Synthesis, Characterization, and Cytotoxicity of a Novel Gold(III) Complex with O,O′-Diethyl Ester of Ethylenediamine-N,N′-Di-2-(4-Methyl)Pentanoic Acid
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Methods
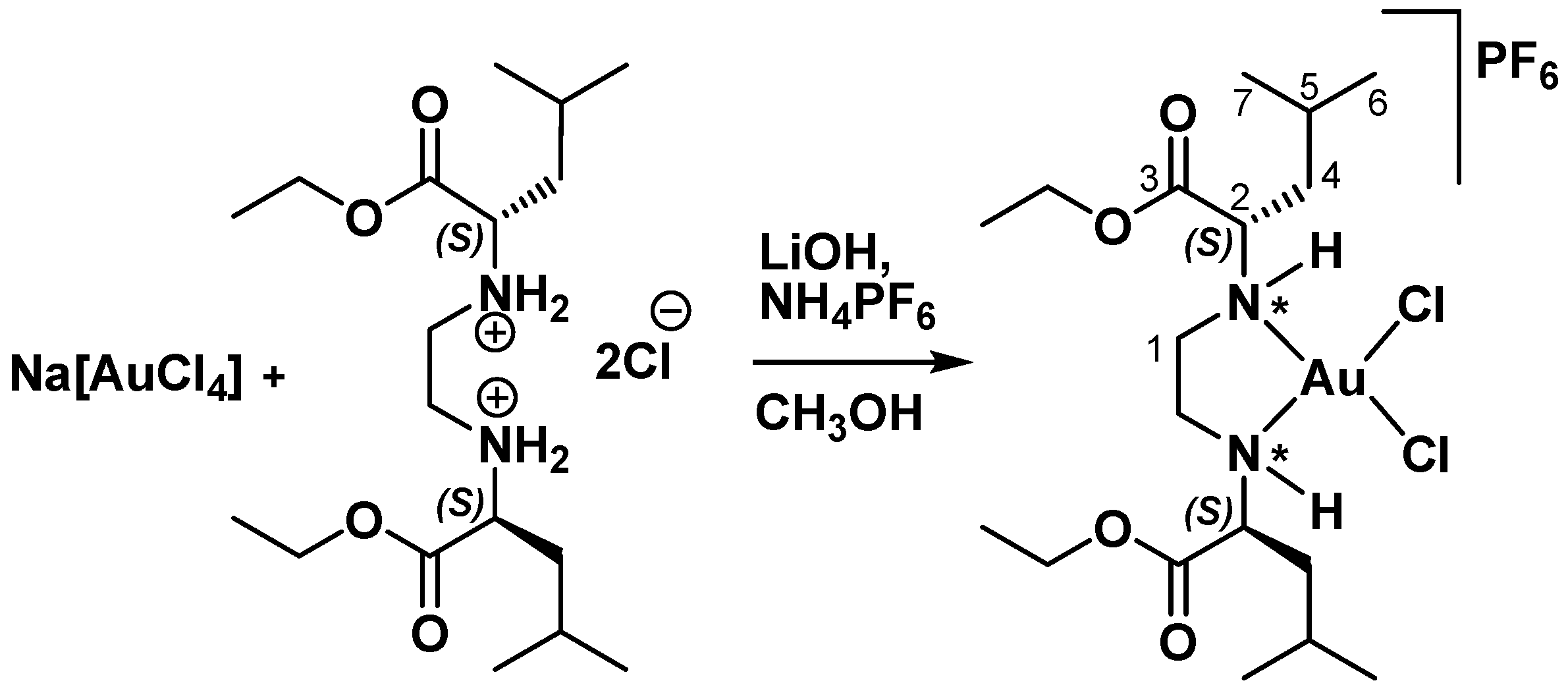
2.2. Synthesis of Complex [AuCl2{(S,S)-Et2eddl}]PF6
2.3. Computational Details
2.4. Biological Studies
2.4.1. Preparation of Drug Solutions
2.4.2. Cell Lines
2.4.3. Determination of Cell Survival
3. Results
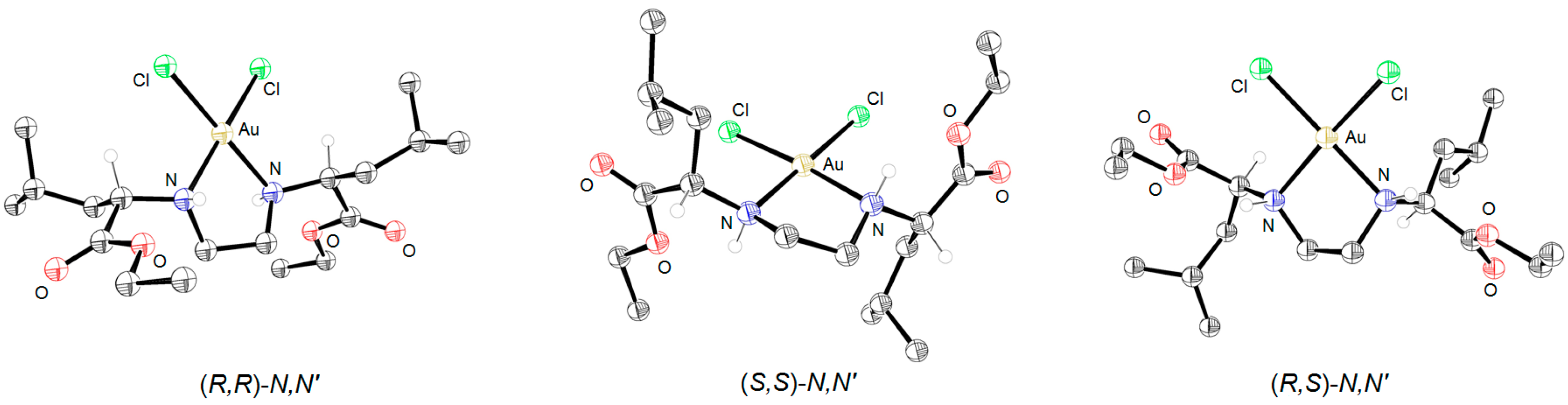
3.1. Synthesis and Characterization
3.2. Quantum Chemical Calculations
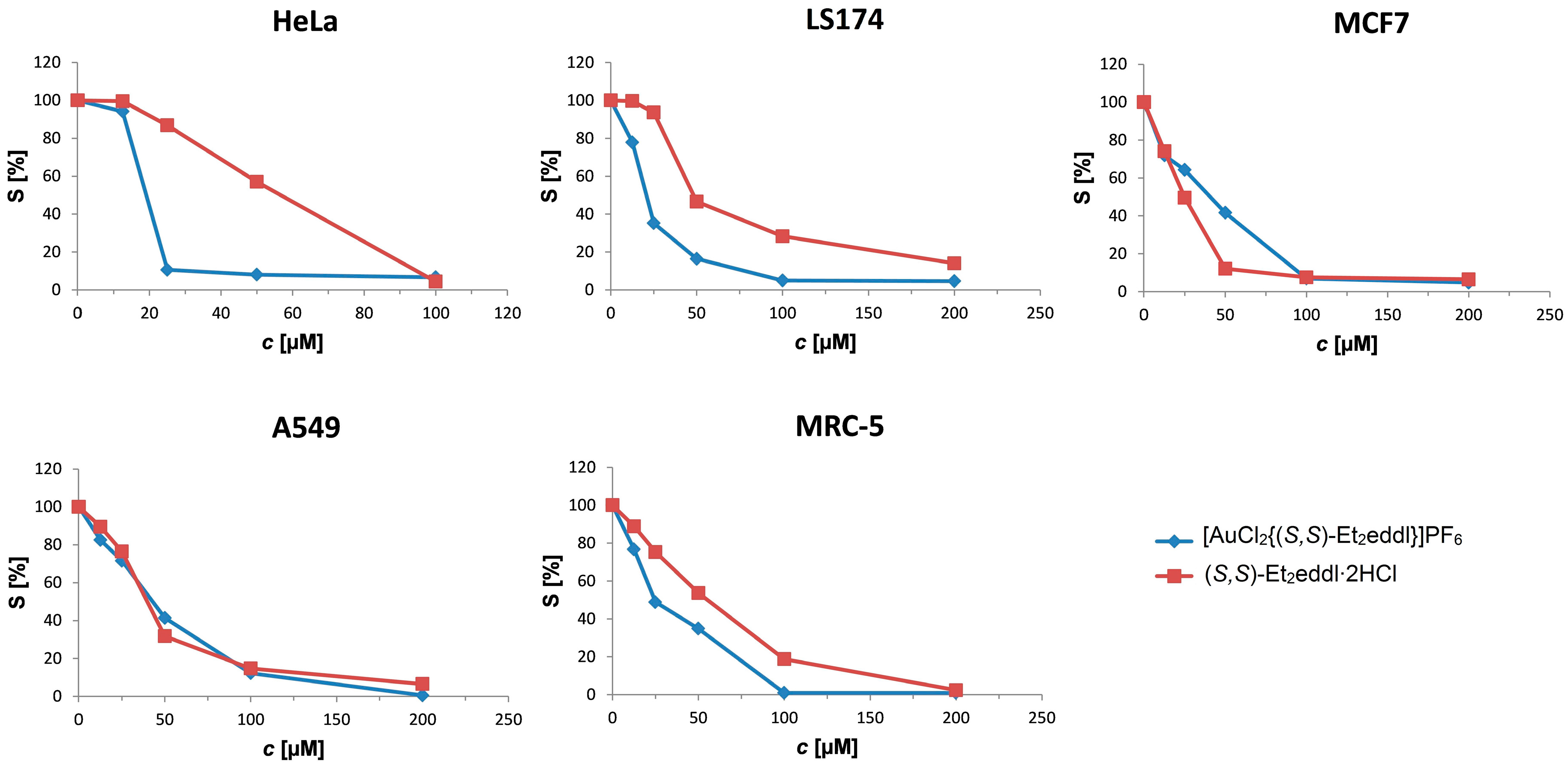
3.3. Biological Activity
3.4. Selectivity Study
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MeOH | Methanol |
| (S,S)-Et2eddl | O,O′-diethyl ester of ethylenediamine-N,N′-di-2-(4-methyl)pentanoic acid |
| HeLa | Human adenocarcinoma cell line |
| LS174 | Human colon carcinoma cell line |
| MCF7 | Human breast cancer cell line |
| A549 | Non-small cell lung carcinoma cell line |
| MRC-5 | Non-cancerous cell line human embryonic lung fibroblast |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| SD | Standard deviation |
| IR | Infrared spectroscopy |
| NMR | Nuclear magnetic resonance spectroscopy |
| HR ESI-MS | High-resolution electrospray ionization mass spectrometry |
References
- Chiang, A.C.; Massagué, J. Molecular basis of metastasis. N. Engl. J. Med. 2008, 359, 2814–2823. [Google Scholar] [CrossRef] [PubMed]
- Mubeen, M.; Kini, S.G. A review on the design and development of EGFR tyrosine kinase inhibitors in cancer therapy. Int. J. Ther. Appl. 2012, 5, 29–37. [Google Scholar]
- Rosenberg, B.; VanCamp, L.; Krigas, T. Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Kidani, Y.; Inagaki, K.; Iigo, M.; Hoshi, A.; Kuretani, K. Antitumor activity of 1,2-diaminocyclohexaneplatinum complexes against Sarcoma-180 ascites form. Med. Chem. 1978, 21, 1315–1318. [Google Scholar] [CrossRef]
