Prevalence and Implications of Shiga Toxin-Producing E. coli in Farm and Wild Ruminants
Abstract
1. Introduction
2. Virulence Factors Associated with STEC
2.1. Virulence Factors Important for Adherence and Colonization
2.2. Shiga Toxins Important for Pathogenesis
2.3. Other Toxigenic Virulence Factors
2.4. Biofilms
3. Major Seropathotypes of STEC and Its Constituents
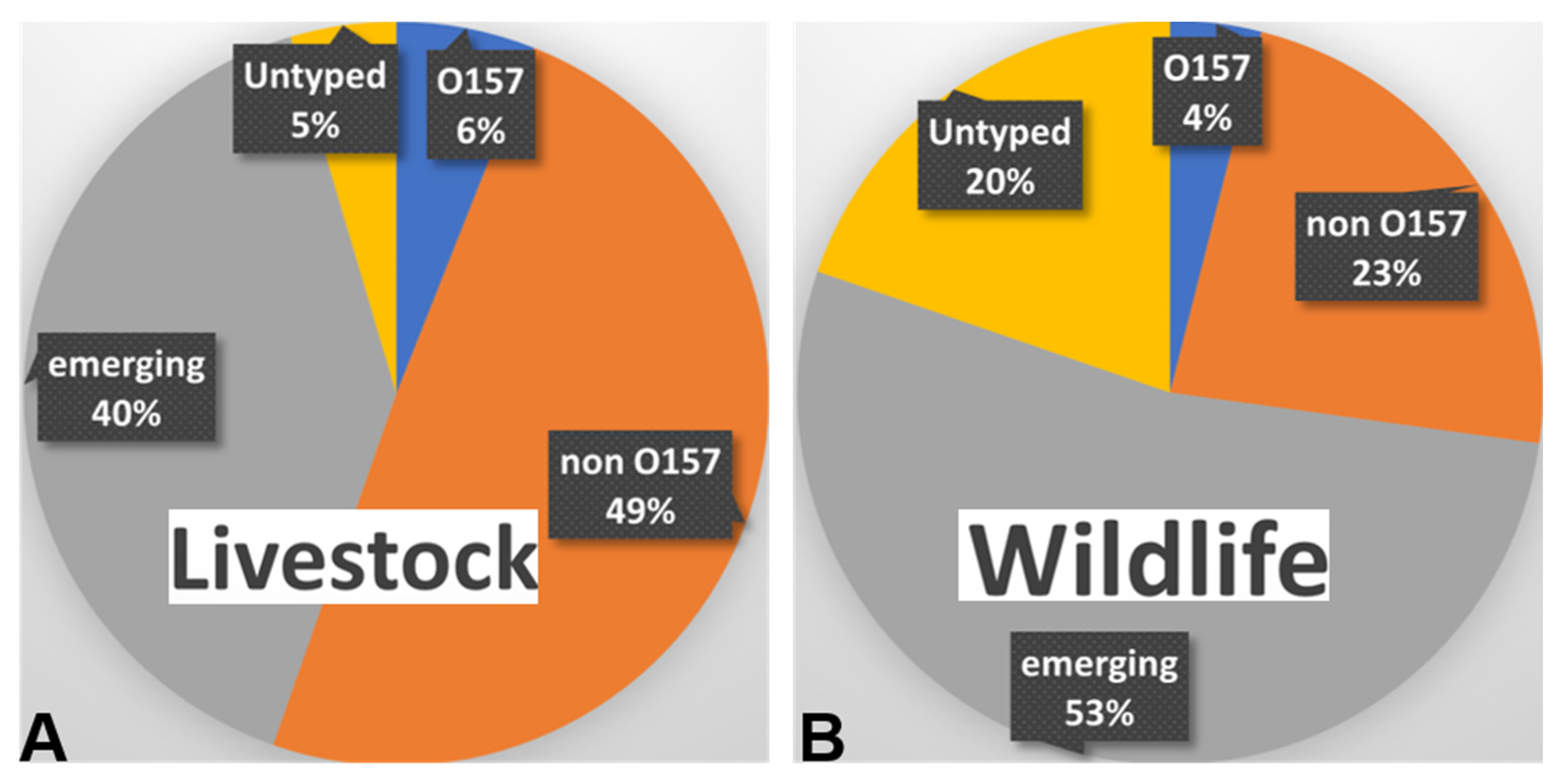
3.1. Serotypes of STEC prevalent in Livestock
3.2. Serotypes of STEC in Wild Animals
3.3. Serotypes of STEC in Environment
4. Emerging STEC Serotypes
4.1. Wildlife as Food Choices (Game Meat)
4.2. Interspecies Transmission
4.3. Rise in Antibiotic-Resistant Strains
5. Control of STEC Transmission
5.1. Transmission Control in Livestock
5.2. Control of Transmission to Wildlife
5.3. Transmission Control in Food Processing Facilities
6. Role of the Intestinal Microbiome in Colonization and Infections
6.1. Intestinal Microbial Communities of Reservoir Animals
6.2. Perturbed Intestinal Microbiome Leads to STEC Infection in Humans
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Majowicz, S.E.; Scallan, E.; Jones-Bitton, A.; Sargeant, J.M.; Stapleton, J.; Angulo, F.J.; Yeung, D.H.; Kirk, M.D. Global incidence of human Shiga toxin–producing Escherichia coli infections and deaths: A systematic review and knowledge synthesis. Foodborne Pathog. Dis. 2014, 11, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Kaper, J.B.; Nataro, J.P.; Mobley, H.L. Pathogenic Escherichia coli. Nat. Rev. Microbiol. 2004, 2, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, S.; Batz, M.B.; Morris Jr, J.G. Annual cost of illness and quality-adjusted life year losses in the United States due to 14 foodborne pathogens. J. Food Prot. 2012, 75, 1292–1302. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Meng, J.; McDermott, P.F.; Ayers, S.; Blickenstaff, K.; Tran, T.-T.; Abbott, J.; Zheng, J.; Zhao, S. Presence and characterization of Shiga toxin-producing Escherichia coli and other potentially diarrheagenic E. coli strains in retail meats. Appl. Environ. Microbiol. 2010, 76, 1709–1717. [Google Scholar] [CrossRef] [PubMed]
- Tarr, P.I. Shiga toxin-associated hemolytic uremic syndrome and thrombotic thrombocytopenic purpura: Distinct mechanisms of pathogenesis. Kidney Int. 2009, 75, S29–S32. [Google Scholar] [CrossRef]
- Crump, J.A.; Sulka, A.C.; Langer, A.J.; Schaben, C.; Crielly, A.S.; Gage, R.; Baysinger, M.; Moll, M.; Withers, G.; Toney, D.M. An outbreak of Escherichia coli O157: H7 infections among visitors to a dairy farm. N. Engl. J. Med. 2002, 347, 555–560. [Google Scholar] [CrossRef]
- Rivas, M.; Sosa-Estani, S.; Rangel, J.; Caletti, M.G.; Vallés, P.; Roldán, C.D.; Balbi, L.; de Mollar, M.C.M.; Amoedo, D.; Miliwebsky, E. Risk factors for sporadic Shiga toxin–producing Escherichia coli infections in children, Argentina. Emerg. Infect. Dis. 2008, 14, 763. [Google Scholar] [CrossRef]
- Kintz, E.; Brainard, J.; Hooper, L.; Hunter, P. Transmission pathways for sporadic Shiga-toxin producing E. coli infections: A systematic review and meta-analysis. Int. J. Hyg. Environ. Health 2017, 220, 57–67. [Google Scholar] [CrossRef]
- Stritt, A.; Tschumi, S.; Kottanattu, L.; Bucher, B.S.; Steinmann, M.; von Steiger, N.; Stephan, R.; Hächler, H.; Simonetti, G.D. Neonatal hemolytic uremic syndrome after mother-to-child transmission of a low-pathogenic stx 2b harboring Shiga toxin–producing Escherichia coli. Clin. Infect. Dis. 2013, 56, 114–116. [Google Scholar] [CrossRef]
- Heiman, K.E.; Mody, R.K.; Johnson, S.D.; Griffin, P.M.; Gould, L.H. Escherichia coli O157 outbreaks in the United States, 2003–2012. Emerg. Infect. Dis. 2015, 21, 1293. [Google Scholar] [CrossRef]
- Karmali, M.A.; Mascarenhas, M.; Shen, S.; Ziebell, K.; Johnson, S.; Reid-Smith, R.; Isaac-Renton, J.; Clark, C.; Rahn, K.; Kaper, J.B. Association of Genomic O Island 122 of Escherichia coli EDL 933 with Verocytotoxin-Producing Escherichia coli Seropathotypes That Are Linked to Epidemic and/or Serious Disease. J. Clin. Microbiol. 2003, 41, 4930–4940. [Google Scholar] [CrossRef]
- Espinosa, L.; Gray, A.; Duffy, G.; Fanning, S.; McMahon, B.J. A scoping review on the prevalence of Shiga-toxigenic Escherichia coli in wild animal species. Zoonoses Public Health 2018, 65, 911–920. [Google Scholar] [CrossRef]
- Marder, E.P.; Cieslak, P.R.; Cronquist, A.B.; Dunn, J.; Lathrop, S.; Rabatsky-Ehr, T.; Ryan, P.; Smith, K.; Tobin-D’Angelo, M.; Vugia, D.J. Incidence and trends of infections with pathogens transmitted commonly through food and the effect of increasing use of culture-independent diagnostic tests on surveillance—Foodborne Diseases Active Surveillance Network, 10 US sites, 2013–2016. Morb. Mortal. Wkly. Rep. 2017, 66, 397. [Google Scholar] [CrossRef] [PubMed]
- Besser, T.; Richards, B.; Rice, D.; Hancock, D. Escherichia coli O157 [ratio] H7 infection of calves: Infectious dose and direct contact transmission. Epidemiol. Infect. 2001, 127, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Mercado, E.; Gioffré, A.; Rodríguez, S.; Cataldi, A.; Irino, K.; Elizondo, A.; Cipolla, A.L.; Romano, M.I.; Malena, R.; Mendez, M. Non-O157 Shiga toxin-producing Escherichia coli isolated from diarrhoeic calves in Argentina. J. Vet. Med. Ser. B 2004, 51, 82–88. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, USDA. FSIS Issues Public Health Alert for Ground Beef Products Due to Possible E. coli O157:H7 Contamina-tion. Available online: https://www.fsis.usda.gov/recalls-alerts/fsis-issues-public-health-alert-ground-beef-products-due-possible-e--coli-o157h7 (accessed on 29 October 2022).
- US Department of Agriculture, USDA. Summary of Recall Cases in Calendar Year 2021. Available online: https://www.fsis.usda.gov/food-safety/recalls-public-health-alerts/annual-recall-summaries/summary-recall-cases-calendar-8 (accessed on 10 September 2022).
- US Department of Agriculture, USDA. Lakeside Refrigerated Services Recalls Ground Beef Products Due to Possible E. coli O103 Con-tamination. Available online: https://www.fsis.usda.gov/recalls-alerts/lakeside-refrigerated-services-recalls-ground-beef-products-due-possible-e--coli (accessed on 25 July 2022).
- US Department of Agriculture, USDA. FSIS Issues Public Health Alert for Specific Ground Beef in HelloFresh Meal Kits Due to Possible E. coli O157:H7 Contamination. Available online: https://www.fsis.usda.gov/recalls-alerts/fsis-issues-public-health-alert-specific-ground-beef-hellofresh-meal-kits-due (accessed on 29 October 2022).
