New Reports of Phytophthora Species in Plant Nurseries in Spain
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Isolation from Plant Tissues, Soil, and Water
2.3. Identification
Molecular Identification
2.4. Conservation of Phytophthora and Pythium Isolates
3. Results
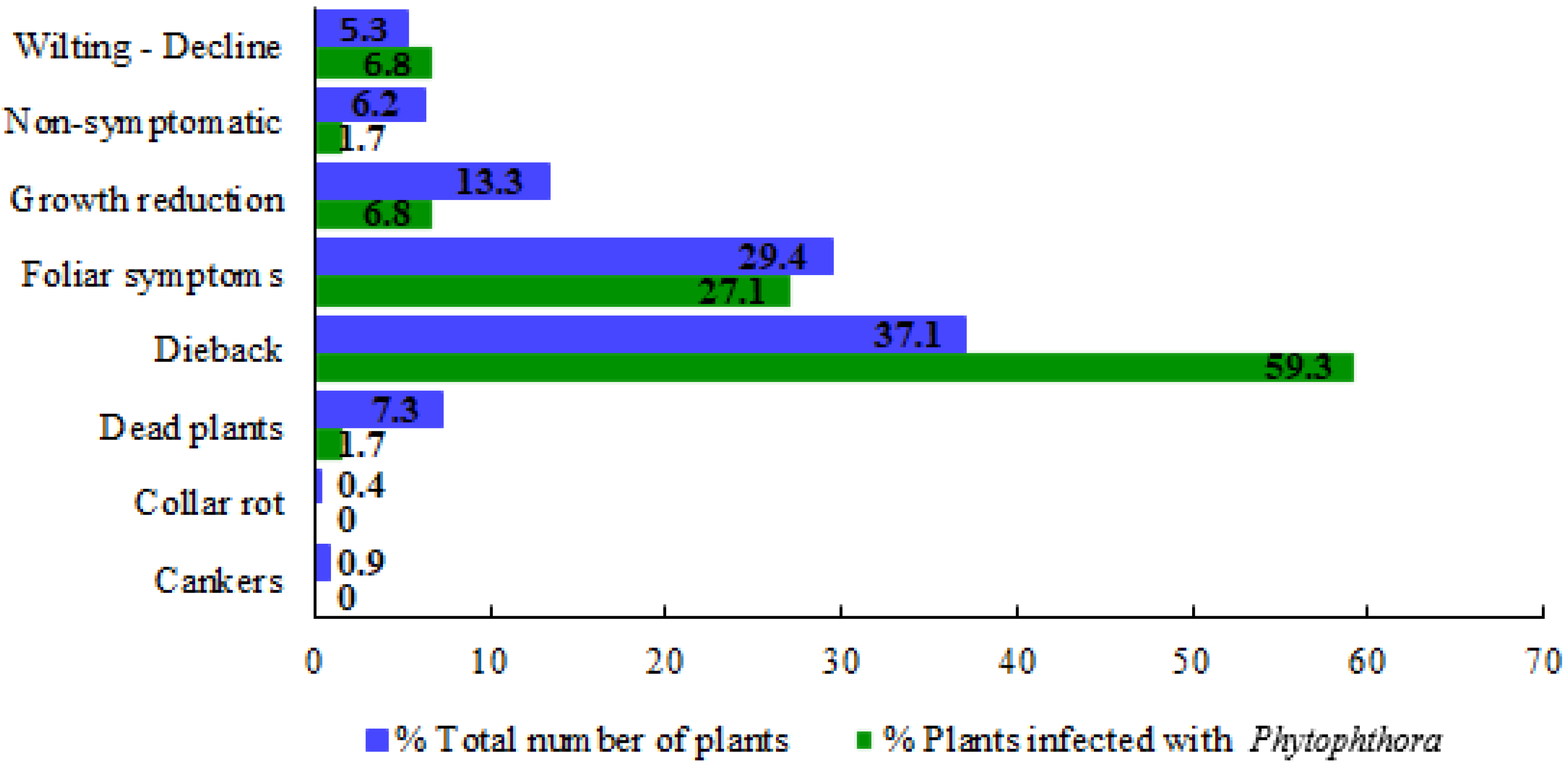
3.1. Symptomatology
3.2. Phytophthora Species Isolated in the Study
3.3. Pythium and Phytopythium Species Isolated in the Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parke, J.L.; Knaus, B.J.; Fieland, V.J.; Lewis, C.; Grünwald, N.J. Phytophthora community structure analyses in Oregon nurseries inform systems approaches to disease management. Phytopathology 2004, 104, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Hyam, L. Plant biosecurity—The way ahead. Farm Policy J. 2008, 5, 47–57. [Google Scholar]
- Tremblay, É.D.; Duceppe, M.O.; Bérubé, J.A.; Kimoto, T.; Lemieux, C.; Bilodeau, G.J. Screening for exotic forest pathogens to increase survey capacity using metagenomics. Phytopathology 2018, 108, 1509–1521. [Google Scholar] [CrossRef] [PubMed]
- Benson, D.M.; Jones, R.K. Etiology of Rhododendron dieback caused by four species of Phytophthora. Plant Dis. 1980, 64, 687–691. [Google Scholar] [CrossRef]
- Erwin, D.C.; Ribeiro, O.K. Phytophthora Diseases Worldwide; APS Press: St. Paul, MN, USA, 1996; 562p, ISBN 0-89054-212-0. [Google Scholar]
- Ferguson, A.J.; Jeffers, S.N. Detecting multiple species of Phytophthora in container mixes from ornamental crop nurseries. Plant Dis. 1999, 83, 1129–1136. [Google Scholar] [CrossRef]
- Werres, S.; Marwitz, R.; Man In’t Veld, W.A.; de Cock, A.W.; Bonants, P.J.M.; de Weerdt, M.; Themann, K.; Ilieva, E.; Baayen, R.P. Phytophthora ramorum sp. nov., a new pathogen on Rhododendron and Viburnum. Mycol. Res. 2001, 105, 1155–1165. [Google Scholar] [CrossRef]
- Osterbauer, N.K.; Griesbach, J.A.; Hedberg, J. Surveying for and eradicating Phytophthora ramorum in agricultural commodities. Plant Health Prog. 2004, 5, 8. [Google Scholar] [CrossRef]
- Pérez-Sierra, A.; Mora-Sala, B.; León, M.; García-Jiménez, J.; Abad-Campos, P. Enfermedades causadas por Phytophthora en viveros de plantas ornamentales. Bol. Sanid. Veg. Plagas 2012, 38, 143–156. [Google Scholar]
- Panabières, F.; Ali, G.S.; Allagui, M.B.; Dalio, R.J.D.; Gudmestad, N.C.; Kuhn, M.L.; Guharoy, S.; Schena, L.; Zampounis, A. Phytophthora nicotianae diseases worldwide: New knowledge of a long-recognised pathogen. Phytopathol. Mediterr. 2016, 55, 20–40. [Google Scholar]
- Jung, T.; Orlikowski, L.; Henricot, B.; Abad-Campos, P.; Aday, A.G.; Aguín Casal, O.; Bakonyi, J.; Cacciola, S.O.; Cech, T.; Chavarriaga, D.; et al. Widespread Phytophthora infestations in European nurseries put forest, semi-natural and horticultural ecosystems at high risk of Phytophthora diseases. For. Pathol. 2016, 46, 134–163. [Google Scholar] [CrossRef]
- Lévesque, C.A. Fifty years of oomycetes—From consolidation to evolutionary and genomic exploration. Fungal Divers. 2011, 50, 35–46. [Google Scholar] [CrossRef]
- Kroon, L.P.N.M.; Brouwer, H.; de Cock, A.W.; Govers, F. The genus Phytophthora anno 2012. Phytopathology 2012, 102, 348–364. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.N.; Abad, Z.G.; Balci, Y.; Ivors, K. Identification and detection of Phytophthora: Reviewing our progress, identifying our needs. Plant Dis. 2012, 96, 1080–1103. [Google Scholar] [CrossRef] [PubMed]
- Brasier, C.M.; Robredo, F.; Ferraz, J.F.P. Evidence for Phytophthora cinnamomi involvement in Iberian oak decline. Plant Pathol. 1993, 42, 140–145. [Google Scholar] [CrossRef]
- Hüberli, D.; Tommerup, I.C.; Dobrowolski, M.P.; Calver, M.C.; Hardy, G.E.S.J. Phenotypic variation in a clonal lineage of two Phytophthora cinnamomi populations from Western Australia. Mycol. Res. 2001, 105, 1053–1064. [Google Scholar] [CrossRef]
- Rizzo, D.M.; Garbelotto, M.; Davidson, J.M.; Slaughter, G.W.; Koike, S.T. Phytophthora ramorum as the cause of extensive mortality of Quercus spp. and Lithocarpus densiflorus in California. Plant Dis. 2002, 86, 205–214. [Google Scholar] [CrossRef]
