The Species at Risk Act (2002) and Transboundary Species Listings along the US–Canada Border
Abstract
1. Introduction
… a similar story template: the idea that modern society has degraded a natural world that used to be beautiful, harmonious, and self-sustaining and that might disappear completely if modern humans do not change their way of life.(p. 7)
2. Reading the Species at Risk Act
The Minister may, in cooperation with one or more provincial governments having an interest therein, take such measures as the Minister deems necessary for the protection of any species of wildlife in danger of extinction.
Legal texts allow us to trace why and how … communities see the fate of nonhuman species as part of their own identity and history, and—given the institutional power of legal texts to shape and enforce social practices …—how they envision the best possible relation between humans and nonhumans now and in the future.
wildlife species means a species, subspecies, variety or geographically or genetically distinct population of animal, plant or other organism, other than a bacterium or virus, that is wild by nature and(a) is native to Canada; or(b) has extended its range into Canada without human intervention and has been present in Canada for at least 50 years.
The purposes of this Act are to prevent wildlife species from being extirpated or becoming extinct, to provide for the recovery of wildlife species that are extirpated, endangered or threatened as a result of human activity and to manage species of special concern to prevent them from becoming endangered or threatened.
3. Methods
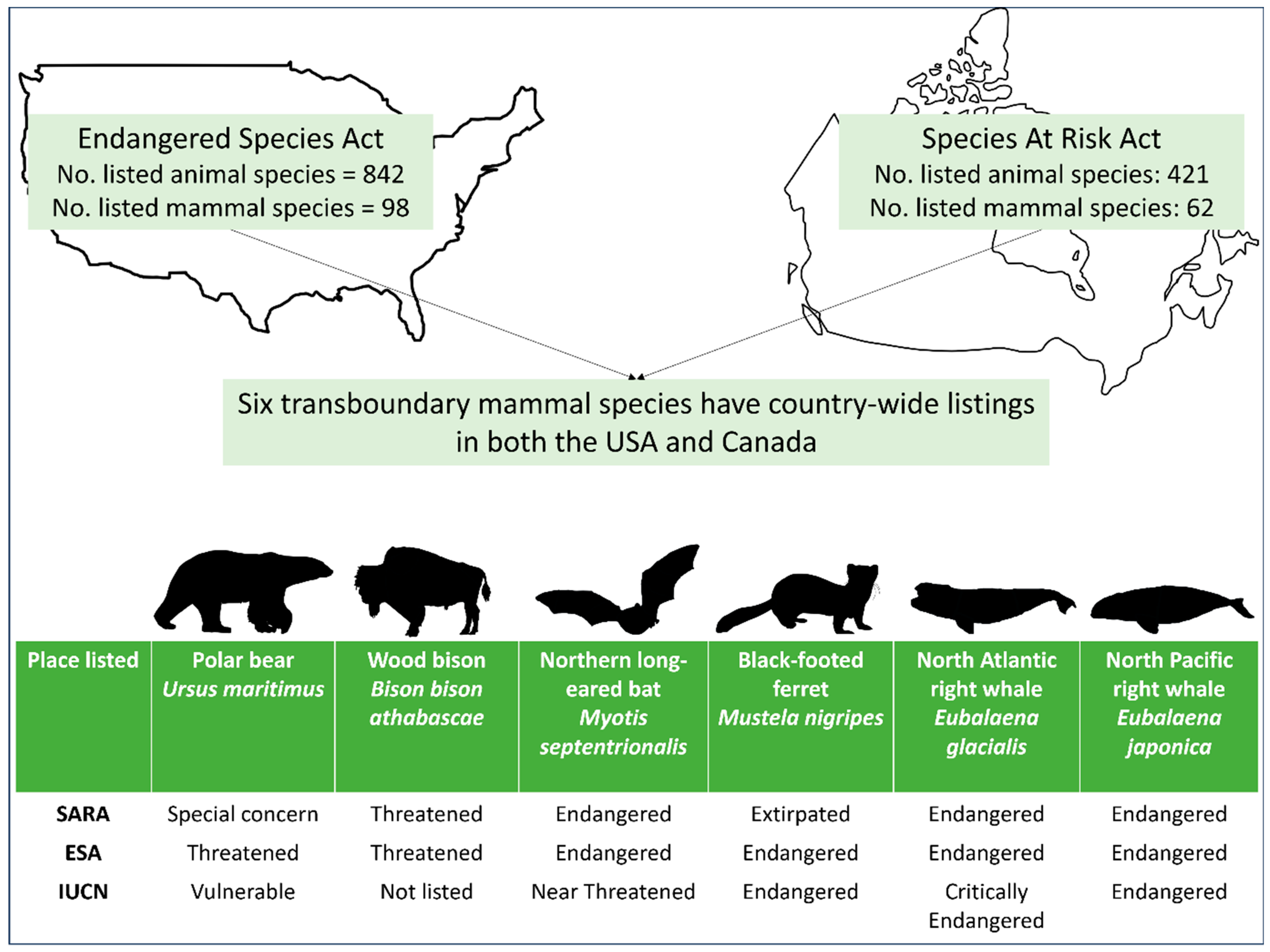
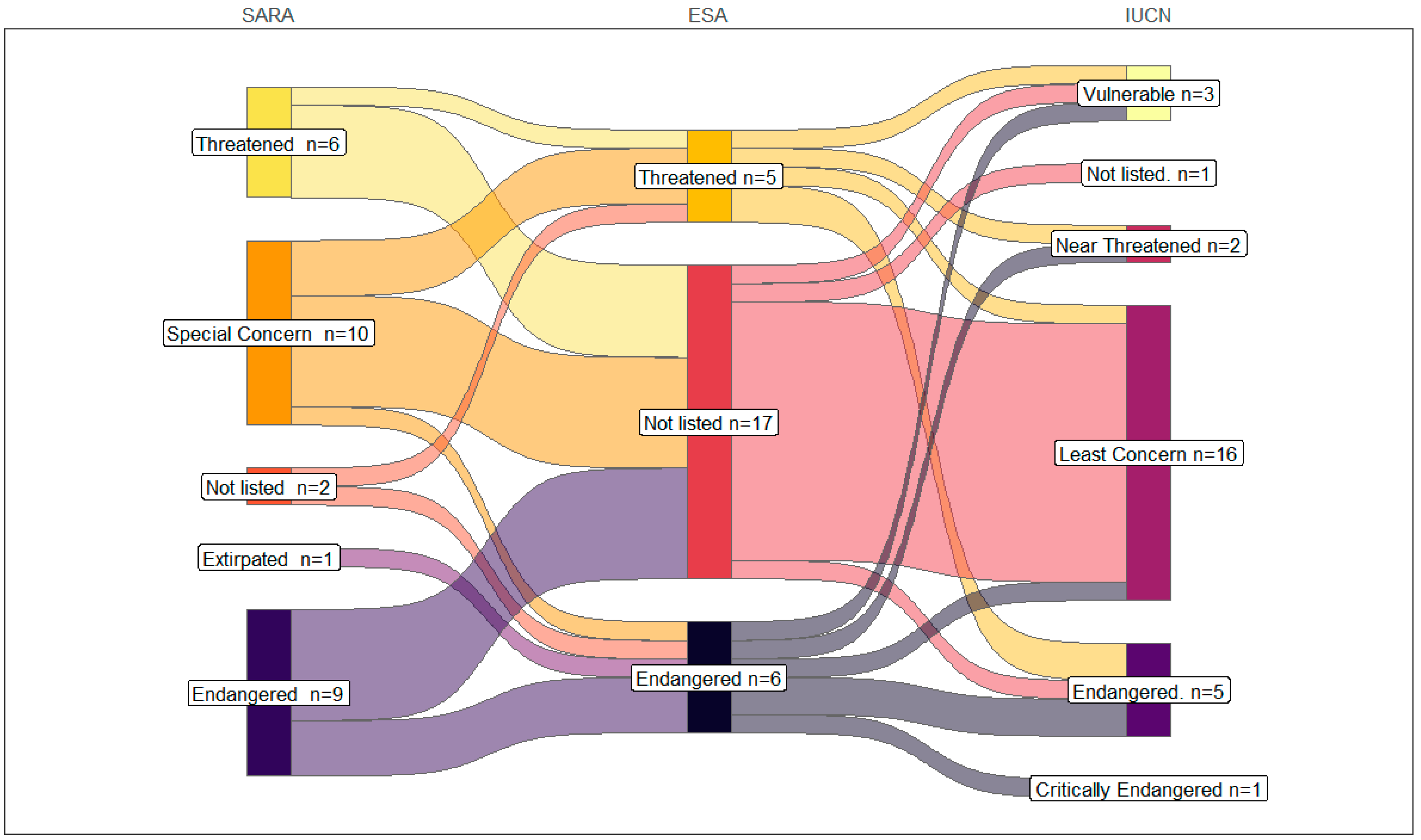
4. Results
Provincial-Scale
5. Discussion
5.1. Differences in Listing Methodology and Terminology
the institutionalized practices by which members of a given society test and deploy knowledge claims used as a basis for making collective choices. Just as any culture has established folkways that give meaning to its social interactions, so I suggest that modern technoscientific cultures have developed tacit knowledge-ways through which they assess the rationality and robustness of claims that seek to order their lives; demonstrations or arguments that fail to meet these tests may be dismissed as illegitimate or irrational.(p. 255)
5.2. Cross-Border Conservation and Jurisdictional Rarity
6. Conclusions
7. Reflections
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
| 1 | In this paper we use this term as shorthand for ‘humanities scholar’. |
| 2 | COSEWIC’s definition of a ‘native wildlife species’ is more restrictive than the Act that the Committee implements: ‘A wildlife species that occurs in Canada naturally, or that has expanded its range into Canada without human intervention from a region where it naturally occurred, has produced viable subpopulations, and has persisted in Canada for at least 50 years.’ So starlings need not apply. |
