Insect Artifacts Are More than Just Altered Bloodstains
Abstract
:1. Introduction
2. Insect Artifacts
2.1. Regurgitate
2.2. Defecatory Stains
2.3. Transfer Patterns
2.4. Meconium
2.5. Larval Stains
2.5.1. Secretions
2.5.2. Excretions
3. Methods of Detection
3.1. Visual Methods
3.2. Contextual Methods
3.3. Chemical Methods
4. New Research
4.1. Chemical Analysis Based on Composition of Fly Fluids
4.2. Molecular Methods
4.3. Fly Microbiome
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Rivers, D.B.; Dahlem, G.A. The Science of Forensic Entomology; Wiley-Blackwell: West Sussex, UK, 2014. [Google Scholar]
- Villet, M.H.; Richards, C.S.; Midgley, J.M. Contemporary precision, bias and accuracy of minimum post-mortem intervals estimated using development of carrion-feeding insects. In Current Concepts in Forensic Entomology; Amendt, J., Campobasso, C.P., Goff, M.L., Grassberger, M., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 109–137. [Google Scholar]
- Byrd, J.H.; Castner, J.L. (Eds.) Forensic Entomology: The Utility of Arthropods in Legal Investigations, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Anderson, G.S. Factors that influence insect succession on carrion. In Forensic Entomology: The Utility of Using Arthropods in Legal Investigations, 2nd ed.; Byrd, J.H., Castner, J.L., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 201–250. [Google Scholar]
- Goff, M.L.; Lord, W.D. Insect as toxicological indicator and the impact of drugs and toxin on insect development. In Forensic Entomology: The Utility of Using Arthropods in Legal Investigations, 2nd ed.; Byrd, J.H., Castner, J.L., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 427–434. [Google Scholar]
- Kulstein, G.; Amendt, J.; Zehner, R. Blow fly artifacts from blood and putrefaction fluid on various surfaces: A source for forensic STR typing. Entomol. Exp. Appl. 2015, 157, 255–262. [Google Scholar] [CrossRef]
- LaGoo, L.; Schaeffer, L.S.; Szymanski, D.W.; Smith, R.W. Detection of gunshot residue in blowfly larvae and decomposing porcine tissue using inductively coupled plasma mass spectrometry (ICP-MS). J. Forensic Sci. 2010, 55, 624–632. [Google Scholar] [CrossRef] [PubMed]
- James, S.H.; Nordby, J.J.; Bell, S. (Eds.) Forensic Science: An Introduction to Scientific and Investigative Techniques, 4th ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Fujikawa, A.; Barksdale, L.; Carter, D.O. Calliphora vicina (Diptera: Calliphoridae) and their ability to alter the morphology and presumptive chemistry of bloodstain patterns. J. Forensic Ident. 2009, 59, 502–512. [Google Scholar]
- Fujikawa, A.; Barskdale, L.; Higley, L.G.; Carter, D.O. Changes in the morphology and presumptive chemistry of impact and pooled bloodstain patterns by Lucilia sericata (Meigen) (Diptera: Calliphoridae). J. Forensic Sci. 2011, 56, 1315–1318. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.A.; Benecke, M.; Byrd, J.H.; Hawkes, R.; Brown, R. Entomological alteration of bloodstain evidence. In Forensic Entomology: The Utility of Using Arthropods in Legal Investigations, 2nd ed.; Byrd, J.H., Castner, J.L., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 539–580. [Google Scholar]