- Brasier, C.M.; Denman, S.; Brown, A.; Webber, J. Sudden Oak Death (Phytophthora ramorum) discovered on trees in Europe. Mycol. Res. 2004, 108, 1107–1110. [Google Scholar] [CrossRef]
- Camilo-Alves, C.S.P.; da Clara, M.I.E.; de Almeida Ribeiro, N.M.C. Decline of Mediterranean oak trees and its association with Phytophthora cinnamomi: A review. Eur. J. For. Res. 2013, 132, 411–432. [Google Scholar] [CrossRef]
- Grünwald, N.J.; Leboldus, J.M.; Hamelin, R.C. Ecology and evolution of the Sudden Oak Death pathogen Phytophthora ramorum. Annu. Rev. Phytopathol. 2019, 57, 301–321. [Google Scholar] [CrossRef]
- Serrano, M.S.; Garbelotto, M. Differential response of four Californian native plants to worldwide Phytophthora cinnamomi genotypes: Implications for the modeling of disease spread in California. Eur. J. Plant Pathol. 2020, 156, 851–866. [Google Scholar] [CrossRef]
- Tooley, P.W. Susceptibility of selected Ericaceous ornamental host species in Phytophthora ramorum. Plant Dis. 2004, 88, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Ivors, K.; Garbelotto, M.; Vries, I.D.E.; Ruyter-Spira, C.; Hekkert, B.T.; Rosenzweig, N.; Bonants, P. Microsatellite markers identify three lineages of Phytophthora ramorum in US nurseries, yet single lineages in US forest and European nursery populations. Mol. Ecol. 2006, 15, 1493–1505. [Google Scholar] [CrossRef] [PubMed]
- Cooke, D.E.L.; Schena, L.; Cacciola, S.O. Tools to detect, identify and monitor Phytophthora species in natural ecosystems. J. Plant Pathol. 2007, 89, 13–28. [Google Scholar]
- Moralejo, E.; Pérez-Sierra, A.M.; Álvarez, L.A.; Belbahri, L.; Lefort, F.; Descals, E. Multiple alien Phytophthora taxa discovered on diseased ornamental plants in Spain. Plant Pathol. 2009, 58, 100–110. [Google Scholar] [CrossRef]
- Leonberger, A.J.; Speers, C.; Ruhl, G.; Creswell, T.; Beckerman, J.L. A survey of Phytophthora spp. in Midwest nurseries, greenhouses, and landscapes. Plant Dis. 2013, 97, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Abad, Z.G.; Abad, J.A.; Cacciola, S.O.; Pane, A.; Faedda, R.; Moralejo, E.; Pérez-Sierra, A.; Abad-Campos, P.; Alvarez-Bernaola, L.A.; Bakonyi, J.; et al. Phytophthora niederhauserii sp. nov., a polyphagous species associated with ornamentals, fruit trees and native plants in 13 countries. Mycologia 2014, 106, 431–447. [Google Scholar] [CrossRef]
- Weste, G. Population dynamics and survival of Phytophthora. In Phytophthora: Its Biology, Taxonomy, Ecology and Pathology; Erwin, D.C., Barnticki-Garcia, S., Tsao, P.H., Eds.; American Phytopathological Society: St. Paul, MN, USA, 1983; pp. 237–257. [Google Scholar]
- Brasier, C.M. The biosecurity threat to the UK and global environment from international trade in plants. Plant Pathol. 2008, 57, 792–808. [Google Scholar] [CrossRef]
- Eschen, R.; Rigaux, L.; Sukovata, L.; Vettraino, A.M.; Marzano, M.; Grégoire, J.C. Phytosanitary inspection of woody plants for planting at European Union entry points: A practical enquiry. Biol. Invasions 2015, 17, 2403–2413. [Google Scholar] [CrossRef]
- Santini, A.; Ghelardini, L.; de Pace, C.; Desprez-Loustau, M.L.; Capretti, P.; Chandelier, A.; Cech, T.; Chira, D.; Diamandis, S.; Gaitniekis, T.; et al. Biogeographical patterns and determinants of invasion by forest pathogens in Europe. New Phytol. 2012, 197, 238–250. [Google Scholar] [CrossRef]
- Jung, T.; Horta Jung, M.; Webber, J.F.; Kageyama, K.; Hieno, A.; Masuya, H.; Uematsu, S.; Pérez-Sierra, A.; Harris, A.R.; Forster, J.; et al. The destructive tree pathogen Phytophthora ramorum originates from the laurosilva forests of East Asia. J. Fungi 2021, 7, 226. [Google Scholar] [CrossRef]
- Prigigallo, M.I.; Mosca, S.; Cacciola, S.O.; Cooke, D.E.L.; Schena, L. Molecular analysis of Phytophthora diversity in nursery-grown ornamental and fruit plants. Plant Pathol. 2015, 64, 1308–1319. [Google Scholar] [CrossRef]
- Hulbert, J.M.; Agne, M.C.; Burgess, T.I.; Roets, F.; Wingfield, M.J. Urban environments provide opportunities for early detections of Phytophthora invasions. Biol. Invasions 2017, 19, 3629–3644. [Google Scholar] [CrossRef]