References
- Bergman, Jordanna N., Allison D. Binley, Rowan E. Murphy, Caitlyn A. Proctor, Thuong Tran Nguyen, Elise S. Urness, Michelle A. Vala, Jaimie G. Vincent, Lenore Fahrig, and Joseph R. Bennett. 2020. How to rescue Ontario’s Endangered Species Act: A biologist’s perspective. FACETS 5: 423–31. [Google Scholar] [CrossRef]
- Bunnell, Fred L., David F. Fraser, and Andrew P. Harcombe. 2009. Increasing Effectiveness of Conservation Decisions: A System and its Application. Natural Areas Journal 29: 79–90. [Google Scholar] [CrossRef]
- Bunnell, Fred L., R. Wayne Campbell, and Kelly Squires. 2004. Allocating Scarce Resources for Conservation in a Species-rich Environment: Guidelines from History and Science. Paper presented at the Species at Risk 2004 Pathways to Recovery Conference, Victoria, BC, Canada, March 2–6. [Google Scholar]
- CDC. 2023. BC Species & Ecosystems Explorer. Available online: https://a100.gov.bc.ca/pub/eswp/ (accessed on 2 August 2023).
- Chen, I-Ching, Jane K. Hill, Ralf Ohlemüller, David B. Roy, and Chris D. Thomas. 2011. Rapid Range Shifts of Species Associated with High Levels of Climate Warming. Science 333: 1024–26. [Google Scholar] [CrossRef]
- Cox, Sarah. 2023. Investigation: B.C. to Feds: Don’t Issue Emergency Order to Save the Endangered Spotted Owl. Available online: https://thenarwhal.ca/spotted-owl-emergency-order-documents/ (accessed on 27 November 2023).