- Rivers, D.B.; McGregor, A. Morphological features of regurgitate and defecatory stains deposited by five species of necrophagous flies are influenced by adult diets and body size. J. Forensic Sci. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Benecke, M.; Barksdale, L. Distinction of bloodstain patterns from fly artifacts. Forensic Sci. Int. 2003, 137, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Durdle, A.; Mitchell, R.J.; van Oorschot, R.A.H. The human DNA content in artifacts deposited by the blowfly Lucilia cuprina fed human blood, semen, and saliva. Forensic Sci. Int. 2013, 233, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Durdle, A.; Mitchell, R.J.; van Oorschot, R.A.H. The use of forensic tests to distinguish blowfly artifacts from human blood, semen, and saliva. J. Forensic Sci. 2015, 60, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Durdle, A.; Mitchell, R.J.; van Oorschot, R.A.H. The change in human DNA content over time in the artefacts of the blowfly Lucilia cuprina (Meigen) (Diptera: Calliphoridae). Forensic Sci. Int. 2011, 3, e289–e290. [Google Scholar] [CrossRef]
- Striman, B.; Fujikawa, A.; Barksdale, L.; Carter, D.O. Alteration of expirated bloodstain patterns by Calliphora vicina and Lucilia sericata (Diptera: Calliphoridae) through ingestion and deposition of artifacts. J. Forensic Sci. 2011, 53, S123–S127. [Google Scholar] [CrossRef] [PubMed]
- Scientific Working Group on Bloodstain Pattern Analysis (SWGSTAIN). Recommended Terminology; I.A.B.P.A. Newsletter: 2008. Available online: http://iabpa.org/uploads/files/iabpa%20publications/June%202008%20News.pdf (accessed on 10 February 2017).
- Durdle, A.; van Oorschot, R.A.H.; Mitchell, R.J. The morphology of fecal and regurgitation artifacts deposited by the blow fly Lucilia cuprina fed a diet of human blood. J. Forensic Sci. 2013, 58, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Zuhu, R.M.; Supriyani, M.; Omar, B. Fly artifact documentation of Chrysomya megacephala (Fabricius) (Diptera: Calliphoridae)—A forensically important blowfly species in Malaysia. Trop. Biomed. 2008, 25, 17–22. [Google Scholar]
- Stoffolano, J.G.; Haselton, A.T. The adult dipteran crop: A unique and overlooked organ. Annu. Rev. Entomol. 2013, 58, 205–225. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, B.; Zunich, J.C. Entomology and the Law; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Stoffolano, J.G.; Acaron, A.; Conway, M. “Bubbling” or droplet regurgitation in both sexes of adult Phormia regina (Diptera: Calliphoridae) fed various concentrations of sugar and protein solutions. Ann. Entomol. Soc. Am. 2008, 101, 964–970. [Google Scholar] [CrossRef]
