Efficient Use of Porous Hybrid Materials in a Selective Detection of Lead(II) from Aqueous Solutions: An Electrochemical Study
Abstract
:1. Introduction
2. Experimental Methods
2.1. Reagents and Apparatus
2.2. Methodology
2.2.1. Synthesis of Hybrid Materials
2.2.2. Fabrication of the Working Electrode
2.2.3. Electrochemical Measurements
2.2.4. Real Tap Water Analysis
3. Results and Discussion
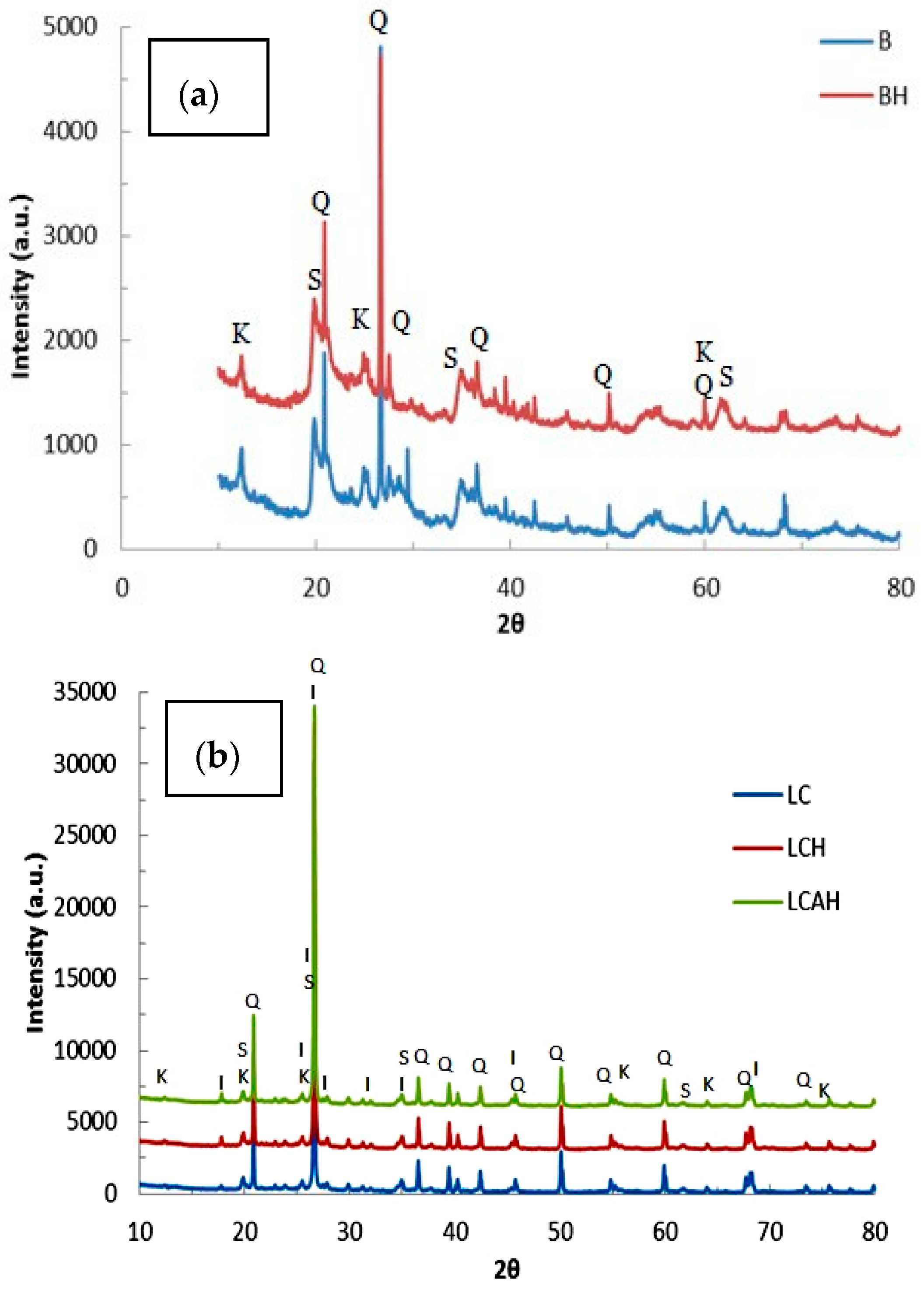
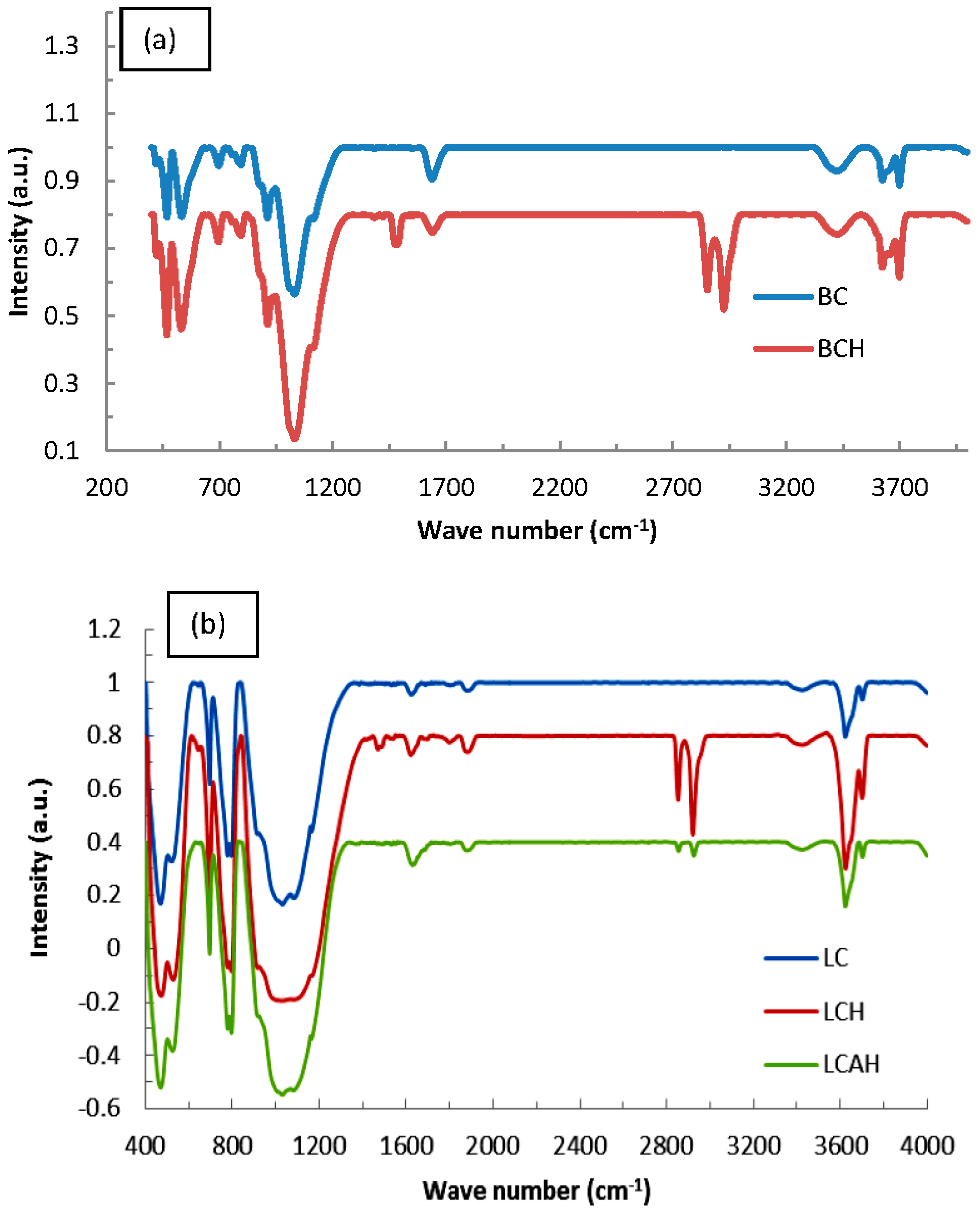
3.1. Characterization of Hybrid Materials
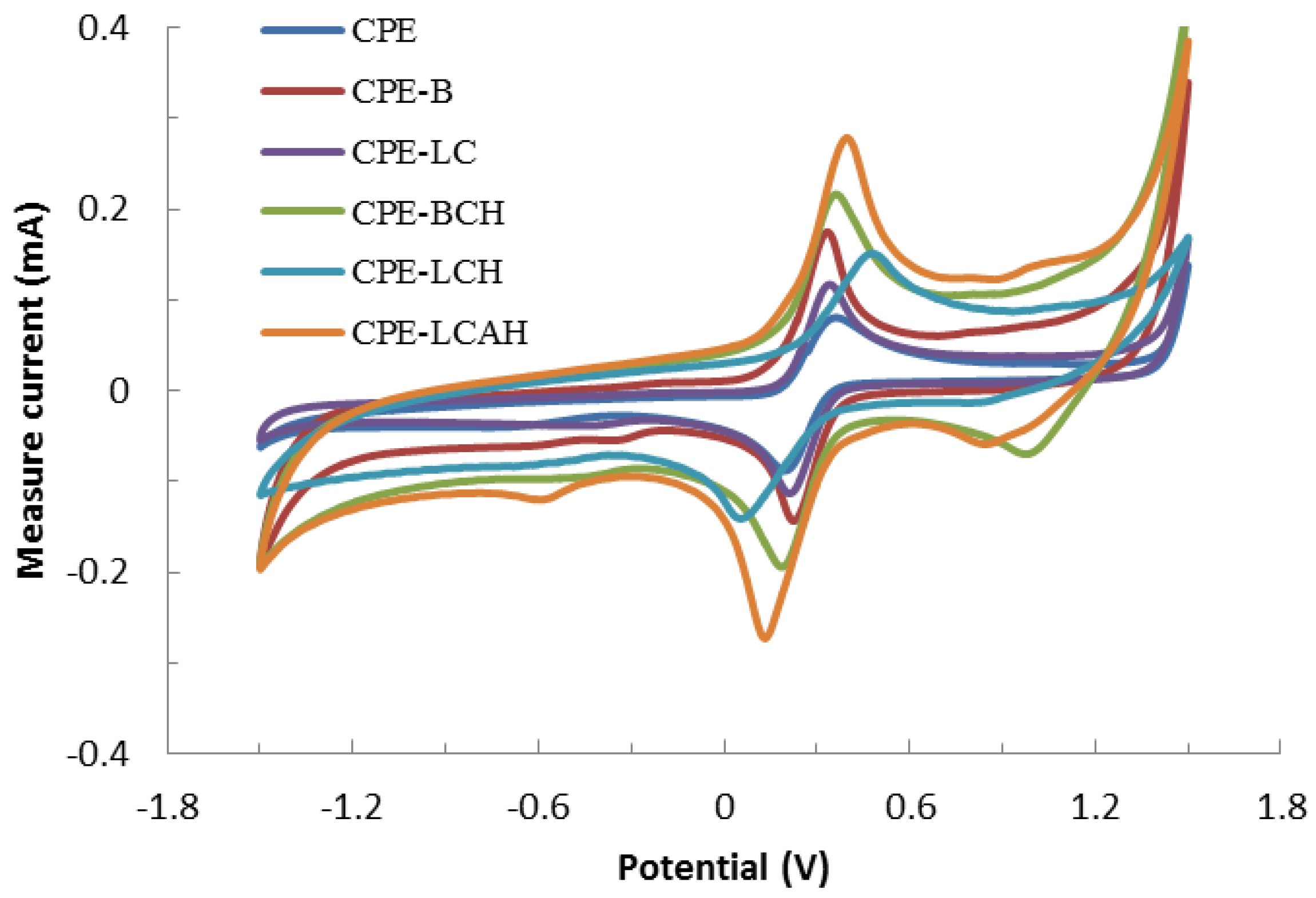
3.2. Electrochemical Response of Modified CPE
3.3. Electroanalytical Response of Various Modified Carbon Paste Electrodes
3.4. Effect of pH
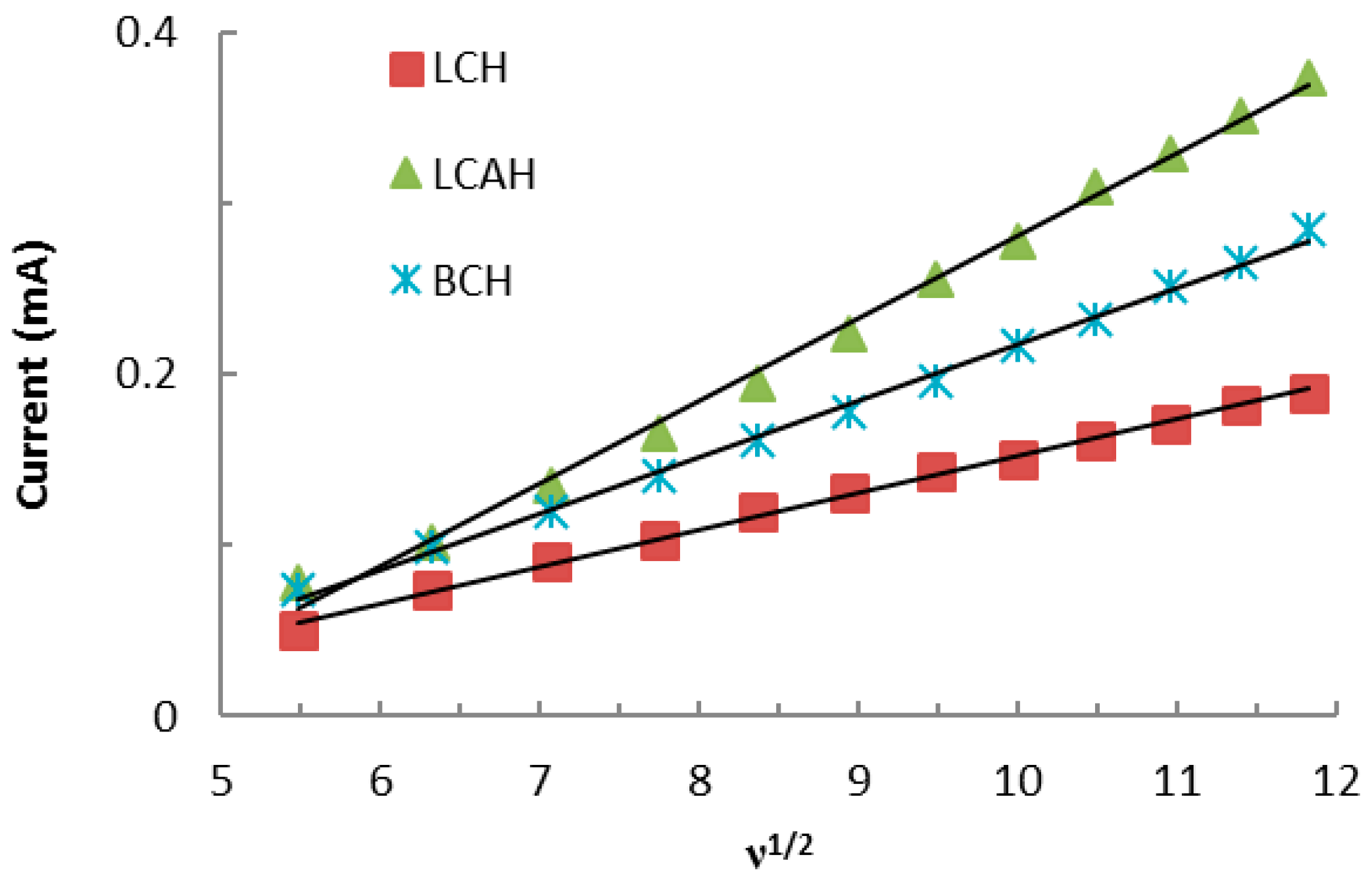
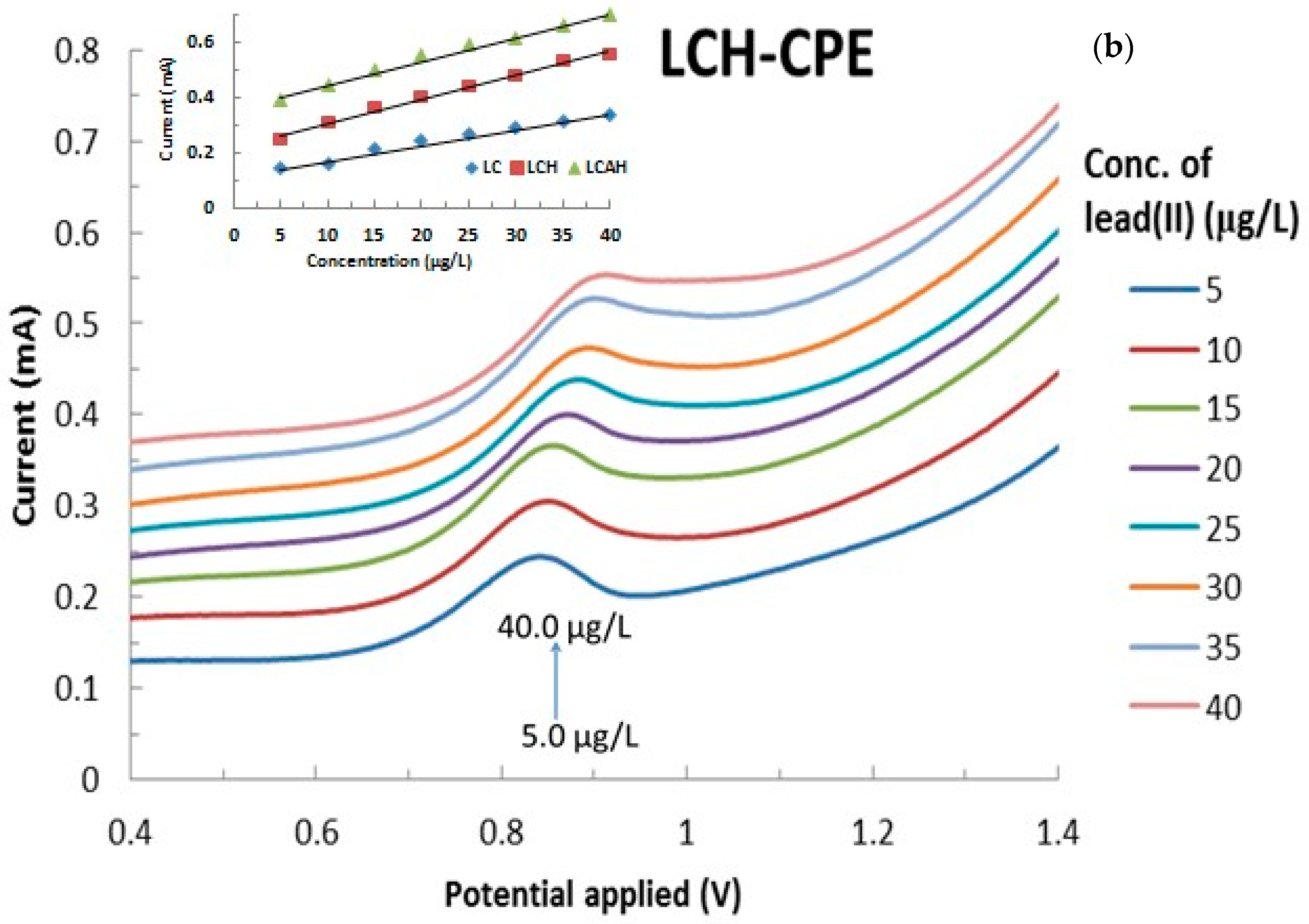
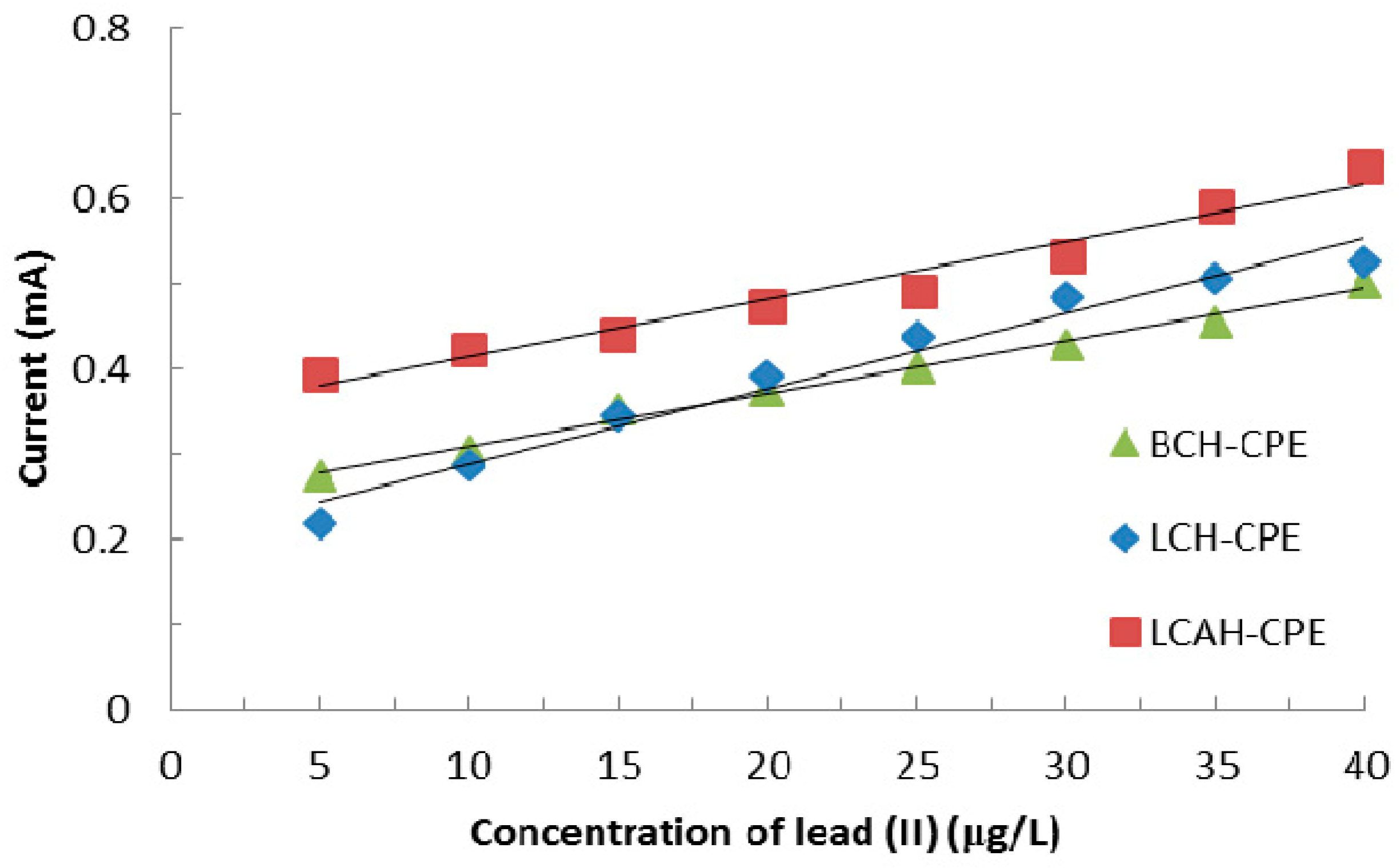
3.5. Electroanalysis Performance of Modified CPEs for Lead(II) Detection
3.6. Interferences Studies
3.7. Detection of Lead in Tap Water Sample
4. Conclusions
Supplementary Materials
Acknowledgment
Author Contributions
Conflicts of Interest
Abbreviations
| BC | Bentonite |
| LC | Local Clay |
| CPE | Carbon paste electrode |
| GCE | Glassy carbon paste electrode |
| HDTMA | hexdadecyl trimethyl ammonium bromide |
| BCH | HDTMA loaded bentonite |
| LCH | HDTMA loaded local clay |
| LCAH | Aluminum pillared local clay |
| XRD | X-ray diffraction |
| SEM | Scanning electron microscopy |
| FT-IR | (Fourier Transform Infra-Red) |
| BET | Brunauer, Emmett and Teller |
| XRF | X-ray fluorescence |
| DL | Detection limit |
| QL | Quantification limit |
References
