Field Trapping Bactrocera latifrons (Diptera: Tephritidae) with Select Eugenol Analogs That Have Been Found to Attract Other ‘Non-Responsive’ Fruit Fly Species
Abstract
:1. Introduction
2. Materials and Methods
2.1. Lures
2.2. Study Site
2.3. Bioassay
2.4. Statistical Analyses
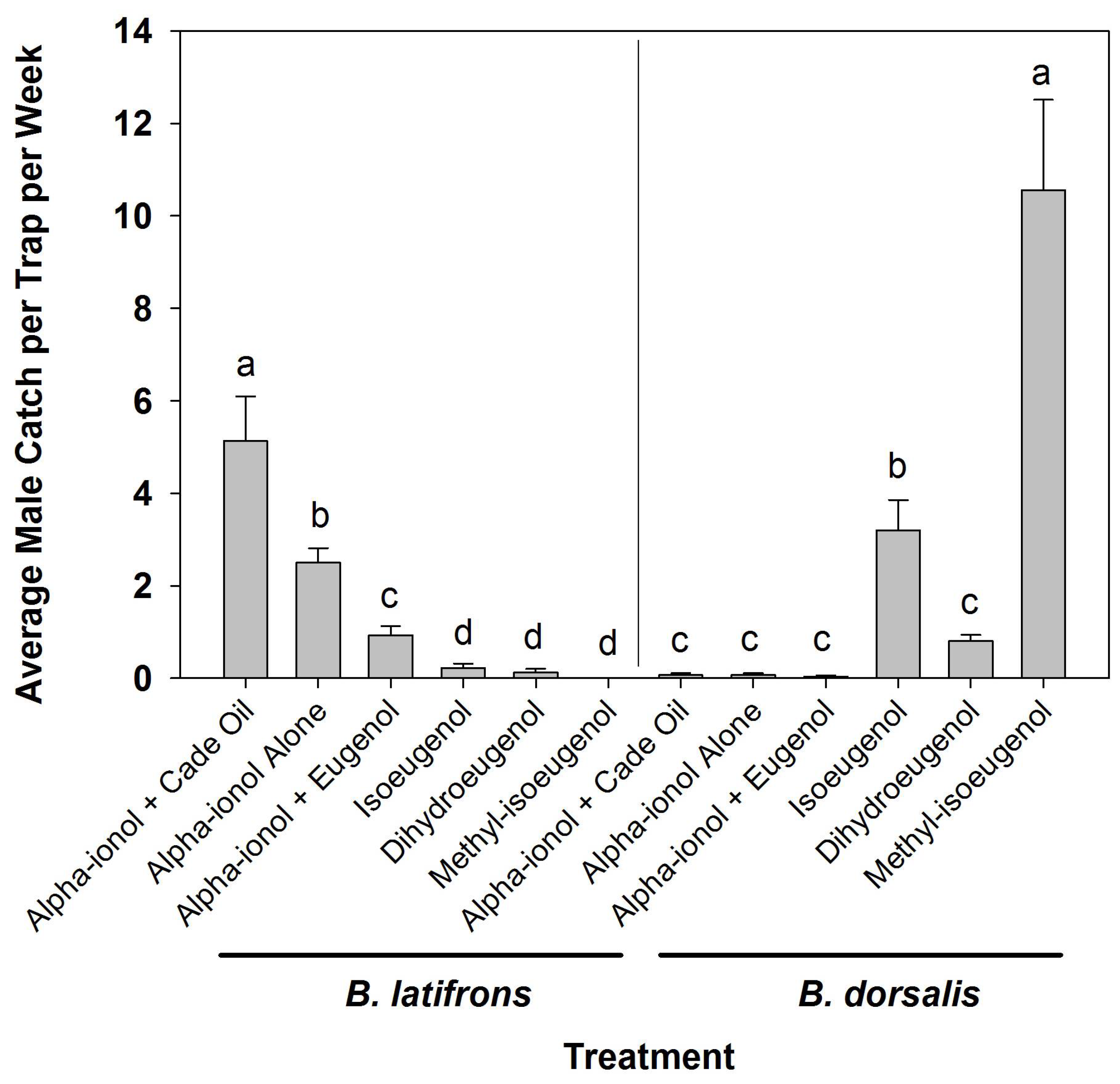
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Schutze, M.K.; Virgilio, M.; Norrbom, A.; Clarke, A.R. Tephritid integrative taxonomy: Where we are now, with a focus on the resolution of three tropical fruit fly complexes. Ann. Rev. Entomol. 2016, 62, 147–164. [Google Scholar] [CrossRef] [PubMed]
- White, I.M.; Elson-Harris, M.M. Fruit Flies of Economic Importance: Their Identification and Bionomics; CAB International: Wallingford, UK, 1992; p. 601. ISBN 0-85198-790-7. [Google Scholar]
- Doorenweerd, C.; Leblanc, L.; Norrbom, A.L.; San Jose, M.; Rubinoff, D. A global checklist of the 932 fruit fly species in the tribe Dacini (Diptera: Tephritidae). ZooKeys 2018, 730, 19–56. [Google Scholar] [CrossRef] [PubMed]
- White, I.M. Morphological features of the Tribe Dacini (Dacinae): Their significance to behavior and classification. In Fruit Flies (Tephritidae): Phylogeny and Evolution of Behavior; Aluja, M., Norrbom, A.L., Eds.; CRC Press LLC: Boca Raton, FL, USA, 2000; pp. 505–533. ISBN 0-8493-1275-2. [Google Scholar]
- Drew, R.A.I. The tropical fruit flies (Diptera: Tephritidae: Dacinae) of the Australasian and Oceanian Regions. Mem. Qld. Mus. 1989, 26, 1–521. [Google Scholar]
- Drew, R.A.I.; Romig, M.C. Pest species in the genera Bactrocera and Dacus. In Tropical Fruit Flies (Tephritidae: Dacinae) of South-East Asia; CAB International: Wallingford, UK, 2013; pp. 408–410. ISBN 978-1-78064-035-8. [Google Scholar]
