Impacts of Dietary Phytochemicals in the Presence and Absence of Pesticides on Longevity of Honey Bees (Apis mellifera)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Insects
2.2. Effects of Dietary Protein, Phytochemicals and Pesticides on Longevity
2.3. Effects of Dietary Protein, Phytochemicals and Pesticides on Diet Consumption
2.4. Statistics
3. Results
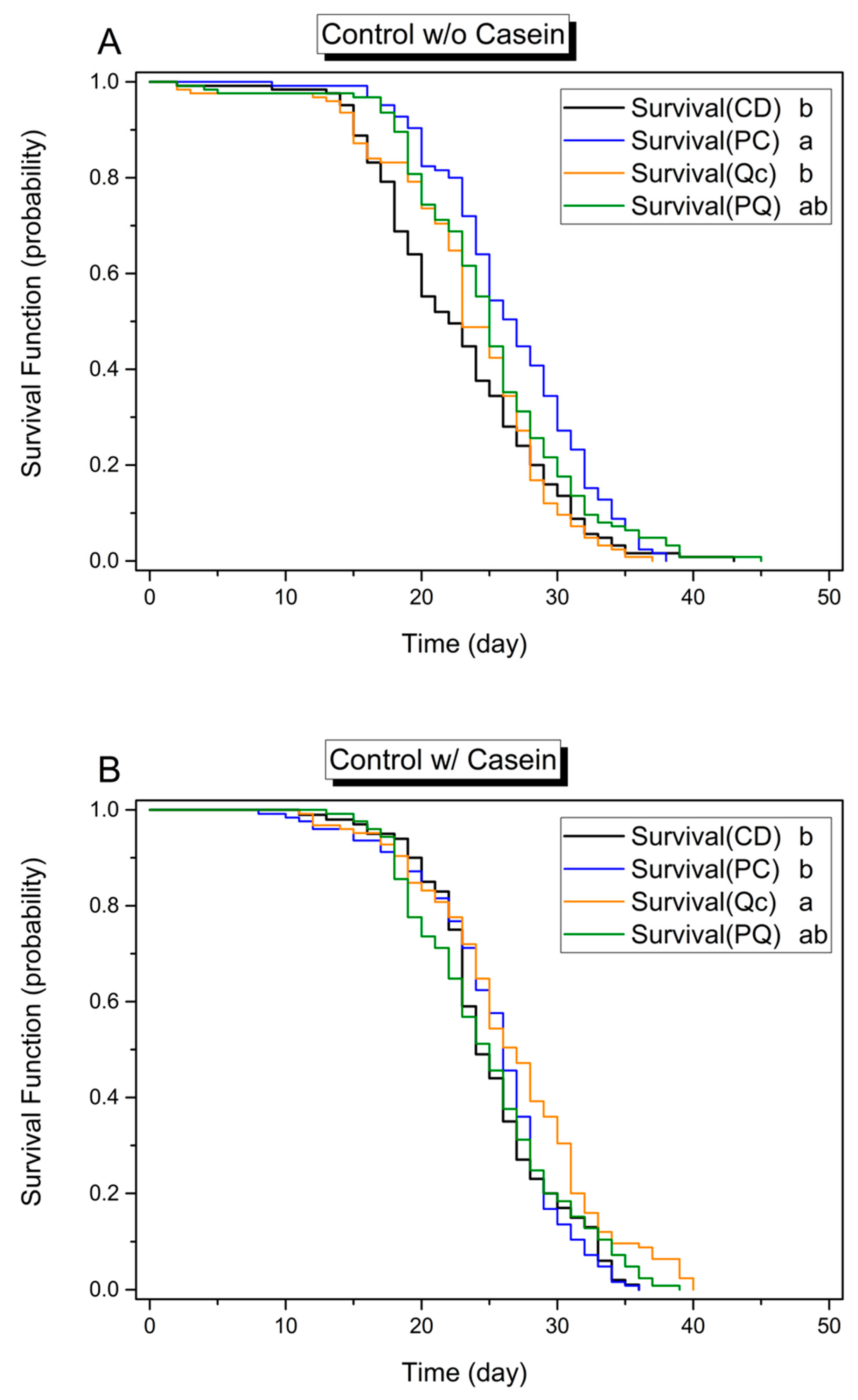
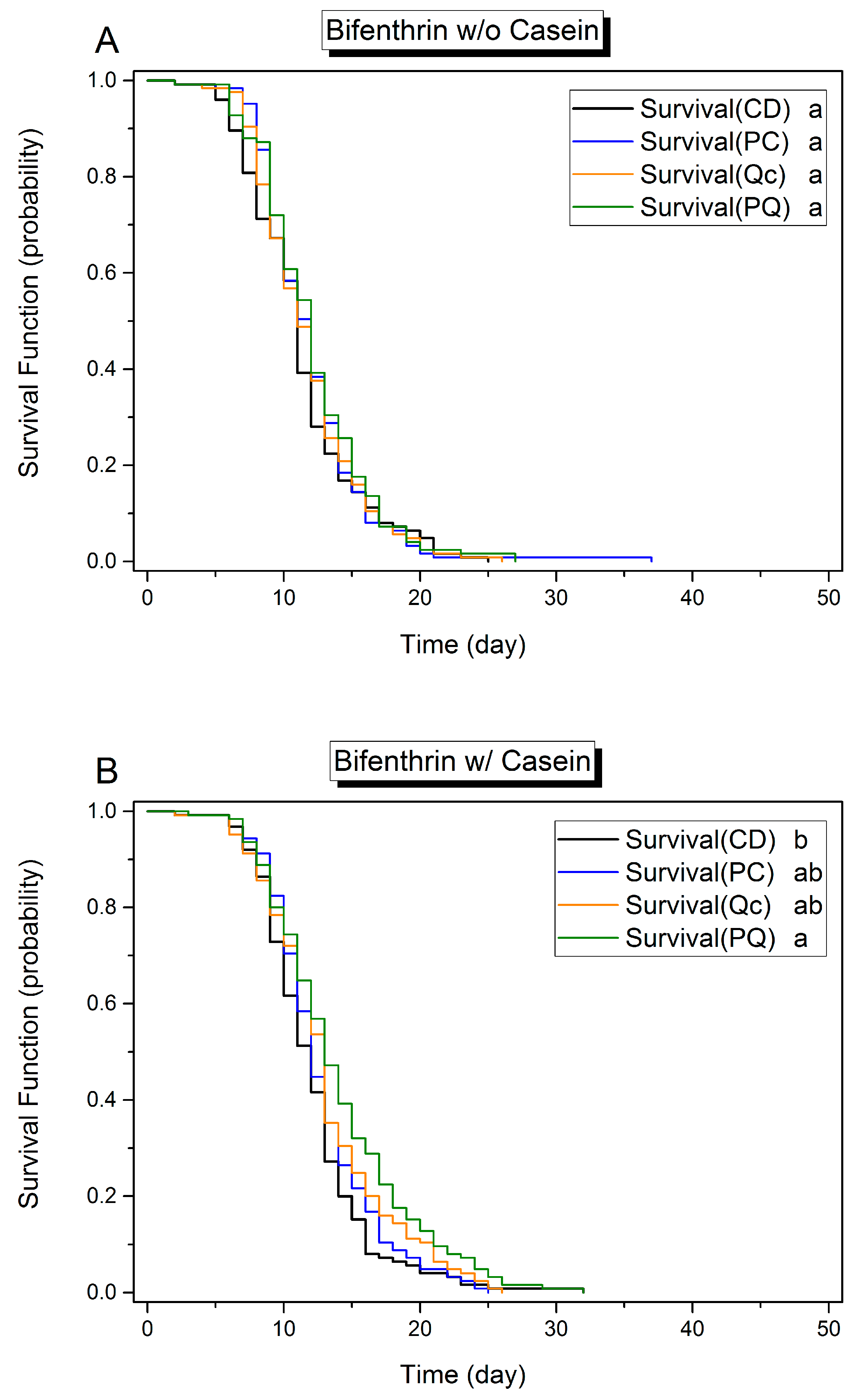
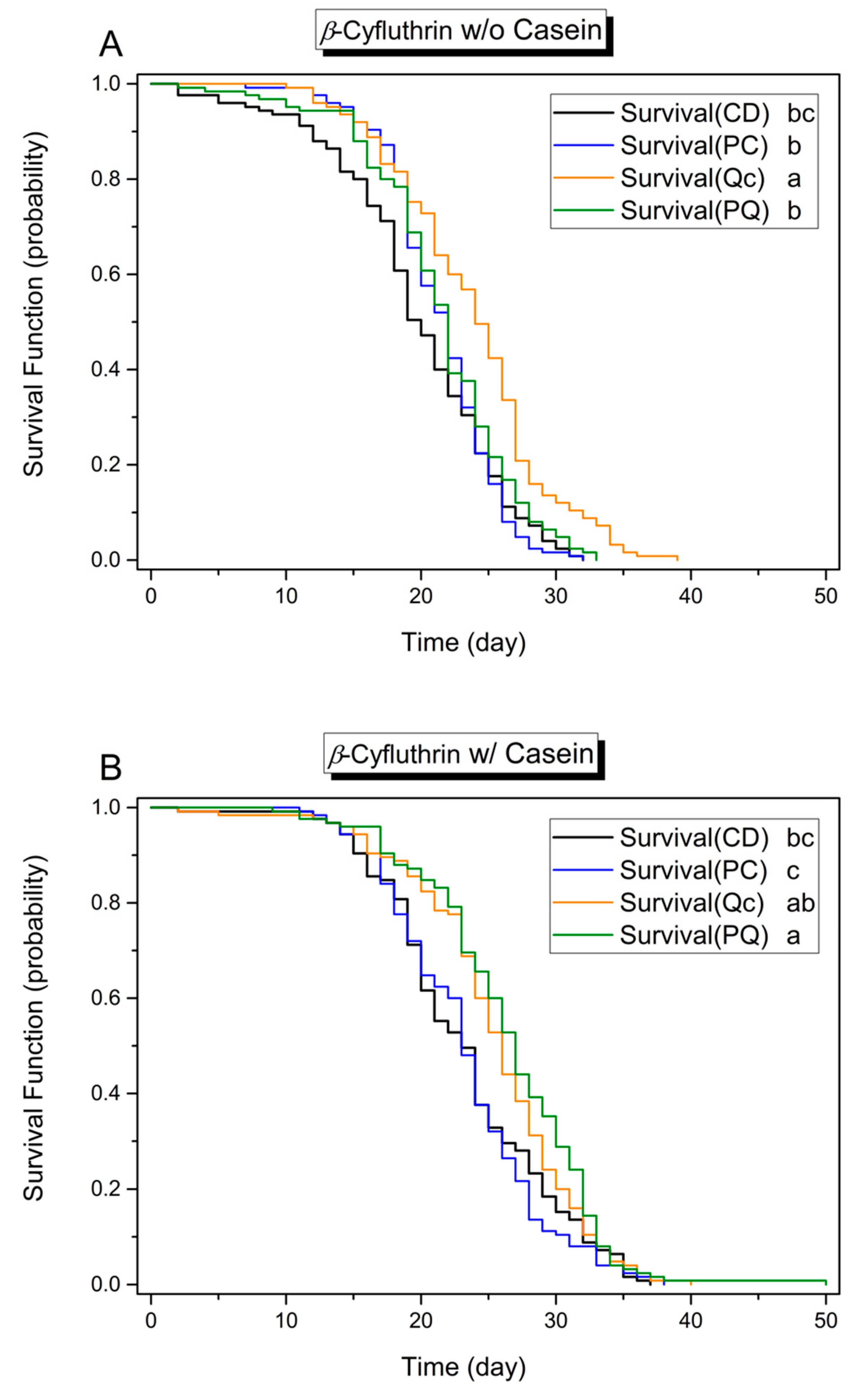
3.1. Effects of Dietary Protein, Phytochemicals and Pesticides on Survival
3.2. Effect of Phytochemicals on Longevity in the Absence of Pesticides
3.3. Effect of Phytochemicals on Survival in the Presence of Pesticides
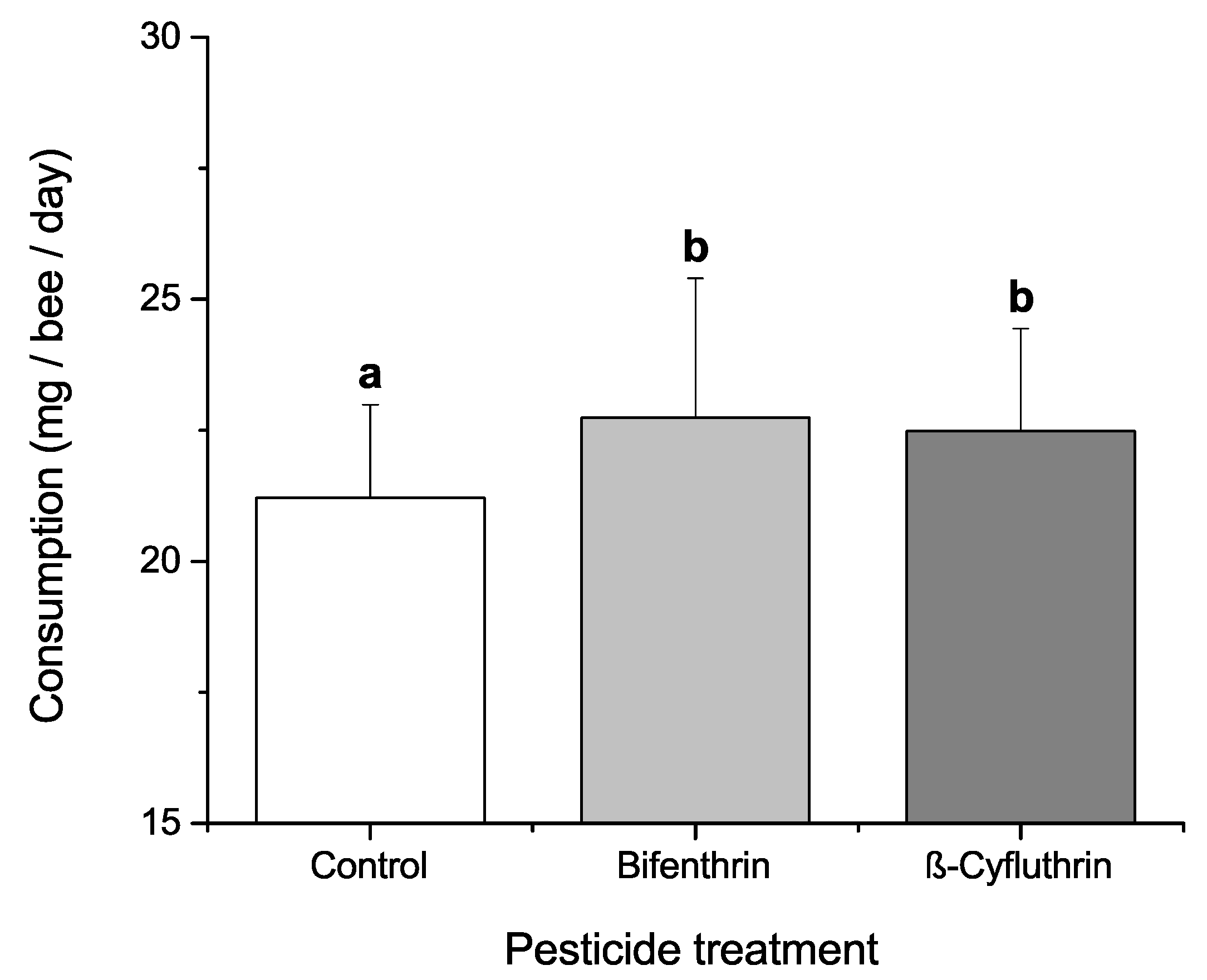
3.4. Effects of Dietary Protein, Phytochemicals and Pesticides on Diet Consumption
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brodschneider, R.; Crailsheim, K. Nutrition and health in honey bees. Apidologie 2010, 41, 278–294. [Google Scholar] [CrossRef]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. A dietary phytochemical alters caste-associated gene expression in honey bees. Sci. Adv. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Honey constituents up-regulate detoxification and immunity genes in the western honey bee Apis mellifera. Proc. Natl. Acad. Sci. USA 2013, 110, 8842–8846. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Rupasinghe, S.G.; Johnson, R.M.; Zangerl, A.R.; Schuler, M.A.; Berenbaum, M.R. Quercetin-metabolizing CYP6AS enzymes of the pollinator Apis mellifera (Hymenoptera: Apidae). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2009, 154, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. CYP9Q-mediated detoxification of acaricides in the honey bee (Apis mellifera). Proc. Natl. Acad. Sci. USA 2011, 108, 12657–12662. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Task-related differential expression of four cytochrome P450 genes in honeybee appendages. Insect Mol. Biol. 2015, 24, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.M.; Mao, W.; Pollock, H.S.; Niu, G.; Schuler, M.A.; Berenbaum, M.R. Ecologically appropriate xenobiotics induce cytochrome P450s in Apis mellifera. PLoS ONE 2012, 7, e31051. [Google Scholar] [CrossRef] [PubMed]
- Leonov, A.; Arlia-Ciommo, A.; Piano, A.; Svistkova, V.; Lutchman, V.; Medkour, Y.; Titorenko, V.I. Longevity extension by phytochemicals. Molecules 2015, 20, 6544–6572. [Google Scholar] [CrossRef] [PubMed]
- Si, H.; Liu, D. Dietary antiaging phytochemicals and mechanisms associated with prolonged survival. J. Nutr. Biochem. 2014, 25, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.M.; Murphy, E.A.; Carmichael, M.D.; Davis, B. Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R1071–R1077. [Google Scholar] [CrossRef] [PubMed]
- Lappalainen, Z. Sirtuins: A family of proteins with implications for human performance and exercise physiology. Res. Sports Med. 2011, 19, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Zhang, X.; Zhang, L.; Bian, H.X.; Xu, N.; Bao, B.; Liu, J. Quercetin reduces obesity-associated ATM infiltration and inflammation in mice: A mechanism including AMPKα1/SIRT1. J. Lipid Res. 2014, 55, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.G.; Rogina, B.; Lavu, S.; Howitz, K.; Helfand, S.L.; Tatar, M.; Sinclair, D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004, 430, 686–689. [Google Scholar] [CrossRef] [PubMed]