- Cornick, N.A.; Matise, I.; Samuel, J.E.; Bosworth, B.T.; Moon, H.W. Shiga toxin-producing Escherichia coli infection: Temporal and quantitative relationships among colonization, toxin production, and systemic disease. J. Infect. Dis. 2000, 181, 242–251. [Google Scholar] [CrossRef]
- Tabaran, F.; Tabaran, A. Edema disease of swine: A review of the pathogenesis. Porc. Res. 2019, 9, 7–14. [Google Scholar]
- Jones, B.A.; Grace, D.; Kock, R.; Alonso, S.; Rushton, J.; Said, M.Y.; McKeever, D.; Mutua, F.; Young, J.; McDermott, J.; et al. Zoonosis emergence linked to agricultural intensification and environmental change. Proc. Natl. Acad. Sci. USA 2013, 110, 8399–8404. [Google Scholar] [CrossRef]
- Shafiq, M.; Huang, J.; Ur Rahman, S.; Shah, J.M.; Chen, L.; Gao, Y.; Wang, M.; Wang, L. High incidence of multidrug-resistant Escherichia coli coharboring mcr-1 and bla (CTX-M-15) recovered from pigs. Infect. Drug Resist 2019, 12, 2135–2149. [Google Scholar] [CrossRef]
- Bertelloni, F.; Cagnoli, G.; Biagini, F.; Poli, A.; Bibbiani, C.; Ebani, V.V. Virulence Genes of Pathogenic Escherichia coli in Wild Red Foxes (Vulpes vulpes). Animals 2022, 12, 1959. [Google Scholar] [CrossRef]
- Munns, K.D.; Selinger, L.B.; Stanford, K.; Guan, L.; Callaway, T.R.; McAllister, T.A. Perspectives on super-shedding of Escherichia coli O157: H7 by cattle. Foodborne Pathog. Dis. 2015, 12, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Matthews, L.; Low, J.; Gally, D.; Pearce, M.; Mellor, D.; Heesterbeek, J.; Chase-Topping, M.; Naylor, S.; Shaw, D.; Reid, S. Heterogeneous shedding of Escherichia coli O157 in cattle and its implications for control. Proc. Natl. Acad. Sci. USA 2006, 103, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Blankenship, H.M.; Carbonell, S.; Mosci, R.E.; McWilliams, K.; Pietrzen, K.; Benko, S.; Gatesy, T.; Grooms, D.; Manning, S.D. Genetic and phenotypic factors associated with persistent shedding of Shiga toxin-producing Escherichia coli by beef cattle. Appl. Environ. Microbiol. 2020, 86, e01292-20. [Google Scholar] [CrossRef]
- Bumunang, E.W.; McAllister, T.A.; Zaheer, R.; Ortega Polo, R.; Stanford, K.; King, R.; Niu, Y.D.; Ateba, C.N. Characterization of non-O157 Escherichia coli from cattle faecal samples in the North-West Province of South Africa. Microorganisms 2019, 7, 272. [Google Scholar] [CrossRef]
- Murphy, R.; Palm, M.; Mustonen, V.; Warringer, J.; Farewell, A.; Parts, L.; Moradigaravand, D. Genomic epidemiology and evolution of Escherichia coli in wild animals in Mexico. Msphere 2021, 6, e00738-20. [Google Scholar] [CrossRef] [PubMed]
- Yar, A.; Choudary, M.A.; Rehman, A.; Hussain, A.; Elahi, A.; ur Rehman, F.; Waqar, A.B.; Alshammari, A.; Alharbi, M.; Nisar, M.A. Genetic Diversity and Virulence Profiling of Multi-Drug Resistant Escherichia coli of Human, Animal, and Environmental Origins. Antibiotics 2022, 11, 1061. [Google Scholar] [CrossRef]
- One Health High-Level Expert, P.; Adisasmito, W.B.; Almuhairi, S.; Behravesh, C.B.; Bilivogui, P.; Bukachi, S.A.; Casas, N.; Cediel Becerra, N.; Charron, D.F.; Chaudhary, A.; et al. One Health: A new definition for a sustainable and healthy future. PLoS Pathog. 2022, 18, e1010537. [Google Scholar] [CrossRef]
- Glaize, A.; Gutierrez-Rodriguez, E.; Hanning, I.; Díaz-Sánchez, S.; Gunter, C.; van Vliet, A.H.M.; Watson, W.; Thakur, S. Transmission of antimicrobial resistant non-O157 Escherichia coli at the interface of animal-fresh produce in sustainable farming environments. Int. J. Food Microbiol. 2020, 319, 108472. [Google Scholar] [CrossRef]
- Paton, J.C.; Paton, A.W. Pathogenesis and diagnosis of Shiga toxin-producing Escherichia coli infections. Clin. Microbiol. Rev. 1998, 11, 450–479. [Google Scholar] [CrossRef]
- Etcheverria, A.I.; Padola, N.L. Shiga toxin-producing Escherichia coli: Factors involved in virulence and cattle colonization. Virulence 2013, 4, 366–372. [Google Scholar] [CrossRef]
- Mahanti, A.; Samanta, I.; Bandyopadhyay, S.; Joardar, S. Molecular characterization and antibiotic susceptibility pattern of caprine Shiga toxin producing-Escherichia coli (STEC) isolates from India. Iran. J. Vet. Res. 2015, 16, 31. [Google Scholar] [PubMed]
- Wong, A.R.C.; Pearson, J.S.; Bright, M.D.; Munera, D.; Robinson, K.S.; Lee, S.F.; Frankel, G.; Hartland, E.L. Enteropathogenic and enterohaemorrhagic Escherichia coli: Even more subversive elements. Mol. Microbiol. 2011, 80, 1420–1438. [Google Scholar] [CrossRef] [PubMed]
- Montero, D.A.; Velasco, J.; Del Canto, F.; Puente, J.L.; Padola, N.L.; Rasko, D.A.; Farfán, M.; Salazar, J.C.; Vidal, R. Locus of adhesion and autoaggregation (LAA), a pathogenicity island present in emerging Shiga toxin–producing Escherichia coli strains. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Colello, R.; Vélez, M.V.; González, J.; Montero, D.A.; Bustamante, A.V.; Del Canto, F.; Etcheverría, A.I.; Vidal, R.; Padola, N.L. First report of the distribution of Locus of Adhesion and Autoaggregation (LAA) pathogenicity island in LEE-negative Shiga toxin-producing Escherichia coli isolates from Argentina. Microb. Pathog. 2018, 123, 259–263. [Google Scholar] [CrossRef]
- Nagy, A.; Xu, Y.; Bauchan, G.R.; Shelton, D.R.; Nou, X. Aggregative adherence fimbriae I (AAF/I) mediate colonization of fresh produce and abiotic surface by Shiga toxigenic enteroaggregative Escherichia coli O104:H4. Int. J. Food Microbiol. 2016, 229, 44–51. [Google Scholar] [CrossRef]
- Boisen, N.; Melton-Celsa, A.R.; Scheutz, F.; O’Brien, A.D.; Nataro, J.P. Shiga toxin 2a and enteroaggregative Escherichia coli–a deadly combination. Gut Microbes 2015, 6, 272–278. [Google Scholar] [CrossRef]
- Paton, A.W.; Srimanote, P.; Woodrow, M.C.; Paton, J.C. Characterization of Saa, a novel autoagglutinating adhesin produced by locus of enterocyte effacement-negative Shiga-toxigenic Escherichia coli strains that are virulent for humans. Infect. Immun. 2001, 69, 6999–7009. [Google Scholar] [CrossRef]
- Lorenz, S.C.; Son, I.; Maounounen-Laasri, A.; Lin, A.; Fischer, M.; Kase, J.A. Prevalence of hemolysin genes and comparison of ehxA subtype patterns in Shiga toxin-producing Escherichia coli (STEC) and non-STEC strains from clinical, food, and animal sources. Appl. Environ. Microbiol. 2013, 79, 6301–6311. [Google Scholar] [CrossRef]
- Herold, S.; Paton, J.C.; Paton, A.W. Sab, a Novel Autotransporter of Locus of Enterocyte Effacement-Negative Shiga-Toxigenic Escherichia coli O113:H21, Contributes to Adherence and Biofilm Formation. Infect. Immun. 2009, 77, 3234–3243. [Google Scholar] [CrossRef]
- Mir, R.A.; Weppelmann, T.A.; Elzo, M.; Ahn, S.; Driver, J.D.; Jeong, K.C. Colonization of Beef Cattle by Shiga Toxin-Producing Escherichia coli during the First Year of Life: A Cohort Study. PLoS ONE 2016, 11, e0148518. [Google Scholar] [CrossRef]
- Vasco, K.; Nohomovich, B.; Singh, P.; Venegas-Vargas, C.; Mosci, R.E.; Rust, S.; Bartlett, P.; Norby, B.; Grooms, D.; Zhang, L. Characterizing the cattle gut microbiome in farms with a high and low prevalence of shiga toxin producing Escherichia coli. Microorganisms 2021, 9, 1737. [Google Scholar] [CrossRef] [PubMed]
- Sapountzis, P.; Segura, A.; Desvaux, M.; Forano, E. An Overview of the Elusive Passenger in the Gastrointestinal Tract of Cattle: The Shiga Toxin Producing Escherichia coli. Microorganisms 2020, 8, 877. [Google Scholar] [CrossRef] [PubMed]
- Sandt, C.H.; Hill, C.W. Four different genes responsible for nonimmune immunoglobulin-binding activities within a single strain of Escherichia coli. Infect. Immun. 2000, 68, 2205–2214. [Google Scholar] [CrossRef]
- Stevens, M.P.; van Diemen, P.M.; Frankel, G.; Phillips, A.D.; Wallis, T.S. Efa1 influences colonization of the bovine intestine by Shiga toxin-producing Escherichia coli serotypes O5 and O111. Infect. Immun. 2002, 70, 5158–5166. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, L.; Grant, T.H.; Robins-Browne, R.M. Identification of a novel genetic locus that is required for in vitro adhesion of a clinical isolate of enterohaemorrhagic Escherichia coli to epithelial cells. Mol. Microbiol. 2000, 35, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Merkel, V.; Ohder, B.; Bielaszewska, M.; Zhang, W.; Fruth, A.; Menge, C.; Borrmann, E.; Middendorf, B.; Müthing, J.; Karch, H. Distribution and phylogeny of immunoglobulin-binding protein G in Shiga toxin-producing Escherichia coli and its association with adherence phenotypes. Infect. Immun. 2010, 78, 3625–3636. [Google Scholar] [CrossRef] [PubMed]
- Stromberg, Z.R.; Masonbrink, R.E.; Mellata, M. Transcriptomic Analysis of Shiga Toxin-Producing Escherichia coli during Initial Contact with Cattle Colonic Explants. Microorganisms 2020, 8, 1662. [Google Scholar] [CrossRef]
- Dziva, F.; Mahajan, A.; Cameron, P.; Currie, C.; McKendrick, I.J.; Wallis, T.S.; Smith, D.G.E.; Stevens, M.P. EspP, a Type V-secreted serine protease of enterohaemorrhagic Escherichia coli O157:H7, influences intestinal colonization of calves and adherence to bovine primary intestinal epithelial cells. FEMS Microbiol. Lett. 2007, 271, 258–264. [Google Scholar] [CrossRef]
