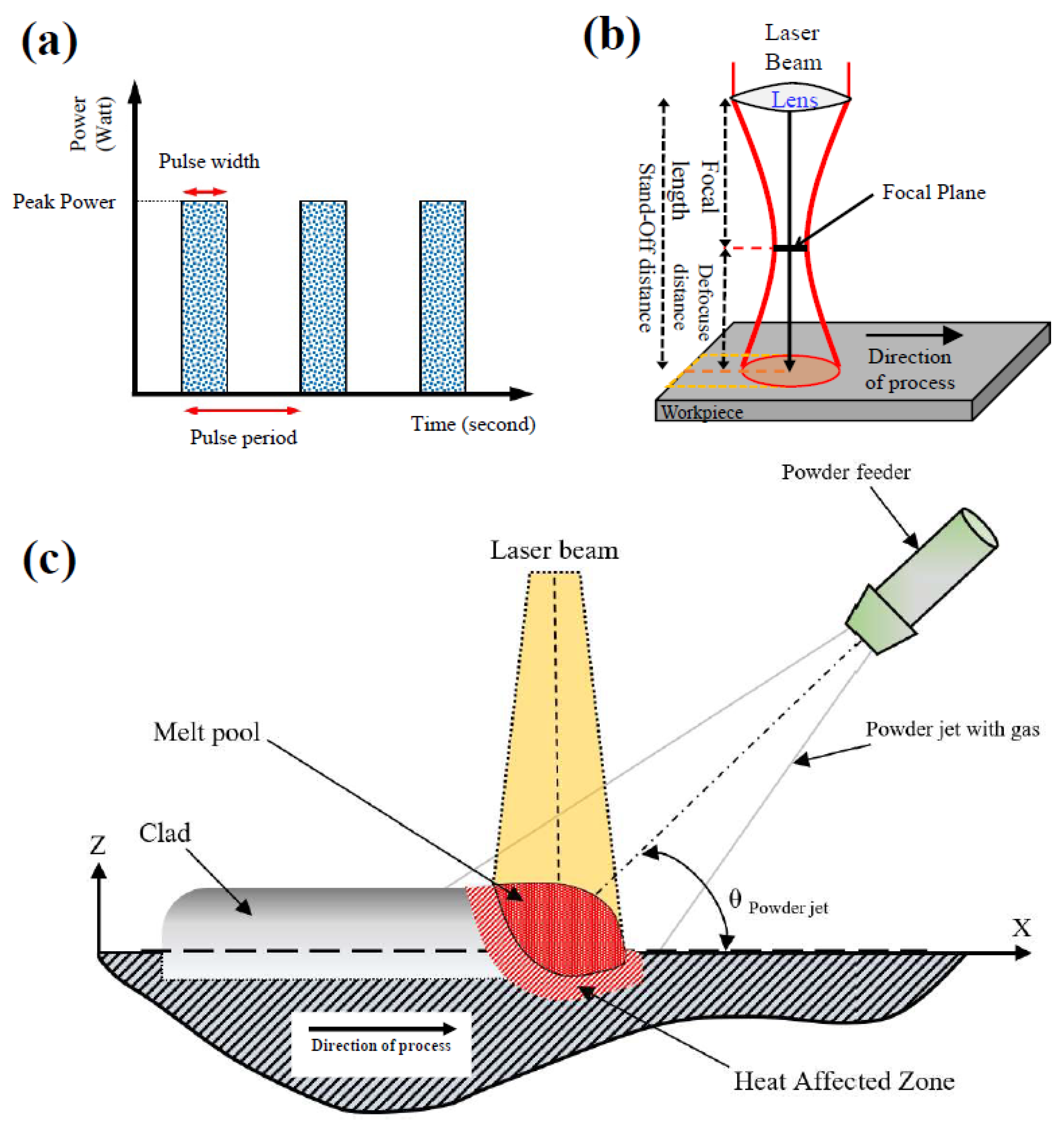
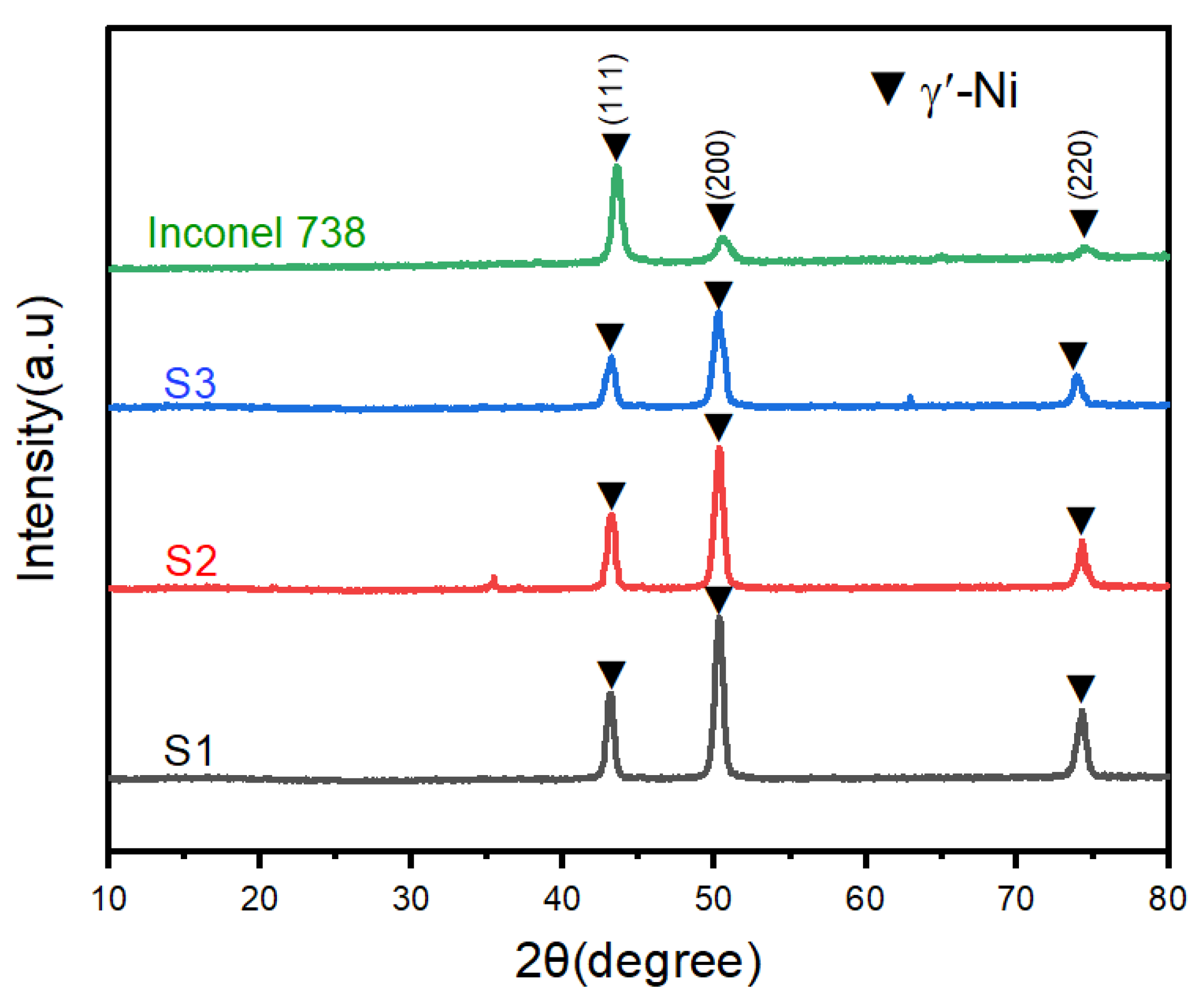
3.3. Corrosion Behavior of Inconel 625 Coating
The corrosion parameters obtained from the potentiodynamic polarization test are summarized in
Table 5.
To evaluate the corrosion resistance, a potentiodynamic polarization method was used for the samples in a 3.5% NaCl solution.