- Rascón, B.; Hubbard, B.P.; Sinclair, D.A.; Amdam, G.V. The lifespan extension effects of resveratrol are conserved in the honey bee and may be driven by a mechanism related to caloric restriction. Aging 2012, 4, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Grandison, R.C.; Wong, R.; Bass, T.M.; Partridge, L.; Piper, M.D.W. Effect of a standardised dietary restriction protocol on multiple laboratory strains of Drosophila melanogaster. PLoS ONE 2009, 4, e4067. [Google Scholar] [CrossRef] [PubMed]
- Lian, T.; Gaur, U.; Yang, D.; Li, D.; Li, Y.; Yang, M. Epigenetic mechanisms of dietary restriction induced aging in Drosophila. Exp. Gerontol. 2015, 72, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Altaye, S.Z.; Pirk, C.W.W.; Crewe, R.M.; Nicolson, S.W. Convergence of carbohydrate-biased intake targets in caged worker honeybees fed different protein sources. J. Exp. Biol. 2010, 213, 3311–3318. [Google Scholar] [CrossRef] [PubMed]
- Amdam, G.V.; Fennern, E.; Havukainen, H. Vitellogenin in honey bee behavior and lifespan. In Honeybee Neurobiology and Behavior. A Tribute to Randolf Menzel, 1st ed.; Galizia, C.G., Eisenhardt, D., Giurfa, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 17–29. [Google Scholar]
- Corona, M.; Velarde, R.A.; Remolina, S.; Moran-Lauter, A.; Wang, Y.; Hughes, K.A.; Robinson, G.E. Vitellogenin, juvenile hormone, insulin signaling, and queen honey bee longevity. Proc. Natl. Acad. Sci. USA 2007, 104, 7128–7133. [Google Scholar] [CrossRef] [PubMed]
- Nicodemo, D.; Malheiros, E.B.; De Jong, D.; Couto, R.H.N. Increased brood viability and longer lifespan of honeybees selected for propolis production. Apidologie 2014, 45, 269–275. [Google Scholar] [CrossRef]
- Meneghelli, C.; Joaquim, L.S.D.; Felix, G.L.Q.; Somensi, A.; Tomazzoli, M.; da Silva, D.A.; Berti, F.V.; Veleirinho, M.B.R.; Recouvreux, D.D.S.; Zeri, A.C.D.; et al. Southern Brazilian autumnal propolis shows anti-angiogenic activity: An in vitro and in vivo study. Microvasc. Res. 2013, 88, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; Vanengelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.-H.; Wu, W.-Y.; Berenbaum, M.R.; University of Illinois, Urbana, IL, USA. Unpublished data. 2017.
