Cherry Gel Supplementation Does Not Attenuate Subjective Muscle Soreness or Alter Wellbeing Following a Match in a Team of Professional Rugby Union players: A Pilot Study
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Experimental Design
2.3. Muscle Soreness
2.4. Daily Wellbeing
2.5. Supplements
2.6. Data Analysis
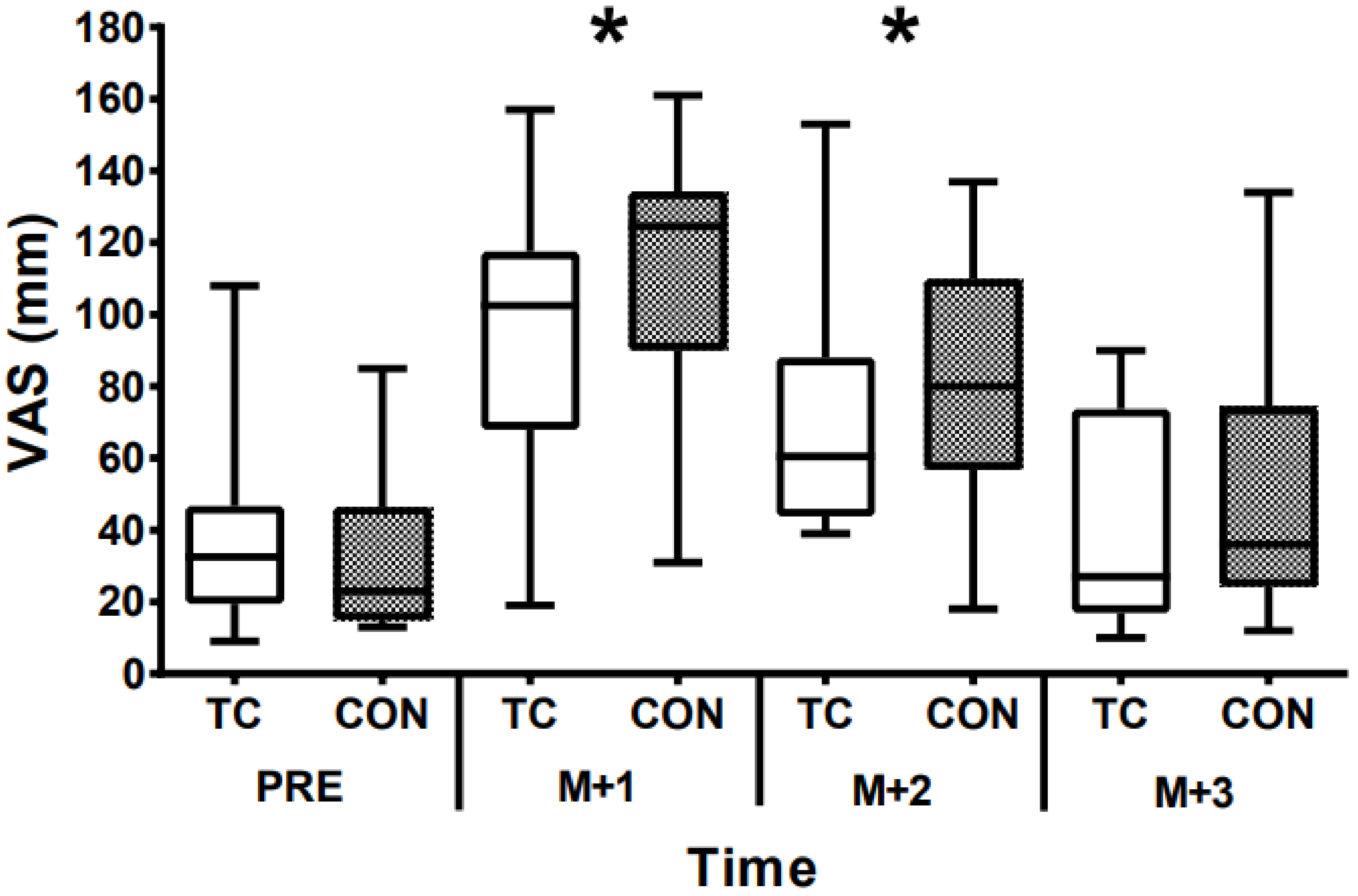
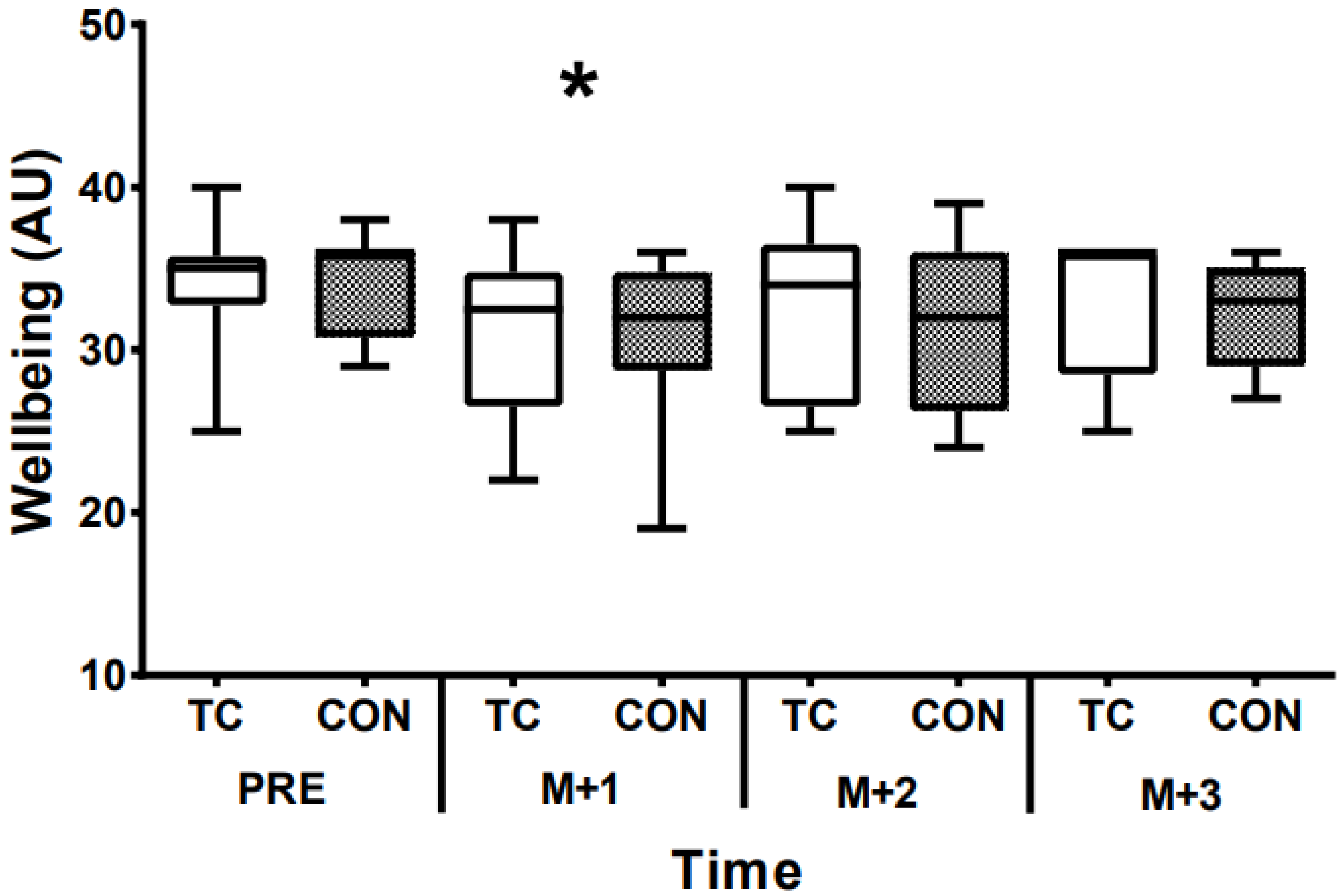
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Paulsen, G.; Ramer, U.; Mikkelsen, U.; Raastad, T.; Peake, J.M. Leucocytes, cytokines and satellite cells: What role do they play in muscle damage and regeneration following eccentric exercise? Exerc. Immunol. Rev. 2012, 18, 42–97. [Google Scholar]
- Peake, J.M.; Suzuki, K.; Coombes, J.S. The influence of antioxidant supplementation on markers of inflammation and the relationship to oxidative stress after exercise. J. Nutr. Biochem. 2007, 18, 357–371. [Google Scholar]
- Bell, P.; Stevenson, E.; Davison, G.; Howatson, G. The effects of montmorency tart cherry concentrate supplementation on recovery following prolonged, intermittent exercise. Nutrients 2016, 8, 441. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K. Involvement of neutrophils in exercise-induced muscle damage. Gen. Intern. Med. Clin. Innov. 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Suzuki, K. Cytokine response to exercise and its modulation. Antioxidants 2018, 7, 17–24. [Google Scholar]
- Connolly, D.A.; McHugh, M.P.; Padilla-Zakour, O.I.; Carlson, L.; Sayers, S.P. Efficacy of a tart cherry juice blend in preventing the symptoms of muscle damage. Brit. J. Sports Med. 2006, 40, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Levers, K.; Dalton, R.; Galvan, E.; Goodenough, C.; O’Connor, A.; Simbo, S.; Barringer, N.; Mertens-Talcott, S.U.; Rasmussen, C.; Greenwood, M.; et al. Effects of powdered Montmorency tart cherry supplementation on an acute bout of intense lower body strength exercise in resistance trained males. J. Int. Soc. Sports Nutr. 2015, 12, 41–52. [Google Scholar] [CrossRef]
