Influence of Elytral Color Pattern, Size, and Sex of Harmonia axyridis (Coleoptera, Coccinellidae) on Parasite Prevalence and Intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales)
Abstract
:1. Introduction
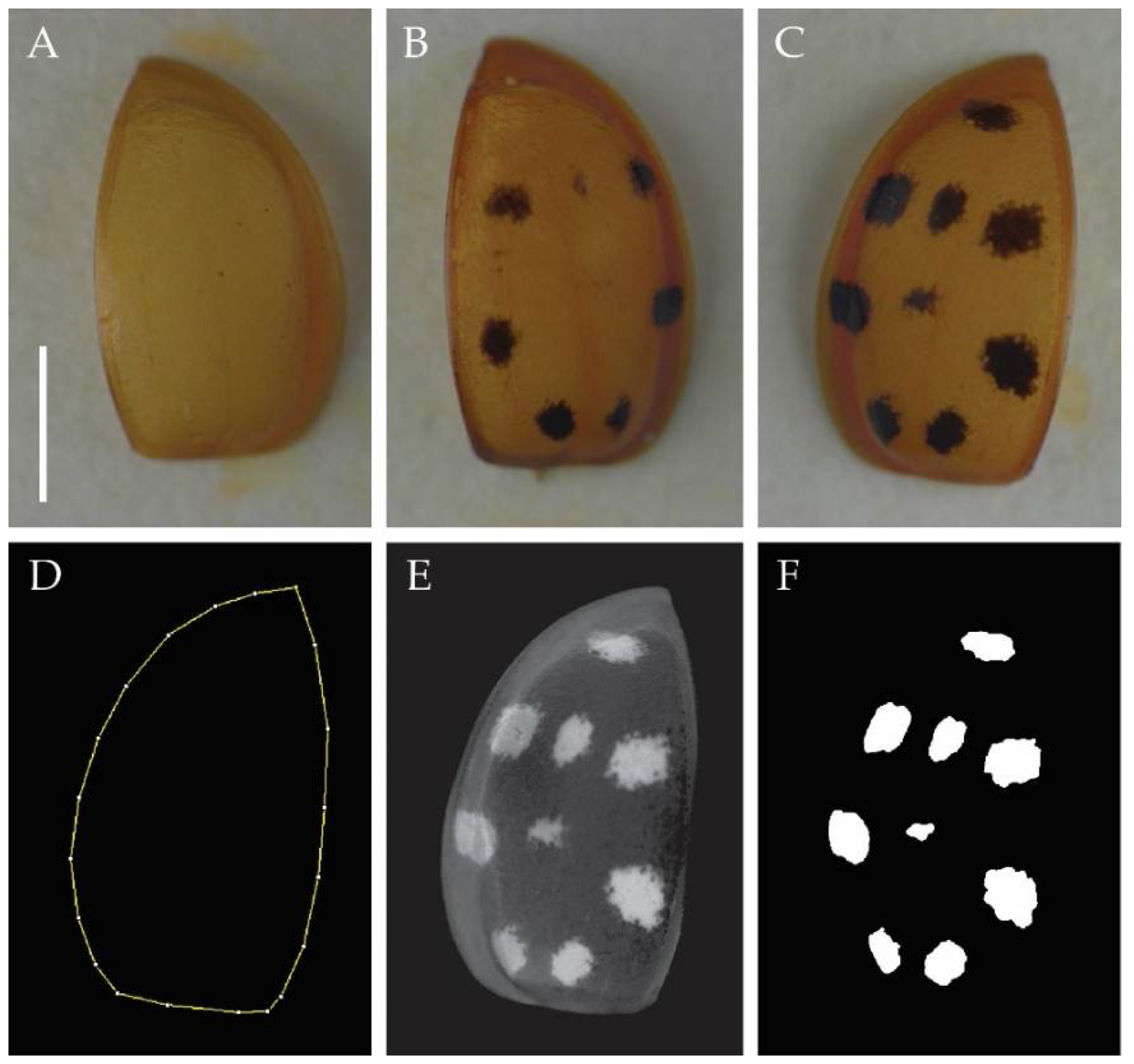
2. Materials and Methods
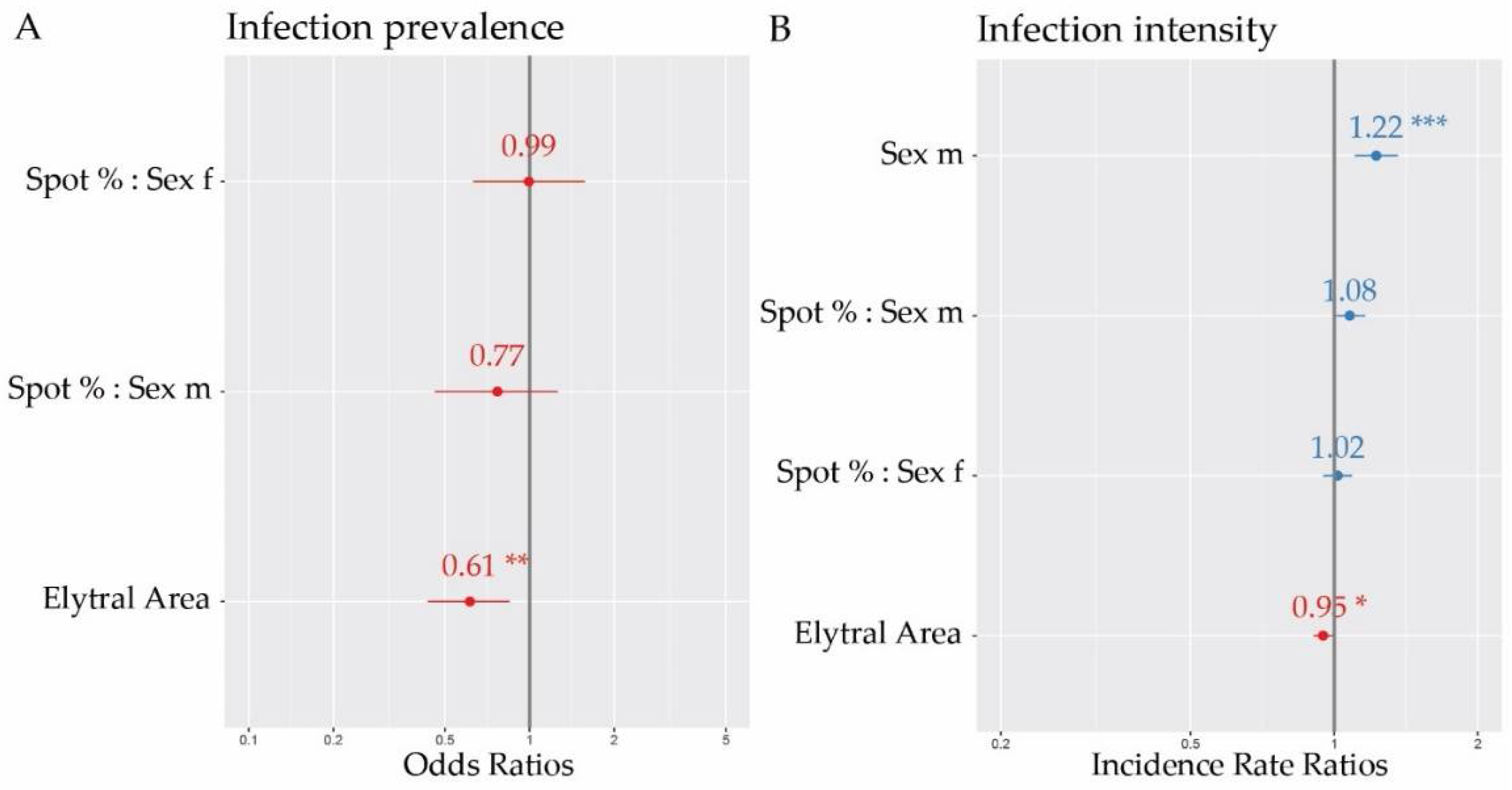
3. Results
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Roy, H.E.; Brown, P.M.J.; Adriaens, T.; Berkvens, N.; Borges, I.; Clusella-Trullas, S.; Comont, R.F.; De Clercq, P.; Eschen, R.; Estoup, A.; et al. The harlequin ladybird, Harmonia axyridis: Global perspectives on invasion history and ecology. Biol. Invasions 2016, 18, 997–1044. [Google Scholar] [CrossRef]
- Brown, P.M.J.; Thomas, C.E.; Lombaert, E.; Jeffries, D.L.; Estoup, A.; Lawson Handley, L.-J. The global spread of Harmonia axyridis (Coleoptera: Coccinellidae): Distribution, dispersal and routes of invasion. Bio. Control 2011, 56, 623–641. [Google Scholar] [CrossRef]
- Nedvěd, O.; Háva, J.; Kulíková, D. Record of the invasive alien ladybird Harmonia axyridis (Coleoptera, Coccinellidae) from Kenya. ZooKeys 2011, 106, 77. [Google Scholar] [CrossRef] [PubMed]
- Nedvěd, O.; Háva, J. New record of the invasive ladybeetle Harmonia axyridis in Afrotropical Region: Tanzania, Zanzibar. Afr. Entomol. 2016, 24, 247–249. [Google Scholar] [CrossRef]
- Kenis, M.; Adriaens, T.; Brown, P.M.; Katsanis, A.; San Martin, G.; Branquart, E.; Maes, D.; Eschen, R.; Zindel, R.; Van Vlaenderen, J. Assessing the ecological risk posed by a recently established invasive alien predator: Harmonia axyridis as a case study. Biol. Control 2017, 62, 341–354. [Google Scholar] [CrossRef]
- Pickering, G.; Lin, J.; Riesen, R.; Reynolds, A.; Brindle, I.; Soleas, G. Influence of Harmonia axyridis on the sensory properties of white and red wine. Am. J. Enol. Vitic. 2004, 55, 153–159. [Google Scholar]
- Haelewaters, D.; Zhao, S.Y.; Clusella-Trullas, S.; Cottrell, T.E.; De Kesel, A.; Fiedler, L.; Herz, A.; Hesketh, H.; Hui, C.; Kleespies, R.G. Parasites of Harmonia axyridis: Current research and perspectives. Biol. Control 2017, 62, 355–371. [Google Scholar] [CrossRef]