- Virgilio, M.; Jordaens, K.; Verwimp, C.; White, I.M.; De Meyer, M. Higher phylogeny of frugivorous flies (Diptera, Tephritidae, Dacini): Localized partition conflicts and a novel generic classification. Mol. Phylogenet. Evol. 2015, 85, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Drew, R.A.I.; Hancock, D.L. Phylogeny of the Tribe Dacini (Dacinae) based on morphological, distributional, and biological data. In Fruit Flies (Tephritidae): Phylogeny and Evolution of Behavior; Aluja, M., Norrbom, A.L., Eds.; CRC Press LLC: Boca Raton, FL, USA, 2000; pp. 491–504. ISBN 0-8493-1275-2. [Google Scholar]
- Royer, J.E.; Agovaua, S.; Bokosou, J.; Kurika, K.; Mararuai, A.; Mayer, D.G.; Niangu, B. Responses of fruit flies (Diptera: Tephritidae) to new attractants in Papua New Guinea. Austral Entomol. 2018, 57, 40–49. [Google Scholar] [CrossRef]
- Royer, J.E. Responses of fruit flies (Tephritidae: Dacinae) to novel male attractants in north Queensland, Australia, and improved lures for some pest species. Austral Entomol. 2015, 54, 411–426. [Google Scholar] [CrossRef]
- Fay, H.A.C. Exploring structure-activity relationships in the phenylpropanoids to procure new male lures for non-responsive Bactrocera and Dacus. In Proceedings of the 8th International Symposium on Fruit Flies of Economic Importance (26 September–1 October 2010, Valencia, Spain); Muñoz, B.S., Llopis, V.N., García, A.U., Eds.; Editorial Universtat Politècnica de València: Valencia, Spain, 2010; pp. 270–280. Available online: https://riunet.upv.es/handle/10251/14530 (accessed on 12 April 2018).
- McQuate, G.T.; Liquido, N.J. Annotated world bibliography of host fruits of Bactrocera latifrons (Hendel) (Diptera: Tephritidae). Insecta Mundi 2013, 289, 1–61. Available online: http://centerforsystematicentomology.org/insectamundi/0289McQuateandLiquido.pdf (accessed on 11 January 2018).