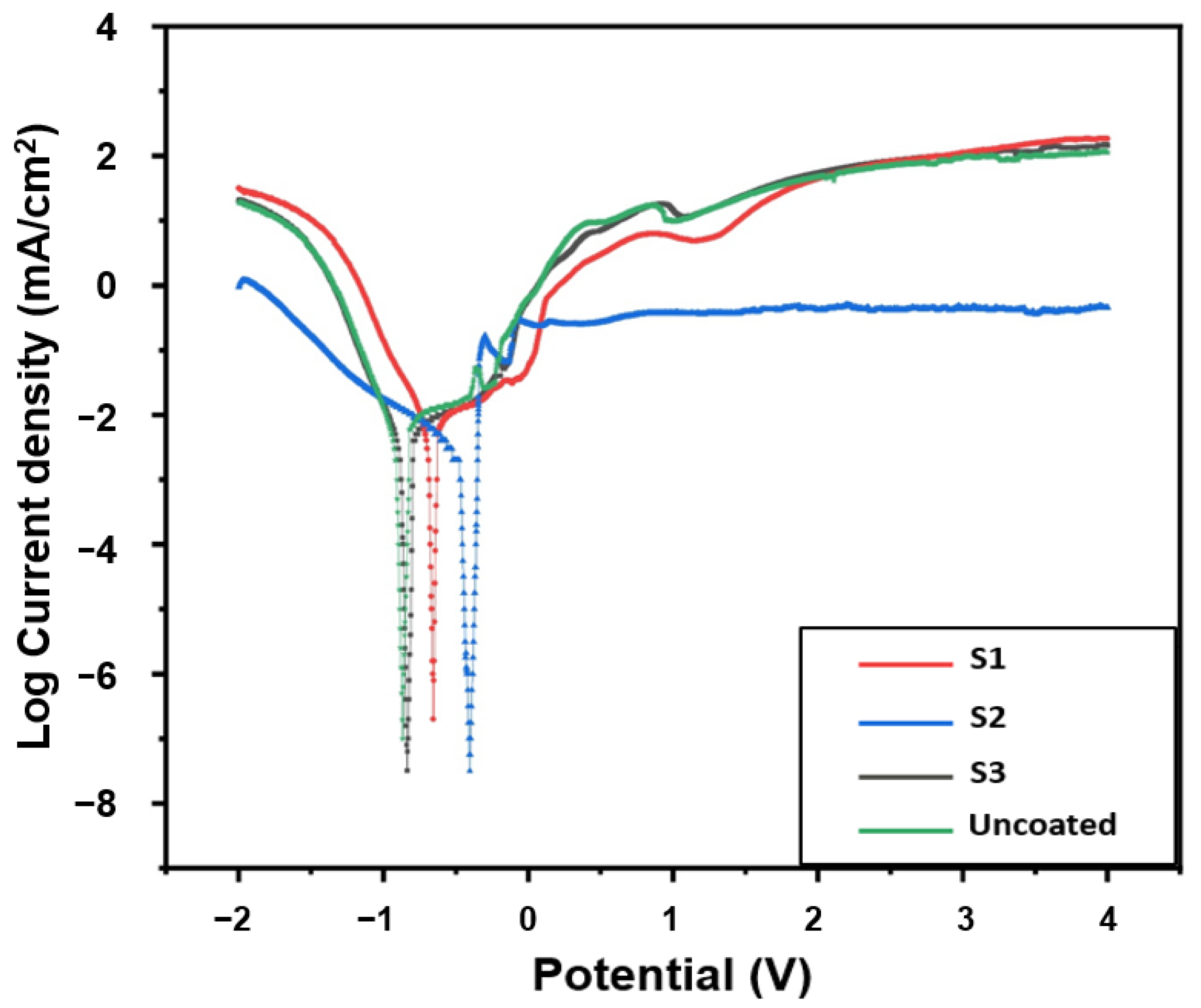
Figure 4 shows the Tofel polarization curves. cathodic (β
c) and anodic (β
a) branches were used to gain the corrosion current density (
Table 5). Low corrosion current density (I
Corr) indicates a slow corrosion rate or excellent corrosion resistance. It can be seen that the cathodic polarization process of the samples is the same. The uncoated samples S1 and S3 show extensive active dissolution behaviour after a small passivation step in the anodic polarization region. However, it can be seen that the S2 sample has a wide deactivation region in the anodic polarization region. The obtained results evince that sample S2 provides better corrosion resistance due to a less negative corrosion potential and lower corrosion current density. The passive layer formation in sample S2 took place more quickly as a consequence of a lower corrosion current density. Additionally, some researchers have pointed out that the good corrosion resistance of Inconel 625 alloy is due to the high content of Mo, Cr, Ni, and Nb, which causes the formation of a stronger passive film on the coating surface [
51,
52,
53]. As can be seen in
Table 6, the content of these elements in sample S2 is higher than other samples, which may be the reason for the better corrosion resistance of this sample than other samples. As can be seen in
Figure 4, the formation of inactive film in sample S2 occurred at a lower current density. The formation of inactive film at lower current density prevents the severe loss of material at higher current density, which also indicates the improvement in corrosion resistance in the S2 sample. Therefore, the performed Inconel 625 coating on sample S2 provides optimal protection against corrosion in the electrolyte solution (3.5% NaCl) at room temperature. Moreover, the measured corrosion current (I
Corr) for sample S2 indicates that the passive layer formed has a better condition compared with other studied samples; hence, better corrosion resistance is expected for sample S2. The Tafel diagram (
Figure 4) shows the relationship between the current generated in an electrochemical cell and the electrode potential of a specific metal. The Tafel curves of S3 and S1 samples display a rapid decrease in their surface corrosion resistance, which might be related to corrosion caused by chloride ions with severe damage to the passive layer [
54]. The corrosion resistance behaviour of the samples improved at the beginning of the corrosion process as a result of the formation of a passive layer of nickel oxide (NiO) [
55,
56]. According to the Tafel diagram of sample S2 (shown in
Figure 4), the passing current decreased more slowly than in other samples, and consequently, the current reached a plateau. The low passing current in sample S2 confirms that the thickness of the protective layer is preserved, and the surface is less corroded. The presented Tafel diagram displayed that the change in potential has led to a sudden change in the current density value for all samples except S2, which verifies the inappropriateness of the passive layer formed on the Inconel 625 coating.
According to the results of the Tafel test, polarization resistance was calculated using the following equation [
57].
In Equation (3), R
p is polarization resistance, β
a is anodic Tafel slope, β
c is cathodic Tafel slope and I
corr is corrosion current. R
p values for different samples are shown in
