.; Bessette, B.; Baron, C.; et al. Acidity and sulfur oxidation intermediate concentrations controlled by O2-driven partitioning of sulfur oxidizing bacteria in a mine tailings impoundment. bioRxiv 2021. [Google Scholar] [CrossRef]
- Bates, S.T.; Berg-Lyons, D.; Caporaso, J.G.; Walters, W.A.; Knight, R.; Fierer, N. Examining the global distribution of dominant archaeal populations in soil. ISME J. 2010, 5, 908. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Buttigieg, P.L.; Ramette, A. A guide to statistical analysis in microbial ecology: A community-focused, living review of multivariate data analyses. FEMS Microbiol. Ecol. 2014, 90, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Ramette, A. Multivariate analyses in microbial ecology. FEMS Microbiol. Ecol. 2007, 62, 142–160. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, G.; Kindt, R.; Legendre, P.; O’Hara, B. Vegan: Community Ecology Package, version 1.17-4; 2010. Available online: https://www.researchgate.net/publication/264237828_H2010_Vegan_Community_Ecology_Package_R_package_version_117-4 (accessed on 10 January 2022).
- Rudderham, S.B. Geomicrobiology and Geochemistry of Fluid Fine Tailings in an Oil Sands End Pit Lake; University of Saskatchewan: Saskatoon, SK, Canada, 2019. [Google Scholar]
- Lin, Q.; Fan, M.; Peng, X.; Ma, J.; Zhang, Y.; Yu, F.; Wu, Z.; Liu, B. Response of Vallisneria natans to aluminum phytotoxicity and their synergistic effect on nitrogen, phosphorus change in sediments. J. Hazard. Mater. 2020, 400, 123167. [Google Scholar] [CrossRef] [PubMed]
- Yeboah, D.; Kingston, S. Tree species richness decreases while species evenness increases with disturbance frequency in a natural boreal forest landscape. Ecol. Evol. 2016, 6, 842–850. [Google Scholar] [CrossRef]
- Klann, J.; McHenry, A.; Montelongo, C.; Goffredi, S. Decomposition of plant-sourced carbon compounds by heterotrophic betaproteobacteria isolated from a tropical Costa Rican bromeliad. Microbiologyopen 2016, 5, 479–489. [Google Scholar] [CrossRef]
- Chen, L.-X.; Zhao, Y.; McMahon, K.D.; Mori, J.; Jessen, G.L.; Nelson, T.C.; Warren, L.A.; Banfield, J.F. Wide Distribution of Phage That Infect Freshwater SAR11 Bacteria. mSystems 2021, 4, e00410–e00419. [Google Scholar] [CrossRef]
- Bernardet, J.; Bowman, J.P. The Genus Flavobacterium. In The Prokaryotes; Springer: Berlin/Heidelberg, Germany, 2006; ISBN 0387307478. [Google Scholar]
- Tamura, T.; Rainey, S. 41 The Family Sporichthyaceae. In The Prokaryotes; Springer: Berlin/Heidelberg, Germany, 2014; pp. 883–888. [Google Scholar] [CrossRef]
- Smith, G.J.; Angle, J.C.; Solden, L.M.; Borton, M.A.; Morin, T.H.; Daly, R.A.; Johnston, M.D.; Stefanik, K.C.; Wolfe, R.; Gil, B.; et al. Members of the Genus Methylobacter Are Inferred To Account for the Majority of Aerobic Methane Oxidation in Oxic Soils from a Freshwater Wetland. MBio 2018, 9, e00815–e00818. [Google Scholar] [CrossRef]
