Efficacy of the Vaccine Candidate Based on the P0 Peptide against Dermacentor nitens and Ixodes ricinus Ticks
Abstract
:1. Introduction
2. Results
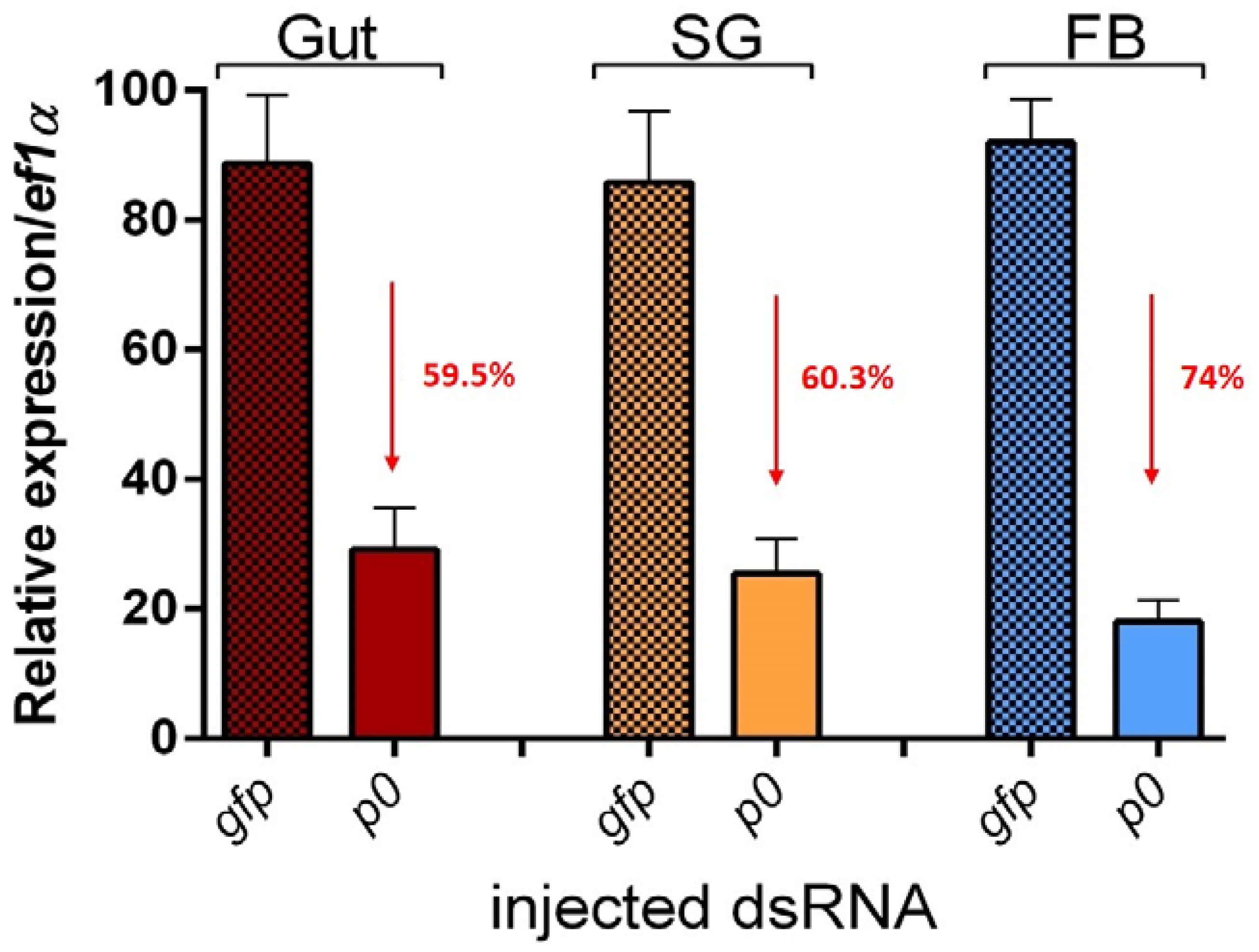
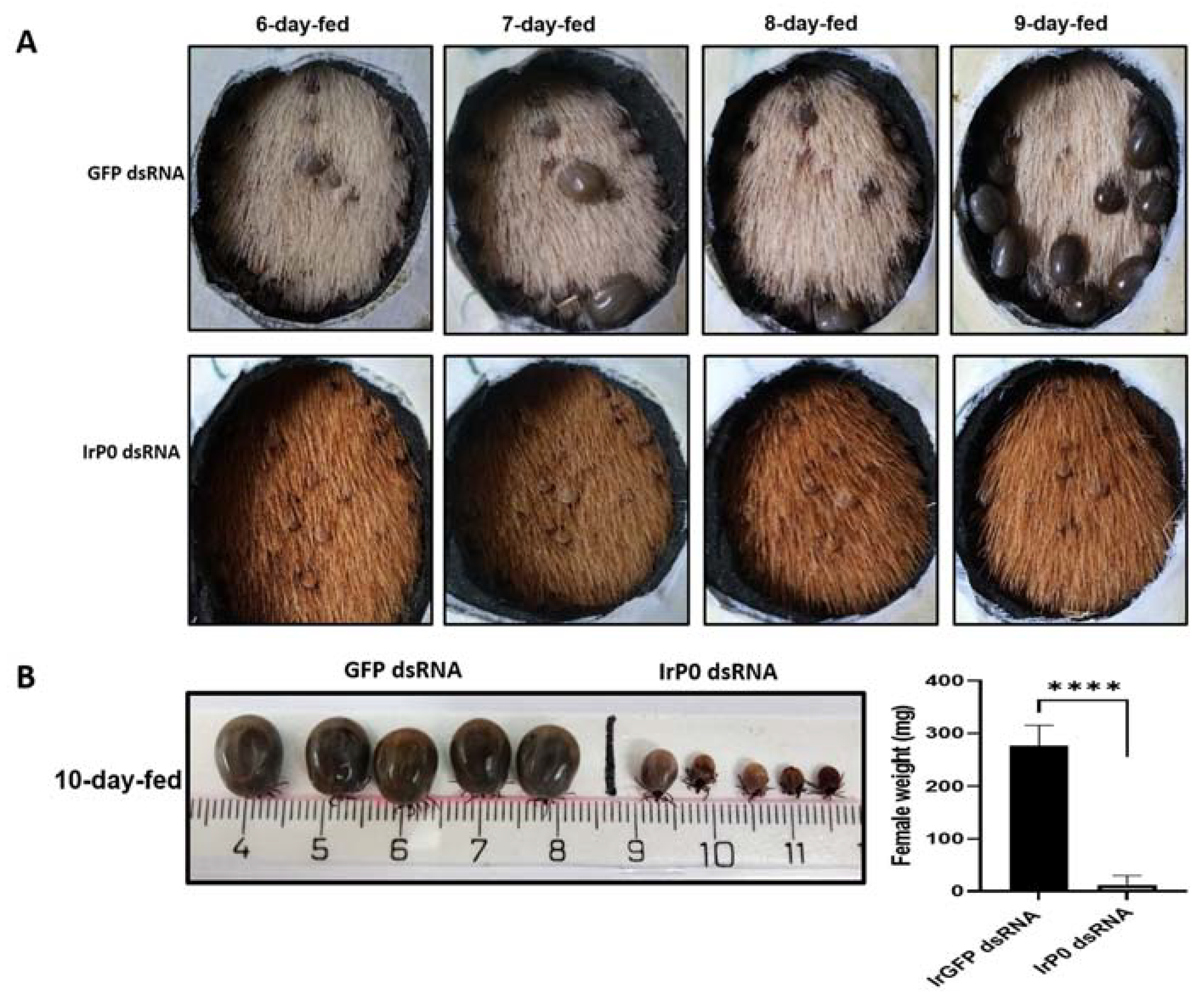
2.1. P0 Knockdown Impairs Feeding of I. ricinus Ticks
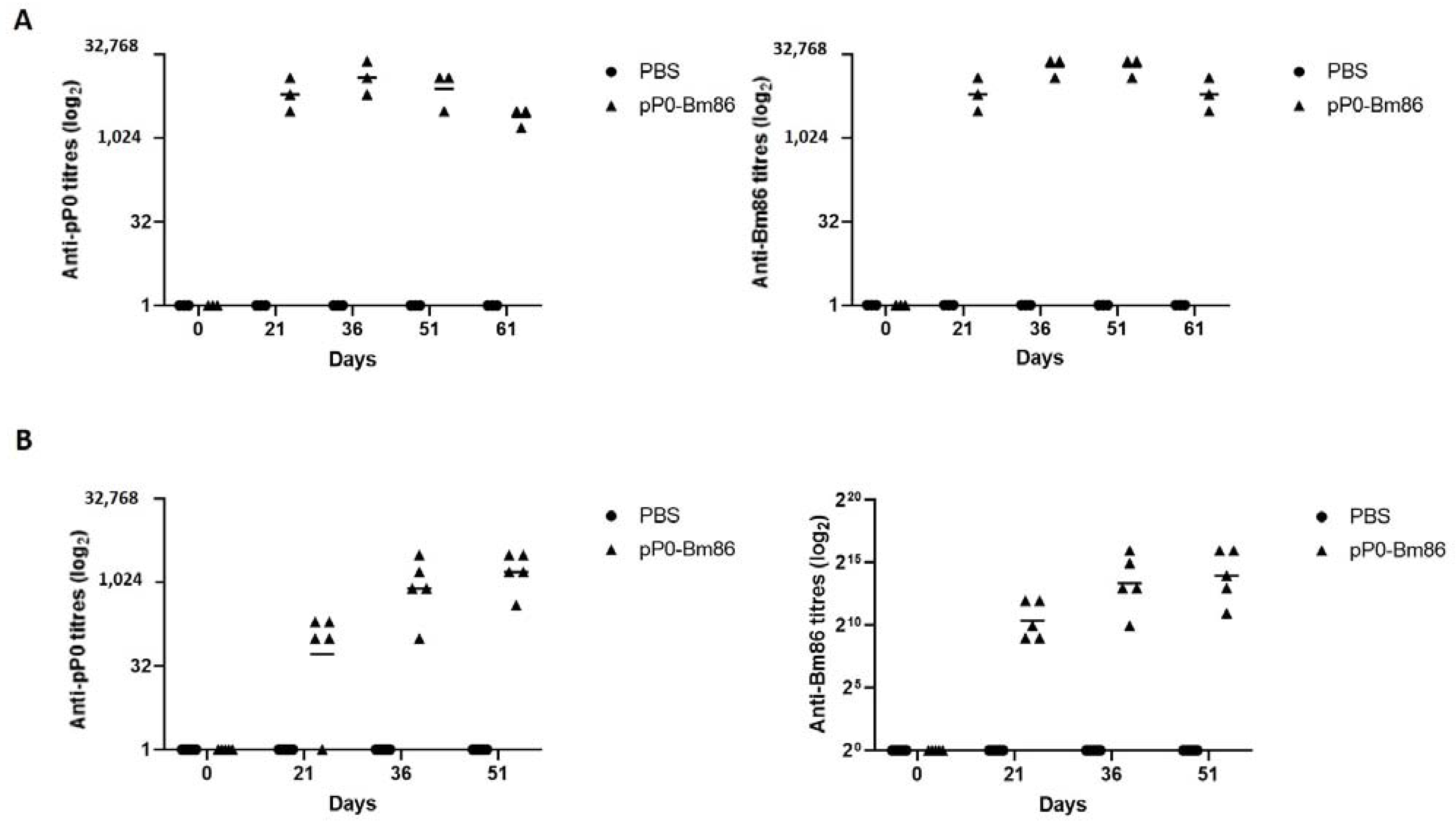
2.2. The Vaccination of Rabbits and Horses with the pP0-Bm86 Conjugate Generates a Specific Antibody Response
2.3. The pP0-Bm86 Conjugate Used as Vaccine Antigen Is Effective against I. ricinus Ticks
2.4. The pP0-Bm86 Conjugate Used as Vaccine Antigen Is Effective against D. nitens Ticks
3. Discussion
4. Materials and Methods
4.1. Ticks and Animals
4.2. RNA Interference
4.3. Synthesis of pP0-Bm86 Conjugate
4.4. Immunization and Challenge Experiment in Rabbits
4.5. Immunization and Challenge Experiment in Horses
4.6. Antibody Response Evaluation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bickerton, M.; Rochlin, I.; González, J.; McSorley, K.; Toledo, A. Field applications of granular and liquid pyrethroids, carbaryl, and IGRs to control the asian longhorned tick (Haemaphysalis longicornis) and impacts on nontarget invertebrates. Ticks Tick-Borne Dis. 2022, 13, 102054. [Google Scholar] [CrossRef] [PubMed]
- Mutavi, F.; Heitkönig, I.; Wieland, B.; Aarts, N.; Van Paassen, A. Tick treatment practices in the field: Access to, knowledge about, and on-farm use of acaricides in Laikipia, Kenya. Ticks Tick-Borne Dis. 2021, 12, 101757. [Google Scholar] [CrossRef]
- Dhang, P.; Koehler, P.; Pereira, R.; Dye II, D.D. Key Questions in Urban Pest Management: A Study and Revision Guide; CAB International: Oxfordshire, UK, 2022. [Google Scholar]
- Kunz, S.E.; Kemp, D.H. Insecticides and acaricides: Resistance and environmental impact. Rev. Sci. Tech. 1994, 13, 1249–1286. [Google Scholar] [CrossRef] [PubMed]
- Agwunobi, D.O.; Yu, Z.; Liu, J. A retrospective review on ixodid tick resistance against synthetic acaricides: Implications and perspectives for future resistance prevention and mitigation. Pestic. Biochem. Physiol. 2021, 173, 104776. [Google Scholar] [CrossRef] [PubMed]
