Towards the Circularity of the EU Steel Industry: Modern Technologies for the Recycling of the Dusts and Recovery of Resources
Abstract
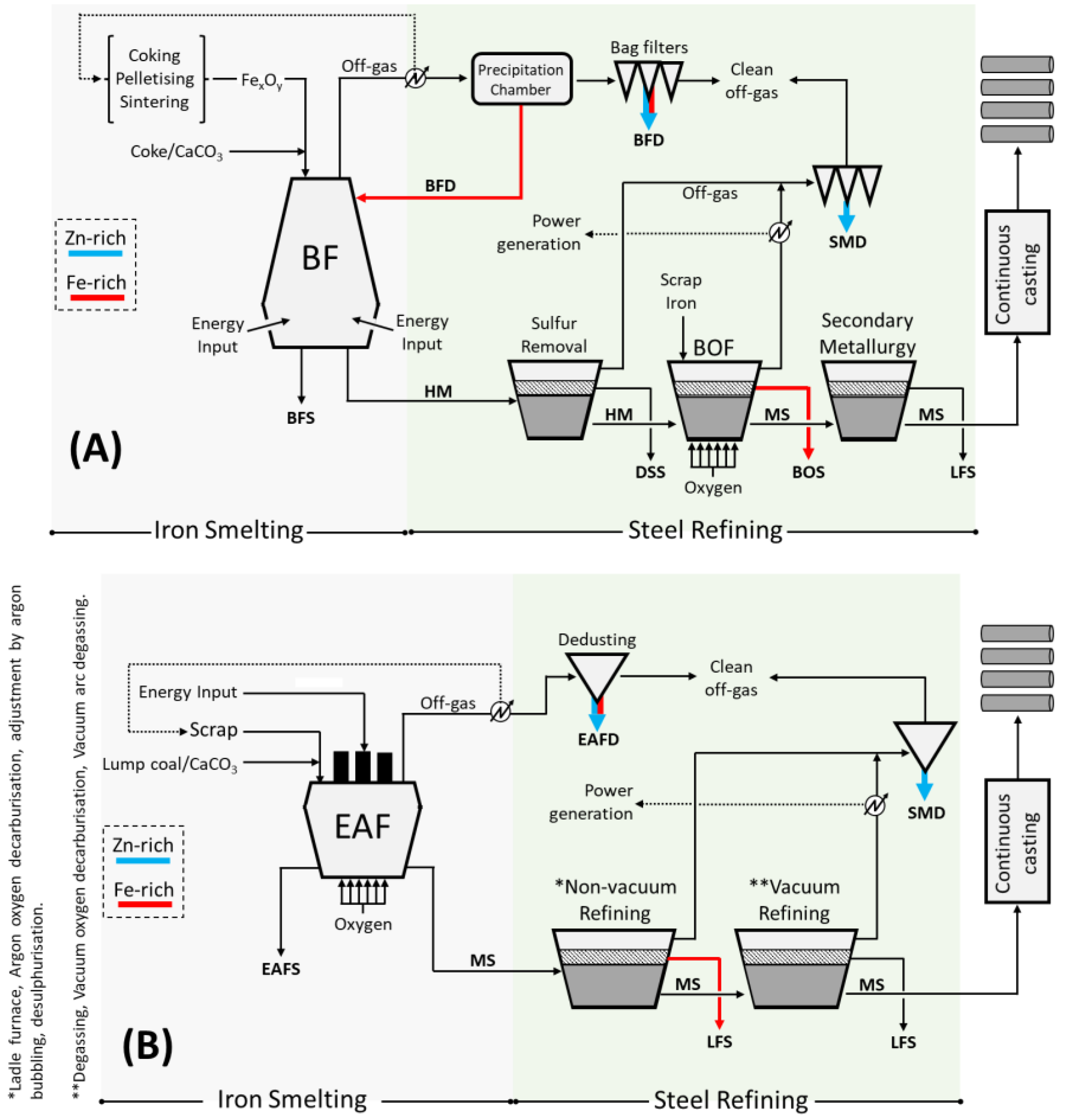
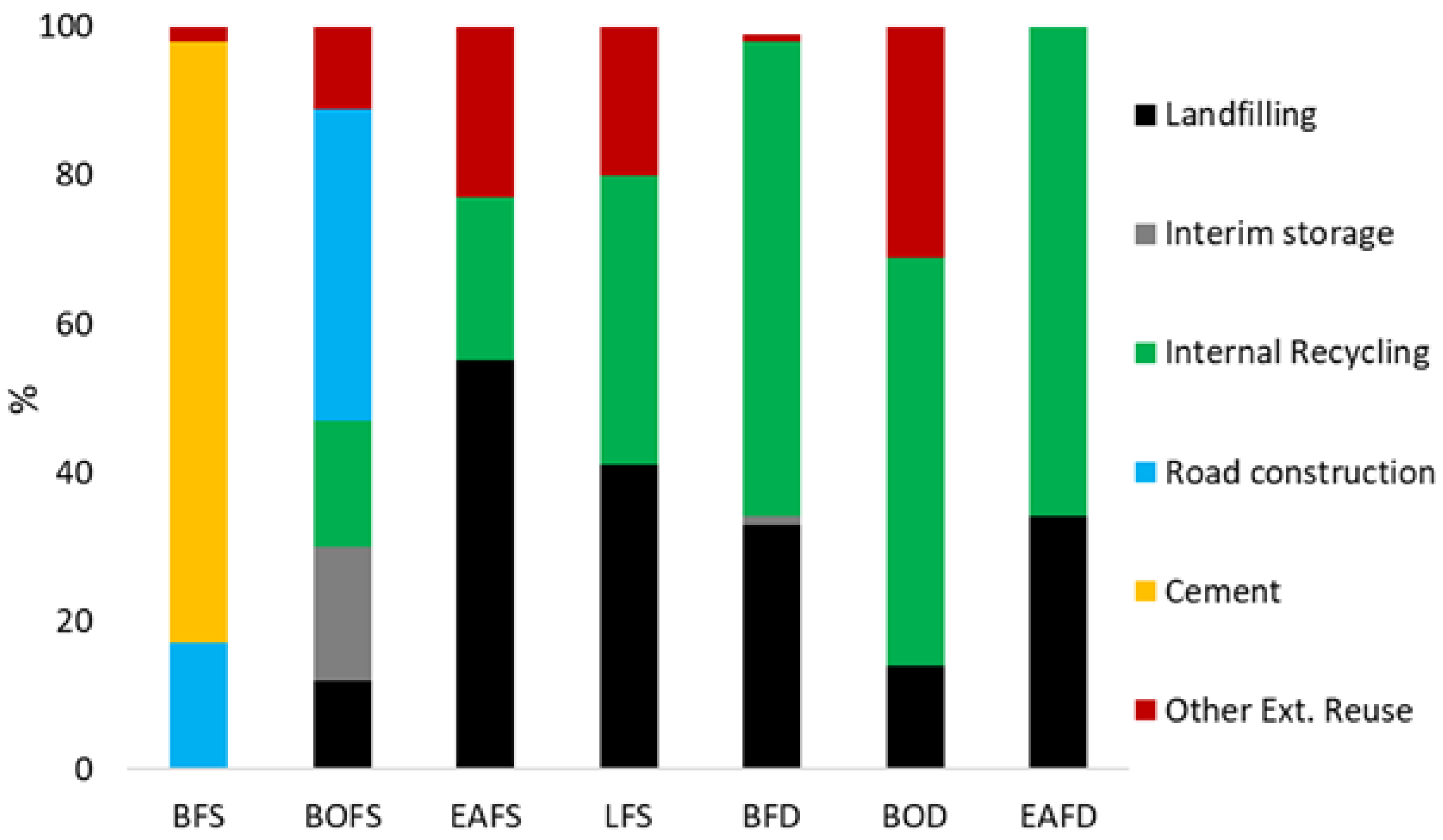
1. Introduction
2. Pyrometallurgical Processes: State-of-the-Art
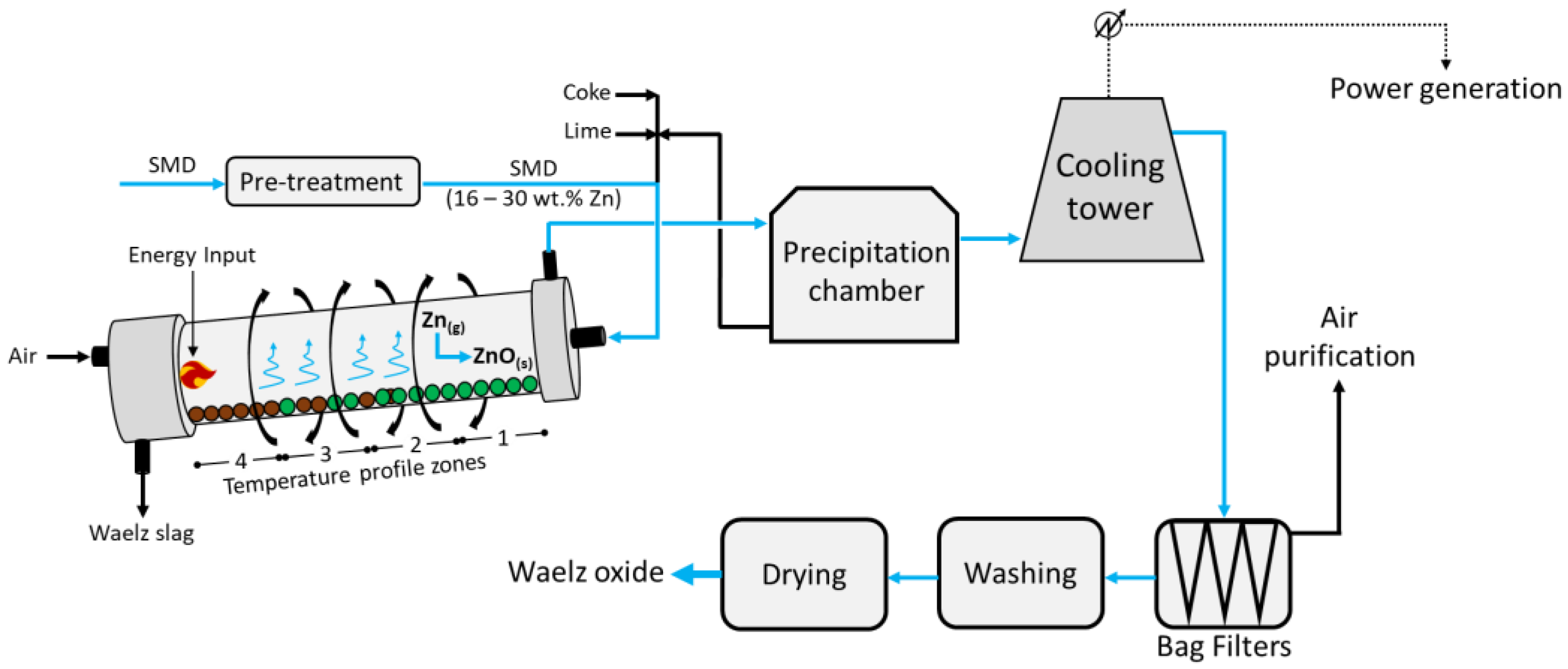
2.1. Waelz Kiln
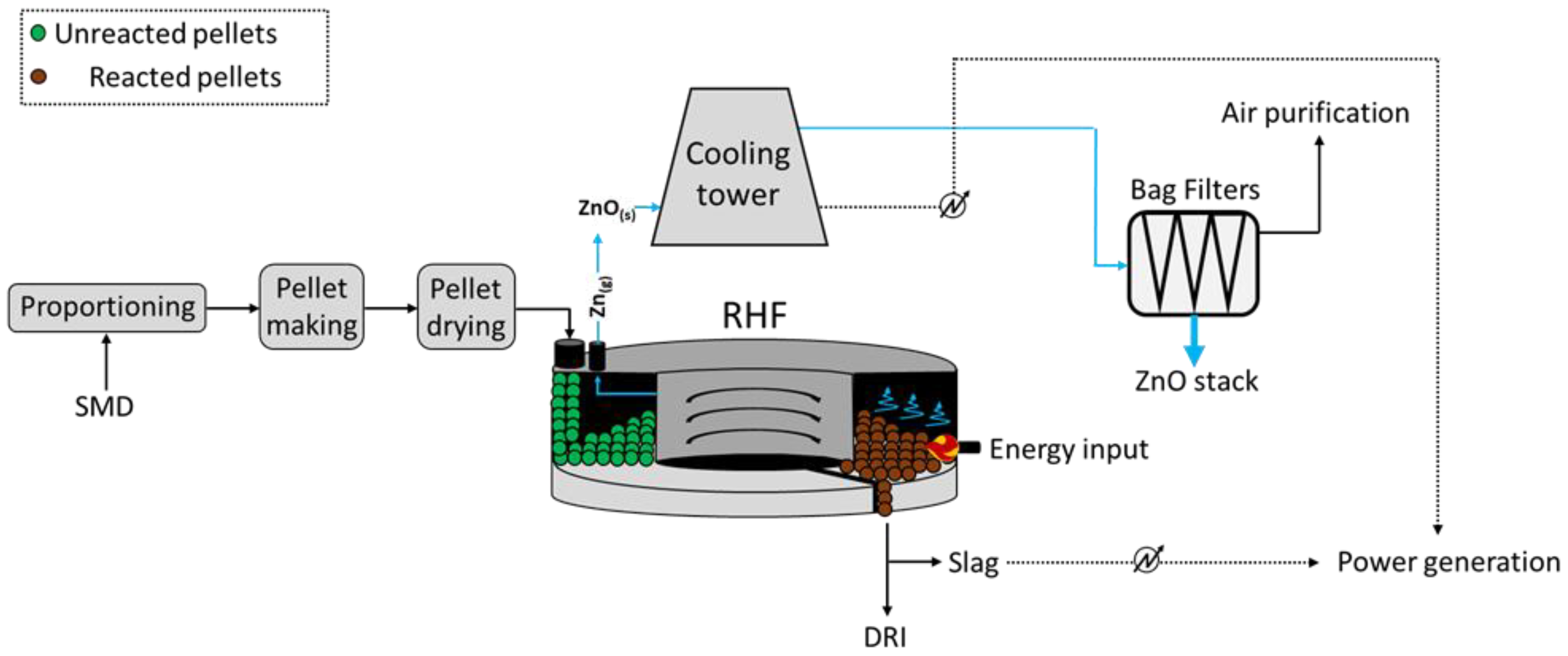
2.2. Rotary Hearth Furnace (RHF)
2.3. OxyCup Furnace
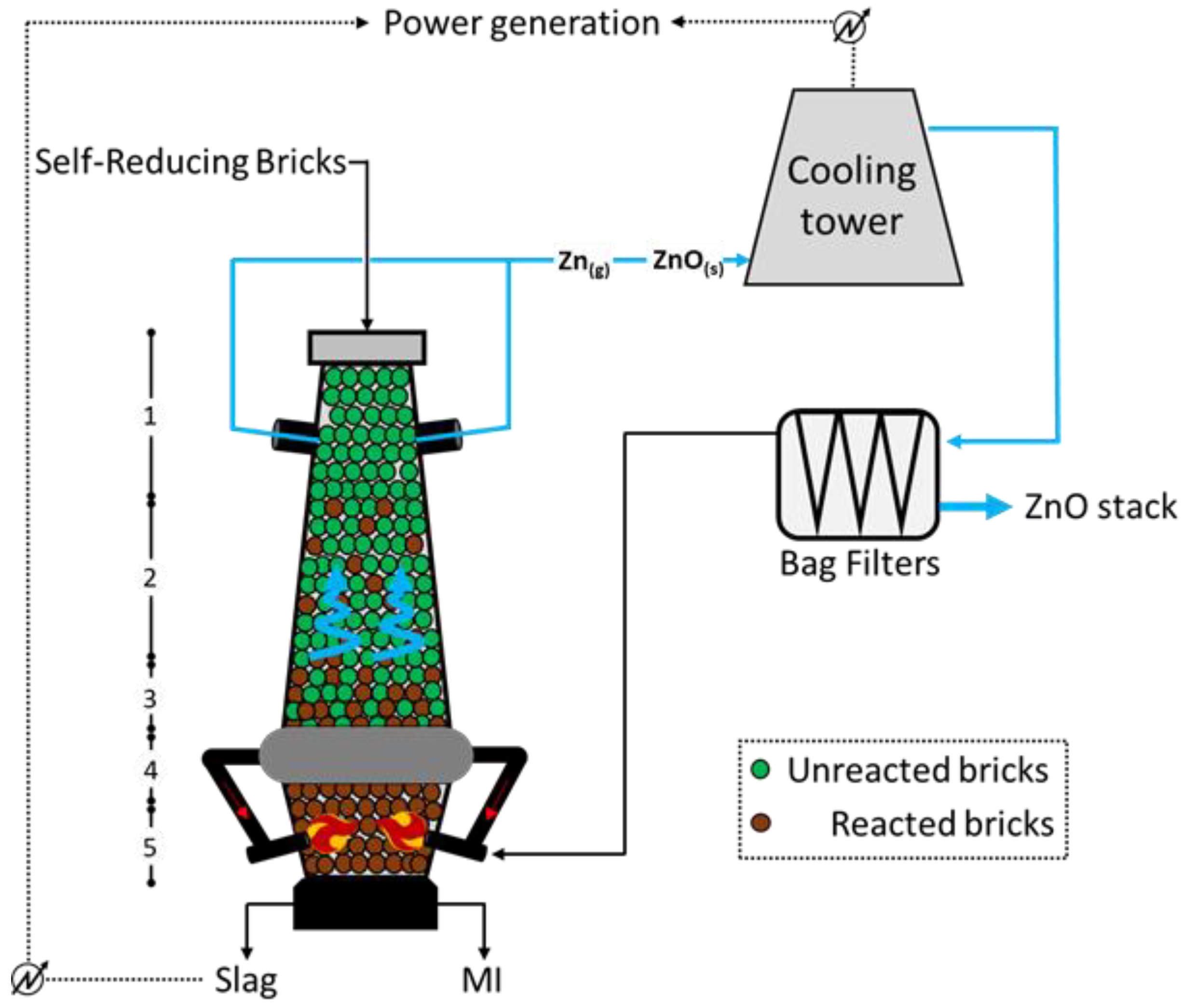
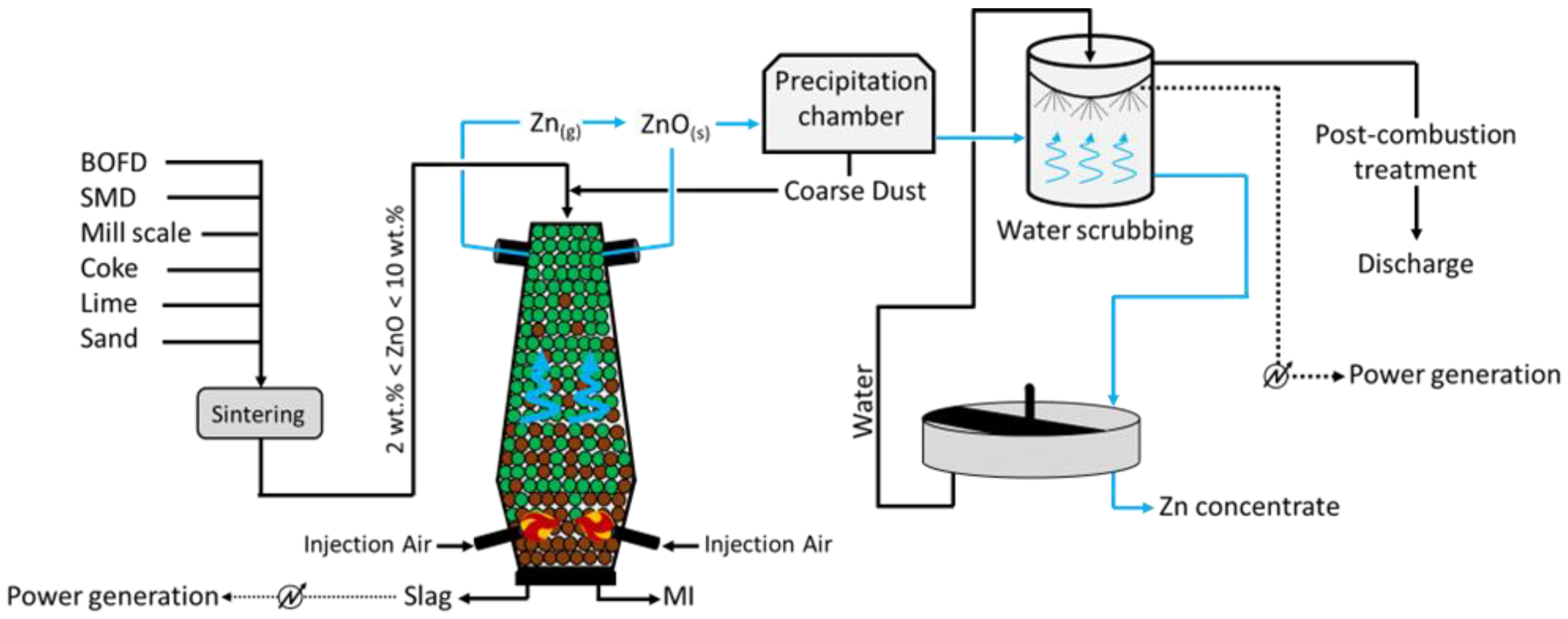
2.4. DK Process (Shaft Furnaces)
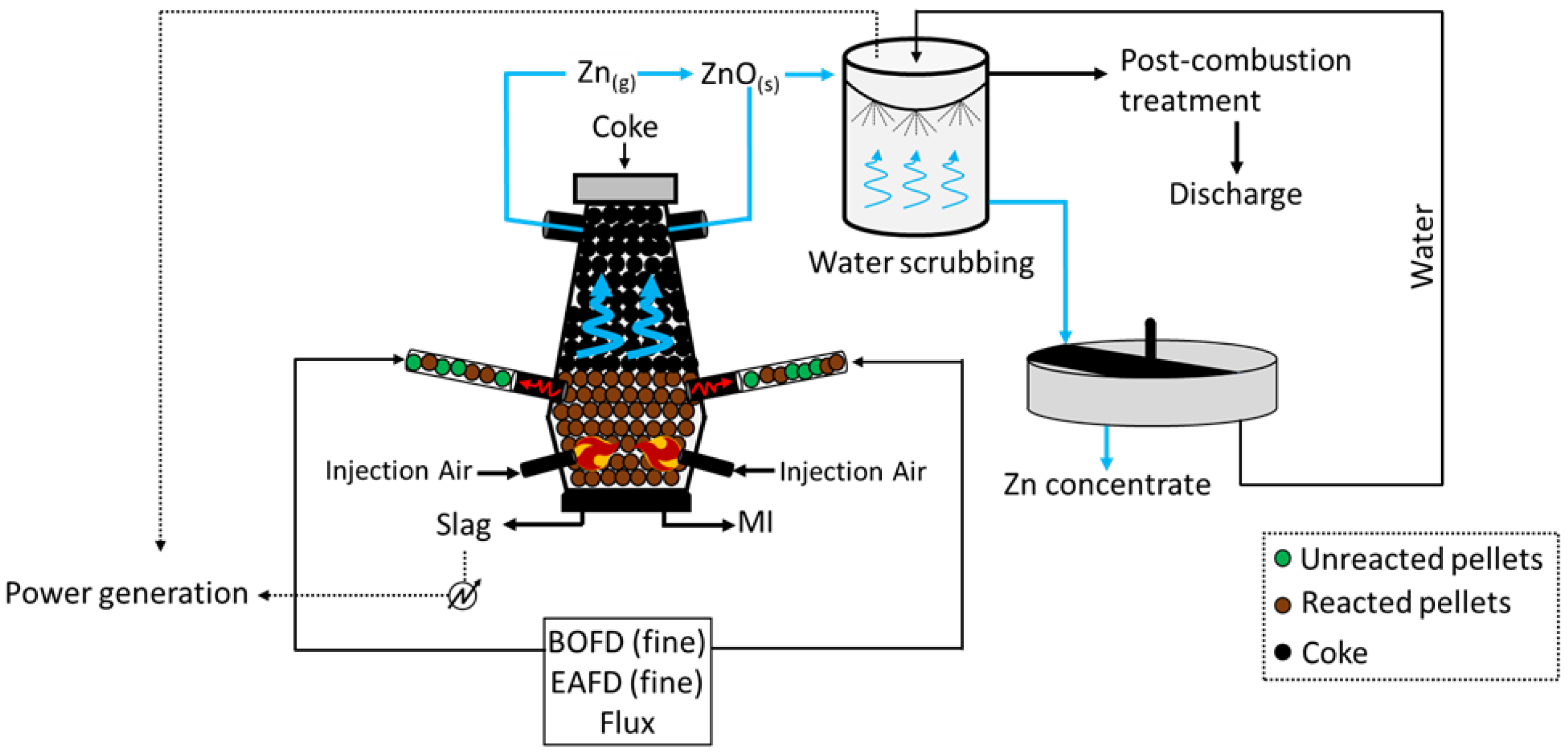
2.5. Coke-Packed Bed Furnace
2.6. Other Processes
3. Novel Routes for the Recovery of Fe and Zn
