Borrelia burgdorferi Migration Assays for Evaluation of Chemoattractants in Tick Saliva
Abstract
1. Introduction
2. Results
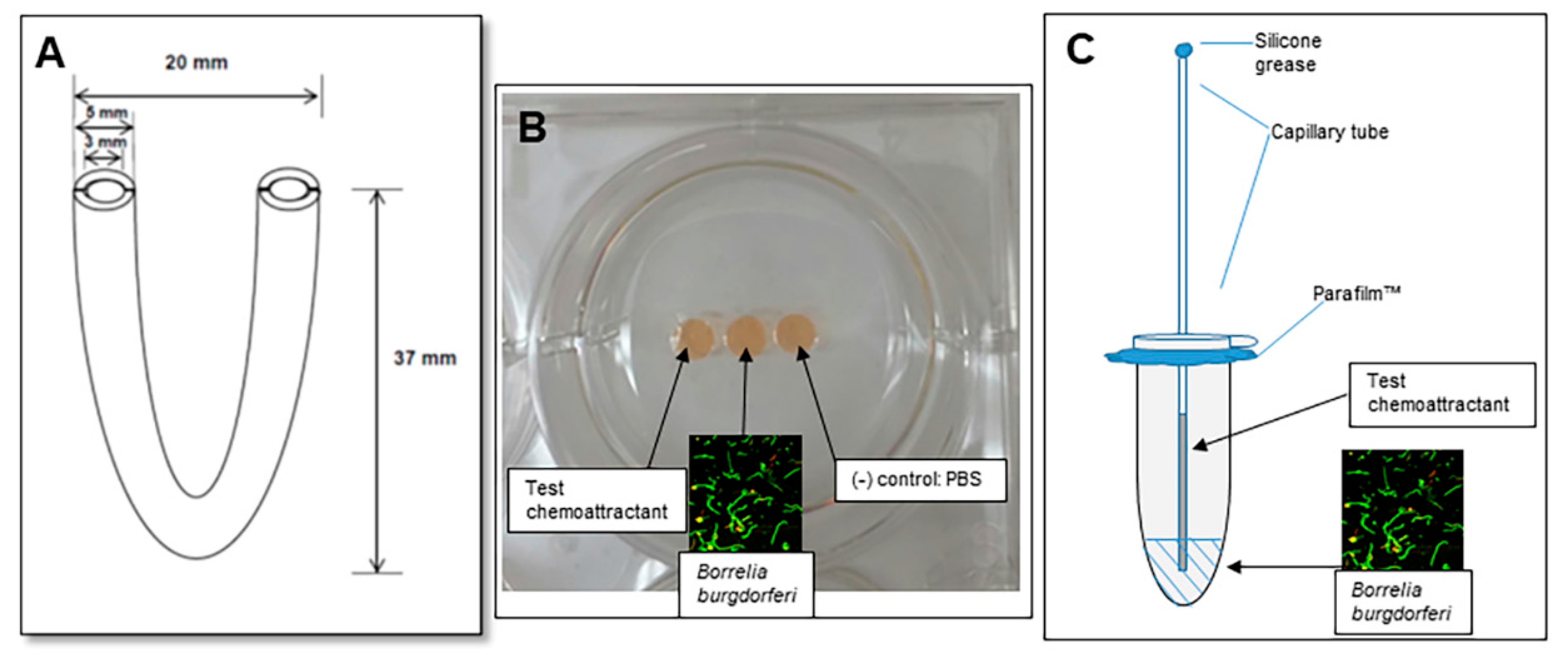
2.1. Assessment of Motility Using the U-Tube
2.2. The Gelatin Well Assay of Spirochete Migration
2.3. The Capillary Tube Migration Assay
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Bacterial Strain, Media and Growth Conditions
4.3. Chemoattractants and Matrices
4.4. Saliva Collection
4.5. Chemotaxis Assays
4.5.1. U-Tube Assay
4.5.2. Gelatin Well Assay
4.5.3. Capillary Tube Assay
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Experimental Model and Subject Details
Appendix A.2. Method Details
Bacterial Strain, Media and Growth Conditions
Appendix A.3. Quantification and Statistical Analyses
Appendix A.4. Key Resources Table
| Reagent or Resource | Source | Identifier |
| Bacterial and Virus Strains | ||
| Borrelia burgdorferi, strain B31.5A19 | S. Norris | Purser, J. E. and S. J. Norris (2000). “Correlation between plasmid content and infectivity in Borrelia burgdorferi.” Proc. Natl. Acad. Sci. U.S.A. 97. |
| Biological Samples | ||
| Ixodes scapularis saliva | B. Grasperge | Produced in-house |
| Chemicals, Peptides, and Recombinant Proteins | ||
| N-acetyl-D-glucosamine (D-GlcNAc) | Sigma chemical | A3286 |
| N,N’-Diacetylchitobiose (chitobiose) | Sigma chemical | D1523 |
| Bovine Serum Albumin (Probumin) | Millipore | 82047 |
| Type A gelatin from porcine skin | Sigma chemical | G2500 |
| Experimental Models: Organisms/Strains | ||
| Ixodes scapularis ticks, adult, female | BEI resources | NR-42510 |
| Ixodes scapularis ticks, adult, female | Wild caught | Covington, LA, USA |
| CD IGS Albino Rats, female, six to eight weeks old | Charles River | Strain Code: 001 |
| New Zealand White Rabbits | Charles River | Strain Code: 052 |
References
- Steere, A.C.; Coburn, J.; Glickstein, L. The Emergence of Lyme Disease. J. Clin. Investig. 2004, 113, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Fallon, B.A.; Levin, E.S.; Schweitzer, P.J.; Hardesty, D. Inflammation and Central Nervous System Lyme Disease. Neurobiol. Dis. 2010, 37, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.C.; Sikand, V.K. The Presenting Manifestations of Lyme Disease and the Outcomes of Treatment. N. Engl. J. Med. 2003, 348, 2472–2474. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.; Baranchuk, A. Diagnosis and Treatment of Lyme Carditis: Jacc Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 73, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Aucott, J.N. Posttreatment Lyme Disease Syndrome. Infect. Dis. Clin. N. Am. 2015, 29, 309–323. [Google Scholar] [CrossRef]
- Logigian, E.L.; Kaplan, R.F.; Steere, A.C. Chronic Neurologic Manifestations of Lyme Disease. N. Engl. J. Med. 1990, 323, 1438–1444. [Google Scholar] [CrossRef]