- Haelewaters, D.; De Kesel, A. De schimmel Hesperomyces virescens, een natuurlijke vijand van lieveheersbeestjes. Entomol. Ber. 2017, 77, 106–118. [Google Scholar]
- Riddick, E. Influence of host gender on infection rate, density and distribution of the parasitic fungus, Hesperomyces virescens, on the multicolored Asian lady beetle, Harmonia axyridis. J. Insect Sci. 2006, 6. [Google Scholar] [CrossRef] [PubMed]
- Nalepa, C.A.; Weir, A. Infection of Harmonia axyridis (Coleoptera: Coccinellidae) by Hesperomyces virescens (Ascomycetes: Laboulbeniales): Role of mating status and aggregation behavior. J. Invertebr. Pathol. 2007, 94, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Garcés, S.; Williams, R. First record of Hesperomyces virescens Thaxter (Laboulbeniales: Ascomycetes) on Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). J. Kans. Entomol. Soc. 2004, 77, 156–158. [Google Scholar] [CrossRef]
- Ceryngier, P.; Twardowska, K. Harmonia axyridis (Coleoptera: Coccinellidae) as a host of the parasitic fungus Hesperomyces virescens (Ascomycota: Laboulbeniales, Laboulbeniaceae): A case report and short review. Eur. J. Entomol. 2013, 110, 549–557. [Google Scholar] [CrossRef]
- Haelewaters, D.; Minnaar, I.A.; Clusella-Trullas, S. First finding of the parasitic fungus Hesperomyces virescens (Laboulbeniales) on native and invasive ladybirds (Coleoptera, Coccinellidae) in South Africa. Parasite 2016, 23. [Google Scholar] [CrossRef] [PubMed]
- Haelewaters, D.; Comont, R.F.; Zhao, S.Y.; Pfister, D.H. Hesperomyces virescens (Fungi, Ascomycota, Laboulbeniales) attacking Harmonia axyridis (Coleoptera, Coccinellidae) in its native range. Chin. Sci. Bull. 2014, 59, 528–532. [Google Scholar] [CrossRef]
- Raak-van den Berg, C.L.; van Wielink, P.S.; de Jong, P.W.; Gort, G.; Haelewaters, D.; Helder, J.; van Lenteren, J.C. Invasive alien species under attack: natural enemies of Harmonia axyridis in the Netherlands. Biol. Control 2014, 59, 229–240. [Google Scholar] [CrossRef]
- Cottrell, T.E.; Riddick, E.W. Limited transmission of the ectoparasitic fungus Hesperomyces virescens between lady beetles. Psyche. 2012, 2012. [Google Scholar] [CrossRef]