- De Rouck, S.; İnak, E.; Dermauw, W.; Van Leeuwen, T. A review of the molecular mechanisms of acaricide resistance in mites and ticks. Insect Biochem. Mol. Biol. 2023, 159, 103981. [Google Scholar] [CrossRef] [PubMed]
- Klafke, G.M.; Miller, R.J.; Tidwell, J.P.; Thomas, D.B.; Sanchez, D.; Feria Arroyo, T.P.; Perez de Leon, A.A. High-resolution melt (HRM) analysis for detection of SNPs associated with pyrethroid resistance in the southern cattle fever tick, Rhipicephalus (Boophilus) microplus (Acari: Ixodidae). Int. J. Parasitol. Drugs Drug Resist. 2019, 9, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Sagar, S.V.; Saini, K.; Sharma, A.K.; Kumar, S.; Kumar, R.; Fular, A.; Shakya, M.; Upadhaya, D.; Nagar, G.; Samanta, S.; et al. Acaricide resistance in Rhipicephalus microplus collected from selected districts of Madhy Pradesh, Uttar Pradesh and Punjab states of India. Trop. Anim. Health Prod. 2019, 52, 611–618. [Google Scholar] [CrossRef]
- Rodríguez-Valle, M.; Mendez, L.; Valdez, M.; Redondo, M.; Montero Espinosa, C.; Vargas, M.; Lleonart Cruz, R.L.; Pérez Barrios, H.; Seoane, G.; Serrano Ramirez, E.; et al. Integrated control of Boophilus microplus ticks in Cuba based on vaccination with the anti-tick vaccine GavacTM. Exp. Appl. Acarol. 2004, 34, 375–382. [Google Scholar] [CrossRef]
- Suarez, M.; Rubi, J.; Pérez, D.; Cordova, V.; Salazar, Y.; Vielma, A.; Barrios, F.; Gil, C.A.; Segura, N.; Carrillo, Y.; et al. High impact and effectiveness of Gavac™ vaccine in the national program for control of bovine ticks Rhipicephalus microplus in Venezuela. Livest. Sci. 2016, 187, 48–52. [Google Scholar] [CrossRef]
- De la Fuente, J.; Estrada-Peña, A. Why New Vaccines for the Control of Ectoparasite Vectors Have Not Been Registered and Commercialized? Vaccines 2019, 7, 75. [Google Scholar] [CrossRef]
- Smith, D.R.; Hungerford, J.; Willadsen, P.; Cobon, G.S. The development of TickGARD: A commercial vaccine against the cattle tick Boophilus microplus. In Proceedings of the 8th International Congress of Parasitology, Kuşadası, Turkey, 10–14 October 1994. [Google Scholar]
