Age and Visual Contribution Effects on Postural Control Assessed by Principal Component Analysis of Kinematic Marker Data
Abstract
1. Introduction
2. Materials and Methods
2.1. Secondary Data Analysis
2.2. Movement Synergy Extraction
2.3. PCA-Based Variable Computation
2.4. Statistical Analysis
3. Results
3.1. Movement Components
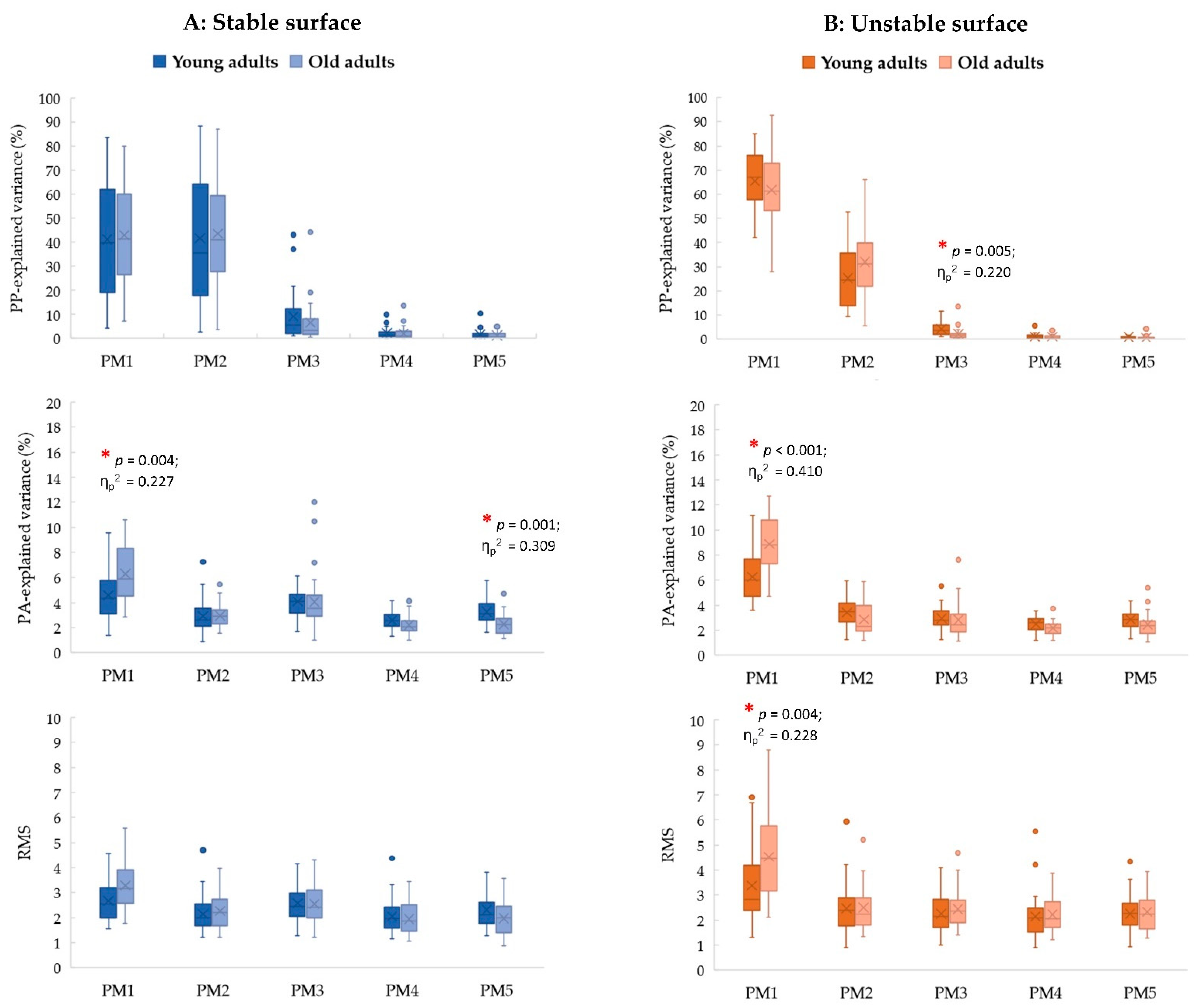
3.2. Age Effects
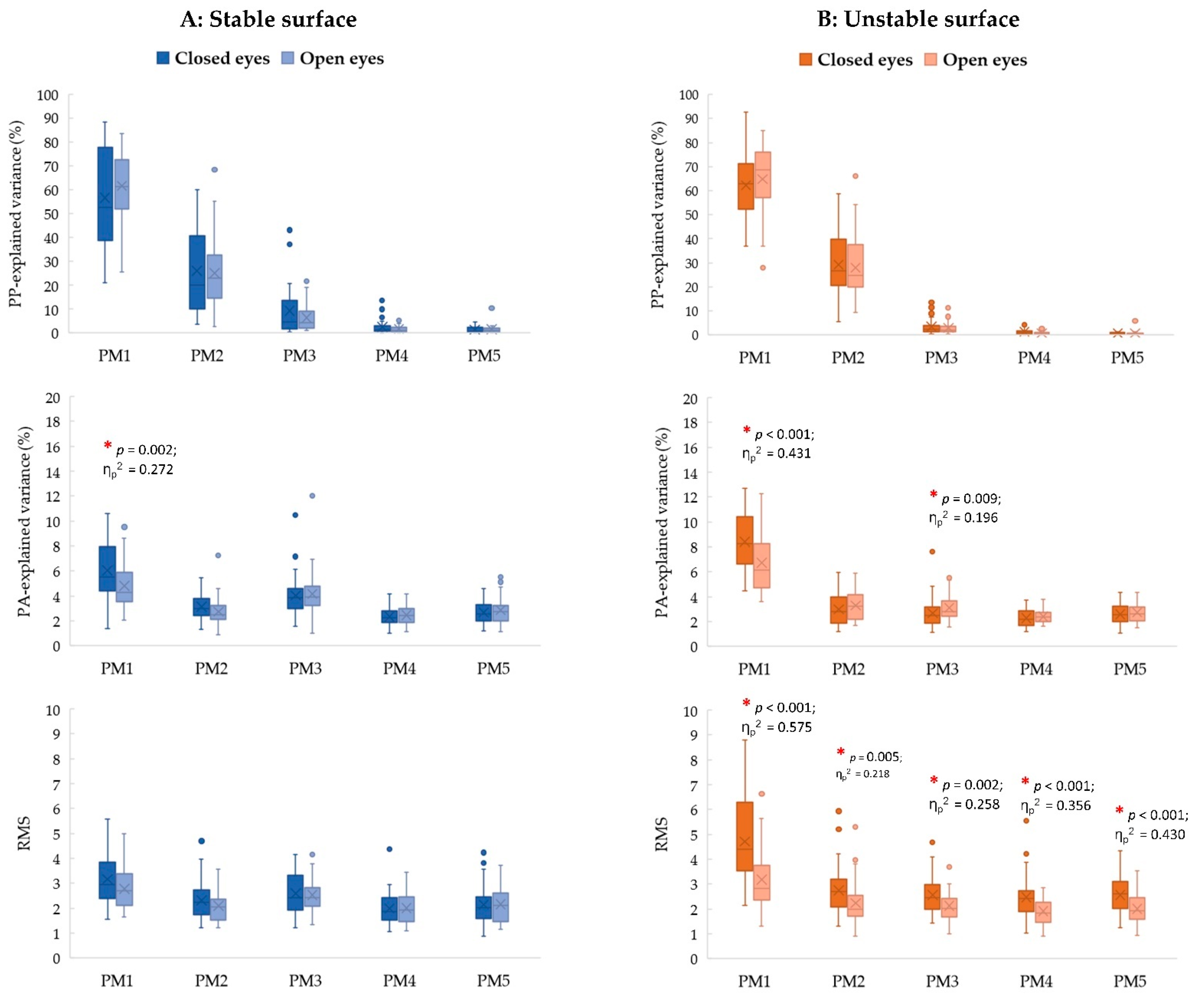
3.3. Visual Contribution Effects
3.4. Interaction Effect
4. Discussion
Limitations and Future Study
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Horak, F.B.; Nashner, L.M. Central Programming of Postural Movements: Adaptation to Altered Support-Surface Configurations. J. Neurophysiol. 1986, 55, 1369–1381. [Google Scholar] [CrossRef]
- Quijoux, F.; Nicolaï, A.; Chairi, I.; Bargiotas, I.; Ricard, D.; Yelnik, A.; Oudre, L.; Bertin-Hugault, F.; Vidal, P.P.; Vayatis, N.; et al. A Review of Center of Pressure (COP) Variables to Quantify Standing Balance in Elderly People: Algorithms and Open-Access Code. Physiol. Rep. 2021, 9, e15067. [Google Scholar] [CrossRef]
- Quijoux, F.; Vienne-Jumeau, A.; Bertin-Hugault, F.; Zawieja, P.; Lefèvre, M.; Vidal, P.-P.; Ricard, D. Center of Pressure Displacement Characteristics Differentiate Fall Risk in Older People: A Systematic Review with Meta-Analysis. Ageing Res. Rev. 2020, 62, 101117. [Google Scholar] [CrossRef]
- Duarte, M.; Freitas, S. Revision of Posturography Based on Force Plate for Balance Evaluation. Rev. Bras. Fisioter. 2010, 14, 183–192. [Google Scholar] [CrossRef]
- Federolf, P.; Angulo-Barroso, R.M.; Busquets, A.; Ferrer-Uris, B.; Gløersen, Ø.; Mohr, M.; Ó’ Reilly, D.; Promsri, A.; van Andel, S.; Wachholz, F.; et al. Letter to the Editor Regarding “The Assessment of Center of Mass and Center of Pressure during Quiet Stance: Current Applications and Future Directions”. J. Biomech. 2021, 128, 110729. [Google Scholar] [CrossRef]
- Promsri, A.; Haid, T.; Federolf, P. Complexity, Composition, and Control of Bipedal Balancing Movements as the Postural Control System Adapts to Unstable Support Surfaces or Altered Feet Positions. Neuroscience 2020, 430, 113–124. [Google Scholar] [CrossRef]
