Abstract
Mineable platinum group metal (PGM) deposits are rare and found in relatively few areas of the world. At the same time, the use of PGM is predicted to expand in green technology and energy applications, and PGMs are consequently currently listed as European Union critical metals. Increased mineralogical complexity, lower grade ores, and recent PGM production expansions give rise to the evaluation of the value chain of the capital-intensive conventional matte smelting treatment and other processing possibilities of the ore. This article will review the processes and value chain developed to treat ores for PGM recovery, highlighting hydrometallurgical refining approaches. It groups processes according to their rationale and discusses the special features of each group.
1. Introduction
The platinum group metals (PGMs) are a family of six metals. They are rare and possess extraordinary physical and chemical properties, e.g., resistance to corrosion and oxidation, electrical conductivity, and catalytic activity. The most economically important of the PGMs are platinum, palladium, and rhodium while ruthenium, iridium, and osmium are less prevalent and less in demand [1].
The demand for PGM is fundamentally strong. The demand for platinum breaks down into four segments: automotive, jewellery, industrial (the chemical and petroleum industries, the dental and medical sectors, the glass industry, and a range of other end uses), and investment [2]. The demand for PGMs has risen significantly after the introduction of catalytic converters to control vehicle exhaust emissions, with the catalyst application accounting for a 6% market share for platinum and 16% for ruthenium in 2012 [3].
Over the last five years, between 72% and 78% of total annual platinum supply has come from primary mining output [4] and the rest originates from secondary materials such as spent catalysts and electronic scrap. Most of the current primary PGM production is derived from sulphide ores that typically also contain nickel, copper, and other metals. PGM annual production amounts to around 400 t, [5] of which platinum accounts for approximately 192 t and ruthenium approximately 12 t [6].
Grandell et al. [6] have predicted a path for the global energy system with an assumed annual 2% increase in global gross domestic product, resulting in 290% and 73% cumulative consumption of the world’s known reserves of platinum and ruthenium, respectively by 2050. This highlights the critical nature of this metal group and justifies the increasing focus on the primary and secondary processing of PGMs. This article will discuss primary PGM production.
2. Regional and Mineralogical Aspects of PGM Production
According to Jones [1], PGMs are commonly associated with nickel-copper sulphides in magmatic rocks. PGMs are produced either as primary products or by-products of the nickel and copper, depending on the relative concentrations of the metals in the ore. Cole and Ferron [7] also describe ores in which PGM values are noted but not recovered due to low concentrations or recovered as a by-product with little or no economic advantage for the primary producer. PGMs originating from these sources are for instance recovered from copper refineries as a by-product in the United States and Canada.
Mineable PGM deposits are very rare and found in relatively few areas of the world. South Africa dominates PGM world production with 58%. Russia accounts for a further 26%, most of this as a co-product of nickel mining. Nearly all of the rest comes from Zimbabwe, Canada, and the United States. One third of PGMs are produced as co-products of nickel mining [5].
The largest known PGM deposit, the Bushveld Complex in South Africa, contains more than two thirds of the world’s reserves of PGMs. The Great Dyke in Zimbabwe is the second largest known deposit of platinum. Other primary PGM-rich deposits include the Stillwater deposit of the United States and the Lac des Isles deposit of Canada. PGMs are produced in significant quantities as by-products from the Norilsk-Talnakh area of Russia and the Sudbury deposit of Canada [1]. Other deposits occur e.g., in Finland and China. Most deposits are fairly small, less than 100 million tons of ore [8].
PGMs have diverse associations and the primary minerals associated with PGMs are pyrrhotite, chalcopyrite, and pentlandite. Chalcopyrite and pentlandite are generally well recovered, as are any PGMs in their lattice or present in any platinum group mineral blebs that they may contain. PGMs associated with pyrrhotite are more challenging to recover and may be largely rejected at the mine site, e.g., in the Sudbury basin, to minimize operating costs and environmental pollution [9].
Liddell and Adams [10] state that pentlandite, chalcopyrite, and cobaltiferrous pyrite are commonly associated with PG mineralisation and that pyrrhotite is also seen in PGM ores. They point out that PG minerals present in the ore have varying associations with base metal sulphide minerals and gangue minerals. A move to increasingly finer grind sizes has occurred as a response to the trend of maximising recovery. Fine gangue particles tend to report to the concentrate more readily than coarse gangue particles.
Gold is often associated with PGM deposits and is treated as part of a family together with platinum, palladium, and rhodium, collectively known as 4E. Nickel and copper are the most prevalent base metals in sulphide ores containing PGMs. Chromite (used to derive chrome) is another significant by-product while cobalt, silver, selenium, and tellurium are found and recovered in trace quantities [11]. Most deposits contain 1–5 g/t 4E [8].
The International Mineralogical Association has recognized nearly 110 different types of platinum group carrying minerals. PGMs form a number of minerals ranging from sulphides to tellurides, antimonides to arsenides, and alloys to native metals [9].
3. PGM Processing and Its Value Chain
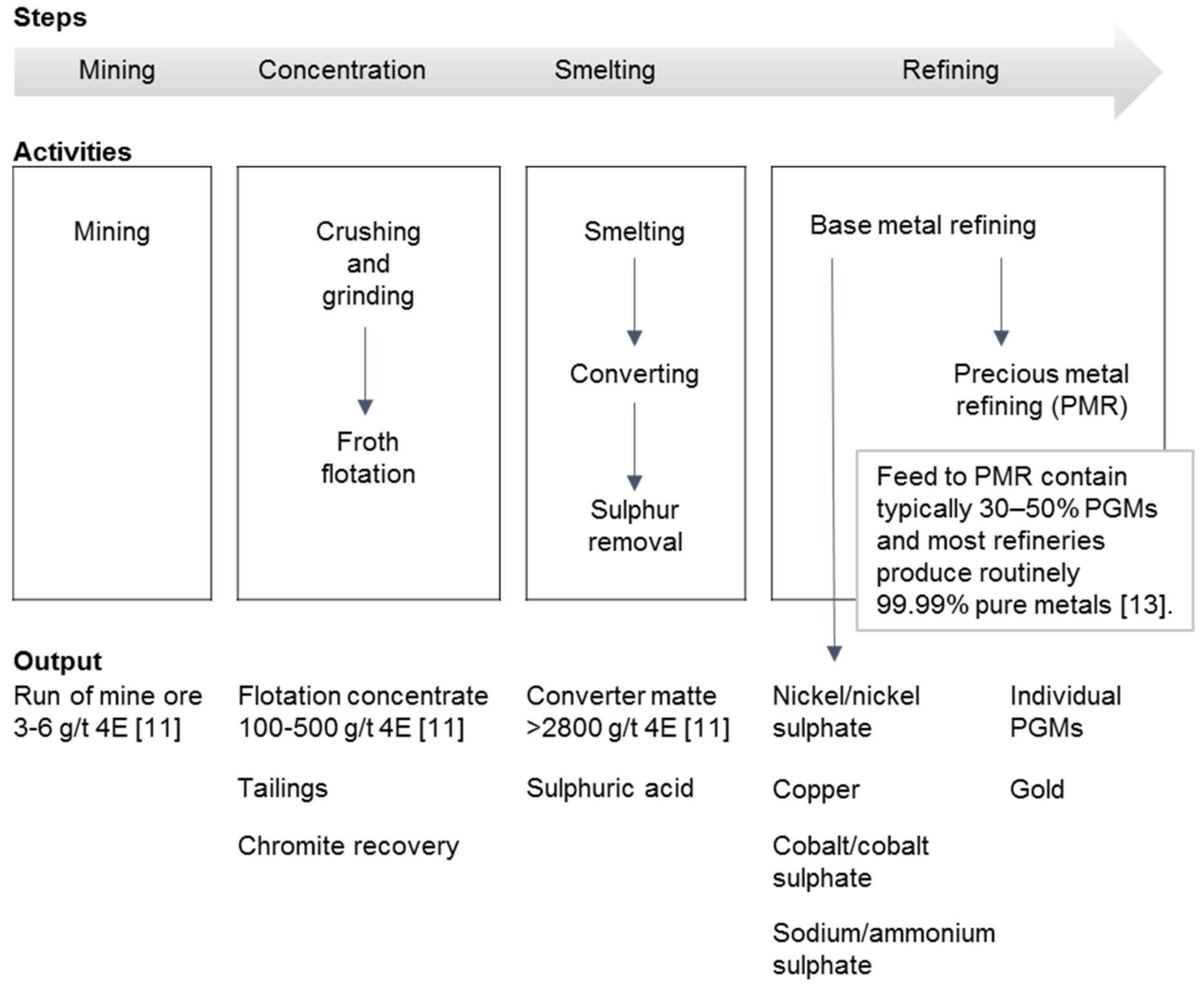
PGM processing consists principally of four steps (Figure 1). PGMs are recovered through underground and open pit mining from poly-metallic sulphide ores [11]. They are initially concentrated by flotation. Conventionally, flotation concentrates are enriched by smelting, i.e., iron is combined with silica and lime, oxidised, and separated as a slag leaving the sulphide minerals in a separate matte phase. The smelter can be a base metal smelter or a PGM smelter [12]. The matte is refined using reverse leaching in which most of the accompanying minerals such as copper, nickel, cobalt, and any residual iron sulphides are dissolved. The leach solutions are processed to recover the base metals. In the case of the PGM smelter and the base metal smelter, an upgraded PGM concentrate is left behind or PGMs eventually find their way into the anodic slimes, respectively. PGM concentrate and anodic slimes are further treated at the PGM refinery or precious metal refinery/plant to recover the PGMs and precious metals [7].