- Alvarez, D.O.; Corona-González, B.; Rodríguez-Mallón, A.; Rodríguez Gonzalez, I.; Alfonso, P.; Noda Ramos, A.A.; Díaz-Sánchez, A.A.; González Navarrete, M.; Rodríguez Fernández, R.; Méndez Mellor, L. Ticks and tick-borne diseases in Cuba, half a century of scientific research. Pathogens 2020, 9, 616. [Google Scholar] [CrossRef] [PubMed]
- Encinosa Guzmán, P.E.; Fernández, C.; Cano Argüelles, A.L.; Fuentes Castillo, A.; García, Y.; Rodríguez, R.; Fernández, Y.; Bello, Y.; González, Y.; Mendez, L.; et al. Characterization of two Cuban colonies of Rhipicephalus microplus ticks. Vet. Parasitol. Reg. Stud. Rep. 2021, 25, 100591. [Google Scholar] [CrossRef] [PubMed]
- Šlapeta, J.; Chandra, S.; Halliday, B. The “tropical lineage” of the brown dog tick Rhipicephalus sanguineus sensu lato identified as Rhipicephalus linnaei (Audouin, 1826). Int. J. Parasitol. 2021, 51, 431–436. [Google Scholar] [CrossRef]
- Encinosa, P.E.; Bello, Y.; Rodríguez-Mallon, A. Genetic and biological characterization of a Cuban tick strain from Rhipicephalus sanguineus complex and its sensitivity to different chemical acaricides. Int. J. Acarol. 2016, 42, 18–25. [Google Scholar] [CrossRef]
- Sanches, G.S.; Evora, P.M.; Mangold, A.J.; Jittapalapong, S.; Rodriguez-Mallon, A.; Guzman, P.E.; Bechara, G.H.; Camargo-Mathias, M.I. Molecular, biological, and morphometric comparisons between different geographical populations of Rhipicephalus sanguineus sensu lato (Acari: Ixodidae). Vet. Parasitol. 2016, 215, 78–87. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Jongejan, F. Ticks feeding on humans: A review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp. Appl. Acarol. 1999, 23, 685–715. [Google Scholar] [CrossRef]
- Dantas-Torres, F.; Figueredo, L.A.; Brandao-Filho, S.P. Rhipicephalus sanguineus (Acari: Ixodidae), the brown dog tick, parasitizing humans in Brazil. Rev. Soc. Bras. Med. Trop. 2006, 39, 64–67. [Google Scholar] [CrossRef]
- Dantas-Torres, F. Biology and ecology of the brown dog tick, Rhipicephalus sanguineus. Parasit. Vectors 2010, 3, 26. [Google Scholar] [CrossRef]
- Encinosa, P.E.; García, Y.; Lleonart, R.; Aliaga, D.; Fernández, Y.; Bello, Y.; la Guardia, C.d.; González, Y.; Díaz, A.; Estrada, M.P.; et al. Morphological and molecular characterization supporting Amblyomma mixtum presence in Cuba. Ticks Tick-Borne Dis. 2021, 12, 101602. [Google Scholar] [CrossRef]
