IL-37 and IL-36 Cytokine Profiles in Chronic Hepatitis Delta During Bulevirtide Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Serological Markers
2.3. IL-37, IL-36α, IL-36β, and IL-36γ Detection
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Longitudinal Analysis of Serum IL-37, IL-36α, IL-36β, and IL-36γ in HBV/HDV-Coinfected Patients Treated with Bulevirtide
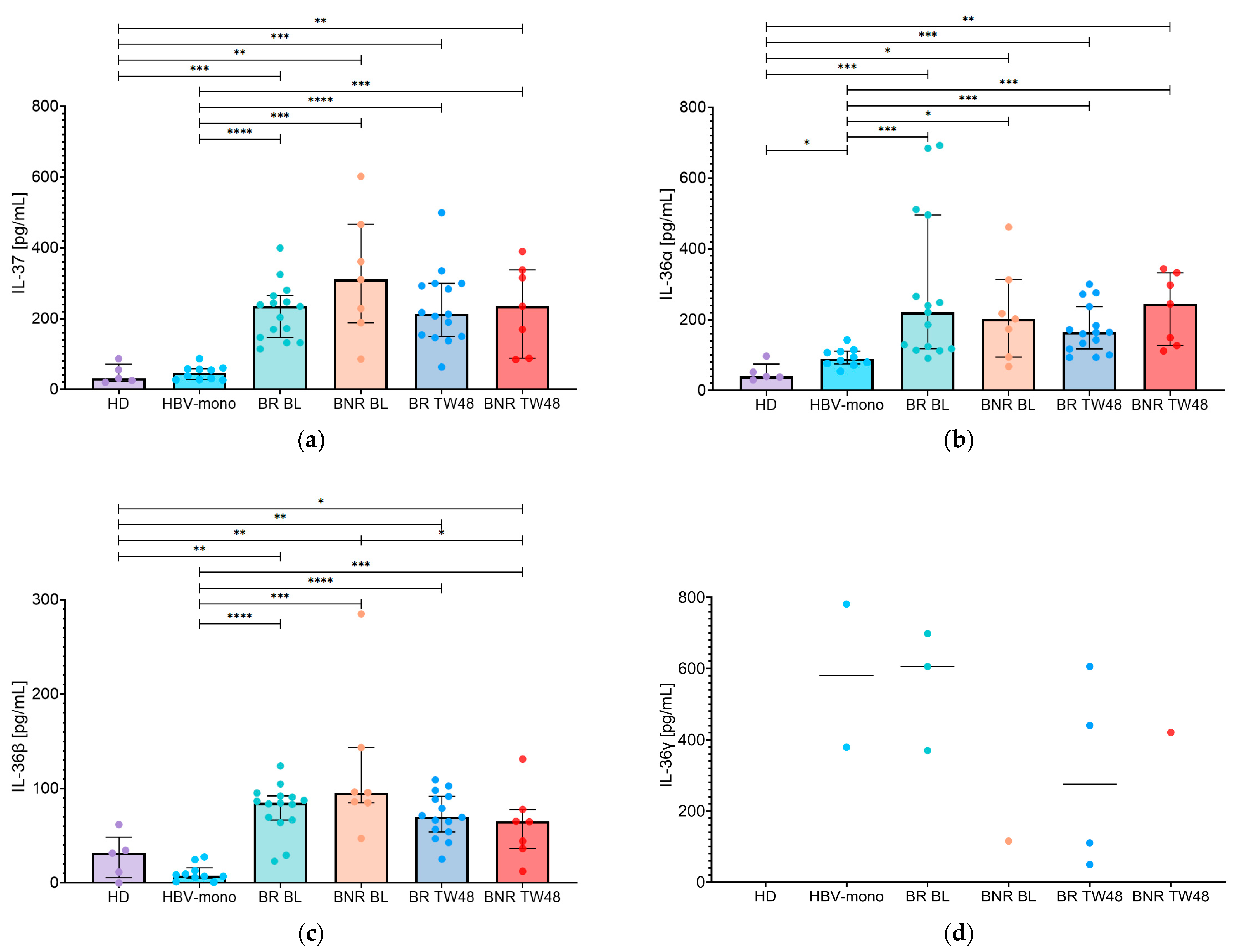
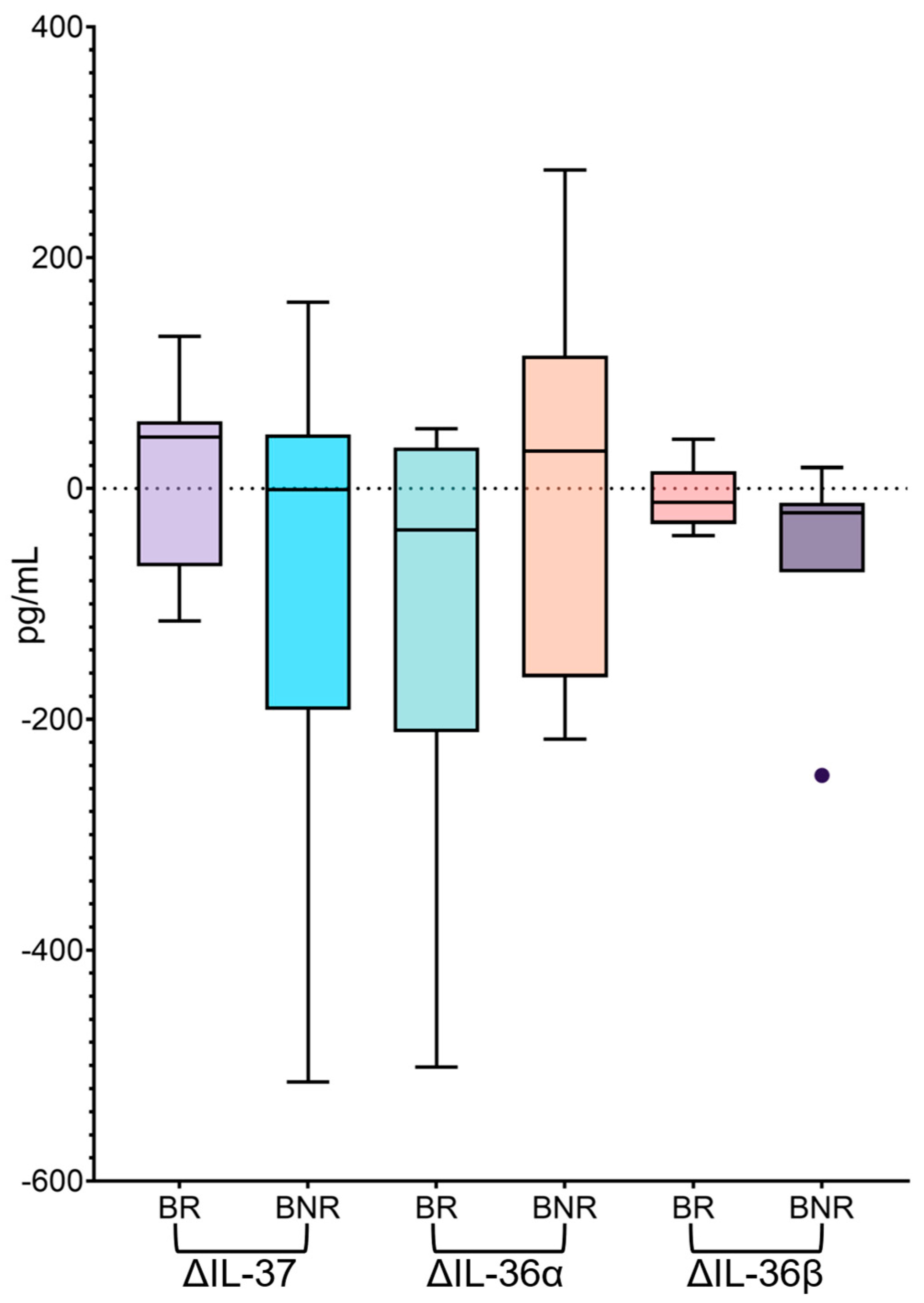
3.3. Changes in IL-37, IL-36α, IL-36β, and IL-36γ in Bulevirtide Treated HBV/HDV-Coinfected Patients According to Biochemical Response
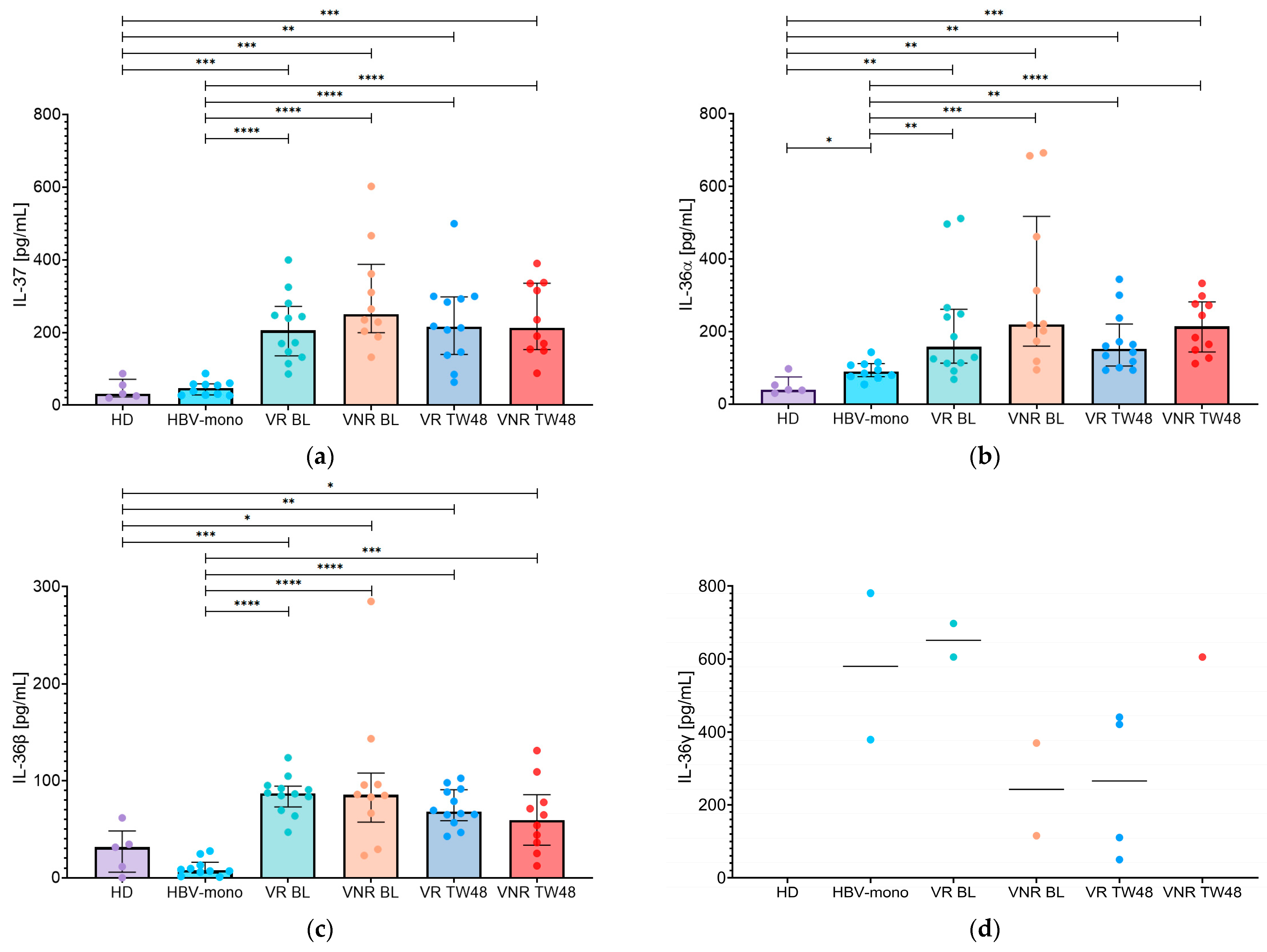
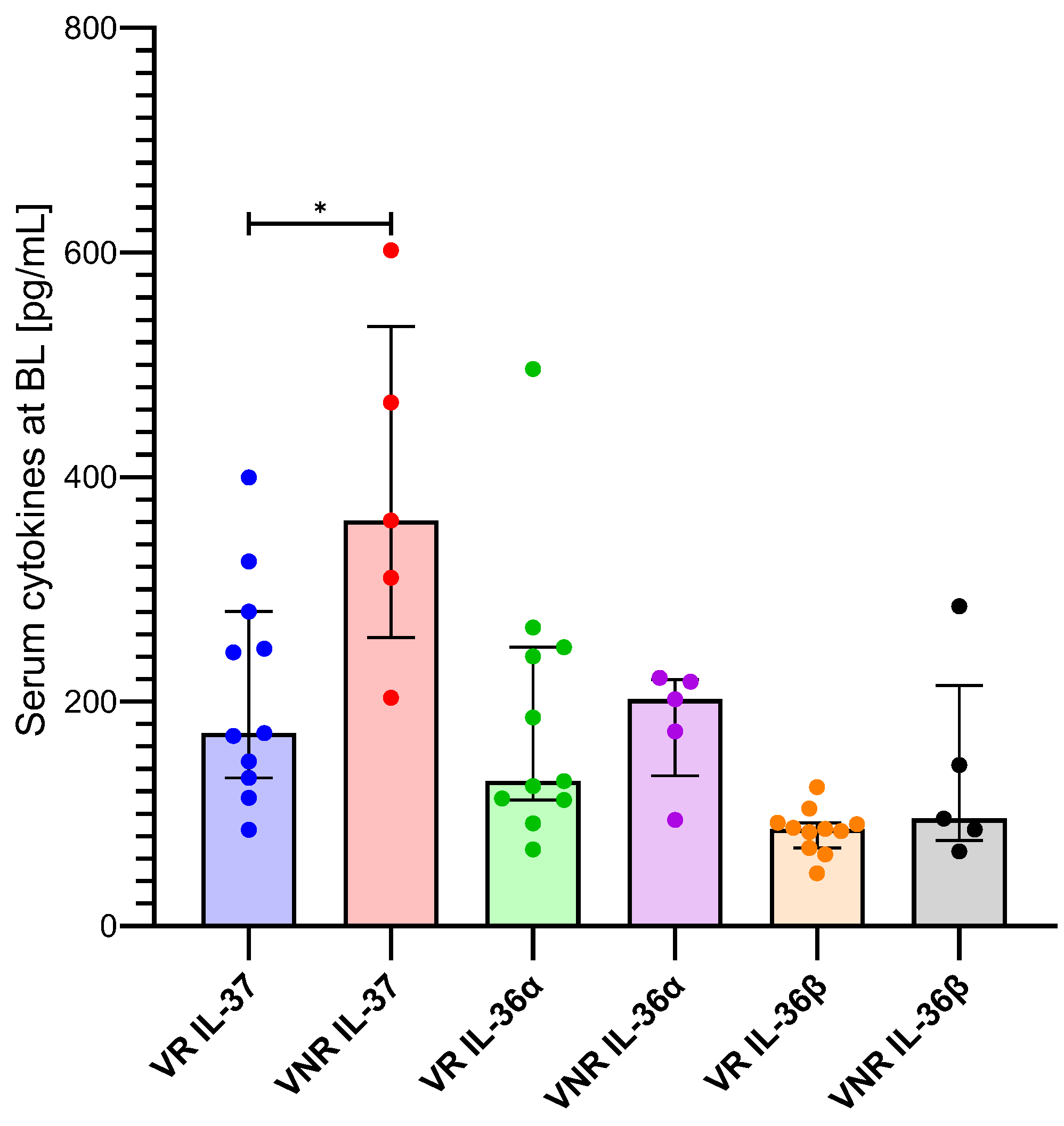
3.4. Changes in IL-37, IL-36α, IL-36β, and IL-36γ in Bulevirtide Treated HBV/HDV-Coinfected Patients According to Virological and Combined Response
3.5. Association Among Cytokine Levels and HDV RNA
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lampertico, P.; Degasperi, E.; Sandmann, L.; Wedemeyer, H. Delta Cure 2022 Working Group Hepatitis D Virus Infection: Pathophysiology, Epidemiology and Treatment. Report from the First International Delta Cure Meeting 2022. JHEP Rep. 2023, 5, 100818. [Google Scholar] [CrossRef]
- Farci, P.; Niro, G.A. Clinical Features of Hepatitis D. Semin. Liver Dis. 2012, 32, 228–236. [Google Scholar] [CrossRef]
- Stockdale, A.J.; Kreuels, B.; Henrion, M.Y.R.; Giorgi, E.; Kyomuhangi, I.; de Martel, C.; Hutin, Y.; Geretti, A.M. The Global Prevalence of Hepatitis D Virus Infection: Systematic Review and Meta-Analysis. J. Hepatol. 2020, 73, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Buti, M.; Stepanova, M.; Palom, A.; Riveiro-Barciela, M.; Nader, F.; Roade, L.; Esteban, R.; Younossi, Z. Chronic Hepatitis D Associated with Worse Patient-Reported Outcomes than Chronic Hepatitis B. JHEP Rep. 2021, 3, 100280. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-Y.; Pang, X.-Y.; Goyal, H.; Yang, R.-X.; Xu, H.-G. Hepatitis D: Challenges in the Estimation of True Prevalence and Laboratory Diagnosis. Gut Pathog. 2021, 13, 66. [Google Scholar] [CrossRef]