- McQuate, G.T.; Liquido, N.J. Host plants of Solanum fruit fly, Bactrocera latifrons (Hendel) (Diptera: Tephritidae). In USDA Compendium of Fruit Fly Host Information, Edition 3.0; Liquido, N.J., McQuate, G.T., Suiter, K.A., Eds.; USDA-APHIS-CPHST: Raleigh, NC, USA, 2017. Available online: https://coffhi.cphst.org/ (accessed on 16 March 2018).
- Liquido, N.J.; Harris, E.J.; Dekker, L.A. Ecology of Bactrocera latifrons (Diptera: Tephritidae) populations: Host plants, natural enemies, distribution, and abundance. Ann. Entomol. Soc. Am. 1994, 87, 71–84. [Google Scholar] [CrossRef]
- Vargas, R.I.; Nishida, T. Survey for Dacus latifrons (Diptera: Tephritidae). J. Econ. Entomol. 1985, 78, 1311–1314. [Google Scholar] [CrossRef]
- Mwatawala, M.W.; De Meyer, M.; White, I.M.; Maerere, A.; Makundi, R.H. Detection of the solanum fruit fly, Bactrocera latifrons (Hendel) in Tanzania (Dipt., Tephritidae). J. Appl. Entomol. 2007, 131, 501–503. [Google Scholar] [CrossRef]
- Papadopoulos, N.T.; Plant, R.E.; Carey, J.R. From trickle to flood: The large-scale, cryptic invasion of California by tropical fruit flies. Proc. R. Soc. B 2013, 280, 20131466. [Google Scholar] [CrossRef] [PubMed]
- McQuate, G.T.; Keum, Y.-S.; Sylva, C.D.; Li, Q.X.; Jang, E.B. Active ingredients in cade oil which synergize the attractiveness of alpha-ionol to male Bactrocera latifrons (Diptera: Tephritidae). J. Econ. Entomol. 2004, 97, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Flath, R.A.; Cunningham, R.T.; Liquido, N.J.; McGovern, T.P. α-Ionol as attractant for trapping Bactrocera latifrons (Diptera: Tephritidae). J. Econ. Entomol. 1994, 87, 1470–1476. [Google Scholar] [CrossRef]
- McQuate, G.T.; Peck, S.L. Enhancement of attraction of alpha-ionol to male Bactrocera latifrons (Diptera: Tephritidae) by addition of a synergist, cade oil. J. Econ. Entomol. 2001, 94, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Bellis, G.A.; Brito, A.A.; De Jesus, H.; Quintao, V.; Sarmento, J.C.; Bere, A.; Rodrigues, J.; Hancock, D.L. A preliminary account of the fruit fly fauna of Timor-Leste (Diptera: Tephritidae: Dacinae). Zootaxa 2017, 4362, 421–432. [Google Scholar] [CrossRef] [PubMed]
- McQuate, G.T.; Jang, E.B.; Siderhurst, M. Detection/Monitoring of Bactrocera latifrons (Diptera: Tephritidae): Assessing the Potential of Prospective New Lures. Proc. Hawaii. Entomol. Soc. 2013, 45, 69–81. [Google Scholar]
- McQuate, G.T.; Bokonon-Ganta, A.H.; Jang, E.B. Use of Alpha-Ionol + Cade Oil for Detection and Monitoring of Bactrocera latifrons (Diptera: Tephritidae) Populations. In Fruit Flies of Economic Importance: From Basic to Applied Knowledge. Proceedings of the 7th International Symposium on Fruit Flies of Economic Importance (10–15 September 2006, Salvador, Bahia, Brazil); Sugayama, R., Zucchi, R.A., Ovruski, S., Sivinski, J., Eds.; SBPC (Sociedade Brasileira para o Progresso da Ciência): São Paulo, Brazil, 2008; pp. 89–95. Available online: http://www.moscamed.org.br/simposium.html (accessed on 11 January 2018).