- Charles, R.A.; Bermúdez, S.; Banović, P.; Alvarez, D.O.; Díaz-Sánchez, A.A.; Corona-González, B.; Etter, E.M.C.; Rodríguez González, I.; Ghafar, A.; Jabbar, A.; et al. Ticks and Tick-Borne Diseases in Central America and the Caribbean: A One Health Perspective. Pathogens 2021, 10, 1273. [Google Scholar] [CrossRef]
- Kahl, O.; Gray, J.S. The biology of Ixodes ricinus with emphasis on its ecology. Ticks Tick-Borne Dis. 2023, 14, 102114. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.; Kahl, O.; Zintl, A. What do we still need to know about Ixodes ricinus? Ticks Tick-Borne Dis. 2021, 12, 101682. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Mallon, A.; Fernández, E.; Encinosa, P.E.; Bello, Y.; Méndez-Pérez, L.; Cepero, L.; Pérez, D.; González, M.; Garay, H.; Reyes, O.; et al. A novel tick antigen shows high vaccine efficacy against the dog tick, Rhipicephalus sanguineus. Vaccine 2012, 30, 1782–1789. [Google Scholar] [CrossRef]
- Sanchez-Madrid, F.; Vidales, F.J.; Ballesta, J.P. Functional role of acidic ribosomal proteins. Interchangeability of proteins from bacterial and eukaryotic cells. Biochemistry 1981, 20, 3263–3266. [Google Scholar] [CrossRef] [PubMed]
- Kurtzman, C.P. Description of Komagataella phaffii sp. nov. and the transfer of Pichia pseudopastoris to the methylotrophic yeast genus Komagataella. Int. J. Syst. Evol. Microbiol. 2005, 55, 973–976. [Google Scholar] [CrossRef]
- Canales, M.; Enriquez, A.; Ramos, E.; Cabrera, D.; Dandie, H.; Soto, A.; Falcon, V.; Rodriguez, M.; de la Fuente, J. Large-scale production in Pichia pastoris of the recombinant vaccine Gavac against cattle tick. Vaccine 1997, 15, 414–422. [Google Scholar] [CrossRef]
- Garcia-Garcia, J.C.; Soto, A.; Nigro, F.; Mazza, M.; Joglar, M.; Hechevarria, M.; Lamberti, J.; de la Fuente, J. Adjuvant and immunostimulating properties of the recombinant Bm86 protein expressed in Pichia pastoris. Vaccine 1998, 16, 1053–1055. [Google Scholar] [CrossRef]