- Levers, K.; Dalton, R.; Galvan, E.; O’Connor, A.; Goodenough, C.; Simbo, S.; Mertens-Talcott, S.U.; Rasmussen, C.; Greenwood, M.; Riechman, S.; et al. Effects of powdered Montmorency tart cherry supplementation on acute endurance exercise performance in aerobically trained individuals. J. Int. Soc. Sports Nutr. 2016, 13, 22–35. [Google Scholar] [CrossRef]
- Bell, P.G.; McHugh, M.P.; Stevenson, E.; Howatson, G. The role of cherries in exercise and health. Scand. J. Med. Sci. Sports 2014, 24, 477–490. [Google Scholar] [CrossRef]
- Bell, P.G.; Walshe, I.H.; Davison, G.W.; Stevenson, E.J.; Howatson, G. Recovery facilitation with Montmorency cherries following high-intensity, metabolically challenging exercise. Appl. Physiol. Nutr. Metab. 2014, 40, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; Van Someren, K.; Howatson, S.A. Influence of tart cherry juice on indices of recovery following marathon running. Scand. J. Med. Sci. Sports Exerc. 2010, 20, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Takarada, Y. Evaluation of muscle damage after a rugby match with special reference to tackle plays. Brit. J. Sports Med. 2003, 37, 416–419. [Google Scholar] [CrossRef]
- Fletcher, B.D.; Twist, C.; Haigh, J.D.; Brewer, C.; Morton, J.P.; Close, G.L. Season-long increases in perceived muscle soreness in professional rugby league players: Role of player position, match characteristics and playing surface. J. Sports Sci. 2016, 34, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Keane, K.M.; Bell, P.G.; Lodge, J.K.; Constantinou, C.L.; Jenkinson, S.E.; Bass, R.; Howatson, G. Phytochemical uptake following human consumption of Montmorency tart cherry (Prunus cerasus L.) and influence of phenolic acids on vascular smooth muscle cells in vitro. Eur. J. Nutr. 2016, 55, 1695–1705. [Google Scholar] [CrossRef] [PubMed]
