Effect of Reprocessing on Microstructure and Corrosion Resistance of Zr-Sn-Nb Alloy
Abstract
1. Introduction
2. Materials and Methods
2.1. Alloy and Materials
2.2. Microscopic Analysis of Zirconium Matrix
2.3. Corrosion Experiment
2.4. Microscopic Analysis of Oxide Films
3. Results
3.1. Microstructure of Zirconium Matrix
3.2. Corrosion Behavior
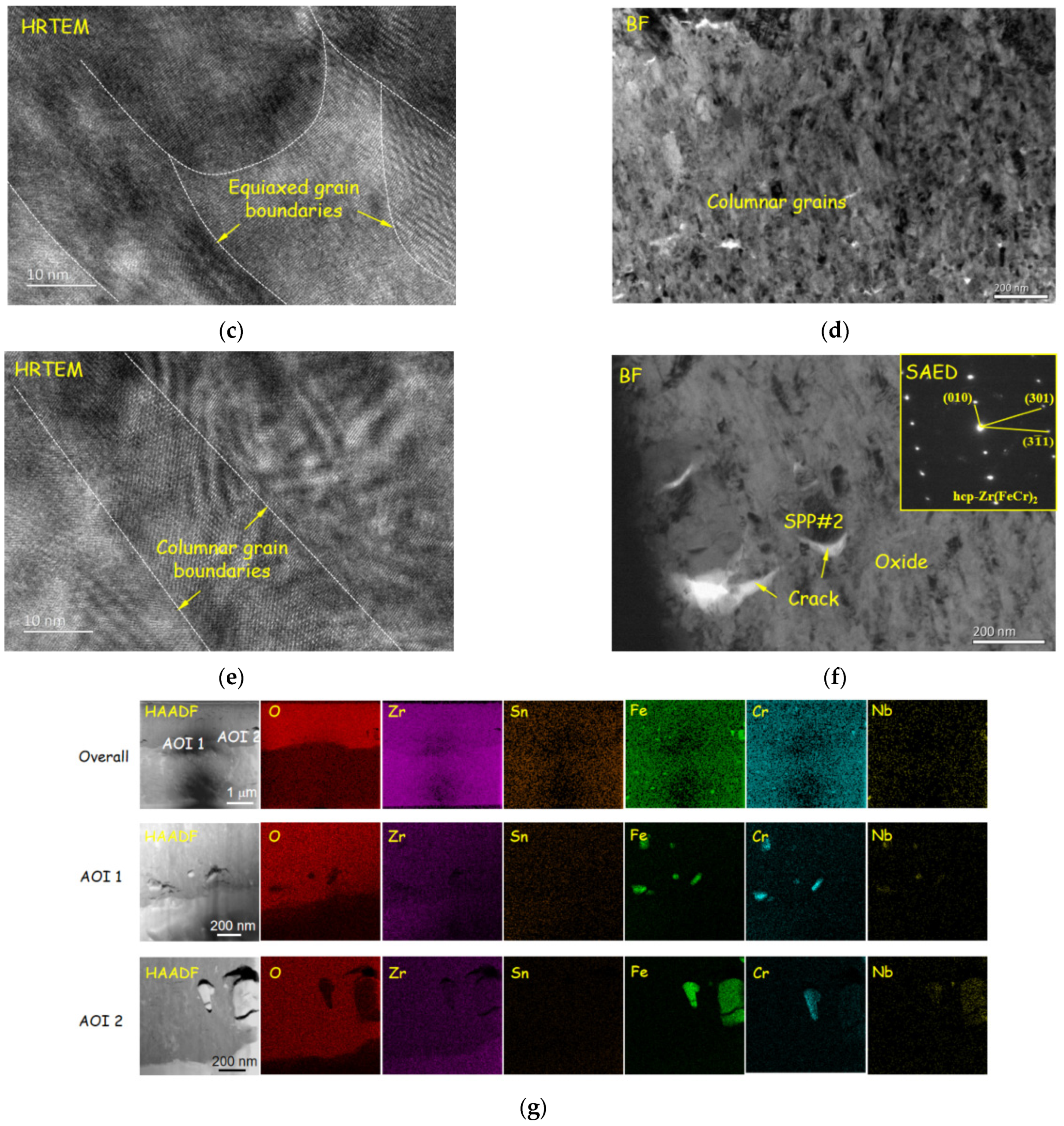
3.3. Microstructure of Oxide Film
4. Discussion
5. Conclusions
- Original plates had a fully recrystallized structure, and the average size of grain was 2.97 μm. Reprocessed plates showed a dynamic-recrystallized structure, which were mainly composed of coarse deformed grains with twins inside and fine equiaxed grain, and the average size of grain was 1.52 μm. Reprocessing might refine the grains.