- Federolf, P.; Roos, L.; Nigg, B.M. Analysis of the Multi-Segmental Postural Movement Strategies Utilized in Bipedal, Tandem and One-Leg Stance as Quantified by a Principal Component Decomposition of Marker Coordinates. J. Biomech. 2013, 46, 2626–2633. [Google Scholar] [CrossRef]
- Federolf, P.; Tecante, K.; Nigg, B. A Holistic Approach to Study the Temporal Variability in Gait. J. Biomech. 2012, 45, 1127–1132. [Google Scholar] [CrossRef]
- Bernstein, N.A. The Co-Ordination and Regulation of Movements: Conclusions towards the Study of Motor Co-Ordination. Biodyn. Locomot. 1967, 104–113. [Google Scholar]
- Torricelli, D.; Barroso, F.; Coscia, M.; Alessandro, C.; Lunardini, F.; Bravo Esteban, E.; D’Avella, A. Muscle Synergies in Clinical Practice: Theoretical and Practical Implications. In Biosystems and Biorobotics; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; Volume 10, pp. 251–272. [Google Scholar]
- D’Avella, A.; Saltiel, P.; Bizzi, E. Combinations of Muscle Synergies in the Construction of a Natural Motor Behavior. Nat. Neurosci. 2003, 6, 300–308. [Google Scholar] [CrossRef]
- Shumway-Cook, A.; Horak, F. Assessing the Influence of Sensory Interaction of Balance. Suggestion from the Field. Phys. Ther. 1986, 66, 1548–1550. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.A. Human Balance and Posture Control during Standing and Walking. Gait Posture 1995, 3, 193–214. [Google Scholar] [CrossRef]
- Troje, N.F. Decomposing Biological Motion: A Framework for Analysis and Synthesis of Human Gait Patterns. J. Vis. 2002, 2, 371–387. [Google Scholar] [CrossRef]
- Daffertshofer, A.; Lamoth, C.J.C.; Meijer, O.G.; Beek, P.J. PCA in Studying Coordination and Variability: A Tutorial. Clin. Biomech. 2004, 19, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Verrel, J.; Lövdén, M.; Schellenbach, M.; Schaefer, S.; Lindenberger, U. Interacting Effects of Cognitive Load and Adult Age on the Regularity of Whole-Body Motion during Treadmill Walking. Psychol. Aging 2009, 24, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Federolf, P.A. A Novel Approach to Study Human Posture Control: “Principal Movements” Obtained from a Principal Component Analysis of Kinematic Marker Data. J. Biomech. 2016, 49, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Promsri, A.; Haid, T.; Federolf, P. How Does Lower Limb Dominance Influence Postural Control Movements during Single Leg Stance? Hum. Mov. Sci. 2018, 58, 165–174. [Google Scholar] [CrossRef]