3.1. Recovery of Fe: Plasma Reactor
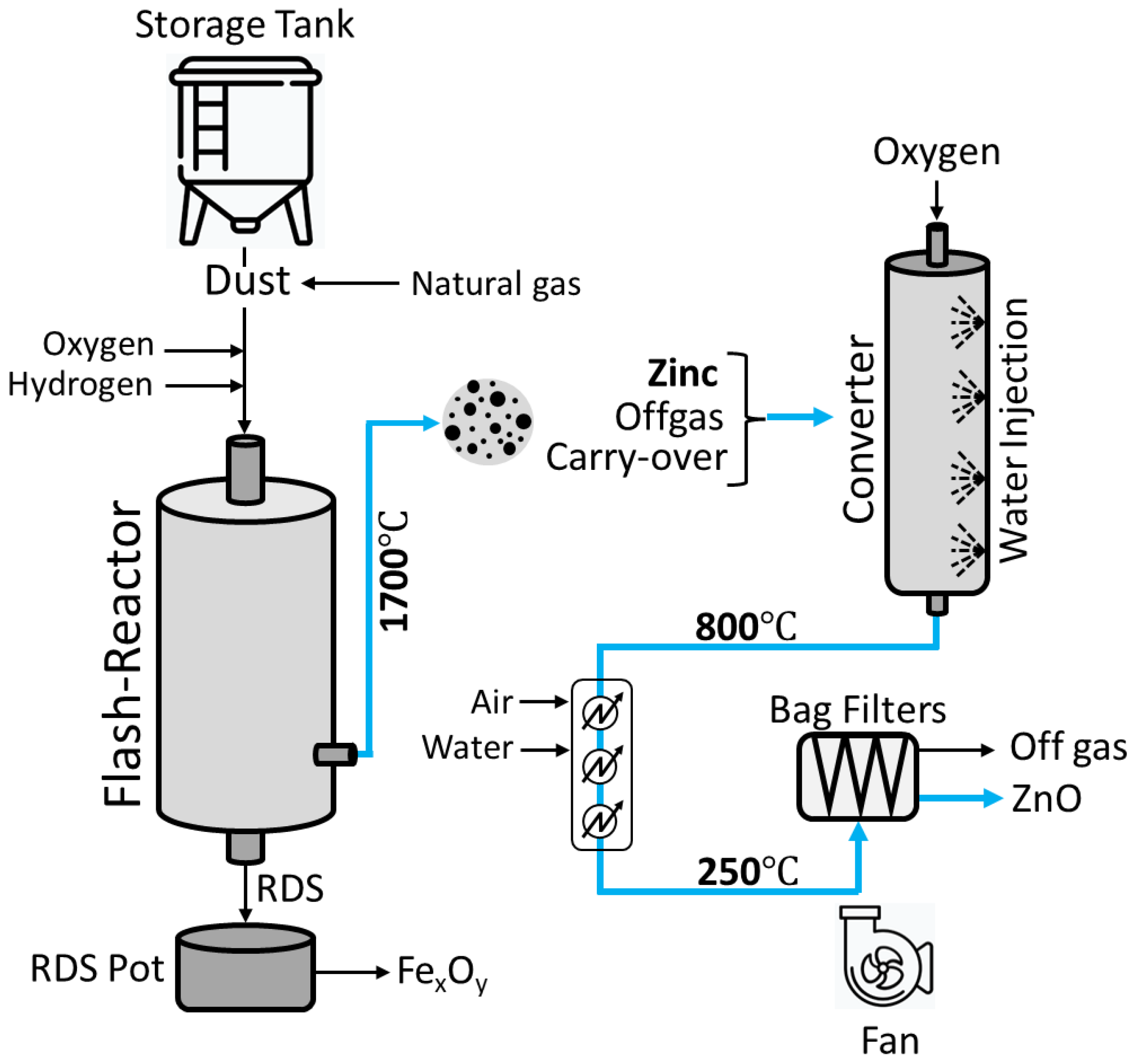
3.2. Recovery of Zn: RecoDust Process
4. Conclusions and Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Steel Association. Available online: https://worldsteel.org/steel-topics/statistics/world-steel-in-figures-2023/ (accessed on 22 September 2023).
- EUROFER—The European Steel Association. Available online: https://www.eurofer.eu/publications/brochures-booklets-and-factsheets/european-steel-in-figures-2023/ (accessed on 22 September 2023).
- Suer, J.; Traverso, M.; Jäger, N. Review of life cycle assessments for steel and environmental analysis of future steel production scenarios. Sustainability 2022, 14, 14131. [Google Scholar] [CrossRef]