- Cruickshank, Ainslie. 2022. ‘Huge Legal Gaps’ Are Driving B.C. Species to Extinction, Conservation Groups Say. Available online: https://thenarwhal.ca/bc-species-at-risk-cop15/ (accessed on 27 November 2023).
- Dallimer, Martin, and Niels Strange. 2015. Why socio-political borders and boundaries matter in conservation. Trends Ecol Evol 30: 132–39. [Google Scholar] [CrossRef] [PubMed]
- Department of Fish and Wildlife. 2023. WAC 220-610-110: Endangered, Threatened, and Sensitive Wildlife Species Classification; Washington: Washington State Legislature, p. 110.
- Donihee, John. 2000. The Evolution of Wildlife Law in Canada. In Occasional Paper No. 9. Calgary: Canadian Institute of Resources Law. [Google Scholar]
- Dorey, Katherine, and Tony R. Walker. 2018. Limitations of threatened species lists in Canada: A federal and provincial perspective. Biological Conservation 217: 259–68. [Google Scholar] [CrossRef]
- ECCC. 2013. Greater Sage-Grouse: Emergency Protection Order. Toronto: Environment and Climate Change Canada. [Google Scholar]
- ECCC. 2014. Categories of Species at Risk: Poster. Available online: https://www.canada.ca/en/environment-climate-change/services/species-risk-education-centre/poster.html (accessed on 4 December 2023).
- ECCC. 2016. Emergency order for the protection of the Western chorus frog (Great Lakes/St. Lawrence—Canadian shield population). In SOR/2016-211. Toronto: Environment and Climate Change Canada. [Google Scholar]
- Garrard, Greg. 2020. Brexit ecocriticism. Green Letters 24: 110–24. [Google Scholar] [CrossRef]
- Garrard, Greg. 2023. Ecocriticism, 3rd ed. The New Critical Idiom. Oxfordshire: Routledge. [Google Scholar]
- Harper, Bill, Sydney Canngings, David Fraser, and William T. Munro. 1994. Provincial lists of species at risk. In Biodiversity in British Columbia. Edited by L. E. Harding and E. McCullum. Delta: Canadian Wildlife Service. [Google Scholar]
- Heise, Ursula K. 2016a. From Arks to ARKive.org: Database, Epic, and Biodiversity. In Imagining Extinction: The Culturual Meanings of Endangered Species. Chicago: University of Chicago Press, pp. 55–86. [Google Scholar]
- Heise, Ursula K. 2016b. Imagining Extinction: The Cultural Meanings of Endangered Species. Chicago: University of Chicago Press. [Google Scholar]
- Heise, Ursula K. 2016c. The Legal Lives of Endangered Species: Biodiversity Laws and Culture. In Imagining Extinction: The Cultural Meanings of Endangered Species. Chicago: University of Chicago Press, pp. 87–126. [Google Scholar]
- Hockings, Marc, James Hardcastle, Stephen Woodley, Trevor Sandwith, Joanne Wilson, Marnie Bammert, Sandra Valenzuela, Beatrice Chataigner, Thierry Lefebvre, Fiona Leverington, and et al. 2019. The IUCN Green List of Protected and Conserved Areas: Setting the standard for effective conservation. Parks 25.2: 57–66. [Google Scholar] [CrossRef]
- Hoffman, Nadine. 2018. Species at Risk Act a Comprehensive Inventory of Legislative Documents 1973–2017. Paper presented at the A Symposium on Environment in the Courtroom: Enforcement Issues in Canadian Wildlife Protection, Calgary, AB, Canada, March 2–3. [Google Scholar]
- Illical, Mary, and Kathryn Harrison. 2007. Protecting Endangered Species in the US and Canada: The Role of Negative Lesson Drawing. Canadian Journal of Political Science/Revue Canadienne de Science Politique 40: 367–94. [Google Scholar] [CrossRef]
- IUCN. 2012. IUCN Red List categories and criteria: Version 3.1, 2nd ed. Gland and Cambridge: IUCN, vol. iv. [Google Scholar]
- IUCN. 2022. Guidelines for Using the IUCN Red List Categories and Criteria. Version 15.1. IUCN Standards and Petitions Subcomittee. Available online: https://www.iucnredlist.org/documents/RedListGuidelines.pdf (accessed on 2 August 2023).