- Boerlin, P.; McEwen, S.A.; Boerlin-Petzold, F.; Wilson, J.B.; Johnson, R.P.; Gyles, C.L. Associations between virulence factors of Shiga toxin-producing Escherichia coli and disease in humans. J. Clin. Microbiol. 1999, 37, 497–503. [Google Scholar] [CrossRef]
- Schmidt, H. Shiga-toxin-converting bacteriophages. Res. Microbiol. 2001, 152, 687–695. [Google Scholar] [CrossRef]
- Allison, H.E. Stx-phages: Drivers and mediators of the evolution of STEC and STEC-like pathogens. Future Med. 2007. [Google Scholar] [CrossRef] [PubMed]
- Ivarsson, M.E.; Leroux, J.C.; Castagner, B. Targeting bacterial toxins. Angew. Chem. Int. Ed. 2012, 51, 4024–4045. [Google Scholar] [CrossRef] [PubMed]
- Karch, H.; Tarr, P.I.; Bielaszewska, M. Enterohaemorrhagic Escherichia coli in human medicine. Int. J. Med. Microbiol. 2005, 295, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K. Shiga toxins. Toxicon 2001, 39, 1629–1635. [Google Scholar] [CrossRef]
- Grau-Leal, F.; Quirós, P.; Martínez-Castillo, A.; Muniesa, M. Free S higa toxin 1-encoding bacteriophages are less prevalent than S higa toxin 2 phages in extraintestinal environments. Environ. Microbiol. 2015, 17, 4790–4801. [Google Scholar] [CrossRef]
- Crespo-Medina, M.; Greaves, I.; Hunter, P.R.; Minnigh, H.; Ramírez-Toro, G. Detection of Shiga toxin-encoding genes in small community water supplies. J. Water Health 2020, 18, 937–945. [Google Scholar] [CrossRef]
- Tozzoli, R.; Grande, L.; Michelacci, V.; Ranieri, P.; Maugliani, A.; Caprioli, A.; Morabito, S. Shiga toxin-converting phages and the emergence of new pathogenic Escherichia coli: A world in motion. Front. Cell. Infect. Microbiol. 2014, 4, 80. [Google Scholar] [CrossRef]
- Scheutz, F.; Teel, L.D.; Beutin, L.; Piérard, D.; Buvens, G.; Karch, H.; Mellmann, A.; Caprioli, A.; Tozzoli, R.; Morabito, S. Multicenter evaluation of a sequence-based protocol for subtyping Shiga toxins and standardizing Stx nomenclature. J. Clin. Microbiol. 2012, 50, 2951–2963. [Google Scholar] [CrossRef]
- Fuller, C.A.; Pellino, C.A.; Flagler, M.J.; Strasser, J.E.; Weiss, A.A. Shiga toxin subtypes display dramatic differences in potency. Infect. Immun. 2011, 79, 1329–1337. [Google Scholar] [CrossRef]
- Krüger, A.; Lucchesi, P.M.A. Shiga toxins and stx phages: Highly diverse entities. Microbiology 2015, 161, 451–462. [Google Scholar] [CrossRef]
- Friesema, I.; Van Der Zwaluw, K.; Schuurman, T.; Kooistra-Smid, M.; Franz, E.; Van Duynhoven, Y.; Van Pelt, W. Emergence of Escherichia coli encoding Shiga toxin 2f in human Shiga toxin-producing E. coli (STEC) infections in the Netherlands, January 2008 to December 2011. Eurosurveillance 2014, 19, 20787. [Google Scholar] [CrossRef] [PubMed]
- Valle, C.-D.; Mariana, D.; la Garza-García, D.; Jorge, A.; Díaz-Aparicio, E.; Valdivia-Flores, A.G.; Cisneros-Guzmán, L.F.; Rosario, C.; Manjarrez-Hernández, Á.H.; Navarro, A. Characterization of Escherichia coli strains from red deer (Cervus elaphus) faeces in a Mexican protected natural area. Eur. J. Wildl. Res. 2016, 62, 415–421. [Google Scholar] [CrossRef]
- Alonso, C.A.; Mora, A.; Díaz, D.; Blanco, M.; González-Barrio, D.; Ruiz-Fons, F.; Simón, C.; Blanco, J.; Torres, C. Occurrence and characterization of stx and/or eae-positive Escherichia coli isolated from wildlife, including a typical EPEC strain from a wild boar. Vet. Microbiol. 2017, 207, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Baines, A.C.; Brodsky, R.A. Complementopathies. Blood Rev 2017, 31, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Orth, D.; Khan, A.B.; Naim, A.; Grif, K.; Brockmeyer, J.; Karch, H.; Joannidis, M.; Clark, S.J.; Day, A.J.; Fidanzi, S. Shiga toxin activates complement and binds factor H: Evidence for an active role of complement in hemolytic uremic syndrome. J. Immunol. 2009, 182, 6394–6400. [Google Scholar] [CrossRef]
- Markiewski, M.M.; Lambris, J.D. The role of complement in inflammatory diseases from behind the scenes into the spotlight. Am. J. Pathol. 2007, 171, 715–727. [Google Scholar] [CrossRef]
- Ekiri, A.B.; Landblom, D.; Doetkott, D.; Olet, S.; Shelver, W.L.; Khaitsa, M.L. Isolation and characterization of Shiga toxin–producing Escherichia coli serogroups O26, O45, O103, O111, O113, O121, O145, and O157 shed from range and feedlot cattle from postweaning to slaughter. J. Food Prot. 2014, 77, 1052–1061. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Beuchat, L.R. Biofilm Formation by Escherichia coli O157:H7 on Stainless Steel: Effect of Exopolysaccharide and Curli Production on Its Resistance to Chlorine. Appl. Environ. Microbiol. 2005, 71, 247–254. [Google Scholar] [CrossRef]
- Barnhart, M.M.; Chapman, M.R. Curli biogenesis and function. Annu. Rev. Microbiol. 2006, 60, 131. [Google Scholar] [CrossRef]
- Vidal, O.; Longin, R.; Prigent-Combaret, C.; Dorel, C.; Hooreman, M.; Lejeune, P. Isolation of an Escherichia coli K-12 mutant strain able to form biofilms on inert surfaces: Involvement of a new ompR allele that increases curli expression. J. Bacteriol. 1998, 180, 2442–2449. [Google Scholar] [CrossRef]
- Carter, M.Q.; Feng, D.; Li, H.H. Curli fimbriae confer shiga toxin-producing Escherichia coli a competitive trait in mixed biofilms. Food Microbiol. 2019, 82, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Bumunang, E.W.; Ateba, C.N.; Stanford, K.; McAllister, T.A.; Niu, Y.D. Biofilm formation by South African non-O157 Shiga toxigenic Escherichia coli on stainless steel coupons. Can. J. Microbiol. 2020, 66, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Jeter, C.; Matthysse, A.G. Characterization of the binding of diarrheagenic strains of E. coli to plant surfaces and the role of curli in the interaction of the bacteria with alfalfa sprouts. Mol. Plant-Microbe Interact. 2005, 18, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Hammar, M.r.; Arnqvist, A.; Bian, Z.; Olsén, A.; Normark, S. Expression of two csg operons is required for production of fibronectin-and congo red-binding curli polymers in Escherichia coli K-12. Mol. Microbiol. 1995, 18, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Hammar, M.; Bian, Z.; Normark, S. Nucleator-dependent intercellular assembly of adhesive curli organelles in Escherichia coli. Proc. Natl. Acad. Sci. USA 1996, 93, 6562–6566. [Google Scholar] [CrossRef]
- Ferrières, L.; Clarke, D.J. The RcsC sensor kinase is required for normal biofilm formation in Escherichia coli K-12 and controls the expression of a regulon in response to growth on a solid surface. Mol. Microbiol. 2003, 50, 1665–1682. [Google Scholar] [CrossRef] [PubMed]
- Jubelin, G.; Vianney, A.; Beloin, C.; Ghigo, J.-M.; Lazzaroni, J.-C.; Lejeune, P.; Dorel, C. CpxR/OmpR interplay regulates curli gene expression in response to osmolarity in Escherichia coli. J. Bacteriol. 2005, 187, 2038–2049. [Google Scholar] [CrossRef]
- Martin, A.; Beutin, L. Characteristics of Shiga toxin-producing Escherichia coli from meat and milk products of different origins and association with food producing animals as main contamination sources. Int. J. Food Microbiol. 2011, 146, 99–104. [Google Scholar] [CrossRef]
- Watanabe, Y.; Ozasa, K.; Mermin, J.H.; Griffin, P.M.; Masuda, K.; Imashuku, S.; Sawada, T. Factory outbreak of Escherichia coli O157: H7 infection in Japan. Emerg. Infect. Dis. 1999, 5, 424. [Google Scholar] [CrossRef]
- Hilborn, E.; Mshar, P.; Fiorentino, T.; Dembek, Z.; Barrett, T.; Howard, R.; Cartter, M. An outbreak of Escherichia coli O157 [ratio] H7 infections and haemolytic uraemic syndrome associated with consumption of unpasteurized apple cider. Epidemiol. Infect. 2000, 124, 31–36. [Google Scholar] [CrossRef]
- Fink, R.C.; Black, E.P.; Hou, Z.; Sugawara, M.; Sadowsky, M.J.; Diez-Gonzalez, F. Transcriptional Responses of Escherichia coli K-12 and O157:H7 Associated with Lettuce Leaves. Appl. Environ. Microbiol. 2012, 78, 1752–1764. [Google Scholar] [CrossRef]
- Silagyi, K.; Kim, S.-H.; Lo, Y.M.; Wei, C.-I. Production of biofilm and quorum sensing by Escherichia coli O157: H7 and its transfer from contact surfaces to meat, poultry, ready-to-eat deli, and produce products. Food Microbiol. 2009, 26, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Nesse, L.L.; Sekse, C.; Berg, K.; Johannesen, K.C.S.; Solheim, H.; Vestby, L.K.; Urdahl, A.M.; Griffiths, M.W. Potentially Pathogenic Escherichia coli Can Form a Biofilm under Conditions Relevant to the Food Production Chain. Appl. Environ. Microbiol. 2014, 80, 2042–2049. [Google Scholar] [CrossRef] [PubMed]
- Buvens, G.; Possé, B.; De Schrijver, K.; De Zutter, L.; Lauwers, S.; Piérard, D. Virulence profiling and quantification of verocytotoxin-producing Escherichia coli O145: H28 and O26: H11 isolated during an ice cream–related hemolytic uremic syndrome outbreak. Foodborne Pathog. Dis. 2011, 8, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Erickson, M.C.; Doyle, M.P. Food as a vehicle for transmission of Shiga toxin–producing Escherichia coli. J. Food Prot. 2007, 70, 2426–2449. [Google Scholar] [CrossRef]
- Lewis, K. Persister Cells. Annu. Rev. Microbiol. 2010, 64, 357–372. [Google Scholar] [CrossRef]
- Ludwig, J.B.; Shi, X.; Shridhar, P.B.; Roberts, E.L.; DebRoy, C.; Phebus, R.K.; Bai, J.; Nagaraja, T.G. Multiplex PCR Assays for the Detection of One Hundred and Thirty Seven Serogroups of Shiga Toxin-Producing Escherichia coli Associated with Cattle. Front. Cell. Infect. Microbiol. 2020, 10, 378. [Google Scholar] [CrossRef]
