Electrostatic Insect Sweeper for Eliminating Whiteflies Colonizing Host Plants: A Complementary Pest Control Device in An Electric Field Screen-Guarded Greenhouse
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Electrostatic Insect Sweeper
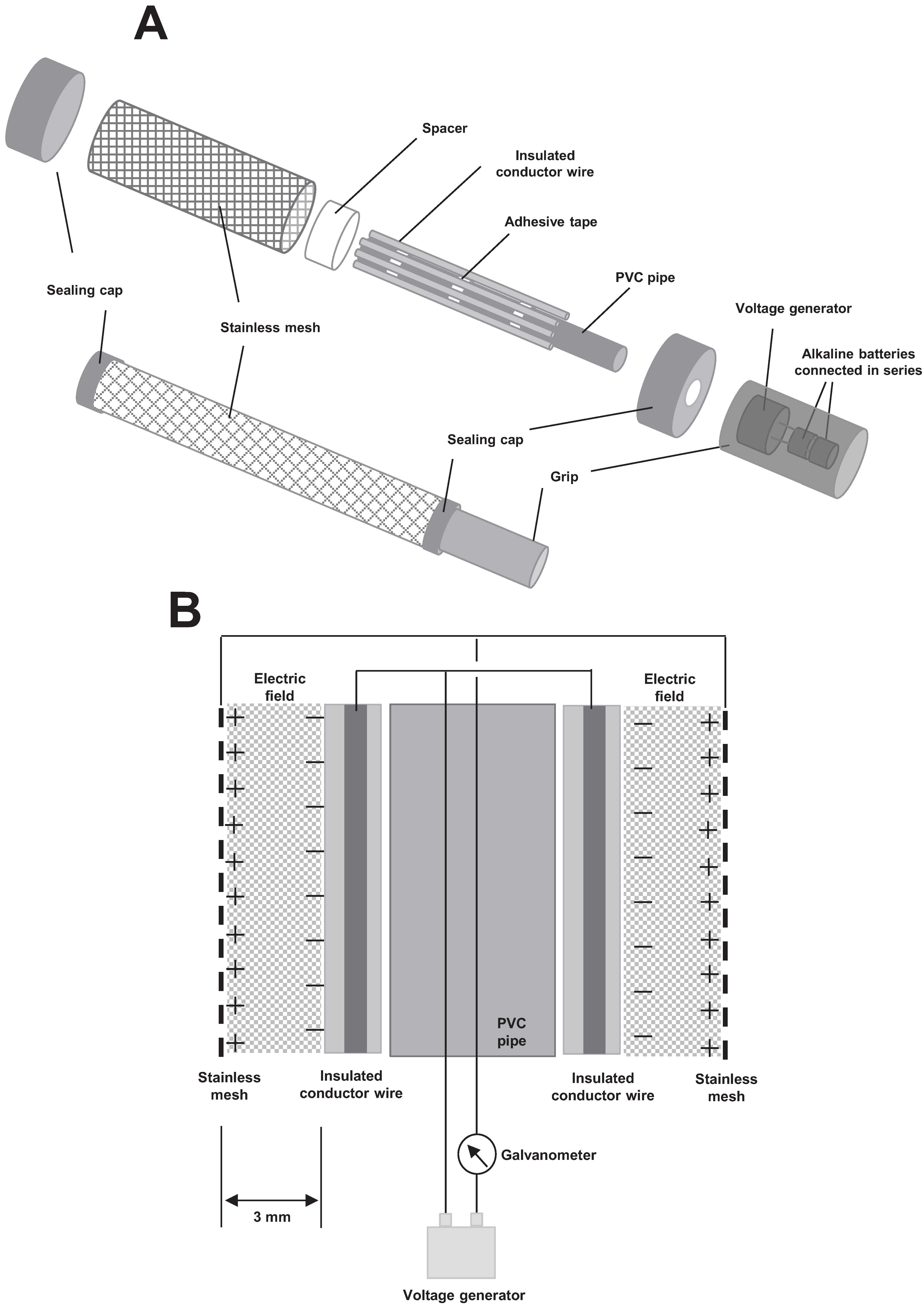
2.3. Insect Capturing Assay
| Pest insects tested | Negative and positive voltages (kV) applied to ICWs | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.5 | 0.8 | 1 | 1.2 | 1.5 | 2 | 3 | 5 | 5.6 | |
| Whitefly | 0 | 0 | 41.6 ± 9.4 a | 91.3 ± 6.7 a | 100 a | 100 | 100 | 100 | 100 | 100 |
| Western flower thrips | 0 | 0 | 0 b | 73.6 ± 4.9 b | 93.5 ± 6.1 b | 100 | 100 | 100 | 100 | 100 |
| Green peach aphid | 0 | 0 | 0 b | 73.3 ± 4.3 b | 92.1 ± 7.2 b | 100 | 100 | 100 | 100 | 100 |
| Shore fly | 0 | 0 | 0 b | 32.1 ± 8.7 c | 60.4 ± 9.6 c | 100 | 100 | 100 | 100 | 100 |
2.4. Greenhouse Setting
2.5. Prevention of Whitefly Entry into the Greenhouse by Window-Installed SD Screens
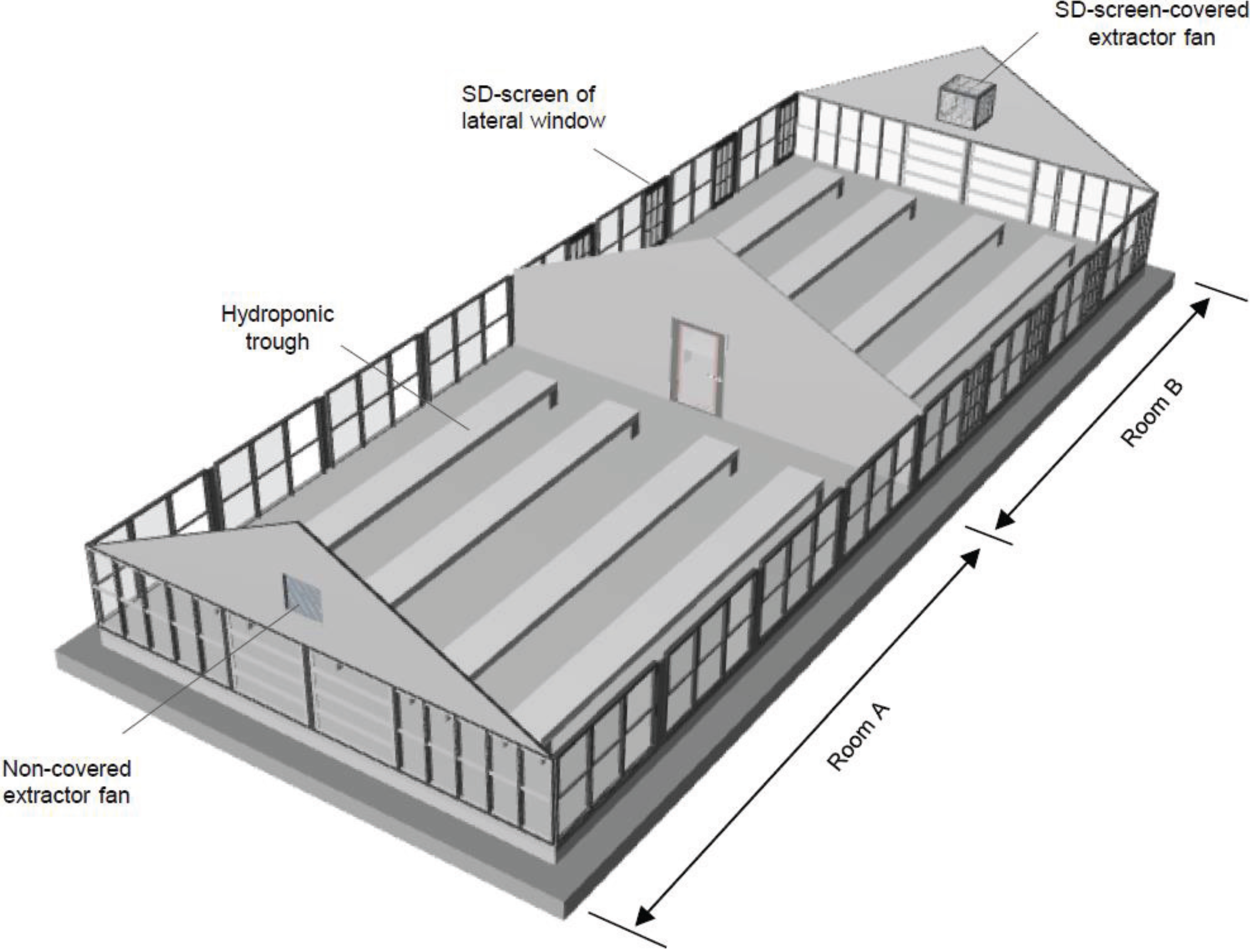
2.6. Greenhouse Assay for Eliminating Whiteflies Entering from an Entrance Door