- IUCN. 2023. The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org/ (accessed on 2 August 2023).
- Jasanoff, Sheila. 2005. Civic Epistemology. In Designs on Nature. Princeton: Princeton University Press, pp. 247–71. [Google Scholar]
- Knorr-Cetina, Karin. 1999. Epistemic Cultures: How the Sciences Make Knowledge. Cambridge: Harvard University Press. [Google Scholar]
- Knorr-Cetina, Karin, and Werner Reichmann. 2015. Epistemic Cultures. International Encyclopedia of the Social & Behavioral Sciences (Second Edition), 873–880. [Google Scholar] [CrossRef]
- Master, Lawrence L., Don Faber-Langendoen, Roxanne Bittman, Geoffrey A. Hammerson, Bonnie Heidel, Leah Ramsay, Kristin Snow, Andy Teucher, and Adele Tomaino. 2012. NatureServe Conservation Status Assessments: Factors for Evaluating Species and Ecosystem Risk. Arlington: NatureServe. [Google Scholar]
- McCune, Jenny L., William L. Harrower, Stephanie Avery-Gomm, Jason M. Brogan, Anna-Mária Csergő, Lindsay N. K. Davidson, Alice Garani, Luke R. Halpin, Linda P. J. Lipsen, Christopher Lee, and et al. 2013. Threats to Canadian species at risk: An analysis of finalized recovery strategies. Biological Conservation 166: 254–65. [Google Scholar] [CrossRef]
- Ministry of Environment. 2006. Order—Category of Species at Risk; Victoria: British Columbia Ministry of Environment.
- NatureServe. 2023. NatureServe Explorer. Available online: https://explorer.natureserve.org/Search (accessed on 2 August 2023).
- Neimanis, V. P. 2011. Crown Land. Available online: https://www.thecanadianencyclopedia.ca/en/article/crown-land (accessed on 20 November 2023).
- NOAA. 2023. Species Directory: ESA Threatened & Endangered. Available online: https://www.fisheries.noaa.gov/species-directory/threatened-endangered (accessed on 2 August 2023).
- Office of Protected Resources. 2023. Candidate Species under the Endangered Species Act. Available online: https://www.fisheries.noaa.gov/endangered-species-conservation/candidate-species-under-endangered-species-act (accessed on 4 December 2023).
- Olive, Andrea. 2014a. Canada’s strategy for species at risk. In Land, Stewardship, and Legitimacy: Endangered Species Policy in Canada and the United States. Toronto: University of Toronto Press, pp. 49–73. [Google Scholar]
- Olive, Andrea. 2014b. Introduction. In Land, Stewardship, and Legitimacy: Endangered Species Policy in Canada and the United States. Toronto: University of Toronto Press, pp. 3–24. [Google Scholar]
- Olive, Andrea. 2014c. Lake Erie Islands Case Study. In Land, Stewardship, and Legitimacy: Endangered Species Policy in Canada and the United States. Toronto: University of Toronto Press, pp. 144–70. [Google Scholar]
- Pecl, Gretta T., Miguel B. Araújo, Johann D. Bell, Julia Blanchard, Timothy C. Bonebrake, I-Ching Chen, Timothy D. Clark, Robert K. Colwell, Finn Danielsen, Birgitta Evengård, and et al. 2017. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 355: eaai9214. [Google Scholar] [CrossRef]
- Ray, Justina C., Jaime Grimm, and Andrea Olive. 2021. The biodiversity crisis in Canada: Failures and challenges of federal and sub-national strategic and legal frameworks. FACETS 6: 1044–68. [Google Scholar] [CrossRef]
- Raymond, Calla V., Lina Wen, Steven J. Cooke, and Joseph R. Bennett. 2018. National attention to endangered wildlife is not affected by global endangerment: A case study of Canada’s species at risk program. Environmental Science & Policy 84: 74–79. [Google Scholar] [CrossRef]
- Raymond, Sarah, Amy L. W. Schwartz, Robert J. Thomas, Elizabeth Chadwick, and Sarah E. Perkins. 2021. Temporal patterns of wildlife roadkill in the UK. PLoS ONE 16: e0258083. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. 2022. R: A Language and Environment for Statistical Computing Version 4.1.1. Vienna: R Foundation for Statistical Computing. [Google Scholar]
- Rüter, Stefan, Claire C. Vos, Michiel van Eupen, and Hilke Rühmkorf. 2014. Transboundary ecological networks as an adaptation strategy to climate change: The example of the Dutch—German border. Basic and Applied Ecology 15: 639–50. [Google Scholar] [CrossRef]
- SARA. 2002. Species at Risk Act, S.C. c. 29. Available online: https://laws.justice.gc.ca/eng/acts/s-15.3/ (accessed on 2 August 2023).
