Oxidation and Corrosion Resistance of NiCr-Re and NiCr-Re-Al2O3 Materials Fabricated by Spark Plasma Sintering
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Preparation
2.2. Thermal Oxidation Tests
2.3. Electrochemical Corrosion Tests
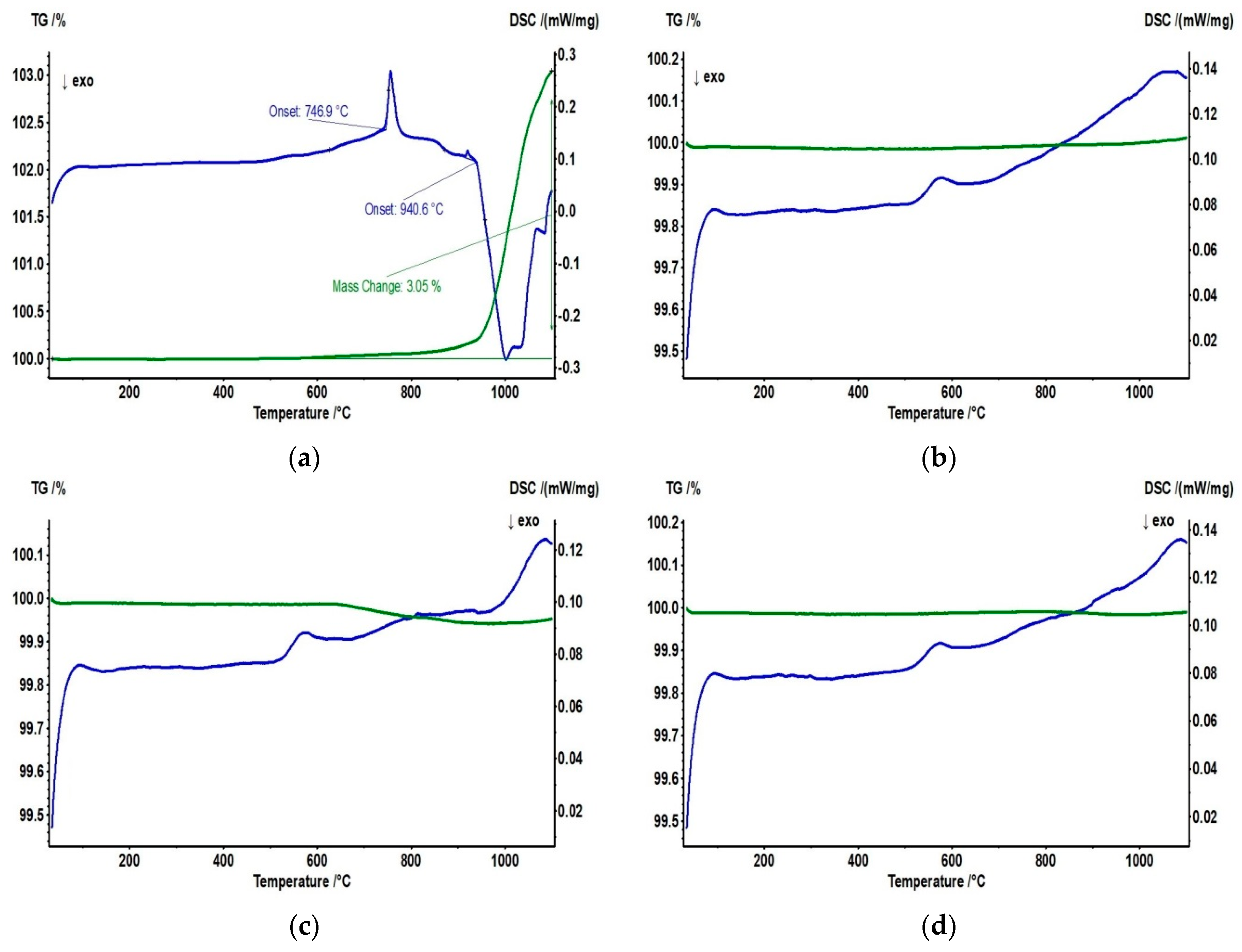
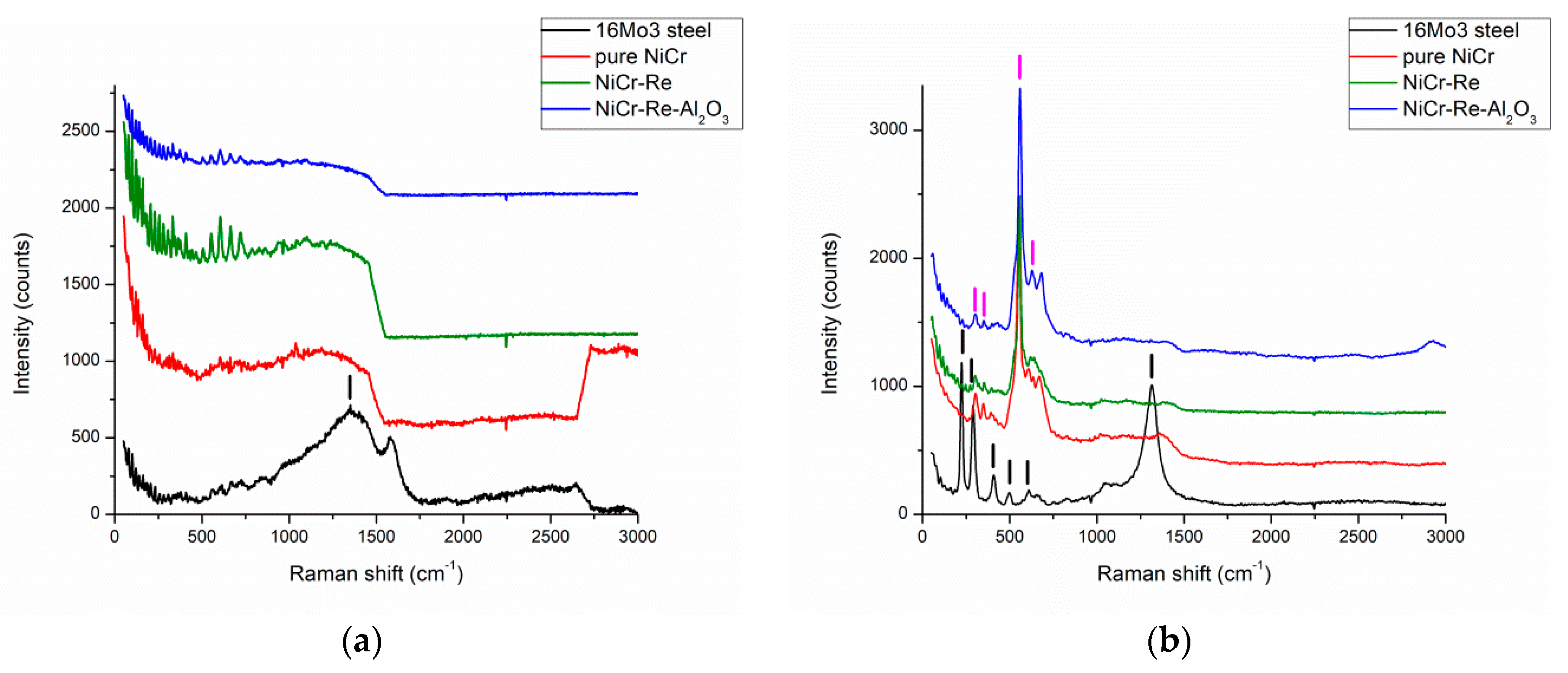
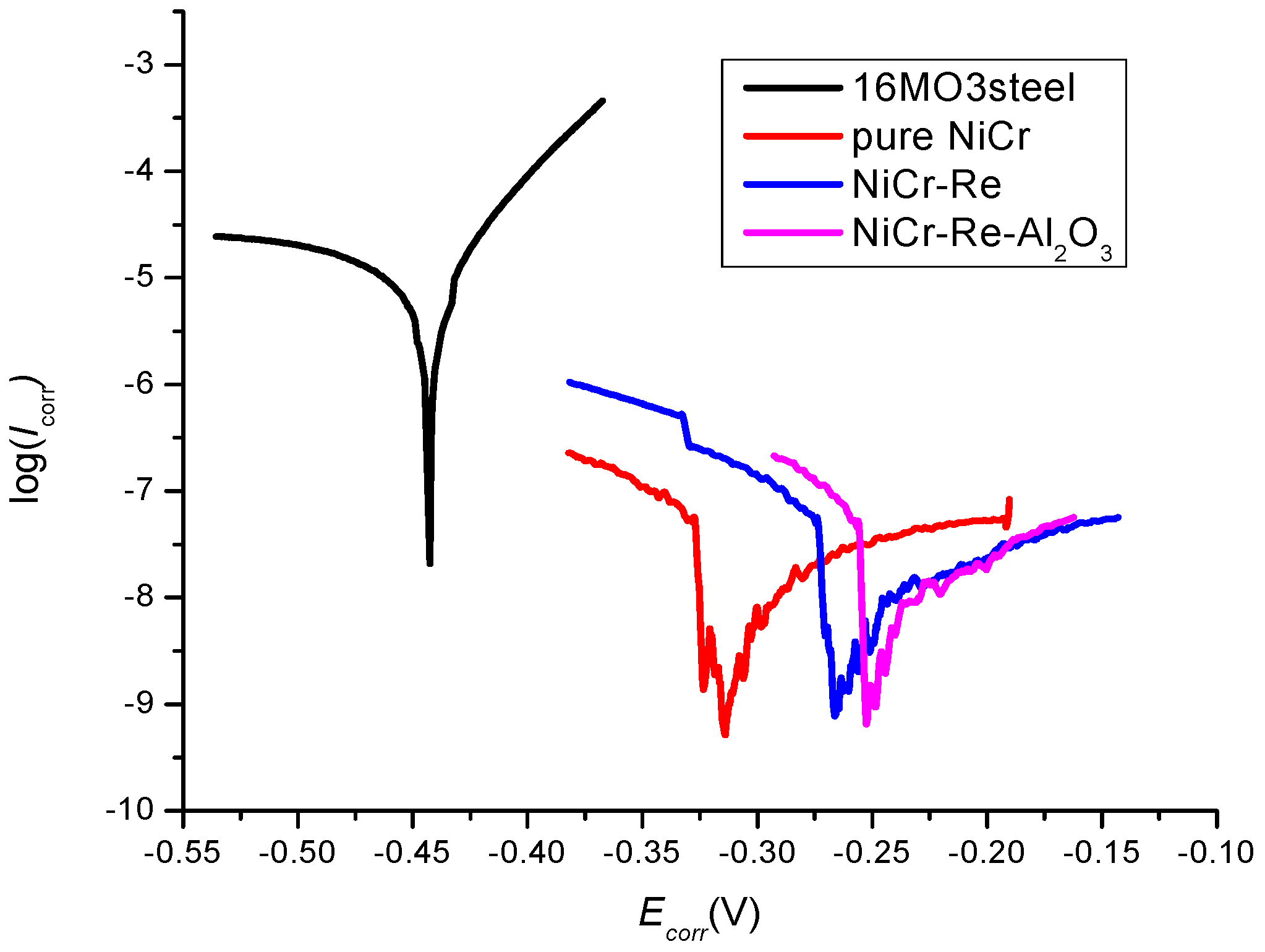
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ghosh, D.; Mitra, S.K. High temperature corrosion problem of boiler components in presence of sulfur and alkali based fuels. High Temp. Mater. Proc. 2011, 30, 81–85. [Google Scholar] [CrossRef]
- Azamata, O.D.; Jibatswen, T.Y.; Michael, O.C. Effect of chlorine and sulphur on stainless steel (AISI 310) due to high temperature corrosion. Am. J. Eng. Res. 2016, 5, 266–270. [Google Scholar]
- Nagarajan, R.; Ambedkar, B.; Gowrisankar, S.; Somasundaram, S. Development of predictive model for fly-ash erosion phenomena in coal-burning boilers. Wear 2009, 267, 122–128. [Google Scholar] [CrossRef]
- Morks, M.F.; Berndt, C.C. Corrosion and oxidation properties of NiCr coatings sprayed in presence of gas shroud system. Appl. Surf. Sci. 2010, 256, 4322–4327. [Google Scholar] [CrossRef]
- Oksa, M.; Metsajoki, J. Optimizing NiCr and FeCr HVOF coating structures for high-temperature corrosion protection applications. J. Therm. Spray Technol. 2015, 24, 436–453. [Google Scholar] [CrossRef]
- Lee, B.J.; Shin, P.K. Fabrication and characterization of Ni-Cr alloy thin films for application to precision thin film resistors. J. Electr. Eng. Technol. 2007, 2, 525–531. [Google Scholar] [CrossRef]
- Yang, Z.; Xia, G.G.; Stevenson, J.W. Evaluation of Ni–Cr-base alloys for SOFC interconnect applications. J. Power Sources 2006, 160, 1104–1110. [Google Scholar] [CrossRef]
- Hamatani, H.; Shimoda, N.; Kitaguchi, S. Effect of the composition profile and density of LPPS sprayed functionally graded coating on the thermal shock resistance. Sci. Technol. Adv. Mat. 2003, 4, 197–203. [Google Scholar] [CrossRef]