- Harrap, K.R. Preclinical studies identifying carboplatin as a viable cisplatin alternative. Cancer Treat. Rev. 1985, 12, 21–33. [Google Scholar] [CrossRef]
- Knox, R.J.; Friedlos, F.; Lydall, D.A.; Roberts, J.J. Mechanism of cytotoxicity of anticancer platinum drugs: Evidence that cis-diamminedichloroplatinum(II) and cis-diammine-(1,1-cyclobutanedicarboxylato) platinum(II) differ only in the kinetics of their interaction with DNA. Cancer Res. 1986, 46, 1972–1979. [Google Scholar] [PubMed]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22, 7265–7279. [Google Scholar] [CrossRef] [PubMed]
- Kalinowska-Lis, U.; Ochocki, J.; Matlawska-Wasowska, K. Trans geometry in platinum antitumor complexes. Coord. Chem. Rev. 2008, 252, 1328–1345. [Google Scholar] [CrossRef]
- Wong, E.; Giandomenico, C.M. Current status of platinum-based antitumor drugs. Chem. Rev. 1999, 99, 2451–2466. [Google Scholar] [CrossRef] [PubMed]
- Lippert, B. Cisplatin: Chemistry and Biochemistry of a Leading Anticancer Drug; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar]
- Gómez, S.; Maksimović-Ivanić, D.; Mijatović, S.; Kaluđerović, G.N. On the Discovery, Biological Effects, and Use of Cisplatin and Metallocenes in Anticancer Chemotherapy. Bioinorg. Chem. Appl. 2012, 2012, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kaluđerović, G.N.; Paschke, R. Anticancer metallotherapeutics in preclinical development. Curr. Med. Chem. 2011, 18, 4738–4752. [Google Scholar] [CrossRef] [PubMed]
- Lakomska, I.; Fandzloch, M.; Muziol, T.; Liz, T.; Jezierska, J. Synthesis, characterization and antitumor properties of two highly cytotoxic ruthenium(III) complexes with bulky triazolopyrimidine ligands. Dalton Trans. 2013, 42, 6219–6226. [Google Scholar] [CrossRef] [PubMed]