- Kamburov, S.; Nadel, D.; Kenneth, R. Observations on Hesperomyces virescens Thaxter (Laboulbeniales) a fungus associated with premature mortality of Chilocorus bipustulatus L. in Israel. Isr. J. Agric. Res. 1967, 17, 131. [Google Scholar]
- Riddick, E.W. Ectoparasitic mite and fungus on an invasive lady beetle: Parasite coexistence and influence on host survival. Bull. Insectol. 2010, 63, 13–20. [Google Scholar]
- Michie, L.J.; Mallard, F.; Majerus, M.E.N.; Jiggins, F.M. Melanic through nature or nurture: genetic polymorphism and phenotypic plasticity in Harmonia axyridis. J. Evol. Biol. 2010, 23, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Osawa, N.; Nishida, T. Seasonal variation in elytral colour polymorphism in Harmonia axyridis (the ladybird beetle): The role of non-random mating. Heredity 1992, 69. [Google Scholar] [CrossRef]
- Awad, M.; Kalushkov, P.; Karabüyük, F.; Nedvĕd, O. Non-random mating activity of colour morphs of ladybird Harmonia axyridis (Coleoptera: Coccinellidae). Acta Soc. Zool. Bohemicae 2015, 79, 11–17. [Google Scholar]
- Su, W.; Michaud, J.P.; Runzhi, Z.; Fan, Z.; Shuang, L. Seasonal cycles of assortative mating and reproductive behaviour in polymorphic populations of Harmonia axyridis in China. Ecol. Entomol. 2009, 34, 483–494. [Google Scholar] [CrossRef]
- Majerus, M. Ladybirds (New Naturalist Series 81), 1st ed.; Harper Collins Publishers: New York, NY, USA, 1994; pp. 1–367. ISBN 978-0002199353. [Google Scholar]
- Honěk, A. Variability and genetic studies. In Ecology of Coccinellidae; Hodek, I., Honěk, A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1996; pp. 33–60. ISBN 978-94-017-1349-8. [Google Scholar]
- Hodek, I.; Ceryngier, P. Sexual activity in Coccinellidae (Coleoptera): A review. Eur. J. Entomol. 2000, 97, 449–456. [Google Scholar] [CrossRef]
- Purse, B.V.; Comont, R.; Butler, A.; Brown, P.M.; Kessel, C.; Roy, H.E. Landscape and climate determine patterns of spread for all colour morphs of the alien ladybird Harmonia axyridis. J. Biogeogr. 2015, 42, 575–588. [Google Scholar] [CrossRef]
- Komai, T. Genetics of Ladybeetles. In Advances in Genetics; Demerec, M., Ed.; Academic Press: Cambridge, MA, USA, 1956; Volume 8, pp. 155–188. ISBN 978-0-12-017608-3. [Google Scholar]