- Uusitalo, M.A.; Vuoristo, P.M.J.; Mantyla, T.A. High-temperature corrosion of coatings and boiler chlorine-containing atmosphere. Surf. Coat. Technol. 2002, 161, 275–285. [Google Scholar] [CrossRef]
- Matthews, S.; James, B.; Hyland, M. High-temperature erosion of Cr3C2-NiCr thermal spray coatings—The role of phase microstructure. Surf. Coat. Technol. 2009, 203, 1144–1153. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Sun, X.F.; Guan, H.R.; Hu, Z.Q. 1050 °C isothermal oxidation behavior of detonation gun sprayed NiCrAlY coating. Surf. Coat. Technol. 2002, 161, 302–305. [Google Scholar] [CrossRef]
- Omoniyi, F.; Olubambi, P.; Sadiku, E. High-temperature oxidation resistance of Ni22Cr11Al bond coat produced by spark plasma sintering as thermal barrier coatings. J. Mater. Sci. Eng. 2016, 5, 1000236. [Google Scholar]
- Chmielewski, T.; Siwek, P.; Chmielewski, M.; Piątkowska, A.; Grabias, A.; Golański, D. Structure and selected properties of arc sprayed coatings containing in-situ fabricated Fe-Al intermetallic phases. Metals 2018, 8, 1059. [Google Scholar] [CrossRef]
- Sartowska, B.; Piekoszewski, J.; Waliś, L.; Senatorski, J.; Stanisławski, J.; Ratajczak, R.; Nowicki, L.; Kopcewicz, M.; Prokert, F.; Barlak, M. Structural and tribological properties of carbon steels modified by plasma pulses containing inert and active ions. Surf. Coat. Technol. 2007, 201, 8295–8298. [Google Scholar] [CrossRef]
- Plasma System. Available online: https://www.plasmasystem.pl/en/shells/highresist (accessed on 24 July 2020).
- Huang, L.; Sun, X.F.; Guan, H.R.; Hu, Z.Q. Effect of rhenium addition on isothermal oxidation behavior of single-crystal Ni-based superalloy. Surf. Coat. Technol. 2006, 200, 6863–6870. [Google Scholar] [CrossRef]
- Wei, W.; Geng, S.; Xie, D.; Wang, F. High temperature oxidation and corrosion behaviours of Ni-Fe-Cr alloys as inert anode for aluminum electrolysis. Corros. Sci. 2019, 157, 382–391. [Google Scholar] [CrossRef]
