Explorative Data Analysis of Drosophila suzukii Trap Catches from a Seven-Year Monitoring Program in Southwest Germany
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area: Topography and Weather
2.2. Trap Designs and Placement
2.3. Species Identification
2.4. Monitoring Data from DrosoMon
2.5. Exploratory Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Swoboda-Bhattarai, K.A.; McPhie, D.R.; Burrack, H.J. Reproductive Status of Drosophila suzukii (Diptera: Drosophilidae) Females Influences Attraction to Fermentation-Based Baits and Ripe Fruits. J. Econ. Entomol. 2017, 110, 1648–1652. [Google Scholar] [CrossRef] [PubMed]
- Hauser, M. A historic account of the invasion of Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) in the continental United States, with remarks on their identification. Pest Manag. Sci. 2011, 67, 1352–1357. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, H.; Beppu, K.; Kimura, M.T. Seasonal life cycles and resource uses of flower- and fruit-feeding drosophilid flies (Diptera: Drosophilidae) in central Japan. Entomol. Sci. 2010, 13, 60–67. [Google Scholar] [CrossRef]
- Farnsworth, D.; Hamby, K.A.; Bolda, M.; Goodhue, R.E.; Williams, J.C.; Zalom, F.G. Economic analysis of revenue losses and control costs associated with the spotted wing drosophila, Drosophila suzukii (Matsumura), in the California raspberry industry. Pest Manag. Sci. 2017, 73, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- Mazzi, D.; Bravin, E.; Meraner, M.; Finger, R.; Kuske, S. Economic Impact of the Introduction and Establishment of Drosophila suzukii on Sweet Cherry Production in Switzerland. Insects 2017, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Asplen, M.K.; Anfora, G.; Biondi, A.; Choi, D.S.; Chu, D.; Daane, K.M.; Gibert, P.; Gutierrez, A.P.; Hoelmer, K.A.; Hutchison, W.D.; et al. Invasion biology of spotted wing Drosophila (Drosophila suzukii): A global perspective and future priorities. J. Pest Sci. 2015, 88, 469–494. [Google Scholar] [CrossRef]
- Briem, F.; Eben, A.; Gross, J.; Vogt, H. An invader supported by a parasite: Mistletoe berries as a host for food and reproduction of Spotted Wing Drosophila in early spring. J. Pest Sci. 2016, 89, 749–759. [Google Scholar] [CrossRef]