- Wang, N.; Kanhere, E.; Miao, J.; Triantafyllou, M.S. Miniaturized chemical sensor with bio-inspired micropillar workingelectrode array for lead detection. Sens. Actuators B 2016, 233, 249–256. [Google Scholar] [CrossRef]
- Goyer, R.A. Lead toxicity: Current concerns. Environ. Health Perspect. 1993, 100, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, R.M.; Raghunath, R.; Mahapatra, S.; Sadasivan, S. Blood lead and its effect on Cd, Cu, Zn, Fe and hemoglobin levels of children. Sci. Total Environ. 2001, 277, 161–168. [Google Scholar] [CrossRef]
- Marsden, P.A. Increased body lead burden-cause or consequence of chronicrenal insufficiency? N. Engl. J. Med. 2003, 348, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M. Fetal death and reduced birth rates associated with exposure tolead-contaminated drinking water. Environ. Sci. Technol. 2014, 48, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Arantes, T.M.; Sardinha, A.; Baldan, M.R.; Cristovan, F.H.; Ferreira, N.G. Lead detection using micro/nanocrystalline boron-doped diamond by square-wave anodic stripping voltammetry. Talanta 2014, 128, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.; Zhang, Y.; Li, D.; Barrow, C.J.; Wang, H.; Yang, W. A biomimetic sensor for the detection of lead in water. Biosens. Bioelectron. 2015, 67, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wei, L.; Liu, X.; Lei, L.; Li, G. Ultrasensitive detection of lead ion based on target induced assembly of DNAzyme modified gold nanoparticle and graphene oxide. Anal. Chim. Acta 2014, 831, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gu, W.; Zhang, C.; Shi, X.; Xian, Y. In situ regulation nanoarchitecture of Au nanoparticles/reduced graphene oxide colloid for sensitive and selective SERS detection of lead ions. J. Colloid Interface Sci. 2016, 465, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Kamaruddin, N.H.; Bakar, A.A.A.; Yaacob, M.H.; Mahdi, M.A.; Zan, M.S.D.; Shaari, S. Enhancement of chitosan-graphene oxide SPR sensor with a multi-metallic layers of Au-Ag-Au nanostructurefor lead(II) ion detection. Appl. Surf. Sci. 2016, 361, 177–184. [Google Scholar] [CrossRef]
- Ganiyu, S.O.; Le, T.X.H.; Bechelany, M.; Esposito, G.; van Hullebusch, E.D.; Oturana, M.A.; Cretin, M. A hierarchical CoFe-layered double hydroxide modified carbon-felt cathode for heterogeneous electro-Fenton process. J. Mater. Chem. A 2017, 5, 3655–3666. [Google Scholar] [CrossRef]
- Lee, S.M.; Zirlianngura; Anjudikkal, J.; Tiwari, D. Electrochemical sensor for trace determination of cadmium(II) from aqueous solutions: Use of hybrid materials precursors to natural clays. Int. J. Environ. Anal. Chem. 2016, 96, 490–504. [Google Scholar] [CrossRef]
- Xiao, L.; Wildgoose, G.G.; Compton, R.G. Sensitive electrochemical detection of Arsenic(III) using gold nanoparticle modified carbon nanotubes via anodic stripping voltammetry. Anal. Chim. Acta 2008, 620, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Mardegan, A.; Scopece, P.; Lamberti, F.; Meneghetti, M.; Moretto, L.M.; Ugo, P. Electroanalysis of trace inorganic arsenic with gold Nanoelectrode ensembles. Electroanalysis 2012, 24, 798–806. [Google Scholar] [CrossRef]