- Peterson, E.; Hansen, E.; Hulbert, J. Source or sink? The role of soil and water borne inoculum in the dispersal of Phytophthora ramorum in Oregon tanoak forests. For. Ecol. Manag. 2014, 322, 48–57. [Google Scholar] [CrossRef]
- Jones, D.R.; Baker, R.H.A. Introductions of non-native plant pathogens into Great Britain, 1970–2004. Plant Pathol. 2007, 56, 891–910. [Google Scholar] [CrossRef]
- Reichard, S.H.; White, P. Horticulture as a pathway of invasive plant introductions in the United States. BioScience 2001, 51, 103–113. [Google Scholar] [CrossRef]
- Harris, A.R.; Webber, J.F. Insights into the potential host range of Phytophthora foliorum. For. Pathol. 2019, 49, e12556. [Google Scholar] [CrossRef]
- Moralejo, E.; Werres, S. First Report of Phytophthora ramorum on Rhododendron sp. in Spain. Plant Dis. 2002, 86, 1052. [Google Scholar] [CrossRef]
- Varela, C.P.; Vázquez, J.P.M.; Casal, O.A. First Report of Phytophthora ramorum on Camellia japonica in Spain. Plant Dis. 2003, 87, 1396. [Google Scholar] [CrossRef]
- Sims, L.L.; Garbelotto, M. Phytophthora species repeatedly introduced in Northern California through restoration projects can spread into adjacent sites. Biol. Invasions 2021, 23, 2173–2190. [Google Scholar] [CrossRef]
- Pérez-Sierra, A.M.; León, M.; Álvarez, L.A.; Alaniz, S.; Berbegal, M.; García-Jiménez, J.; Abad-Campos, P. Outbreak of a new Phytophthora sp. associated with severe decline of almond trees in Eastern Spain. Plant Dis. 2010, 94, 534–541. [Google Scholar] [CrossRef]
- Jeffers, S.N.; Aldwinckle, H.S. Enhancing detection of Phytophthora cactorum in naturally infested soil. Phytopathology 1987, 77, 1475–1482. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, S.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Cooke, D.E.L.; Drenth, A.; Duncan, J.M.; Wagels, G.; Brasier, C.M. A molecular phylogeny of Phytophthora and related Oomycetes. Fungal Genet. Biol. 2000, 30, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Robideau, G.P.; de Cock, A.W.A.M.; Coffey, M.D.; Voglmayr, H.; Brouwer, H.; Bala, K.; Chitty, D.W.; Désaulniers, N.; Eggertson, Q.A.; Gachon, C.M.M.; et al. DNA barcoding of oomycetes with cytochrome c oxidase subunit I and internal transcribed spacer. Mol. Ecol. Resour. 2011, 11, 1002–1011. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Lemoine, F.; Correia, D.; Lefort, V.; Doppelt-Azeroual, O.; Mareuil, F.; Cohen-Boulakia, S.; Gascuel, O. NGPhylogeny.fr: New generation phylogenetic services for non-specialists. Nucleic Acids Res. 2019, 47, W260–W265. [Google Scholar] [CrossRef]
- Alix, B.; Boubacar, D.A.; Vladimir, M. T-REX: A web server for inferring, validating and visualizing phylogenetic trees and networks. Nucleic Acids Res. 2012, 40, W573–W579. [Google Scholar]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Jung, T.; Scanu, B.; Brasier, C.M.; Webber, J.; Milenković, I.; Corcobado, T.; Tomšovský, M.; Pánek, m.; Bakonyi, J.; Maia, C.; et al. A survey in natural forest ecosystems of Vietnam reveals high diversity of both new and described Phytophthora taxa including P. ramorum. Forests 2020, 11, 93. [Google Scholar] [CrossRef]
- Cacciola, S.O.; Pane, A.; Polizzi, G. Due specie di Phytophthora agenti di marciume radicale e del colletto del rosmarino. Inf. Fitopatol. 1997, 47, 35–42. [Google Scholar]
- Schwingle, B.W.; Smith, J.A.; Blanchette, R.A. Phytophthora species associated with diseased woody ornamentals in Minnesota nurseries. Plant Dis. 2007, 91, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Grünwald, N.J.; Goss, E.M.; Press, C.M. Phytophthora ramorum: A pathogen with a remarkably wide host range causing Sudden Oak Death on oaks and ramorum blight on woody ornamentals. Mol. Plant Pathol. 2008, 9, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Bienapfl, J.C.; Balci, Y. Movement of Phytophthora spp. in Maryland’s nursery trade. Plant Dis. 2014, 98, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Cacciola, S.O.; Polizzi, G. Phytophthora nicotianae e P. palmivora agenti di marciume radicale e del colletto del Pittosporo. Inf. Fitopatol. 1996, 46, 25–29. [Google Scholar]