- Zhang, X.; Payne, M.; Kaur, S.; Lan, R. Improved Genomic Identification, Clustering, and Serotyping of Shiga Toxin-Producing Escherichia coli Using Cluster/Serotype-Specific Gene Markers. Front. Cell. Infect. Microbiol. 2022, 11, 772574. [Google Scholar] [CrossRef]
- GROUP, F.W.S.E. Hazard identification and characterization: Criteria for categorizing Shiga toxin–producing Escherichia coli on a risk basis. J. Food Prot. 2019, 82, 7–21. [Google Scholar] [CrossRef]
- Lerouge, I.; Vanderleyden, J. O-antigen structural variation: Mechanisms and possible roles in animal/plant–microbe interactions. FEMS Microbiol. Rev. 2002, 26, 17–47. [Google Scholar] [CrossRef]
- Beutin, L.; Krause, G.; Zimmermann, S.; Kaulfuss, S.; Gleier, K. Characterization of Shiga toxin-producing Escherichia coli strains isolated from human patients in Germany over a 3-year period. J. Clin. Microbiol. 2004, 42, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- DiLuzio, W.R.; Turner, L.; Mayer, M.; Garstecki, P.; Weibel, D.B.; Berg, H.C.; Whitesides, G.M. Escherichia coli swim on the right-hand side. Nature 2005, 435, 1271–1274. [Google Scholar] [CrossRef]
- Erdem, A.L.; Avelino, F.; Xicohtencatl-Cortes, J.; Girón, J.A. Host protein binding and adhesive properties of H6 and H7 flagella of attaching and effacing Escherichia coli. J. Bacteriol. 2007, 189, 7426–7435. [Google Scholar] [CrossRef] [PubMed]
- Momtaz, H.; Farzan, R.; Rahimi, E.; Safarpoor Dehkordi, F.; Souod, N. Molecular characterization of Shiga toxin-producing Escherichia coli isolated from ruminant and donkey raw milk samples and traditional dairy products in Iran. Sci. World J. 2012, 2012, 231342. [Google Scholar] [CrossRef] [PubMed]
- Perera, A.; Clarke, C.M.; Dykes, G.A.; Fegan, N. Characterization of Shiga toxigenic Escherichia coli O157 and Non-O157 isolates from ruminant feces in Malaysia. BioMed Res. Int. 2015, 2015, 382403. [Google Scholar] [CrossRef]
- Brusa, V.; Restovich, V.; Galli, L.; Teitelbaum, D.; Signorini, M.; Brasesco, H.; Londero, A.; García, D.; Padola, N.L.; Superno, V. Isolation and characterization of non-O157 Shiga toxin-producing Escherichia coli from beef carcasses, cuts and trimmings of abattoirs in Argentina. PLoS ONE 2017, 12, e0183248. [Google Scholar] [CrossRef]
- Essendoubi, S.; Stashko, N.; So, I.; Gensler, G.; Rolheiser, D.; Mainali, C. Prevalence of Shiga toxin-producing Escherichia coli (STEC) O157: H7, six non-O157 STECs, and Salmonella on beef carcasses in provincially licensed abattoirs in Alberta, Canada. Food Control 2019, 105, 226–232. [Google Scholar] [CrossRef]
- Gould, L.H.; Walsh, K.A.; Vieira, A.R.; Herman, K.; Williams, I.T.; Hall, A.J.; Cole, D. Surveillance for foodborne disease outbreaks—United States, 1998–2008. Morb. Mortal. Wkly. Rep. Surveill. Summ. 2013, 62, 1–34. [Google Scholar]
- Hussein, H.S. Prevalence and pathogenicity of Shiga toxin-producing Escherichia coli in beef cattle and their products1,2. J. Anim. Sci. 2007, 85, E63–E72. [Google Scholar] [CrossRef]
- Gökmen, M.; İlhan, Z.; Tavşanlı, H.; Önen, A.; Ektik, N.; Göçmez, E.B. Prevalence and molecular characterization of shiga toxin-producing Escherichia coli in animal source foods and green leafy vegetables. Food Sci. Technol. Int. 2022, 10820132221125104. [Google Scholar] [CrossRef]
- Bai, X.; Wang, H.; Xin, Y.; Wei, R.; Tang, X.; Zhao, A.; Sun, H.; Zhang, W.; Wang, Y.; Xu, Y.; et al. Prevalence and characteristics of Shiga toxin-producing Escherichia coli isolated from retail raw meats in China. Int. J. Food Microbiol. 2015, 200, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Nong, F.; Zhang, P.; Meng, J.; Xie, Q.; Li, Y.; Pan, Y.; Zhao, Y.; Liu, H. Characterization of Shiga-toxin producing Escherichia coli (STEC) isolated from retail raw meats in Southeast China. Food Control 2021, 126, 108061. [Google Scholar] [CrossRef]
- The European Union One Health 2019 Zoonoses Report. Efsa J. 2021, 19, e06406. [CrossRef]
- Feng, P.C.; Delannoy, S.; Lacher, D.W.; Bosilevac, J.M.; Fach, P.; Beutin, L. Shiga toxin-producing serogroup O91 Escherichia coli strains isolated from food and environmental samples. Appl. Environ. Microbiol. 2017, 83, e01231-17. [Google Scholar] [CrossRef]
- Feng, P.C.; Delannoy, S.; Lacher, D.W.; Dos Santos, L.F.; Beutin, L.; Fach, P.; Rivas, M.; Hartland, E.L.; Paton, A.W.; Guth, B.E. Genetic diversity and virulence potential of Shiga toxin-producing Escherichia coli O113: H21 strains isolated from clinical, environmental, and food sources. Appl. Environ. Microbiol. 2014, 80, 4757–4763. [Google Scholar] [CrossRef]
- Valilis, E.; Ramsey, A.; Sidiq, S.; DuPont, H.L. Non-O157 Shiga toxin-producing Escherichia coli—A poorly appreciated enteric pathogen: Systematic review. Int. J. Infect. Dis. 2018, 76, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Sokolovic, M.; Šimpraga, B.; Amšel-Zelenika, T.; Berendika, M.; Krstulović, F. Prevalence and Characterization of Shiga Toxin Producing Escherichia coli Isolated from Animal Feed in Croatia. Microorganisms 2022, 10, 1839. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, Q.; Bai, X.; Hu, B.; Jiang, D.; Jiao, H.; Lu, L.; Fan, R.; Hou, P.; Matussek, A.; et al. High Prevalence and Persistence of Escherichia coli Strains Producing Shiga Toxin Subtype 2k in Goat Herds. Microbiol. Spectr. 2022, 10, e01571-01522. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Bai, X.; Zhang, J.; Sun, H.; Fu, S.; Fan, R.; He, X.; Scheutz, F.; Matussek, A.; Xiong, Y. Escherichia coli strains producing a novel Shiga toxin 2 subtype circulate in China. Int. J. Med. Microbiol. 2020, 310, 151377. [Google Scholar] [CrossRef]
- Pinto, G.; Sampaio, M.; Dias, O.; Almeida, C.; Azeredo, J.; Oliveira, H. Insights into the genome architecture and evolution of Shiga toxin encoding bacteriophages of Escherichia coli. BMC Genom. 2021, 22, 1–13. [Google Scholar] [CrossRef]
- Rodwell, E.V.; Chan, Y.-W.; Sawyer, C.; Carroll, A.; McNamara, E.; Allison, L.; Browning, L.; Holmes, A.; Godbole, G.; McCarthy, N.; et al. Shiga toxin-producing Escherichia coli clonal complex 32, including serotype O145:H28, in the UK and Ireland. J. Med. Microbiol. 2022, 71. [Google Scholar] [CrossRef] [PubMed]
- Dewsbury, D.M.; Renter, D.G.; Shridhar, P.B.; Noll, L.W.; Shi, X.; Nagaraja, T.G.; Cernicchiaro, N. Summer and winter prevalence of Shiga toxin–producing Escherichia coli (STEC) O26, O45, O103, O111, O121, O145, and O157 in feces of feedlot cattle. Foodborne Pathog. Dis. 2015, 12, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Cull, C.A.; Renter, D.G.; Dewsbury, D.M.; Noll, L.W.; Shridhar, P.B.; Ives, S.E.; Nagaraja, T.G.; Cernicchiaro, N. Feedlot-and pen-level prevalence of enterohemorrhagic Escherichia coli in feces of commercial feedlot cattle in two major US cattle feeding areas. Foodborne Pathog. Dis. 2017, 14, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.C.; Reddy, S. Prevalences of Shiga toxin subtypes and selected other virulence factors among Shiga-toxigenic Escherichia coli strains isolated from fresh produce. Appl. Environ. Microbiol. 2013, 79, 6917–6923. [Google Scholar] [CrossRef] [PubMed]
- Fayemi, O.E.; Akanni, G.B.; Elegbeleye, J.A.; Aboaba, O.O.; Njage, P.M. Prevalence, characterization and antibiotic resistance of Shiga toxigenic Escherichia coli serogroups isolated from fresh beef and locally processed ready-to-eat meat products in Lagos, Nigeria. Int. J. Food Microbiol. 2021, 347, 109191. [Google Scholar] [CrossRef]
- Mora, A.; López, C.; Dhabi, G.; López-Beceiro, A.M.; Fidalgo, L.E.; Díaz, E.A.; Martínez-Carrasco, C.; Mamani, R.; Herrera, A.; Blanco, J.E.; et al. Seropathotypes, Phylogroups, Stx Subtypes, and Intimin Types of Wildlife-Carried, Shiga Toxin-Producing Escherichia coli Strains with the Same Characteristics as Human-Pathogenic Isolates. Appl. Environ. Microbiol. 2012, 78, 2578–2585. [Google Scholar] [CrossRef]
- Miko, A.; Pries, K.; Haby, S.; Steege, K.; Albrecht, N.; Krause, G.; Beutin, L. Assessment of Shiga toxin-producing Escherichia coli isolates from wildlife meat as potential pathogens for humans. Appl. Environ. Microbiol. 2009, 75, 6462–6470. [Google Scholar] [CrossRef]
- Hussein, M.A.; Merwad, A.M.; Elabbasy, M.T.; Suelam, I.I.; Abdelwahab, A.M.; Taha, M.A. Prevalence of Enterotoxigenic Staphylococcus aureus and Shiga Toxin Producing Escherichia coli in fish in Egypt: Quality parameters and public health hazard. Vector-Borne Zoonotic Dis. 2019, 19, 255–264. [Google Scholar] [CrossRef]
- Arthur, T.M.; Barkocy-Gallagher, G.A.; Rivera-Betancourt, M.; Koohmaraie, M. Prevalence and characterization of non-O157 Shiga toxin-producing Escherichia coli on carcasses in commercial beef cattle processing plants. Appl. Environ. Microbiol. 2002, 68, 4847–4852. [Google Scholar] [CrossRef]
- Singh, P.; Sha, Q.; Lacher, D.W.; Del Valle, J.; Mosci, R.E.; Moore, J.A.; Scribner, K.T.; Manning, S.D. Characterization of enteropathogenic and Shiga toxin-producing Escherichia coli in cattle and deer in a shared agroecosystem. Front. Cell. Infect. Microbiol. 2015, 5, 29. [Google Scholar] [CrossRef]