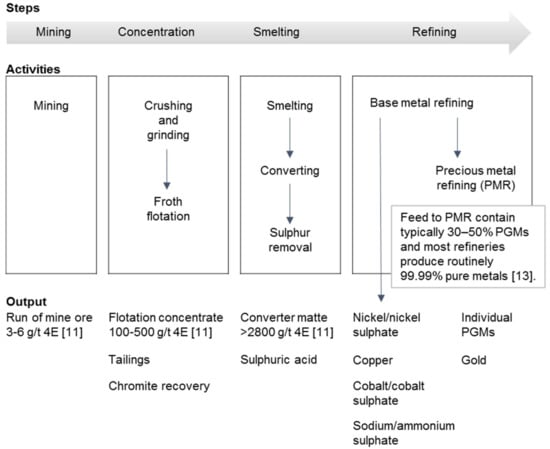
Figure 1.
Simplified platinum group metal (PGM) processing chain (conventional matte smelting treatment). 4E = Pt, Pd, Rh, and Au. Adapted from Ndlovu [11] and Cramer [13].
In the case of the PGM smelter, electric furnaces are typically used and operated at 1350 °C. Temperatures of up to 1600 °C may be necessary for chromite-containing concentrates owing to the higher content of chromium and magnesium oxides. The furnace matte is further processed by converting, i.e., air is blown into the molten charge to oxidise and remove the iron and its associated sulphur. The converter matte is typically granulated. Anglo Platinum slow-cools the matte, which results in PGM concentrate on the grain boundaries and tends to form ferromagnetic species that are recovered by magnetic separation. The ferromagnetic fraction is treated to dissolve the associated base metals leaving an enriched PGM concentrate for the precious metal refinery [7].
The four major PGM mining companies, namely Anglo Platinum, Norilsk, Impala Platinum, and Lonmin Platinum, all South African apart from the Russian Norilsk, are integrated from mining to refining and account for 80% of the market [14]. One of them, Lonmin, outsources the refining of excess base metals [11]. According to Cramer [15], their nickel production is sold to other nickel refiners as nickel sulphate. Levine et al. [16] state that Norilsk outsources PGM-containing copper solids at the Harjavalta refinery. Norilsk relies on third party PGM refiners according to Ndlovu [11].
Cramer [13] stated in 2001 that several of the South African PGM producers reviewed their refining capacities in order to align their base metals capacities with the major PGM production expansions planned over the next several years. Anglo Platinum found that PGM processing capacity (for PGM dominant ores) was not easily globally traded, with very few concentrated alternatives, namely other integrated PGM processing facilities. Their choice was to keep deploying capacity downstream [11]. In 2011, an expansion project at Anglo Platinum’s base metals refinery in Rustenburg was completed and operational [17].
Cramer [15] stated in 2008 that a couple of dozen minor platinum mining companies were being set up in Southern Africa. Most of them intended to mine, concentrate the PG minerals to flotation concentrate, and sell the product. There was also several ongoing studies, in particular smelter studies, on further upgrading the product as another intermediate and saleable product. Further investment in process capacity was often considered in conjunction with other smaller mining companies faced with limited capacity for concentrate smelting and base metal refining within the local platinum industry or together with one of the major platinum companies.
The major factors of process value come down to capital and operating efficiency. Generally, the focus to drive value is largely in the mining and concentration steps in which the most losses are incurred. For example, total losses in the mining step before the process, account for 27% compared with 15% overall processing losses, of which the concentration step accounts for 13% at an underground platinum mine. Non-integrated mining companies produce approximately 10% of the major PGM mining companies’ processing input. Of the operating costs, smelting and refining account for 15% (Table 1). [11] The capital investments to process PGMs from flotation concentrate are very large and a significant barrier to entry for smaller mining companies [15].

Table 1.
Value chain for PGM processing (adapted from Ndlovu [11]).
Platinum producers evaluate process routes and their value chain alternative to conventional matte smelting treatment, which is presented in Figure 1. Projects are evaluated for cost-effective processing with low-grade ores and in some cases higher content of deleterious elements such as chromium [18]. Hydrometallurgical plants are best suited for small operations and this is the direction usually taken [7]. Smelters have a high capital cost and favour large capacities. In addition, sulphur dioxide generation at the smelters requires conversion to sulphuric acid due to increasingly stringent regulations, which have a large negative value in the overall economics [19] and a considerably positive environmental impact.
4. Base Metal Refining
According to Cole and Ferron [7], all PGM base metal refineries in the western world use Sherrit-Gordon’s sulphuric acid pressure leach process. The process flow sheets vary in each operation. The now closed Hartley Platinum operations applied the Outokumpu process. Both processes have the same rationale and the chemistry underlying both processes is the same. In general, the same processes used in nickel and copper-nickel refineries are also used for PGM base metal refineries. The following focuses on the latter.
Finely ground matte (or the non-magnetic matte at the Anglo Platinum operation) is leached in a number of stages under increasingly more oxidizing conditions to produce a residue containing PGMs and typically nickel- and copper-rich solutions. Copper is recovered as electrowon cathode and nickel as metal powder by hydrogen reduction or electrowon cathode. At the Stillwater refinery, a bulk nickel-copper solution is produced and shipped to an outside nickel refinery. The residue can be subjected to further treatment to upgrade the PGM content before the final residue passes to the PGM refinery [7].
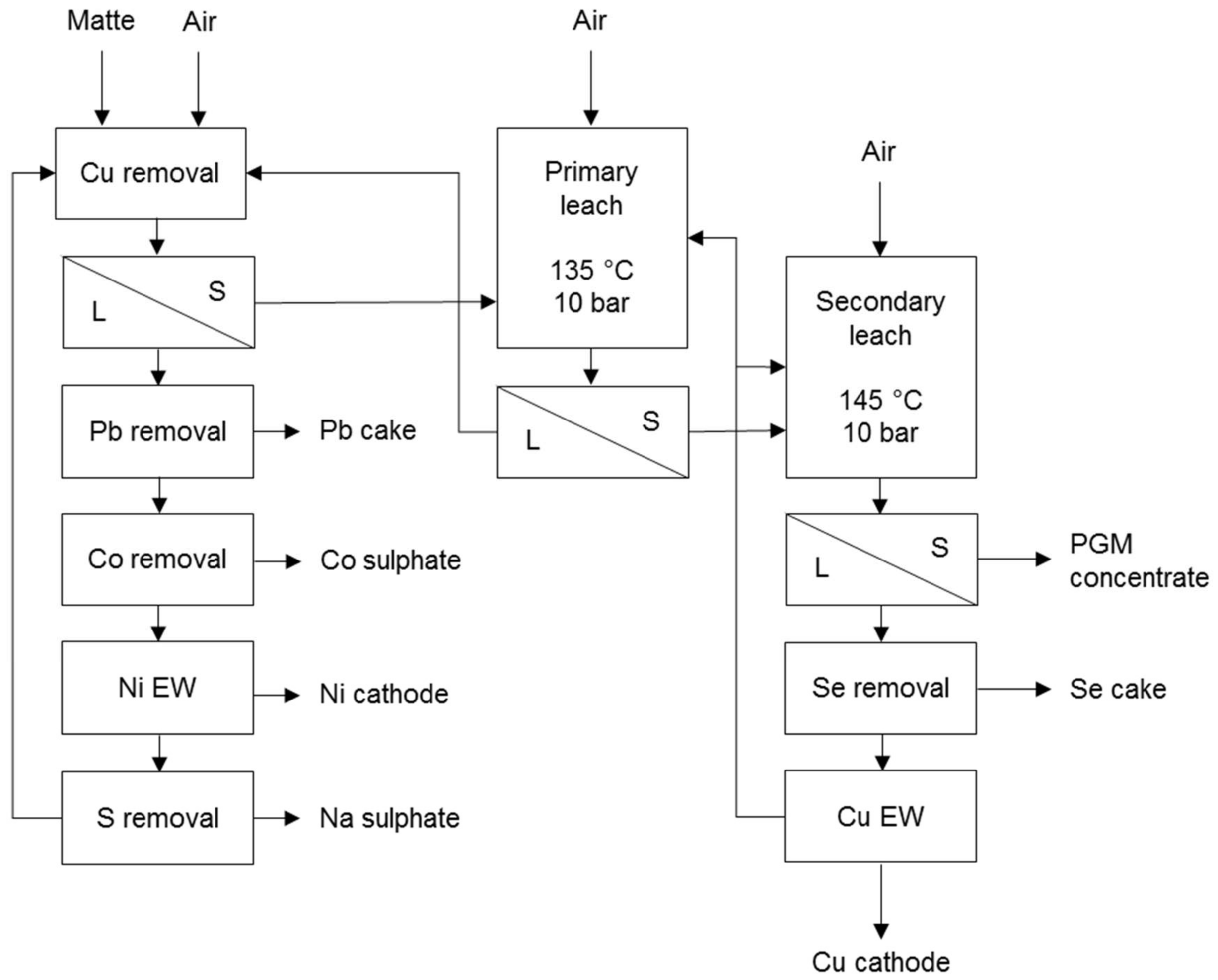
A typical example is the process used at Rustenburg Base Metals Refiners that treats the non-magnetic nickel-copper matte from a slow cooling matte separation process (Figure 2). The initial stage, i.e., copper removal, operates under mild conditions to leach some nickel and cobalt and aims to precipitate copper from the nickel sulphate solution with fresh matte. The primary leach stage aims to dissolve all the nickel and some copper from the matte while the secondary leach stage aims to dissolve all the remaining copper and some iron from the matte [7].