- Scanu, B.; Linaldeddu, B.T.; Deidda, A.; Jung, T. Diversity of Phytophthora species from declining Mediterranean maquis vegetation, including two new species, Phytophthora crassamura and P. ornamentata sp. nov. PLoS ONE 2015, 10, e0143234. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; La Spada, F.; Pane, A.; Aloi, F.; Evoli, M.; Horta Jung, M.; Scanu, B.; Faedda, R.; Rizza, C.; Puglisi, I. Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily. Forests 2019, 10, 259. [Google Scholar] [CrossRef]
- Sims, L.; Tjosvold, S.; Chambers, D.; Garbelotto, M. Control of Phytophthora species in plant stock for habitat restoration through best management practices. Plant Pathol. 2019, 68, 196–204. [Google Scholar] [CrossRef]
- Safaiefarahani, B.; Mostowfizadeh-Ghalamfarsa, R.; Hardy, G.E.S.J.; Burgess, T.I. Re-evaluation of Phytophthora cryptogea species complex and the description of a new species, Phytophthtora pseudocryptogea sp. nov. Mycol. Prog. 2015, 14, 108. [Google Scholar] [CrossRef]
- Mora-Sala, B.; Berbegal, M.; Abad-Campos, P. The Use of qPCR Reveals a High Frequency of Phytophthora quercina in Two Spanish Holm Oak Areas. Forests 2018, 9, 697. [Google Scholar] [CrossRef]
- Mora-Sala, B.; Abad-Campos, P.; Berbegal, M. Response of Quercus ilex seedlings to Phytophthora spp. root infection in a soil infestation test. Eur. J. Plant Pathol. 2019, 154, 215–225. [Google Scholar] [CrossRef]
- Hansen, E.M.; Wilcox, W.F.; Reeser, P.W.; Sutton, W. Phytophthora rosacearum and P. sansomeana, new species segregated from the Phytophthora megasperma “complex”. Mycologia 2009, 101, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.H.; Gao, F.; Li, G.Y.; Wang, H.; Zheng, X.B.; Wang, Y.C. First report of root rot caused by Phytophthora sansomeana on soybean in China. Plant Dis. 2010, 94, 378. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.F.; Hwang, S.F.; Ahmed, H.U.; Fu, H.; Zhou, Q.; Strelkov, S.E.; Turnbull, G.D. First report of Phytophthora sansomeana causing root rot in field pea in Alberta, Canada. Crop Prot. 2017, 101, 1–4. [Google Scholar] [CrossRef]
- Sanchez, M.E.; Andicoberry, S.; Trapero, A. Pathogenicity of three Phytophthora spp. causing late seedling rot of Quercus ilex ssp. ballota. For. Pathol. 2005, 35, 115–125. [Google Scholar] [CrossRef]
- Donahoo, R.S.; Lamour, K.H. Characterization of Phytophthora species from leaves of nursery woody ornamentals in Tennessee. Hort. Science 2008, 43, 1833–1837. [Google Scholar] [CrossRef]
- Olson, H.A.; Benson, D.M. Characterization of Phytophthora spp. on floriculture crops in North Carolina. Plant Dis. 2011, 95, 1013–1020. [Google Scholar] [CrossRef]
- Prospero, S.; Vercauteren, A.; Heungens, K.; Belbahri, L.; Rigling, D. Phytophthora diversity and the population structure of Phytophthora ramorum in Swiss ornamental nurseries. Plant Pathol. 2013, 62, 1063–1071. [Google Scholar] [CrossRef]
- Prigigallo, M.I.; Abdelfattah, A.; Cacciola, S.O.; Faedda, R.; Sanzani, S.M.; Cooke, D.E.L.; Schena, L. Metabarcoding analysis of Phytophthora diversity using genus-specific primers and 454 pyrosequencing. Phytopathology 2016, 106, 305–313. [Google Scholar] [CrossRef]
- Rooney-Latham, S.; Blomquist, C.L.; Kosta, K.L.; Gou, Y.Y.; Woods, P.W. Phytophthora species are common on nursery stock grown for restoration and revegetation purposes in California. Plant Dis. 2019, 103, 448–455. [Google Scholar] [CrossRef]
- Hwang, J.; Oak, S.W.; Jeffers, S.N. Detecting Phytophthora ramorum and other species of Phytophthora in streams in natural ecosystems using baiting and filtration methods. In Proceedings of the Sudden Oak Death Third Science Symposium, Santa Rosa, CA, USA, 5–9 March 2007; Frankel, S.J., Kliejunas, J.T., Palmieri, K.M., Eds.; US Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2008. [Google Scholar]
- Reeser, P.W.; Sutton, W.; Hansen, E.M.; Remigi, P.; Adams, G.C. Phytophthora species in forest streams in Oregon and Alaska. Mycologia 2011, 103, 22–35. [Google Scholar] [CrossRef]
- Huai, W.X.; Tian, G.; Hansen, E.M.; Zhao, W.X.; Goheen, E.M.; Grünwald, N.J.; Cheng, C. Identification of Phytophthora species baited and isolated from forest soil and streams in northwestern Yunnan province, China. For. Pathol. 2013, 43, 87–103. [Google Scholar] [CrossRef]