- Holappa, L. A general vision for reduction of energy consumption and CO2 emissions from the steel industry. Metals 2020, 10, 1117. [Google Scholar] [CrossRef]
- Olabi, A.; Wilberforce, T.; Elsaid, K.; Sayed, E.T.; Maghrabie, H.M.; Abdelkareem, M.A. Large scale application of carbon capture to process industries—A review. J. Clean. Prod. 2022, 362, 132300. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, L.; Cui, K.; Wang, H.; Fu, T. Carbon capture and storage technology by steel-making slags: Recent progress and future challenges. Chem. Eng. J. 2023, 455, 140552. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, W.; Wang, Y.; Shen, J.; Wang, Y.; Geng, Z.; Wang, Q.; Zhu, T. Progress of CCUS technology in the iron and steel industry and the suggestion of the integrated application schemes for China. Chem. Eng. J. 2022, 450, 138438. [Google Scholar] [CrossRef]
- Shahabuddin, M.; Brooks, G.; Rhamdhani, M.A. Decarbonisation and hydrogen integration of steel industries: Recent development, challenges and technoeconomic analysis. J. Clean. Prod. 2023, 395, 13639. [Google Scholar] [CrossRef]
- Rieger, J.; Schenk, J. Residual processing in the European steel industry: A technological overview. J. Sustain. Metall. 