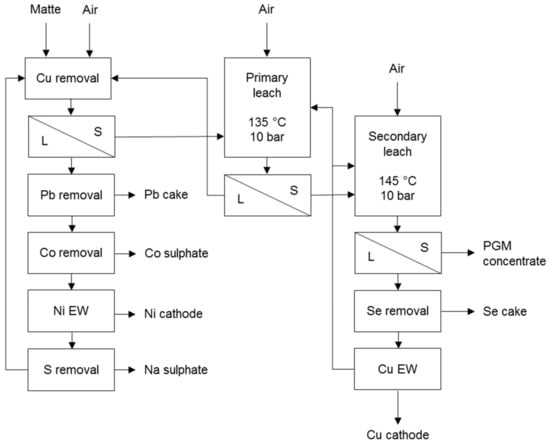
Figure 2.
Simplified flow sheet of Rustenburg Base Metals Refinery (adapted from Hofirek & Halton [20]).
Fresh matte consumes the available acid and iron precipitates during the decopperisation. Iron can also be removed in a special high temperature step prior to copper removal. The neutralised iron- and copper-free nickel solution is suitable for the recovery of nickel and cobalt products. The copper solution undergoes a selenium removal stage that also removes any co-dissolved PGM prior to electrowinning [7].
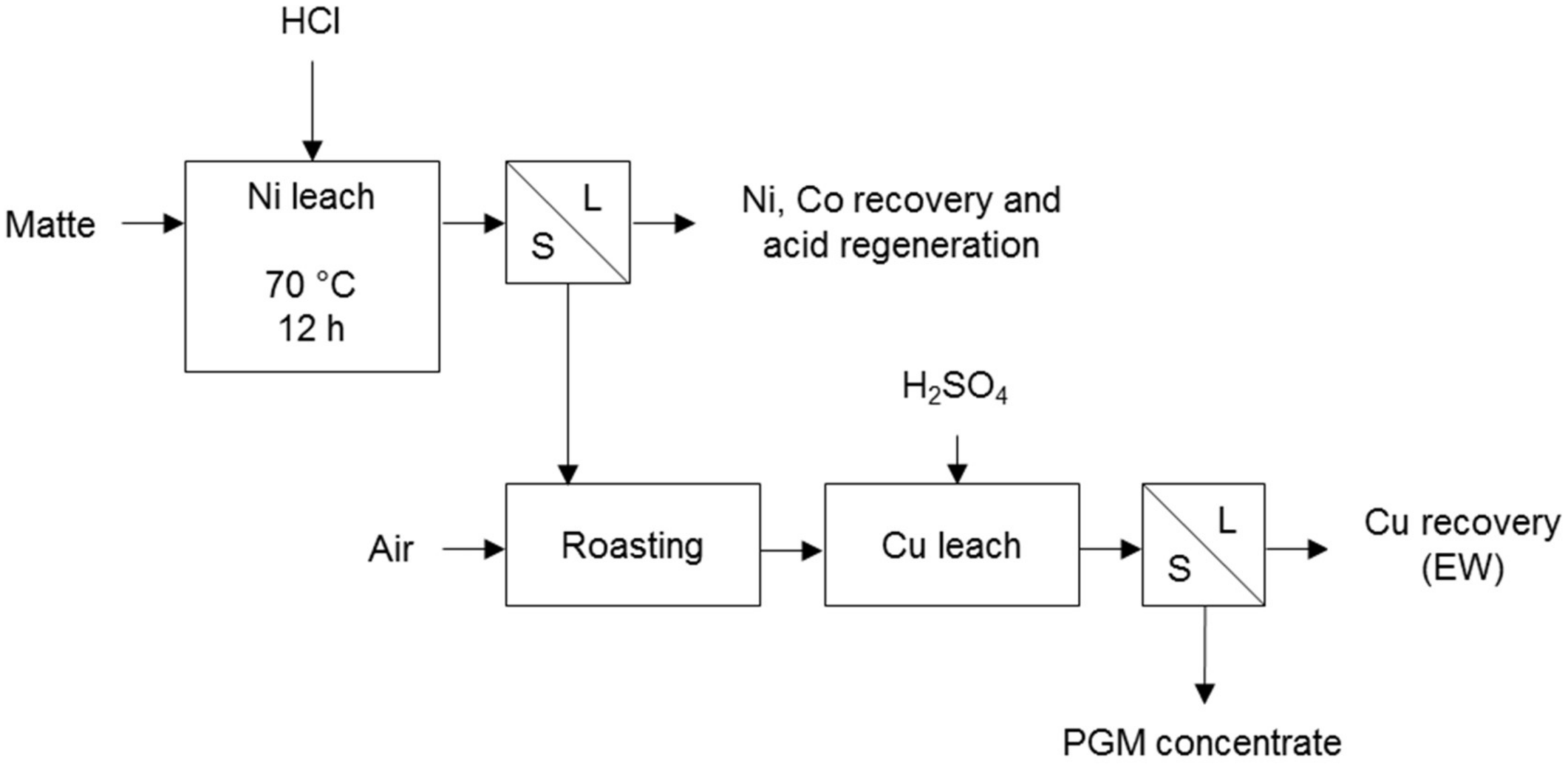
Liddell et al. [21] also present the Falconbridge process for the removal of base metals from matte (Figure 3). The nickel is dissolved with hydrochloric acid in a non-oxidative leach and the solution is purified, i.e., sulphur is removed by oxidation, and iron and cobalt by solvent extraction. The nickel is then recovered and the acid regenerated. The leach residue is roasted and copper is then leached with sulphuric acid. The copper is electrowon from the solution and the residue passes to the PGM refinery. PGMs are a by-product of mostly nickel, copper, and cobalt in this process according to Cole & Ferron [7].
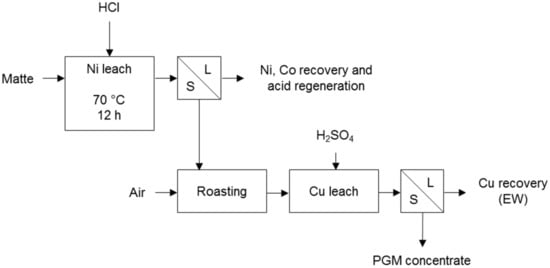
Figure 3.
Falconbridge method for base metal removal (adapted from Thornhill et al. [22]).
5. PGM Refining
The individual PGMs are refined totally or partially using a classical refining process with solvent extraction and molecular recognition developments. The classical refining process is based on PGM chloride chemistry. It involves successive steps of precipitation and redissolution to purify the metals, followed by thermal reduction to metal. However, it gives a poor first time yield for refined metals and requires lengthy refining times, e.g., up to six months for rhodium [7].
Crundwell et al. [23] classify PGM refining processes by the technique used to separate platinum and palladium from one another. Of the major South African PGM mining companies, Lonmin Platinum uses the precipitation process, Anglo Platinum the solvent extraction process, and Impala Platinum the ion-exchange process (Table 2). The solvent extraction process is also used, by Johnson Matthey and Vale for instance.

Table 2.
Extraction methods in a few PGM refineries (adapted from Crundwell et al. [23]).
6. Non-Smelter-Based Processes
Non-smelter-based processes developed for PGM extraction typically include sequential base metal leach and PGM leach steps or leaching the PGMs together with base metals. Both approaches are followed by solution purification operations for the recovery of metal or intermediate products.
The processes include sulphide treatment to break down the sulphide matrix to liberate the base and PG metals locked in the matrix. Consequently, process flow sheets commonly comprise at least roasting, pressure oxidation, or fine grinding prior to leaching, or the matrix is attacked by bio or chemical oxidation during leaching. Pressure oxidation is used at conventional or mild temperatures to completely oxidise sulphides to sulphate or to oxidise them predominantly to sulphur, respectively. The latter requires a finer feed according to Milbourne et al. [12]. This may be done by ultrafine grinding, which produces particles sized within the 1 to 20 um range. Small enough particles allow disintegration of the leached mineral before the sulphur layer becomes thick enough to passivate it [8].