- James, S.H.; Sutton, T.P. Medium- and high-velocity impact blood spatter. In Interpretation of Bloodstain Evidence at Crime Scenes, 2nd ed.; James, S.H., Eckert, W.G., Eds.; CRC Press: Boca Raton, FL, USA, 1998; pp. 59–83. [Google Scholar]
- Benecke, M.; Barksdale, L. In response to: “Commentary on: Mark Benecke and Larry Barksdale, Distinction of bloodstain patterns from fly artifacts: Forensic Science International 137 (2003) 152–159” [Forensic Sci. Int. 149 (2/3) (2005) 293–294]. Forensic Sci. Int. 2007. [Google Scholar] [CrossRef] [PubMed]
- Rivers, D.B.; Acca, G.; Fink, M.; Brogan, R.; Schoeffield, A. Spatial characterization of proteolytic enzyme activity in the foregut region of the adult necrophagous fly, Protophormia terraenovae. J. Insect Physiol. 2014, 67, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Langer, S.V.; Illes, M. Confounding factors of fly artefacts in bloodstain pattern analysis. Can. Soc. Forensic Sci. J. 2015. [Google Scholar] [CrossRef]
- Chapman, R.F. The Insects: Structure and Function, 4th ed.; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Wigglesworth, V.B. The Principles of Insect Physiology, 7th ed.; Chapman and Hall: London, UK, 1982. [Google Scholar]
- Evans, W.A.L. Studies on the digestive enzymes of the blowfly Calliphora erythrocephala: I. The carbohydrates. Exp. Parasitol. 1956, 5, 191–206. [Google Scholar] [CrossRef]
- Terra, W.R.; Ferreira, C. Insect digestive enzymes: Properties, compartmentalization and function. Comp. Biochem. Physiol. B 1994, 109, 1–62. [Google Scholar] [CrossRef]
- Weiss, M.R. Defecation behavior and ecology of insects. Annu. Rev. Entomol. 2006, 51, 635–661. [Google Scholar] [CrossRef] [PubMed]
- O'Donnell, M.J.; Maddrell, S.H.P.; Gardiner, B.O.C. Transport of uric acid by the Malpighian tubules of Rhodnius prolixus and other insects. J. Exp. Biol. 1983, 103, 169–184. [Google Scholar]
- Rivers, D.B.; Thompson, C.; Brogan, R. Physiological trade-offs of forming maggot masses by necrophagous flies on vertebrate carrion. Bull. Entomol. Res. 2011, 101, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Casu, R.E.; Eisemann, C.H.; Vuocolo, T.; Tellam, R.L. The major excretory/secretory protease from Lucilia cuprina larvae is also a gut digestive protease. Int. Parasitol. 1996, 26, 623–628. [Google Scholar] [CrossRef]
- Vistnes, L.M.; Lee, R.; Ksander, G.A. Proteolytic activity of blowfly larvae secretions in experimental burns. Surgery 1981, 90, 835–841. [Google Scholar] [PubMed]
- Chambers, L.; Woodrow, S.; Brown, A.P.; Harris, P.D.; Philips, D.; Hall, M.; Church, J.C.T.; Pritichard, D.I. Degradation of extracellular matrix components by defined proteinases from the greenbottle larvae Lucilia sericata used for the clinical debridement of non-healing wounds. Br. J. Dermatol. 2003, 148, 14–23. [Google Scholar] [CrossRef]
- Pinilla, Y.T.; Moreno-Perez, D.A.; Patarroyo, M.A.; Bello, F.J. Proteolytic activity regarding Sarconesiopsis magellanica (Diptera: Calliphoridae) larval excretions and secretions. Acta Trop. 2013, 128, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Ziffren, S.E.; Heist, H.E.; May, S.C.; Womack, N.A. The secretion of collagenase by maggots and its implication. Ann. Surg. 1953, 138, 932–934. [Google Scholar] [CrossRef] [PubMed]
