A Better Understanding of Bee Nutritional Ecology Is Needed to Optimize Conservation Strategies for Wild Bees—The Application of Ecological Stoichiometry
Abstract
1. Background
1.1. An Improved Understanding of Wild-Bee Nutritional Ecology Is Needed
1.2. The Framework of Ecological Stoichiometry May Be Applied for A Better Understanding of Bee Nutritional Ecology
2. Adult Bee Individuals Need Food Quantity, but Bee Populations Need Food Quality to Thrive and Prosper
3. The Application of Ecological Stoichiometry to Gain Knowledge on the Nutritional Ecology of Wild Bees
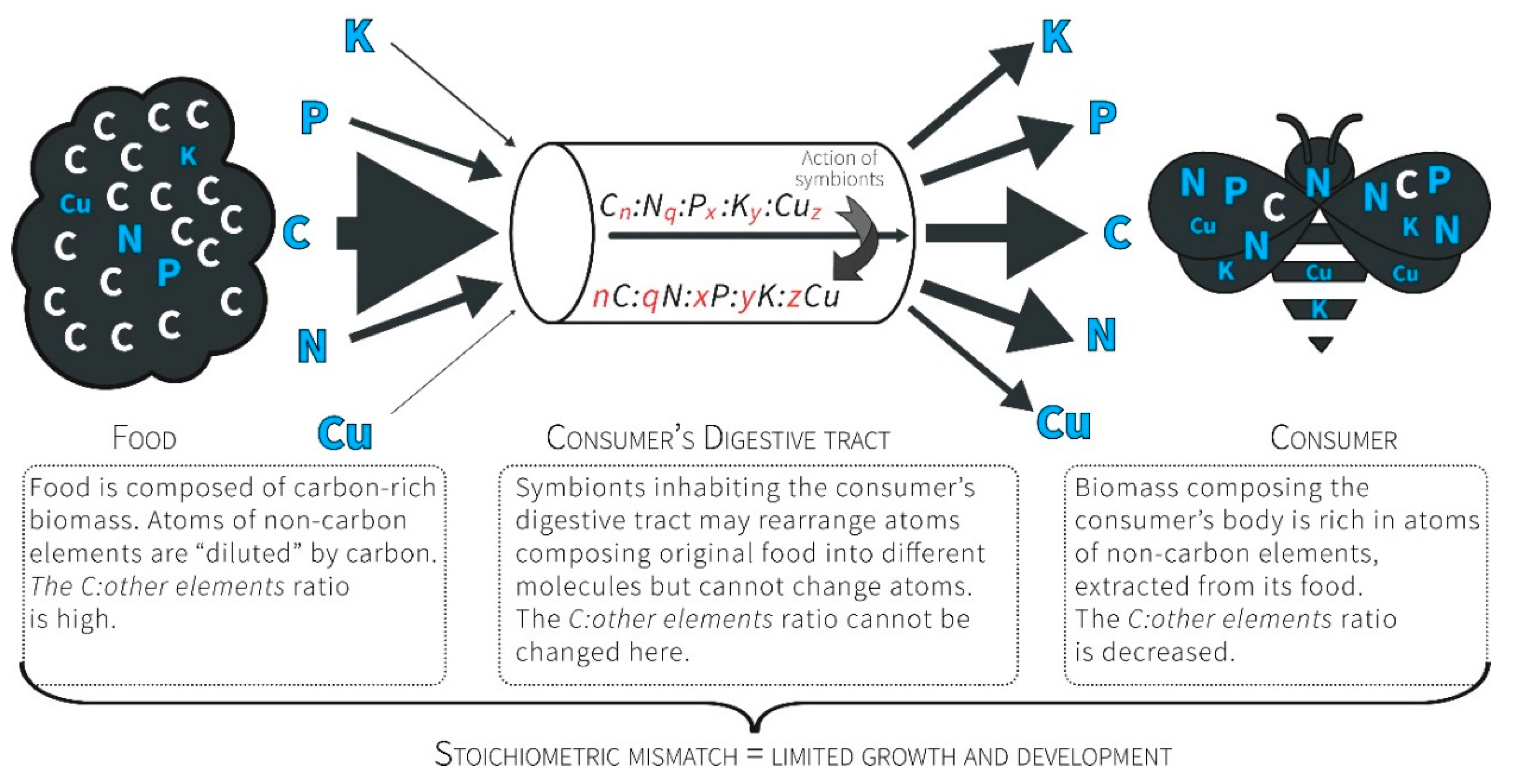
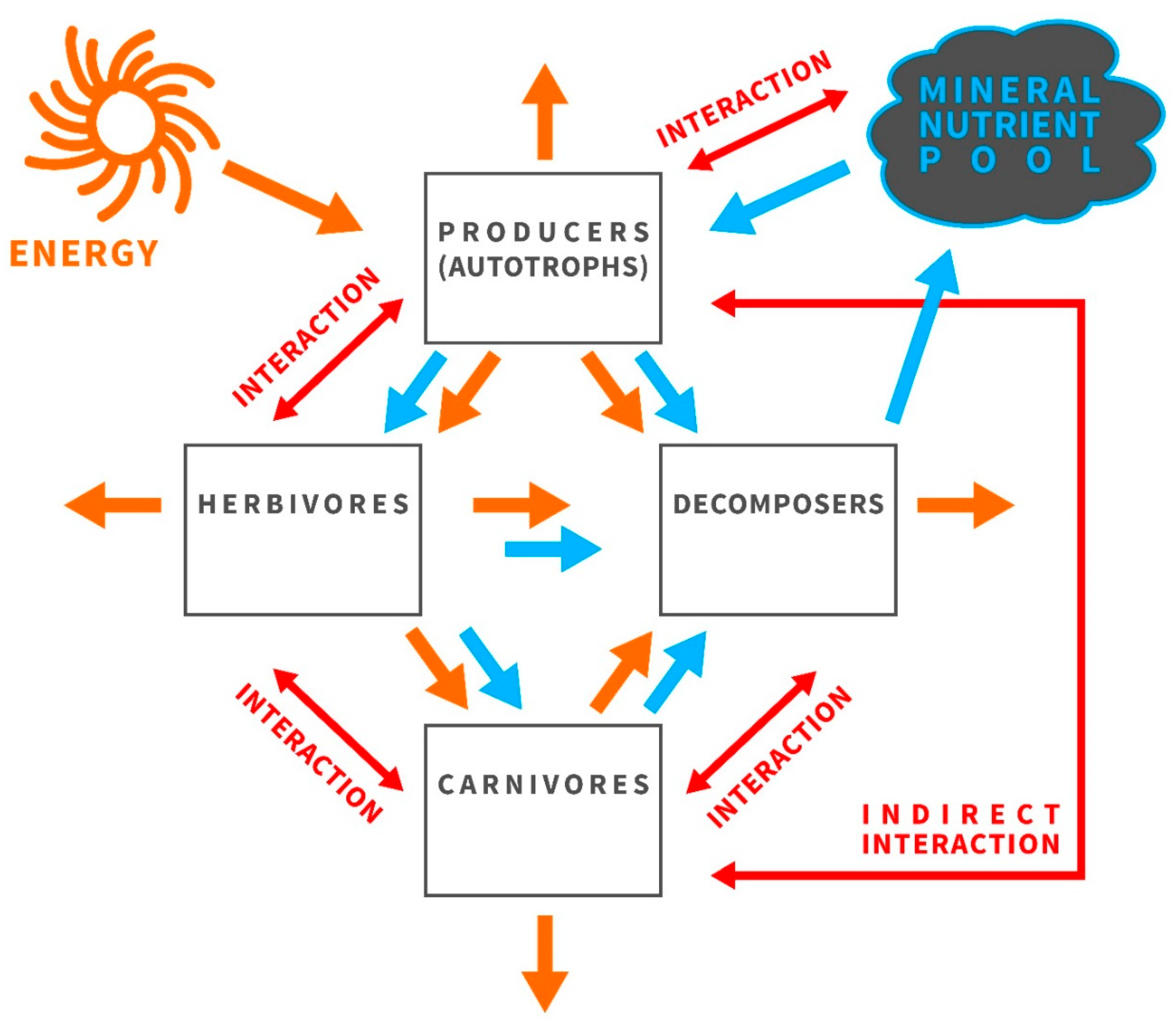
3.1. Ecological Stoichiometry Reflects the Economy of Nature
3.2. Changes in the Floral Composition of Bee Habitats Impact the Nutritional Balance of Bee Larvae Diets, Thereby Shaping Bee Populations
3.3. Stoichiometric Niche
3.4. Sexual Dimorphism in Nutritional Needs May Shape Bee Populations
3.5. Solitary Bee—A Model Organism for Ecological Stoichiometry Studies
4. Conclusions and Avenues for Future Research
- How do various taxa of pollen differ in their stoichiometry?