- Jiang, C.; Xie, Y.; Kong, C.; Zhang, J.; Young, D.J. Corrosion behaviour of Ni-Cr alloys in mixed oxidising gases at 650 °C. Corros. Sci. 2020, 174, 108801. [Google Scholar] [CrossRef]
- Xing, X.; Li, Z.X.; Li, M.; Zhou, C. Comparison of the corrosion resistance of Ni2Al3 coating with and without Ni-Re interlayer in dry and wet CO2 gas. Corros. Sci. 2019, 159, 108121. [Google Scholar]
- Ai, H.; Yea, X.X.; Jiang, L.; Lenga, B.; Shen, M.; Li, Z.; Jia, Y.; Wang, J.Q.; Zhou, X.; Xie, Y.; et al. On the possibility of severe corrosion of a Ni-W-Cr alloy influoride moltensalts at high temperature. Corros. Sci. 2019, 149, 218–225. [Google Scholar] [CrossRef]
- Chmielewski, M.; Pietrzak, K.; Basista, M.; Weglewski, W. Rhenium doped chromium–alumina composites for high-temperature applications. Int. J. Refract. Met. Hard Mater. 2016, 54, 196–202. [Google Scholar] [CrossRef]
- Petrovich, V.; Haurylau, M.; Volchek, S. Rhenium deposition on silicon surface at the room temperature for application in microsystems. Sens. Actuator 2002, 99, 45–48. [Google Scholar] [CrossRef]
- Kojima, R.; Enomoto, H.; Muhler, M.; Aika, K. Cesium-promoted rhenium catalysts supported on alumina for ammonia synthesis. Appl. Catal. A Gen. 2003, 246, 311–322. [Google Scholar] [CrossRef]
- Roger, J.; Audubert, F.; Petitcorps, Y.L. Thermal reactions of SiC films with Mo, Re and Mo-Re alloys. J. Alloy Compd. 2009, 475, 635–642. [Google Scholar] [CrossRef]
- Kindrachuk, V.; Wanderka, N.; Banhart, J.; Mukherji, D.; Del Genovese, D.; Rösler, J. Effect of rhenium addition on the microstructure of the superalloy Inconel 706. Acta Mater. 2008, 56, 1609–1618. [Google Scholar] [CrossRef]
- Brynk, T.; Pakiela, Z.; Ludwichowska, K.; Romelczyk, B.; Molak, R.M.; Plocinska, M.; Kurzac, J.; Kurzynowski, T.; Chlebus, E. Fatigue crack growth rate and tensile strength of Re modified Inconel 718 produced by means of selective laser melting. Mat. Sci. Eng. A Struct. 2017, 698, 289–301. [Google Scholar] [CrossRef]
