Stress Corrosion Cracking Behavior of Fine-Grained AZ61 Magnesium Alloys Processed by Equal-Channel Angular Pressing
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Material
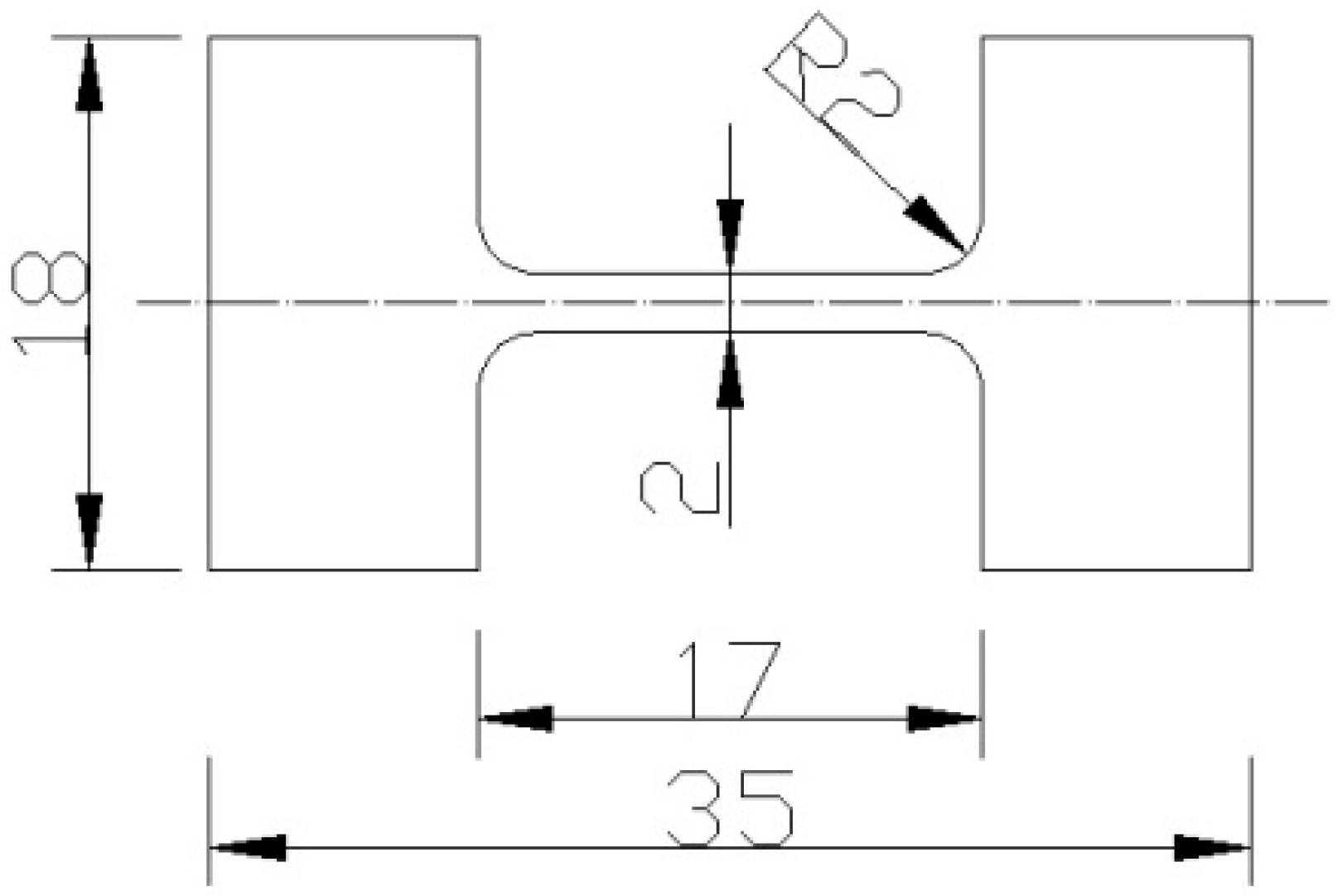
2.2. Slow Strain Rate Tensile Testing
2.3. Microstructure Observation
3. Results
3.1. Microstructure
3.2. Slow Strain Rate Tensile Testing
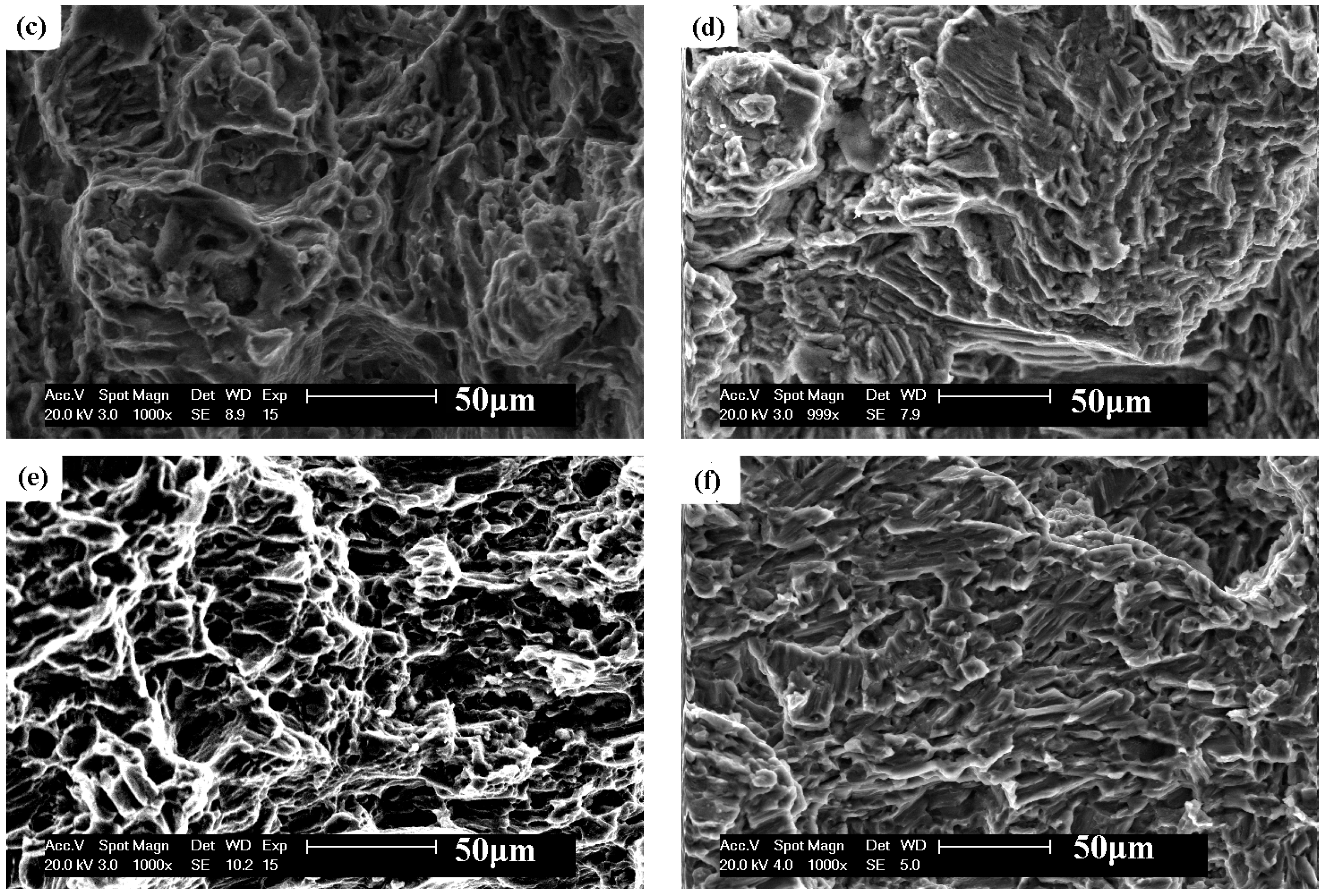
3.3. Fractography
4. Discussion
5. Conclusions
- Fine-grained AZ61 Mg alloys were fabricated via multi-pass ECAP processing both at 623 K and 673 K. The decrease in processing temperature is favourable to the refinement of the microstructure, resulting in the enhancement of ultimate tensile strength and even elongation in distilled water.
- The as-cast AZ61 Mg alloys with about 6% Al content have a great tendency to SCC in distilled water and the ECAPed processing increased the susceptibility to SCC at room temperature.