- Guerroro, E.; Miranda, S.; Luttenberg, S.; Frohlich, N.; Koenen, J.; Mohr, F.; Cerrada, E.; Laguna, M. trans-Thionate Derivatives of Pt(II) and Pd(II) with Water-Soluble Phosphane PTA and DAPTA Ligands: Antiproliferative Activity against Human Ovarian Cancer Cell Lines. Inorg. Chem. 2013, 52, 6635–6647. [Google Scholar] [CrossRef] [PubMed]
- Matesans, A.I.; Leitao, I.; Souza, P. Palladium(II) and platinum(II) bis (thiosemicarbazone) complexes of the 2,6-diacetylpyridine series with high cytotoxic activity in cisplatin resistant A2780cisR tumor cells and reduced toxicity. J. Inorg. Biochem. 2013, 125, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gust, R. Metal N-heterocyclic carbene complexes as potential antitumor metallodrugs. Chem. Soc. Rev. 2013, 42, 755–773. [Google Scholar] [CrossRef] [PubMed]
- Casini, A.; Cinellu, M.A.; Minghetti, G.; Gabbiani, C.; Coronnello, M.; Mini, E.; Messori, L. Structural and solution chemistry, antiproliferative effects, and DNA and protein binding properties of a series of dinuclear gold(III) compounds with bipyridyl ligands. J. Med. Chem. 2006, 49, 5524–5531. [Google Scholar] [CrossRef] [PubMed]
- Aldinucci, D.; Lorenzon, D.; Stefani, L.; Giovagnini, L.; Colombatti, A.; Fregona, D. Antiproliferative and apoptotic effects of two new gold(III) methylsarcosinedithiocarbamate derivatives on human acute myeloid leukemia cells in vitro. Anticancer Drugs 2007, 18, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, N.; Zmejkovski, B.B.; Trifunović-Macedoljan, J.; Savić, A.; Stanković, D.; Damjanović, A.; Juranić, Z.; Kaluđerović, G.N.; Sabo, T.J. Gold(III) complexes with esters of cyclohexyl-functionalized ethylenediamine-N,N′-diacetate. J. Inorg. Biochem. 2013, 128, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, N.; Stanojković, T.P.; Zmejkovski, B.B.; Sabo, T.J.; Kaluđerović, G.N. In vitro anticancer activity of gold(III) complexes with some esters of (S,S)-ethylenediamine-N,N′-di-2-propanoic acid. Eur. J. Med. Chem. 2015, 90, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Casini, A.; Hartinger, C.; Gabbiani, C.; Mini, E.; Dyson, P.J.; Keppler, B.K.; Messori, L. Gold(III) compounds as anticancer agents: Relevance of gold-protein interactions for their mechanism of action. J. Inorg. Biochem. 2005, 102, 564–575. [Google Scholar] [CrossRef] [PubMed]