- Diepenbrock, L.M.; McPhie, D.R. Use of a Novel Host Plant Family, Basallaceae, by Drosophila suzukii (Diptera: Drosophilidae). Proc. Entomol. Soc. Wash. 2018, 120, 255–259. [Google Scholar] [CrossRef]
- Kenis, M.; Tonina, L.; Eschen, R.; van der Sluis, B.; Sancassani, M.; Mori, N.; Haye, T.; Helsen, H. Non-crop plants used as hosts by Drosophila suzukii in Europe. J. Pest Sci. 2016, 89, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Dreves, A.J.; Cave, A.M.; Kawai, S.; Isaacs, R.; Miller, J.C.; Van Timmeren, S.; Bruck, D.J. Infestation of Wild and Ornamental Noncrop Fruits by Drosophila suzukii (Diptera: Drosophilidae). Ann. Entomol. Soc. Am. 2015, 108, 117–129. [Google Scholar] [CrossRef]
- Poyet, M.; Le Roux, V.; Gibert, P.; Meirland, A.; Prevost, G.; Eslin, P.; Chabrerie, O. The Wide Potential Trophic Niche of the Asiatic Fruit Fly Drosophila suzukii: The Key of Its Invasion Success in Temperate Europe? PLoS ONE 2015, 10, e0142785. [Google Scholar] [CrossRef] [PubMed]
- Briem, F.; Zeisler, C.; Guenay, Y.; Staudacher, K.; Vogt, H.; Traugott, M. Identifying plant DNA in the sponging–feeding insect pest Drosophila suzukii. J. Pest Sci. 2018, 91, 985–994. [Google Scholar] [CrossRef]
- Fountain, M.T.; Bennett, J.; Cobo-Medina, M.; Conde Ruiz, R.; Deakin, G.; Delgado, A.; Harrison, R.; Harrison, N. Alimentary microbes of winter-form Drosophila suzukii. Insect Mol. Biol. 2018, 27, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Calabria, G.; Maca, J.; Bachli, G.; Serra, L.; Pascual, M. First records of the potential pest species Drosophila suzukii (Diptera: Drosophilidae) in Europe. J. Appl. Entomol. 2012, 136, 139–147. [Google Scholar] [CrossRef]
- Walsh, D.B.; Bolda, M.P.; Goodhue, R.E.; Dreves, A.J.; Lee, J.; Bruck, D.J.; Walton, V.M.; O’Neal, S.D.; Zalom, F.G. Drosophila suzukii (Diptera: Drosophilidae): Invasive Pest of Ripening Soft Fruit Expanding its Geographic Range and Damage Potential. J. Integr. Pest Manag. 2011, 2, G1–G7. [Google Scholar] [CrossRef]
- Heuck, P. Drosophila suzukii erstmals für Deutschland (Diptera: Drosophildae). NachrBl. Bayer. Ent. 2012, 61, 46. [Google Scholar]