- Smith, David R., Nathan L. Allan, Conor P. McGowan, Jennifer A. Szymanski, Susan R. Oetker, and Heather M. Bell. 2018. Development of a Species Status Assessment Process for Decisions under the U.S. Endangered Species Act. Journal of Fish and Wildlife Management 9: 302–20. [Google Scholar] [CrossRef]
- Thornton, Daniel H., Aaron J. Wirsing, Carlos Lopez-Gonzalez, John R. Squires, Scott Fisher, Karl W. Larsen, Alan Peatt, Matt A. Scrafford, Ron A. Moen, Arthur E. Scully, and et al. 2018. Asymmetric cross-border protection of peripheral transboundary species. Conservation Letters 11: e12430. [Google Scholar] [CrossRef]
- Titley, Mark A., Stuart H. M. Butchart, Victoria R. Jones, Mark J. Whittingham, and Stephen G. Willis. 2021. Global inequities and political borders challenge nature conservation under climate change. Proceedings of the National Academy of Sciences 118: e2011204118. [Google Scholar] [CrossRef]
- Turcotte, Audrey, Natalie Kermany, Sharla Foster, Caitlyn A. Proctor, Sydney M. Gilmour, Maria Doria, James Sebes, Jeannette Whitton, Steven J. Cooke, and Joseph R. Bennett. 2021. Fixing the Canadian Species at Risk Act: Identifying major issues and recommendations for increasing accountability and efficiency. FACETS 6: 1474–94. [Google Scholar] [CrossRef]
- USFWS. 1973. Endangered Species Act of 1973 as Amended through the 108th Congress. U.S Department of the Interior. Available online: https://www.fws.gov/sites/default/files/documents/endangered-species-act-accessible.pdf (accessed on 27 November 2023).
- USFWS. 2017. U.S. Fish & Wildlife Service: Candidate Species: Section 4 of the Endangered Species Act. Available online: https://www.fws.gov/sites/default/files/documents/Candidate-Species.pdf (accessed on 2 August 2023).
- Vincent, C. H., L. A. Hanson, and L. F. Bermejo. 2020. Federal Land Ownership: Overview and Data. Available online: https://sgp.fas.org/crs/misc/R42346.pdf (accessed on 27 November 2023).
- Waples, Robin S., Marta Nammack, Jean Fitts Cochrane, and Jeffrey A. Hutchings. 2013. A Tale of Two Acts: Endangered Species Listing Practices in Canada and the United States. BioScience 63: 723–34. [Google Scholar] [CrossRef]
- WDFW. 2023. Washington State Listed Species. Available online: https://wdfw.wa.gov/species-habitats/at-risk/listed (accessed on 2 August 2023).