- Hamsawahini, K.; Sathishkumar, P.; Ahamad, R.; Yusoff, A.R.M. PVDF–ErGO–GRC electrode: A single set up electrochemical system for separation, pre-concentration and detection of lead ions in complex aqueous samples. Talanta 2016, 148, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.M.; Wang, Z.; Liu, X.; Chen, Z.; Liu, E. Glassy carbon electrode modified by graphene-gold nanocomposite coating for detection of trace lead ions in acetate buffer solution. Thin Solid Films 2015, 584, 85–89. [Google Scholar] [CrossRef]
- Han, X.J.; Zhou, S.F.; Fan, H.L.; Zhang, Q.X.; Liu, Y.Q. Mesoporous MnFe2O4 nanocrystal clusters for electrochemistry detection of lead by stripping voltammetry. J. Electroanal. Chem. 2015, 755, 203–209. [Google Scholar] [CrossRef]
- Li, X.; Wen, H.; Fu, Q.; Peng, D.; Yu, J.; Zhang, Q.; Huang, X. Morphology-dependent NiO modified glassy carbon electrode surfacefor lead(II) and cadmium(II) detection. Appl. Surf. Sci. 2016, 363, 7–12. [Google Scholar] [CrossRef]
- Xie, Y.L.; Zhao, S.Q.; Ye, H.L.; Yuan, J.; Song, P.; Hu, S.Q. Graphene/CeO2 hybrid materials for the simultaneous electrochemical detection of cadmium(II), lead(II), copper(II), and mercury(II). J. Electroanal. Chem. 2015, 757, 235–242. [Google Scholar] [CrossRef]
- Chaiyo, S.; Apiluk, A.; Siangproh, W.; Chailapakula, O. High sensitivity and specificity simultaneous determination of lead, cadmium and copper using μPAD with dual electrochemical and colorimetric detection. Sens. Actuators B 2016, 233, 540–549. [Google Scholar] [CrossRef]