- Vogt, H.; Hoffmann, C.; Baufeld, P. Ein neuer Schädling, die Kirschessigfliege, Drosophila suzukii (Matsumura, 1931), bedroht Obst- und Weinkulturen (Diptera, Drosophilidae). Entomol. Nachr. Ber. 2012, 56, 191–196. [Google Scholar]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World Map of the Köppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar] [CrossRef]
- Thistlewood, H.M.A.; Gill, P.; Beers, E.H.; Shearer, P.W.; Walsh, D.B.; Rozema, B.M.; Acheampong, S.; Castagnoli, S.; Yee, W.L.; Smytheman, P.; et al. Spatial Analysis of Seasonal Dynamics and Overwintering of Drosophila suzukii (Diptera: Drosophilidae) in the Okanagan-Columbia Basin, 2010–2014. Environ. Entomol. 2018, 47, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Dalton, D.T.; Walton, V.M.; Shearer, P.W.; Walsh, D.B.; Caprile, J.; Isaacs, R. Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Manag. Sci. 2011, 67, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.W.; Hamby, K.A.; Wilson, H.E.; Zalom, F.G. Seasonal monitoring of Drosophila suzukii (Diptera: Drosophilidae) in a mixed fruit production system. J. Asia-Pacif. Entomol. 2014, 17, 857–864. [Google Scholar] [CrossRef]
- Tochen, S.; Woltz, J.M.; Dalton, D.T.; Lee, J.C.; Wiman, N.G.; Walton, V.M. Humidity affects populations of Drosophila suzukii (Diptera: Drosophilidae) in blueberry. J. Appl. Entomol. 2016, 140, 47–57. [Google Scholar] [CrossRef]
- Wang, X.G.; Stewart, T.J.; Biondi, A.; Chavez, B.A.; Ingels, C.; Caprile, J.; Grant, J.A.; Walton, V.M.; Daane, K.M. Population dynamics and ecology of Drosophila suzukii in Central California. J. Pest Sci. 2016, 89, 701–712. [Google Scholar] [CrossRef]
- Wiman, N.G.; Walton, V.M.; Dalton, D.T.; Anfora, G.; Burrack, H.J.; Chiu, J.C.; Daane, K.M.; Grassi, A.; Miller, B.; Tochen, S.; et al. Integrating temperature-dependent life table data into a matrix projection model for Drosophila suzukii population estimation. PLoS ONE 2014, 9, e106909. [Google Scholar] [CrossRef] [PubMed]
- Cini, A.; Anfora, G.; Escudero-Colomar, L.A.; Grassi, A.; Santosuosso, U.; Seljak, G.; Papini, A. Tracking the invasion of the alien fruit pest Drosophila suzukii in Europe. J. Pest Sci. 2014, 87, 559–566. [Google Scholar] [CrossRef]