- Rodríguez-Mallon, A.; Encinosa, P.E.; Méndez-Pérez, L.; Bello, Y.; Rodríguez Fernández, R.; Garay, H.; Cabrales, A.; Méndez, L.; Borroto, C.; Estrada, M. High efficacy of a 20 amino acid peptide of the acidic ribosomal protein P0 against the cattle tick, Rhipicephalus microplus. Ticks Tick-Borne Dis. 2015, 6, 530–537. [Google Scholar] [CrossRef]
- Rodríguez-Mallon, A.; Encinosa Guzmán, P.E.; Bello Soto, Y.; Rosales Perdomo, K.; Montero Espinosa, C.; Vargas, M.; Estrada García, M.P. A chemical conjugate of the tick P0 peptide is efficacious against Amblyomma mixtum. Transbound. Emerg. Dis. 2019, 67, 175–177. [Google Scholar] [CrossRef]
- Rodriguez Mallon, A.; Javier Gonzalez, L.; Encinosa Guzman, P.E.; Bechara, G.H.; Sanches, G.S.; Pousa, S.; Cabrera, G.; Cabrales, A.; Garay, H.; Mejias, R.; et al. Functional and mass spectrometric evaluation of an anti-tick antigen based on the P0 peptide conjugated to Bm86 protein. Pathogens 2020, 9, 513. [Google Scholar] [CrossRef]
- González, L.J.; Encinosa Guzmán, P.E.; Machado, W.; Pousa, S.; Leyva, A.; Arguelles, A.L.C.; Cabrera, G.; Espinosa, L.A.; Parra, R.; Hernández, R. Synthesis, LC-MS/MS analysis, and biological evaluation of two vaccine candidates against ticks based on the antigenic P0 peptide from R. sanguineus linked to the p64K carrier protein from Neisseria meningitidis. Anal. Bioanal. Chem. 2021, 413, 5885–5900. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Liao, M.; Zhou, J.; Hatta, T.; Huang, P.; Zhang, G.; Kanuka, H.; Nishikawa, Y.; Xuan, X.; Fujisaki, K. Gene silencing of ribosomal protein P0 is lethal to the tick Haemaphysalis longicornis. Vet. Parasitol. 2008, 151, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cui, J.; Zhou, Y.; Cao, J.; Gong, H.; Zhang, H.; Zhou, J. Liposome mediated double-stranded RNA delivery to silence ribosomal protein P0 in the tick Rhipicephalus haemaphysaloides. BMC Ecol. 2018, 9, 638–644. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, J.; Manzano-Roman, R.; Naranjo, V.; Kocan, K.M.; Zivkovic, Z.; Blouin, E.F.; Canales, M.; Almazan, C.; Galindo, R.C.; Step, D.L.; et al. Identification of protective antigens by RNA interference for control of the lone star tick, Amblyomma americanum. Vaccine 2010, 28, 1786–1795. [Google Scholar] [CrossRef]