- Yoon, K.E.; Noebe, R.D.; Seidman, D.N. Effects of rhenium addition on the temporal evolution of the nanostructure and chemistry of a model Ni-Cr-Al superalloy. I: Experimental observations. Acta Mater. 2007, 55, 1145–1157. [Google Scholar] [CrossRef]
- Yoon, K.E.; Noebe, R.D.; Seidman, D.N. Effects of rhenium addition on the temporal evolution of the nanostructure and chemistry of a model Ni–Cr–Al superalloy. II: Analysis of the coarsening behaviour. Acta Mater. 2007, 55, 1159–1169. [Google Scholar] [CrossRef]
- Bochenek, K.; Węglewski, W.; Morgiel, J.; Maj, M.; Basista, M. Enhancement of fracture toughness of hot-pressed NiAl-Re material by aluminum oxide addition. Mat. Sci. Eng. A Struct. 2020, 790, 139670-1-6. [Google Scholar] [CrossRef]
- Kruszewski, M.J.; Zybała, R.; Ciupinski, Ł.; Chmielewski, M.; Adamczyk-Cieślak, B.; Michalski, A.; Rajska, M.; Kurzydłowski, K.J. Microstructure and thermoelectric properties of bulk cobalt antimonide (CoSb3) skutterudites obtained by pulse plasma sintering. J. Electron. Mater. 2016, 45, 1369–1376. [Google Scholar] [CrossRef]
- Orru, R.; Licheri, R.; Locci, A.M.; Cincotti, A.; Cao, G. Consolidation/synthesis of materials by electric current activated/assisted sintering. Mat. Sci. Eng. R. 2009, 63, 127–287. [Google Scholar] [CrossRef]
- Chmielewski, M.; Pietrzak, K.; Teodorczyk, M.; Nosewicz, S.; Jarząbek, D.; Zybała, R.; Bazarnik, P.; Lewandowska, M.; Strojny-Nędza, A. Effect of metallic coating on the properties of copper-silicon carbide composites. Appl. Surf. Sci. 2017, 421A, 159–169. [Google Scholar] [CrossRef]
- Caron, P.; Khan, T. Evolution of Ni-based superalloys for single crystal gas turbine blade applications. Aerosp. Sci. Technol. 1999, 3, 513–523. [Google Scholar] [CrossRef]
- Osório, W.R.; Peixoto, L.C.; Canté, M.V.; Garcia, A. Electrochemical corrosion characterization of Al-Ni alloys in a dilute sodium chloride solution. Electrochim. Acta 2010, 55, 4078–4085. [Google Scholar] [CrossRef]