- Grassi, A.; Gottardello, A.; Dalton, D.T.; Tait, G.; Rendon, D.; Ioriatti, C.; Gibeaut, D.; Rossi Stacconi, M.V.; Walton, V.M. Seasonal Reproductive Biology of Drosophila suzukii (Diptera: Drosophilidae) in Temperate Climates. Environ. Entomol. 2018, 47, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Mazzetto, F.; Pansa, M.G.; Ingegno, B.L.; Tavella, L.; Alma, A. Monitoring of the exotic fly Drosophila suzukii in stone, pome and soft fruit orchards in NW Italy. J. Asia-Pacif. Entomol. 2015, 18, 321–329. [Google Scholar] [CrossRef]
- Kremmer, L.; Thaon, M.; Borowiec, N.; David, J.; Poirie, M.; Gatti, J.L.; Ris, N. Field Monitoring of Drosophila suzukii and Associated Communities in South Eastern France as a Pre-Requisite for Classical Biological Control. Insects 2017, 8, 124. [Google Scholar] [CrossRef] [PubMed]
- Tochen, S.; Dalton, D.T.; Wiman, N.; Hamm, C.; Shearer, P.W.; Walton, V.M. Temperature-related development and population parameters for Drosophila suzukii (Diptera: Drosophilidae) on cherry and blueberry. Environ. Entomol. 2014, 43, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Wallingford, A.K.; Lee, J.C.; Loeb, G.M. The influence of temperature and photoperiod on the reproductive diapause and cold tolerance of spotted-wing drosophila, Drosophila suzukii. Entomol. Exp. Appl. 2016, 159, 327–337. [Google Scholar] [CrossRef]
- Shearer, P.W.; West, J.D.; Walton, V.M.; Brown, P.H.; Svetec, N.; Chiu, J.C. Seasonal cues induce phenotypic plasticity of Drosophila suzukii to enhance winter survival. BMC Ecol. 2016, 16, 11. [Google Scholar] [CrossRef] [PubMed]
- Briem, F.; Just, J.; Vogt, H. JKI-Trap to Monitor the Spotted Wing Drosophila, Drosophila suzukii; Institut für Pflanzenschutz in Obst- und Weinbau: Quedlinburg Germany, 2018. [Google Scholar]
- Vlach, J. Identifying Drosophila suzukii; Oregon Department of Agriculture: Salem, OR, USA, 2013.
- Briem, F.; Dominic, A.R.; Golla, B.; Hoffmann, C.; Englert, C.; Herz, A.; Vogt, H. Trap Captures Data from the 7-Year Drosophila suzukii Monitoring Program in Southwest Germany [Data Set], 1.0st ed.; Julius Kühn-Institut, Bundesforschungsinstitut für Kulturpflanzen: Quedlinburg, Germany, 2018. [Google Scholar]
- Briem, F.; Breuer, M.; Köppler, K.; Vogt, H. Phenology and occurrence of spotted wing drosophila in Germany and case studies for its control in berry crops. IOBC-WPRS Bull. 2015, 109, 233–237. [Google Scholar]