Table 5. As can be seen, the results are consistent with other corrosion tests. Based on the calculated values, the S2 sample has the most optimal polarization resistance along with low corrosion current.
Figure 5 shows SEM micrographs taken from the surface of all studied samples after the corrosion test. There are two main reasons for the corrosion in the coating: (i) the formation of a non-uniform passive layer on the coating, and (ii) the improper fixation and placement of the Inconel 625 coating on the Inconel 738 substrate.
Figure 5b shows a quite different aspect for sample 2, as compared with the other samples, with very limited and localized corrosion points. This shows that a uniform layer of Inconel 625 coating with the appropriate thickness was placed on sample S2. Such a behaviour can be rationalized by the higher amount of molybdenum found on Inconel 625 laser coating on the S2 sample (
Table 6). The presence of molybdenum ions in the coating surface’s cavities helps to protect the coating against pitting corrosion by forming sodium molybdate, as a result of the reaction of sodium in the polarization solution with molybdenum [
58]. Similar behaviour was observed regarding the anti-corrosion properties of (S235JR) steel with Inconel 625 laser cladding coatings elsewhere [
59,
60].
As can be seen in
Figure 5a,c, pitting and groove corrosion severely damaged the coating surface of samples S1 and S3, respectively. The presence of corrosion products and crevice corrosion caused the surface to become spongy in some places, which was confirmed by the SEM images. A, B, C, and D areas in
Figure 5 were analysed by energy dispersive spectrometry (EDS), and the results are displayed in
Table 6. The molybdenum and chromium elements are critical parameters for corrosion behaviour as ions are absorbed into the formed holes and protect the surface against pitting corrosion. Accordingly, the surface corrosion rate decreased for the S2 sample, as it contains a higher amount of chromium and molybdenum within the electrolyte solution (see
Table 5 and
Table 6). In addition, the amount of these protective coating elements in the uncoated sample is remarkable. The uncoated sample contains a significant amount of Ti element, which improves the hardness of the alloy against wear, but increases the susceptibility of the alloy to corrosion [
61]. The purpose of applying the coating is to increase the corrosion resistance of the alloy by increasing the amount of Mo, Cr, and Nb elements on the surface of the alloy. For this reason, the application of Inconel 625 coating, in addition to maintaining the mechanical strength of the alloy, significantly improves its corrosion resistance. The chromium and molybdenum in sample S3 were oxidized after receiving the highest average laser power, which reduces the corrosion rate (
Table 5 and
Table 6) [
48]. The corrosion process was initiated with the passive layer’s local failure, and the localized corrosive attack can be originated in the Inconel 738 substrate (
Figure 5c). While uniform corrosion can be seen on the surface, pitting corrosion mainly appears as small holes. The amount of material removed underneath the holes is generally unknown because hidden pits can form, making pitting corrosion more difficult to detect and/or predict [
62]. Various forms of corrosion are evident in the microscopic images, which apply to pitting corrosion. The through pits were seen as shallow and elliptical (
Figure 5a), and sideway pits occurred in the subsurface due to the increase in average laser power (
Figure 5c) [
63].