- USGS. The Universal Transverse Mercator (UTM) Grid. Fact Sheet 077-01 (August 2001); 2001. Available online: https://pubs.usgs.gov/fs/2001/0077/report.pdf (accessed on 5 January 2018).
- Shelly, T.E.; Pahio, E.; Edu, J. Synergistic and inhibitory interactions between methyl eugenol and cue lure influence trap catch of male fruit flies, Bactrocera dorsalis (Hendel) and B. cucurbitae (Diptera: Tephritidae). Fla. Entomol. 2004, 87, 481–486. [Google Scholar] [CrossRef]
- SAS Institute Inc. JMP 11.2.0; SAS Institute Inc.: Cary, NC, USA, 2013. [Google Scholar]
- Metcalf, R.L.; Metcalf, E.R. Plant Kairomones in Insect Ecology and Control; Chapman and Hall: New York, NY, USA, 1992; p. 168. ISBN 0-412-01991-4. [Google Scholar]
- Royer, J.E.; Kahn, M.; Mayer, D.G. Methyl-isoeugenol, a highly attractive male lure for the cucurbit flower pest Zeugodacus diversus (Coquillett) (syn. Bactrocera diversa) (Diptera: Tephritidae: Dacinae). J. Econ. Entomol. 2018, 111. [Google Scholar] [CrossRef] [PubMed]
- Royer, J.E.; Teakle, G.; Mayer, D.G. Methyl-isoeugenol, a significantly more attractive male lure for the methyl eugenol-responsive Pacific fruit fly, Bactrocera xanthodes (Broun) (Diptera: Tephritidae: Dacinae). 2018; manuscript in preparation. [Google Scholar]
- Royer, J.E.; Mille, C.; Cazeres, S.; Brinon, J.; Mayer, D.G. Isoeugenol, a more attractive male lure for the cuelure-responsive pest fruit fly Bactrocera curvipennis (Froggatt) (Tephritidae: Dacinae), and new records of species responding to zingerone in New Caledonia. 2018; manuscript in preparation. [Google Scholar]
- Inskeep, J.; Macquarie University, Sydney, New South Wales, Australia. Personal communication, 2018.
- Enomoto, H.; Ishida, T.; Hamagami, A.; Nishida, R. 3-Oxygenated α-ionone derivatives as potent male attractants for the solanaceous fruit fly, Bactrocera latifrons (Diptera: Tephritidae), and sequestered metabolites in the rectal gland. Appl. Entomol. Zool. 2010, 45, 551–556. [Google Scholar] [CrossRef]
- Nishida, R.; Kyoto University, Kyoto, Kansai, Japan; Tan, K.H.; Tan Hak Heng Co., Ulu Tiram, Johor, Malaysia. Personal communication, 2016.
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McQuate, G.T.; Royer, J.E.; Sylva, C.D. Field Trapping Bactrocera latifrons (Diptera: Tephritidae) with Select Eugenol Analogs That Have Been Found to Attract Other ‘Non-Responsive’ Fruit Fly Species. Insects 2018, 9, 50. https://doi.org/10.3390/insects9020050
McQuate GT, Royer JE, Sylva CD. Field Trapping Bactrocera latifrons (Diptera: Tephritidae) with Select Eugenol Analogs That Have Been Found to Attract Other ‘Non-Responsive’ Fruit Fly Species. Insects. 2018; 9(2):50. https://doi.org/10.3390/insects9020050
Chicago/Turabian StyleMcQuate, Grant T., Jane E. Royer, and Charmaine D. Sylva. 2018. "Field Trapping Bactrocera latifrons (Diptera: Tephritidae) with Select Eugenol Analogs That Have Been Found to Attract Other ‘Non-Responsive’ Fruit Fly Species" Insects 9, no. 2: 50. https://doi.org/10.3390/insects9020050
APA StyleMcQuate, G. T., Royer, J. E., & Sylva, C. D. (2018). Field Trapping Bactrocera latifrons (Diptera: Tephritidae) with Select Eugenol Analogs That Have Been Found to Attract Other ‘Non-Responsive’ Fruit Fly Species. Insects, 9(2), 50. https://doi.org/10.3390/insects9020050