- Sun, Y.; Li, C.; Yu, L.; Gao, Z.; Xia, X.; Liu, Y. Corrosion behavior of Al-15%Mg2Si alloy with 1% Ni addition. Results Phys. 2020, 17, 103129. [Google Scholar] [CrossRef]
- Deo, Y.; Guha, S.; Sarkar, K.; Mohanta, P.; Pradhan, D.; Mondal, A. Electrodeposited Ni-Cu alloy coatings on mild steel for enhanced corrosion properties. Appl. Surf. Sci. 2020, 515, 146078. [Google Scholar] [CrossRef]
- Liu, S.; Xia, C.; Yang, T.; Yang, Z.; Liu, N.; Li, Q. High strength and superior corrosion resistance of the Ti-Ni-Cu-Zr crystal/glassy alloys with superelasticity. Mater. Lett. 2020, 260, 126938. [Google Scholar] [CrossRef]
- Li, Y.; Wei, S.; Cheng, X.; Zhang, T.; Cheng, G. Corrosion behavior and surface characterization of tantalum implanted TiNi alloy. Surf. Coat. Technol. 2008, 202, 3017–3022. [Google Scholar] [CrossRef]
- Cheng, Y.; Zheng, Y.F. The corrosion behavior and hemocompatibility of TiNi alloys coated with DLC by plasma based ion implantation. Surf. Coat. Technol. 2006, 200, 4543–4548. [Google Scholar] [CrossRef]
- Fu, Y.; Wu, X.; Wang, Y.; Li, B.; Yang, S.Z. Study of corrosion resistance property and microstructure of TiNi shape memory alloy modified by pulsed high-energy density plasma. Appl. Surf. Sci. 2000, 157, 167–177. [Google Scholar] [CrossRef]
- Kanyane, L.R.; Adesina, O.S.; Popoola, A.P.I.; Farotade, G.A.; Malatji, N. Microstructural evolution and corrosion properties of laser clad TiNi on titanium alloy (Ti6Al4V). Procedia Manuf. 2019, 35, 1267–1272. [Google Scholar] [CrossRef]
- Chmielewski, M.; Pietrzak, K. Processing, microstructure and mechanical properties of Al2O3-Cr nanocomposites. J. Eur. Ceram. Soc. 2007, 27, 1273–1279. [Google Scholar] [CrossRef]





| Material Composition | Theoretical Density (g/cm3) | Measured Density (g/cm3) | Relative Density (%) |
|---|---|---|---|
| NiCr | 8.50 | 8.42 | 99.1 ± 0.2 |
| NiCr-Re | 8.62 | 8.52 | 98.8 ± 0.3 |