- How do various species, and different sexes and castes of bees differ in their stoichiometry, and therefore, how different are their nutritional demands? Which scarce elements limit the growth and development of different sexes and castes, and of various species of bees?
- Following (1) and (2), which specific key host plant species that produce stoichiometrically desirable pollen allow bees to balance their diets?
Funding
Acknowledgments
Conflicts of Interest
References
- Giannini, T.C.; Cordeiro, G.D.; Freitas, B.M.; Saraiva, A.M.; Imperatriz-Fonseca, V.L. The dependence of crops for pollinators and the economic value of pollination in Brazil. J. Econ. Entomol. 2015, 108, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.M.; Vaissiere, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D.; Nicholls, E.; Botias, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347. [Google Scholar] [CrossRef] [PubMed]
- Vanbergen, A.J. Threats to an ecosystem service: Pressures on pollinators. Front. Ecol. Environ. 2013, 11, 251–259. [Google Scholar] [CrossRef]
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissière, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- IPEBS. IPBES: The Assessment Report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on Pollinators, Pollination and Food Production; Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services: Bonn, Germany, 2016. [Google Scholar]
- Ollerton, J. Pollinator diversity: Distribution, ecological function, and conservation. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 353–376. [Google Scholar] [CrossRef]
- Roulston, T.A.H.; Goodell, K. The role of resources and risks in regulating wild bee populations. Annu. Rev. Entomol. 2011, 56, 293–312. [Google Scholar] [CrossRef] [PubMed]
- Woodard, S.H.; Jha, S. Wild bee nutritional ecology: Predicting pollinator population dynamics, movement, and services from floral resources. Curr. Opin. Insect Sci. 2017, 21, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Matias, D.M.S.; Borgemeister, C.; von Wehrden, H. Thinking beyond Western commercial honeybee hives: Towards improved conservation of honey bee diversity. Biodivers. Conserv. 2017, 26, 3499–3504. [Google Scholar] [CrossRef]
- Rader, R.; Bartomeus, I.; Garibaldi, L.A.; Garratt, M.P.D.; Howlett, B.G.; Winfree, R.; Cunningham, S.A.; Mayfield, M.M.; Arthur, A.D.; Andersson, G.K.S.; et al. Non-bee insects are important contributors to global crop pollination. Proc. Natl. Acad. Sci. USA 2016, 113, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Geldmann, J.; González-Varo, J.P. Conserving honey bees does not help wildlife. Science 2018, 359, 392–393. [Google Scholar] [CrossRef] [PubMed]
- Wright, G.A.; Nicolson, S.W.; Shafir, S. Nutritional physiology and ecology of honey bees. Annu. Rev. Entomol. 2018, 63, 327–344. [Google Scholar] [CrossRef] [PubMed]
- Vit, P.; Pedro, S.R.M.; Roubik, D.W. (Eds.) Pot-Pollen in Stingless Bee Melittology; Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-61838-8. [Google Scholar]
- Filipiak, M.; Weiner, J. Plant-insect interactions: The role of ecological stoichiometry. Acta Agrobot. 2017, 70. [Google Scholar] [CrossRef]
- Filipiak, M.; Kuszewska, K.; Asselman, M.; Denisow, B.; Stawiarz, E.; Woyciechowski, M.; Weiner, J. Ecological stoichiometry of the honeybee: Pollen diversity and adequate species composition are needed to mitigate limitations imposed on the growth and development of bees by pollen quality. PLoS ONE 2017, 12, e0183236. [Google Scholar] [CrossRef] [PubMed]
- Cripps, C.; Rust, R.W. Pollen preferences of seven osmia species (Hymenoptera: Megachilidae). Environ. Entomol. 1989, 18, 133–138. [Google Scholar] [CrossRef]
- Kraemer, M.E.; Favi, F.D.; Niedziela, C.E. Nesting and pollen preference of Osmia lignaria lignaria (Hymenoptera: Megachilidae) in Virginia and North Carolina Orchards. Environ. Entomol. 2014, 43, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Raw, A. Pollen preferences of three osmia species (Hymenoptera). Oikos 1974, 25. [Google Scholar] [CrossRef]