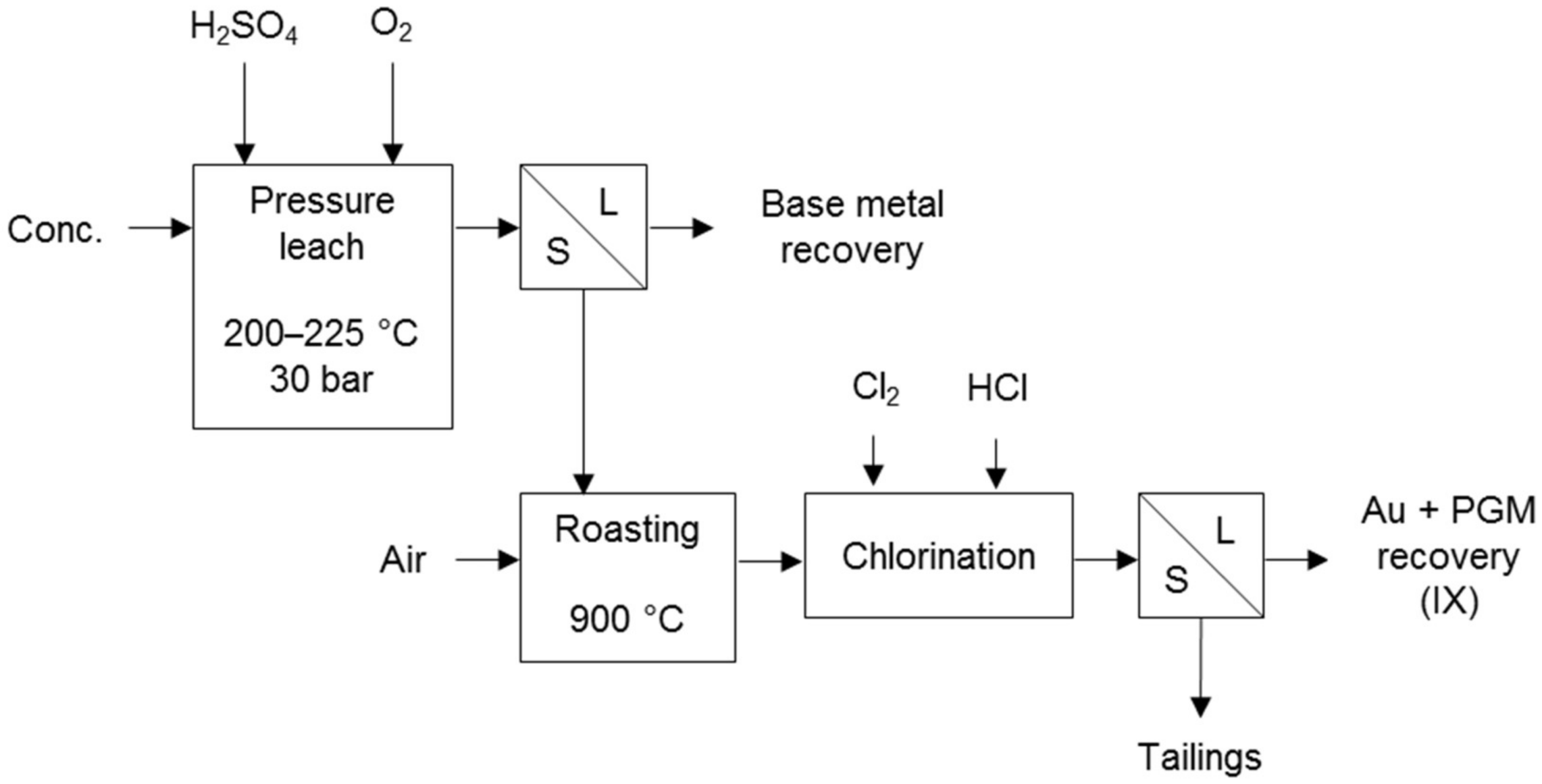
The Kell process (Figure 4) first selectively removes base metals and sulphur by pressure oxidation. The residue is subjected to roasting to render the PGMs amenable to recovery by chlorination. The conventional pressure oxidation of the concentrate converts all the sulphur to sulphate, avoiding sulphur dioxide in the roaster off-gas. This process was developed to treat PGM-bearing concentrates and has been successively tested on several concentrates [10], including Platreef concentrate from the Bushveld Complex of South Africa [18].
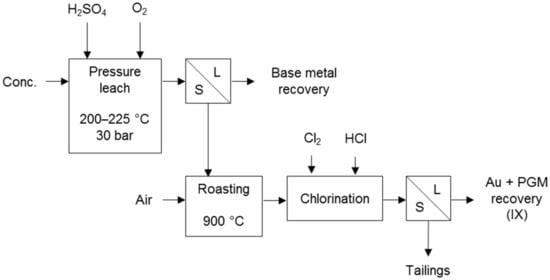
Figure 4.
Block diagram of the Kell process (adapted from Liddell & Adams [10]).
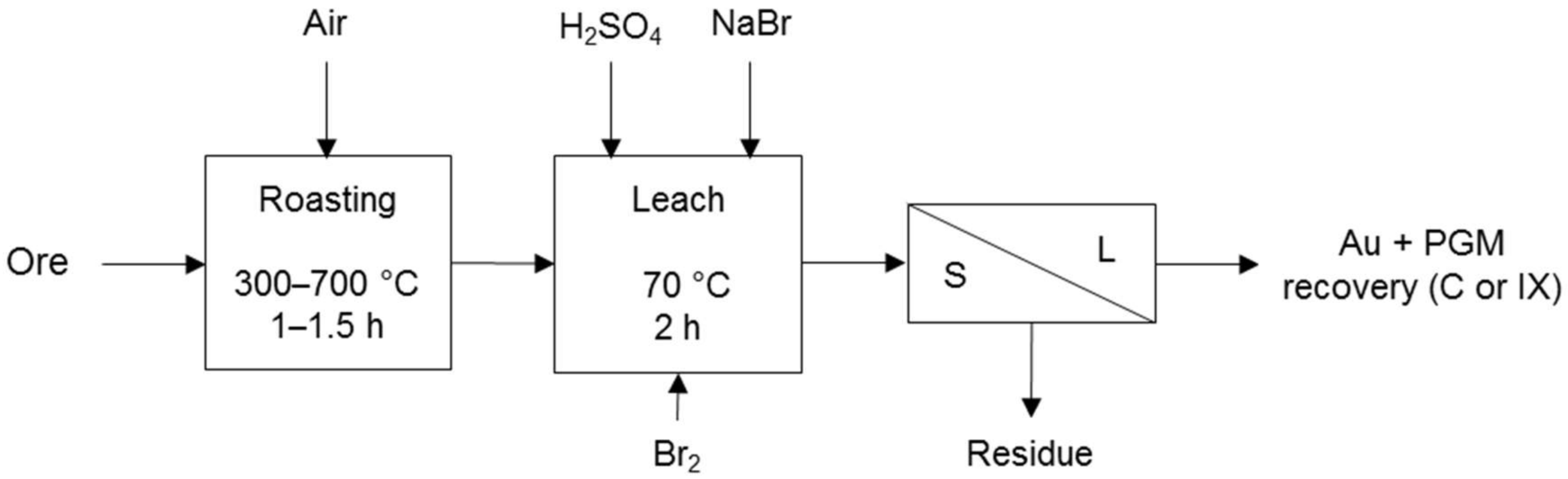
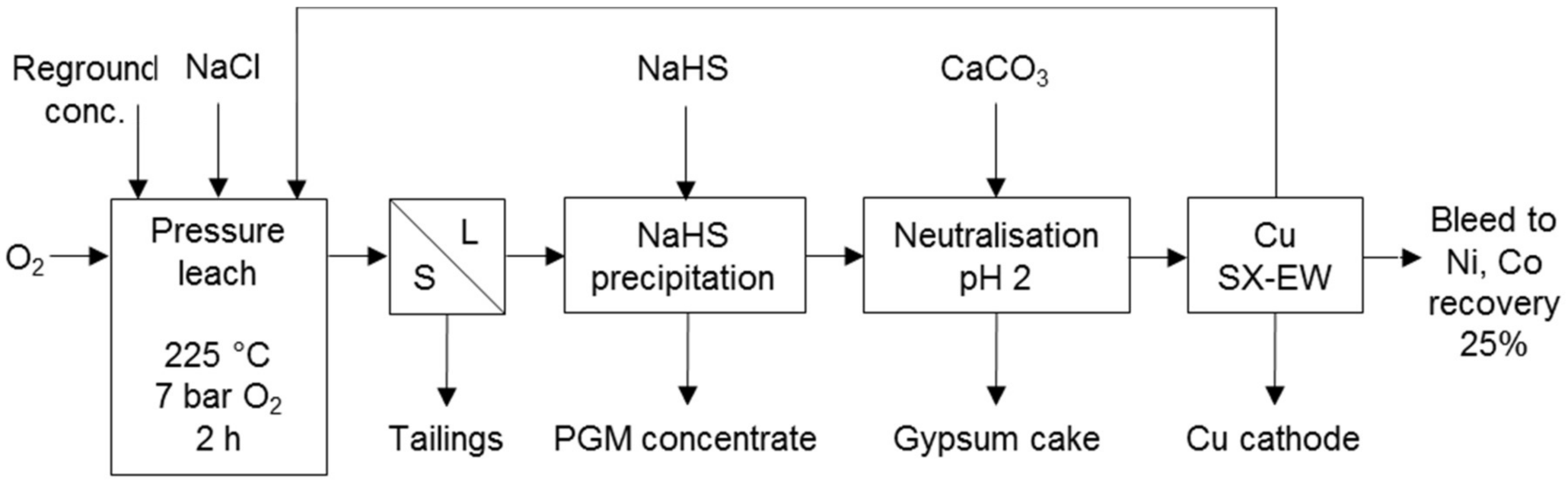
Processes that co-dissolve PG and base metals in a sulphate environment make use of halogens to promote PGM dissolution. These processes include the TML process (Figure 5) and the Platsol process (Figure 6). The former was developed to recover PGM from the oxidised portion of the Hartley deposit in Zimbabwe. The latter was developed to treat the NorthMet deposit in Minnesota, USA and has also been applied to treat other PGM/Cu–Ni concentrates, Cu–Au concentrates, Cu–Ni–PGM matte, Pt laterites and auto-catalysts [7]. Both processes use halogens for the formation of PGM complexes and the TML process also for redox potential control.