- Asakura, H.; Makino, S.i.; Shirahata, T.; Tsukamoto, T.; Kurazono, H.; Ikeda, T.; Takeshi, K. Detection and genetical characterization of Shiga toxin-producing Escherichia coli from wild deer. Microbiol. Immunol. 1998, 42, 815–822. [Google Scholar] [CrossRef] [PubMed]
- GM Gonzalez, A.; MF Cerqueira, A. Shiga toxin-producing Escherichia coli in the animal reservoir and food in Brazil. J. Appl. Microbiol. 2020, 128, 1568–1582. [Google Scholar] [CrossRef]
- Díaz-Sánchez, S.; Sánchez, S.; Herrera-León, S.; Porrero, C.; Blanco, J.; Dahbi, G.; Blanco, J.; Mora, A.; Mateo, R.; Hanning, I. Prevalence of Shiga toxin-producing Escherichia coli, Salmonella spp. and Campylobacter spp. in large game animals intended for consumption: Relationship with management practices and livestock influence. Vet. Microbiol. 2013, 163, 274–281. [Google Scholar] [CrossRef]
- Fakih, I.; Thiry, D.; Duprez, J.-N.; Saulmont, M.; Iguchi, A.; Piérard, D.; Jouant, L.; Daube, G.; Ogura, Y.; Hayashi, T. Identification of Shiga toxin-producing (STEC) and enteropathogenic (EPEC) Escherichia coli in diarrhoeic calves and comparative genomics of O5 bovine and human STEC. Vet. Microbiol. 2017, 202, 16–22. [Google Scholar] [CrossRef]
- Beutin, L.; Martin, A. Outbreak of Shiga toxin–producing Escherichia coli (STEC) O104: H4 infection in Germany causes a paradigm shift with regard to human pathogenicity of STEC strains. J. Food Prot. 2012, 75, 408–418. [Google Scholar] [CrossRef]
- Shridhar, P.B.; Siepker, C.; Noll, L.W.; Shi, X.; Nagaraja, T.; Bai, J. Shiga toxin subtypes of non-O157 Escherichia coli serogroups isolated from cattle feces. Front. Cell. Infect. Microbiol. 2017, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Capps, K.M.; Ludwig, J.B.; Shridhar, P.B.; Shi, X.; Roberts, E.; DebRoy, C.; Cernicchiaro, N.; Phebus, R.K.; Bai, J.; Nagaraja, T. Identification, Shiga toxin subtypes and prevalence of minor serogroups of Shiga toxin-producing Escherichia coli in feedlot cattle feces. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lauzi, S.; Luzzago, C.; Chiani, P.; Michelacci, V.; Knijn, A.; Pedrotti, L.; Corlatti, L.; Buccheri Pederzoli, C.; Scavia, G.; Morabito, S. Free-ranging red deer (Cervus elaphus) as carriers of potentially zoonotic Shiga toxin-producing Escherichia coli. Transbound. Emerg. Dis. 2022, 69, 1902–1911. [Google Scholar] [CrossRef]
- Milton, A.A.P.; Agarwal, R.K.; Priya, G.B.; Aravind, M.; Athira, C.K.; Rose, L.; Saminathan, M.; Sharma, A.K.; Kumar, A. Captive wildlife from India as carriers of Shiga toxin-producing, enteropathogenic and enterotoxigenic Escherichia coli. J. Vet. Med. Sci. 2019, 81, 321–327. [Google Scholar] [CrossRef]
- Schmidt, H.; Scheef, J.r.; Morabito, S.; Caprioli, A.; Wieler, L.H.; Karch, H. A new Shiga toxin 2 variant (Stx2f) from Escherichia coli isolated from pigeons. Appl. Environ. Microbiol. 2000, 66, 1205–1208. [Google Scholar] [CrossRef]
- Friesema, I.H.; Keijzer-Veen, M.G.; Koppejan, M.; Schipper, H.S.; van Griethuysen, A.J.; Heck, M.E.; van Pelt, W. Hemolytic uremic syndrome associated with Escherichia coli O8: H19 and Shiga toxin 2f gene. Emerg. Infect. Dis. 2015, 21, 168. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority, European Centre for Disease Prevention and Control. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA J. 2016, 14, e04634. [Google Scholar] [CrossRef]
- Dias, D.; Costa, S.; Fonseca, C.; Baraúna, R.; Caetano, T.; Mendo, S. Pathogenicity of Shiga toxin-producing Escherichia coli (STEC) from wildlife: Should we care? Sci. Total Environ. 2022, 812, 152324. [Google Scholar] [CrossRef] [PubMed]
- Cooley, M.B.; Jay-Russell, M.; Atwill, E.R.; Carychao, D.; Nguyen, K.; Quiñones, B.; Patel, R.; Walker, S.; Swimley, M.; Pierre-Jerome, E. Development of a robust method for isolation of Shiga toxin-positive Escherichia coli (STEC) from fecal, plant, soil and water samples from a leafy greens production region in California. PLoS ONE 2013, 8, e65716. [Google Scholar] [CrossRef]
- Keen, J.E.; Wittum, T.E.; Dunn, J.R.; Bono, J.L.; Durso, L.M. Shiga-toxigenic Escherichia coli O157 in agricultural fair livestock, United States. Emerg. Infect. Dis. 2006, 12, 780. [Google Scholar] [CrossRef]
- Duris, J.W.; Haack, S.K.; Fogarty, L.R. Gene and Antigen Markers of Shiga-toxin Producing E. coli from Michigan and Indiana River Water: Occurrence and Relation to Recreational Water Quality Criteria. J. Environ. Qual. 2009, 38, 1878–1886. [Google Scholar] [CrossRef]
- Prakasan, S.; Prabhakar, P.; Lekshmi, M.; Kumar, S.; Nayak, B.B. Isolation of Shiga toxin-producing Escherichia coli harboring variant Shiga toxin genes from seafood. Vet. World 2018, 11, 379. [Google Scholar] [CrossRef]
- Feng, P.C.; Councell, T.; Keys, C.; Monday, S.R. Virulence characterization of Shiga-toxigenic Escherichia coli isolates from wholesale produce. Appl. Environ. Microbiol. 2011, 77, 343–345. [Google Scholar] [CrossRef]
- Ramaite, K.; Ekwanzala, M.D.; Momba, M.N.B. Prevalence and Molecular Characterisation of Extended-Spectrum Beta-Lactamase-Producing Shiga Toxin-Producing Escherichia coli, from Cattle Farm to Aquatic Environments. Pathogens 2022, 11, 674. [Google Scholar] [CrossRef]
- Elmonir, W.; Shalaan, S.; Tahoun, A.; Mahmoud, S.F.; Remela, E.M.A.; Eissa, R.; El-Sharkawy, H.; Shukry, M.; Zahran, R.N. Prevalence, antimicrobial resistance, and genotyping of Shiga toxin-producing Escherichia coli in foods of cattle origin, diarrheic cattle, and diarrheic humans in Egypt. Gut Pathog. 2021, 13, 1–11. [Google Scholar] [CrossRef]
- Bai, X.; Hu, B.; Xu, Y.; Sun, H.; Zhao, A.; Ba, P.; Fu, S.; Fan, R.; Jin, Y.; Wang, H. Molecular and phylogenetic characterization of non-O157 Shiga toxin-producing Escherichia coli strains in China. Front. Cell. Infect. Microbiol. 2016, 6, 143. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.; Shao, K.; Yang, X.; Bai, X.; Fu, S.; Sun, H.; Xu, Y.; Wang, H.; Li, Q.; Hu, B. High prevalence of non-O157 Shiga toxin-producing Escherichia coli in beef cattle detected by combining four selective agars. BMC Microbiol. 2019, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Liang, W.; Hu, Z.; Li, X.; Guo, R.; Hua, L.; Tang, X.; Tan, C.; Chen, H.; Wang, X.; et al. O-serogroups, virulence genes, antimicrobial susceptibility, and MLST genotypes of Shiga toxin-producing Escherichia coli from swine and cattle in Central China. BMC Vet. Res. 2019, 15, 427. [Google Scholar] [CrossRef] [PubMed]
- Fadel, H.M.; Afifi, R.; Al-Qabili, D.M. Characterization and zoonotic impact of Shiga toxin producing Escherichia coli in some wild bird species. Vet World 2017, 10, 1118–1128. [Google Scholar] [CrossRef]
- Probert, W.S.; Miller, G.M.; Ledin, K.E. Contaminated stream water as source for Escherichia coli O157 illness in children. Emerg. Infect. Dis. 2017, 23, 1216. [Google Scholar] [CrossRef]
- Ahlstrom, C.; Manuel, C.; Den Bakker, H.; Wiedmann, M.; Nightingale, K. Molecular ecology of Listeria spp., Salmonella, Escherichia coli O157: H7 and non-O157 Shiga toxin-producing E. coli in pristine natural environments in Northern Colorado. J. Appl. Microbiol. 2018, 124, 511–521. [Google Scholar] [CrossRef]
- Dias, D.; Caetano, T.; Torres, R.; Fonseca, C.; Mendo, S. Shiga toxin-producing Escherichia coli in wild ungulates. Sci. Total Environ. 2019, 651, 203–209. [Google Scholar] [CrossRef]
- Szczerba-Turek, A.; Socha, P.; Bancerz-Kisiel, A.; Platt-Samoraj, A.; Lipczynska-Ilczuk, K.; Siemionek, J.; Kończyk, K.; Terech-Majewska, E.; Szweda, W. Pathogenic potential to humans of Shiga toxin-producing Escherichia coli isolated from wild boars in Poland. Int. J. Food Microbiol. 2019, 300, 8–13. [Google Scholar] [CrossRef]
- Beutin, L.; Hammerl, J.A.; Reetz, J.; Strauch, E. Shiga toxin-producing Escherichia coli strains from cattle as a source of the Stx2a bacteriophages present in enteroaggregative Escherichia coli O104:H4 strains. Int. J. Med. Microbiol. 2013, 303, 595–602. [Google Scholar] [CrossRef]
- Patel, P.N.; Lindsey, R.L.; Garcia-Toledo, L.; Rowe, L.A.; Batra, D.; Whitley, S.W.; Drapeau, D.; Stoneburg, D.; Martin, H.; Juieng, P.; et al. High-Quality Whole-Genome Sequences for 77 Shiga Toxin-Producing Escherichia coli Strains Generated with PacBio Sequencing. Genome Announc. 2018, 6, e00391-00318. [Google Scholar] [CrossRef]
- Mellor, G.E.; Fegan, N.; Duffy, L.L.; McMILLAN, K.E.; Jordan, D.; Barlow, R.S. National survey of Shiga toxin–producing Escherichia coli serotypes O26, O45, O103, O111, O121, O145, and O157 in Australian beef cattle feces. J. Food Prot. 2016, 79, 1868–1874. [Google Scholar] [CrossRef] [PubMed]
- Puri-Giri, R.; Ghosh, A.; Thomson, J.; Zurek, L. House flies in the confined cattle environment carry non-O157 Shiga toxin-producing Escherichia coli. J. Med. Entomol. 2017, 54, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Mainga, A.O.; Cenci-Goga, B.T.; Malahlela, M.N.; Tshuma, T.; Kalake, A.; Karama, M. Occurrence and characterization of seven major Shiga toxin-producing Escherichia coli serotypes from healthy cattle on cow–calf operations in South Africa. Zoonoses Public Health 2018, 65, 777–789. [Google Scholar] [CrossRef]