- Salih, F.E.; Ouarzane, A.; Rhazi, M.E. Electrochemical detection of lead (II) at bismuth/Poly(1,8-diaminonaphthalene) modified carbon paste electrode. Arab. J. Chem. 2015. [Google Scholar] [CrossRef]
- Zirlianngura; Jamsheera, A.; Tiwari, D.; Lee, S.M. Efficient use of novel hybrid materials in the ultra-trace determination of arsenic from aqueous solutions: An electrochemical study. Desalination Water Treat. 2015, 57, 18730–18738. [Google Scholar] [CrossRef]
- Thanhmingliana; Tiwari, D. Efficient use of hybrid materials in the remediation of aquatic environment contaminated with micro-pollutant diclofenac sodium. Chem. Eng. J. 2015, 263, 364–373. [Google Scholar] [CrossRef]
- Thanhmingliana; Lee, S.M.; Tiwari, D.; Prasad, S.K. Efficient attenuation of 17α-ethynylestradiol (EE2) and tetracycline using novel hybrid materials: Batch and column reactor studies. RSC Adv. 2015, 5, 46834–46842. [Google Scholar] [CrossRef]
- Aroke, U.O.; El-Nafaty, U.A. XRF, XRD and FTIR properties and characterization of HDTMA-Br surface modified organo-kaolinite clay. Int. J. Emerg. Technol. Adv. Eng. 2014, 4, 817–825. [Google Scholar]
- Eisazadeh, A.; Kassim, K.A.; Nur, H. Solid–state NMR and FTIR studies of lime stabilized montmorillonitic and lateritic clays. Appl. Clay Sci. 2012, 67–68, 5–10. [Google Scholar] [CrossRef]
- Tabak, A.; Yilmaz, N.; Eren, E.; Caglar, B.; Afsin, B.; Sarihan, A. Structural analysis of naproxen–intercalated bentonite (Unye). Chem. Eng. J. 2011, 174, 281–288. [Google Scholar] [CrossRef]
- Slimani, M.S.; Ahlafi, H.; Moussout, H.; Boukhlifi, F.; Zegaoui, O. Adsorption of hexavalent chromium and phenol onto bentonite modified with hexadecyltrimethylammonium bromide (HDTMABr). J. Adv. Chem. 2014, 8, 1602–1611. [Google Scholar]