- Glišić, B.Ð.; Djuran, M.I.; Stanić, Z.D.; Rajković, S. Oxidation of methionine residue in Gly-Met dipeptide induced by [Au(en)Cl2]+ and influence of the chelated ligand on the rate of this redox process. Gold Bull. 2014, 47, 33–40. [Google Scholar] [CrossRef]
- Messori, L.; Marcon, G. Gold complexes as antitumor agents. Met. Ions Biol. 2004, 42, 385–424. [Google Scholar]
- Bindoli, A.; Rigobello, M.P.; Scutari, G.; Gabbiani, C.; Casini, A.; Messori, L. Thioredoxin reductase: A target for gold compounds acting as potential anticancer drug. Coord. Chem. Rev. 2009, 253, 1692–1707. [Google Scholar] [CrossRef]
- Vujić, J.M.; Cvijović, M.; Kaluđerović, G.N.; Milovanović, M.; Zmejkovski, B.B.; Volarević, V.; Arsenijević, N.; Sabo, T.J.; Trifunović, S.R. Palladium(II) complexes with R2edda derived ligands. Part IV. O,O′-dialkyl esters of (S,S)-ethylenediamine-N,N′-di-2-(4-methyl)-pentanoic acid dihydrochloride and their palladium(II) complexes: Synthesis, characterization and in vitro antitumoral activity against chronic lymphocytic leukemia (CLL) cells. Eur. J. Med. Chem. 2010, 45, 3601–3606. [Google Scholar]
- Brauer, G. Handbook of Preparative Inorganic Chemistry; Academic Press: New York, NY, USA, 1963; Volume 1. [Google Scholar]
- Gaussian 09; Revision D.01; Gaussian Inc.: Wallingford, CT, USA, 2009.
- Adamo, C.; Barone, V. Toward reliable adiabatic connection models free from adjustable parameters. Chem. Phys. Lett. 1997, 274, 242–250. [Google Scholar] [CrossRef]
- Dunning, T.H., Jr.; Hay, P.J. Modern Theoretical Chemistry, 3rd ed.; Plenum: New York, NY, USA, 1976; Volume 3, pp. 1–28. [Google Scholar]