- Bukovinszky, T.; Rikken, I.; Evers, S.; Wäckers, F.L.; Biesmeijer, J.C.; Prins, H.H.T.; Kleijn, D. Effects of pollen species composition on the foraging behaviour and offspring performance of the mason bee Osmia bicornis (L.). Basic Appl. Ecol. 2016, 18, 21–30. [Google Scholar] [CrossRef]
- Sterner, R.W.; Elser, J.J. Ecological stoichiometry: Overview. In Encyclopedia of Ecology; Jorgensen, S.E., Fath, B.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1101–1116. [Google Scholar]
- Sterner, R.W.; Elser, J.J. Ecological Stoichiometry: The Biology of Elements from Molecules to the Biosphere; Princeton University Press: Princeton, NJ, USA, 2002. [Google Scholar]
- Elser, J.J.; Hamilton, A. Stoichiometry and the new biology: The future is now. PLoS Biol. 2007, 5. [Google Scholar] [CrossRef] [PubMed]
- Kay, A.D.; Ashton, I.W.; Gorokhova, E.; Kerkhoff, A.J.; Liess, A.; Litchman, E. Toward a stoichiometric framework for evolutionary biology. Oikos 2005, 109, 6–17. [Google Scholar] [CrossRef]
- Kay, A.D.; Vrede, T. Ecological stoichiometry: Evolutionary and biochemical aspects. Encycl. Ecol. 2008, 1–5, 1472–1481. [Google Scholar] [CrossRef]
- Jeyasingh, P.D.; Goos, J.M.; Thompson, S.K.; Godwin, C.M.; Cotner, J.B. Ecological stoichiometry beyond redfield: An ionomic perspective on elemental homeostasis. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Cherif, M. Biological stoichiometry: The elements at the heart of biological interactions. In Stoichiometry and Research—The Importance of Quantity in Biomedicine; Innocenti, D.A., Ed.; InTech: Rijeka, Croatia, 2012; pp. 357–376. [Google Scholar]
- Denno, R.F.; Fagan, W.F. Might nitrogen limitation promote omnivory among carnivorous arthropods? Ecology 2003, 84, 2522–2531. [Google Scholar] [CrossRef]
- Jones, R.H.; Flynn, K.J. Nutritional status and diet composition affect the value of diatoms as copepod prey. Science 2005, 307, 1457–1459. [Google Scholar] [CrossRef] [PubMed]
- Slansky, F.; Rodriguez, J.G. Nutritional Ecology of Insects, Mites, Spiders, and Related Invertebrates; Wiley-Interscience: New York, NY, USA, 1987. [Google Scholar]
- Rotheray, E.L.; Osborne, J.L.; Goulson, D. Quantifying the food requirements and effects of food stress on bumble bee colony development. J. Apic. Res. 2017, 56, 288–299. [Google Scholar] [CrossRef]
- Bowers, M.A. Resource availability and timing of reproduction in bumble bee colonies (Hymenoptera: Apidae). Environ. Entomol. 1986, 15, 750–755. [Google Scholar] [CrossRef]
- Riedinger, V.; Mitesser, O.; Hovestadt, T.; Steffan-Dewenter, I.; Holzschuh, A. Annual dynamics of wild bee densities: Attractiveness and productivity effects of oilseed rape. Ecology 2015, 96, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Holzschuh, A.; Dainese, M.; González-Varo, J.P.; Mudri-Stojnić, S.; Riedinger, V.; Rundlöf, M.; Scheper, J.; Wickens, J.B.; Wickens, V.J.; Bommarco, R.; et al. Mass-flowering crops dilute pollinator abundance in agricultural landscapes across Europe. Ecol. Lett. 2016, 19, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Todd, K.J.; Gardiner, M.M.; Lindquist, E.D. Mass flowering crops as a conservation resource for wild pollinators (Hymenoptera: Apoidea). J. Kansas Entomol. Soc. 2016, 89, 158–167. [Google Scholar] [CrossRef]
- Kaspari, M.; Powers, J.S. Biogeochemistry and geographical ecology: Embracing all twenty-five elements required to build organisms. Am. Nat. 2016, 188, S62–S73. [Google Scholar] [CrossRef] [PubMed]
- Sterner, R.W.; Hessen, D.O. Algal nutrient limitation and the nutrition of aquatic herbivores. Annu. Rev. Ecol. Evol. Syst. 1994, 25, 1–29. [Google Scholar] [CrossRef]
- Hessen, D.O.; Elser, J.J.; Sterner, R.W.; Urabe, J. Ecological stoichiometry: An elementary approach using basic principles. Limnol. Oceanogr. 2013, 58, 2219–2236. [Google Scholar] [CrossRef]