- Caccavo, F.; Lonergan, D.J.; Lovley, D.R.; Davis, M.; Stolz, J.F.; McInerney, M.J. Geobacter sulfurreducens sp. nov., a hydrogen- and acetate-oxidizing dissimilatory metal-reducing microorganism. Appl. Environ. Microbiol. 1994, 60, 3752–3759. [Google Scholar] [CrossRef]
- Engel, C.E.A.; Vorländer, D.; Biedendieck, R.; Krull, R.; Dohnt, K. Quantification of microaerobic growth of Geobacter sulfurreducens. PLoS ONE 2020, 15, e0215341. [Google Scholar] [CrossRef] [PubMed]
- Dyksma, S.; Lenk, S.; Sawicka, J.E.; Mußmann, M. Uncultured Gammaproteobacteria and Desulfobacteraceae Account for Major Acetate Assimilation in a Coastal Marine Sediment. Front. Microbiol. 2018, 9, 3124. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, M.; Kurisu, F.; Kasuga, I.; Furumai, H. Time-resolved DNA stable isotope probing links Desulfobacterales- and Coriobacteriaceae-related bacteria to anaerobic degradation of benzene under methanogenic conditions. Microbes Environ. 2014, 29, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Cabello-Yeves, P.J.; Ghai, R.; Mehrshad, M.; Picazo, A.; Camacho, A.; Rodriguez-Valera, F. Reconstruction of diverse verrucomicrobial genomes from metagenome datasets of freshwater reservoirs. Front. Microbiol. 2017, 8, 2131. [Google Scholar] [CrossRef]
- Slater, G.F.; Goad, C.A.; Lindsay, M.B.J.; Mumford, K.G.; Colenbrander Nelson, T.E.; Brady, A.L.; Jessen, G.L.; Warren, L.A. Isotopic and Chemical Assessment of the Dynamics of Methane Sources and Microbial Cycling during Early Development of an Oil Sands Pit Lake. Microorganisms 2021, 9, 2509. [Google Scholar] [CrossRef]



| Year | Depth [m] | Oxygen (µM) | Temp (C) | pH | EC (mScm−1) | TOC (mgL−1) | DOC (mgL−1) | Methane (µM) | Ammonia (µM) | Nitrite (µM) | Nitrate (µM) | Sulfate (µM) | Sulfide (µM) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | 0.00 | 214.2 | 20.8 | 8.2 | 2.82 | 99.9 | 87.7 | 0.6 | 9.9 | 1.5 | 38.8 | 2026.7 | 0.0 |
| 2016 | 0.50 | 200.8 | 20.7 | 8.3 | 2.82 | ||||||||
| 2016 | 1.00 | 197.7 | 20.5 | 8.3 | 2.82 | ||||||||
| 2016 | 1.50 | 196.9 | 20.4 | 8.3 | 2.82 | 58.8 | 57.5 | 0.5 | 8.6 | 2.0 | 36.0 | 2212.1 | 0.0 |
| 2016 | 2.00 | 194.7 | 20.3 | 8.3 | 2.82 | ||||||||
| 2016 | 2.50 | 194.0 | 20.3 | 8.3 | 2.82 | ||||||||
| 2016 | 3.00 | 192.5 | 20.2 | 8.3 | 2.82 | ||||||||
| 2016 | 3.50 | 187.7 | 20.0 | 8.3 | 2.82 | ||||||||
| 2016 | 4.00 | 179.0 | 19.8 | 8.3 | 2.82 | ||||||||
| 2016 | 4.25 | 167.9 | 19.5 | 8.3 | 2.82 | 6.7 | 1.9 | 34.0 | 2280.9 | 0.0 | |||
| 2016 | 4.50 | 156.7 | 19.3 | 8.2 | 2.82 | 58.4 | 58.3 | 0.4 | 10.4 | 2.3 | 39.4 | 1709.5 | 0.0 |
| 2016 | 4.75 | 144.0 | 19.1 | 8.2 | 2.82 | 8.1 | 1.4 | 39.4 | 2314.4 | 0.0 | |||
| 2016 | 5.00 | 131.3 | 18.9 | 8.2 | 2.82 | ||||||||
| 2016 | 5.50 | 120.9 | 18.6 | 8.2 | 2.82 | 0.7 | 6.0 | 1.1 | 39.0 | 2333.8 | 0.0 | ||
| 2016 | 6.00 | 68.6 | 17.5 | 8.1 | 2.84 | 60.7 | 60.9 | 0.6 | 6.2 | 6.2 | 47.2 | 2115.4 | 0.0 |
