Effect of Hot Mill Scale on Hydrogen Embrittlement of High Strength Steels for Pre-Stressed Concrete Structures
Abstract
:1. Introduction
2. Materials and Methods
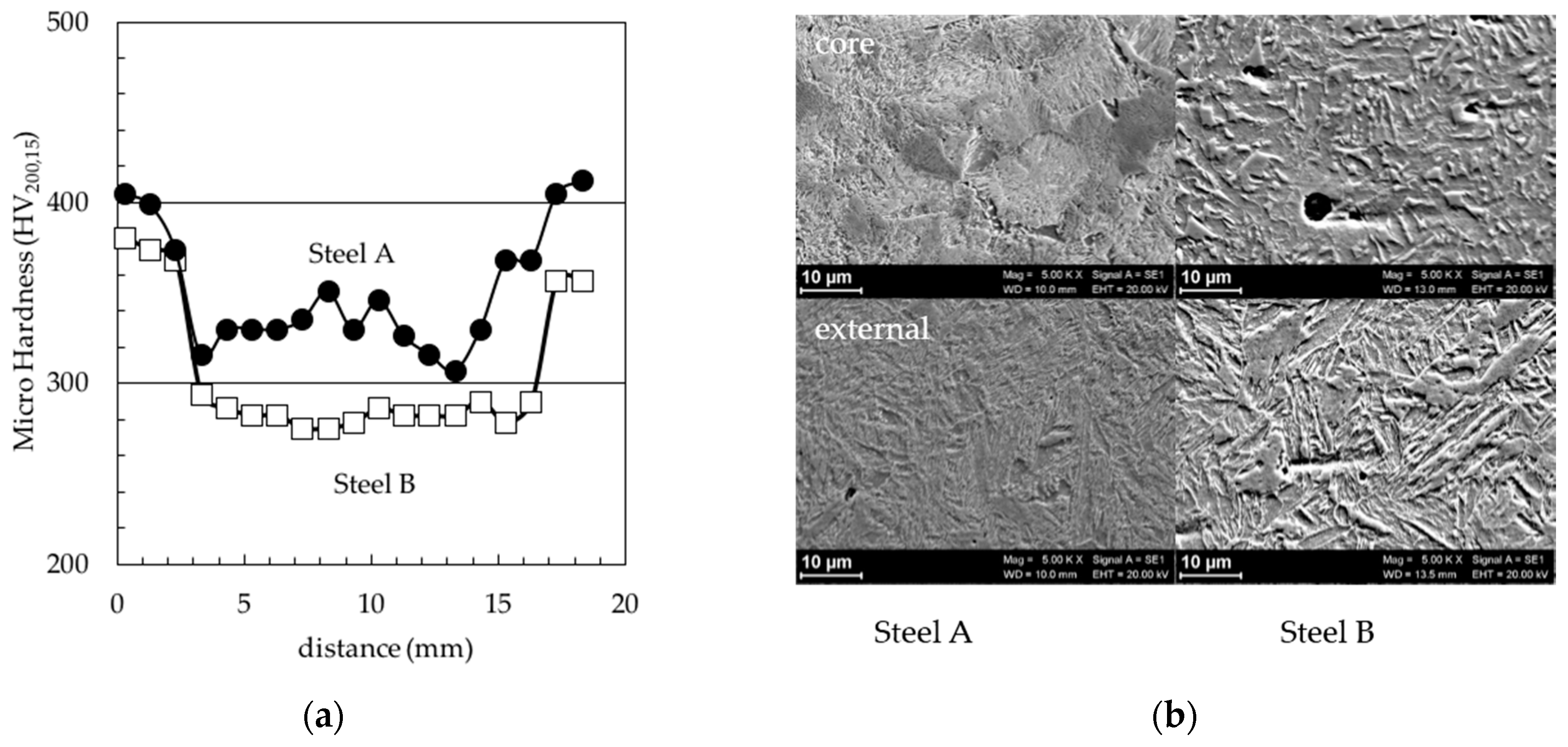
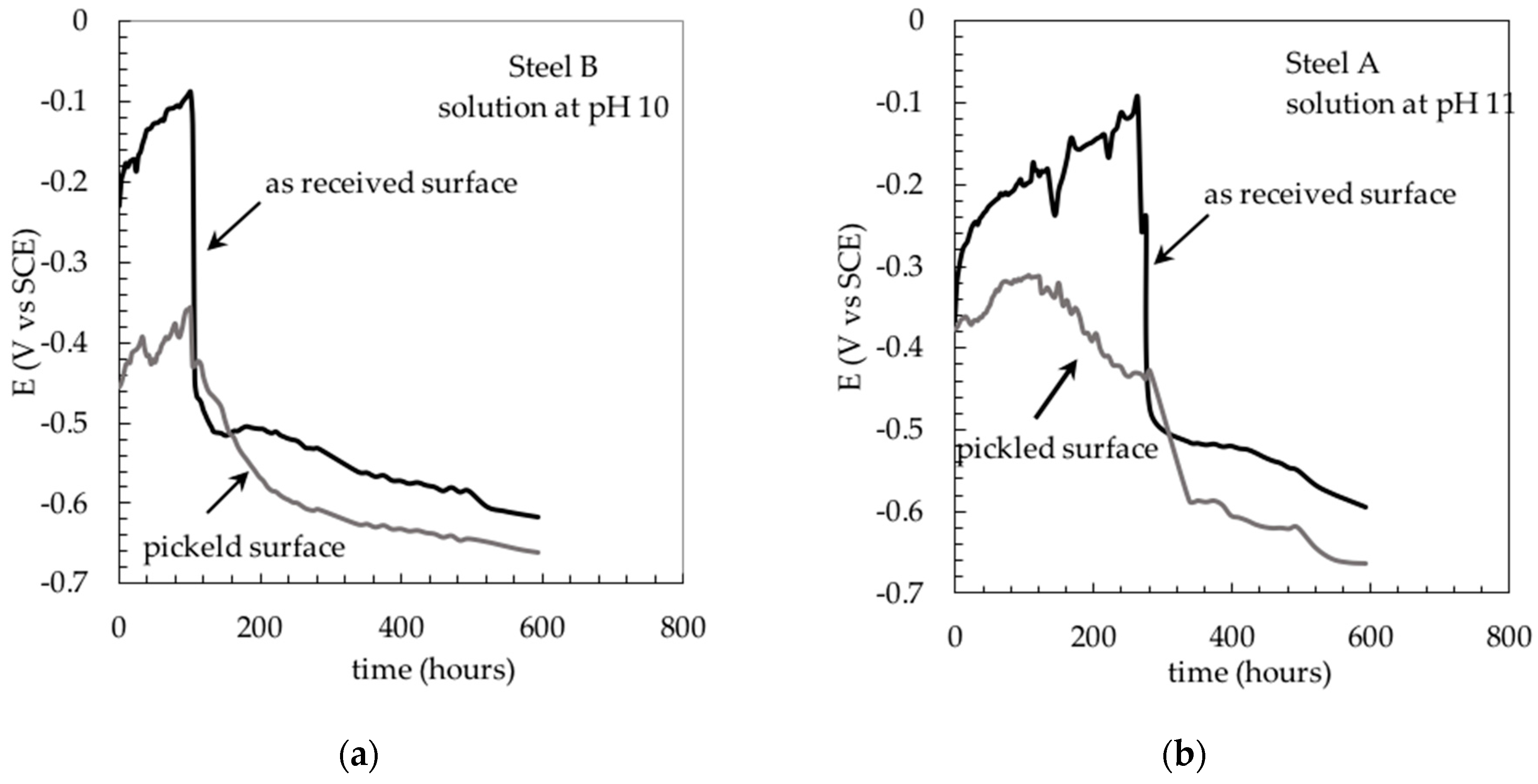
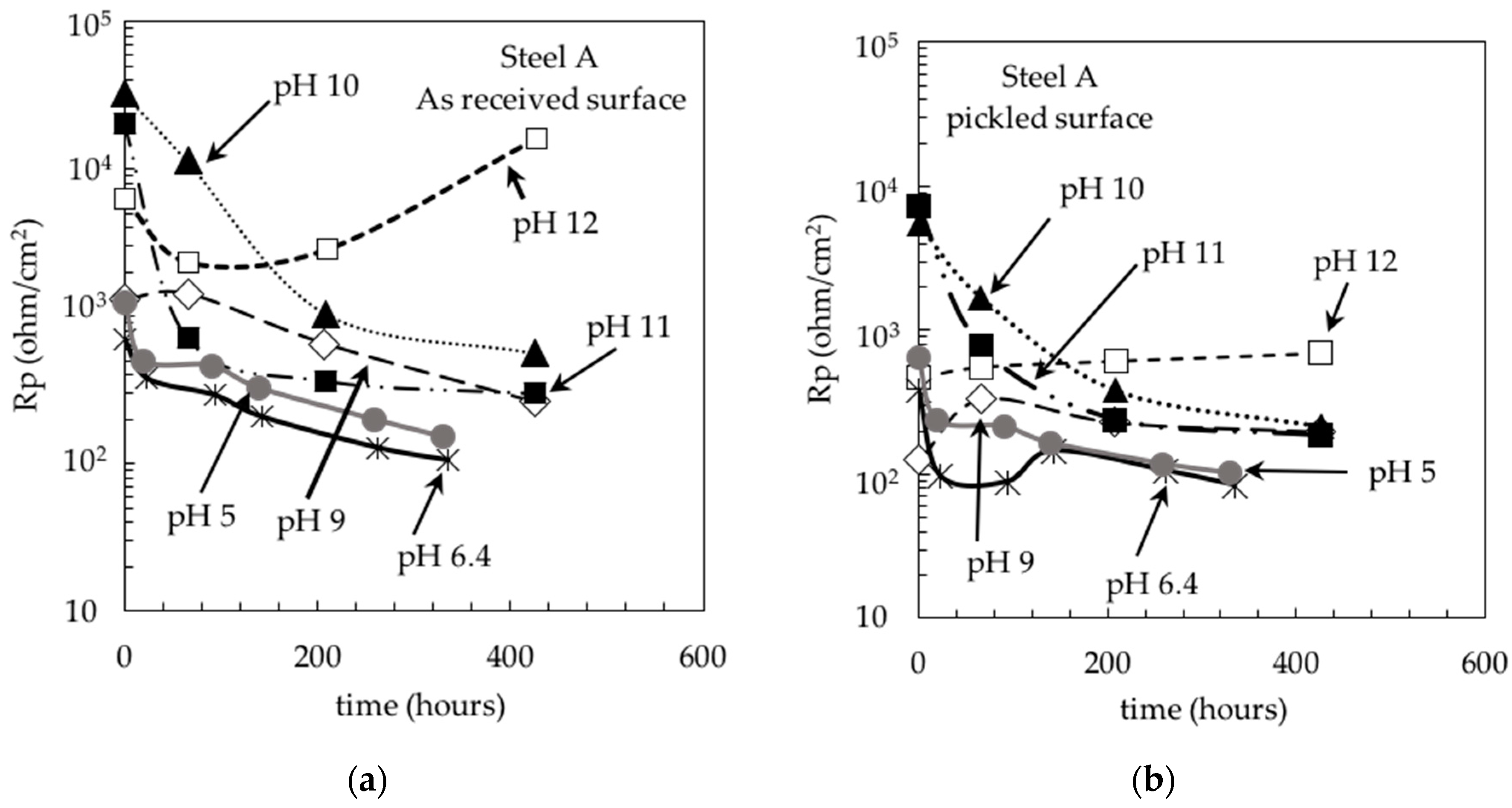
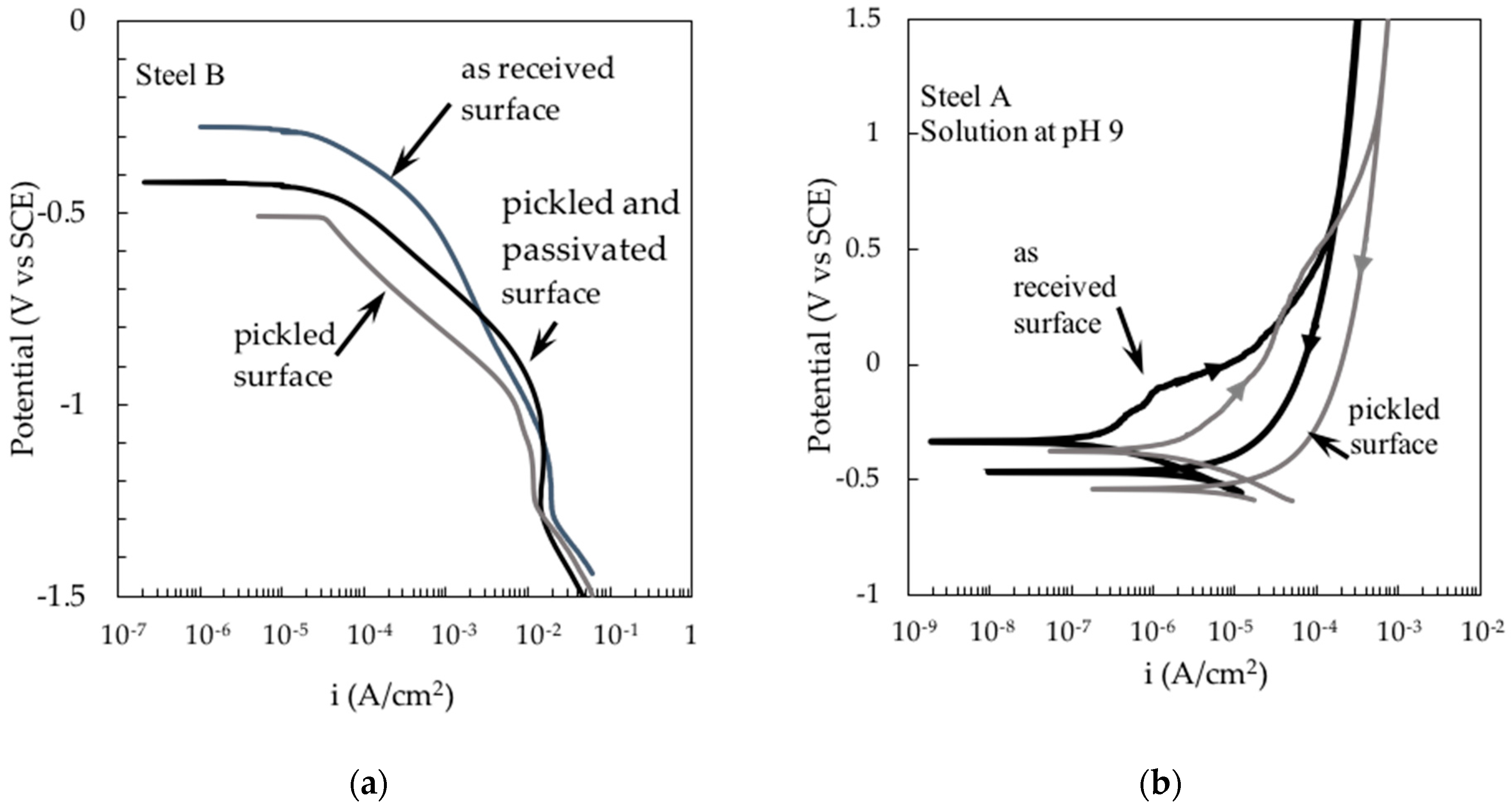
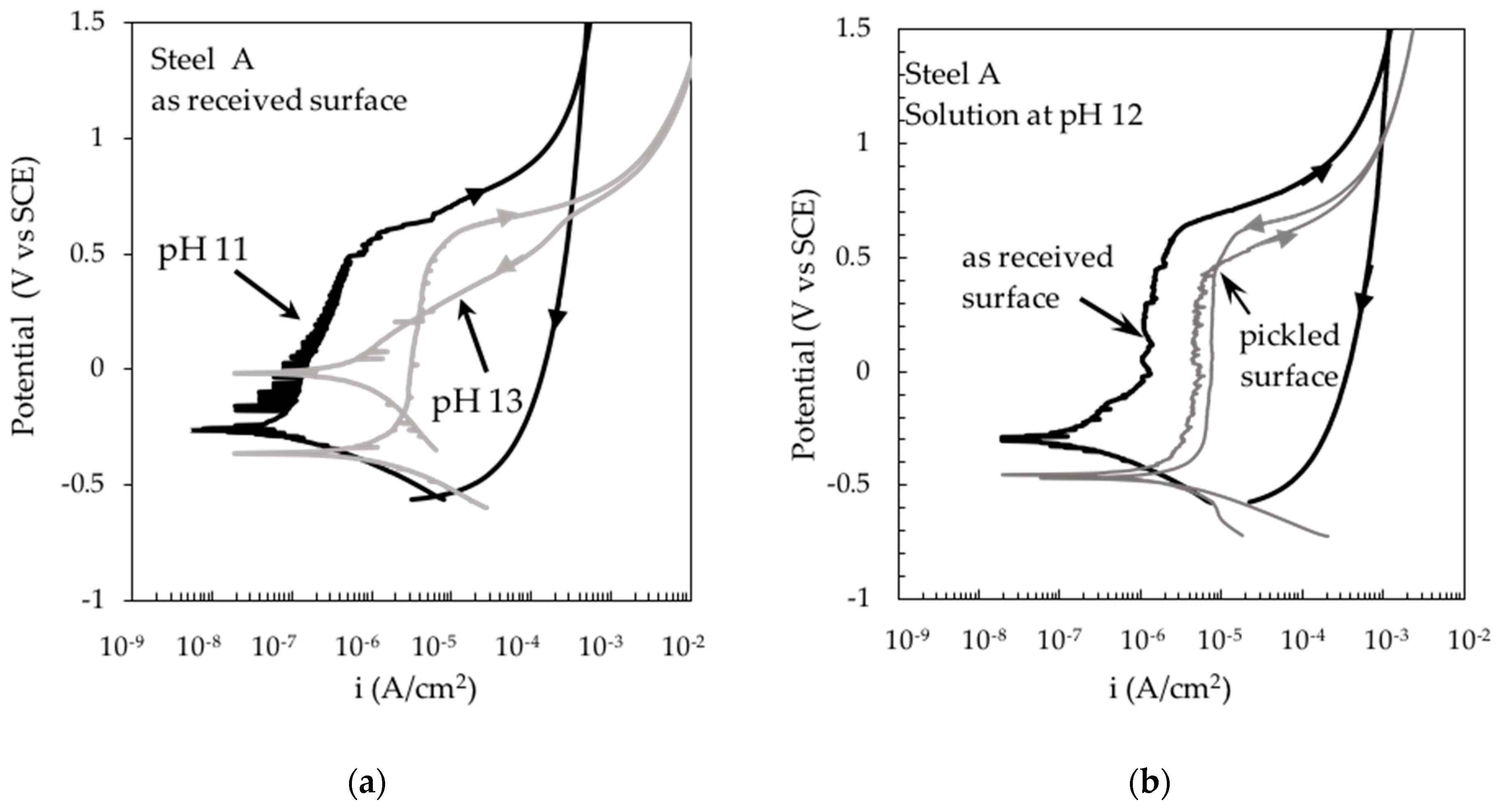
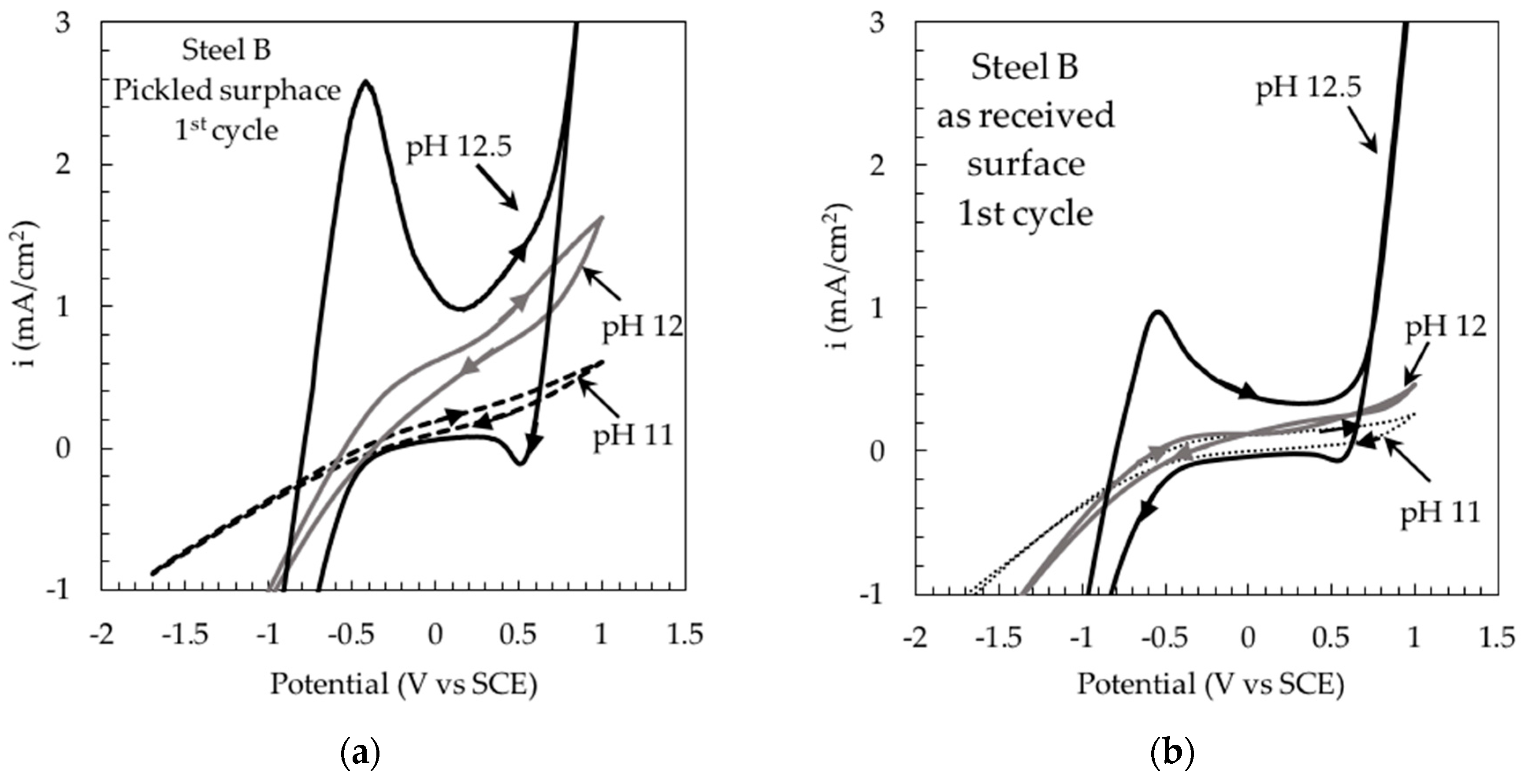
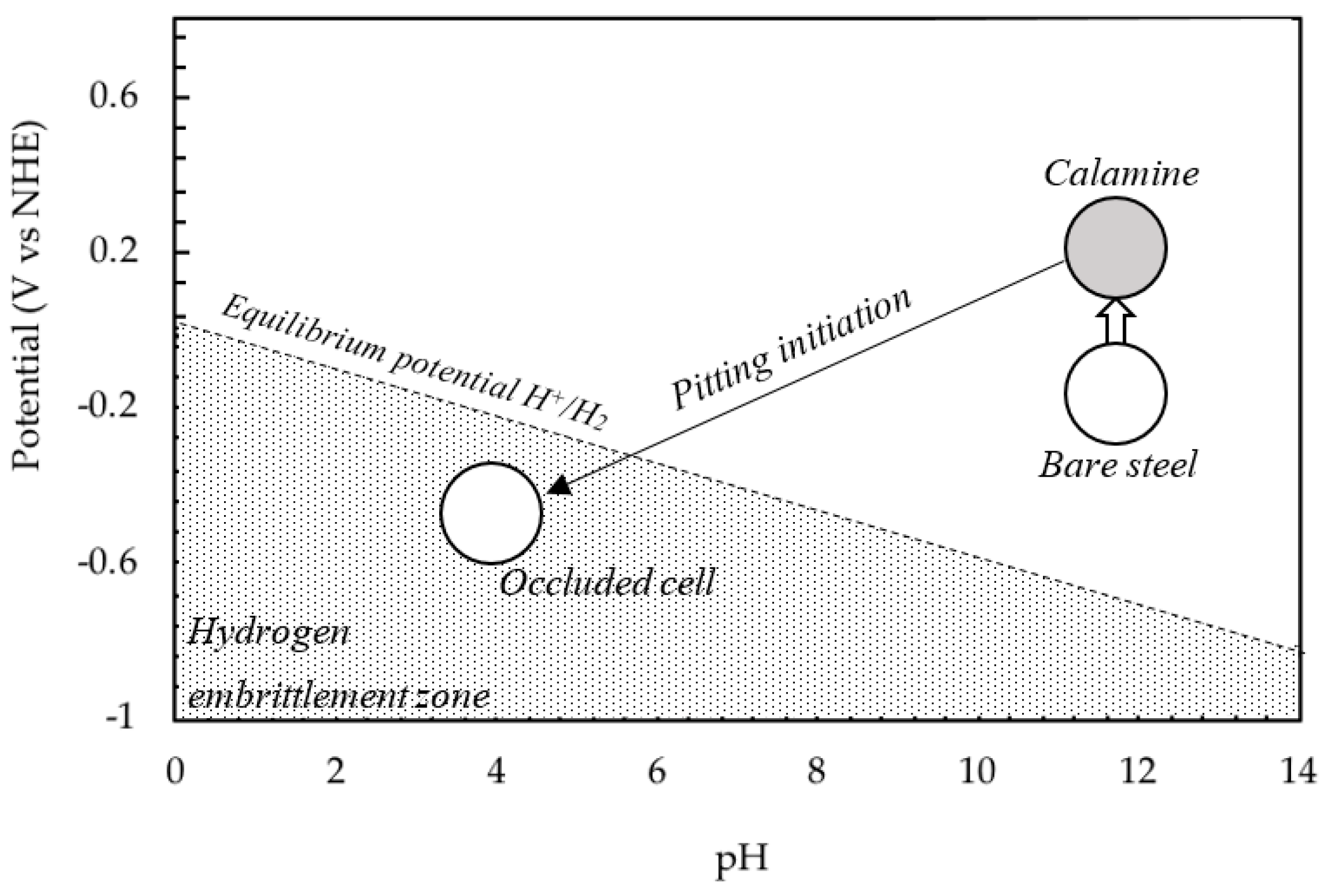
3. Results and Discussion
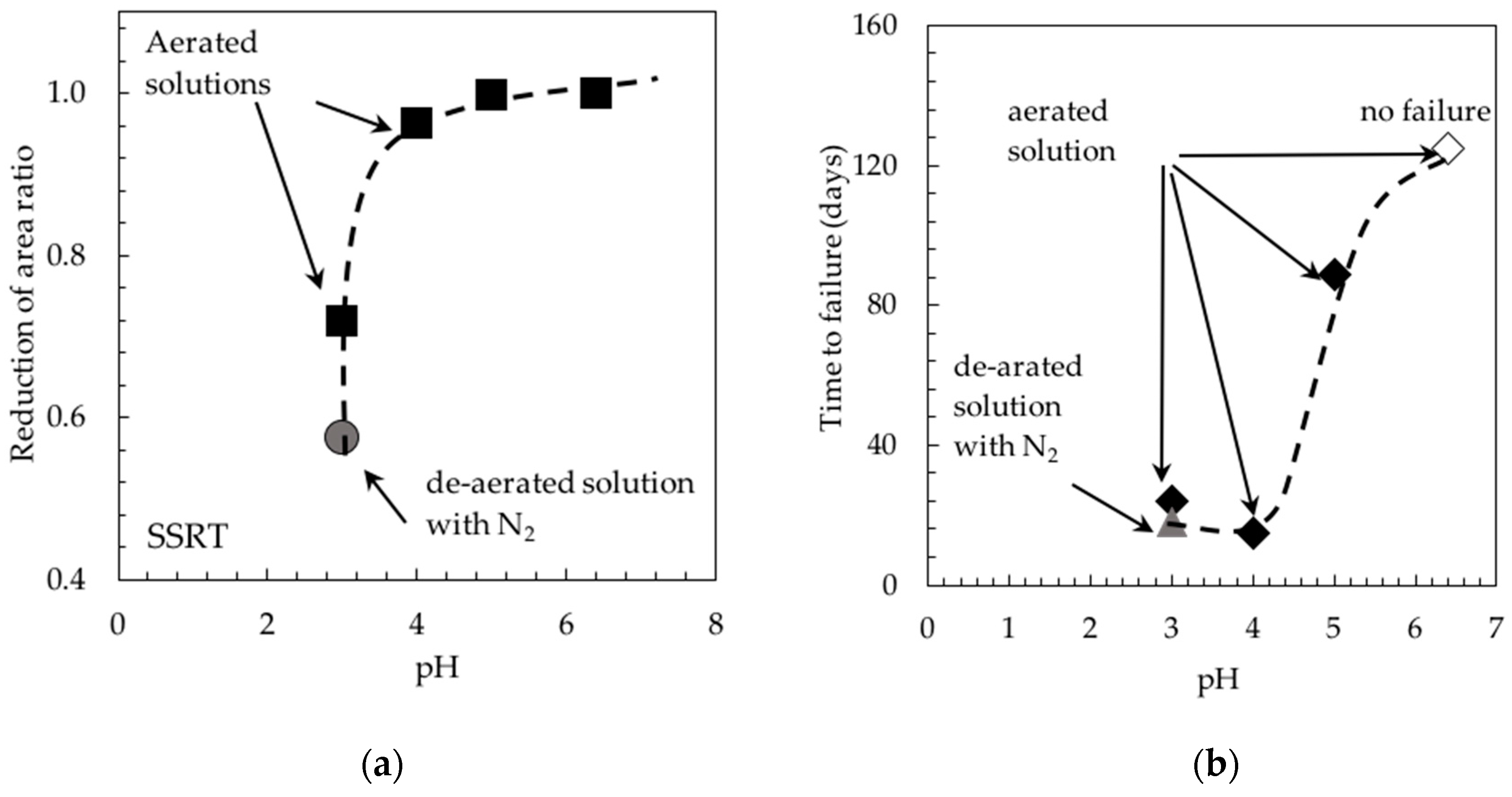
Stress Corrosion Tests
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Valient, A.; Elices, M. Premature failure of prestressed steel bars. Fail. Anal. Case Stud. II 1998, 5, 219–227. [Google Scholar] [CrossRef]
- Monfore, G.; Verbeck, C. Corrosion of prestressed wire in concrete. J. Am. Concr. Inst. 1960, 37, 491–516. [Google Scholar]