- The greater SCC tendency of ECAPed AZ61 Mg alloys was related to the combined effect of grain refinement, introduced defects and refined β-phase following ECAP processing. Fine grains of the Mg matrix could retard cracks propagation to some extent, whereas enormous crystalline defects introduced by severe plastic deformation accelerated corrosion destruction and diffusion of hydrogen along with crumbled barrier action for a transition from net-like β-phase to isolated fine β-phase particles.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Winzer, N.; Atrens, A.; Song, G.; Ghali, E.; Dietzel, W.; Kainer, K.U.; Hort, N.; Blawert, C. A Critical Review of the Stress Corrosion Cracking (SCC) of Magnesium Alloys. Adv. Eng. Mater. 2005, 7, 659–693. [Google Scholar] [CrossRef]
- Uematsu, Y.; Kakiuchi, T.; Nakajima, M. Stress corrosion cracking behavior of the wrought magnesium alloy AZ31 under controlled cathodic potentials. Mater. Sci. Eng. A 2012, 531, 171–177. [Google Scholar] [CrossRef]
- Li, X.C.; Zhang, Y.K.; Chen, J.F.; Lu, Y.L. Effect of laser shock processing on stress corrosion cracking behaviour of AZ31 magnesium alloy at slow strain rate. Mater. Sci. Technol. 2013, 29, 626–630. [Google Scholar] [CrossRef]
- Bala Srinivasan, P.; Blawert, C.; Dietzel, W. Effect of plasma electrolytic oxidation coating on the stress corrosion cracking behaviour of wrought AZ61 magnesium alloy. Corros. Sci. 2008, 50, 2415–2418. [Google Scholar] [CrossRef]
- Winzer, N.; Atrens, A.; Dietzel, W.; Song, G.; Kainer, K.U. Evaluation of the delayed hydride cracking mechanism for transgranular stress corrosion cracking of magnesium alloys. Mater. Sci. Eng. A 2007, 466, 18–31. [Google Scholar] [CrossRef]
- Tomashov, N.D.; Modestova, N.V. Intercrystalline Corrosion and Corrosion of Metals under Stress; L. Hill: London, UK, 1963. [Google Scholar]
- Padekar, B.S.; Singh Raman, R.K.; Raja, V.S.; Paul, L. Stress corrosion cracking of a recent rare-earth containing magnesium alloy, EV31A, and a common Al-containing alloy, AZ91E. Corros. Sci. 2013, 71, 1–9. [Google Scholar] [CrossRef]
- Pu, Z.; Song, G.L.; Yang, S.; Outeiro, J.C.; Dillion, O.W., Jr.; Puleo, D.A.; Jawahir, I.S. Grain refined and basal textured surface produced by burnishing for improved corrosion performance of AZ31B Mg alloy. Corros. Sci. 2012, 57, 192–201. [Google Scholar] [CrossRef]
- Argade, G.R.; Yuan, W.; Kandasamy, K.; Mishra, R.S. Stress corrosion cracking susceptibility of ultrafine grained AZ31. J. Mater. Sci. 2012, 47, 6812–6822. [Google Scholar] [CrossRef]
- López, H.F.; Cisneros, M.M.; Mancha, H.; García, O.; Pérez, M.J. Grain size effects on the SCC susceptibility of a nitrogen steel in hot NaCl solutions. Corros. Sci. 2006, 48, 913–924. [Google Scholar] [CrossRef]
- Fairman, L.; Bray, H.J. Transgranular see in Mg-Al alloys. Corros. Sci. 1971, 11, 533–541. [Google Scholar] [CrossRef]
- Pelensky, M.; Gallaccio, A. Stress Corrosion of Magnesium Alloys—Environmental Factors. J. Oral Res. 1967, 3, 257–261. [Google Scholar]
- Kim, W.J.; Kim, J.K.; Park, T.Y.; Hong, S.I.; Kim, D.I.; Kim, Y.S.; Lee, J.D. Enhancement of strength and superplasticity in a 6061 Al alloy processed by equal-channel-angular-pressing. Metall. Mater. Trans. A 2002, 33, 3155–3164. [Google Scholar] [CrossRef]
- Horita, Z.; Fujinami, T.; Nemoto, M.; Langdon, T.G. Improvement of mechanical properties for Al alloys using equal-channel angular pressing. J. Mater. Process. Technol. 2001, 117, 288–292. [Google Scholar] [CrossRef]
- Iwahashi, Y.; Horita, Z.; Nemoto, M.; Langdon, T.G. An investigation of microstructural evolution during equal-channel angular pressing. Acta Mater. 1997, 45, 4733–4741. [Google Scholar] [CrossRef]
- Iwahashi, Y.; Furukawa, M.; Horita, Z.; Nemoto, M.; Langdon, T.G. Microstructural characteristics of ultrafine-grained aluminum produced using equal-channel angular pressing. Metall. Mater. Trans. A 1998, 29, 2245–2252. [Google Scholar] [CrossRef]