- Seo, M.J.; Kim, G.H.; Youn, Y.N. Differences in biological and behavioral characteristics of Harmonia axyridis (Coleoptera: Coccinellidae) according to colour patterns of elytra. J. Appl. Entomol. 2008, 132, 239–247. [Google Scholar] [CrossRef]
- Bezzerides, A.L.; McGraw, K.J.; Parker, R.S.; Husseini, J. Elytra color as a signal of chemical defense in the Asian ladybird beetle Harmonia axyridis. Behav. Ecol. Sociobiol. 2007, 61, 1401–1408. [Google Scholar] [CrossRef]
- Röhrich, C.R.; Ngwa, C.J.; Wiesner, J.; Schmidtberg, H.; Degenkolb, T.; Kollewe, C.; Fischer, R.; Pradel, G.; Vilcinskas, A. Harmonine, a defence compound from the harlequin ladybird, inhibits mycobacterial growth and demonstrates multi-stage antimalarial activity. Biol. Lett. 2011. [Google Scholar] [CrossRef] [PubMed]
- Haelewaters, D.; Hiller, T.; Gorczak, M.; Pfister, D.H. Data from “Influence of elytral color pattern, size, and sex of Harmonia axyridis (Coleoptera, Coccinellidae) on parasite prevalence and intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales)”. Available online: https://figshare.com/collections/Data_from_Influence_of_elytral_colour_pattern_size_and_sex_of_Harmonia_axyridis_Coleoptera_Coccinellidae_on_parasite_prevalence_and_intensity_of_Hesperomyces_virescens_Ascomycota_Laboulbeniales_/4103450/1 (accessed on 18 May 2018).
- Collins, T.J. ImageJ for microscopy. Biotechniques 2007, 43, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. Available online: http://www.R-project.org (accessed on 6 March 2018).
- Lüdecke, D. Sjstats: Statistical Functions for Regression Models. R Package Version 0.14.3. 2018. Available online: https://CRAN.R-project.org/package= sjstats (accessed on 17 May 2018).
- Nalepa, C.; Kidd, K.; Ahlstrom, K. Biology of Harmonia axyridis (Coleoptera: Coccinellidae) in winter aggregations. Ann. Entomol. Soc. Am. 1996, 89, 681–685. [Google Scholar] [CrossRef]
- Riddick, E.W.; Schaefer, P.W. Occurrence, density and distribution of parasitic fungus Hesperomyces virescens (Laboulbeniales: Laboulbeniaceae) on multicolored Asian lady beetle (Coleoptera: Coccinellidae). Ann. Entomol. Soc. Am. 2005, 98, 615–624. [Google Scholar] [CrossRef]