- Wojda, I.; Cytrynska, M.; Frajnt, M.; Jakubowicz, T. Protein kinases CKI and CKII are implicated in modification of ribosomal proteins of the yeast Trichosporon cutaneum. Acta Biochim. Pol. 2002, 49, 947–957. [Google Scholar] [CrossRef]
- Yacoub, A.; Kelley, M.R.; Deutsch, W.A. Drosophila ribosomal protein PO contains apurinic/apyrimidinic endonuclease activity. Nucleic Acids Res. 1996, 24, 4298–4303. [Google Scholar] [CrossRef]
- Sehgal, A.; Kumar, N.; Carruthers, V.B.; Sharma, S. Translocation of ribosomal protein P0 onto the Toxoplasma gondii tachyzoite surface. Int. J. Parasitol. 2003, 33, 1589–1594. [Google Scholar] [CrossRef]
- Singh, S.; Sehgal, A.; Waghmare, S.; Chakraborty, T.; Goswami, A.; Sharma, S. Surface expression of the conserved ribosomal protein P0 on parasite and other cells. Mol. Biochem. Parasitol. 2002, 119, 121–124. [Google Scholar] [CrossRef]
- Radulović, Ž.M.; Kim, T.K.; Porter, L.M.; Sze, S.H.; Lewis, L.; Mulenga, A. A 24-48 h fed Amblyomma americanum tick saliva immuno-proteome. BMC Genom. 2014, 15, 518. [Google Scholar] [CrossRef]
- Tirloni, L.; Reck, J.; Soares Terra, R.M.; Martins, J.R.; Mulenga, A.; Sherman, N.E.; Fox, J.W.; Yates, J.R.; Termignoni, C.; Pinto, A.F.M.; et al. Proteomic analysis of cattle tick Rhipicephalus (Boophilus) microplus saliva: A comparison between partially and fully engorged females. PLoS ONE 2014, 9, e94831. [Google Scholar] [CrossRef]
- Coumou, J.; Wagemakers, A.; Trentelman, J.J.; Nijhof, A.M.; Hovius, J.W. Vaccination against Bm86 homologues in rabbits does not impair Ixodes ricinus feeding or oviposition. PLoS ONE 2014, 10, e0123495. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, J.C.; Gonzalez, I.L.; Gonzalez, D.M.; Valdes, M.; Mendez, L.; Lamberti, J.; D’Agostino, B.; Citroni, D.; Fragoso, H.; Ortiz, M.; et al. Sequence variations in the Boophilus microplus Bm86 locus and implications for immunoprotection in cattle vaccinated with this antigen. Exp. Appl. Acarol. 1999, 23, 883–895. [Google Scholar] [CrossRef] [PubMed]