Among the reasons for the spongy spots, the selectiveness of the corrosion site on the coating can be identified, as well as the improper stabilization and placement of the Inconel 625 coating on the Inconel 738 substrate, and the formation of a non-uniform layer on the coating.
The pH of corrosive solution around the Inconel 738 substrate greatly affects the corrosion of the Inconel 625 coating [
59]. The pH of the polarization solution is increased when nickel ions are added to the polarization solution. According to
Figure 6, related to the electrochemical phase diagram for nickel (Pourbaix diagram), nickel oxide is a passive coating-resistant layer at high pH [
64].
The spinel phase (NiCr
2O
4), which prevents oxygen from entering the coating, is not included in the Inconel 625 surface at low pH [
65]; accordingly, the corrosion resistance increased [
66]. The Inconel 738 substrates without Inconel 625 coating are also resistant to corrosion [
67,
68,
69]; however, the coated sample S2 has a lower corrosion rate than the uncoated substrate owing to a proper formation of a passive nickel layer on the surface of S2 sample.
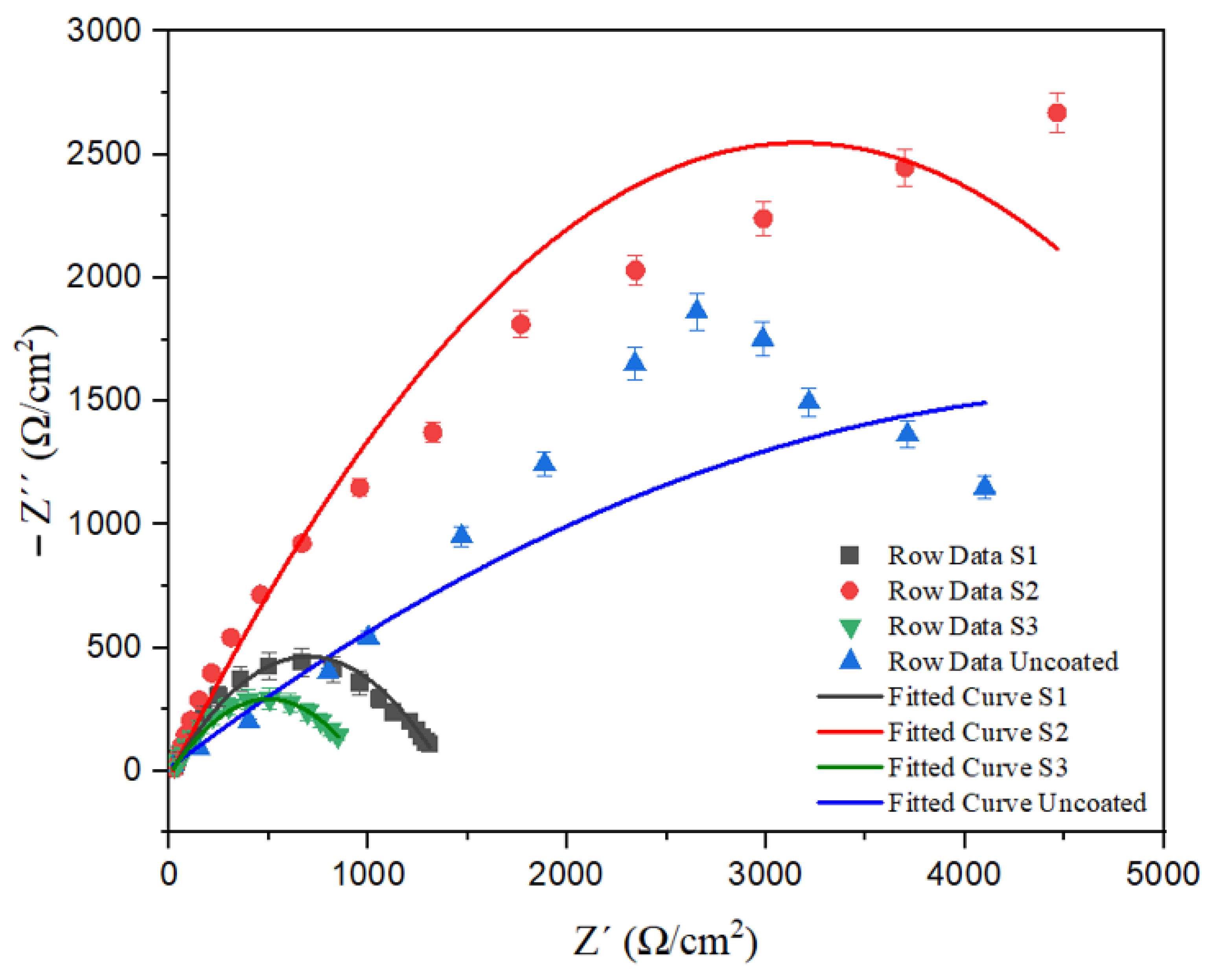
Figure 7 represents the impedance spectra for Nyquist plots by fitting the experimental data to the equivalent circuit model. Based on
Figure 7 curves, capacitive semi-circular loops are similar in shape for samples, although the diameters are different. Increasing the diameter of the Nyquist diagrams shows the high charge transfer resistance (R
ct). In total, a larger diameter in the semicircle in the Nyquist curve means better corrosion resistance. As can be seen, the S2 sample has the highest charge transfer resistance and the highest corrosion resistance. Additionally, if the peak frequency of these semicircles is measured, the amount of corrosion capacitance can be calculated using Equation (4):
Equation (5) is for the circle with a centre of (R
s + R
p/2) Z′ located on the Z′ axis, and its radius is R