- Farci, P.; Niro, G.A.; Zamboni, F.; Diaz, G. Hepatitis D Virus and Hepatocellular Carcinoma. Viruses 2021, 13, 830. [Google Scholar] [CrossRef]
- Choi, S.-H.; Jeong, S.-H.; Hwang, S.B. Large Hepatitis Delta Antigen Modulates Transforming Growth Factor-Beta Signaling Cascades: Implication of Hepatitis Delta Virus-Induced Liver Fibrosis. Gastroenterology 2007, 132, 343–357. [Google Scholar] [CrossRef]
- Park, C.-Y.; Oh, S.-H.; Kang, S.M.; Lim, Y.-S.; Hwang, S.B. Hepatitis Delta Virus Large Antigen Sensitizes to TNF-Alpha-Induced NF-kappaB Signaling. Mol. Cells 2009, 28, 49–55. [Google Scholar] [CrossRef]
- Giersch, K.; Allweiss, L.; Volz, T.; Helbig, M.; Bierwolf, J.; Lohse, A.W.; Pollok, J.M.; Petersen, J.; Dandri, M.; Lütgehetmann, M. Hepatitis Delta Co-Infection in Humanized Mice Leads to Pronounced Induction of Innate Immune Responses in Comparison to HBV Mono-Infection. J. Hepatol. 2015, 63, 346–353. [Google Scholar] [CrossRef]
- Gu, Y.; Mohammad, I.S.; Liu, Z. Overview of the STAT-3 Signaling Pathway in Cancer and the Development of Specific Inhibitors. Oncol. Lett. 2020, 19, 2585–2594. [Google Scholar] [CrossRef]
- Asselah, T.; Razavi, H.; Fontaine, H.; Agarwal, K. Bulevirtide for Chronic Hepatitis Delta: From Clinical Trials to Real Life Data: An Expert Opinion Report. Antiviral Res. 2026, 245, 106311. [Google Scholar] [CrossRef]
- Yurdaydin, C.; Abbas, Z.; Buti, M.; Cornberg, M.; Esteban, R.; Etzion, O.; Gane, E.J.; Gish, R.G.; Glenn, J.S.; Hamid, S.; et al. Treating Chronic Hepatitis Delta: The Need for Surrogate Markers of Treatment Efficacy. J. Hepatol. 2019, 70, 1008–1015. [Google Scholar] [CrossRef]
- Wranke, A.; Hardtke, S.; Heidrich, B.; Dalekos, G.; Yalçin, K.; Tabak, F.; Gürel, S.; Çakaloğlu, Y.; Akarca, U.S.; Lammert, F.; et al. Ten-Year Follow-up of a Randomized Controlled Clinical Trial in Chronic Hepatitis Delta. J. Viral Hepat. 2020, 27, 1359–1368. [Google Scholar] [CrossRef] [PubMed]