- Zago, M.; Codari, M.; Iaia, F.M.; Sforza, C. Multi-Segmental Movements as a Function of Experience in Karate. J. Sports Sci. 2017, 35, 1515–1522. [Google Scholar] [CrossRef]
- Promsri, A.; Mohr, M.; Federolf, P. Principal Postural Acceleration and Myoelectric Activity: Interrelationship and Relevance for Characterizing Neuromuscular Function in Postural Control. Hum. Mov. Sci. 2021, 77, 102792. [Google Scholar] [CrossRef]
- Binda, S.M.; Culham, E.G.; Brouwer, B. Balance, Muscle Strength, and Fear of Falling in Older Adults. Exp. Aging Res. 2003, 29, 205–219. [Google Scholar] [CrossRef]
- Pollock, A.S.; Durward, B.R.; Rowe, P.J.; Paul, J.P. What Is Balance? Clin. Rehabil. 2000, 14, 402–406. [Google Scholar] [CrossRef]
- Haid, T.H.; Doix, A.-C.M.; Nigg, B.M.; Federolf, P.A. Age Effects in Postural Control Analyzed via a Principal Component Analysis of Kinematic Data and Interpreted in Relation to Predictions of the Optimal Feedback Control Theory. Front. Aging Neurosci. 2018, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Wachholz, F.; Tiribello, F.; Mohr, M.; Van Andel, S.; Federolf, P. Adolescent Awkwardness: Alterations in Temporal Control Characteristics of Posture with Maturation and the Relation to Movement Exploration. Brain Sci. 2020, 10, 216. [Google Scholar] [CrossRef] [PubMed]
- Wachholz, F.; Tiribello, F.; Promsri, A.; Federolf, P. Should the Minimal Intervention Principle Be Considered When Investigating Dual-Tasking Effects on Postural Control? Brain Sci. 2019, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Promsri, A.; Longo, A.; Haid, T.; Doix, A.-C.M.; Federolf, P. Leg Dominance as a Risk Factor for Lower-Limb Injuries in Downhill Skiers—A Pilot Study into Possible Mechanisms. Int. J. Environ. Res. Public Health 2019, 16, 3399. [Google Scholar] [CrossRef]
- Maki, B.E.; McIlroy, W.E. Postural Control in the Older Adult. Clin. Geriatr. Med. 1996, 12, 635–658. [Google Scholar] [CrossRef]
- Montell, C. Coordinated Movement: Watching Proprioception Unfold. Curr. Biol. 2019, 29, R202–R205. [Google Scholar] [CrossRef]
- Henry, M.; Baudry, S. Age-Related Changes in Leg Proprioception: Implications for Postural Control. J. Neurophysiol. 2019, 122, 525–538. [Google Scholar] [CrossRef]