- Jović-Jovičić, N.; Milutinović-Nikolić, A.; Banković, P.; Dojčinović, B.; Nedić, B.; Gržetić, I.; Jovanović, D. Synthesis, characterization and adsorptive properties of organobentonites. Acta Phys. Pol. A 2010, 117, 849–854. [Google Scholar] [CrossRef]
- Jiang, L.H.; Yao, M.G.; Liu, B.; Li, Q.J.; Liu, R.; Lv, H.; Lu, S.C.; Chen, C.; Zou, B.; Cui, T.; et al. Controlled synthesis of CeO2/graphene nanocomposites with highly enhanced optical and catalytic properties. J. Phys. Chem. C 2012, 116, 11741–11745. [Google Scholar] [CrossRef]
- Ndlovu, T.; Mamba, B.B.; Sampath, S.; Krause, R.W.; Arotiba, O.A. Voltammetric detection of arsenic on a bismuth modified exfoliated graphite electrode. Electrochim. Acta 2014, 128, 48–53. [Google Scholar] [CrossRef]
- Salinas-Torres, D.; Huerta, F.; Montilla, F.; Morallon, E. Study on electroactive and electrocatalytic surfaces of single walled carbon nanotube-modified electrodes. Electrochim. Acta 2011, 56, 2464–2670. [Google Scholar] [CrossRef]
- Tiwari, D.; Kim, H.U.; Lee, S.M. Removal behavior of sericite for Cu(II) and Pb(II) from aqueous solutions: Batch and column studies. Sep. Purif. Technol. 2007, 57, 11–16. [Google Scholar] [CrossRef]
- Wei, Y.; Yang, R.; Yu, X.Y.; Wang, L.; Liu, J.H.; Huang, X.J. Stripping voltammetry study of ultra-trace toxic metal ions on highly selectively adsorptive porous magnesium oxide nanoflowers. Analyst 2012, 137, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Liu, Z.G.; Yu, X.Y.; Wang, L.; Liu, J.H.; Huang, X.J. O2-plasma oxidized multi-walled carbon nanotubes for Cd(II) and Pb(II) detection: Evidence of adsorption capacity for electrochemical sensing. Electrochem. Commun. 