- Hüberli, D.; Hardy, G.E.S.J.; White, D.; Williams, N.; Burgess, T.I. Fishing for Phytophthora from Western Australia’s waterways: A distribution and diversity survey. Australas. Plant Pathol. 2013, 42, 251–260. [Google Scholar] [CrossRef]
- Nagel, J.H.; Gryzenhout, M.; Slippers, B.; Wingfield, M.J.; Hardy, G.E.S.J.; Stukely, M.J.C.; Burgess, T.I. Characterization of Phytophthora hybrids from ITS clade 6 associated with riparian ecosystems in South Africa and Australia. Fungal Biol. 2013, 117, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Zappia, R.E.; Hüberli, D.; Hardy, G.E.S.J.; Bayliss, K.L. Fungi and oomycetes in open irrigation systems: Knowledge gaps and biosecurity implications. Plant Pathol. 2014, 63, 961–972. [Google Scholar] [CrossRef]
- Català, S.; Pérez-Sierra, A.; Abad-Campos, P. The use of genus-specific amplicon pyrosequencing to assess Phytophthora species diversity using eDNA from soil and water in northern Spain. PLoS ONE 2015, 10, e0119311. [Google Scholar] [CrossRef]
- Migliorini, D.; Ghelardini, L.; Tondini, E.; Luchi, N.; Santini, A. The potential of symptomless potted plants for carrying invasive soilborne plant pathogens. Divers. Distrib. 2015, 21, 1218–1229. [Google Scholar] [CrossRef]
- Sims, L.L.; Sutton, W.; Reeser, P.; Hansen, E.M. The Phytophthora species assemblage and diversity in riparian alder ecosystems of Western Oregon, USA. Mycologia 2015, 107, 889–902. [Google Scholar] [CrossRef]
- Redondo, M.A.; Boberg, J.; Stenlid, J.; Oliva, J. Contrasting distribution patterns between aquatic and terrestrial Phytophthora species along a climatic gradient are linked to functional traits. ISME J. 2018, 12, 2967–2980. [Google Scholar] [CrossRef]
- Hansen, E.M.; Goheen, D.J.; Jules, E.S.; Ullian, B. Managing Port-Orford-Cedar and the introduced pathogen Phytophthora lateralis. Plant Dis. 2000, 84, 4–14. [Google Scholar] [CrossRef]
- Brasier, C.M.; Kirk, S.A.; Delcan, J.; Cooke, D.E.L.; Jung, T.; Man In’t Veld, W.A. Phytophthora alni sp. nov. and its variants: Designation of emerging heteroploid hybrid pathogens spreading on Alnus trees. Mycol. Res. 2004, 108, 1172–1184. [Google Scholar] [CrossRef]
- Crous, P.W.; Summerell, B.A.; Shivas, R.G.; Burgess, T.I.; de Cock, C.A.; Dreyer, L.L.; Granke, L.L.; Guest, D.I.; Hardy, G.E.S.J.; Hausbeck, M.K.; et al. Fungal planet description sheets: 107–127. Persoonia 2012, 28, 138–182. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Tyler, B.M.; Hong, C. An expanded phylogeny for the genus Phytophthora. IMA Fungus 2017, 8, 355–384. [Google Scholar] [CrossRef] [PubMed]
- Brasier, C.M.; Cooke, D.E.L.; Duncan, J.M.; Hansen, E.M. Multiple new phenotypic taxa from trees and riparian ecosystems in Phytophthora gonapodyides-P. megasperma ITS clade 6, which tend to be high-temperature tolerant and either inbreeding or sterile. Mycol. Res. 2003, 107, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Stukely, M.J.C.; Hardy, G.E.S.J.; White, D.; Paap, T.; Dunstan, W.A.; Burgess, T.I. Multiple new Phytophthora species from ITS clade 6 associated with natural ecosystems in Australia: Evolutionary and ecological implications. Persoonia 2011, 26, 13–39. [Google Scholar] [CrossRef]
- Guha Roy, S.; Grünwald, N.J. The plant destroyer genus Phytophthora in the 21st century. Rev. Plant Pathol. 2012, 6, 388–412. [Google Scholar]
- Jung, T.; Blaschke, H.; Neumann, P. Isolation, identification and pathogenicity of Phytophthora species from declining oak stands. Eur. J. Forest Pathol. 1996, 26, 253–272. [Google Scholar] [CrossRef]
- Romero, M.A.; Sánchez, J.E.; Jiménez, J.J.; Belbahri, L.; Trapero, A.; Lefort, F.; Sánchez, M.E. New Pythium taxa causing root rot in Mediterranean Quercus species in southwest Spain and Portugal. J. Phytopathol. 2007, 115, 289–295. [Google Scholar] [CrossRef]
- Ivors, K.L.; Abad, Z.G.; Benson, D.M. Evaluating the pathogenicity of Pythium vexans isolates from Fraser fir in North Carolina. Plant Health Prog. 2008, 9, 8. [Google Scholar] [CrossRef]
- Weiland, J.E.; Beck, B.R.; Davis, A. Pathogenicity and virulence of Pythium species obtained from forest nursery soils on Douglas-fir seedlings. Plant Dis. 2013, 97, 744–748. [Google Scholar] [CrossRef]