- Vilcinskas, A.; Mukherjee, K.; Vogel, H. Expansion of the antimicrobial peptide repertoire in the invasive ladybird Harmonia axyridis. Proc. R. Soc. B 2013, 280. [Google Scholar] [CrossRef] [PubMed]
- Beckert, A.; Wiesner, J.; Baumann, A.; Pöppel, A.-K.; Vogel, H.; Vilcinskas, A. Two c-type lysozymes boost the innate immune system of the invasive ladybird Harmonia axyridis. Dev. Comp. Immunol. 2015, 49, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Evans, E.W. Morphology of invasion: Body size patterns associated with establishment of Coccinella septempunctata (Coleoptera: Coccinellidae) in western North America. Eur. J. Entomol. 2000, 97, 469–474. [Google Scholar] [CrossRef]
- Honěk, A. Factors affecting the distribution of larvae of aphid predators (Col., Coccinellidae and Dipt., Syrphidae) in cereal stands. J. Appl. Entomol. 1983, 95, 336–345. [Google Scholar] [CrossRef]
- Majewski, T. The Laboulbeniales of Poland. Polish Bot. Stud. 1994, 7, 1–466. [Google Scholar]
- Peeters, C.; Molet, M.; Lin, C.-C.; Billen, J. Evolution of cheaper workers in ants: A comparative study of exoskeleton thickness. Biol. J. Linn. Soc. 2017, 121, 556–563. [Google Scholar] [CrossRef]
- Richards, A.G.; Smith, M.N. Infection of cockroaches with Herpomyces (Laboulbeniales) II. Histology and histopathology. Ann. Entomol. Soc. Am. 1956, 49, 85–93. [Google Scholar] [CrossRef]
- Thornhill, R.; Alcock, J. The Evolution of Insect Mating Systems; Harvard University Press: Cambridge, MA, USA, 1983; pp. 1–547. ISBN 0-674-27180-7. [Google Scholar]
- Juliano, S.A. The effects of body size on mating and reproduction in Brachinus lateralis (Coleoptera: Carabidae). Ecol. Entomol. 1985, 10, 271–280. [Google Scholar] [CrossRef]
- Ueno, H.; Sato, Y.; Tsuchida, K. Colour-associated mating success in a polymorphic ladybird beetle, Harmonia axyridis. Funct. Ecol. 1998, 12, 757–761. [Google Scholar] [CrossRef]
- Honek, A.; Dixon, A.F.; Martinkova, Z. Body size, reproductive allocation, and maximum reproductive rate of two species of aphidophagous Coccinellidae exploiting the same resource. Entomol. Exp. Appl. 2008, 127, 1–9. [Google Scholar] [CrossRef]