- Bremer, B.; Anastasiou, O.E.; Hardtke, S.; Caruntu, F.A.; Curescu, M.G.; Yalcin, K.; Akarca, U.S.; Gürel, S.; Zeuzem, S.; Erhardt, A.; et al. Residual Low HDV Viraemia Is Associated HDV RNA Relapse after PEG-IFNa-Based Antiviral Treatment of Hepatitis Delta: Results from the HIDIT-II Study. Liver Int. 2021, 41, 295–299. [Google Scholar] [CrossRef]
- Watashi, K.; Shionoya, K.; Kobayashi, C.; Morita, T. Hepatitis B and D Virus Entry. Nat. Rev. Microbiol. 2025, 23, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Degasperi, E.; Anolli, M.P.; Jachs, M.; Reiberger, T.; De Ledinghen, V.; Metivier, S.; D’Offizi, G.; Di Maria, F.; Schramm, C.; Schmidt, H.; et al. Real-World Effectiveness and Safety of Bulevirtide Monotherapy for up to 96 Weeks in Patients with HDV-Related Cirrhosis. J. Hepatol. 2025, 82, 1012–1022. [Google Scholar] [CrossRef]
- Lampertico, P.; Anolli, M.P.; Steppich, K.; Wedemeyer, H. Bulevirtide Monotherapy or in Combination for Chronic Hepatitis Delta: 2025 Update. J. Viral Hepat. 2025, 32, e70056. [Google Scholar] [CrossRef]
- Degasperi, E.; De Silvestri, A.; Anolli, M.P.; Sambarino, D.; Borghi, M.; Perbellini, R.; Facchetti, F.; Soffredini, R.; Monico, S.; De Lédinghen, V.; et al. OS-120 Bulevirtide Monotherapy Prevents Liver Decompensation and Reduces Mortality in Patients with HDV-Related Cirrhosis: A Case Control Study with Propensity Score Weighted Analysis. J. Hepatol. 2024, 80, S73–S74. [Google Scholar] [CrossRef]
- Jachs, M.; Panzer, M.; Hartl, L.; Schwarz, M.; Balcar, L.; Camp, J.V.; Munda, P.; Mandorfer, M.; Trauner, M.; Aberle, S.W.; et al. Long-Term Follow-up of Patients Discontinuing Bulevirtide Treatment upon Long-Term HDV-RNA Suppression. JHEP Rep. 2023, 5, 100751. [Google Scholar] [CrossRef]
- Jiang, B.; Zhou, Y.; Liu, Y.; He, S.; Liao, B.; Peng, T.; Yao, L.; Qi, L. Research Progress on the Role and Mechanism of IL-37 in Liver Diseases. Semin. Liver Dis. 2023, 43, 336–350. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wang, B.; Ma, Z.; Sun, X.; Tang, Y.; Li, X.; Wu, X. Detection of the Novel IL-1 Family Cytokines by QAH-IL1F-1 Assay in Rheumatoid Arthritis. Cell. Mol. Biol. 2016, 62, 31–34. [Google Scholar]