- Yang, X.; Balci, Y.; Brazee, N.J.; Loyd, A.L.; Hong, C.X. A unique species in Phytophthora clade 10, Phytophthora intercalaris sp. nov., recovered from stream and irrigation water in the eastern USA. Int. J. Syst. Evol. Microbiol. 2016, 66, 845–855. [Google Scholar] [CrossRef]
- Li, D.W.; Schultes, N.P.; LaMondia, J.A.; Cowles, R.S. Phytophthora abietivora, a new species isolated from diseased Christmas trees in Connecticut, U.S.A. Plant Dis. 2019, 103, 3057–3064. [Google Scholar] [CrossRef] [PubMed]
- Brasier, C.M. The rise of the hybrid fungi. Nature 2000, 405, 134–135. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, P.A.; Williams, N.; Hardy, G.E.S.J. Detecting Phytophthora. Crit. Rev. Microbiol. 2009, 35, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Gonthier, P.; Nicolotti, G. Infectious Forest Diseases; CAB International: Wallingford, UK, 2013; 704p. [Google Scholar]
- Gullino, M.L.; Garibaldi, A. Critical aspects in management of fungal diseases of ornamental plants and directions in research. Phytopathol. Mediterr. 2007, 46, 135–149. [Google Scholar]
- Mora-Sala, B.; Gramaje, D.; Abad-Campos, P.; Berbegal, M. Diversity of Phytophthora species associated with Quercus ilex L. in three Spanish regions evaluated by NGS. Forests 2019, 10, 979. [Google Scholar] [CrossRef]
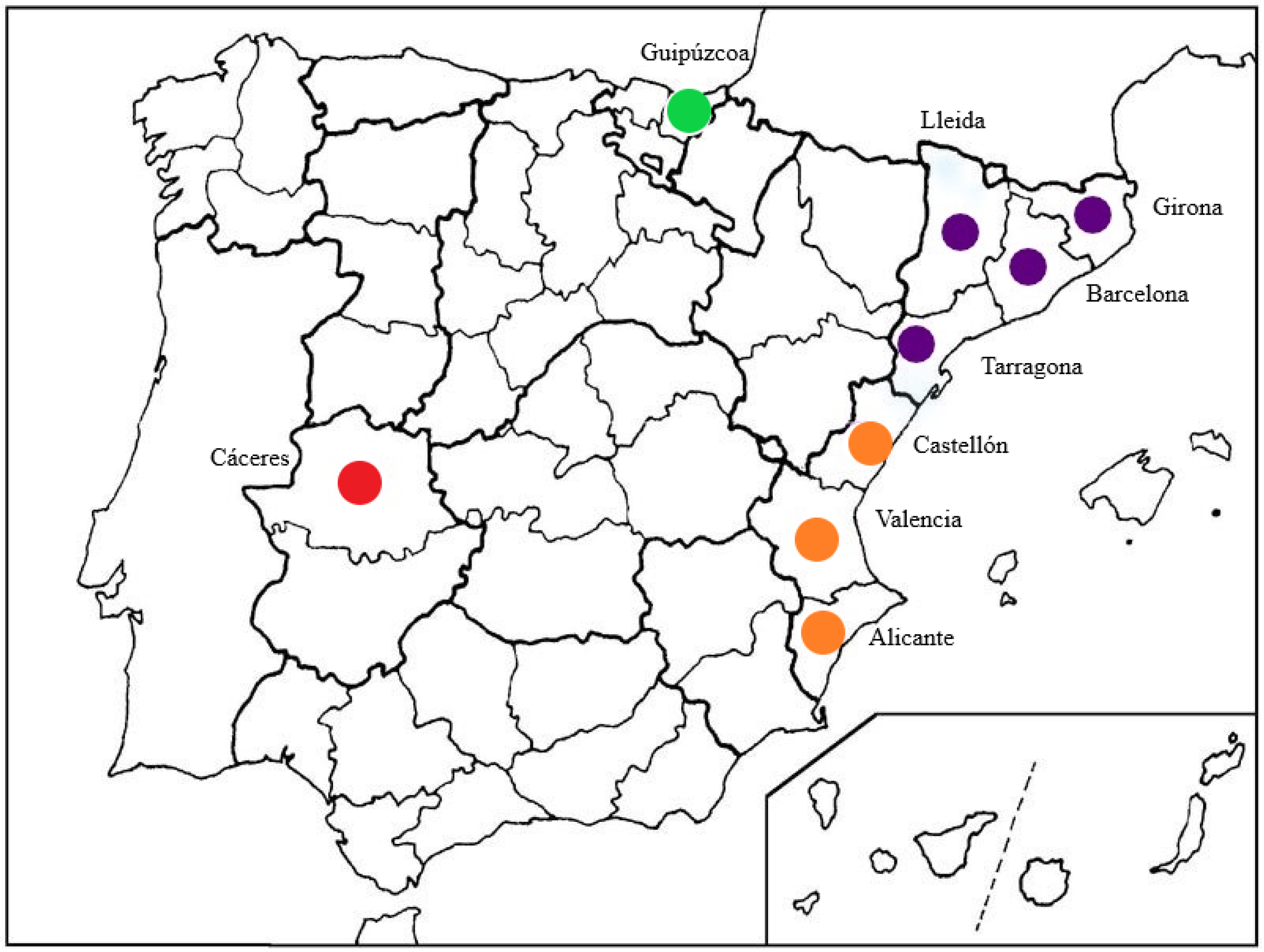


| Species | Strain Number | Host | Location | GenBank Accession Numbers | |
|---|---|---|---|---|---|
| ITS | COI | ||||
| P. bilorbang | PS-1420 | Nursery water pond | Lleida, Spain | MW314333 | - |
| P. cactorum | PS-1463 | Photinia x fraseri “Red Robin” (roots) | Barcelona, Spain | MW314274 | - |
| PS-1411 | Nursery water pond | Girona, Spain | MW314275 | - | |
| PS-1417 | Nursery water pond | Lleida, Spain | MW314276 | - | |
| PS-1447 | Photinia x fraseri “Red Robin” (roots) | Barcelona, Spain | MW314277 | - | |
| PS-1396 | Nursery water pond | Barcelona, Spain | MW314278 | - | |
| PS-1395 | Nursery water pond | Barcelona, Spain | MW314279 | - | |
| PS-1445 | Nursery water pond | Barcelona, Spain | MW314280 | - | |
| PS-1428 | Photinia x fraseri “Red Robin” (roots) | Barcelona, Spain | MW314281 | - | |
| PS-1397 | Nursery water pond | Barcelona, Spain | MW314282 | - | |
| PS-1400 | Nursery water pond | Barcelona, Spain | MW314283 | - | |
| P. cactorum | PS-1600 | Fagus sylvatica (soil) | Guipúzcoa, Spain | MW314287 | - |
| PS-1393 | Nursery water pond | Barcelona, Spain | MW314286 | - | |
| PS-1399 | Nursery water pond | Barcelona, Spain | MW314285 | - | |
| PS-1444 | Nursery water pond | Barcelona, Spain | MW314284 | - | |
| P. cambivora | PS-1394 | Nursery water pond | Barcelona, Spain | MW314353 | - |
| PS-1392 | Nursery water pond | Barcelona, Spain | MW314354 | - | |
| PS-1389 | Nursery water pond | Barcelona, Spain | MW314356 | - | |
| PS-1599 | Quercus ilex (roots) | Valencia, Spain | MW314355 | - | |
| P. cinnamomi | PS-1448 | Arbutus unedo (roots) | Barcelona, Spain | MW314360 | - |
| PS-1470 | Arbutus unedo (roots) | Barcelona, Spain | MW314357 | - | |
| P. cinnamomi | PS-1602 | Pseudotsuga menziesii (roots) | Guipúzcoa, Spain | MW314358 | - |
| PS-1603 | Pinus radiata (roots) | Guipúzcoa, Spain | MW314359 | - | |
| P. citrophthora | PS-1419 | Nursery water pond | Lleida, Spain | MW314301 | - |
| PS-954 | Quercus faginea (soil) | Castellón, Spain | MW314302 | - | |
| PS-1407 | Nursery water pond | Barcelona, Spain | MW314303 | - | |
| PS-1418 | Nursery water pond | Lleida, Spain | MW314304 | - | |
| PS-1426 | Escallonia sp. (soil) | Barcelona, Spain | MW314305 | - | |
| PS-1429 | Picea pungens “Glauca Globosa” (roots) | Girona, Spain | MW314306 | - | |
| PS-955 | Quercus faginea (roots) | Castellón, Spain | MW314307 | - | |
| PS-1474 | Rosmarinus sp. (roots) | Tarragona, Spain | MW314308 | - | |
| PS-1437 | Citrus sinensis (soil) | Tarragona, Spain | MW314309 | - | |
| P. crassamura | 2308 | Pinus pinea (soil) | Cáceres, Spain | MW314336 | - |
| 2309 | Pinus pinea (soil) | Cáceres, Spain | MW314335 | - | |
| P. crassamura | 2310 | Pinus pinea (soil) | Cáceres, Spain | MW314334 | - |