- Venables, W.N.; Ripley, B.D. Modern Applied Statistics with S, Fourth ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Emiljanowicz, L.M.; Ryan, G.D.; Langille, A.; Newman, J. Development, reproductive output and population growth of the fruit fly pest Drosophila suzukii (Diptera: Drosophilidae) on artificial diet. J. Econ. Entomol. 2014, 107, 1392–1398. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, S.L.; Mehlferber, E.; Moore, P.J. Life-history trade-offs under different larval diets in Drosophila suzukii (Diptera: Drosophilidae). Physiol. Entomol. 2015, 40, 2–9. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Elsensohn, J.E.; Loeb, G.M. Non-Crop Host Sampling Yields Insights into Small-Scale Population Dynamics of Drosophila suzukii (Matsumura). Insects 2018, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Wiman, N.G.; Dalton, D.T.; Anfora, G.; Biondi, A.; Chiu, J.C.; Daane, K.M.; Gerdeman, B.; Gottardello, A.; Hamby, K.A.; Isaacs, R.; et al. Drosophila suzukii population response to environment and management strategies. J. Pest Sci. 2016, 89, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Poyet, M.; Eslin, P.; Heraude, M.; Le Roux, V.; Prevost, G.; Gibert, P.; Chabrerie, O. Invasive host for invasive pest: When the Asiatic cherry fly (Drosophila suzukii) meets the American black cherry (Prunus serotina) in Europe. Agric. For. Entomol. 2014, 16, 251–259. [Google Scholar] [CrossRef]
- Hamby, K.A.; Bolda, M.P.; Sheehan, M.E.; Zalom, F.G. Seasonal monitoring for Drosophila suzukii (Diptera: Drosophilidae) in California commercial raspberries. Environ. Entomol. 2014, 43, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Zerulla, F.N.; Schmidt, S.; Streitberger, M.; Zebitz, C.P.W.; Zelger, R. On the overwintering ability of Drosophila suzukii in South Tyrol. J. Berry Res. 2015, 5, 41–48. [Google Scholar] [CrossRef]
- Tait, G.; Grassi, A.; Pfab, F.; Crava, C.M.; Dalton, D.T.; Magarey, R.; Ometto, L.; Vezzulli, S.; Rossi-Stacconi, M.V.; Gottardello, A.; et al. Large-scale spatial dynamics of Drosophila suzukii in Trentino, Italy. J. Pest Sci. 2018, 91, 1213–1224. [Google Scholar] [CrossRef]