2019, 2019, 295–309. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Cui, K.; Fu, T.; Gao, J.; Hussain, S.; AlGarni, T.S. Pyrometallurgical recovery of zinc and valuable metals from electric arc furnace dust–a review. J. Clean. Prod. 2021, 298, 126788. [Google Scholar] [CrossRef]
- Remus, R.; Roudier, S.; Delgado Sancho, L.; Aguado-Monsonet, M.A. Best Available Techniques (BAT) Reference Document for Iron and Steel Production—Industrial Emissions Directive 2010/75/EU—Integrated Pollution Prevention and Control, Publications Office. 2013. Available online: https://data.europa.eu/doi/10.2791/97469 (accessed on 30 January 2024).
- Pilz, K. Online-Analysis for Process Control at voestalpine Stahl GmbH. BHM Berg Hüttenmännische Monatshefte 2012, 157, 250–257. [Google Scholar] [CrossRef]
- Rudiger, D.; Hillmann, C. Metallurgie-Das Verhalten von Zink beim Recycling von eisenhaltigen Stauben und Schlammen-Eisenhaltige Staube und Schlamme konnen gut uber den Sinter-und Hochofenprozess wiederverwertet werden. Stahl Und Eisen 1999, 119, 53–60. [Google Scholar]
- EUROFER—European Steel Association The European Steel in Figures 2022. Available online: https://www.eurofer.eu/assets/publications/brochures-booklets-and-factsheets/european-steel-in-figures-2022/European-Steel-in-Figures-2022-v2.pdf (accessed on 22 September 2023).