p/2. At the circle peak (maximum value |−Z″|), it can be shown that by differentiating from −Z″ respect to Z′ and equating to zero:
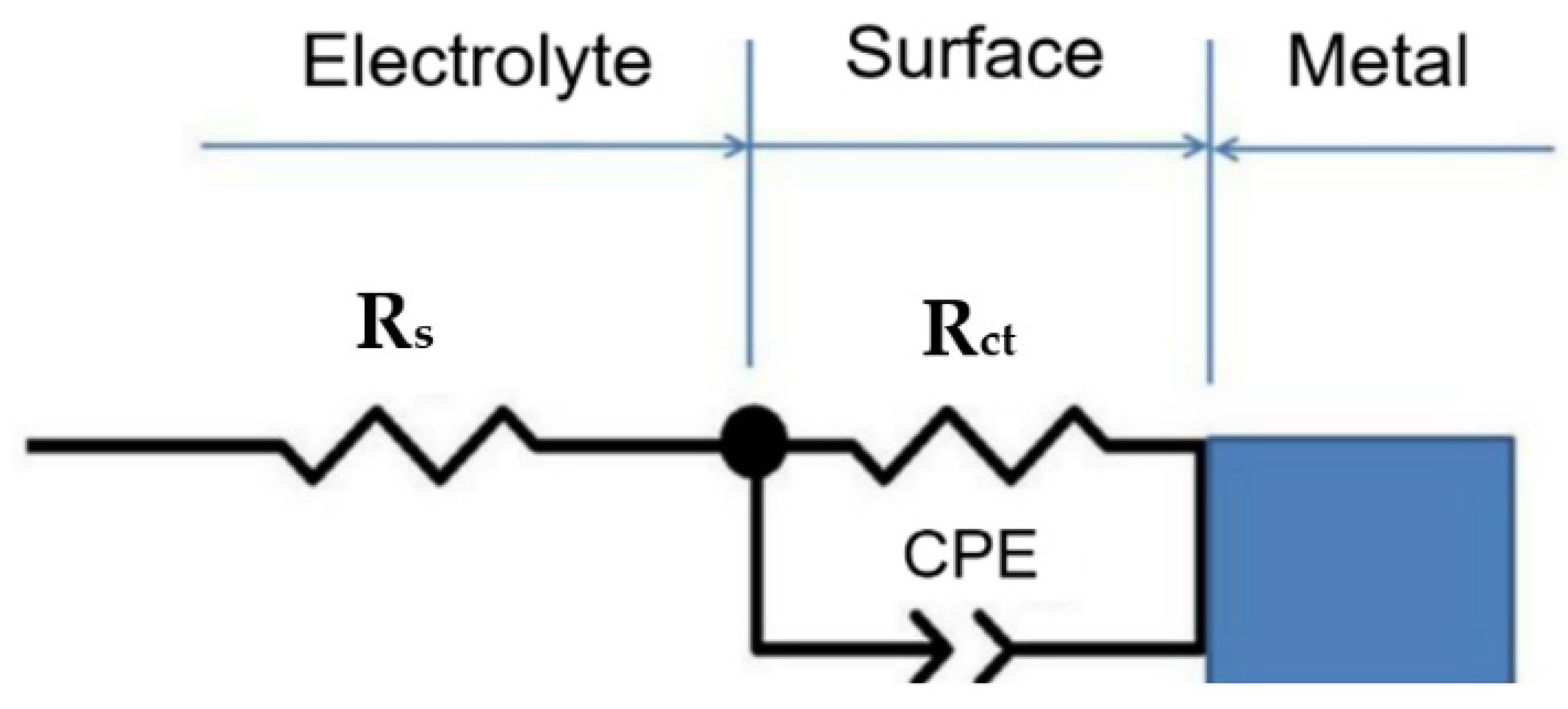
In the equivalent circuit shown in
Figure 8 (as selected in ZView software) and
Table 7, R
s is the solution resistor, R
ct is the charge transfer resistance, and CPE is the constant phase element. According to the parameters of
Table 7, the corrosion resistance increased in S2 sample in the impedance test.
The graph related to the coating of sample S2 has a significant resistance up to the range of 3000 to 3500 Ω/cm
2. However, the coating resistance started to decrease after some time, indicating the beginning of the corrosion process and the formation of holes. The passive layer on the surface created anti-corrosion properties when the cladding method was used for coating [
69]. The graphs related to the Inconel 625 coating of the S1 and S3 samples have assumed a semi-circular state, which indicates a rapid drop in their surface resistance against corrosion. According to previous studies [
40], this decrease in resistance in these samples is due to the development of corrosion by chloride ions and as a result of the severe damage of the passive layer due to the penetration of chloride ions. According to the Nyquist curve, the value of R
ct for the S1 and S3 samples is lower than the uncoated sample, which, according to the EDS analysis results reported in
Table 6, can be attributed to the lower amount of molybdenum and chromium elements in these samples than in the uncoated sample.
Constant phase elements (CPE) are widely used in electrical equivalent circuits to fit experimental impedance data [
70]. CPE behaviour is generally attributed to distributed surface reactivity, surface inhomogeneity, roughness or fractal geometry, electrode porosity, and current/potential distributions associated with electrode geometry [
71]. The coatings of samples S1, S2, and S3 with high-frequency range for total impedance are shown in