| NiCrRe-Al2O3 | 8.16 | 8.08 | 99.0 ± 0.2 |
| Material | Ecorr (V) | jcorr (μA/cm2) | Reference |
|---|---|---|---|
| 16Mo3 steel | −0.410 | 1.35 | Reference material |
| pure NiCr | −0.296 | 0.0792 | This work |
| NiCr-Re | −0.279 | 0.0455 | This work |
| NiCr-Re-Al2O3 | −0.253 | 0.0134 | This work |
| Al-15%Mg2Si-1%Ni | −0.819 | 0.407 | [35] |
| Ni-Cu alloy | −0.404 | 22.9 | [36] |
| Ti38Ni38Cu12Zr12 | −0.285 | 0.289 | [37] |
| Ti42Ni42Cu8Zr8 | −0.271 | 0.147 | [37] |
| TiNi | −0.288 | 0.087 | [38] |
| Diamond-like carbon/TiNi | −0.088 | 0.02 | [39] |
| Plasma treated TiNi | −0.052 | 0.013 | [40] |
| Ti-10Ni1.2 | −0.076 | 1700 | [41] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzak, K.; Strojny-Nędza, A.; Kaszyca, K.; Shepa, I.; Mudra, E.; Vojtko, M.; Dusza, J.; Antal, V.; Hovancova, J.; Chmielewski, M. Oxidation and Corrosion Resistance of NiCr-Re and NiCr-Re-Al2O3 Materials Fabricated by Spark Plasma Sintering. Metals 2020, 10, 1009. https://doi.org/10.3390/met10081009
Pietrzak K, Strojny-Nędza A, Kaszyca K, Shepa I, Mudra E, Vojtko M, Dusza J, Antal V, Hovancova J, Chmielewski M. Oxidation and Corrosion Resistance of NiCr-Re and NiCr-Re-Al2O3 Materials Fabricated by Spark Plasma Sintering. Metals. 2020; 10(8):1009. https://doi.org/10.3390/met10081009
Chicago/Turabian StylePietrzak, Katarzyna, Agata Strojny-Nędza, Kamil Kaszyca, Ivan Shepa, Erika Mudra, Marek Vojtko, Jan Dusza, Vitaliy Antal, Jana Hovancova, and Marcin Chmielewski. 2020. "Oxidation and Corrosion Resistance of NiCr-Re and NiCr-Re-Al2O3 Materials Fabricated by Spark Plasma Sintering" Metals 10, no. 8: 1009. https://doi.org/10.3390/met10081009
APA StylePietrzak, K., Strojny-Nędza, A., Kaszyca, K., Shepa, I., Mudra, E., Vojtko, M., Dusza, J., Antal, V., Hovancova, J., & Chmielewski, M. (2020). Oxidation and Corrosion Resistance of NiCr-Re and NiCr-Re-Al2O3 Materials Fabricated by Spark Plasma Sintering. Metals, 10(8), 1009. https://doi.org/10.3390/met10081009