- Perrin, M.; Gaillet, L.; Tessier, C.; Idrissi, H. Hydrogen embrittlement of prestressing cables. Corros. Sci. 2010, 52, 1915–1926. [Google Scholar] [CrossRef]
- Yuyama, S.; Yokoyama, K.; Niitani, K.; Ohtsu, M.; Uomoto, T. Detection and evaluation of failures in high-strength tendon of prestressed concrete bridges by acoustic emission. Constr. Build. Mater. 2007, 21, 491–500. [Google Scholar] [CrossRef]
- Stoll, F.; Saliba, J.; Casper, L. Experimental study of CFRP-prestressed high-strength concrete bridge beams. Compos. Struct. 2000, 49, 191–200. [Google Scholar] [CrossRef]
- Woodward, R.; Williams, F. Collapse of Ynys-y-Gwas Bridge, west Glamorgan. Proc. Instn. Civ. Engrs. 1988, 84, 635–669. [Google Scholar] [CrossRef]
- Helmerich, R.; Zunkel, A. Partial collapse of the Berlin Congress Hall on May 21st 1980. Eng. Failure Anal. 2014, 43, 107–119. [Google Scholar] [CrossRef]
- Senigaglia, D.; Re, G.; Pedeferri, P. Cedimento per Fatica e Ambientale dei Materiali Metallici; CLUP: Milano, Italy, 1979. [Google Scholar]
- Pedeferri, P.; Bertolini, L. La Durabilità del Calcestruzzo Armato; Mc Graw-Hill: Milano, Italy, 2000; p. 199. [Google Scholar]
- Woodtli, J.; Kieselbach, R. Damage due to hydrogen embrittlement and stress corrosion cracking. Eng. Failure Anal. 2000, 7, 427–450. [Google Scholar] [CrossRef]
- Cabrini, M.; Lorenzi, S.; Marcassoli, P.; Pastore, T. Hydrogen embrittlement behavior of HSLA line pipe steel under cathodic protection. Corros. Rev. 2011, 29, 261–270. [Google Scholar] [CrossRef]
- Cabrini, M.; Lorenzi, S.; Pellegrini, S.; Pastore, T. Environmentally assisted cracking and hydrogen diffusion in traditional and high-strength pipeline steels. Corros. Rev. 2015, 33, 529–545. [Google Scholar] [CrossRef]