- Osawa, N. The effect of prey availability on ovarian development and oosorption in the ladybird beetle Harmonia axyridis (Coleoptera: Coccinellidae). Eur. J. Entomol. 2005, 102, 503–511. [Google Scholar] [CrossRef]
| Explanatory Variable | Estimate | Std. Error | z Value | p Value | |
|---|---|---|---|---|---|
| (Intercept) | −0.0486 | 0.1776 | −0.2740 | 0.7843 | |
| Elytral Area | −0.4896 | 0.1700 | −2.8790 | 0.0040 | ** |
| Spot Percentage : Sex f | −0.0061 | 0.2313 | −0.0260 | 0.9791 | |
| Spot Percentage : Sex m | −0.2642 | 0.2558 | −1.0330 | 0.3016 |
| Explanatory Variable | Estimate | Std. Error | z Value | p Value | |
|---|---|---|---|---|---|
| (Intercept) | 3.0004 | 0.0385 | 77.9380 | <2 × 10−16 | *** |
| Sex m | 0.2028 | 0.0524 | 3.8690 | 0.0001 | *** |
| Elytral Area | −0.0534 | 0.0240 | −2.2230 | 0.0262 | * |
| Spot Percentage : Sex f | 0.0165 | 0.0357 | 0.4620 | 0.6438 | |
| Spot Percentage : Sex m | 0.0745 | 0.0383 | 1.9490 | 0.0513 | (*) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haelewaters, D.; Hiller, T.; Gorczak, M.; Pfister, D.H. Influence of Elytral Color Pattern, Size, and Sex of Harmonia axyridis (Coleoptera, Coccinellidae) on Parasite Prevalence and Intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales). Insects 2018, 9, 67. https://doi.org/10.3390/insects9020067
Haelewaters D, Hiller T, Gorczak M, Pfister DH. Influence of Elytral Color Pattern, Size, and Sex of Harmonia axyridis (Coleoptera, Coccinellidae) on Parasite Prevalence and Intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales). Insects. 2018; 9(2):67. https://doi.org/10.3390/insects9020067
Chicago/Turabian StyleHaelewaters, Danny, Thomas Hiller, Michał Gorczak, and Donald H. Pfister. 2018. "Influence of Elytral Color Pattern, Size, and Sex of Harmonia axyridis (Coleoptera, Coccinellidae) on Parasite Prevalence and Intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales)" Insects 9, no. 2: 67. https://doi.org/10.3390/insects9020067
APA StyleHaelewaters, D., Hiller, T., Gorczak, M., & Pfister, D. H. (2018). Influence of Elytral Color Pattern, Size, and Sex of Harmonia axyridis (Coleoptera, Coccinellidae) on Parasite Prevalence and Intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales). Insects, 9(2), 67. https://doi.org/10.3390/insects9020067