- Haid, T.; Federolf, P. Human Postural Control: Assessment of Two Alternative Interpretations of Center of Pressure Sample Entropy through a Principal Component Factorization of Whole-Body Kinematics. Entropy 2018, 20, 30. [Google Scholar] [CrossRef]
- Haid, T.; Federolf, P. The Effect of Cognitive Resource Competition Due to Dual-Tasking on the Irregularity and Control of Postural Movement Components. Entropy 2019, 21, 70. [Google Scholar] [CrossRef]
- Wachholz, F.; Kockum, T.; Haid, T.; Federolf, P. Changed Temporal Structure of Neuromuscular Control, Rather than Changed Intersegment Coordination, Explains Altered Stabilographic Regularity after a Moderate Perturbation of the Postural Control System. Entropy 2019, 21, 614. [Google Scholar] [CrossRef] [PubMed]
- Zago, M.; Condoluci, C.; Manzia, C.M.; Pili, M.; Manunza, M.E.; Galli, M. Multi-Segmental Postural Control Patterns in down Syndrome. Clin. Biomech. 2021, 82, 105271. [Google Scholar] [CrossRef] [PubMed]
- Fazle Rabbi, M.; Pizzolato, C.; Lloyd, D.G.; Carty, C.P.; Devaprakash, D.; Diamond, L.E. Non-Negative Matrix Factorisation Is the Most Appropriate Method for Extraction of Muscle Synergies in Walking and Running. Sci. Rep. 2020, 10, 8266. [Google Scholar] [CrossRef]
- Haid, T.H.; Zago, M.; Promsri, A.; Doix, A.-C.M.; Federolf, P.A. PManalyzer: A Software Facilitating the Study of Sensorimotor Control of Whole-Body Movements. Front. Neuroinform. 2019, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Promsri, A.; Haid, T.; Werner, I.; Federolf, P. Leg Dominance Effects on Postural Control When Performing Challenging Balance Exercises. Brain Sci. 2020, 10, 128. [Google Scholar] [CrossRef]
- dos Santos, D.A.; Fukuchi, C.A.; Fukuchi, R.K.; Duarte, M. A Data Set with Kinematic and Ground Reaction Forces of Human Balance. PeerJ 2017, 2017, e3626. [Google Scholar] [CrossRef]
- Rethwilm, R.; Böhm, H.; Dussa, C.U.; Federolf, P. Excessive Lateral Trunk Lean in Patients With Cerebral Palsy: Is It Based on a Kinematic Compensatory Mechanism? Front. Bioeng. Biotechnol. 2019, 7, 345. [Google Scholar] [CrossRef]
- Błaszczyk, J.W.; Cieślinska-Świder, J.; Plewa, M.; Zahorska-Markiewicz, B.; Markiewicz, A. Effects of Excessive Body Weight on Postural Control. J. Biomech. 2009, 42, 1295–1300. [Google Scholar] [CrossRef]