- Burrack, H.J.; Asplen, M.; Bahder, L.; Collins, J.; Drummond, F.A.; Guedot, C.; Isaacs, R.; Johnson, D.; Blanton, A.; Lee, J.C.; et al. Multistate Comparison of Attractants for Monitoring Drosophila suzukii (Diptera: Drosophilidae) in Blueberries and Caneberries. Environ. Entomol. 2015, 44, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Cha, D.H.; Hesler, S.P.; Park, S.; Adams, T.B.; Zack, R.S.; Rogg, H.; Loeb, G.M.; Landolt, P.J. Simpler is better: Fewer non-target insects trapped with a four-component chemical lure vs. a chemically more complex food-type bait for Drosophila suzukii. Entomol. Exp. Appl. 2015, 154, 251–260. [Google Scholar] [CrossRef]
- Lasa, R.; Tadeo, E.; Toledo-Hernandez, R.A.; Carmona, L.; Lima, I.; Williams, T. Improved capture of Drosophila suzukii by a trap baited with two attractants in the same device. PLoS ONE 2017, 12, e0188350. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, D.M.; Gut, L.J.; Miller, J.R. Estimating Monitoring Trap Plume Reach and Trapping Area for Drosophila suzukii (Diptera: Drosophilidae) in Michigan Tart Cherry. J. Econ. Entomol. 2018, 111, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Beers, E.H.; Van Steenwyk, R.A.; Shearer, P.W.; Coates, W.W.; Grant, J.A. Developing Drosophila suzukii management programs for sweet cherry in the western United States. Pest Manag. Sci. 2011, 67, 1386–1395. [Google Scholar] [CrossRef] [PubMed]
- Ryan, G.D.; Emiljanowicz, L.; Wilkinson, F.; Kornya, M.; Newman, J.A. Thermal Tolerances of the Spotted-Wing Drosophila Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Entomol. 2016, 109, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Eben, A.; Reifenrath, M.; Briem, F.; Pink, S.; Vogt, H. Response of Drosophila suzukii (Diptera: Drosophilidae) to extreme heat and dryness. Agric. For. Entomol. 2018, 20, 113–121. [Google Scholar] [CrossRef]
- Wong, J.S.; Wallingford, A.K.; Loeb, G.M.; Lee, J.C. Physiological status of Drosophila suzukii (Diptera: Drosophilidae) affects their response to attractive odours. J. Appl. Entomol. 2018, 142, 473–482. [Google Scholar] [CrossRef]