- Original plates and reprocessed plates both exhibited a <0001>//ND texture. Reprocessing did not changed the texture too much but made the texture more concentrated.
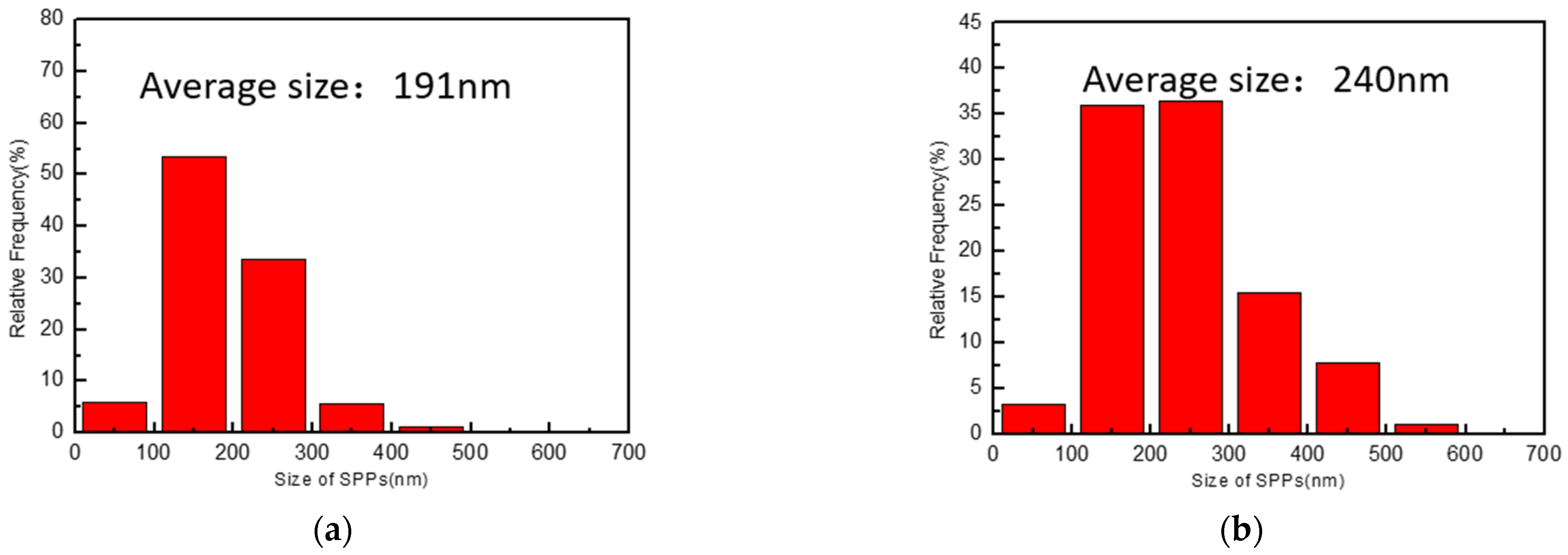
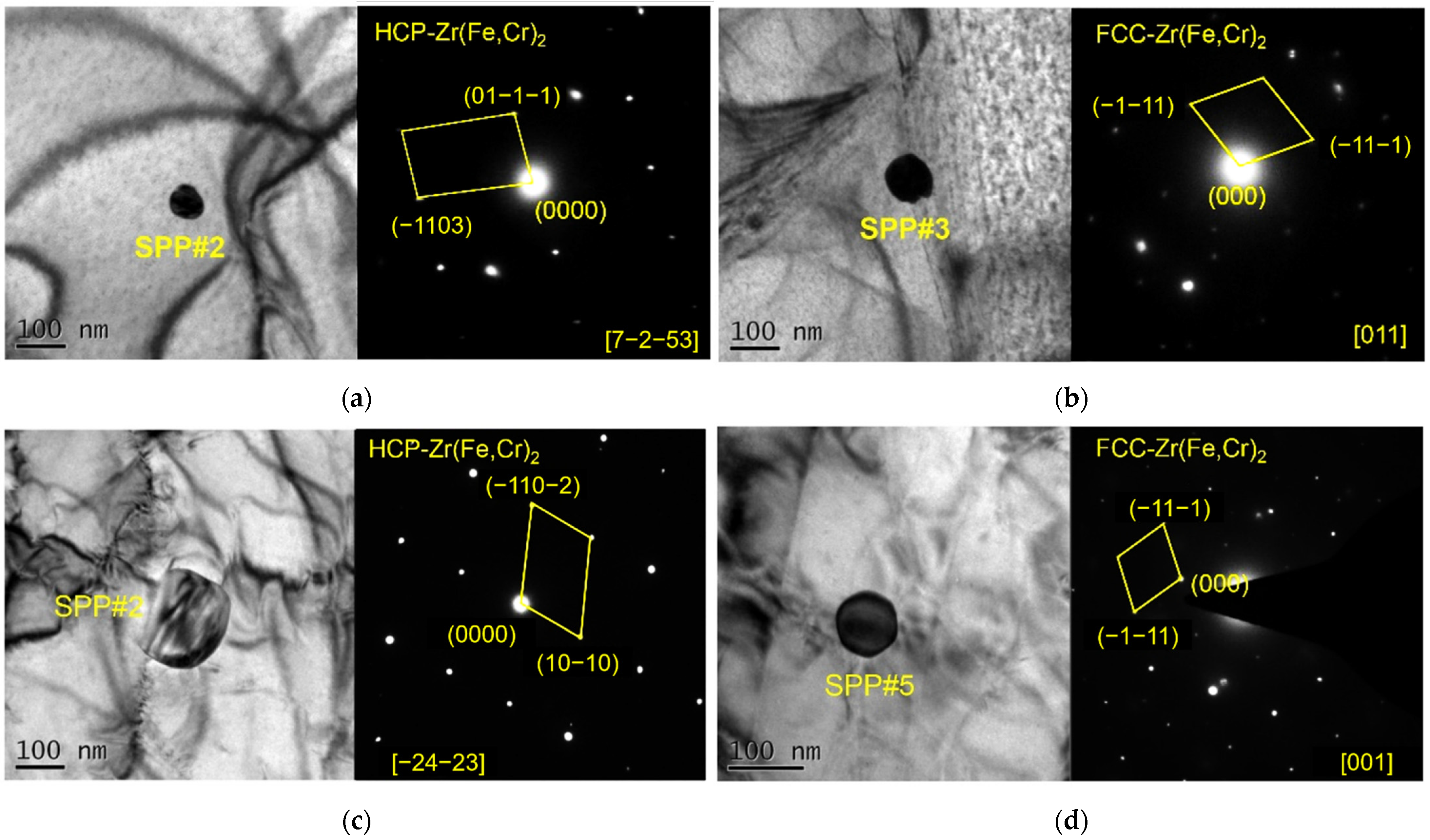
- Original plates and reprocessed plates both showed fine distribution of SPPs. The SPP size of the original plates was 191 nm, while that of reprocessed plates was 240 nm. It seemed that reprocessing coarsened SPPs and changed the structure of SPPs (HCP to FCC).
- Original plates and reprocessed plates both displayed typical corrosion kinetics. Compared with reprocessed plates, the transition of original plates was 40 days later. The corrosion rate of original plates was also slower than that of reprocessed plates. Reprocessing worsened the corrosion resistance of Zr-Sn-Nb alloy.