- Suarez, H.; Angeli, S.; Suarez, A.; Rosales, B.; Carrera, X.; Alonso, R. Balance Sensory Organization in Children with Profound Hearing Loss and Cochlear Implants. Int. J. Pediatr. Otorhinolaryngol. 2007, 71, 629–637. [Google Scholar] [CrossRef]
- Wiernicka, M.; Kotwicki, T.; Kamińska, E.; Łochyński, D.; Kozinoga, M.; Lewandowski, J.; Kocur, P. Postural Stability in Adolescent Girls with Progressive Idiopathic Scoliosis. Biomed Res. Int. 2019, 2019, 7103546. [Google Scholar] [CrossRef]
- Leardini, A.; Sawacha, Z.; Paolini, G.; Ingrosso, S.; Nativo, R.; Benedetti, M.G. A New Anatomically Based Protocol for Gait Analysis in Children. Gait Posture 2007, 26, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Leardini, A.; Biagi, F.; Merlo, A.; Belvedere, C.; Benedetti, M.G. Multi-Segment Trunk Kinematics during Locomotion and Elementary Exercises. Clin. Biomech. 2011, 26, 562–571. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, I.T.; Cadima, J. Principal Component Analysis: A Review and Recent Developments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 2065. [Google Scholar] [CrossRef]
- de Leva, P. Adjustments to Zatsiorsky-Seluyanov’s Segment Inertia Parameters. J. Biomech. 1996, 29, 1223–1230. [Google Scholar] [CrossRef]
- Promsri, A.; Federolf, P. Analysis of Postural Control Using Principal Component Analysis: The Relevance of Postural Accelerations and of Their Frequency Dependency for Selecting the Number of Movement Components. Front. Bioeng. Biotechnol. 2020, 8, 480. [Google Scholar] [CrossRef] [PubMed]
- Promsri, A. Sex Difference in Running Stability Analyzed Based on a Whole-Body Movement: A Pilot Study. Sports 2022, 10, 138. [Google Scholar] [CrossRef]
- Wang, X.; O’Dwyer, N.; Halaki, M. A Review on the Coordinative Structure of Human Walking and the Application of Principal Component Analysis. Neural Regen. Res. 2013, 8, 662–670. [Google Scholar] [CrossRef]
- Promsri, A.; Cholamjiak, P.; Federolf, P. Walking Stability and Risk of Falls. Bioengineering 2023, 10, 471. [Google Scholar] [CrossRef]
- Promsri, A. Assessing Walking Stability Based on Whole-Body Movement Derived from a Depth-Sensing Camera. Sensors 2022, 22, 7542. [Google Scholar] [CrossRef]
- Longo, A.; Haid, T.; Meulenbroek, R.; Federolf, P. Biomechanics in Posture Space: Properties and Relevance of Principal Accelerations for Characterizing Movement Control. J. Biomech. 2019, 82, 397–403. [Google Scholar] [CrossRef]