- Dixon, A.; Cernicchiaro, N.; Amachawadi, R.G.; Shi, X.; Cull, C.A.; Renter, D.G. Longitudinal characterization of prevalence and concentration of Shiga toxin–producing Escherichia coli serogroups in feces of individual feedlot cattle. Foodborne Pathog. Dis. 2020, 17, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Engelen, F.; Thiry, D.; Devleesschauwer, B.; Mainil, J.; De Zutter, L.; Cox, E. Occurrence of ‘gang of five’ Shiga toxin-producing Escherichia coli serogroups on Belgian dairy cattle farms by overshoe sampling. Lett. Appl. Microbiol. 2021, 72, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Cernicchiaro, N.; Oliveira, A.R.; Hoehn, A.; Noll, L.W.; Shridhar, P.B.; Nagaraja, T.G.; Ives, S.E.; Renter, D.G.; Sanderson, M.W. Associations between season, processing plant, and hide cleanliness scores with prevalence and concentration of major shiga toxin–producing Escherichia coli on beef cattle hides. Foodborne Pathog. Dis. 2020, 17, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Ballem, A.; Gonçalves, S.; Garcia-Meniño, I.; Flament-Simon, S.C.; Blanco, J.E.; Fernandes, C.; Saavedra, M.J.; Pinto, C.; Oliveira, H.; Blanco, J. Prevalence and serotypes of Shiga toxin-producing Escherichia coli (STEC) in dairy cattle from Northern Portugal. PLoS ONE 2020, 15, e0244713. [Google Scholar] [CrossRef]
- Sanches, L.A.; Gomes, M.d.S.; Teixeira, R.H.F.; Cunha, M.P.V.; Oliveira, M.G.X.d.; Vieira, M.A.M.; Gomes, T.A.T.; Knobl, T. Captive wild birds as reservoirs of enteropathogenic E. coli (EPEC) and Shiga-toxin producing E. coli (STEC). Braz. J. Microbiol. 2017, 48, 760–763. [Google Scholar] [CrossRef]
- Rahman, M.M.; Lim, S.J.; Kim, W.H.; Cho, J.Y.; Park, Y.C. Prevalence data of diarrheagenic E. coli in the fecal pellets of wild rodents using culture methods and PCR assay. Data Brief 2020, 33, 106439. [Google Scholar] [CrossRef]
- Haindongo, N.; Nkandi, J.; Hamatui, N.; Akai, L.A.; Hemberger, M.Y.; Khaiseb, S.; Molini, U. The prevalence of non-O157: H7 Shiga toxin-producing Escherichia coli (STEC) in Namibian game meat. Vet. Ital. 2018, 54, 185–188. [Google Scholar]
- Reimoser, F.; Putman, R. Impacts of wild ungulates on vegetation: Costs and benefits. In Ungulate Management in Europe: Problems and Practices; Cambridge University Press: Cambridge, UK, 2011; pp. 144–191. [Google Scholar]
- Niewiadomska, K.; Kosicka-Gębska, M.; Gębski, J.; Gutkowska, K.; Jeżewska-Zychowicz, M.; Sułek, M. Game meat consumption—Conscious choice or just a game? Foods 2020, 9, 1357. [Google Scholar] [CrossRef] [PubMed]
- Hedman, H.D.; Varga, C.; Duquette, J.; Novakofski, J.; Mateus-Pinilla, N.E. Food Safety Considerations Related to the Consumption and Handling of Game Meat in North America. Vet. Sci. 2020, 7, 188. [Google Scholar] [CrossRef]
- Díaz-Sánchez, S.; Sánchez, S.; Sánchez, M.; Herrera-León, S.; Hanning, I.; Vidal, D. Detection and characterization of Shiga toxin-producing Escherichia coli in game meat and ready-to-eat meat products. Int. J. Food Microbiol. 2012, 160, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Reinstein, S.; Fox, J.; Shi, X.; Alam, M.; Nagaraja, T. Prevalence of Escherichia coli O157: H7 in the American bison (Bison bison). J. Food Prot. 2007, 70, 2555–2560. [Google Scholar] [CrossRef] [PubMed]
- Vu-Khac, H.; Cornick, N.A. Prevalence and genetic profiles of Shiga toxin-producing Escherichia coli strains isolated from buffaloes, cattle, and goats in central Vietnam. Vet. Microbiol. 2008, 126, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Roug, A.; Byrne, B.A.; Conrad, P.A.; Miller, W. Zoonotic fecal pathogens and antimicrobial resistance in county fair animals. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 303–308. [Google Scholar] [CrossRef]
- Strachan, N.J.C.; Rotariu, O.; Lopes, B.; MacRae, M.; Fairley, S.; Laing, C.; Gannon, V.; Allison, L.J.; Hanson, M.F.; Dallman, T.; et al. Whole Genome Sequencing demonstrates that Geographic Variation of Escherichia coli O157 Genotypes Dominates Host Association. Sci. Rep. 2015, 5, 14145. [Google Scholar] [CrossRef]
- Booher, S.; Cornick, N.; Moon, H. Persistence of Escherichia coli O157: H7 in experimentally infected swine. Vet. Microbiol. 2002, 89, 69–81. [Google Scholar] [CrossRef]
- Siddhnath, K.; Majumdar, R.; Parhi, J.; Sharma, S.; Mehta, N.; Laishram, M. Detection and characterization of Shiga toxin-producing Escherichia coli from carps from integrated aquaculture system. Aquaculture 2018, 487, 97–101. [Google Scholar] [CrossRef]
- Bertelloni, F.; Lunardo, E.; Rocchigiani, G.; Ceccherelli, R.; Ebani, V. Occurrence of Escherichia coli virulence genes in feces of wild birds from Central Italy. Asian Pac. J. Trop. Med. 2019, 12, 142–146. [Google Scholar] [CrossRef]
- Smith, O.M.; Olimpi, E.M.; Navarro-Gonzalez, N.; Cornell, K.A.; Frishkoff, L.O.; Northfield, T.D.; Bowles, T.M.; Edworthy, M.; Eilers, J.; Fu, Z. A trait-based framework for predicting foodborne pathogen risk from wild birds. Ecol. Appl. 2022, 32, e2523. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, M.; Hikoda, Y.; Fujii, Y.; Murata, M.; Miyoshi, H.; Ogura, Y.; Gotoh, Y.; Iwata, T.; Hayashi, T.; Akiba, M. Emergence of a multidrug-resistant Shiga toxin-producing enterotoxigenic Escherichia coli lineage in diseased swine in Japan. J. Clin. Microbiol. 2016, 54, 1074–1081. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.-Y.I.; Abu, J.; Zakaria, Z.; Khan, A.R.; Abdul Aziz, S.; Bitrus, A.A.; Habib, I. Multi-Drug Resistant Pathogenic Escherichia coli Isolated from Wild Birds, Chicken, and the Environment in Malaysia. Antibiotics 2022, 11, 1275. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.; Evenson, A.; Luckey, T.; McCoy, E.; Elvehjem, C.; Hart, E. Use of sulfasuxidine, streptothricin, and streptomycin in nutritional studies with the chick. J. Biol. Chem 1946, 165, 437–441. [Google Scholar] [CrossRef]
- Castanon, J. History of the use of antibiotic as growth promoters in European poultry feeds. Poult. Sci. 2007, 86, 2466–2471. [Google Scholar] [CrossRef]
- Cameron, A.; McAllister, T.A. Antimicrobial usage and resistance in beef production. J. Anim. Sci. Biotechnol. 2016, 7, 68. [Google Scholar] [CrossRef]
- Kim, J.C.; Chui, L.; Wang, Y.; Shen, J.; Jeon, B. Expansion of Shiga Toxin-Producing Escherichia coli by Use of Bovine Antibiotic Growth Promoters. Emerg. Infect. Dis. 2016, 22, 802–809. [Google Scholar] [CrossRef]
- Iweriebor, B.C.; Iwu, C.J.; Obi, L.C.; Nwodo, U.U.; Okoh, A.I. Multiple antibiotic resistances among Shiga toxin producing Escherichia coli O157 in feces of dairy cattle farms in Eastern Cape of South Africa. BMC Microbiol. 2015, 15, 213. [Google Scholar] [CrossRef]
- Srinivasan, V.; Nguyen, L.T.; Headrick, S.I.; Murinda, S.E.; Oliver, S.P. Antimicrobial resistance patterns of Shiga toxin-producing Escherichia coli O157: H7 and O157: H7− from different origins. Microb. Drug Resist. 2007, 13, 44–51. [Google Scholar] [CrossRef]
- Schroeder, C.M.; Meng, J.; Zhao, S.; DebRoy, C.; Torcolini, J.; Zhao, C.; McDermott, P.F.; Wagner, D.D.; Walker, R.D.; White, D.G. Antimicrobial resistance of Escherichia coli O26, O103, O111, O128, and O145 from animals and humans. Emerg. Infect. Dis. 2002, 8, 1409. [Google Scholar] [CrossRef]
- Tamang, M.D.; Sunwoo, H.; Jeon, B. Phage-mediated dissemination of virulence factors in pathogenic bacteria facilitated by antibiotic growth promoters in animals: A perspective. Anim. Health Res. Rev. 2017, 18, 160–166. [Google Scholar] [CrossRef] [PubMed]
- McGannon, C.M.; Fuller, C.A.; Weiss, A.A. Different classes of antibiotics differentially influence Shiga toxin production. Antimicrob. Agents Chemother. 2010, 54, 3790–3798. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Shafiq, M.; Rahman, S.U.; Bilal, H.; Ullah, A.; Noman, S.M.; Zeng, M.; Yuan, Y.; Xie, Q.; Li, X.; Jiao, X. Incidence and molecular characterization of ESBL-producing and colistin-resistant Escherichia coli isolates recovered from healthy food-producing animals in Pakistan. J. Appl. Microbiol. 2022, 133, 1169–1182. [Google Scholar] [CrossRef]
- Shafiq, M.; Huang, J.; Shah, J.M.; Ali, I.; Rahman, S.U.; Wang, L. Characterization and resistant determinants linked to mobile elements of ESBL-producing and mcr-1-positive Escherichia coli recovered from the chicken origin. Microb. Pathog. 2021, 150, 104722. [Google Scholar] [CrossRef]
- Rahal, E.A.; Fadlallah, S.M.; Nassar, F.J.; Kazzi, N.; Matar, G.M. Approaches to treatment of emerging Shiga toxin-producing Escherichia coli infections highlighting the O104: H4 serotype. Front. Cell. Infect. Microbiol. 2015, 5, 24. [Google Scholar] [CrossRef]
- Ochoa, T.J.; Chen, J.; Walker, C.M.; Gonzales, E.; Cleary, T.G. Rifaximin does not induce toxin production or phage-mediated lysis of Shiga toxin-producing Escherichia coli. Antimicrob. Agents Chemother. 2007, 51, 2837–2841. [Google Scholar] [CrossRef]
- Kurioka, T.; Yunou, Y.; Harada, H.; Kita, E. Efficacy of antibiotic therapy for infection with Shiga-like toxin-producing Escherichia coli O157: H7 in mice with protein-calorie malnutrition. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 561–571. [Google Scholar] [CrossRef]