| 2016 | 6.25 | 46.5 | 17.0 | 8.1 | 2.85 | 7.5 | 1.6 | 31.4 | 1869.1 | 0.0 | |||
| 2016 | 6.50 | 24.3 | 16.6 | 8.0 | 2.87 | 0.2 | 15.9 | 10.3 | 45.7 | 1824.1 | 0.0 | ||
| 2016 | 7.00 | 13.9 | 15.6 | 8.0 | 2.88 | 61.0 | 59.0 | 11.1 | 22.1 | 5.0 | 43.4 | 2039.3 | 0.0 |
| 2016 | 7.50 | 11.4 | 15.0 | 7.9 | 2.89 | 9.5 | 16.8 | 5.8 | 34.2 | 1802.5 | 0.0 | ||
| 2016 | 8.00 | 10.5 | 14.5 | 7.9 | 2.90 | 94.5 | 87.5 | 39.0 | 27.9 | 4.7 | 12.8 | 2007.5 | 0.0 |
| 2016 | 8.50 | 10.5 | 14.3 | 7.9 | 2.91 | ||||||||
| 2016 | 9.00 | 9.1 | 14.1 | 7.9 | 2.89 | 126.3 | 49.7 | 0.8 | 15.5 | 2049.8 | 0.0 | ||
| 2016 | (FFT)−0.05 | 0.0 | 8.1 | 2.96 | 72.0 | >1200 | >600 | <10 | <20 | <100 | >10 | ||
| 2017 | 0.00 | 238.2 | 20.9 | 8.3 | 2.79 | 54.8 | 48.2 | 0.8 | 16.9 | 4.7 | 48.9 | 2627.4 | 0.0 |
| 2017 | 1.00 | 236.6 | 21.1 | 8.3 | 2.79 | ||||||||
| 2017 | 1.50 | 237.2 | 20.8 | 8.3 | 2.79 | 54.5 | 48.9 | 0.8 | 13.0 | 4.2 | 54.5 | 2685.6 | 0.0 |
| 2017 | 2.00 | 236.6 | 21.0 | 8.3 | 2.79 | ||||||||
| 2017 | 3.00 | 235.8 | 20.7 | 8.3 | 2.79 | ||||||||
| 2017 | 4.00 | 234.7 | 20.9 | 8.3 | 2.79 | ||||||||
| 2017 | 4.50 | 234.5 | 20.5 | 8.3 | 2.78 | 53.8 | 49.8 | 1.0 | 17.2 | 4.5 | 55.1 | 2601.0 | 0.0 |
| 2017 | 5.00 | 224.6 | 20.2 | 8.3 | 2.78 | ||||||||
| 2017 | 5.50 | 195.9 | 19.6 | 8.3 | 2.79 | 55.8 | 0.8 | 16.5 | 4.3 | 52.3 | 2690.9 | 0.0 | |
| 2017 | 5.75 | 213.5 | 19.7 | 8.3 | 2.79 | ||||||||
| 2017 | 6.00 | 135.4 | 18.4 | 8.3 | 2.80 | ||||||||
| 2017 | 6.50 | 93.7 | 17.4 | 8.2 | 2.80 | 52.8 | 48.1 | 0.6 | 25.4 | 3.5 | 35.8 | 2606.3 | 0.0 |
| 2017 | 7.00 | 59.1 | 16.8 | 8.2 | 2.80 | ||||||||
| 2017 | 7.25 | 48.1 | 16.3 | 8.1 | 2.80 | ||||||||
| 2017 | 7.50 | 16.5 | 15.8 | 8.1 | 2.80 | 54.3 | 49.5 | 0.7 | 31.3 | 2.6 | 22.5 | 2475.8 | 0.0 |
| 2017 | 7.75 | 11.9 | 15.6 | 8.1 | 1.80 | 38.4 | 38.6 | 1.3 | 33.2 | 2.7 | 21.3 | 2559.5 | 0.0 |
| 2017 | 8.00 | 1.0 | 15.1 | 8.1 | 2.80 | 50.3 | 48.9 | 3.1 | 34.0 | 2.3 | 14.7 | 2601.0 | 0.0 |
| 2017 | 8.50 | 0.7 | 14.9 | 8.0 | 2.80 | ||||||||
| 2017 | 9.00 | 0.1 | 14.5 | 8.0 | 2.80 | 52.1 | |||||||
| 2017 | 9.50 | 0.0 | 14.1 | 8.0 | 2.80 | ||||||||
| 2017 | 9.75 | 0.0 | 14.3 | 8.0 | 2.81 | 49.7 | 28.9 | 40.2 | 0.8 | 0.0 | 2579.8 | 0.0 | |
| 2017 | (FFT)−0.08 | 0.0 | 7.8 | 3.08 | 75.0 | >1200 | >600 | <10 | <20 | <100 | >10 | ||
| 2018 | 0.00 | 251.6 | 20.9 | 8.3 | 2.53 | 32.5 | 32.0 | 0.3 | 16.4 | 1.2 | 0.0 | 2056.0 | 0.0 |
| 2018 | 1.50 | 247.5 | 20.0 | 8.3 | 2.48 | 48.9 | 47.5 | 0.4 | 24.9 | 1.3 | 0.0 | 2064.8 | 0.0 |
| 2018 | 4.00 | 227.8 | 18.2 | 8.2 | 2.41 | ||||||||
| 2018 | 4.50 | 214.4 | 17.6 | 8.2 | 2.34 | 48.6 | 44.9 | 0.4 | 21.7 | 1.4 | 0.0 | 2045.4 | 0.0 |
| 2018 | 5.50 | 211.3 | 17.4 | 8.2 | 2.33 | ||||||||
| 2018 | 6.00 | 206.3 | 17.3 | 8.2 | 2.33 | ||||||||
| 2018 | 6.50 | 191.9 | 17.0 | 8.2 | 2.32 | ||||||||
| 2018 | 7.00 | 181.6 | 16.8 | 8.2 | 2.31 | ||||||||
| 2018 | 7.50 | 125.6 | 16.1 | 8.1 | 2.29 | 37.1 | 35.8 | 0.3 | 27.2 | 1.3 | 0.0 | 2096.6 | 0.0 |