- Andrae, D.; Häußermann, U.; Dolg, M.; Stoll, H.; Preuß, H. Energy-adjusted ab initio pseudopotentials for the second and third row transition elements. Theor. Chem. Acta 1990, 77, 123–141. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Ohno, M.; Abe, T. Rapid colorimetric assay for the quantification of leukemia inhibitory factor (LIF) and interleukin-6 (IL-6). J. Immunol. Methods 1991, 145, 199–203. [Google Scholar] [CrossRef]
- Kaluđerović, G.N.; Kommera, H.; Schwieger, S.; Schmidt, H.; Paethanom, A.; Kunze, M.; Paschke, R.; Steinborn, D. Synthesis, characterization, in vitro antitumoral investigations and interaction with plasmid pBR322 DNA of R2eddp-platinum(IV) complexes (R = Et, n-Pr). Dalton Trans. 2009, 48, 10720–10726. [Google Scholar] [CrossRef] [PubMed]
- Krajčinović, B.B.; Kaluđerović, G.N.; Steinborn, D.; Schmidt, H.; Wagner, C.; Žižak, Ž.; Juranić, Z.D.; Trifunović, S.R.; Sabo, T.J. Synthesis and in vitro antitumoral activity of novel O,O′-di-2-alkyl-(S,S)-ethylenediamine-N,N′-di-2-propanoate ligands and corresponding platinum(II/IV) complexes. J. Inorg. Biochem. 2008, 102, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Vujić, J.M.; Kaluđerović, G.N.; Milovanović, M.; Zmejkovski, B.B.; Volarević, V.; Živić, D.; Đurđević, P.; Arsenijević, N.; Trifunović, S.R. Stereospecific ligands and their complexes. Part VII. Synthesis, characterization and in vitro antitumoral activity of platinum(II) complexes with O,O′-dialkyl esters of (S,S)-ethylenediamine-N,N′-di-2-(4-methyl)pentanoic acid. Eur. J. Med. Chem. 2011, 46, 4559–4565. [Google Scholar] [CrossRef] [PubMed]
| Compounds | IC50 (µM) | ||||
|---|---|---|---|---|---|
| HeLa | LS174 | MCF7 | A549 | MRC5 | |
| [(S,S)-H2Et2eddl]Cl2 | 53.97 ± 2.72 | 53.23 ± 4.12 | 46.97 ± 2.19 | 38.44 ± 1.39 | 55.91 ± 0.61 |
| [AuCl2{(S,S)-Et2eddl}]PF6 | 18.25 ± 0.87 | 7.44 ± 1.19 | 41.10 ± 1.96 | 36.35 ± 1.75 | 24.94 ± 0.43 |
| Na[AuCl4] | 52.80 ± 2.93 | 39.89 ± 3.60 | 75.70 ± 0.38 | 45.66 ± 2.35 | 54.60 ± 3.11 |