2011, 13, 1506–1509. [Google Scholar] [CrossRef]
- Tiwari, D.; Zirlianngura; Lee, S.M. Fabrication of efficient and selective total arsenic sensor using the hybrid materials modified carbon paste electrodes. J. Electroanal. Chem. 2017, 784, 109–114. [Google Scholar] [CrossRef]




| Components | Mass Percentage | |
|---|---|---|
| Bentonite | Local Clay | |
| SiO2 | 54.51 | 54.57 |
| Al2O3 | 21.87 | 21.89 |
| Fe2O3 | 12.46 | 12.45 |
| Na2O | 2.11 | 2.17 |
| MgO | 2.77 | 2.72 |
| P2O5 | 0.13 | 0.13 |
| K2O | 1.39 | 1.38 |
| CaO | 0.98 | 0.95 |
| TiO2 | 2.51 | 2.47 |
| MnO2 | 0.17 | 0.19 |
| Materials | Pore Size (nm) | BET Specific Surface Area (m2/g) |
|---|---|---|
| BC | 4.98 | 80.86 |
| BCH | 36.60 | 4.68 |
| LC | 30.67 | 2.94 |
| LCH | 13.12 | 11.87 |
| LCAH | 8.04 | 4.55 |
| Modified CPEs Used | Linear Equation Obtained | R2 | Surface Area Estimated (cm2) |
|---|---|---|---|
| BCH | y = 0.034x − 0.1124 | 0.997 | 4.739 |
| LCH | y = 0.0214x − 0.0626 | 0.998 | 3.088 |
| LCAH | y = 0.0484x − 0.2019 | 0.995 | 6.937 |
| Interfering Ions | Concentration of Pb(II) ± S.E. Obtained Using Different Electrodes | ||
|---|---|---|---|
| BCH | LCH | LCAH | |
| Pb(II) (Blank) | 28.40 ± 0.88 | 19.35 ± 0.93 | 27.70 ± 0.78 |
| Cd(II) | 26.86 ± 0.89 | 15.67 ± 0.99 | 27.80 ± 0.92 |
| Cu(II) | 31.23 ± 0.90 | 17.15 ± 0.79 | 29.30 ± 0.86 |
| Mn(II) | 34.94 ± 0.81 | 18.52 ± 0.92 | 29.40 ± 0.93 |