- Mietz, J. Investigation on hydrogen-induced embrittlement of quenched and tempered prestressing steels. Mater. Corros. 2000, 51, 2–80. [Google Scholar] [CrossRef]
- Darmawan, M.; Stewart, M. Effect of pitting corrosion on capacity of prestressing wires. Mag. Concr. Res. 2007, 59, 131–139. [Google Scholar] [CrossRef]
- Toribio, J.; Ovejero, E. Effect of Cold Drawing on Microstructure and Corrosion Performance of High-Strength Steel. Mech. Time-Depend. Mater. 1998, 1, 307–319. [Google Scholar] [CrossRef]
- Enos, D.; Scully, J. A Critical-Strain Criterion for Hydrogen Embrittlement of Cold-Drawn, Ultrafine Pearlitic Steel. Metall. Mater. Trans. A 2002, 33A, 1151–1166. [Google Scholar] [CrossRef]
- Zitrou, E.; Nikolaou, J.; Tsakiridis, P.; Papadimitriou, G. Atmospheric corrosion of steel reinforcing bars produced by various manufacturing processes. Constr. Build. Mater. 2007, 21, 1161–1169. [Google Scholar] [CrossRef]
- Vehovar, L.; Kuhar, K.; Vehovar, A. Hydrogen-assisted stress corrosion of prestressing wires in a motorway viaduct. Eng. Failure Anal. 1998, 5, 21–27. [Google Scholar] [CrossRef]
- Nürnberger, U. Korrosion und Korrosionsschutz im Bauwesen; Grundlagen, B., Metallbau, K., Eds.; Bauverlag BV GmbH: Gütersloh, Germany, 1995. [Google Scholar]
- Hirth, J. Effects of Hydrogen on the Properties of Iron and Steel. Metall. Trans. A 1980, 11A, 861–890. [Google Scholar] [CrossRef]
- Lynch, S. Hydrogen embrittlement phenomena and mechanisms. Corros. Rev. 2012, 30, 105–123. [Google Scholar] [CrossRef]
- Bockris, J.O.; McBreen, J.; Nanis, L. The Hydrogen Evolution Kinetics and Hydrogen Entry into a-Iron. J. Appl. Electrochem. 1965, 112, 1025–1031. [Google Scholar] [CrossRef]
- Cabrini, M.; Lorenzi, S. Pipeline Steels: Hydrogen Diffusion and Environmentally Assisted Cracking. In Encyclopedia of Iron, Steel, and Their