- Ó’Reilly, D.; Federolf, P. Identifying Differences in Gait Adaptability across Various Speeds Using Movement Synergy Analysis. PLoS ONE 2021, 16, e0244582. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Nomura, T.; Casadio, M.; Morasso, P. Intermittent Control with Ankle, Hip, and Mixed Strategies during Quiet Standing: A Theoretical Proposal Based on a Double Inverted Pendulum Model. J. Theor. Biol. 2012, 310, 55–79. [Google Scholar] [CrossRef] [PubMed]
- Sturnieks, D.L.; St George, R.; Lord, S.R. Balance Disorders in the Elderly. Neurophysiol. Clin. 2008, 38, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Fernie, G.R.; Gryfe, C.I.; Holliday, P.J.; Llewellyn, A. The Relationship of Postural Sway in Standing to the Incidence of Falls in Geriatric Subjects. Age Ageing 1982, 11, 11–16. [Google Scholar] [CrossRef]
- Williams, G.N.; Higgins, M.J.; Lewek, M.D. Aging Skeletal Muscle: Physiologic Changes and the Effects of Training. Phys. Ther. 2002, 82, 62–68. [Google Scholar] [CrossRef]
- Menz, H.B.; Morris, M.E.; Lord, S.R. Foot and Ankle Risk Factors for Falls in Older People: A Prospective Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 866–870. [Google Scholar] [CrossRef]
- Enderlin, C.; Rooker, J.; Ball, S.; Hippensteel, D.; Alderman, J.; Fisher, S.J.; McLeskey, N.; Jordan, K. Summary of Factors Contributing to Falls in Older Adults and Nursing Implications. Geriatr. Nurs. 2015, 36, 397–406. [Google Scholar] [CrossRef]
- Granacher, U.; Gollhofer, A.; Hortobágyi, T.; Kressig, R.W.; Muehlbauer, T. The Importance of Trunk Muscle Strength for Balance, Functional Performance, and Fall Prevention in Seniors: A Systematic Review. Sport. Med. 2013, 43, 627–641. [Google Scholar] [CrossRef]
- Major, M.J. Fall Prevalence and Contributors to the Likelihood of Falling in Persons With Upper Limb Loss. Phys. Ther. 2019, 99, 377–387. [Google Scholar] [CrossRef]
- Zech, A.; Hübscher, M.; Vogt, L.; Banzer, W.; Hänsel, F.; Pfeifer, K. Balance Training for Neuromuscular Control and Performance Enhancement: A Systematic Review. J. Athl. Train. 2010, 45, 392–403. [Google Scholar] [CrossRef]