- Oxide films exhibited a double-layer structure. The inner layer was mainly a columnar crystal structure, and the outer oxide layer was composed of equiaxed crystals. Compared with reprocessed plates, original plates had smaller cracks and lower O/M-interface roughness, which might be the reason for the slower corrosion rate of the original plates.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Motta, A.; Couet, A.; Comstock, R. Corrosion of zirconium alloys used for nuclear fuel cladding. Annu. Rev. Mater. Res. 2015, 45, 311–343. [Google Scholar] [CrossRef]
- Sabol, G.; Comstock, R.; Weiner, R.; Larouere, P.; Stanutz, R. In-reactor corrosion performance of ZIRLO™ and zircaloy-4. In Zirconium in the Nuclear Industry: Tenth International Symposium; ASTM International: Baltimore, MD, USA, 1994; Volume 1, pp. 724–743. [Google Scholar]
- Novikov, V.; Markelov, V.; Tselishchev, A.; Kon’Kov, V.; Sinelnikov, L.; Panchenko, V. Structure-phase changes and corrosion behavior of E110 and E635 claddings of fuels in water cooled reactors. J. Nucl. Sci. Technol. 2006, 43, 991–997. [Google Scholar] [CrossRef]
- Jeong, Y.; Park, S.; Lee, M.; Choi, B.; Baek, J.; Park, J.; Kim, J.; Kim, H. Out-of-pile and in-pile perfomance of advanded zirconium alloys (HANA) for high burn-up fuel. J. Nucl. Sci. Technol. 2006, 43, 977–983. [Google Scholar] [CrossRef]
- Zhou, B.; Yao, M.; Li, Z.; Wang, X.; Zhou, J.; Long, C.; Liu, Q.; Luan, B. Optimization of N18 zirconium alloy for fuel cladding of water reactors. J. Mater. Sci. Technol. 2012, 28, 606–613. [Google Scholar] [CrossRef]
- Aldeen, A.; Chen, Z.; Disher, I.; Zhu, Y.; Yan, K. Growth kinetics of second phase particles in N36 zirconium alloy: Zr-Sn–Nb–Fe. J. Mater. Res. Technol. 2022, 17, 2038–2046. [Google Scholar] [CrossRef]
- Liao, J.; Yang, Z.; Qiu, S.; Peng, Q.; Li, Z.; Zhou, M.; Liu, H. Corrosion of new zirconium claddings in 500 °C/10.3 MPa steam: Effects of alloying and metallography. Acta Metall. Sin. (Engl. Lett.) 2019, 32, 981–994. [Google Scholar] [CrossRef]
- Cox, B. Some thoughts on the mechanisms of in-reactor corrosion of zirconium alloys. J. Nucl. Mater. 2005, 336, 331–368. [Google Scholar] [CrossRef]
- Ni, N. Study of Oxidation Mechanisms of Zirconium Alloys by Electron Microscopy. Ph.D. Thesis, Oxford University, Oxford, UK, 2011. [Google Scholar]
- Ahmed, T.; Keys, L. The breakaway oxidation of zirconium and its alloys: A review. J. Less-Common Met. 1975, 39, 99–107. [Google Scholar] [CrossRef]
- Park, D.; Park, J.; Jeong, Y.; Lee, J. Microstructural characterization of ZrO2 layers formed during the transition to breakaway oxidation. J. Nucl. Mater. 2010, 399, 208–211. [Google Scholar] [CrossRef]
- Dawson, J.; Baugh, U.; White, J. Observations on the Early Stages of Oxidation of Zirconium and Zircaloy-2. Electrochem. Technol. (US) Absorbed J. Electrochem. Soc. 1966, 4, 137. [Google Scholar]
- Tupin, M.; Pijolat, M.; Valdivieso, F.; Soustelle, M. Differences in reactivity of oxide growth during the oxidation of Zircaloy-4 in water vapour before and after the kinetic transition. J. Nucl. Mater. 2003, 317, 130–144. [Google Scholar] [CrossRef]
- Wei, J.; Frankel, P.; Polatidis, E.; Blat, M.; Ambard, A.; Comstock, R.; Hallstadius, L.; Hudson, D.; Smith, G.; Grovenor, C.; et al. The effect of Sn on autoclave corrosion performance and corrosion mechanisms in Zr-Sn–Nb alloys. Acta Mater. 2013, 61, 4200–4214. [Google Scholar] [CrossRef]
- Jeong, Y.; Kim, H.; Kim, D.; Choi, B.; Kim, J. Influence of Nb concentration in the α-matrix on the corrosion behavior of Zr-xNb binary alloys. J. Nucl. Mater. 2003, 323, 72–80. [Google Scholar] [CrossRef]
- Kim, H.; Park, J.; Jeong, Y. Ex-reactor corrosion and oxide characteristics of Zr–Nb–Fe alloys with the Nb/Fe ratio. J. Nucl. Mater. 2005, 345, 1–10. [Google Scholar] [CrossRef]