- Colla, V.; Branca, T.A.; Pietruck, R.; Wölfelschneider, S.; Morillon, A.; Algermissen, D.; Snaet, D. Future Research and Developments on Reuse and Recycling of Steelmaking By-Products. Metals 2023, 13, 676. [Google Scholar] [CrossRef]
- RECOVERY—Recycling Technology Worldwide. Available online: https://www.recovery-worldwide.com/en/artikel/slag-recycling-3528047.html (accessed on 22 September 2023).
- Sabnavis, M.; Jagasheth, U.H.; Avachat, H.; Mishra, M. Zinc Industry: The Unsung Metal of the Economy; CARE Ratings: Professional Risk Opinion Report; CARE Ratings: Maharashtra, India, 31 October 2018; Available online: https://www.careratings.com/uploads/newsfiles/Zinc%20Industry.pdf (accessed on 30 January 2024).
- Statista. Available online: https://de.statista.com/statistik/daten/studie/520464/umfrage/verbrauchsanteile-von-zink-weltweit-nach-verwendungszweck/ (accessed on 22 September 2023).
- Khaliq, A.; Rhamdahini, M.A.; Brooks, G.; Masood, S. Metal extraction processes for electronic waste and existing industrial routes: A review and Australian perspective. Resources 2014, 3, 152–179. [Google Scholar] [CrossRef]
- Maccagni, M.G. INDUTEC®/EZINEX® Integrate Process on Secondary Zinc-Bearing Materials. J. Sustain. Metall. 2016, 2, 133–140. [Google Scholar] [CrossRef]
- Kelebek, S.; Yoruk, S.; Davis, B. Characterization of basic oxygen furnace dust and zinc removal by acid leaching. Miner. Eng. 2004, 17, 285–291. [Google Scholar] [CrossRef]
- Binnemans, K.; Jones, P.T.; Fernandze, A.M.; Torres, V.M. Hydrometallurgical processes for the recovery of metals from steel industry by-products: A critical review. J. Sustain. Metall. 2020, 6, 505–540. [Google Scholar] [CrossRef]
- Thottempudi, K.; Roy, G.G. A review on processing of electric arc furnace dust (EAFD) by pyro-metallurgical processes. Trans. Indian Inst. Met. 2022, 75, 1101–1112. [Google Scholar]
- Xue, Y.; Hao, X.; Liu, X.; Zhang, N. Recovery of zinc and iron from steel mill dust—An overview of available technologies. Materials 2022, 15, 4127. [Google Scholar] [CrossRef]
- Antrekowitsch, J.; Rösler, G.; Steinacker, S. State of the art in steel mill dust recycling. Chem. Ing. Tech. 2015, 87, 1498–1503. [Google Scholar] [CrossRef]
- Grudinsky, P.I.; Zinoveev, D.V.; Dyubanov, V.G.; Kozlov, P.A. State of the art and prospect for recycling of Waelz slag from electric arc furnace dust processing. Inorg. Mater. Appl. Res. 2019, 10, 1220–1226. [Google Scholar] [CrossRef]
- Strohmeier, G.; Bonestell, J. Steelworks residues and the Waelz kiln treatment of electric arc furnace dust. Iron Steel Eng. 1996, 73, 4. [Google Scholar]
- Chipman, J. Thermodynamics and phase diagram of the Fe-C system. Metall. Mater. Trans. B 1972, 3, 55–64. [Google Scholar] [CrossRef]
- Beyzavi, A.L.; Mattich, C. Operational practice with the Waelz kiln and leaching plant of TSU in Taiwan. In Recycling of Metals and Engineered Materials; Stewart, D.L., Jr., Daley, J.C., Stephens, R.L., Eds.; The Minerals, Metals and Materials Society: Pittsburgh, PA, USA, 2000. [Google Scholar] [CrossRef]
- EN 19694-2; Stationary Source Emissions-Greenhouse Gas (GHG) Emissions in Energy-Intensive Industries-Part 2: Iron and Steel Industry. International Organization for Standardization: Geneva, Switzerland, 2017.
- Anameric, B.; Kawatra, S.K. Properties and features of direct reduced iron. Miner. Process. Extr. Metall. Rev. 2007, 28, 59–116. [Google Scholar] [CrossRef]
- Ishikawa, H.; Kopfle, J.; Mcclelland, J.; Ripke, J. Rotary hearth furnace technologies for iron ore and recycling applications. Arch. Metall. Mater. 2008, 53, 541–545. [Google Scholar]
- Hiroshi, O.; Ibaraki, T.; Abe, Y. Dust recycling system by the rotary hearth furnace. Shinnittetsu Giho 2006, 384, 134. [Google Scholar]
- Kim, J.; Son, M.S.; Park, J.K.; Kim, J. Optimized rotary hearth furnace utilization with blast furnace and electric arc furnace: Techno-economics, CO2 reduction. Fuel Process. Technol. 2022, 237, 107450. [Google Scholar] [CrossRef]
- Tenova. Available online: https://tenova.com/technologies/rotary-hearth-furnace (accessed on 22 September 2023).
- Holtzer, M.; Kmita, A.; Roczniak, A. The recycling of materials containing iron and zinc in the OxyCup process. Arch. Foundry Eng. 2015, 15, 126–130. [Google Scholar]
- Kuttner. Available online: https://www.kuttner.com.br/Files/Filer/Downloads/en/Kuettner_shaft-furnace.pdf (accessed on 22 September 2023).
- Lamperle, M.; Jennes, R.; Cappel, J. Oxycup (R) Furnace Operation at TISCO, China. 2012. Available online: https://www.cappel-consult.com/fileadmin/user_upload/OxyCup_TISCO_Lemperle_2012v002.pdf (accessed on 22 September 2023).