- Shadick, N.A.; Phillips, C.B.; Logigian, E.L.; Steere, A.C.; Kaplan, R.F.; Berardi, V.P.; Duray, P.H.; Larson, M.G.; Wright, E.A.; Ginsburg, K.S.; et al. The Long-Term Clinical Outcomes of Lyme Disease. A Population-Based Retrospective Cohort Study. Ann. Intern. Med. 1994, 121, 560–567. [Google Scholar] [CrossRef]
- Rebman, A.W.; Aucott, J.N. Post-Treatment Lyme Disease as a Model for Persistent Symptoms in Lyme Disease. Front. Med. 2020, 7, 57. [Google Scholar] [CrossRef]
- Embers, M.E.; Barthold, S.W.; Borda, J.T.; Bowers, L.; Doyle, L.; Hodzic, E.; Jacobs, M.B.; Hasenkampf, N.R.; Martin, D.S.; Narasimhan, S.; et al. Persistence of Borrelia burgdorferi in Rhesus Macaques Following Antibiotic Treatment of Disseminated Infection. PLoS ONE 2012, 7, e29914. [Google Scholar] [CrossRef]
- Ferreira, A.W.; Ávila, S. Laboratory Diagnosis of Chagas’ Heart Disease. Sao Paulo Med. J. 1995, 113, 767–771. [Google Scholar] [CrossRef]
- Hovius, J.W.; van Dam, A.P.; Fikrig, E. Tick-Host-Pathogen Interactions in Lyme Borreliosis. Trends Parasitol. 2007, 23, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Pal, U.; Fikrig, E. Adaptation of Borrelia burgdorferi in the Vector and Vertebrate Host. Microbes Infect. 2003, 5, 659–666. [Google Scholar] [CrossRef]
- Charon, N.W.; Goldstein, S.F. Genetics of Motility and Chemotaxis of a Fascinating Group of Bacteria: The Spirochetes. Ann. Rev. Genet. 2002, 36, 47–73. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.E.; Patricia, A.R. Physiologic and Genetic Factors Influencing the Zoonotic Cycle of Borrelia burgdorferi. In Spirochete Biology: The Post Genomic Era; Adler, B., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 63–82. [Google Scholar]
- Akins, D.R.; Bourell, K.W.; Caimano, M.J.; Norgard, M.V.; Radolf, J.D. A New Animal Model for Studying Lyme Disease Spirochetes in a Mammalian Host-Adapted State. J. Clin. Investig. 1998, 101, 2240–2250. [Google Scholar] [CrossRef]
- Shih, C.M.; Chao, L.L.; Yu, C.P. Chemotactic Migration of the Lyme Disease Spirochete (Borrelia burgdorferi) to Salivary Gland Extracts of Vector Ticks. Am. J. Trop. Med. Hyg. 2002, 66, 616–621. [Google Scholar] [CrossRef]
- Shi, W.; Yang, Z.; Geng, Y.; Wolinsky, L.E.; Lovett, M.A. Chemotaxis in Borrelia burgdorferi. J. Bacterial. 1998, 180, 231–235. [Google Scholar] [CrossRef]
- Bakker, R.G.; Li, C.; Miller, M.R.; Cunningham, C.; Charon, N.W. Identification of Specific Chemoattractants and Genetic Complementation of a Borrelia burgdorferi Chemotaxis Mutant: Flow Cytometry-Based Capillary Tube Chemotaxis Assay. Appl. Environ. Microbiol. 2007, 73, 1180–1188. [Google Scholar] [CrossRef]
- Murfin, K.E.; Kleinbard, R.; Aydin, M.; Salazar, S.A.; Fikrig, E. Borrelia burgdorferi Chemotaxis toward Tick Protein Salp12 Contributes to Acquisition. Tick. Tick Borne Dis. 2019, 10, 1124–1134. [Google Scholar] [CrossRef]
- Van Gundy, T.J.; Ullmann, A.J.; Brandt, K.S.; Gilmore, R.D. A Transwell Assay Method to Evaluate Borrelia burgdorferi Sensu Stricto Migratory Chemoattraction toward Tick Saliva Proteins. Tick. Tick Borne Dis. 2021, 12, 101782. [Google Scholar] [CrossRef]