- Ben Said, M.; Galai, Y.; Mhadhbi, M.; Jedidi, M.; de la Fuente, J.; Darghouth, M.A. Molecular characterization of Bm86 gene orthologs from Hyalomma excavatum, Hyalomma dromedarii and Hyalomma marginatum marginatum and comparison with a vaccine candidate from Hyalomma scupense. Vet. Parasitol. 2012, 190, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, J.C.; Montero, C.; Rodriguez, M.; Soto, A.; Redondo, M.; Valdes, M.; Mendez, L.; de la Fuente, J. Effect of particulation on the immunogenic and protective properties of the recombinant Bm86 antigen expressed in Pichia pastoris. Vaccine 1998, 16, 374–380. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, J.; Rodriguez, M.; Montero, C.; Redondo, M.; Garcia-Garcia, J.C.; Mendez, L.; Serrano, E.; Valdes, M.; Enriquez, A.; Canales, M.; et al. Vaccination against ticks (Boophilus spp.): The experience with the Bm86-based vaccine GavacTM. Genet. Anal. 1999, 15, 143–148. [Google Scholar] [CrossRef] [PubMed]
- NRC. Guide for the Care and Use of Laboratory Animals: Eighth Edition; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Fuentes Castillo, A.; Armenteros Zaldívar, Y.; Ledesma Bravo, F.L.; Bello Soto, Y.; Rodríguez Fernández, R.; Méndez, L. Obtaining Dermacentor nitens Ticks (Neumann, 1897) (Acari: Ixodidae) in Cattle in BioTicks; Elfos Scientiae: Varadero, Cuba, 2022. [Google Scholar]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [PubMed]
- Hajdusek, O.; Sojka, D.; Kopacek, P.; Buresova, V.; Franta, Z.; Sauman, I.; Winzerling, J.; Grubhoffer, L. Knockdown of proteins involved in iron metabolism limits tick reproduction and development. Proc. Natl. Acad. Sci. USA 2009, 106, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Perez-Perez, D.; Bechara, G.H.; Machado, R.Z.; Andrade, G.M.; Del Vecchio, R.E.; Pedroso, M.S.; Hernandez, M.V.; Farnos, O. Efficacy of the Bm86 antigen against immature instars and adults of the dog tick Rhipicephalus sanguineus (Latreille, 1806) (Acari: Ixodidae). Vet. Parasitol. 2010, 167, 321–326. [Google Scholar] [CrossRef]
| Group | Larva Yield | Mortality in Molt (%) | Nymph Yield | Mortality in Molt (%) | Female Yield | Female Weight (mg) | Egg Mass (mg) | Hatching (%) | E (%) |