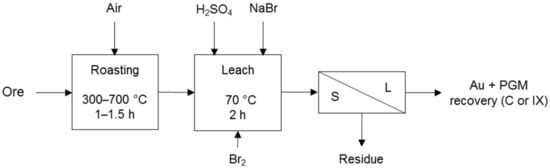
Figure 5.
Simplified TML process diagram for Hartley oxide ore. Other leach parameters were 100 g/L H2SO4, 10 g/L NaBr, >800 mV ORP with Br2 using Geobrom 3400 as oxidant, and 24–40% solids. Adapted from Cole and Ferron [7]. Geobrom 3400 is a sodium bromide solution in which liquid bromine is dissolved.
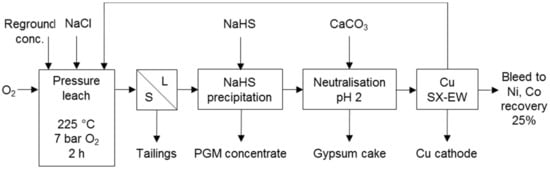
Figure 6.
Flow sheet for the Platsol treatment of NorthMet concentrate. NaCl addition was 5–20 g/L. Adapted from Cole and Ferron [7].
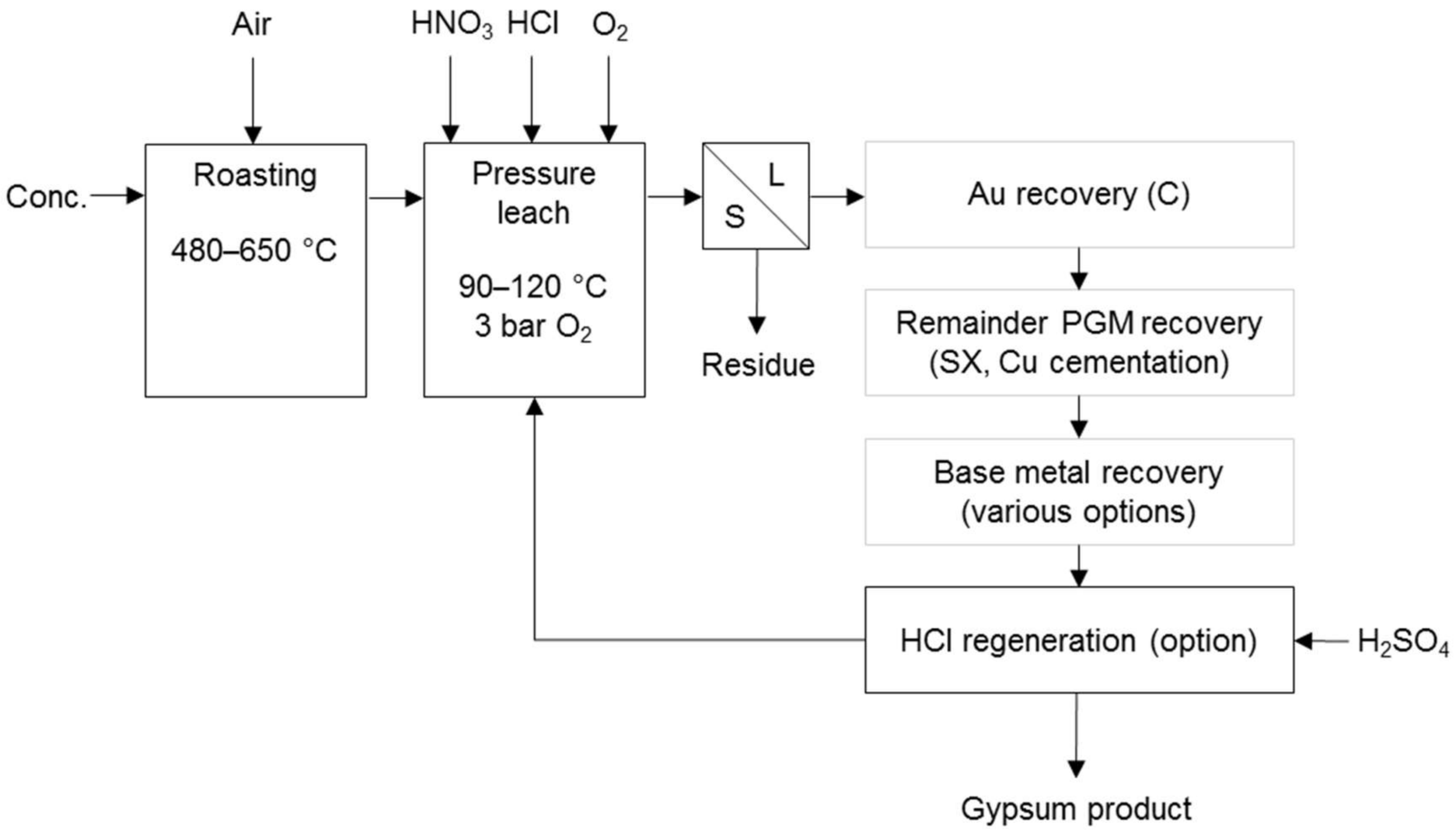
Chloride-based processes co-dissolve PG and base metals, promoting dissolution by other means. Cole and Ferron [7] state that the North American Palladium process (Figure 7) was developed for the treatment of Lac des Isles PGM sulphide concentrate. It uses pressure and hydrochloric acid with small amounts of nitric acid. Soluble nitrogen species transport oxygen to the surface of the solid particle, which enhances the rate of dissolution of metal sulphides. The nitric acid is continuously regenerated by the oxygen gas applied. Partial oxidising roasting was included in the flow sheet, as it increased the platinum recovery significantly [24].
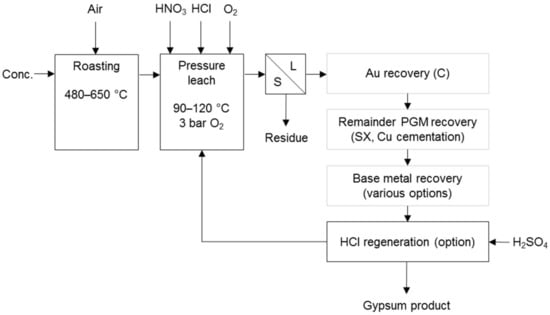
Figure 7.
Block diagram for the treatment of the Lac des Iles PGM concentrate (adapted from McDoulett & Reschke [24]).
Several chloride-based or chloride-aided sulphate processes such as the CESL [25], Outotec [26] and Intec [19] copper processes and HydroCopper [27] have been developed to treat copper sulphide concentrates. They exclude PGMs from consideration. This is also the case for the chloride-based Outotec gold process [28] developed to treat gold-bearing raw materials. Based on the literature, they have not been applied to treat PGM-dominant concentrates apart from some laboratory batch tests carried out with the Intec process and mentioned by Milbourne et al. [12].
Other direct leaching processes for the treatment of chalcopyrite copper concentrate have also been developed. These include the nitrogen species catalysed process and several other sulphate- based processes. Typically none focus on PGM recovery. Milbourne et al. [12] reviewed and theoretically discussed the dissolution and deportment of PGMs in many of these processes. Mpinga et al. [8] also reviewed these processes. However, test work is required to determine the behaviour of PGMs and the method of recovery among other process modifications needed. We will concentrate on reviewing the tested technology for PGM extraction.
Cyanide-based processes use elevated temperature or pressure during cyanidation to promote PGM dissolution. The processes that co-dissolve PG and base metals were apparently mainly developed to process oxide material with low copper concentrations, as the presence of sulphide minerals and high concentrations of copper in the cyanidation feed typically have an adverse effect on cyanidation.