- Keane, K.M.; George, T.W.; Constantinou, C.L.; Brown, M.A.; Clifford, T.; Howatson, G. Effects of Montmorency tart cherry (Prunus cerasus L.) consumption on vascular function in men with early hypertension. Am. J. Clin. Nutr. 2016, 103, 531–1539. [Google Scholar] [CrossRef] [PubMed]
- Clifford, T.; Allerton, D.M.; Brown, M.A.; Harper, L.; Horsburgh, S.; Keane, K.M.; Stevenson, E.J.; Howatson, G. Minimal muscle damage after a marathon and no influence of beetroot juice on inflammation and recovery. Appl. Physiol. Nutr. Metab. 2016, 42, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Bowtell, J.L.; Sumners, D.P.; Dyer, A.; Fox, P.; Mileva, K.N. Montmorency cherry juice reduces muscle damage caused by intensive strength exercise. Med. Sci. Sports Exerc. 2011, 43, 1544–1551. [Google Scholar] [CrossRef]
- Kuehl, K.S.; Perrier, E.T.; Elliot, D.L.; Chesnutt, J.C. Efficacy of tart cherry juice in reducing muscle pain during running: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2010, 7, 17–24. [Google Scholar] [CrossRef]
- Boorsma, R.K.; Whitfield, J.; Spriet, L.L. Beetroot juice supplementation does not improve performance of elite 1500-m runners. Med. Sci. Sports Exerc. 2014, 46, 2326–2334. [Google Scholar] [CrossRef]
- Moore, D.R. Nutrition to support recovery from endurance exercise: Optimal carbohydrate and protein replacement. Curr. Sports Med. Rep. 2015, 14, 294–300. [Google Scholar] [CrossRef]