- Lee, K.P. The interactive effects of protein quality and macronutrient imbalance on nutrient balancing in an insect herbivore. J. Exp. Biol. 2007, 210, 3236–3244. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Schuler, M.A.; Berenbaum, M.R. Disruption of quercetin metabolism by fungicide affects energy production in honey bees Apis mellifera. Proc. Natl. Acad. Sci. USA 2017, in press. [Google Scholar] [CrossRef]
- Martos, I.; Ferreres, F.; Tomás-Barberán, F.A. Identification of flavonoid markers for the botanical origin of Eucalyptus Honey. J. Agric. Food Chem. 2000, 48, 1498–1502. [Google Scholar] [CrossRef] [PubMed]
- Bonvehí, S.J.; Torrentó, S.M.; Lorente, C.E. Evaluation of polyphenolic and flavonoid compounds in honeybee-collected pollen produced in Spain. J. Agric. Food Chem. 2001, 49, 1848–1853. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Ruočkuvienė, G.; Kaškonas, P.; Akuneca, I.; Maruška, A. Chemometric analysis of bee pollen based on volatile and phenolic compound compositions and antioxidant properties. Food Anal. Methods 2015, 8, 1150–1163. [Google Scholar] [CrossRef]
- Ceksteryte, V.; Kazlauskas, S.; Racys, J. Composition of flavonoids in Lithuanian honey and beebread. Biologija 2006, 2, 28–33. [Google Scholar]
- Case, D.L.; Kimmick, G.; Paskett, E.D.; Lohman, K.; Tucker, R. Interpreting measures of treatment effect in cancer clinical trials. Oncologist 2002, 7, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Boots, A.W.; Haenen, G.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, B.D.; Seth, V.; Ahmed, R.S. Pesticide-induced oxidative stress: Perspectives and trends. Rev. Environ. Health 2001, 16, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Dahlgren, L.; Johnson, R.M.; Siegfried, B.D.; Ellis, M.D. Comparative toxicity of acaricides to honey bee (Hymenoptera: Apidae) workers and queens. J. Econ. Entomol. 2012, 105, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Pritsos, C.A.; Ahmad, S.; Bowen, S.M.; Elliott, A.J.; Blomquist, G.J.; Pardini, R.S. Antioxidant enzymes of the black swallowtail butterfly, Papilio polyxenes, and their response to the prooxidant allelochemical, quercetin. Arch. Insect Biochem. Physiol. 1988, 8, 101–112. [Google Scholar] [CrossRef]
- Dai, P.L.; Wang, Q.; Sun, J.H.; Liu, F.; Wang, X.; Wu, Y.Y.; Zhou, T. Effects of sublethal concentrations of bifenthrin and deltamethrin on fecundity, growth, and development of the honeybee Apis mellifera ligustica. Environ. Toxicol. Chem. 2010, 29, 644–649. [Google Scholar] [CrossRef] [PubMed]
- Wahl, O.; Ulm, K. Influence of pollen feeding and physiological condition on pesticide sensitivity of the honey bee Apis mellifera carnica. Oecologia 1983, 59, 106–128. [Google Scholar] [CrossRef] [PubMed]