- Gunde, P.; Hänzi, A.C.; Sologubenko, A.S.; Uggowitzer, P.J. High-strength magnesium alloys for degradable implant applications. Mater. Sci. Eng. A 2011, 528, 1047–1054. [Google Scholar] [CrossRef]
- Wu, S.; Wang, D.; Deng, C.; Wang, Y. Investigation on Pop-in phenomenon and its causes in CTOD test for weld metal. Trans. China Weld. Inst. 2012, 33, 105–108. [Google Scholar]
- Chakrapani, D.G.; Pugh, E.N. The transgranular SCC of a Mg-Al alloy: Crystallographic, fractographic and acoustic-emission studies. Metall. Trans. A 1975, 6, 1155–1163. [Google Scholar] [CrossRef]
- Chakrapani, D.G.; Pugh, E.N. Hydrogen embrittlement in a Mg-Al alloy. Metall. Trans. A 1976, 7, 173–178. [Google Scholar] [CrossRef]
- Winzer, N.; Atrens, A.; Dietzel, W.; Song, G.; Kainer, K.U. Comparison of the linearly increasing stress test and the constant extension rate test in the evaluation of transgranular stress corrosion cracking of magnesium. Mater. Sci. Eng. A 2008, 472, 97–106. [Google Scholar] [CrossRef]
- Tan, J.C.; Tan, M.J. Dynamic continuous recrystallization characteristics in two stage deformation of Mg–3Al–1Zn alloy sheet. Mater. Sci. Eng. A 2003, 339, 124–132. [Google Scholar] [CrossRef]
- Song, D.; Ma, A.B.; Jiang, J.H.; Lin, P.H.; Yang, D.H.; Fan, J.F. Corrosion behaviour of bulk ultra-fine grained AZ91D magnesium alloy fabricated by equal-channel angular pressing. Corros. Sci. 2011, 53, 362–373. [Google Scholar] [CrossRef]
- Schino, A.D.; Kenny, J.M. Effects of the grain size on the corrosion behavior of refined AISI 304 austenitic stainless steels. J. Mater. Sci. Lett. 2002, 21, 1631–1634. [Google Scholar] [CrossRef]
- Aung, N.N.; Zhou, W. Effect of grain size and twins on corrosion behaviour of AZ31B magnesium alloy. Corros. Sci. 2010, 52, 589–594. [Google Scholar] [CrossRef]
- Makar, G.L.; Kruger, J. Corrosion Studies of Rapidly Solidified Magnesium Alloys. J. Electrochem. Soc. 1990, 137, 414–421. [Google Scholar] [CrossRef]
- Baliga, C.B.; Tsakiropoulos, P. Development of corrosion resistant magnesium alloys Part 2 Structure of corrosion products on rapidly solidified Mg–16Al alloys. Mater. Sci. Technol. 1993, 9, 513–519. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; Wu, X.; Zhang, B. Corrosion behaviour of AZ21, AZ501 and AZ91 in sodium chloride. Corros. Sci. 1998, 40, 1769–1791. [Google Scholar] [CrossRef]





| Alloy | Test Medium | Yield Strength (MPa) | Ultimate Tensile Strength (MPa) | Elongation (%) | Absorbed Energy to Failure (KJ/m3) | SCC Susceptibility Index, ISCC (%) |
|---|---|---|---|---|---|---|
| 1# | AIR | 64 | 186 | 12.12 | 15,805.21 | 42.50 |
| DW | 71 | 151 | 8.28 | 9087.45 | ||
| 2# | AIR | 153 | 267 | 11.86 | 24,760.22 | 54.83 |
| DW | 138 | 212 | 6.96 | 11,184.32 | ||
| 3# | AIR | 106 | 235 | 12.50 | 21,401.24 | 69.66 |
| DW | 118 | 186 | 5.01 | 6492.35 | ||
| 4# | AIR | 96 | 261 | 25.14 | 50,594.78 | 67.45 |
| DW | 105 | 206 | 10.77 | 16,467.80 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, Q.; Ma, A.; Jiang, J.; Cheng, Z.; Song, D.; Yuan, Y.; Liu, H. Stress Corrosion Cracking Behavior of Fine-Grained AZ61 Magnesium Alloys Processed by Equal-Channel Angular Pressing. Metals 2017, 7, 343. https://doi.org/10.3390/met7090343
Xie Q, Ma A, Jiang J, Cheng Z, Song D, Yuan Y, Liu H. Stress Corrosion Cracking Behavior of Fine-Grained AZ61 Magnesium Alloys Processed by Equal-Channel Angular Pressing. Metals. 2017; 7(9):343. https://doi.org/10.3390/met7090343
Chicago/Turabian StyleXie, Qiuyuan, Aibin Ma, Jinghua Jiang, Zhaojun Cheng, Dan Song, Yuchun Yuan, and Huan Liu. 2017. "Stress Corrosion Cracking Behavior of Fine-Grained AZ61 Magnesium Alloys Processed by Equal-Channel Angular Pressing" Metals 7, no. 9: 343. https://doi.org/10.3390/met7090343
APA StyleXie, Q., Ma, A., Jiang, J., Cheng, Z., Song, D., Yuan, Y., & Liu, H. (2017). Stress Corrosion Cracking Behavior of Fine-Grained AZ61 Magnesium Alloys Processed by Equal-Channel Angular Pressing. Metals, 7(9), 343. https://doi.org/10.3390/met7090343