- Li, P.; Guo, H.; Wu, K.; Su, L.; Huang, K.; Lai, R.; Deng, Z.; Li, S.; Ouyang, P.; Wang, Y.; et al. Decreased IL-37 Expression in Hepatocellular Carcinoma Tissues and Liver Cancer Cell Lines. Oncol. Lett. 2020, 19, 2639–2648. [Google Scholar] [CrossRef]
- Buhl, A.-L.; Wenzel, J. Interleukin-36 in Infectious and Inflammatory Skin Diseases. Front. Immunol. 2019, 10, 1162. [Google Scholar] [CrossRef] [PubMed]
- Ngo, V.L.; Kuczma, M.; Maxim, E.; Denning, T.L. IL-36 Cytokines and Gut Immunity. Immunology 2021, 163, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Madonna, S.; Girolomoni, G.; Dinarello, C.A.; Albanesi, C. The Significance of IL-36 Hyperactivation and IL-36R Targeting in Psoriasis. Int. J. Mol. Sci. 2019, 20, 3318. [Google Scholar] [CrossRef] [PubMed]
- Bridgewood, C.; Fearnley, G.W.; Berekmeri, A.; Laws, P.; Macleod, T.; Ponnambalam, S.; Stacey, M.; Graham, A.; Wittmann, M. IL-36γ Is a Strong Inducer of IL-23 in Psoriatic Cells and Activates Angiogenesis. Front. Immunol. 2018, 9, 200. [Google Scholar] [CrossRef]
- Sachen, K.L.; Arnold Greving, C.N.; Towne, J.E. Role of IL-36 Cytokines in Psoriasis and Other Inflammatory Skin Conditions. Cytokine 2022, 156, 155897. [Google Scholar] [CrossRef]
- Sugiura, K.; Fujita, H.; Komine, M.; Yamanaka, K.; Akiyama, M. The Role of Interleukin-36 in Health and Disease States. Acad. Dermatol. Venereol. 2024, 38, 1910–1925. [Google Scholar] [CrossRef]
- Obeagu, E.; Obeagu, G.; Okwuanaso, C. Optimizing Immune Health in HIV Patients through Nutrition: A Review. Elite J. Immunol. 2024, 2, 14–33. [Google Scholar]
- Qi, F.; Yan, Y.; Liu, M.; Lv, Q.; Xu, Y.; Liu, M.; Li, F.; Deng, R.; Liang, X.; Li, S.; et al. IL-37 Mitigates the Inflammatory Response in Macrophages Induced by SARS-CoV-2 Omicron Infection Through the NF-κB Signaling Pathway. MedComm 2025, 6, e70229. [Google Scholar] [CrossRef]