- Westwood, Alana R., Sarah P. Otto, Arne Mooers, Chris Darimont, Karen E. Hodges, Chris Johnson, Brian M. Starzomski, Cole Burton, Kai M. A. Chan, Marco Festa-Bianchet, and et al. 2019. Protecting biodiversity in British Columbia: Recommendations for developing species at risk legislation. FACETS 4: 136–60. [Google Scholar] [CrossRef]
| Species | SARA | ESA | IUCN |
|---|---|---|---|
| Pallid bat Antrozous pallidus | Threatened | Not listed | Least Concern |
| Black-tailed prairie dog Cynomys ludovicianus | Threatened | Not listed | Least Concern |
| Ord’s kangaroo rat Dipodomys ordii | Endangered | Not listed | Least Concern |
| Spotted bat Euderma maculatum | Special Concern | Not listed | Least Concern |
| Wolverine Gulo gulo | Special Concern | Not listed | Least Concern |
| Sowerby’s beaked whale Mesoplodon bidens | Special Concern | Not listed | Least Concern |
| Woodland vole Microtus pinetorum | Special Concern | Not listed | Least Concern |
| Haida ermine Mustela haidarum | Threatened | Not listed | Not listed |
| Little brown myotis Myotis lucifugus | Endangered | Not listed | Endangered |
| Collared pika Ochotona collaris | Special Concern | Not listed | Least Concern |
| Tri-coloured bat Perimyotis subflavus | Endangered | Not listed | Vulnerable |
| Western harvest mouse Reithrodontomys megalotis (dychei) | Endangered * | Not listed | Not listed |
| Eastern mole Scalopus aquaticus | Special Concern | Not listed | Least Concern |
| Townsend’s mole Scapanus townsendii | Endangered | Not listed | Least Concern |
| Pacific water shrew Sorex bendirii | Endangered | Not listed | Least Concern |
| Grey fox Urocyon cinereoargenteus | Threatened | Not listed | Least Concern |
| Swift fox Vulpes velox | Threatened | Not listed | Least Concern |
| Canada lynx Lynx canadensis | Not listed | Threatened | Least Concern |
| Pacific marten Martes caurina | Not listed | Threatened | Least Concern |
| Sperm whale Physeter macrocephalus | Not listed | Endangered | Vulnerable |
| Mountain beaver Aplodontia rufa (nigra) | Special Concern | Endangered * | Least Concern |
| Sea otter Enhydra lutris (kenyoni) | Special Concern | Threatened * | Endangered |
| Sea otter Enhydra lutris (nereis) | Special Concern | Threatened * | Endangered |
| Polar bear Ursus maritimus | Special Concern | Threatened | Vulnerable |
| Wood bison Bison bison athabascae | Threatened | Threatened | Not listed |
| Northern myotis Myotis septentrionalis | Endangered | Endangered | Near Threatened |
| Black-footed ferret Mustela nigripes | Extirpated | Endangered | Endangered |
| North Atlantic right whale Eubalaena glacialis | Endangered | Endangered | Critically Endangered |
| North Pacific right whale Eubalaena japonica | Endangered | Endangered | Endangered |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raymond, S.; Perkins, S.E.; Garrard, G. The Species at Risk Act (2002) and Transboundary Species Listings along the US–Canada Border. Humanities 2024, 13, 38. https://doi.org/10.3390/h13010038
Raymond S, Perkins SE, Garrard G. The Species at Risk Act (2002) and Transboundary Species Listings along the US–Canada Border. Humanities. 2024; 13(1):38. https://doi.org/10.3390/h13010038
Chicago/Turabian StyleRaymond, Sarah, Sarah E. Perkins, and Greg Garrard. 2024. "The Species at Risk Act (2002) and Transboundary Species Listings along the US–Canada Border" Humanities 13, no. 1: 38. https://doi.org/10.3390/h13010038
APA StyleRaymond, S., Perkins, S. E., & Garrard, G. (2024). The Species at Risk Act (2002) and Transboundary Species Listings along the US–Canada Border. Humanities, 13(1), 38. https://doi.org/10.3390/h13010038