| P. gonapodyides | PS-1415 | Nursery water pond | Girona, Spain | MW314338 | - |
| PS-1434 | Juniperus hibernicus (roots) | Lleida, Spain | MW314337 | - | |
| PS-1410 | Nursery water pond | Girona, Spain | MW314342 | - | |
| PS-1414 | Nursery water pond | Girona, Spain | MW314339 | - | |
| PS-1423 | Leaves floating in nursery water pond | Barcelona, Spain | MW314341 | - | |
| PS-972 | Quercus ilex (soil) | Valencia, Spain | MW314340 | - | |
| PS-1409 | Nursery water pond | Girona, Spain | MW314344 | - | |
| PS-1402 | Nursery water pond | Barcelona, Spain | MW349609 | - | |
| PS-1413 | Nursery water pond | Girona, Spain | MW314343 | - | |
| P. hedraiandra | PS-1449 | Quercus ilex (soil) | Cáceres, Spain | MW314288 | - |
| 2300 | Quercus ilex (soil) | Cáceres, Spain | MW314290 | - | |
| 2301 | Quercus ilex (soil) | Cáceres, Spain | MW314293 | - | |
| P. hedraiandra | PS-1480 | Juniperus phoenicia (soil) | Alicante, Spain | MW314289 | - |
| PS-1427 | Viburnum tinus (collar) | Barcelona, Spain | MW314291 | - | |
| PS-1422 | Viburnum tinus (roots) | Girona, Spain | MW314292 | - | |
| PS-1421 | Viburnum tinus (roots) | Barcelona, Spain | MW314294 | - | |
| P. lacustris | PS-1408 | Nursery water pond | Barcelona, Spain | MW314345 | - |
| PS-1424 | Leaves floating in nursery water pond | Barcelona, Spain | MW314346 | - | |
| PS-1412 | Nursery water pond | Girona, Spain | MW314347 | - | |
| PS-1401 | Nursery water pond | Barcelona, Spain | MW314348 | - | |
| PS-1446 | Nursery water pond | Girona, Spain | MW314349 | - | |
| PS-961 | Nursery fountain | Valencia, Spain | MW314350 | - | |
| PS-1404 | Nursery water pond | Barcelona, Spain | MW314351 | - | |
| P. lacustris | PS-1403 | Nursery water pond | Barcelona, Spain | MW314352 | - |
| P. nicotianae | PS-1441 | Pistacia lentiscus (roots) | Barcelona, Spain | MW314298 | - |
| PS-1425 | Escallonia sp. (roots) | Barcelona, Spain | MW314299 | - | |
| PS-1439 | Citrus sinensis (roots) | Tarragona, Spain | MW314297 | - | |
| PS-1435 | Rosmarinus sp. (roots) | Tarragona, Spain | MW314295 | - | |
| PS-1442 | Myrtus communis “Tarentina” (roots) | Barcelona, Spain | MW314296 | - | |
| PS-1443 | Buxus sempervirens (roots) | Barcelona, Spain | MW314300 | - | |
| P. niederhauserii | PS-1606 | Arbutus unedo (soil) | Cáceres, Spain | MW314361 | - |
| P. palmivora | PS-1465 | Pistacia lentiscus (roots) | Barcelona, Spain | MW314330 | - |
| PS-1475 | Cupressus sempervirens (roots) | Tarragona, Spain | MW314331 | - | |
| PS-1416 | Nursery water pond | Lleida, Spain | MW314332 | - | |
| P. plurivora | 1978 | Quercus faginea (roots) | Castellón, Spain | MW314325 | - |
| P. plurivora | PS-1405 | Nursery water pond | Barcelona, Spain | MW314314 | - |
| PS-1432 | Chamaecyparis lawsoniana “Elwoodii” (soil) | Girona, Spain | MW314326 | MW314042 | |
| PS-1476 | Cupressus sempervirens (roots) | Tarragona, Spain | MW314327 | MW314043 | |
| PS-957 | Quercus faginea (soil) | Castellón, Spain | MW314315 | - | |
| PS-959 | Quercus faginea (roots) | Castellón, Spain | MW314316 | - | |
| PS-1431 | Chamaecyparis lawsoniana “Elwoodii” (soil) | Girona, Spain | MW314317 | - | |
| PS-1390 | Nursery water pond | Barcelona, Spain | MW314318 | - | |
| PS-1391 | Nursery water pond | Barcelona, Spain | MW314319 | - | |
| PS-958 | Quercus faginea (roots) | Castellón, Spain | MW314320 | - | |
| PS-960 | Quercus faginea (roots) | Castellón, Spain | MW314321 | - | |
| PS-969 | Juniperus sp. (soil) | Valencia, Spain | MW314322 | - | |
| P. plurivora | PS-971 | Quercus ilex (roots) | Valencia, Spain | MW314323 | - |
| 2213 | Juniperus chinensis “Expansa Variegata” (roots) | Barcelona, Spain | MW314324 | - | |
| P. pseudocryptogea | PS-1469 | Ilex x meserveae “Blue Maid” (roots) | Barcelona, Spain | MW314362 | - |
| PS-1450 | Chamaecyparis obtusa “Nana gracilis” (roots) | Barcelona, Spain | MW314363 | - | |
| PS-1430 | Chamaecyparis lawsoniana “Elwoodii” (roots) | Girona, Spain | MW314364 | - | |
| PS-967 | Quercus ilex (soil) | Valencia, Spain | MW314365 | - | |
| PS-966 | Quercus ilex (roots) | Valencia, Spain | MW314366 | - | |