- Wein, A.N.; Dunbar, P.R.; McMaster, S.R.; Li, Z.-R.T.; Denning, T.L.; Kohlmeier, J.E. IL-36γ Protects against Severe Influenza Infection by Promoting Lung Alveolar Macrophage Survival and Limiting Viral Replication. J. Immunol. 2018, 201, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Gamero, A.M.; Jensen, L.E. IL-36 Promotes Anti-Viral Immunity by Boosting Sensitivity to IFN-α/β in IRF1 Dependent and Independent Manners. Nat. Commun. 2019, 10, 4700. [Google Scholar] [CrossRef]
- Liu, Q.; Zhou, Q.; Wang, M.; Pang, B. Interleukin-37 Suppresses the Cytotoxicity of Hepatitis B Virus Peptides-Induced CD8+ T Cells in Patients with Acute Hepatitis B. Biomol. Biomed. 2023, 23, 527–534. [Google Scholar] [CrossRef]
- Pu, X.-Y.; Zheng, D.-F.; Shen, A.; Gu, H.-T.; Wei, X.-F.; Mou, T.; Zhang, J.-B.; Liu, R. IL-37b Suppresses Epithelial Mesenchymal Transition in Hepatocellular Carcinoma by Inhibiting IL-6/STAT3 Signaling. Hepatobiliary Pancreat. Dis. Int. 2018, 17, 408–415. [Google Scholar] [CrossRef]
- Zhao, J.-J.; Pan, Q.-Z.; Pan, K.; Weng, D.-S.; Wang, Q.-J.; Li, J.-J.; Lv, L.; Wang, D.-D.; Zheng, H.-X.; Jiang, S.-S.; et al. Interleukin-37 Mediates the Antitumor Activity in Hepatocellular Carcinoma: Role for CD57+ NK Cells. Sci. Rep. 2014, 4, 5177. [Google Scholar] [CrossRef]
- Zulian, V.; Salichos, L.; Taibi, C.; Pauciullo, S.; Dong, L.; D’Offizi, G.; Biliotti, E.; Rianda, A.; Federici, L.; Bibbò, A.; et al. Exploring Predictive Factors for Bulevirtide Treatment Response in Hepatitis Delta-Positive Patients. Biomedicines 2025, 13, 280. [Google Scholar] [CrossRef] [PubMed]
- Fattovich, G.; Giustina, G.; Christensen, E.; Pantalena, M.; Zagni, I.; Realdi, G.; Schalm, S.W. Influence of Hepatitis Delta Virus Infection on Morbidity and Mortality in Compensated Cirrhosis Type B. The European Concerted Action on Viral Hepatitis (Eurohep). Gut 2000, 46, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Roulot, D.; Brichler, S.; Layese, R.; BenAbdesselam, Z.; Zoulim, F.; Thibault, V.; Scholtes, C.; Roche, B.; Castelnau, C.; Poynard, T.; et al. Origin, HDV Genotype and Persistent Viremia Determine Outcome and Treatment Response in Patients with Chronic Hepatitis Delta. J. Hepatol. 2020, 73, 1046–1062. [Google Scholar] [CrossRef]