- Hudson, D.; Smith, G. Initial observation of grain boundary solute segregation in a zirconium alloy (ZIRLO) by the three dimensional atom probe. Scr. Mater. 2009, 61, 411–414. [Google Scholar] [CrossRef]
- Yang, Z.; Zhao, W. Review of corrosion and oxide characterization for Zr alloys. Mater. Rev. 2010, 24, 120–125. (In Chinese) [Google Scholar]
- Sundell, G.; Thuvander, M.; Andren, H. Barrier oxide chemistry and hydrogen pick-up mechanisms in zirconium alloys. Corros. Sci. 2016, 102, 490–502. [Google Scholar] [CrossRef]
- Yardley, S.; Moore, K.; Ni, N.; Wei, J.; Lyon, S.; Preuss, M.; Lozano-Perez, S.; Grovenor, C. An investigation of the oxidation behaviour of zirconium alloys using isotopic tracers and high resolution SIMS. J. Nucl. Mater. 2013, 443, 436–443. [Google Scholar] [CrossRef]
- Pêcheur, D. Oxidation of β-Nb and Zr (Fe,V)2 precipitates in oxide films formed on advanced Zr-based alloys. J. Nucl. Mater. 2000, 278, 195–201. [Google Scholar] [CrossRef]
- Motta, A.; Yilmazbayhan, A.; da Silva, M.; Comstock, R.; Was, G.; Busby, J.; Gartner, E.; Peng, Q.; Jeong, Y.; Park, J. Zirconium alloys for supercritical water reactor applications: Challenges and possibilities. J. Nucl. Mater. 2007, 371, 61–75. [Google Scholar] [CrossRef]
- Fan, Q.; Yang, Z.; Zhou, J.; Shi, M.; Chen, X.; Li, Z. Research progress on second phase particles on Zr-Sn-Nb-Fe zirconim alloy. J. Mater. Eng. 2016, 44, 110–118. (In Chinese) [Google Scholar]
- Liu, H.; Dai, X.; Wang, Y.; Zhao, W. EBSD analysis of the microstructure for reprocessed N18 alloy plates. J. Chin. Electron Microsc. Soc. 2010, 39, 437–441. (In Chinese) [Google Scholar]
- Chen, B.; Qiu, S.; Peng, Q.; Dai, X.; Wang, P.; Liu, H.; Wei, T. Effect of Re-Working on Secondary Phases in Modified N18 Sheet. Nucl. Power Eng. 2018, 39, 32–36. (In Chinese) [Google Scholar]
- Murty, K.; Charit, I. Texture development and anisotropic deformation of zircaloys. Prog. Nucl. Energy 2006, 48, 325–359. [Google Scholar] [CrossRef]
- Park, J.; Jeong, Y.; Jung, Y. Effects of precipitation characteristics on the out-of-pile corrosion behavior of niobium-containing zirconium alloys. Met. Mater. Int. 2001, 7, 447–455. [Google Scholar] [CrossRef]
- Voorhees, P. The theory of Ostwald ripening. J. Stat. Phys. J. Stat. Phys. 1985, 38, 231–252. [Google Scholar] [CrossRef]
- Chemelle, P.; Knorr, D.; Van Der Sande, J.; Pelloux, R. Morphology and composition of second phase particles in Zircaloy-2. J. Nucl. Mater. 1983, 113, 58–64. [Google Scholar] [CrossRef]
- Arias, D.; Palacios, T.; Turrillo, C. Composition of precipitates present in Zircaloy-2 and 4. J. Nucl. Mater. 1987, 148, 227–229. [Google Scholar] [CrossRef]
- Shen, Y.; Paasche, O. On the Transformation of ZrCr2; Wah Chang Corp.: Albany, OR, USA, 1968. [Google Scholar]
- Gosmain, L.; Valot, C.; Ciosmak, D.; Sicardy, O. Study of stress effects in the oxidation of Zircaloy-4. Solid State Ion. 2001, 141, 633–640. [Google Scholar] [CrossRef]
- Couet, A.; Motta, A.; Ambard, A. The coupled current charge compensation model for zirconium alloy fuel cladding oxidation: I. Parabolic oxidation of zirconium alloys. Corros. Sci. 2015, 100, 73–84. [Google Scholar] [CrossRef]
- Couet, A.; Motta, A.; Ambard, A.; Livigni, D. In-situ electrochemical impedance spectroscopy measurements of zirconium alloy oxide conductivity: Relationship to hydrogen pickup. Corros. Sci. 2017, 119, 1–13. [Google Scholar] [CrossRef]
- Wei, J.; Frankel, P.; Blat, M.; Ambard, A.; Comstock, R.; Hallstadius, L.; Lyon, S.; Cottis, R.; Preuss, M. Autoclave study of zirconium alloys with and without hydride rim. Corros. Eng. Sci. Technol. 2012, 47, 516–528. [Google Scholar] [CrossRef]