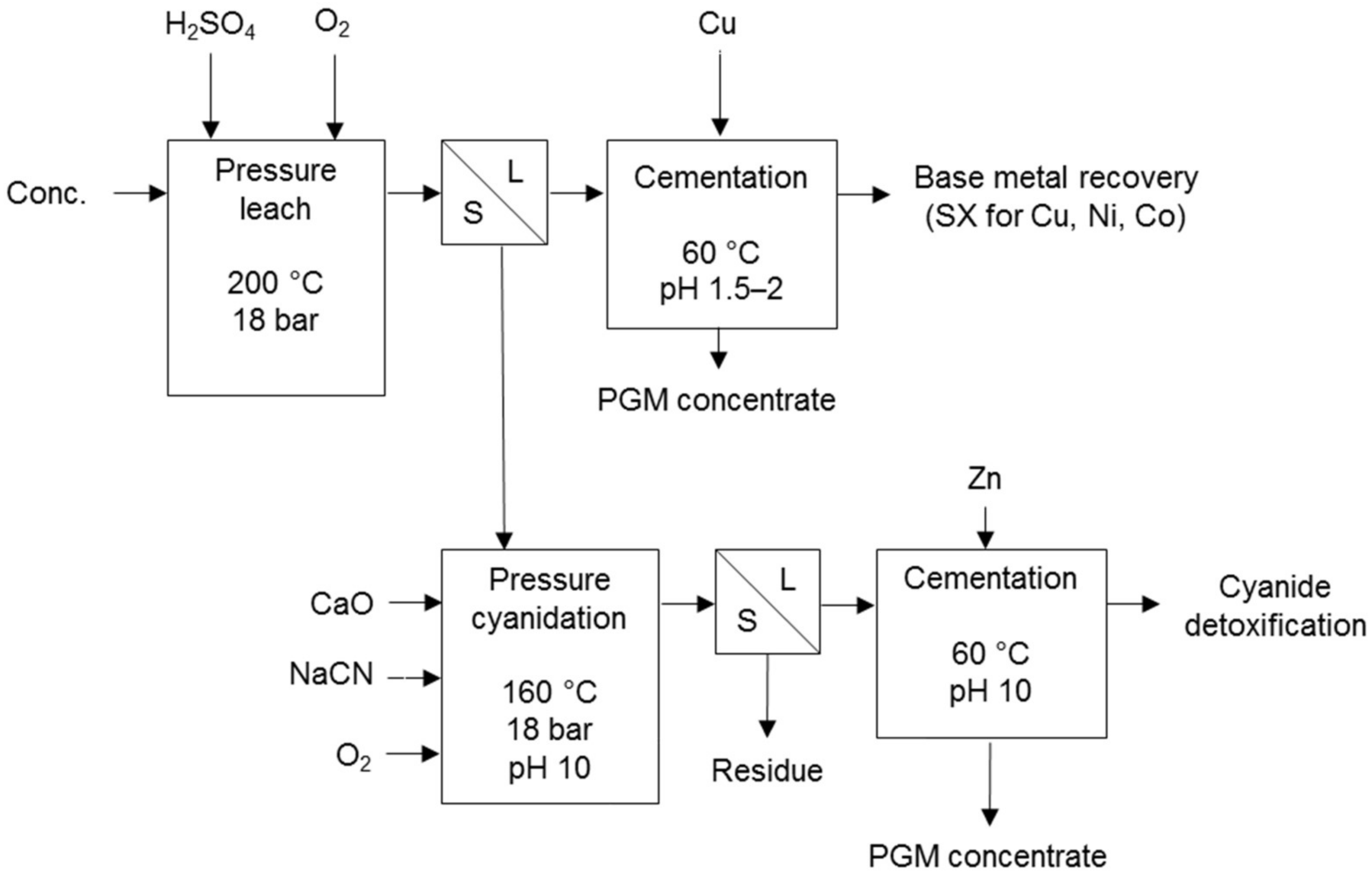
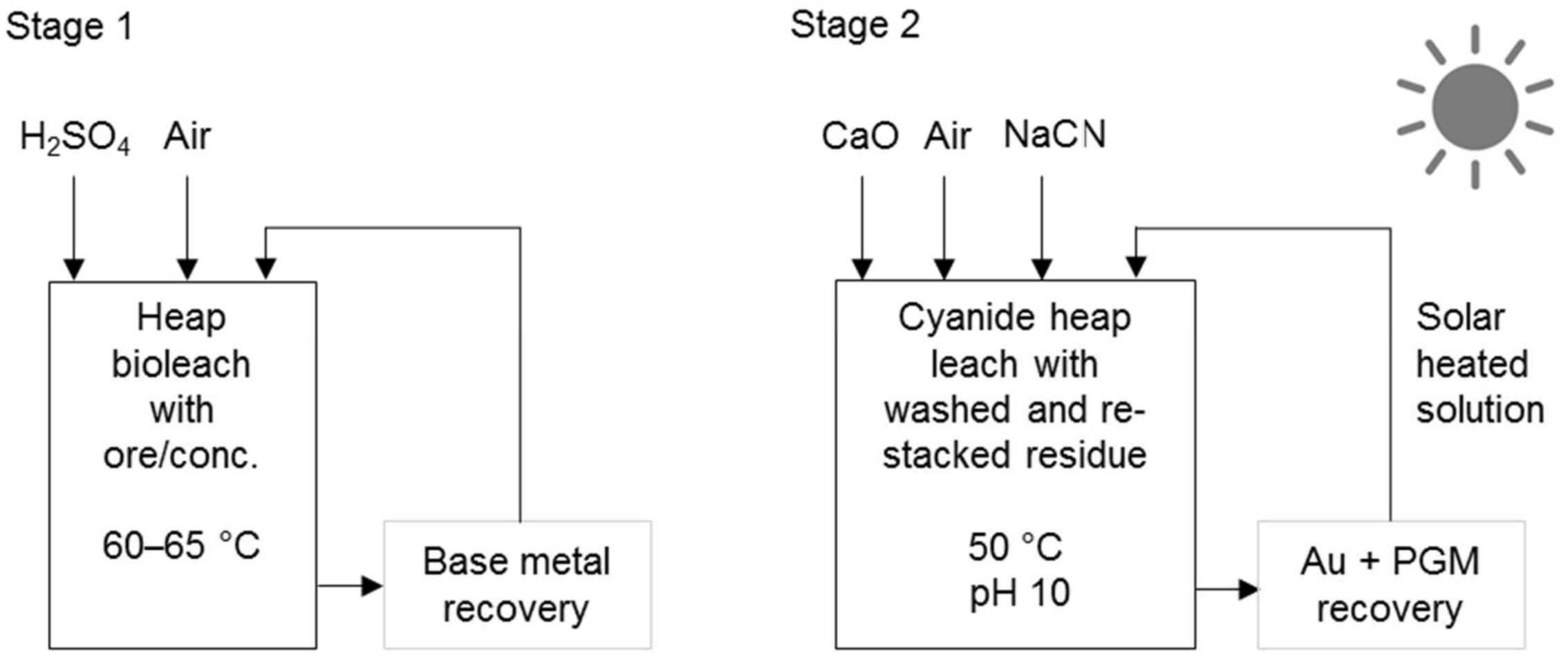
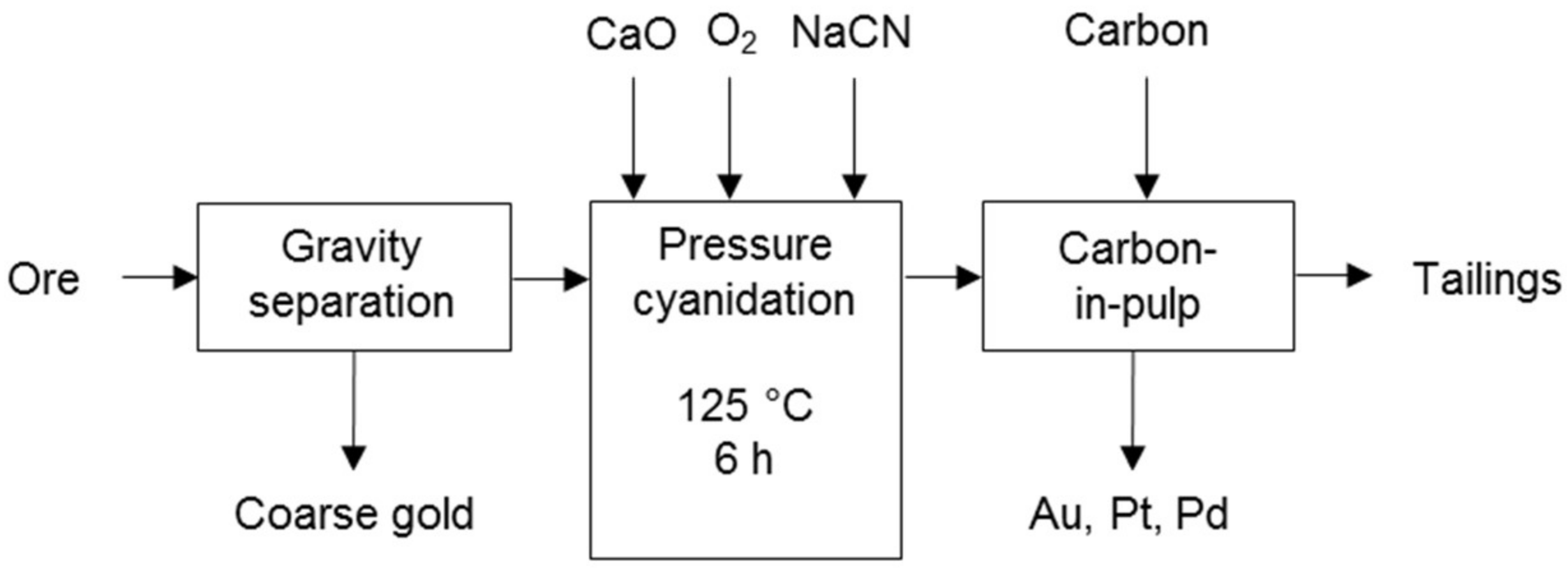
Processes that use cyanidation following base metal leach in a sulphate environment include the two-stage selective pressure leach process (Figure 8) and sequential heap leach process (Figure 9). The former has been tested for treating Jinbaoshan mine’s concentrate from China [29] and the latter for treating Platreef ore and its low-grade concentrate [30]. Conventional pressure oxidation avoids and bioleaching decreases the formation of ineffective thiocyanates in these processes.