- Kamal, H.; Westman, G.; Falconer, K.; Duberg, A.-S.; Weiland, O.; Haverinen, S.; Wejstål, R.; Carlsson, T.; Kampmann, C.; Larsson, S.B.; et al. Long-Term Study of Hepatitis Delta Virus Infection at Secondary Care Centers: The Impact of Viremia on Liver-Related Outcomes. Hepatology 2020, 72, 1177–1190. [Google Scholar] [CrossRef]
- Heller, T.; Rotman, Y.; Koh, C.; Clark, S.; Haynes-Williams, V.; Chang, R.; McBurney, R.; Schmid, P.; Albrecht, J.; Kleiner, D.E.; et al. Long-Term Therapy of Chronic Delta Hepatitis with Peginterferon Alfa. Aliment. Pharmacol. Ther. 2014, 40, 93–104. [Google Scholar] [CrossRef]
- Wedemeyer, H.; Aleman, S.; Blank, A.; Andreone, P.; Bogomolov, P.; Chulanov, V.; Mamonova, N.; Geyvandova, N.; Morozov, V.; Sagalova, O.; et al. LBO-004 Final Results of MYR301: A Randomised Phase 3 Study Evaluating the Efficacy and Safety of up to 144 Weeks of Bulevirtide Monotherapy for Chronic Hepatitis Delta and 96 Weeks of Posttreatment Follow-Up. J. Hepatol. 2025, 82, S10–S11. [Google Scholar] [CrossRef]
- Ding, S.; Ma, J.; Hu, Y.; Hu, A.; Shen, Q.; Gao, G. Outcomes of Interferon/Ribavirin Therapy in Patients with HCV Defined by Expression of Plasma Soluble Human Leukocyte Antigen-G but Not IL-37. Med. Sci. Monit. 2016, 22, 1398–1402. [Google Scholar] [CrossRef]
- Salpini, R.; Piermatteo, L.; Caviglia, G.P.; Bertoli, A.; Brunetto, M.R.; Bruzzone, B.; Callegaro, A.; Caudai, C.; Cavallone, D.; Chessa, L.; et al. Comparison of Diagnostic Performances of HDV-RNA Quantification Assays Used in Clinical Practice: Results from a National Quality Control Multicenter Study. J. Clin. Virol. 2025, 180, 105850. [Google Scholar] [CrossRef]
- Zulian, V.; Taibi, C.; Coppola, A.; Bibbò, A.; Federici, L.; De Sanctis, M.; Pauciullo, S.; D’Offizi, G.; Biliotti, E.; McPhee, F.; et al. Improving Virological Monitoring of HDV Infection: A Proof-of-Concept Comparative Study of Bosphore and AltoStar® Assays in Patients Treated with Bulevirtide. Biomedicines 2025, 13, 1564. [Google Scholar] [CrossRef]