| Fe(III) | 28.34 ± 0.88 | 20.24 ± 0.89 | 27.20 ± 0.79 |
| EDTA | 37.44 ± 0.81 | 17.10 ± 0.90 | 27.90 ± 0.79 |
| Glycine | 41.98 ± 0.97 | 20.74 ± 0.87 | 24.17 ± 0.94 |
| PO43− | 34.10 ± 0.84 | 27.67 ± 0.95 | 26.17 ± 0.95 |
| Elements Present | Amount Analyzed (mg/L) | |
|---|---|---|
| AAS Analysis | Copper | Not detected |
| Cadmium | Not detected | |
| Lead | Not detected | |
| Calcium | 11.92 | |
| Magnesium | 4.62 | |
| TOC Analysis | TOC (total organic carbon) | 1.998 |
| IC (inorganic carbon) | 7.478 | |
| TC (total carbon) | 9.476 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zirlianngura; Tiwari, D.; Ha, J.-H.; Lee, S.-M. Efficient Use of Porous Hybrid Materials in a Selective Detection of Lead(II) from Aqueous Solutions: An Electrochemical Study. Metals 2017, 7, 124. https://doi.org/10.3390/met7040124
Zirlianngura, Tiwari D, Ha J-H, Lee S-M. Efficient Use of Porous Hybrid Materials in a Selective Detection of Lead(II) from Aqueous Solutions: An Electrochemical Study. Metals. 2017; 7(4):124. https://doi.org/10.3390/met7040124
Chicago/Turabian StyleZirlianngura, Diwakar Tiwari, Jeong-Hyub Ha, and Seung-Mok Lee. 2017. "Efficient Use of Porous Hybrid Materials in a Selective Detection of Lead(II) from Aqueous Solutions: An Electrochemical Study" Metals 7, no. 4: 124. https://doi.org/10.3390/met7040124
APA StyleZirlianngura, Tiwari, D., Ha, J.-H., & Lee, S.-M. (2017). Efficient Use of Porous Hybrid Materials in a Selective Detection of Lead(II) from Aqueous Solutions: An Electrochemical Study. Metals, 7(4), 124. https://doi.org/10.3390/met7040124