- Yilmazbayhan, A.; Motta, A.; Comstock, R.; Sabol, G.; Lai, B.; Cai, Z. Structure of zirconium alloy oxides formed in pure water studied with synchrotron radiation and optical microscopy: Relation to corrosion rate. J. Nucl. Mater. 2004, 324, 6–22. [Google Scholar] [CrossRef]
- Garner, A.; Gholinia, A.; Frankel, P.; Gass, M.; Maclaren, I.; Preuss, M. The microstructure and microtexture of zirconium oxide films studied by transmission electron backscatter diffraction and automated crystal orientation mapping with transmission electron microscopy. Acta Mater. 2014, 80, 159–171. [Google Scholar] [CrossRef]
- Vermaak, N.; Parry, G.; Estevez, R.; Bréchet, Y. New insight into crack formation during corrosion of zirconium-based metal-oxide systems. Acta Mater. 2013, 61, 4374–4383. [Google Scholar] [CrossRef]
- Liao, J.; Yang, Z.; Qiu, S.; Peng, Q.; Li, Z.; Zhang, J. The correlation between tetragonal phase and the undulated metal/oxide interface in the oxide films of zirconium alloys. J. Nucl. Mater. 2019, 524, 101–110. [Google Scholar] [CrossRef]
- Ni, N.; Lozano-Perez, S.; Sykes, J.; Grovenor, C. Transmission EELS quantification study of oxygen content at zirconium alloy metal/oxide interface. Microsc. Microanal. 2010, 16, 1614–1615. [Google Scholar] [CrossRef]
- Skovgaard, M.; Ahniyaz, A.; Sørensen, B.; Almdal, K.; Van Lelieveld, A. Effect of microscale shear stresses on the martensitic phase transformation of nanocrystalline tetragonal zirconia powders. J. Eur. Ceram. Soc. 2010, 30, 2749–2755. [Google Scholar] [CrossRef]
- Zhang, X. Understanding the corrosion resistance of nanocrystalline materials: The influence of grain size. In Corrosion Protection and Control Using Nanomaterials; Woodhead Publishing: Sawston, UK, 2012. [Google Scholar]
- Ni, N.; Lozano-Perez, S.; Sykes, J.; Smith, G.; Grovenor, C. Focussed ion beam sectioning for the 3D characterisation of cracking in oxide scales formed on commercial ZIRLO™ alloys during corrosion in high temperature pressurised water. Corros. Sci. 2011, 53, 4073–4083. [Google Scholar] [CrossRef]
- Bossis, P.; Thomazet, J.; Lefebvre, F. Study of the mechanisms controlling the oxide growth under irradiation: Characterization of irradiated Zircaloy-4 and Zr-1Nb-O oxide scales. In Zirconium in the Nuclear Industry: Thirteenth International Symposium; ASTM International: Annecy, France, 2002; Volume 1, pp. 10–14. [Google Scholar]








| Element | Sn | Nb | Fe | Cr | Zr |
|---|---|---|---|---|---|
| Content | 0.8–1.2 | 0.25–0.35 | 0.3–0.4 | 0.05–0.10 | Bal. |
| Materials | Size/nm | Proportion of SPPs with a Size Greater than 200 nm/% |
|---|---|---|
| Original plates | 191 | 42.2 |
| Reprocessed plates | 240 | 62.1 |
| Materials | Pre-Transition | Linear Period | Transition Time (days) | Maximum ΔW (mg/dm2) | ||
|---|---|---|---|---|---|---|
| k1 | n | k2 | R2 | |||
| Original plates | 6.08 | 0.330 | 0.21 | 0.9825 | 210 | 125.37 ± 8.34 |
| Reprocessed plates | 6.76 | 0.335 | 0.26 | 0.9911 | 170 | 154.39 ± 10.26 |
| Zr-4 | 5.68 | 0.360 | 0.37 | 0.9351 | 170 | 240.85 ± 12.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Jia, Y.; Dai, X.; Yi, W. Effect of Reprocessing on Microstructure and Corrosion Resistance of Zr-Sn-Nb Alloy. Metals 2022, 12, 1822. https://doi.org/10.3390/met12111822
Wu Z, Jia Y, Dai X, Yi W. Effect of Reprocessing on Microstructure and Corrosion Resistance of Zr-Sn-Nb Alloy. Metals. 2022; 12(11):1822. https://doi.org/10.3390/met12111822
Chicago/Turabian StyleWu, Zongpei, Yuzhen Jia, Xun Dai, and Wei Yi. 2022. "Effect of Reprocessing on Microstructure and Corrosion Resistance of Zr-Sn-Nb Alloy" Metals 12, no. 11: 1822. https://doi.org/10.3390/met12111822
APA StyleWu, Z., Jia, Y., Dai, X., & Yi, W. (2022). Effect of Reprocessing on Microstructure and Corrosion Resistance of Zr-Sn-Nb Alloy. Metals, 12(11), 1822. https://doi.org/10.3390/met12111822