- Cherif, M.; Loreau, M. Plant-herbivore-decomposer stoichiometric mismatches and nutrient cycling in ecosystems. Proc. Biol. Sci. 2013, 280, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wilder, S.M.; Jeyasingh, P.D. Merging elemental and macronutrient approaches for a comprehensive study of energy and nutrient flows. J. Anim. Ecol. 2016, 85, 1427–1430. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Jansen, M.; De Meester, L.; Stoks, R. Energy storage and fecundity explain deviations from ecological stoichiometry predictions under global warming and size-selective predation. J. Anim. Ecol. 2016, 85, 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Bujan, J.; Wright, S.J.; Kaspari, M. Biogeochemical drivers of neotropical ant activity and diversity. Ecosphere 2016, 7. [Google Scholar] [CrossRef]
- Schade, J.D.; Kyle, M.; Hobbie, S.E.; Fagan, W.F.; Elser, J.J. Stoichiometric tracking of soil nutrients by a desert insect herbivore. Ecol. Lett. 2003, 6, 96–101. [Google Scholar] [CrossRef]
- Elser, J.J.; Fagan, W.F.; Denno, R.F.; Dobberfuhl, D.R.; Folarin, A.; Huberty, A.; Interlandi, S.; Kilham, S.S.; McCauley, E.; Schulz, K.L.; et al. Nutritional constraints in terrestrial and freshwater food webs. Nature 2000, 408, 578–580. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T. Ecological stoichiometry and the shape of resource-based tradeoffs. Oikos 2006, 112, 406–411. [Google Scholar] [CrossRef]
- Zera, A.J.; Harshman, L.G. The physiology of life history trade-offs in animals. Annu. Rev. Ecol. Evol. Syst. 2001, 32, 95–126. [Google Scholar] [CrossRef]
- Morehouse, N.I.; Nakazawa, T.; Booher, C.M.; Jeyasingh, P.D.; Hall, M.D. Sex in a material world: Why the study of sexual reproduction and sex-specific traits should become more nutritionally-explicit. Oikos 2010, 119, 766–778. [Google Scholar] [CrossRef]
- Boggs, C.L. Understanding insect life histories and senescence through a resource allocation lens. Funct. Ecol. 2009, 23, 27–37. [Google Scholar] [CrossRef]
- Jeyasingh, P.D.; Cothran, R.D.; Tobler, M. Testing the ecological consequences of evolutionary change using elements. Ecol. Evol. 2014, 4, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Frost, P.C.; Evans-White, M.A.; Finkel, Z.V.; Jensen, T.C.; Matzek, V. Are you what you eat? Physiological constraints on organismal stoichiometry in an elementally imbalanced world. Oikos 2005, 109, 18–28. [Google Scholar] [CrossRef]
- Donkersley, P.; Rhodes, G.; Pickup, R.W.; Jones, K.C.; Power, E.F.; Wright, G.A.; Wilson, K. Nutritional composition of honey bee food stores vary with floral composition. Oecologia 2017, 185, 749–761. [Google Scholar] [CrossRef] [PubMed]
- Scheper, J.; Reemer, M.; van Kats, R.; Ozinga, W.A.; van der Linden, G.T.J.; Schaminée, J.H.J.; Siepel, H.; Kleijn, D. Museum specimens reveal loss of pollen host plants as key factor driving wild bee decline in The Netherlands. Proc. Natl. Acad. Sci. USA 2014, 111, 17552–17557. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, D.; Diekötter, T.; Wolters, V.; Jauker, F. Attractiveness of wildflower mixtures for wild bees and hoverflies depends on some key plant species. Insect Conserv. Divers. 2017, 11, 32–41. [Google Scholar] [CrossRef]
- Cole, L.J.; Brocklehurst, S.; Robertson, D.; Harrison, W.; McCracken, D.I. Exploring the interactions between resource availability and the utilisation of semi-natural habitats by insect pollinators in an intensive agricultural landscape. Agric. Ecosyst. Environ. 2017, 246, 157–167. [Google Scholar] [CrossRef]
- Jachuła, J.; Denisow, B.; Wrzesień, M. Validation of floral food resources for pollinators in agricultural landscape in SE Poland. J. Sci. Food Agric. 2017, 98, 2672–2680. [Google Scholar] [CrossRef] [PubMed]