- Marques, A.; Telford, S.R., III; Turk, S.P.; Chung, E.; Williams, C.; Dardick, K.; Krause, P.J.; Brandeburg, C.; Crowder, C.D.; Carolan, H.E.; et al. Xenodiagnosis to Detect Borrelia burgdorferi Infection: A First-in-Human Study. Clin. Infect. Dis. 2014, 58, 937–945. [Google Scholar] [CrossRef]
- Liveris, D.; Wang, G.; Girao, G.; Byrne, D.W.; Nowakowski, J.; McKenna, D.; Nadelman, R.; Wormser, G.P.; Schwartz, I. Quantitative Detection of Borrelia burgdorferi in 2-Millimeter Skin Samples of Erythema Migrans Lesions: Correlation of Results with Clinical and Laboratory Findings. J. Clin. Microbiol. 2002, 40, 1249–1253. [Google Scholar] [CrossRef] [PubMed]
- Embers, M.E.; Narasimhan, S. Vaccination against Lyme Disease: Past, Present and Future. Front. Cell. Infect. Microbiol. 2013, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, M.B.; Purcell, J.E.; Philipp, M.T. Ixodes Scapularis Ticks (Acari: Ixodidae) from Louisiana Are Competent to Transmit Borrelia burgdorferi, the Agent of Lyme Borreliosis. J. Med. Entomol. 2003, 40, 964–967. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Caimano, M.J. Cultivation of Borrelia burgdorferi in Dialysis Membrane Chambers in Rat Peritonea. Curr. Protoc. Microbiol. 2005, 12, 12C-3. [Google Scholar] [CrossRef] [PubMed]
- Patton, T.G.; Dietrich, G.; Brandt, K.; Dolan, M.C.; Piesman, J.; Gilmore, R.D., Jr. Saliva, Salivary Gland, and Hemolymph Collection from Ixodes Scapularis Ticks. J. Vis. Exp. 2012, 60, e3894. [Google Scholar] [CrossRef]
- Mousseau, Y.; Leclers, D.; Faucher-Durand, K.; Cook-Moreau, J.; Lia-Baldini, A.S.; Rigaud, M.; Sturtz, F.G. Improved Agarose Gel Assay for Quantification of Growth Factor-Induced Cell Motility. Biotechniques 2007, 43, 509–516. [Google Scholar] [CrossRef]
- Adler, J. A Method for Measuring Chemotaxis and Use of the Method to Determine Optimum Conditions for Chemotaxis by Escherichia coli. J. Gen. Microbiol. 1973, 74, 77–91. [Google Scholar] [CrossRef]
- Adler, J. Chemotaxis in Bacteria. Annu. Rev. Biochem. 1966, 153, 708–716. [Google Scholar] [CrossRef]
- Fraser, C.M.; Casjens, S.; Huang, W.M.; Sutton, G.G.; Clayton, R.; Lathigra, R.; White, O.; Ketchum, K.A.; Dodson, R.; Hickey, E.K.; et al. Genomic Sequence of a Lyme Disease Spirochaete, Borrelia burgdorferi. Nature 1997, 390, 580–586. [Google Scholar] [CrossRef]
- Purser, J.E.; Norris, S.J. Correlation between Plasmid Content and Infectivity in Borrelia burgdorferi. Proc. Natl. Acad. Sci. USA 2000, 97, 13865–13870. [Google Scholar] [CrossRef]
- Zeidner, N.S.; Schneider, B.S.; Dolan, M.C.; Piesman, J. An Analysis of Spirochete Load, Strain, and Pathology in a Model of Tick-Transmitted Lyme Borreliosis. Vector Borne Zoonotic Dis. 2001, 1, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.; Kalu, O.; Purser, J.; Norris, S.; Stevenson, B.; Schwartz, I. Linear and Circular Plasmid Content in Borrelia burgdorferi Clinical Isolates. Infect. Immun. 2003, 71, 3699–3706. [Google Scholar] [CrossRef] [PubMed]