| PS-1406 | Nursery water pond | Barcelona, Spain | MW314367 | - | |
| PS-1436 | Yucca rostrate (collar) | Tarragona, Spain | MW314368 | - | |
| PS-1466 | Chamaecyparis lawsoniana “Elwoodii” (roots) | Girona, Spain | MW314369 | - | |
| P. pseudocryptogea | PS-970 | Quercus ilex (soil) | Valencia, Spain | MW314370 | - |
| PS-964 | Quercus ilex (roots) | Valencia, Spain | MW314371 | - | |
| PS-978 | Quercus ilex (soil) | Valencia, Spain | MW314372 | - | |
| PS-973 | Quercus ilex (soil) | Valencia, Spain | MW314373 | - | |
| PS-962 | Quercus ilex (roots) | Valencia, Spain | MW314374 | - | |
| PS-965 | Quercus ilex (roots) | Valencia, Spain | MW314376 | - | |
| PS-968 | Quercus ilex (soil) | Valencia, Spain | MW314377 | - | |
| PS-1398 | Nursery water pond | Barcelona, Spain | MW314375 | - | |
| PS-1605 | Quercus ilex (soil) | Cáceres, Spain | MW314378 | - | |
| P. sansomeana | PS-974 | Quercus ilex (soil) | Valencia, Spain | MW314379 | - |
| PS-979 | Quercus ilex (soil) | Valencia, Spain | MW314380 | MW314044 | |
| P. sansomeana | PS-980 | Quercus ilex (roots) | Valencia, Spain | MW314381 | MW314045 |
| Phytophthora sp. tropicalis-like 2 | PS-1433 | Juniperus hibernicus (soil) | Lleida, Spain | MW314329 | MW314040 |
| PS-1604 | Arbutus unedo (roots) | Cáceres, Spain | MW314328 | MW314041 | |
| Phytophthora sp. 1 clade 2 | PS-1477 | Citrus sinensis (soil) | Tarragona, Spain | MW314310 | MW314036 |
| PS-1460 | Citrus sinensis (soil) | Tarragona, Spain | MW314311 | MW314037 | |
| PS-1478 | Citrus sinensis (roots) | Tarragona, Spain | MW314312 | MW314038 | |
| PS-1479 | Citrus sinensis (roots) | Tarragona, Spain | MW314313 | MW314039 | |
| Phytophthora spp. | Host | Nursery | Source | N. Samples | Region |
|---|---|---|---|---|---|
| BIL | 13 | 1 | |||
| CAC | Fagus sylvatica, Photinia “Red Robin”, Pinus pinea | 6, 7, 9, 10, 12, 13, 19 | R, S, W | 14 | Cat., Com. Val., Bas. C. |
| CAM | Quercus ilex | 3, 7, 9 | R, W | 9 | Com. Val. |
| CIN | Arbutus unedo, Pinus radiata, Pseudotsuga menziesii | 9, 18 | R, S | 4 | Cat., Bas. C. |
| CIP | Citrus sinensis, Escallonia sp., Picea pungens “Glauca Globosa”, Quercus faginea, Rosmarinus officinalis | 4, 10, 11, 13, 15, 16 | R, S, W | 9 | Cat., Com. Val. |
| CRA | Pinus pinea | 17 | S | 3 | Ext. |
| HED | Juniperus phoenicea, Q. ilex, Viburnum tinus | 5, 7, 8, 10, 17 | R, S | 7 | Cat., Com. Val., Ext. |
| GON | Juniperus communis “Hibernica”, Q. ilex | 2, 8, 9, 10, 12, 13 | R, S, L, W | 9 | Cat., Com. Val. |
| LAC | 2, 8, 10 | L, W | 8 | Cat. | |
| NIC | Buxus sempervirens, Citrus sinensis, Escallonia sp., Myrtus communis “Tarentina”, Pistacia lentiscus, Rosmarinus sp. | 7, 10, 15, 16 | R | 6 | Cat. |
| NIE | Arbutus unedo | 24 | S | 1 | Ext. |
| PAL | Cupressus sempervirens, Pistacia lentiscus | 7, 13, 16 | R, W | 3 | Cat. |
| PLU | Chamaecyparis lawsoniana “Elwoodii”, Cupressus sempervirens, Juniperus chinensis “Expansa”, Q. faginea, Q. ilex | 2, 4, 9, 10, 12, 16 | R, S, W | 14 | Cat., Com. Val. |
| PSC | Chamaecyparis lawsoniana “Elwoodii”, Q. ilex, Yucca rostrata | 2, 8, 10, 12,15, 23 | R, S, W | 17 | Cat., Com. Val., Ext. |
| SAN | Quercus ilex | 2 | R, S | 3 | Com. Val. |
| TRO | Arbutus unedo, Juniperus communis “Hibernica” | 13, 23 | R | 2 | Cat., Ext. |
| SP. 1 | Citrus sinensis | 16 | R | 3 | Cat. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mora-Sala, B.; León, M.; Pérez-Sierra, A.; Abad-Campos, P. New Reports of Phytophthora Species in Plant Nurseries in Spain. Pathogens 2022, 11, 826. https://doi.org/10.3390/pathogens11080826
Mora-Sala B, León M, Pérez-Sierra A, Abad-Campos P. New Reports of Phytophthora Species in Plant Nurseries in Spain. Pathogens. 2022; 11(8):826. https://doi.org/10.3390/pathogens11080826
Chicago/Turabian StyleMora-Sala, Beatriz, Maela León, Ana Pérez-Sierra, and Paloma Abad-Campos. 2022. "New Reports of Phytophthora Species in Plant Nurseries in Spain" Pathogens 11, no. 8: 826. https://doi.org/10.3390/pathogens11080826
APA StyleMora-Sala, B., León, M., Pérez-Sierra, A., & Abad-Campos, P. (2022). New Reports of Phytophthora Species in Plant Nurseries in Spain. Pathogens, 11(8), 826. https://doi.org/10.3390/pathogens11080826