- Anzaghe, M.; Niles, M.A.; Korotkova, E.; Dominguez, M.; Kronhart, S.; Ortega Iannazzo, S.; Bechmann, I.; Bachmann, M.; Mühl, H.; Kochs, G.; et al. Interleukin-36γ Is Causative for Liver Damage upon Infection with Rift Valley Fever Virus in Type I Interferon Receptor-Deficient Mice. Front. Immunol. 2023, 14, 1194733. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, L.; Du, Y.; Zhu, P.; Huang, G.; Luo, J.; Yan, X.; Ye, B.; Li, C.; Xia, P.; et al. The Long Noncoding RNA lncTCF7 Promotes Self-Renewal of Human Liver Cancer Stem Cells through Activation of Wnt Signaling. Cell Stem Cell 2015, 16, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Dong, X.; Chen, Y.; Wang, X. Serum Exosomal hnRNPH1 mRNA as a Novel Marker for Hepatocellular Carcinoma. Clin. Chem. Lab. Med. CCLM 2018, 56, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Tong, Y.; Fang, H.; Tang, J.; Liu, L.; Hu, Y.; Li, J.; Zhong, L. IL36 Indicating Good Prognosis in Human Hepatocellular Carcinoma. J. Cancer 2020, 11, 6248–6255. [Google Scholar] [CrossRef]
- Elias, M.; Zhao, S.; Le, H.T.; Wang, J.; Neurath, M.F.; Neufert, C.; Fiocchi, C.; Rieder, F. IL-36 in Chronic Inflammation and Fibrosis—Bridging the Gap? J. Clin. Investig. 2021, 131, e144336. [Google Scholar] [CrossRef]
- Feng, X.-X.; Chi, G.; Wang, H.; Gao, Y.; Chen, Q.; Ru, Y.-X.; Luo, Z.-L.; Yan, W.; Li, P.-Y.; Liu, M.; et al. IL-37 Suppresses the Sustained Hepatic IFN-γ/TNF-α Production and T Cell-Dependent Liver Injury. Int. Immunopharmacol. 2019, 69, 184–193. [Google Scholar] [CrossRef]
- Schümann, J.; Wolf, D.; Pahl, A.; Brune, K.; Papadopoulos, T.; van Rooijen, N.; Tiegs, G. Importance of Kupffer Cells for T-Cell-Dependent Liver Injury in Mice. Am. J. Pathol. 2000, 157, 1671–1683. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Sig. Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Shen, S.; Verma, I.M. NF-κB, an Active Player in Human Cancers. Cancer Immunol. Res. 2014, 2, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Siveen, K.S.; Sikka, S.; Surana, R.; Dai, X.; Zhang, J.; Kumar, A.P.; Tan, B.K.H.; Sethi, G.; Bishayee, A. Targeting the STAT3 Signaling Pathway in Cancer: Role of Synthetic and Natural Inhibitors. Biochim. Biophys. Acta 2014, 1845, 136–154. [Google Scholar] [CrossRef]