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Tooker, J.F.; Grozinger, C.M. Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc. Natl. Acad. Sci. USA 2016, 113, E4035–E4042. [Google Scholar] [CrossRef] [PubMed]
- Kriesell, L.; Hilpert, A.; Leonhardt, S.D. Different but the same: Bumblebee species collect pollen of different plant sources but similar amino acid profiles. Apidologie 2017, 48, 102–116. [Google Scholar] [CrossRef]
- González, A.L.; Dézerald, O.; Marquet, P.A.; Romero, G.Q.; Srivastava, D.S. The multidimensional stoichiometric niche. Front. Ecol. Evol. 2017, 5, 1–17. [Google Scholar] [CrossRef]
- Goos, J.M.; Cothran, R.D.; Jeyasingh, P.D. Within-population variation in the chemistry of life: The stoichiometry of sexual dimorphism in multiple dimensions. Evol. Ecol. 2017, 31, 635–651. [Google Scholar] [CrossRef]
- Lee, K.P. Sex-specific differences in nutrient regulation in a capital breeding caterpillar, Spodoptera litura (Fabricius). J. Insect Physiol. 2010, 56, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Halvorson, H.M.; Sperfeld, E.; Evans-White, M.A. Quantity and quality limit detritivore growth: Mechanisms revealed by ecological stoichiometry and co-limitation theory. Ecology 2017, 98, 2995–3002. [Google Scholar] [CrossRef] [PubMed]
- Cherif, M.; Faithfull, C.; Guo, J.; Meunier, C.L.; Sitters, J.; Uszko, W.; Vasconcelos, F.R. An operational framework for the advancement of a molecule-to-biosphere stoichiometry theory. Front. Mar. Sci. 2017, 4, 1–16. [Google Scholar] [CrossRef]
- Moe, S.J.; Stelzer, R.S.; Forman, M.R.; Harpole, W.S.; Daufresne, T.; Yoshida, T. Recent advances in ecological stoichiometry: Insights for population and community ecology. Oikos 2005, 109, 29–39. [Google Scholar] [CrossRef]
- Raw, A. The biology of the solitary bee Osmia rufa (L.) (Megachilidae). Trans. R. Entomol. Soc. Lond. 2009, 124, 213–229. [Google Scholar] [CrossRef]
- Ivanov, S.P. The nesting of Osmia rufa (L.) (Hymenoptera, Megachilidae) in the Crimea: Structure and composition of nests. Entomol. Rev. 2006, 86, 524–533. [Google Scholar] [CrossRef]
- Cane, J.H.; Griswold, T.; Parker, F.D. Substrates and materials used for nesting by North American osmia bees (Hymenoptera: Apiformes: Megachilidae). Ann. Entomol. Soc. Am. 2007, 100, 350–358. [Google Scholar] [CrossRef]
- Giejdasz, K.; Fliszkiewicz, M.; Bednárová, A.; Krishnan, N. Reproductive potential and nesting effects of Osmia rufa (syn. bicornis) female (Hymenoptera: Megachilidae). J. Apic. Res. 2016, 60, 75–86. [Google Scholar] [CrossRef]
- Carvell, C.; Roy, D.B.; Smart, S.M.; Pywell, R.F.; Preston, C.D.; Goulson, D. Declines in forage availability for bumblebees at a national scale. Biol. Conserv. 2006, 132, 481–489. [Google Scholar] [CrossRef]
- Winfree, R.; Bartomeus, I.; Cariveau, D.P. Native pollinators in anthropogenic habitats. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 1–22. [Google Scholar] [CrossRef]
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filipiak, M. A Better Understanding of Bee Nutritional Ecology Is Needed to Optimize Conservation Strategies for Wild Bees—The Application of Ecological Stoichiometry. Insects 2018, 9, 85. https://doi.org/10.3390/insects9030085
Filipiak M. A Better Understanding of Bee Nutritional Ecology Is Needed to Optimize Conservation Strategies for Wild Bees—The Application of Ecological Stoichiometry. Insects. 2018; 9(3):85. https://doi.org/10.3390/insects9030085
Chicago/Turabian StyleFilipiak, Michał. 2018. "A Better Understanding of Bee Nutritional Ecology Is Needed to Optimize Conservation Strategies for Wild Bees—The Application of Ecological Stoichiometry" Insects 9, no. 3: 85. https://doi.org/10.3390/insects9030085
APA StyleFilipiak, M. (2018). A Better Understanding of Bee Nutritional Ecology Is Needed to Optimize Conservation Strategies for Wild Bees—The Application of Ecological Stoichiometry. Insects, 9(3), 85. https://doi.org/10.3390/insects9030085