- Lambert-Shirzad, N.; Van der Loos, H.F.M. On Identifying Kinematic and Muscle Synergies: A Comparison of Matrix Factorization Methods Using Experimental Data from the Healthy Population. J. Neurophysiol. 2017, 117, 290–302. [Google Scholar] [CrossRef] [PubMed]
| Young (n = 17) | Older (n = 17) | p-Value | |
|---|---|---|---|
| Age (yrs.) | 26.6 ± 3.3 | 67.8 ± 6.6 | <0.001 * |
| Weight (kg) | 66.3 ± 12.3 | 65.4 ± 8.3 | 0.807 |
| Height (cm) | 172.2 ± 1.1 | 161.3 ± 0.8 | 0.002 * |
| Body mass index (kg/m2) | 22.3 ± 3.3 | 25.1 ± 2.3 | 0.007 * |
| PMk | PPk_rVAR | PPk_rVAR | Descriptive Characteristics |
|---|---|---|---|
| A: Stable surface | |||
| 1 | 59.1 ± 17.8 | 5.4 ± 2.2 | Anteroposterior sway around the ankle joint |
| 2 | 25.5 ± 15.5 | 2.9 ± 1.1 | Mediolateral sway around the ankle joint |
| 3 | 7.9 ± 9.2 | 4.1 ± 1.8 | Anteroposterior sway around the hip joint |
| 4 | 2.1 ± 2.5 | 2.4 ± 0.7 | Whole-body rotation |
| 5 | 1.5 ± 1.7 | 2.8 ± 1.0 | Head flexion and extension combined with the hip rotation |
| B: Unstable surface | |||
| 1 | 63.6 ± 13.0 | 7.6 ± 2.4 | Anteroposterior sway around the ankle joint combined with the support surface movement |
| 2 | 28.6 ± 12.5 | 3.1 ± 1.2 | Mediolateral sway around the ankle joint |
| 3 | 3.2 ± 2.8 | 2.9 ± 1.1 | Anteroposterior sway around the ankle joint combined with anteroposterior sway around the hip joint |
| 4 | 1.1 ± 1.1 | 2.3 ± 0.6 | Whole-body rotation |
| 5 | 0.8 ± 1.0 | 2.7 ± 0.9 | Whole-body rotation combined with knee flexion and extension |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Promsri, A. Age and Visual Contribution Effects on Postural Control Assessed by Principal Component Analysis of Kinematic Marker Data. Sports 2023, 11, 98. https://doi.org/10.3390/sports11050098
Promsri A. Age and Visual Contribution Effects on Postural Control Assessed by Principal Component Analysis of Kinematic Marker Data. Sports. 2023; 11(5):98. https://doi.org/10.3390/sports11050098
Chicago/Turabian StylePromsri, Arunee. 2023. "Age and Visual Contribution Effects on Postural Control Assessed by Principal Component Analysis of Kinematic Marker Data" Sports 11, no. 5: 98. https://doi.org/10.3390/sports11050098
APA StylePromsri, A. (2023). Age and Visual Contribution Effects on Postural Control Assessed by Principal Component Analysis of Kinematic Marker Data. Sports, 11(5), 98. https://doi.org/10.3390/sports11050098