- Howatson, G.; Bell, P.G.; Tallent, J.; Middleton, B.; McHugh, M.P.; Ellis, J. Effect of tart cherry juice (Prunus cerasus) on melatonin levels and enhanced sleep quality. Eur. J. Nutr. 2012, 8, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Hyldahl, R.D.; Hubal, M.J. Lengthening our perspective: Morphological, cellular, and molecular responses to eccentric exercise. Muscle Nerve 2014, 49, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.L. Causes of delayed onset muscle soreness and the impact on athletic performance: A review. J. Strength Cond. Res. 1992, 6, 135–141. [Google Scholar]
| Variable | TC | CON | p-Value |
|---|---|---|---|
| Total distance (m) | 6869.4 ± 1009.0 | 6822.0 ± 749.9 | 0.911 |
| High speed distance (m) | 530.5 ± 238.0 | 543.6 ± 245.6 | 0.798 |
| Sprint distance (m) | 82.1 ± 36.3 | 91.7 ± 39.4 | 0.506 |
| Player load (AU) | 712.7 ± 115.2 | 749.0 ± 126.7 | 0.542 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kupusarevic, J.; McShane, K.; Clifford, T. Cherry Gel Supplementation Does Not Attenuate Subjective Muscle Soreness or Alter Wellbeing Following a Match in a Team of Professional Rugby Union players: A Pilot Study. Sports 2019, 7, 84. https://doi.org/10.3390/sports7040084
Kupusarevic J, McShane K, Clifford T. Cherry Gel Supplementation Does Not Attenuate Subjective Muscle Soreness or Alter Wellbeing Following a Match in a Team of Professional Rugby Union players: A Pilot Study. Sports. 2019; 7(4):84. https://doi.org/10.3390/sports7040084
Chicago/Turabian StyleKupusarevic, Joe, Kevin McShane, and Tom Clifford. 2019. "Cherry Gel Supplementation Does Not Attenuate Subjective Muscle Soreness or Alter Wellbeing Following a Match in a Team of Professional Rugby Union players: A Pilot Study" Sports 7, no. 4: 84. https://doi.org/10.3390/sports7040084
APA StyleKupusarevic, J., McShane, K., & Clifford, T. (2019). Cherry Gel Supplementation Does Not Attenuate Subjective Muscle Soreness or Alter Wellbeing Following a Match in a Team of Professional Rugby Union players: A Pilot Study. Sports, 7(4), 84. https://doi.org/10.3390/sports7040084