| Parameter | N = 22 |
|---|---|
| Age, years | 50 (35–68) |
| Male sex | 11 (50.0) |
| Body mass index | 24.5 (23.0–29.7) |
| Cirrhosis | 13 (59.1) |
| Previous interferon therapy | 16 (72.7) |
| Concomitant NUC therapy | 22 (100.0) |
| ALT, U/L | 87.5 (55.3–103.8) |
| AST, U/L | 72 (52.3–88.5) |
| Albumin, g/dL | 4.2 (4.0–4.4) |
| Bile acids, µmol/L | 11.7 (6.4–18.9) |
| Total bilirubin, mg/dL | 0.7 (0.7–0.9) |
| Platelet count, ×103/µL | 113.5 (90.1–190.0) |
| AFP, ng/mL | 4.9 (3.5–6.7) |
| HDV RNA, Log cp/mL | 4.9 (3.9–5.9) |
| HBV DNA undetectable 1 | 21 (95.5) |
| HBsAg, Log IU/mL | 3.6 (3.3–4.1) |
| HBcrAg, Log U/mL | 3.4 (2.6–4.1) |
| Anti-HBc IgG, COI | 58.5 (40.8–264.2) |
| HBeAg, positive | 1 (4.5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zulian, V.; De Sanctis, M.; Pauciullo, S.; Sciamanna, R.; Cimini, E.; Del Porto, P.; Garbuglia, A.R. IL-37 and IL-36 Cytokine Profiles in Chronic Hepatitis Delta During Bulevirtide Therapy. Pathogens 2026, 15, 198. https://doi.org/10.3390/pathogens15020198
Zulian V, De Sanctis M, Pauciullo S, Sciamanna R, Cimini E, Del Porto P, Garbuglia AR. IL-37 and IL-36 Cytokine Profiles in Chronic Hepatitis Delta During Bulevirtide Therapy. Pathogens. 2026; 15(2):198. https://doi.org/10.3390/pathogens15020198
Chicago/Turabian StyleZulian, Verdiana, Martina De Sanctis, Silvia Pauciullo, Roberta Sciamanna, Eleonora Cimini, Paola Del Porto, and Anna Rosa Garbuglia. 2026. "IL-37 and IL-36 Cytokine Profiles in Chronic Hepatitis Delta During Bulevirtide Therapy" Pathogens 15, no. 2: 198. https://doi.org/10.3390/pathogens15020198
APA StyleZulian, V., De Sanctis, M., Pauciullo, S., Sciamanna, R., Cimini, E., Del Porto, P., & Garbuglia, A. R. (2026). IL-37 and IL-36 Cytokine Profiles in Chronic Hepatitis Delta During Bulevirtide Therapy. Pathogens, 15(2), 198. https://doi.org/10.3390/pathogens15020198