- Jakobs, R.; Gariepy, T.D.; Sinclair, B.J. Adult plasticity of cold tolerance in a continental-temperate population of Drosophila suzukii. J. Insect Physiol. 2015, 79, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.T. Cold and heat tolerance of drosophilid flies with reference to their latitudinal distributions. Oecologia 2004, 140, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Enriquez, T.; Colinet, H. Basal tolerance to heat and cold exposure of the spotted wing drosophila, Drosophila suzukii. PeerJ 2017, 5, e3112. [Google Scholar] [CrossRef] [PubMed]
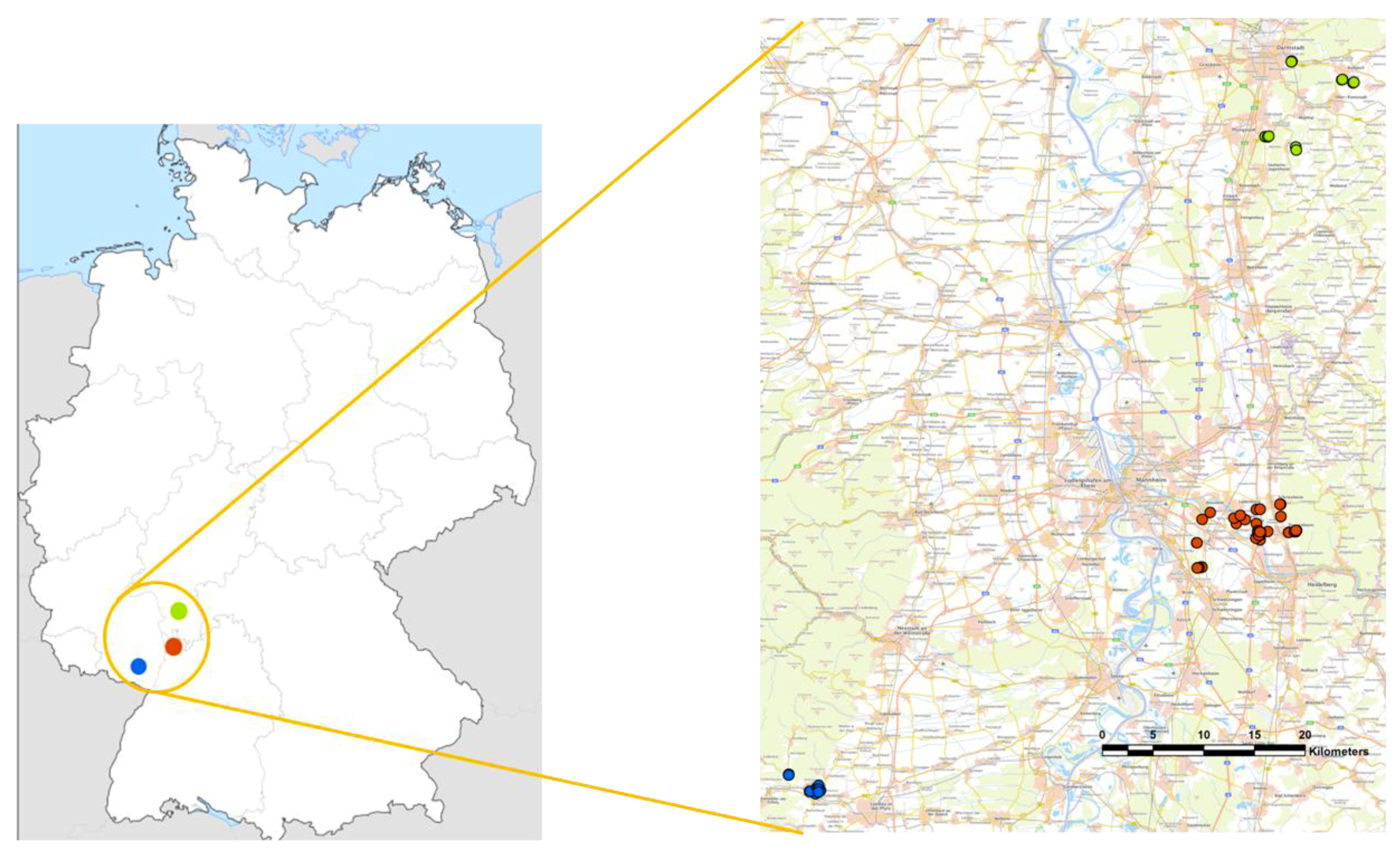

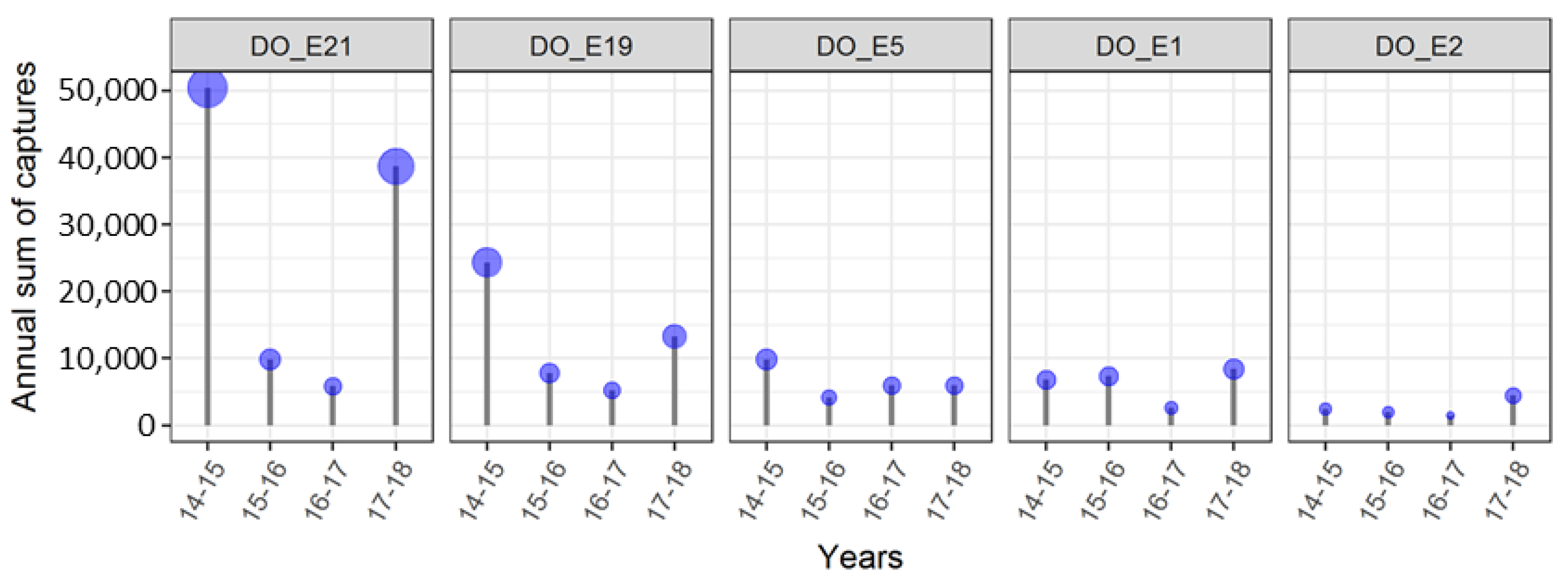
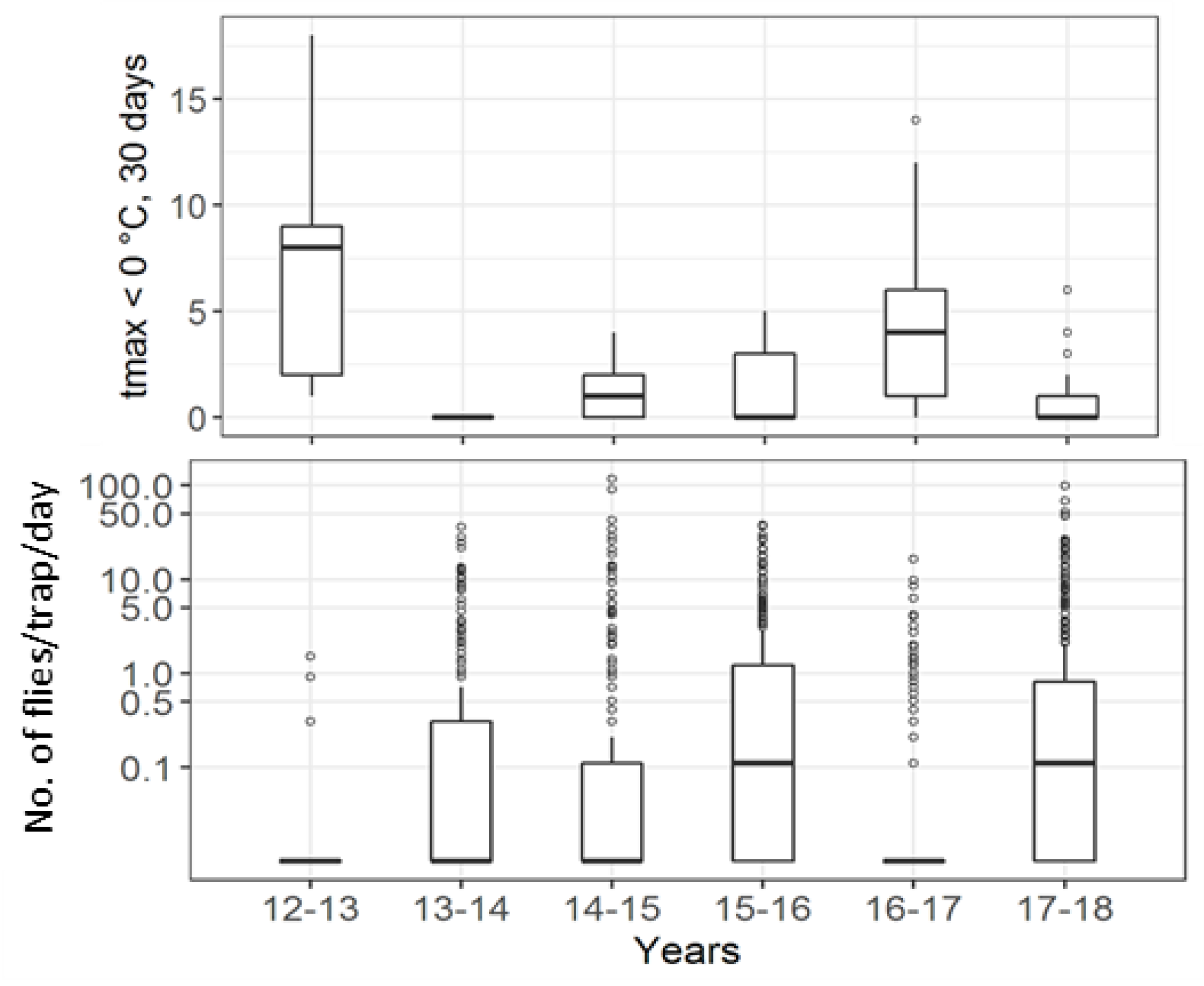
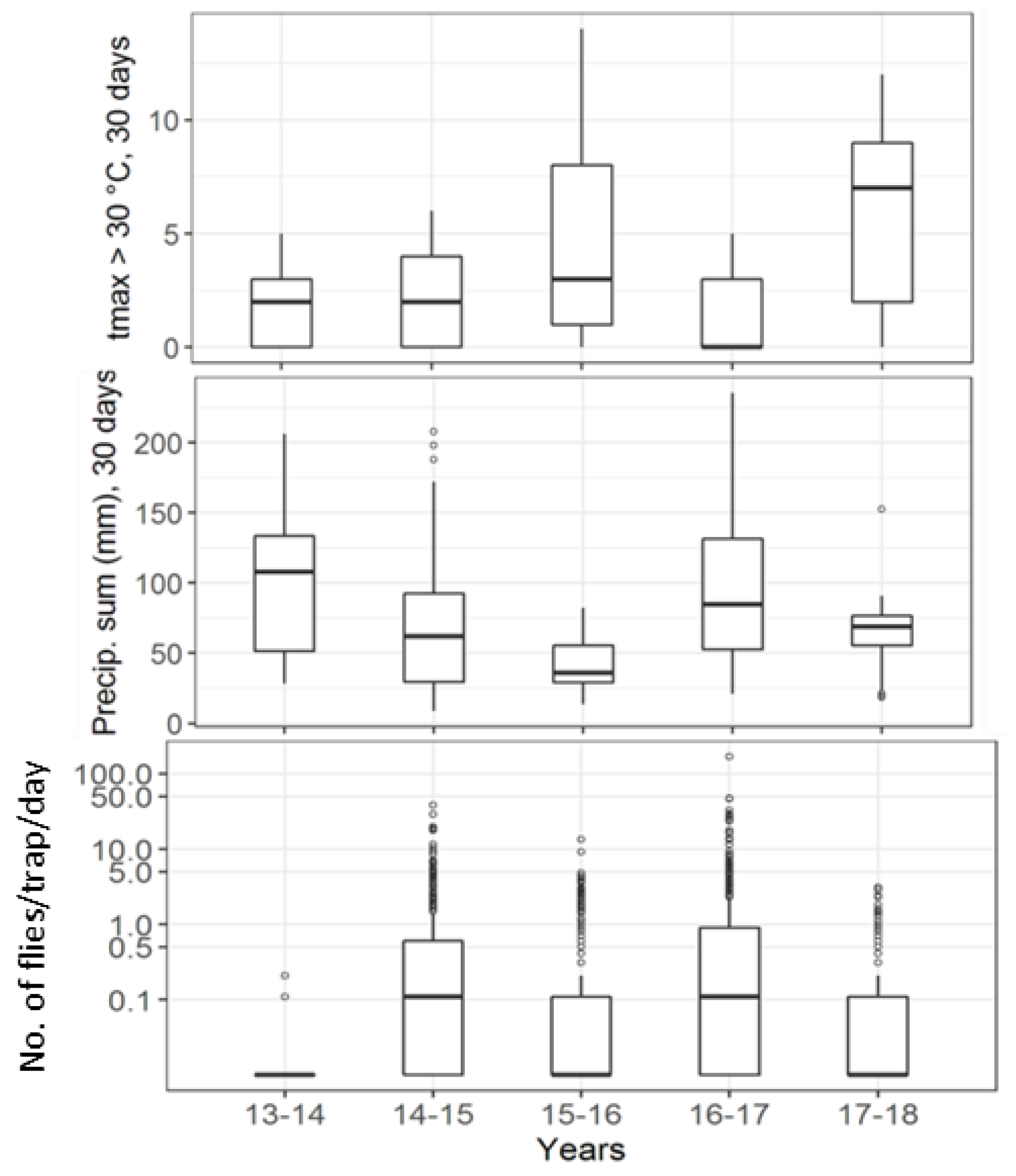
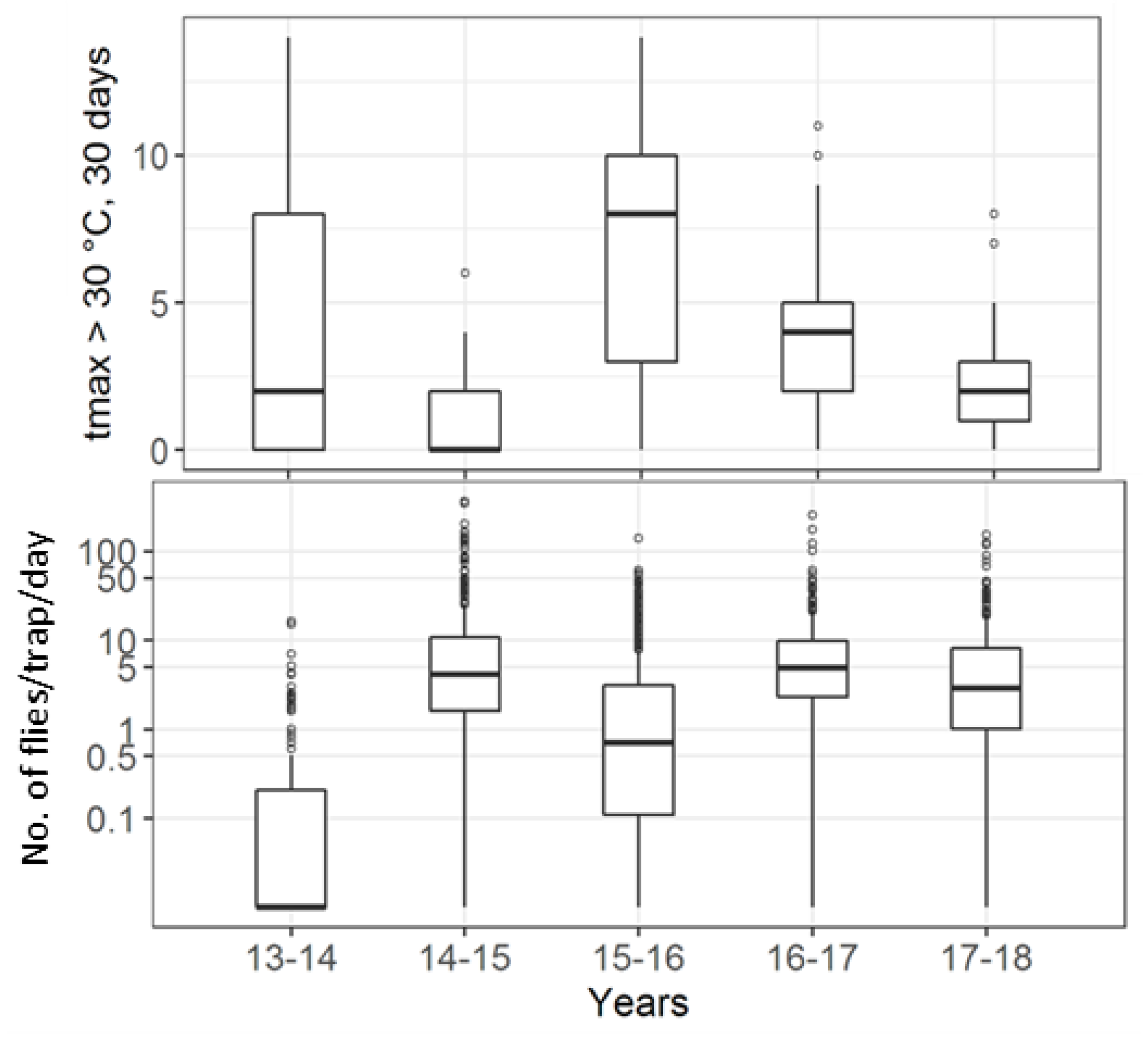
| Institute | Forest | Forest Border | Hedge | Orchards | Vineyard | Urban Area | Total |
|---|---|---|---|---|---|---|---|
| JKI Dos | 33 | 2 | 18 | 15 | 0 | 0 | 68 |
| JKI Sie | 1 | 1 | 5 | 6 | 4 | 2 | 19 |
| JKI Dar | 3 | 1 | 3 | 4 | 0 | 2 | 13 |
| Total No. of traps | 37 | 4 | 26 | 25 | 4 | 4 | 100 |
| Seasonal Classes | Start-End | Day of Year | Observation |
|---|---|---|---|
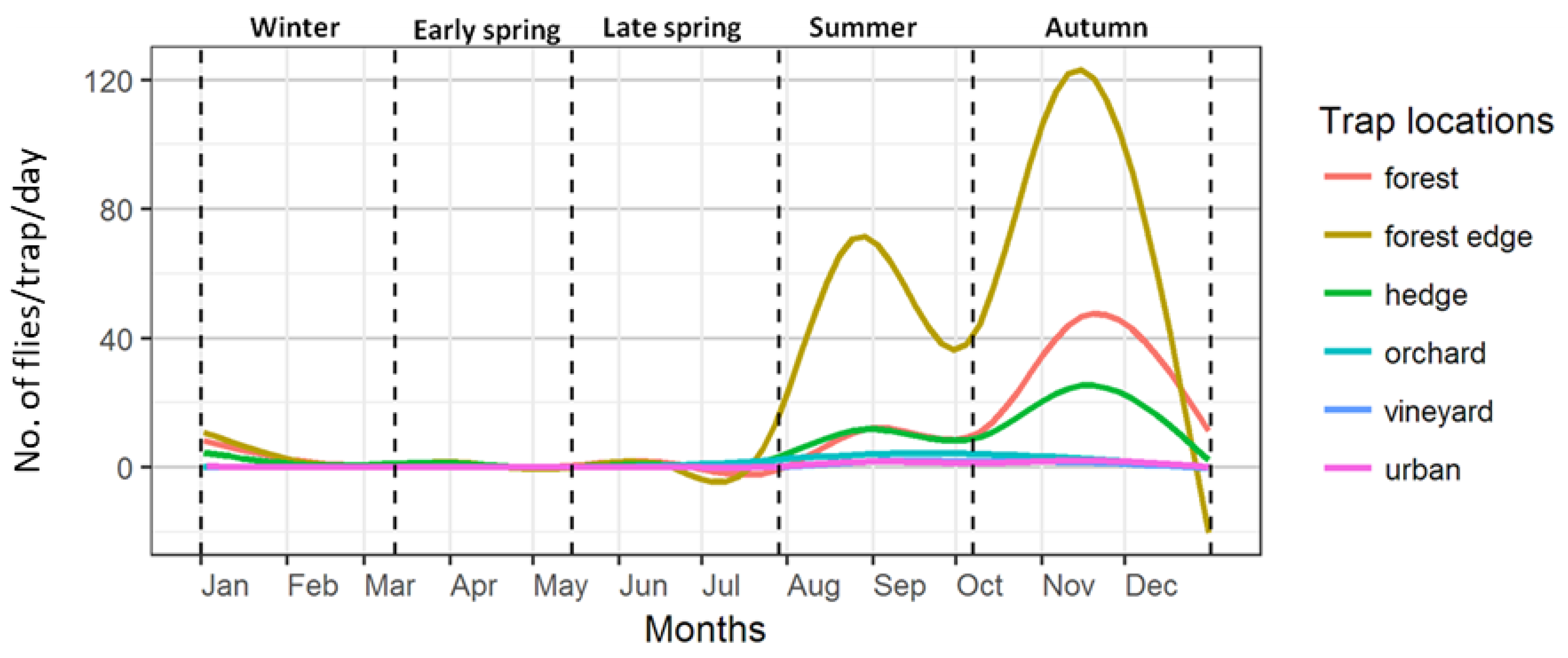
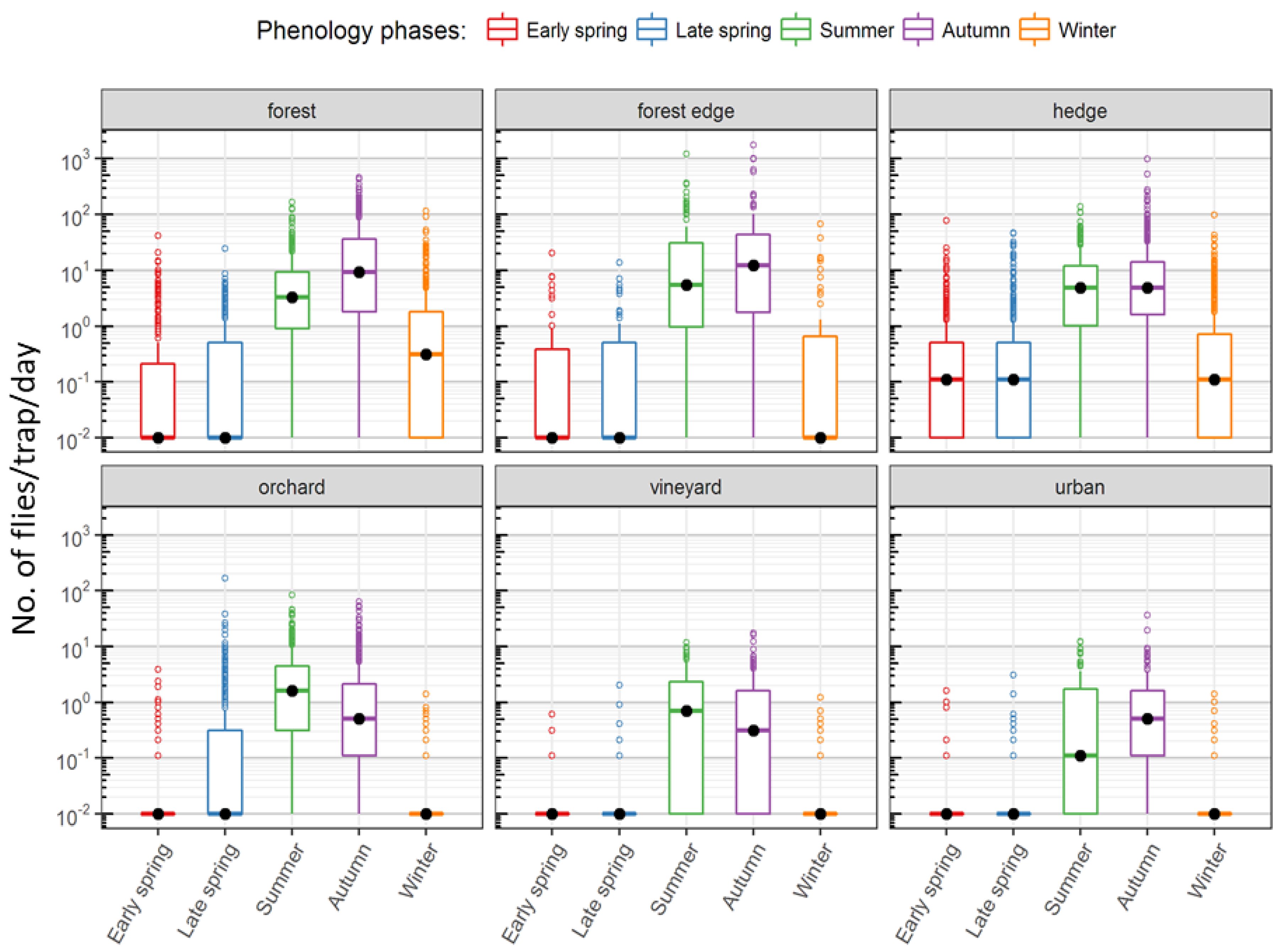
| Early spring | March 12–May 15 | 71–135 | Lowest capture rate |
| Late spring | May 16–July 29 | 136–210 | Increase in trap captures, emergence of summer morph |
| Summer | July 30–October 7 | 211–280 | First peak in number of captures |
| Autumn | October 8–December 31 | 281–366 | Second peak in number of captures, emergence of winter morph |
| Winter | January 1–March 11 | 1–70 | Winter decline of captures |
| Weather Indices | Description | Expected Effect on D. suzukii |
|---|---|---|
| Tmax < 0 °C | Ice days | − |
| Tmin < 0 °C | Frost days | − |
| Tmax ≤ 8 °C | Cold days | − |
| 20 °C ≤ Tmean ≤ 30 °C | Optimal development range | + |
| Tmax > 30 °C | Hot days | − |
| Tmean °C | Average temperature | Varying with season |
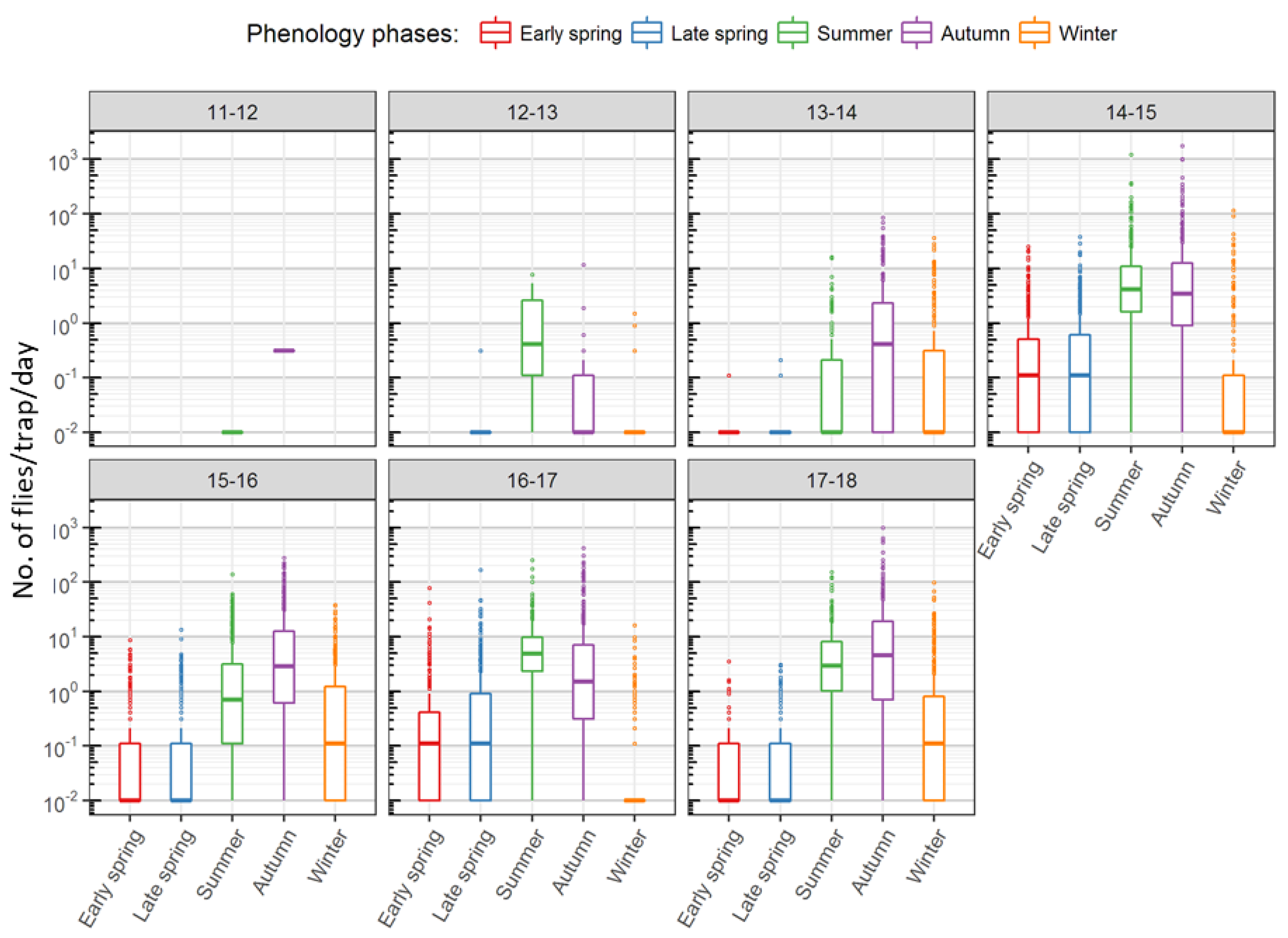
| Operator | 2011–2012 | 2012–2013 | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 | 2017–2018 |
|---|---|---|---|---|---|---|---|
| JKI Dar | (0) 0 | (8) 0 | (10) 3 | 10 | 13 | 13 | 6 |
| JKI Dos | (5) 1 | 11 | 23 | 60 | 56 | 61 | 45 |
| JKI Sie | (19) 0 | 16 | 17 | 17 | 19 | 19 | 17 |
| Total | (24) 1 | 27 | 43 | 87 | 88 | 93 | 68 |