- Kerridge, A.; Lappin-Scott, H.; Stevens, J.R. Antibacterial properties of larval secretions of the blowfly, Lucilia sericata. Med. Vet. Entomol. 2005, 19, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Bexfield, A.; Nigam, Y.; Thomas, S.; Ratcliffe, N.A. Detection and partial characterisation of two antibacterial factors from the excretions/secretions of the medicinal maggot Lucilia sericata and their activity against methicillin-resistant Staphylococcus aureus (MRSA). Microbes Infect. 2004, 6, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- Poppel, A.-K.; Vogel, H.; Wiesner, J.; Vilcinskas, A. Antimicrobial peptides expressed in medicinal maggots of the blow fly Lucilia sericata show combinatorial activity against bacteria. Antimicrob. Agents Chemother. 2015, 59, 2508–2514. [Google Scholar] [CrossRef] [PubMed]
- Čeřovský, V.; Žďárek, J.; Fučík, V.; Monincová, L.; Voburka, Z.; Bém, R. Lucifensin, the long-sought antimicrobial factor of medicinal maggots of the blowfly Lucilia sericata. Cell. Mole. Life Sci. 2010, 67, 455–466. [Google Scholar] [CrossRef] [PubMed]
- El Shazely, B.; Veverka, V.; Fučík, V.; Voburka, Z.; Žďárek, J.; Čeřovský, V. Lucifensin II, a defensin of medicinal maggots of the blowfly Lucilia cuprina (Diptera: Calliphoridae). J. Med. Entomol. 2013, 50, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, B. Flies as forensic indicators. J. Med. Entomol. 1991, 28, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Christopherson, C.; Gibo, D.L. Foraging by food deprived larvae of Neobellieria bullata (Diptera: Sarcophagidae). J. Forensic Sci. 1997, 42, 71–73. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, B. Model for destruction of bacteria in the midgut of blow fly maggots. J. Med. Entomol. 1968, 5, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, V.H. Excretion of ammonia by Lucilia cuprina larvae suppresses immunity in sheep. Vet. Immunol. Immunopathol. 1997, 56, 311–317. [Google Scholar] [CrossRef]
- Fraenkel, G.; Bhaskaran, G. Pupariation and pupation in cyclorrhaphous flies (Diptera): Terminology and interpretation. Ann. Entomol. Soc. Am. 1973, 66, 418–422. [Google Scholar] [CrossRef]
- Greenberg, B. Behavior of postfeeding larvae of some Calliphoridae and a muscid (Diptera). Ann. Entomol. Soc. Am. 1990, 83, 1210–1214. [Google Scholar] [CrossRef]
- Hobson, R.P. On an enzyme from blow-fly larvae (Lucilia sericata) which digests collagen in alkaline solution. Biochem. J. 1931, 25, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, D.F.; Irzkiewicz, H. An examination of proteolytic enzymes from several insects for collagenase activity. J. Insect Physiol. 1957, 1, 18–22. [Google Scholar] [CrossRef]
- Pendola, S.; Greenberg, B. Substrate-specific analysis of proteolytic enzymes in the larval midgut of Calliphora vicina. Ann. Entomol. Soc. Am. 1975, 68, 341–345. [Google Scholar] [CrossRef]