- Mir, R.A.; Kudva, I.T. Antibiotic-resistant Shiga toxin-producing Escherichia coli: An overview of prevalence and intervention strategies. Zoonoses Public Health 2019, 66, 1–13. [Google Scholar] [CrossRef]
- Jost, C.; Bidet, P.; Carrere, T.; Mariani-Kurkdjian, P.; Bonacorsi, S. Susceptibility of enterohaemorrhagic Escherichia coli to azithromycin in France and analysis of resistance mechanisms. J. Antimicrob. Chemother. 2016, 71, 1183–1187. [Google Scholar] [CrossRef]
- Wang, Y.; Subedi, D.; Li, J.; Wu, J.; Ren, J.; Xue, F.; Dai, J.; Barr, J.J.; Tang, F. Phage Cocktail Targeting STEC O157: H7 Has Comparable Efficacy and Superior Recovery Compared with Enrofloxacin in an Enteric Murine Model. Microbiol. Spectr. 2022, 10, e00232-00222. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zeng, H.; Qian, X.; Li, Y.; Xue, F.; Ren, J.; Dai, J.; Tang, F. Pre-treatment with phages achieved greater protection of mice against infection with Shiga toxin-producing Escherichia coli than post-treatment. Res. Vet. Sci. 2022, 150, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.; Krishnasamy, V.; Saw, L.; Smith, L.; Wagner, J.; Weigand, J.; Tewell, M.; Kellis, M.; Penev, R.; McCullough, L. Outbreak of E. coli O157: H7 infections associated with exposure to animal manure in a rural community—Arizona and Utah, June–July 2017. Morb. Mortal. Wkly. Rep. 2018, 67, 659. [Google Scholar] [CrossRef]
- Venegas-Vargas, C.; Henderson, S.; Khare, A.; Mosci, R.E.; Lehnert, J.D.; Singh, P.; Ouellette, L.M.; Norby, B.; Funk, J.A.; Rust, S. Factors associated with Shiga toxin-producing Escherichia coli shedding by dairy and beef cattle. Appl. Environ. Microbiol. 2016, 82, 5049–5056. [Google Scholar] [CrossRef]
- Mechie, S.; Chapman, P.; Siddons, C. A fifteen month study of Escherichia coli O157: H7 in a dairy herd. Epidemiol. Infect. 1997, 118, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Braden, K.; Blanton Jr, J.; Allen, V.; Pond, K.; Miller, M. Ascophyllum nodosum supplementation: A preharvest intervention for reducing Escherichia coli O157: H7 and Salmonella spp. in feedlot steers. J. Food Prot. 2004, 67, 1824–1828. [Google Scholar] [CrossRef]
- Walle, K.V.; Vanrompay, D.; Cox, E. Bovine innate and adaptive immune responses against Escherichia coli O157: H7 and vaccination strategies to reduce faecal shedding in ruminants. Vet. Immunol. Immunopathol. 2013, 152, 109–120. [Google Scholar] [CrossRef]
- Snedeker, K.G.; Campbell, M.; Sargeant, J.M. A systematic review of vaccinations to reduce the shedding of Escherichia coli O157 in the faeces of domestic ruminants. Zoonoses Public Health 2012, 59, 126–138. [Google Scholar] [CrossRef]
- Blazar, J.; Allard, M.; Lienau, E.K. Insects as vectors of foodborne pathogenic bacteria. Terr. Arthropod Rev. 2011, 4, 5–16. [Google Scholar] [CrossRef]
- Ray, R.; Potts, R.; Pietri, J.E. The persistence of Escherichia coli infection in German cockroaches (Blattodea: Blattellidae) varies between host developmental stages and is influenced by the gut microbiota. J. Med. Entomol. 2020, 57, 1964–1971. [Google Scholar] [CrossRef]
- Sánchez, S.; Martínez, R.; García, A.; Vidal, D.; Blanco, J.; Blanco, M.; Blanco, J.; Mora, A.; Herrera-León, S.; Echeita, A. Detection and characterisation of O157: H7 and non-O157 Shiga toxin-producing Escherichia coli in wild boars. Vet. Microbiol. 2010, 143, 420–423. [Google Scholar] [CrossRef] [PubMed]
- Bakry, N.; Awad, W.; Ahmed, S.; Kamel, M. The role of Musca domestica and milk in transmitting pathogenic multidrug-resistant Escherichia coli and associated phylogroups to neonatal calves. Environ. Sci. Pollut. Res. 2022, 29, 39593–39609. [Google Scholar] [CrossRef] [PubMed]
- Perrat, A.; Branchu, P.; Decors, A.; Turci, S.; Bayon-Auboyer, M.-H.; Petit, G.; Grosbois, V.; Brugère, H.; Auvray, F.; Oswald, E. Wild Boars as Reservoir of Highly Virulent Clone of Hybrid Shiga Toxigenic and Enterotoxigenic Escherichia coli Responsible for Edema Disease, France. Emerg. Infect. Dis. 2022, 28, 382. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.J.; Li, A.Y.; Pritchard, S.; Tangtrakulwanich, K.; Baxendale, F.P.; Brewer, G. Contact and fumigant toxicity of a botanical-based feeding deterrent of the stable fly, Stomoxys calcitrans (Diptera: Muscidae). J. Agric. Food Chem. 2011, 59, 10394–10400. [Google Scholar] [CrossRef]
- Pavela, R. Lethal and sublethal effects of thyme oil (Thymus vulgaris L.) on the house fly (Musca domestica Lin.). J. Essent. Oil Bear. Plants 2007, 10, 346–356. [Google Scholar] [CrossRef]
- Weerarathne, P.; Payne, J.; Saha, J.; Kountoupis, T.; Jadeja, R.; Jaroni, D. Evaluating the efficacy of sodium acid sulfate to reduce Escherichia coli O157: H7 and its biofilms on food-contact surfaces. LWT 2021, 139, 110501. [Google Scholar] [CrossRef]
- Sloup, R.E.; Cieza, R.J.; Needle, D.B.; Abramovitch, R.B.; Torres, A.G.; Waters, C.M. Polysorbates prevent biofilm formation and pathogenesis of Escherichia coli O104: H4. Biofouling 2016, 32, 1131–1140. [Google Scholar] [CrossRef]
- Barbieri, R.; Coppo, E.; Marchese, A.; Daglia, M.; Sobarzo-Sánchez, E.; Nabavi, S.F.; Nabavi, S.M. Phytochemicals for human disease: An update on plant-derived compounds antibacterial activity. Microbiol. Res. 2017, 196, 44–68. [Google Scholar] [CrossRef]
- Lillehoj, H.; Liu, Y.; Calsamiglia, S.; Fernandez-Miyakawa, M.E.; Chi, F.; Cravens, R.L.; Oh, S.; Gay, C.G. Phytochemicals as antibiotic alternatives to promote growth and enhance host health. Vet. Res. 2018, 49, 76. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Y.; Hu, L.; Suo, Y.; Zhang, L.; Jin, F.; Feng, X.; Teng, N.; Li, Y. Effects of dietary supplementation of quercetin on performance, egg quality, cecal microflora populations, and antioxidant status in laying hens. Poult. Sci. 2014, 93, 347–353. [Google Scholar] [CrossRef]
- Lee, J.-H.; Regmi, S.C.; Kim, J.-A.; Cho, M.H.; Yun, H.; Lee, C.-S.; Lee, J. Apple flavonoid phloretin inhibits Escherichia coli O157: H7 biofilm formation and ameliorates colon inflammation in rats. Infect. Immun. 2011, 79, 4819–4827. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Cho, H.S.; Joo, S.W.; Chandra Regmi, S.; Kim, J.-A.; Ryu, C.-M.; Ryu, S.Y.; Cho, M.H.; Lee, J. Diverse plant extracts and trans-resveratrol inhibit biofilm formation and swarming of Escherichia coli O157: H7. Biofouling 2013, 29, 1189–1203. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, Y.-G.; Ryu, S.Y.; Cho, M.H.; Lee, J. Ginkgolic acids and Ginkgo biloba extract inhibit Escherichia coli O157: H7 and Staphylococcus aureus biofilm formation. Int. J. Food Microbiol. 2014, 174, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, Y.-G.; Cho, H.S.; Ryu, S.Y.; Cho, M.H.; Lee, J. Coumarins reduce biofilm formation and the virulence of Escherichia coli O157: H7. Phytomedicine 2014, 21, 1037–1042. [Google Scholar] [CrossRef]
- Kim, Y.-G.; Lee, J.-H.; Gwon, G.; Kim, S.-I.; Park, J.G.; Lee, J. Essential oils and eugenols inhibit biofilm formation and the virulence of Escherichia coli O157: H7. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Moretti, M.D.; Sanna-Passino, G.; Demontis, S.; Bazzoni, E. Essential oil formulations useful as a new tool for insect pest control. AAPs PharmSciTech 2002, 3, 64–74. [Google Scholar] [CrossRef]
- Kholif, A.E.; Olafadehan, O.A. Essential oils and phytogenic feed additives in ruminant diet: Chemistry, ruminal microbiota and fermentation, feed utilization and productive performance. Phytochem. Rev. 2021, 20, 1087–1108. [Google Scholar] [CrossRef]
- Xu, Y.; Dugat-Bony, E.; Zaheer, R.; Selinger, L.; Barbieri, R.; Munns, K.; McAllister, T.A.; Selinger, L.B. Escherichia coli O157:H7 Super-Shedder and Non-Shedder Feedlot Steers Harbour Distinct Fecal Bacterial Communities. PLoS ONE 2014, 9, e98115. [Google Scholar] [CrossRef]
- Zaheer, R.; Dugat-Bony, E.; Holman, D.; Cousteix, E.; Xu, Y.; Munns, K.; Selinger, L.J.; Barbieri, R.; Alexander, T.; McAllister, T.A. Changes in bacterial community composition of Escherichia coli O157: H7 super-shedder cattle occur in the lower intestine. PLoS ONE 2017, 12, e0170050. [Google Scholar]
- Mir, R.A.; Schaut, R.G.; Looft, T.; Allen, H.K.; Sharma, V.K.; Kudva, I.T. Recto-anal junction (RAJ) and fecal microbiomes of cattle experimentally challenged with Escherichia coli O157: H7. Front. Microbiol. 2020, 11, 693. [Google Scholar] [CrossRef]
- Wang, O.; McAllister, T.A.; Plastow, G.; Stanford, K.; Selinger, B.; Guan, L.L. Interactions of the Hindgut Mucosa-Associated Microbiome with Its Host Regulate Shedding of Escherichia coli O157:H7 by Cattle. Appl. Environ. Microbiol. 2018, 84, e01738-17. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yan, F.; Liu, T.; Zhang, Y.; Li, X.; Wang, M.; Zhang, C.; Xu, X.; Deng, L.; Yao, J.; et al. Ruminal Microbiota Determines the High-Fiber Utilization of Ruminants: Evidence from the Ruminal Microbiota Transplant. Microbiol. Spectr. 2022, 10, e00446-00422. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.; Hu, J.; Shi, B.; Dingkao, R.; Wang, J.; Li, S.; Zhang, W.; Luo, Y.; Liu, X. Characteristics and functions of the rumen microbial community of Cattle-Yak at different ages. BioMed Res. Int. 2020, 2020, 3482692. [Google Scholar] [CrossRef] [PubMed]