- Schwan, T.G.; Burgdorfer, W.; Garon, C.F. Changes in Infectivity and Plasmid Profile of the Lyme Disease Spirochete, Borrelia burgdorferi, as a Result of in vitro Cultivation. Infect. Immun. 1988, 56, 1831–1836. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.G. Isolation and Cultivation of Lyme Disease Spirochetes. Yale J. Biol. Med. 1984, 57, 521–525. [Google Scholar]
| Matrix: Gelatin (%) | Well Diameter (mm) | Well Depth (mm) | Distance between Wells (mm) | Cells | [Cell] (×107/mL) | Lateral Well Inocula | [CA] | Volume per Well (µL) | Incubation Time (Hours) | Final Cell Count * | Ratio (CA:Control) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 | 6 | 2 | 4 | in vitro Bb | 8 | chitobiose | 10 mM | 35 | 21 | 0.45/field | |
| PBS | 0 | ||||||||||
| 3 | 6 | 2 | 4 | in vitro Bb | 8 | chitobiose | 10 mM | 35 | 21 | 0.9/field | (-) |
| BSK-II | 1/field | ||||||||||
| 5 | 6 | 2 | 2 | in vitro Bb | 8 | chitobiose | 10 mM | 35 | 21 | 1.5/field | |
| PBS | 0 | ||||||||||
| 5 | 6 | 2 | 2 | in vitro Bb | 8 | chitobiose | 10 mM | 35 | 21 | 14/cs | 7:1 |
| BSK-II | 2/cs | ||||||||||
| 5 | 6 | 2 | 2 | in vitro Bb | 8 | tick saliva | 5% | 40 | 24 | 1/cs | 1:1 |
| PBS | 1/cs | ||||||||||
| 5 | 6 | 2 | 2 | in vitro Bb | 4.6 | chitobiose | 10 mM | 40 | 21 | 1/cs | 1:1 |
| PBS | 1/cs | ||||||||||
| 5 | 6 | 2 | 2 | in vitro Bb | 4.6 | tick saliva | 5% | 40 | 21 | 3/cs | (-) |
| PBS | 8/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 40 | 26 | 20/cs | 10:1 |
| PBS | 2/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | tick saliva | 9% | 45 | 27 | 2/cs | 1:1 |
| PBS | 2/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 45 | 27 | 24/cs | 3.5:1 |
| PBS | 7/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 45 | 27 | 25/cs | 13:1 |
| PBS | 2/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | D-GlcNAc | 100 mM | 45 | 26 | 64/cs | 4:1 |
| PBS | 16/cs | ||||||||||
| 5 | 6 | 2 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 45 | 28 | 62/cs | 1.6:1 |
| PBS | 40/cs | ||||||||||
| 5 | 6 | 4 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 46 | 27 | 27/cs | 2.5:1 |
| PBS | 11/cs | ||||||||||
| 4 | 6 | 2 | 1 | in vitro Bb | 7 | chitobiose | 10 mM | 40 | 23 | 37/cs | 5.3:1 |
| 4 | 4 | 3 | 1 | in vitro Bb | 7 | D-GlcNAc | 100 mM | 30 | 22 | 57/cs | 3.2:1 |
| PBS | 18/cs | ||||||||||
| 5 | 4 | 3 | 1 | in vitro Bb | 7 | D-GlcNAc | 100 mM | 30 | 22 | 21/cs | 7:1 |
| PBS | 3/cs | ||||||||||
| 4 | 4 | 3 | 1 | Host-adapted Bb | 2 | chitobiose | 10 mM | 30 | 22 | 3/cs | |
| PBS | 0 | ||||||||||
| 4 | 4 | 3 | 1 | Host-adapted Bb | 2 | chitobiose | 10 mM | 30 | 22 | 3/cs | |
| PBS | 1/cs | ||||||||||
| 4 | 4 | 3 | 1 | Host-adapted Bb | 2 | tick saliva | 5% | 30 | 27 | 15/cs | 7.5:1 |
| PBS | 2/cs | ||||||||||