| D. suzukii Phenology | Males% | Females% |
|---|---|---|
| Early spring | 39.9 | 60.1 |
| Late spring | 44.5 | 55.5 |
| Summer | 52 | 48 |
| Autumn | 55.1 | 44.9 |
| Winter | 40.3 | 59.7 |
| Season | Weather Parameter | Coeff | se | p-Value |
|---|---|---|---|---|
| Late spring | 20 °C ≤ Tmean ≤ 30 °C (days) | 0.134 | 0.008 | <0.001 |
| Late spring | Mean of tmean (°C) | 0.309 | 0.020 | <0.001 |
| Summer | 20 °C ≤ Tmean ≤ 30 °C (days) | −0.045 | 0.005 | <0.001 |
| Summer | Mean of tmean (°C) | −0.163 | 0.014 | <0.001 |
| Summer | tmax > 30 °C (days) | −0.131 | 0.009 | <0.001 |
| Late spring and autumn | tmax > 30 °C (days) | −0.102 | 0.009 | <0.001 |
| Autumn, winter and early spring | tmin < 0 °C (days) | −0.092 | 0.005 | <0.001 |
| Autumn, winter and early spring | Precipitation sum (mm) | 0.014 | 0.001 | <0.001 |
| Autumn, winter and early spring | Mean of tmean (°C) | 0.110 | 0.009 | <0.001 |
| Autumn, winter and early spring | tmax < 0 °C (days) | −0.350 | 0.026 | <0.001 |
| Autumn, winter and early spring | tmean ≤ 8 °C (days) | −0.026 | 0.003 | <0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Briem, F.; Dominic, A.R.; Golla, B.; Hoffmann, C.; Englert, C.; Herz, A.; Vogt, H. Explorative Data Analysis of Drosophila suzukii Trap Catches from a Seven-Year Monitoring Program in Southwest Germany. Insects 2018, 9, 125. https://doi.org/10.3390/insects9040125
Briem F, Dominic AR, Golla B, Hoffmann C, Englert C, Herz A, Vogt H. Explorative Data Analysis of Drosophila suzukii Trap Catches from a Seven-Year Monitoring Program in Southwest Germany. Insects. 2018; 9(4):125. https://doi.org/10.3390/insects9040125
Chicago/Turabian StyleBriem, Felix, Anto Raja Dominic, Burkhard Golla, Christoph Hoffmann, Camilla Englert, Annette Herz, and Heidrun Vogt. 2018. "Explorative Data Analysis of Drosophila suzukii Trap Catches from a Seven-Year Monitoring Program in Southwest Germany" Insects 9, no. 4: 125. https://doi.org/10.3390/insects9040125
APA StyleBriem, F., Dominic, A. R., Golla, B., Hoffmann, C., Englert, C., Herz, A., & Vogt, H. (2018). Explorative Data Analysis of Drosophila suzukii Trap Catches from a Seven-Year Monitoring Program in Southwest Germany. Insects, 9(4), 125. https://doi.org/10.3390/insects9040125