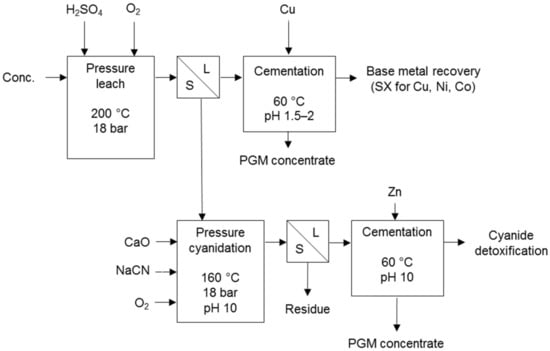
Figure 8.
Two-stage selective pressure leaching process diagram (adapted from Huang et al. [29]).
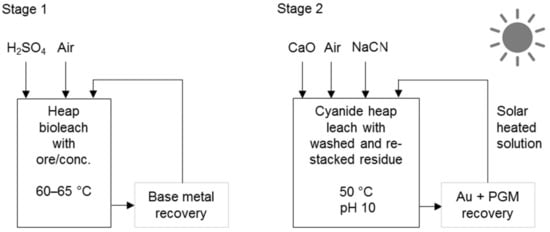
Figure 9.
Schematic diagram of the sequential heap leach process (adapted from Mwase et al. [30]).
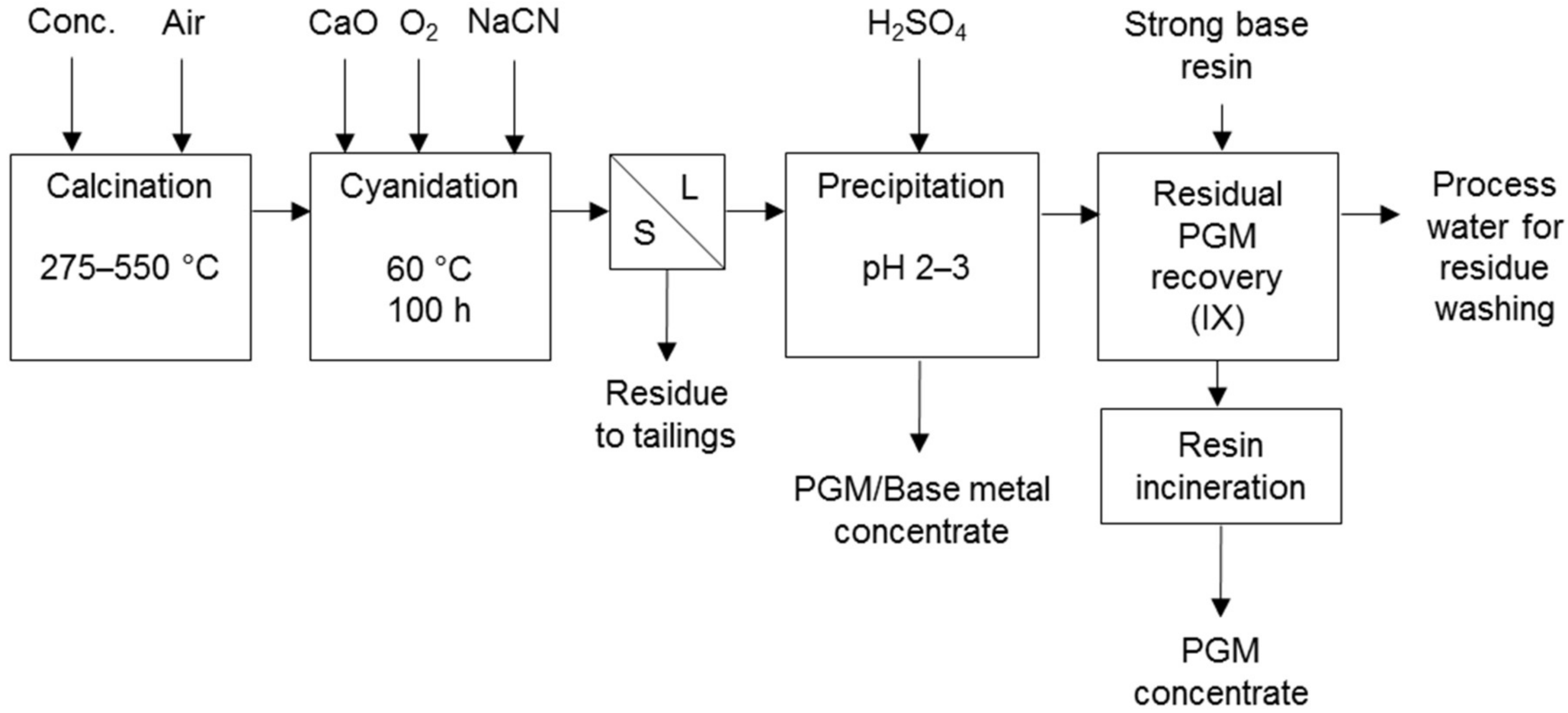
Cyanidation processes co-dissolving PG and base metals were developed to treat Coronation Hill ore (Figure 10) [7] and Panton flotation concentrate (Figure 11) in Australia. The Panton process comprises cyanidation following calcination and the dissolved metals are co-precipitated. The precipitate is the final product or is upgraded to PGM concentrate and a separate base metal concentrate [31]. The current focus of the project is away from the Panton process [32].
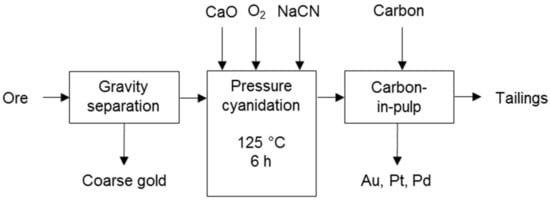
Figure 10.
Cyanidation process diagram for Coronation Hill ore (adapted from Cole & Ferron [7]).
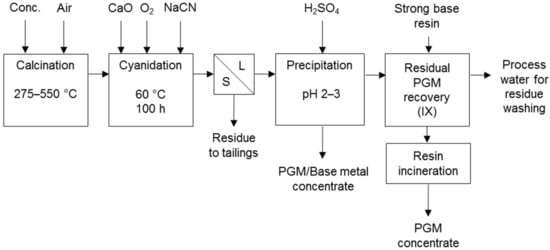
Figure 11.
Panton process flow sheet (adapted from Lewins & Greenaway [31]).
7. Conclusions
Due to the critical nature of PGMs and the increasing need for clean technology applications, the recovery of PGMs from both primary and secondary raw materials is of increasing importance. The current article focused on the PGM processes of the former and their value chain, highlighting hydrometallurgical refining approaches.
PGMs are recovered from low-grade resources and from a variety of host minerals due to their high value. Annual production of PGMs is around 400 t, with Anglo Platinum having the biggest share. One third of PGMs are produced as co-products of nickel mining. More than 80% of PGMs originate from two countries, namely South Africa and Russia. The former produces PGMs as primary products and the latter mainly as by-products of nickel. The geographical concentration of the resources increases the supply risk of this metal group.
The major four PGM mining companies are integrated from mining to refining. They account for 80% of the market; conventional matte smelting treatment being the prevailing processing method. Capital investments to process PGMs from flotation concentrate are very large and a significant barrier to entry for smaller mining companies. They typically produce flotation concentrate and sell the product for further refining. Other solutions for their value chain may emerge as a result of several studies—these may lead to joint ventures in the near future.