- Espinoza-Fuentes, F.P.; Terra, W.R. Physiological adaptations for digesting bacteria. Water fluxes and distribution of digestive enzymes in Musca domestica larval midgut. Insect Biochem. 1987, 17, 809–817. [Google Scholar] [CrossRef]
- Ristenblatt, R.R., III; Pizzola, P.A.; Shaler, R.C.; Sorkin, L.N. Commentary on: Mark Benecke and Larry Barksdale, Distinction of bloodstain patterns from fly artifacts. Forensic Sci. International. 137 (2003) 152–159. Forensic Sci. Int. 2005, 14, 293–294. [Google Scholar]
- Bevel, T.; Gardner, R. Bloodstain Pattern Analysis: With an Introduction to Crime Scene Reconstruction, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Li, R. Forensic Biology; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Dametto, M.; David, A.P.; Azzolini, S.S.; Campos, I.T.N.; Tanaka, A.M.; Gomes, A.; Andreotti, R.; Tanaka, A.S. Purification and characterization of a trypsin-like enzyme with fibrinolytic activity present in the abdomen of horn fly, Haematobia irritans irritans (Diptera: Muscidae). J. Protein Chem. 2000, 19, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Terra, W.R. Physiology and biochemistry of insect digestion: An evolutionary perspective. Braz. J. Med. Biol. Res. 1988, 21, 675–734. [Google Scholar] [PubMed]
- Padilha, M.H.P.; Pimentel, A.C.; Ribeiro, A.F.; Terra, W.R. Sequence and function of lysosomal and digestive cathepsin D-like proteinases of Musca domestica midgut. Insect Biochem. Mol. Biol. 2009, 39, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Barrilas-Mury, C.V.; Noriega, F.G.; Wells, M.A. Early trypsin activity is part of the signal transduction system that activates transcription of the late trypsin gene in the midgut of the mosquito, Aedes aegypti. Insect Biochem. Mol. Biol. 1995, 35, 241–246. [Google Scholar] [CrossRef]
- Young, E.G.; Wentworth, H.P.; Hawkins, W.W. The absorption and excretion of allantoin in mammals. J. Pharmacol. Exp. Ther. 1944, 81, 1–9. [Google Scholar]
- Fujiwara, S.; Noguchi, T. Degradation of purines: Only ureidoglycollate lyase out of four allantoin-degrading enzymes is present in mammals. Biochem. J. 1995, 312 Pt 1, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Elkins, K.M. Forensic DNA Biology: A Laboratory Manual; Academic Press: New York, NY, USA, 2013. [Google Scholar]
- Young, E.G.; Conway, C.F. On the estimation of allantoin by the Rimini-Schruver reaction. J. Biol. Chem. 1942, 142, 839–853. [Google Scholar]
- Li, H.; Jasper, H. Gastrointestinal stem cells in health and disease: From flies to humans. Dis. Model Mech. 2016, 9, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Buchon, N.; Broderick, N.A.; Lemaitre, B. Gut homeostasis in a microbial world: Insights from Drosophila melanogaster. Nat. Rev. Microbiol. 2013, 11, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Tauc, H.M.; Tasdogan, A.; Pandur, P. Isolating intestinal stem cells from adult Drosophila midguts by FACS to study stem cell behavior during aging. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Patel, P.H.; Kohlmaier, A.; Grenley, M.O.; McEwen, D.G.; Edgar, B.A. Cytokine/Jak/Stat signaling mediates regeneration and homeostasis in the Drosophila midgut. Cell 2009, 137, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Edgar, B.A. Intestinal stem cells in the adult Drosophila midgut. Exp. Cell Res. 2011, 317, 2780–2788. [Google Scholar] [CrossRef] [PubMed]
- Dowd, S.E.; Callaway, T.R.; Wolcott, R.D.; Sun, Y.; McKeehan, T.; Hagevoort, R.G.; Edrington, T.S. Evaluation of the bacterial diversity in the feces of cattle using 16S rDNA bacterial tag-encoded FLX amplicon pyrosequencing (bTEFAP). BMC Microbiol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Crippen, T.L.; Zheng, L.; Fields, A.T.; Yu, Z.; Ma, Q.; Wood, T.K.; Dowd, S.E.; Flores, M.; Tomberlin, J.K.; et al. A metagenomic assessment of the bacteria associated with Lucilia sericata and Lucilia cuprina (Diptera: Calliphoridae). Appl. Microbiol. Biotechnol. 2015, 99, 869–883. [Google Scholar] [CrossRef] [PubMed]
- Javan, G.T.; Finley, S.J.; Can, I.; Wilkinson, J.E.; Hanson, J.D.; Tarone, A.M. Human thanatomicrobiome succession and the time since death. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
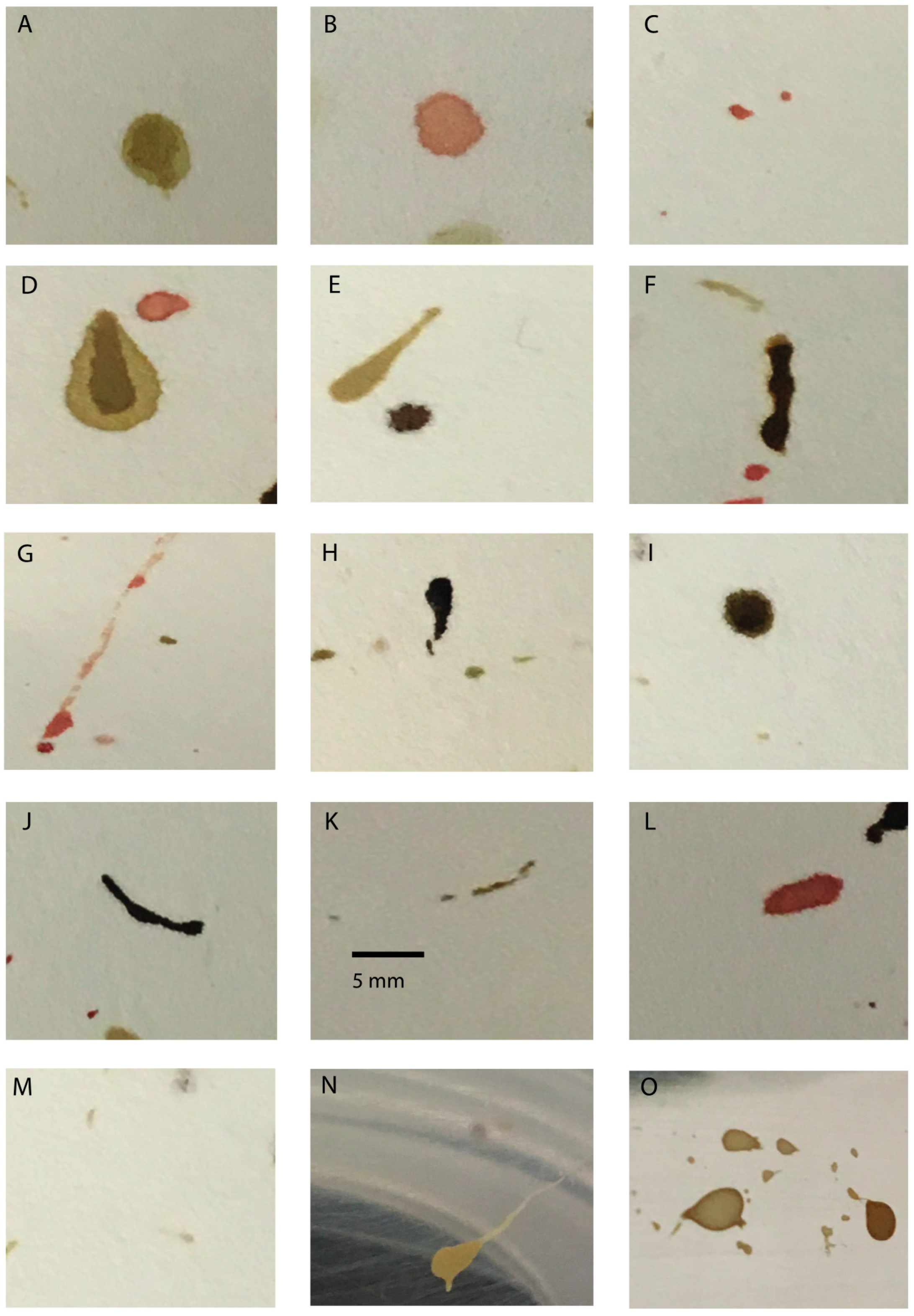
| Stain Type | Shape | Color | Dimensions * |
|---|---|---|---|
| Regurgitate | Area in mm3 | ||
| round to asymmetrically round, occasionally with small tails to form tear drop shape | highly variable and dependent on food source. May be clear, red, green, gray tan/light, dark brown/black | 3.0–16.2 (human blood) | |
| 1.9–19.2 (bovine blood) | |||
| Defecatory | Tail length (mm) | ||
| round to asymmetrically round, some possessing long tails that form tad pole, tear-drop, and sperm-like shapes | highly variable and dependent on food source. May appear creamy, dark brown/black, gray, tan/light # | 3.0–16.2 (human blood) | |
| 1.9–19.2 (bovine blood) | |||
| 4.8–9.2 (chicken blood) + | |||
| Translocation | Stain length (mm) | ||
| asymmetrically linear | same as food source | 0.98–10.4 (human blood) | |
| 0.74–9.7 (bovine blood) | |||
| Tarsal tracks | small and round, impressions of tarsi or pulvilli | same as food source | >0.2 mm in diameter |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivers, D.; Geiman, T. Insect Artifacts Are More than Just Altered Bloodstains. Insects 2017, 8, 37. https://doi.org/10.3390/insects8020037
Rivers D, Geiman T. Insect Artifacts Are More than Just Altered Bloodstains. Insects. 2017; 8(2):37. https://doi.org/10.3390/insects8020037
Chicago/Turabian StyleRivers, David, and Theresa Geiman. 2017. "Insect Artifacts Are More than Just Altered Bloodstains" Insects 8, no. 2: 37. https://doi.org/10.3390/insects8020037
APA StyleRivers, D., & Geiman, T. (2017). Insect Artifacts Are More than Just Altered Bloodstains. Insects, 8(2), 37. https://doi.org/10.3390/insects8020037