| cisplatin | 6.90 ± 1.71 | 22.40 ± 0.44 | 18.13 ± 0.57 | 17.20 ± 0.82 | 14.21 ± 1.54 |
| Compounds | IC50 (MRC-5)/IC50 (cell line) | |||
|---|---|---|---|---|
| HeLa | LS174 | MCF7 | A549 | |
| [(S,S)-H2Et2eddl]Cl2 | 0.93 ± 0.04 | 1.05 ± 0.08 | 1.19 ± 0.06 | 1.45 ± 0.05 |
| [AuCl2{(S,S)-Et2eddl}]PF6 | 1.37 ± 0.07 | 3.35 ± 0.54 | 0.61 ± 0.03 | 0.69 ± 0.04 |
| Na[AuCl4] | 1.03 ± 0.08 | 1.37 ± 0.15 | 0.72 ± 0.04 | 1.20 ± 0.09 |
| cisplatin | 2.06 ± 0.56 | 0.63 ± 0.07 | 0.78 ± 0.09 | 0.83 ± 0.10 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pantelić, N.; Zmejkovski, B.B.; Marković, D.D.; Vujić, J.M.; Stanojković, T.P.; Sabo, T.J.; Kaluđerović, G.N. Synthesis, Characterization, and Cytotoxicity of a Novel Gold(III) Complex with O,O′-Diethyl Ester of Ethylenediamine-N,N′-Di-2-(4-Methyl)Pentanoic Acid. Metals 2016, 6, 226. https://doi.org/10.3390/met6090226
Pantelić N, Zmejkovski BB, Marković DD, Vujić JM, Stanojković TP, Sabo TJ, Kaluđerović GN. Synthesis, Characterization, and Cytotoxicity of a Novel Gold(III) Complex with O,O′-Diethyl Ester of Ethylenediamine-N,N′-Di-2-(4-Methyl)Pentanoic Acid. Metals. 2016; 6(9):226. https://doi.org/10.3390/met6090226
Chicago/Turabian StylePantelić, Nebojša, Bojana B. Zmejkovski, Dragana D. Marković, Jelena M. Vujić, Tatjana P. Stanojković, Tibor J. Sabo, and Goran N. Kaluđerović. 2016. "Synthesis, Characterization, and Cytotoxicity of a Novel Gold(III) Complex with O,O′-Diethyl Ester of Ethylenediamine-N,N′-Di-2-(4-Methyl)Pentanoic Acid" Metals 6, no. 9: 226. https://doi.org/10.3390/met6090226
APA StylePantelić, N., Zmejkovski, B. B., Marković, D. D., Vujić, J. M., Stanojković, T. P., Sabo, T. J., & Kaluđerović, G. N. (2016). Synthesis, Characterization, and Cytotoxicity of a Novel Gold(III) Complex with O,O′-Diethyl Ester of Ethylenediamine-N,N′-Di-2-(4-Methyl)Pentanoic Acid. Metals, 6(9), 226. https://doi.org/10.3390/met6090226