- Alaux, C.; Ducloz, F.; Crauser, D.; Conte, Y. Diet effects on honeybee immunocompetence. Biol. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.M.; Robinson, G.E. Diet-dependent gene expression in honey bees: Honey vs. sucrose or high fructose corn syrup. Sci. Rep. 2014. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhao, G.; Yu, Y.; Liu, F. High concentration of nectar quercetin enhances worker resistance to queen’s signals in bees. J. Chem. Ecol. 2010, 36, 1241–1243. [Google Scholar] [CrossRef] [PubMed]
| Protein | Protein-free (casein−) | Protein-rich (casein+) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Phytochemical | Control (CD) | 0.5 mM | 0.25 mM quercetin (Qc) | 0.5 mM | Control (CD) | 0.5 mM | 0.25 mM quercetin (Qc) | 0.5 mM | |
| p-coumaric acid (PC) | p-coumaric acid and 0.25 mM quercetin (PQ) | p-coumaric acid (PC) | p-coumaric acid and 0.25 mM quercetin (PQ) | ||||||
| Pesticide | Pesticide-free | Treatment 1 | Treatment 2 | Treatment 3 | Treatment 4 | Treatment 5 | Treatment 6 | Treatment 7 | Treatment 8 |
| β-Cyfluthrin | Treatment 9 | Treatment 10 | Treatment 11 | Treatment 12 | Treatment 13 | Treatment 14 | Treatment 15 | Treatment 16 | |
| Bifenthrin | Treatment 17 | Treatment 18 | Treatment 19 | Treatment 20 | Treatment 21 | Treatment 22 | Treatment 23 | Treatment 24 | |
| Experimental factor | df | Estimate | Standard Error | χ2 | p | Hazard Ratio |
|---|---|---|---|---|---|---|
| Casein | 1 | −0.30 | 0.04 | 66.31 | < 0.001 | 0.739 *** |
| Quercetin | 1 | −0.20 | 0.04 | 27.93 | < 0.001 | 0.823 *** |
| p-Coumaric acid | 1 | −0.09 | 0.04 | 5.93 | 0.015 | 0.914 * |
| β-cyfluthrin | 1 | 0.30 | 0.05 | 42.16 | < 0.001 | 1.345 *** |
| Bifenthrin | 1 | 2.22 | 0.05 | 1741.64 | < 0.001 | 9.171 *** |
| Protein-free (casein−) | Protein-rich (casein+) | ||
|---|---|---|---|
| Overall | <a | ||
| Pesticide-free dietb | Treatment 1–4 | = | Treatment 5–8 |
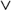 | |||
| β-cyfluthrin diet | Treatment 9–12 | < | Treatment 13–16 |
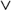 | |||
| Bifenthrin diet | Treatment 17–20 | < | Treatment 21–24 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, L.-H.; Wu, W.-Y.; Berenbaum, M.R. Impacts of Dietary Phytochemicals in the Presence and Absence of Pesticides on Longevity of Honey Bees (Apis mellifera). Insects 2017, 8, 22. https://doi.org/10.3390/insects8010022
Liao L-H, Wu W-Y, Berenbaum MR. Impacts of Dietary Phytochemicals in the Presence and Absence of Pesticides on Longevity of Honey Bees (Apis mellifera). Insects. 2017; 8(1):22. https://doi.org/10.3390/insects8010022
Chicago/Turabian StyleLiao, Ling-Hsiu, Wen-Yen Wu, and May R. Berenbaum. 2017. "Impacts of Dietary Phytochemicals in the Presence and Absence of Pesticides on Longevity of Honey Bees (Apis mellifera)" Insects 8, no. 1: 22. https://doi.org/10.3390/insects8010022
APA StyleLiao, L.-H., Wu, W.-Y., & Berenbaum, M. R. (2017). Impacts of Dietary Phytochemicals in the Presence and Absence of Pesticides on Longevity of Honey Bees (Apis mellifera). Insects, 8(1), 22. https://doi.org/10.3390/insects8010022