| 2018 | 7.75 | 68.8 | 15.3 | 8.1 | 2.27 | ||||||||
| 2018 | 8.00 | 35.9 | 14.9 | 8.0 | 2.22 | ||||||||
| 2018 | 8.50 | 0.3 | 13.4 | 8.0 | 2.20 | ||||||||
| 2018 | 8.75 | 0.0 | 13.2 | 8.0 | 2.19 | 51.6 | 49.4 | 6.4 | 41.5 | 1.0 | 0.0 | 2096.6 | 0.0 |
| 2018 | 9.00 | 0.0 | 13.1 | 7.9 | 2.19 | ||||||||
| 2018 | 9.50 | 0.0 | 13.0 | 7.9 | 2.18 | 6.1 | |||||||
| 2018 | 9.75 | 0.0 | 12.9 | 7.9 | 2.18 | 52.2 | 43.0 | 13.6 | 34.6 | 0.9 | 0.0 | 2075.4 | 4.4 |
| 2018 | 10.00 | 0.0 | 12.8 | 7.9 | 2.17 | ||||||||
| 2018 | 10.50 | 0.0 | 12.4 | 7.9 | 2.16 | 52.2 | 48.5 | 26.9 | 28.6 | 0.9 | 0.0 | 2029.6 | 9.0 |
| TCF | TCU | TOC | DOC | TOC/TCU | DOC/TCF | |
|---|---|---|---|---|---|---|
| mg L−1 | mg L−1 | mg L−1 | mg L−1 | % | % | |
| (av) H2O cap | (av) H2O cap | (av) H2O cap | ||||
| 2016 | 214 | 219.8 | 72.2 | 68.5 | 32 | 32 |
| 2017 | 182.5 | 179.3 | 51.2 | 54.2 | 28 | 29 |
| 2018 | 172.5 | 176.1 | 45 | 43 | 25 | 26 |
| (av) hypolimnion | (av) hypolimnion | (av) hypolimnion | ||||
| 2016 | 224.9 | 231.4 | 77.8 | 73.2 | 34 | 34 |
| 2017 | 181.9 | 162.2 | 47.7 | 46.7 | 29 | 26 |
| 2018 | 180.6 | 185.7 | 46.3 | 44.2 | 25 | 26 |
| ∆ TCF (%) | ∆ TCU (%) | ∆ TOC (%) | ∆ DOC (%) | ∆ TOC/TCU (%) | ∆ DOC/TCF (%) | |
| ∆ (av) H2O cap decrease | ||||||
| 2016–2017 | 15 | 18 | 29 | 21 | 13 | 9 |
| 2017–2018 | 5 | 2 | 12 | 21 | 11 | 10 |
| 2016–2018 | 24 | 25 | 60 | 59 | 28 | 23 |
| ∆ (av) hypolimnion decrease | ||||||
| 2016–2017 | 19 | 30 | 39 | 36 | 15 | 24 |
| 2017–2018 | 1 | −14 | 3 | 5 | 14 | 0 |
| 2016–2018 | 5 | 25 | 68 | 66 | 36 | 31 |
| ∆ (av) epilimnetic (O2) increase | ||||||
| ∆ (O2) | ∆ (O2) | ∆ (O2) | ||||
| μM | mg L−1 | % | ||||
| 2016–2018 | 24 | 0.75 | 11 | |||
| 2016–2018 | 13 | 0.41 | 5 | |||
| 2016–2018 | 37 | 1.16 | 17 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jessen, G.L.; Chen, L.-X.; Mori, J.F.; Nelson, T.E.C.; Slater, G.F.; Lindsay, M.B.J.; Banfield, J.F.; Warren, L.A. Alum Addition Triggers Hypoxia in an Engineered Pit Lake. Microorganisms 2022, 10, 510. https://doi.org/10.3390/microorganisms10030510
Jessen GL, Chen L-X, Mori JF, Nelson TEC, Slater GF, Lindsay MBJ, Banfield JF, Warren LA. Alum Addition Triggers Hypoxia in an Engineered Pit Lake. Microorganisms. 2022; 10(3):510. https://doi.org/10.3390/microorganisms10030510
Chicago/Turabian StyleJessen, Gerdhard L., Lin-Xing Chen, Jiro F. Mori, Tara E. Colenbrander Nelson, Gregory F. Slater, Matthew B. J. Lindsay, Jillian F. Banfield, and Lesley A. Warren. 2022. "Alum Addition Triggers Hypoxia in an Engineered Pit Lake" Microorganisms 10, no. 3: 510. https://doi.org/10.3390/microorganisms10030510
APA StyleJessen, G. L., Chen, L.-X., Mori, J. F., Nelson, T. E. C., Slater, G. F., Lindsay, M. B. J., Banfield, J. F., & Warren, L. A. (2022). Alum Addition Triggers Hypoxia in an Engineered Pit Lake. Microorganisms, 10(3), 510. https://doi.org/10.3390/microorganisms10030510