- Sassen, K.J.; Hillmann, C. The DK Process—For the Recovery of Iron and Zinc from BOF Dusts and Sludges. April 2011. Available online: https://www.researchgate.net/publication/295165370_The_DK_process_-_For_the_recovery_of_iron_and_zinc_from_BOF_dusts_and_sludges (accessed on 18 January 2023).
- Fröhling, M.; Rentz, O. A case study on raw material blending for the recycling of ferrous wastes in a blast furnace. J. Clean. Prod. 2010, 18, 161–173. [Google Scholar] [CrossRef]
- Hamada, T.; Takeuchi, S.; Igawa, K.; Katayama, H.; Itaya, H.; Nakanishi, K. Pyrometallurgy ’87 Symposium; The Institute of Mining Metallurgy: London, UK, 1987. [Google Scholar]
- Yoshiaki, H.; Ishiwata, N.; Matsumoto, T. Smelting Reduction Process with a Coke Packed Bed for Steelmaking Dust Recycling. ISIJ Int. 2000, 40, 231–237. [Google Scholar]
- George, H.L.; Longbottom, R.J.; Chew, S.J.; Monaghan, B.J. Flow of molten slag through a coke packed bed. ISIJ Int. 2014, 54, 820–826. [Google Scholar] [CrossRef][Green Version]
- Orre, J.; Ökvist, L.S.; Bodén, A.; Björkman, B. Understanding of blast furnace performance with biomass introduction. Minerals 2021, 11, 157. [Google Scholar] [CrossRef]
- Roth, J.L.; Frieden, R.; Hansmann, T.; Monai, J.; Solvi, M. PRIMUS®, un nouveau procédé pour le recyclage des coproduits et la production de fonte. La Rev. De Métallurgie-CIT 2001, 988–996. [Google Scholar]
- Romenets, V.; Valavin, V.; Pokhvisnev, Y.; Vandariev, S. Processing industrial wastes with the liquid-phase reduction Romelt process. JOM 1999, 51, 33–37. [Google Scholar] [CrossRef]
- Reiter, W.; Rieger, J.; Raupenstrauch, H.; Cattini, L.; Maystrenko, N.; Kovalev, D.; Alexey, A.; Mitrofanov, A. Recovery of valuable materials with the RecoDust process. Metals 2023, 13, 1191. [Google Scholar] [CrossRef]

| CaO | Fe | SiO2 | C | Zn | Pb | Mn | Al2O3 | S | P2O5 | MgO | Others (4) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BFS (3) | 42.0 | 0.4 | 35.1 | - | - | - | 0.7 | 11.5 | 1.4 | - | 9.0 | 2.7 |
| BOFS | 49.6 | 14.0 | 15.0 | - | - | - | 3.4 | 1.6 | - | 7.0 | 3.1 | 1.5 |
| EAFS (2) | 28.0 | 32.0 | 19.0 | - | - | - | 5.0 | 7.0 | - | 0.4 | 7.0 | 2.1 |
| LFS | 27.0 | 20.0 | 18.0 | - | - | - | <0.5 | 8.0 | <4 | <0.2 | 10.0 | <0.1 |
| BFD | 10.5 | 21.0 | 6.0 | 31.0 | 5.5 | 1.4 | 0.13 | 2.7 | 2.4 | 0.23 | 10.2 | 0.4 |
| BOD (1) | 14.9 | 57.6 | - | 1.9 | 3.1 | 0.3 | - | - | 0.3 | - | - | <0.2 |
| EAFD (2) | 10.0 | 28.0 | 2.9 | 1.9 | 32.0 | 5.2 | 3.5 | 1.6 | 1.5 | 0.2 | 3.2 | 4.1 |
| CO2 Equivalent | MWh | |
|---|---|---|
| Waelz kiln | 1.2 | 3.0 |
| Rotary Hearth Furnace | 1.3 | 5.5 |
| OxyCup Furnace | 1.1 | 2.8 |
| DK process | 2.5 | N/A (*) |
| Coke-Packed Bed Furnace | 3.5 | 8.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simoni, M.; Reiter, W.; Suer, J.; Di Sante, L.; Cirilli, F.; Praolini, F.; Mosconi, M.; Guzzon, M.; Malfa, E.; Algermissen, D.; et al. Towards the Circularity of the EU Steel Industry: Modern Technologies for the Recycling of the Dusts and Recovery of Resources. Metals 2024, 14, 233. https://doi.org/10.3390/met14020233
Simoni M, Reiter W, Suer J, Di Sante L, Cirilli F, Praolini F, Mosconi M, Guzzon M, Malfa E, Algermissen D, et al. Towards the Circularity of the EU Steel Industry: Modern Technologies for the Recycling of the Dusts and Recovery of Resources. Metals. 2024; 14(2):233. https://doi.org/10.3390/met14020233
Chicago/Turabian StyleSimoni, Marco, Wolfgang Reiter, Julian Suer, Loredana Di Sante, Filippo Cirilli, Fabio Praolini, Manuel Mosconi, Marta Guzzon, Enrico Malfa, David Algermissen, and et al. 2024. "Towards the Circularity of the EU Steel Industry: Modern Technologies for the Recycling of the Dusts and Recovery of Resources" Metals 14, no. 2: 233. https://doi.org/10.3390/met14020233
APA StyleSimoni, M., Reiter, W., Suer, J., Di Sante, L., Cirilli, F., Praolini, F., Mosconi, M., Guzzon, M., Malfa, E., Algermissen, D., & Rieger, J. (2024). Towards the Circularity of the EU Steel Industry: Modern Technologies for the Recycling of the Dusts and Recovery of Resources. Metals, 14(2), 233. https://doi.org/10.3390/met14020233