Alternative process routes are typically evaluated when the conventional method is not cost-effective. The direction is usually towards hydrometallurgical plants, as they are best suited for small operations. The technology developed makes use of metallurgy diversely and takes into account a specific raw material. Consequently, the proposed processes differ from each other. The proposed unit operations are predominantly proven and in common use. The chemistry underlying them is generally known in quite a detail. The suggested leaching media are sulphide-, chloride- or cyanide-based. These processes have a lack of industrial reference, which make it more challenging for them to gain a foothold.
Acknowledgments
The authors greatly acknowledge the Circular Metal Ecosystem (CMEco) project (7405-31-2016) for funding the research.
Author Contributions
Pia Sinisalo planned the content of the paper, conducted the literature research and the majority of the manuscript writing; Mari Lundström participated in the writing and was the instructor and supervisor of the work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jones, R.T. An overview of Southern African PGM smelting. In Nickel and Cobalt 2005, Challenges in Extraction and Production; Donald, J., Shoneville, R., Eds.; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, Quebec, Canada, 2005; pp. 147–178. [Google Scholar]
- Jollie, D. Forecasting Platinum Supply and Demand. 2016. Available online: https://www.platinuminvestment.com/files/Platinum_Fundamentals_forecast_Glaux_2016.pdf (accessed on 31 May 2017).
- Butler, J. Platinum 2012. Available online: http://www.platinum.matthey.com/publications/pgm-market-reviews/market-review-archive/platinum-2012 (accessed on 16 November 2017).
- Platinum Mining Supply Predicted to Fall Further in 2017. World Platinum Investment Council Press Release, 15 May 2017, 1–3. Available online: https://www.platinuminvestment.com/files/224537/WPIC_PR_PQ%20Q1%202017_15052017.pdf (accessed on 9 February 2018).
- The Primary Production of Platinum Group Metals (PGMs). International Platinum Group Metals Association Fact Sheet, 1–7. Available online: http://ipa-news.com/assets/sustainability/Primary%20Production%20Fact%20Sheet_LR.pdf (accessed on 9 February 2018).
- Grandell, L.; Lehtilä, A.; Kivinen, M.; Koljonen, T.; Kihlman, S.; Lauri, L.S. Role of critical metals in the future markets of clean energy technologies. Renew. Energy 2016, 95, 53–62. [Google Scholar] [CrossRef]
- Cole, S.; Ferron, J. A Review of the Beneficiation and Extractive Metallurgy of the Platinum Group Elements, Highlighting Recent Process Innovations. SGS Minerals Services Technical Paper 2002-03, 1–43. Available online: http://www.sgs.com/-/media/global/documents/technical-documents/sgs-technical-papers/sgs-min-tp2002-03-beneficiation-and-extractive-metallurgy-of-pge.pdf (accessed on 9 February 2018).
- Mpinga, C.N.; Eksteen, J.J.; Aldrich, C.; Dyer, L. Direct leach approaches to platinum group metal (PGM) ores and concentrates: A review. Miner. Eng. 2015, 78, 93–113. [Google Scholar] [CrossRef]
- Xiao, Z.; Laplante, A.R. Characterizing and recovering the platinum group minerals—A review. Miner. Eng. 2004, 17, 961–979. [Google Scholar] [CrossRef]
- Liddell, K.S.; Adams, M.D. Kell hydrometallurgical process for extraction of platinum group metals and base metals from flotation concentrates. J. South. Afr. Inst. Min. Metall. 2012, 112, 31–36. [Google Scholar]
- Ndlovu, J. Overview of PGM Processing. Anglo Platinum. Available online: http://www.angloamericanplatinum.com/~/media/Files/A/Anglo-American-Platinum/investor-presentation/standardbankconference-anglo-american-platinum-processing-111114.pdf (accessed on 31 May 2017).
- Milbourne, J.; Tomlinson, M.; Gormely, L. Use of hydrometallurgy in direct processing of base metal/PGM concentrates. In Hydrometallurgy 2003; Young, C.A., Alfantazi, A.M., Anderson, C.G., Dreisinger, D.B., Harris, B., James, A., Eds.; TMS: Pittsburgh, PA, USA, 2003; Volume 1, pp. 617–630. [Google Scholar]
- Cramer, L.A. The extractive metallurgy of South Africa’s platinum ore. JOM 2001, 53, 14–18. [Google Scholar] [CrossRef]
- Ndlovu, J. Precious Metals Supply. Anglo Platinum, 2015. Available online: https://www.google.fi/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&cad=rja&uact=8&ved=0ahUKEwjw36qOv7HXAhVREewKHe1OCD8QFgglMAA&url=http%3A%2F%2Fec.europa.eu%2FDocsRoom%2Fdocuments%2F14045%2Fattachments%2F1%2Ftranslations%2Fen%2Frenditions%2Fnative&usg=AOvVaw16kK3iMz6QoKTO3MH90wyl (accessed on 2 June 2017).
- Cramer, L.A. What is your PGM concentrate worth? In Third International Platinum Conference ‘Platinum in Transformation’; The Southern African Institute of Mining and Metallurgy: Johannesburg, South Africa, 2008; pp. 387–394. [Google Scholar]
- Levine, R.M.; Brininstool, M.; Wallace, G.J. The mineral industry of Russia—Strategic information. In Russia and Newly Independent States (NIS), Mineral Industry Handbook; International Business Publications: Washington DC, USA, 2015; Volume 1, pp. 40–61. [Google Scholar]
- Goosen, S. Rustenburg Base Metals Refinery Completed on Time and on Budget. Mining Weekly, 16 December 2011. Available online: http://www.miningweekly.com/article/rustenburg-base-metals-refinery-998-complete-2011-12-16 (accessed on 9 February 2018).
- Adams, M.; Liddell, K.; Holohan, T. Hydrometallurgical processing of Platreef flotation concentrate. Miner. Eng. 2011, 24, 545–550. [Google Scholar] [CrossRef]
- Everett, P.K. Development of Intec Copper Process by an international consortium. In Hydrometallurgy ’94; Springer: Dordrecht, The Netherlands, 1994; pp. 913–922. [Google Scholar]
- Hofirek, Z.; Halton, P. Production of high quality electrowon nickel at Rustenburg Base Metals Refiners (Pty.) Ltd. In Proceedings of the International Symposium on Electrometallurgical Plant Practice; Claessens, P.L., Harris, G.B., Eds.; Pergamon Press: New York, NY, USA, 1990; pp. 233–252. [Google Scholar] [CrossRef]
- Liddell, K.S.; McRae, L.B.; Dunne, R.C. Process routes for beneficiation of noble metals from Merensky and UG-2 ores. Mintek Rev. 1986, 4, 33–44. [Google Scholar]
- Thornhill, P.G.; Wigstol, E.; Van Weert, G. The Falconbridge matte leach process. JOM 1971, 23, 13–18. [Google Scholar] [CrossRef]
- Crundwell, F.; Moats, M.; Ramachandran, V.; Robinson, T.; Davenport, W. Extractive Metallurgy of Nickel, Cobalt and Platinum Group Metals; Elsevier: Oxford, UK, 2011; pp. 489–534. ISBN 978-0-08-096809-4. [Google Scholar]
- McDoulett, C.D.; Reschke, G.W. Metal Leaching and Recovery Process. Patent EP 0637635 A2, 8 February 1995. [Google Scholar]
- Jones, D.L. Chloride Assisted Hydrometallurgical Extraction of Metal. Patent US 5902474 A, 11 May 1999. [Google Scholar]
- Valkama, K.; Sinisalo, P.; Karonen, J.; Hietala, K. Method of Recovering Copper and Precious Metals. Patent WO 2014195586 A1, 11 December 2014. [Google Scholar]
- Sinisalo, P.; Tiihonen, M.; Hietala, K. Gold recovery from chalcopyrite concentrates in the HydroCopper® process. In ALTA 2008 Copper Conference; ALTA Metallurgical Services: Melbourne, Australia, 2008; pp. 1–8. [Google Scholar]
- Miettinen, V.; Ahtiainen, R.; Valkama, K. Method of Preparing a Gold-Containing Solution and Process Arrangement for Recovering Gold and Silver. Patent US 20160068927 A1, 10 March 2016. [Google Scholar]
- Huang, K.; Chen, J.; Chen, Y.R.; Zhao, J.C.; Li, Q.W.; Yang, Q.X.; Zhang, Y. Enrichment of platinum group metals (PGMs) by two-stage selective pressure leaching cementation from low-grade Pt-Pd sulfide concentrates. Metall. Mater. Trans B 2006, 37, 697–701. [Google Scholar] [CrossRef]
- Mwase, J.M.; Petersen, J.; Eksteen, J.J. A novel sequential heap leach process for treating crushed Platreef ore. Hydrometallurgy 2014, 141, 97–104. [Google Scholar] [CrossRef]
- Lewins, J.; Greenaway, T. The Panton platinum palladium project. Aust. Inst. Min. Metall. Bull. 2004, 24–34. [Google Scholar]
- Ferron, C.J. Recovery of gold as by-product from the base-metals industries. In Gold Ore Processing: Project Development and Operations, 2nd ed.; Adams, M.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 831–856. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).