Table 7. Sample S2 has passed a lower amount of CPE as the geometry of the etched surface is more regular, and this caused less distribution of stationary-phase elements on the surface (
Figure 5) [
72].
R
ct in the fitted equivalent circuit is related to the charge transfer resistance on the surface of the sample under the impedance test, which indicates the kinetics of charge transfer at the interface between the sample and the corrosive solution and is related to corrosion reactions. The size of the diameter of the semicircle in circuits with one constant phase element indicates the value of R
ct. The higher this value is, the lower the kinetics of the corrosion reaction on the surface of the sample, which, as a result, indicates higher corrosion resistance [
73]. So, the higher this R
ct, the more corrosion-resistant the coating and the lower the corrosion rate. According to
Table 7, sample S2 has the highest value of R
ct, indicating the greater corrosion resistance of this sample; on the opposite side, sample S1 has the lowest value of R
ct, followed by the lowest corrosion resistance. On the other hand, by measuring the diameter of the semicircles in
Figure 5, it is also possible to determine the value of R
ct in the samples. CPE shows the amount of penetration into the passive layer formed on the samples; with the increase in CPE, the amount of defects in the passive layer on the samples increased. The corrosive solution caused the surface corrosion of the samples to occur more easily. As is clear in
Table 7, sample S2 had the lowest amount of CPE and, as a result, the highest amount of corrosion resistance.
Due to the presence of nickel, molybdenum, and niobium—which has different conductivity ratio [
74]—sample S2 did not pass much electrical current, increasing the initial resistance of the solution to almost 3500 Ω·cm
−2. Measuring the diameter of the semicircles in
Figure 5, it can be verified that the increase in CPE has a direct relationship with the number of defects (cavities) in the passive layer on the samples, and the electrolyte solution caused their surface corrosion.