Alloys; George, E., Totten, R.C., Eds.; CRC Press Taylor and Francis Group: Boca Raton, FL, USA, 2016; pp. 2547–2599. [Google Scholar]
- Camuffo, D.; Bernardi, A.; Zanetti, M. Analysis of the Real-Time Measurement of the oH of Rainfall at Padova, Italy: Seasonal Variatino and Meteorological Aspects. Sci. Total. Environ. 1988, 71, 187–200. [Google Scholar] [CrossRef]
- Mrose, H. Measurements of pH, and chemical analyses of rain-, snow-, and fog-water. Tellus 1966, 18, 266–270. [Google Scholar] [CrossRef]
- Singh, A.; Agrawal, M. Acid rain and its ecological consequences. J. Environ. Biol. 2008, 15–24. [Google Scholar]
- Nürnberger, U. Corrosion induced failure mechanisms of prestressing steel. Mater. Corros. 2002, 53, 591–601. [Google Scholar] [CrossRef]
- Schrebler Guzmán, R.S.; Vilche, J.R.; Arvia, A.J. The potentiodynamic behaviour of iron in alkaline solutions. Electrochim. Acta 1979, 24, 395–403. [Google Scholar] [CrossRef]
- Cabrini, M.; Lorenzi, S.; Pastore, T. Cyclic voltammetry evaluation of inhibitors for localised corrosion in alkaline solutions. Electrochim. Acta 2014, 124, 156–164. [Google Scholar] [CrossRef]
- Schrebler Guzman, R.S.; Vilche, J.R.; Arvia, A.J. The voltammetric detection of intermediate electrochemical processes related to iron in alkaline aqueous solutions. J. Appl. Electrochem. 1981, 11, 5. [Google Scholar] [CrossRef]





| Steel | Chemical Composition (% Weight) | Mechanical Properties | |||||
|---|---|---|---|---|---|---|---|
| C | Mn | Si | P | S | RS (MPa) | R (MPa) | |
| A | 0.71 | 0.63 | 0.22 | 0.016 | 0.016 | 996 | 1103 |
| B | 0.2 | 1.45 | 0.45 | 0.022 | 0.017 | 713 | 818 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabrini, M.; Lorenzi, S.; Pastore, T.; Pesenti Bucella, D. Effect of Hot Mill Scale on Hydrogen Embrittlement of High Strength Steels for Pre-Stressed Concrete Structures. Metals 2018, 8, 158. https://doi.org/10.3390/met8030158
Cabrini M, Lorenzi S, Pastore T, Pesenti Bucella D. Effect of Hot Mill Scale on Hydrogen Embrittlement of High Strength Steels for Pre-Stressed Concrete Structures. Metals. 2018; 8(3):158. https://doi.org/10.3390/met8030158
Chicago/Turabian StyleCabrini, Marina, Sergio Lorenzi, Tommaso Pastore, and Diego Pesenti Bucella. 2018. "Effect of Hot Mill Scale on Hydrogen Embrittlement of High Strength Steels for Pre-Stressed Concrete Structures" Metals 8, no. 3: 158. https://doi.org/10.3390/met8030158
APA StyleCabrini, M., Lorenzi, S., Pastore, T., & Pesenti Bucella, D. (2018). Effect of Hot Mill Scale on Hydrogen Embrittlement of High Strength Steels for Pre-Stressed Concrete Structures. Metals, 8(3), 158. https://doi.org/10.3390/met8030158