|---|---|---|---|---|---|---|---|---|---|
| pP0-Bm86 | 142 ± 107 a | 65 ± 21 a | 16 ± 1 a | 86 ± 3 a | 17 ± 2 a | 207.5 ± 66.1 a | 40.57 ± 22.3 a | ND | 63 |
| PBS | 163 ± 50 a | 54 ± 5 a | 21 ± 2 b | 71 ± 8 b | 18 ± 1 a | 185.5 ± 55.3 a | 47.25 ± 20.3 a | ND |
| Group | Female Yield | Female Weight (mg) | Egg Mass (mg) | IEC (%) | Hatching (%) | E (%) |
|---|---|---|---|---|---|---|
| pP0-Bm86 | 373 ± 500 a | 302.2 ± 39.67 b | 142.3 ± 27.38 a | 47.42± 6.5 a | 68.67 ± 22.94 a | 55 |
| PBS | 693 ± 567 a | 276.1 ± 35.06 a | 142.5 ± 20.85 a | 49.97 ± 5.7 b | 82.71 ± 14.23 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Mallon, A.; Encinosa Guzmán, P.E.; Bello, Y.; Domingos, A.; Antunes, S.; Kopacek, P.; Santos, A.S.; Velez, R.; Perner, J.; Ledesma Bravo, F.L.; et al. Efficacy of the Vaccine Candidate Based on the P0 Peptide against Dermacentor nitens and Ixodes ricinus Ticks. Pathogens 2023, 12, 1365. https://doi.org/10.3390/pathogens12111365
Rodríguez-Mallon A, Encinosa Guzmán PE, Bello Y, Domingos A, Antunes S, Kopacek P, Santos AS, Velez R, Perner J, Ledesma Bravo FL, et al. Efficacy of the Vaccine Candidate Based on the P0 Peptide against Dermacentor nitens and Ixodes ricinus Ticks. Pathogens. 2023; 12(11):1365. https://doi.org/10.3390/pathogens12111365
Chicago/Turabian StyleRodríguez-Mallon, Alina, Pedro E. Encinosa Guzmán, Yamil Bello, Ana Domingos, Sandra Antunes, Petr Kopacek, Ana Sofia Santos, Rita Velez, Jan Perner, Frank L. Ledesma Bravo, and et al. 2023. "Efficacy of the Vaccine Candidate Based on the P0 Peptide against Dermacentor nitens and Ixodes ricinus Ticks" Pathogens 12, no. 11: 1365. https://doi.org/10.3390/pathogens12111365
APA StyleRodríguez-Mallon, A., Encinosa Guzmán, P. E., Bello, Y., Domingos, A., Antunes, S., Kopacek, P., Santos, A. S., Velez, R., Perner, J., Ledesma Bravo, F. L., Frantova, H., Erhart, J., Rodríguez, R., Fuentes, A., Diago, D., Joglar, M., Méndez, L., & Estrada, M. P. (2023). Efficacy of the Vaccine Candidate Based on the P0 Peptide against Dermacentor nitens and Ixodes ricinus Ticks. Pathogens, 12(11), 1365. https://doi.org/10.3390/pathogens12111365