| 4 | 4 | 3 | 1 | Host-adapted Bb | 2 | tick saliva | 10% | 30 | 28 | 17/cs | 17:1 |
| PBS | 1/cs | ||||||||||
| 4 | 4 | 3 | 1 | Host-adapted Bb | 2 | D-GlcNAc | 100 mM | 30 | 26 | 3/cs | 1:1 |
| Matrix | Cells | Cap Tube Contents: CA or Control | [CA] | Cap Tube Volume (µL) | No. Replicates | Incubation Environment | Incubation Time (Hours) | AVG. Cell Count per Field | Ratio (CA:Control) |
|---|---|---|---|---|---|---|---|---|---|
| Motility | Host-adapted | D-GlcNAc | 100 mM | 20 | 1 | 1.4 | 3.5:1 | ||
| Buffer | Bb | tick saliva | 10% | 20 | 3 | RT | 2 | 3.1 | 7.8:1 |
| MB buffer | 20 | 3 | 0.4 | ||||||
| Motility | Host-adapted | D-GlcNAc | 100 mM | 20 | 1 | 4.8 | 5.3:1 | ||
| Buffer | Bb | tick saliva | 10% | 20 | 4 | RT | 2 | 6.6 | 7.3:1 |
| MB buffer | 20 | 3 | 0.9 | ||||||
| Motility | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | RT | 2 | 20.8 | 14.9:1 |
| Buffer | MB buffer | 20 | 3 | 1.4 | |||||
| Motility | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | 22.3 | 9.7:1 | ||
| Buffer | tick saliva | 10% | 20 | 3 | RT | 2 | 3.9 | 1.7:1 | |
| MB buffer | 20 | 3 | 2.3 | ||||||
| Motility | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | RT | 2 | 13 | 26:1 |
| Buffer | MB buffer | 20 | 3 | 0.5 | |||||
| Motility Buffer+ | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | 34 °C trigas | 3 | 4.9 | 5.5:1 |
| 3% gelatin | MB/gelatin | 20 | 3 | 0.9 | |||||
| Motility Buffer+ | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | 34 °C trigas | 3 | 9.4 | 5.9:1 |
| 3% gelatin | tick saliva | 10% | 20 | 3 | 2.1 | 1.3:1 | |||
| MB/gelatin | 20 | 3 | 1.6 | ||||||
| Motility Buffer+ | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | 34 °C trigas | 3 | 7.4 | 5.7:1 |
| 3% gelatin | MB/gelatin | 20 | 3 | 1.3 | |||||
| Motility Buffer+ | in vitro Bb | D-GlcNAc | 100 mM | 20 | 3 | 27 | 1.5 | 1.5:1 | |
| 3% gelatin | tick saliva | 10% | 20 | 3 | RT | 1.1 | 1.1:1 | ||
| MB/gelatin | 20 | 3 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacobs, M.B.; Grasperge, B.J.; Doyle-Meyers, L.A.; Embers, M.E. Borrelia burgdorferi Migration Assays for Evaluation of Chemoattractants in Tick Saliva. Pathogens 2022, 11, 530. https://doi.org/10.3390/pathogens11050530
Jacobs MB, Grasperge BJ, Doyle-Meyers LA, Embers ME. Borrelia burgdorferi Migration Assays for Evaluation of Chemoattractants in Tick Saliva. Pathogens. 2022; 11(5):530. https://doi.org/10.3390/pathogens11050530
Chicago/Turabian StyleJacobs, Mary B., Britton J. Grasperge, Lara A. Doyle-Meyers, and Monica E. Embers. 2022. "Borrelia burgdorferi Migration Assays for Evaluation of Chemoattractants in Tick Saliva" Pathogens 11, no. 5: 530. https://doi.org/10.3390/pathogens11050530
APA StyleJacobs, M. B., Grasperge, B. J., Doyle-Meyers, L. A., & Embers, M. E. (2022). Borrelia burgdorferi Migration Assays for Evaluation of Chemoattractants in Tick Saliva. Pathogens, 11(5), 530. https://doi.org/10.3390/pathogens11050530