- Saleem, F.; Ametaj, B.N.; Bouatra, S.; Mandal, R.; Zebeli, Q.; Dunn, S.M.; Wishart, D.S. A metabolomics approach to uncover the effects of grain diets on rumen health in dairy cows. J. Dairy Sci. 2012, 95, 6606–6623. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, F.; Cai, W.; Jia, C.; Bai, Y.; He, Y.; Zhu, W.; Li, R.W.; Song, J. Diet-induced changes in bacterial communities in the jejunum and their associations with bile acids in Angus beef cattle. Anim. Microbiome 2020, 2, 33. [Google Scholar] [CrossRef] [PubMed]
- Keen, J.; Uhlich, G.; Elder, R. Effects of hay-and grain-based diets on fecal shedding in naturally-acquired enterohemorrhagic E. coli (EHEC) O157 in beef feedlot cattle. In Proceedings of the 80th Conference Research Workers in Animal Diseases, Chicago, IL, USA, 7–9 November 1999. [Google Scholar]
- Jacob, M.; Paddock, Z.; Renter, D.G.; Lechtenberg, K.; Nagaraja, T. Inclusion of dried or wet distillers’ grains at different levels in diets of feedlot cattle affects fecal shedding of Escherichia coli O157: H7. Appl. Environ. Microbiol. 2010, 76, 7238–7242. [Google Scholar] [CrossRef][Green Version]
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current understanding of the human microbiome. Nat. Med. 2018, 24, 392–400. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Kim, A. Dysbiosis: A review highlighting obesity and inflammatory bowel disease. J. Clin. Gastroenterol. 2015, 49, S20–S24. [Google Scholar] [CrossRef]
- Singh, P.; Teal, T.K.; Marsh, T.L.; Tiedje, J.M.; Mosci, R.; Jernigan, K.; Zell, A.; Newton, D.W.; Salimnia, H.; Lephart, P.; et al. Intestinal microbial communities associated with acute enteric infections and disease recovery. Microbiome 2015, 3, 45. [Google Scholar] [CrossRef]
- Mendes, R.; Raaijmakers, J.M. Cross-kingdom similarities in microbiome functions. ISME J. 2015, 9, 1905–1907. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.C.-H.; Shih, Y.-A.; Wu, L.-L.; Lin, Y.-D.; Kuo, W.-T.; Peng, W.-H.; Lu, K.-S.; Wei, S.-C.; Turner, J.R.; Ni, Y.-H. Enteric dysbiosis promotes antibiotic-resistant bacterial infection: Systemic dissemination of resistant and commensal bacteria through epithelial transcytosis. Am. J. Physiol.-Gastrointest. Liver Physiol. 2014, 307, G824–G835. [Google Scholar] [CrossRef] [PubMed]
- Winter, S.E.; Winter, M.G.; Xavier, M.N.; Thiennimitr, P.; Poon, V.; Keestra, A.M.; Laughlin, R.C.; Gomez, G.; Wu, J.; Lawhon, S.D. Host-derived nitrate boosts growth of E. coli in the inflamed gut. Science 2013, 339, 708–711. [Google Scholar] [CrossRef]
- Tarr, G.; Shringi, S.; Oltean, H.; Mayer, J.; Rabinowitz, P.; Wakefield, J.; Tarr, P.; Besser, T.; Phipps, A. Importance of case age in the purported association between phylogenetics and hemolytic uremic syndrome in Escherichia coli O157: H7 infections. Epidemiol. Infect. 2018, 146, 1550–1555. [Google Scholar] [CrossRef]
- Gigliucci, F.; von Meijenfeldt, F.B.; Knijn, A.; Michelacci, V.; Scavia, G.; Minelli, F.; Dutilh, B.E.; Ahmad, H.M.; Raangs, G.C.; Friedrich, A.W. Metagenomic characterization of the human intestinal microbiota in fecal samples from STEC-infected patients. Front. Cell. Infect. Microbiol. 2018, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-S.; Jeong, Y.-J.; Lee, M.-S. Escherichia coli Shiga toxins and gut microbiota interactions. Toxins 2021, 13, 416. [Google Scholar] [CrossRef]
- Saito, K.; Suzuki, R.; Koyanagi, Y.; Isogai, H.; Yoneyama, H.; Isogai, E. Inhibition of enterohemorrhagic Escherichia coli O157: H7 infection in a gnotobiotic mouse model with pre-colonization by Bacteroides strains. Biomed. Rep. 2019, 10, 175–182. [Google Scholar] [CrossRef]
- Zumbrun, S.D.; Melton-Celsa, A.R.; Smith, M.A.; Gilbreath, J.J.; Merrell, D.S.; O’Brien, A.D. Dietary choice affects Shiga toxin-producing Escherichia coli (STEC) O157:H7 colonization and disease. Proc. Natl. Acad. Sci. USA 2013, 110, E2126–E2133. [Google Scholar] [CrossRef]
- Byres, E.; Paton, A.W.; Paton, J.C.; Löfling, J.C.; Smith, D.F.; Wilce, M.C.; Talbot, U.M.; Chong, D.C.; Yu, H.; Huang, S.; et al. Incorporation of a non-human glycan mediates human susceptibility to a bacterial toxin. Nature 2008, 456, 648–652. [Google Scholar] [CrossRef]
- Paton, A.W.; Paton, J.C. Escherichia coli Subtilase Cytotoxin. Toxins 2010, 2, 215–228. [Google Scholar] [CrossRef]
- Varki, N.M.; Varki, A. Diversity in cell surface sialic acid presentations: Implications for biology and disease. Lab. Investig. 2007, 87, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Hwang, S.W.; Kim, S.; Lee, Y.-S.; Kim, T.-Y.; Lee, S.-H.; Kim, S.J.; Yoo, H.J.; Kim, E.N.; Kweon, M.-N. Dietary cellulose prevents gut inflammation by modulating lipid metabolism and gut microbiota. Gut Microbes 2020, 11, 944–961. [Google Scholar] [CrossRef] [PubMed]
- Beddoe, T.; Paton, A.W.; Le Nours, J.; Rossjohn, J.; Paton, J.C. Structure, biological functions and applications of the AB5 toxins. Trends Biochem. Sci. 2010, 35, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Zhang, L.; Sun, H.; Hu, Y.; Fanning, S.; Du, P.; Cui, S.; Bai, L. Characterization of non-O157 Shiga toxin-producing Escherichia coli cultured from cattle farms in Xinjiang Uygur Autonomous Region, China, during 2016–2017. Foodborne Pathog. Dis. 2021, 18, 761–770. [Google Scholar] [CrossRef]
- Brooks, J.T.; Sowers, E.G.; Wells, J.G.; Greene, K.D.; Griffin, P.M.; Hoekstra, R.M.; Strockbine, N.A. Non-O157 Shiga toxin–producing Escherichia coli infections in the United States, 1983–2002. J. Infect. Dis. 2005, 192, 1422–1429. [Google Scholar] [CrossRef]
- Khan, A.; Das, S.; Ramamurthy, T.; Sikdar, A.; Khanam, J.; Yamasaki, S.; Takeda, Y.; Nair, G.B. Antibiotic resistance, virulence gene, and molecular profiles of Shiga toxin-producing Escherichia coli isolates from diverse sources in Calcutta, India. J. Clin. Microbiol. 2002, 40, 2009–2015. [Google Scholar] [CrossRef]
- Mora, A.; Herrrera, A.; López, C.; Dahbi, G.; Mamani, R.; Pita, J.M.; Alonso, M.P.; Llovo, J.; Bernárdez, M.I.; Blanco, J.E. Characteristics of the Shiga-toxin-producing enteroaggregative Escherichia coli O104: H4 German outbreak strain and of STEC strains isolated in Spain. Int Microbiol 2011, 14, 121–141. [Google Scholar]
- Withenshaw, S.M.; Smith, R.P.; Davies, R.; Smith, A.E.; Gray, E.; Rodgers, J. A systematized review and qualitative synthesis of potential risk factors associated with the occurrence of non-O157 Shiga toxin-producing Escherichia coli (STEC) in the primary production of cattle. Compr. Rev. Food Sci. Food Saf. 2022, 21, 2363–2390. [Google Scholar] [CrossRef]
- Menrath, A.; Wieler, L.H.; Heidemanns, K.; Semmler, T.; Fruth, A.; Kemper, N. Shiga toxin producing Escherichia coli: Identification of non-O157: H7-Super-Shedding cows and related risk factors. Gut Pathog. 2010, 2, 1–9. [Google Scholar] [CrossRef]
- Allué-Guardia, A.; Martínez-Castillo, A.; Muniesa, M. Persistence of infectious Shiga toxin-encoding bacteriophages after disinfection treatments. Appl. Environ. Microbiol. 2014, 80, 2142–2149. [Google Scholar] [CrossRef]
- Solheim, H.; Sekse, C.; Urdahl, A.M.; Wasteson, Y.; Nesse, L.L. Biofilm as an environment for dissemination of stx genes by transduction. Appl. Environ. Microbiol. 2013, 79, 896–900. [Google Scholar] [CrossRef] [PubMed]


| Serotypes | Livestock | Wildlife | Agriculture | Food Industry | Continental Prevalence | Source |
|---|---|---|---|---|---|---|
| O157 | + | + | + | + | EU, NA, SA, AS, AF | [34,116,117,118,119,120] |
| Non-O157 (Big-6) | ||||||
| O26 | + | + | + | + | EU, NA, SA, AS, AF | [34,116,117,118,119,120,121,122] |
| O45 | + | + | + | + | EU, NA, SA, AS, AF | [116,117,123,124] |
| O103 | + | + | + | + | EU, NA, SA, AS, AF | [116,117,119,120,124] |
| O111 | + | + | + | EU, NA, SA, AS, AF | [116,117,119,121,122,125] | |
| O121 | + | + | + | EU, NA, SA, AS, AF | [116,120,123] | |
| O145 | + | + | + | EU, NA, SA, AS, AF | [34,116,117,119,120,124] | |
| Other Non-O157 | ||||||
| O113 | + | + | + | EU, NA, SA, AS, AF | [34,109,118,126] | |
| O2 | + | + | + | + | EU, NA, SA, AS, AF | [118,120,123,127] |
| O5 | + | + | EU, AF, AA, NA, SA | [120,128] | ||
| O8 | + | + | + | + | NA, EU, SA, AS | [34,118,120,121,123,127,129] |
| O22 | + | + | EU, NA, AS, SA | [67,100,121,130] | ||
| O91 | + | + | + | EU, NA, SA, AS, AF | [34,119,121] | |
| O171 | + | + | EU, SA | [34,131] | ||
| O15 | + | + | NA, SA | [4,123] | ||
| O174 | + | + | EU, SA | [120,121,127] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ray, R.; Singh, P. Prevalence and Implications of Shiga Toxin-Producing E. coli in Farm and Wild Ruminants. Pathogens 2022, 11, 1332. https://doi.org/10.3390/pathogens11111332
Ray R, Singh P. Prevalence and Implications of Shiga Toxin-Producing E. coli in Farm and Wild Ruminants. Pathogens. 2022; 11(11):1332. https://doi.org/10.3390/pathogens11111332
Chicago/Turabian StyleRay, Ritesh, and Pallavi Singh. 2022. "Prevalence and Implications of Shiga Toxin-Producing E. coli in Farm and Wild Ruminants" Pathogens 11, no. 11: 1332. https://doi.org/10.3390/pathogens11111332
APA StyleRay, R., & Singh, P. (2022). Prevalence and Implications of Shiga Toxin-Producing E. coli in Farm and Wild Ruminants. Pathogens, 11(11), 1332. https://doi.org/10.3390/pathogens11111332