2.7. Biotype Determination of Trapped Whiteflies and Virus Detection in Whiteflies
3. Results and Discussion
3.1. Current Pest and Pathogen Situation in Our Greenhouse Tomatoes
3.2. Confirmation of Successful Exclusion of Whiteflies from a Greenhouse by SD Screens

3.3. Experimental Setup for Increasing Whitefly Entry through the Greenhouse Entrance Door
3.4. Electrostatic Characteristics of the Sweeper and Its Ability to Capture Insects on Tomato Leaves
3.5. Practical Application of the Sweeper to Greenhouse Tomatoes
4. Conclusions
Supplementary Files
Supplementary File 1Supplementary File 2Acknowledgements
Author Contributions
Conflicts of Interest
References
- Cohen, S.; Berlinger, M.J. Transmission and cultural control of white-fly borne viruses. Agric. Ecosys. Environ. 1986, 17, 89–97. [Google Scholar] [CrossRef]
- Oliveira, M.R.V.; Henneberry, T.J.; Anderson, P. History, current status, and collaborative research projects of Bemisia tabaci. Crop Prot. 2001, 20, 709–723. [Google Scholar] [CrossRef]
- Sharaf, N. Chemical control of Bemisia tabaci. Agric. Ecosys. Environ. 1986, 17, 111–127. [Google Scholar] [CrossRef]
- Prabhaker, N.; Coudriet, D.L.; Meyerdirk, D.E. Insecticide resistance in the sweetpotato whitefly, Bemisia tabaci (Homoptera: Aleyrodiae). J. Econ. Entomol. 1985, 78, 748–752. [Google Scholar] [CrossRef]
- Palumbo, J.C.; Horowitz, A.R.; Prabhaker, N. Insecticidal control and resistance management for Bemisia tabaci. Crop Prot. 2001, 20, 739–765. [Google Scholar] [CrossRef]
- Horowitz, A.R.; Kontsedalov, S.; Ishaaya, I. Dynamics of resistance to the neonicotinoids acetamiprid and thiamethoxam in Bemisia tabacci (Homoptera: Aleyrodidae). J. Econ. Entomol. 2004, 97, 2051–2056. [Google Scholar] [CrossRef] [PubMed]
- Nauen, R.; Denholm, I. Resistance of insect pests to neonicotinoid insecticides: Current status and future prospects. Arch. Insect Biochem. Physiol. 2005, 58, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Weintraub, P.G.; Berlinger, M.J. Physical control in greenhouses and field crops. In Insect Pest Management; Horowitz, A.R., Ishaaya, I., Eds.; Springer-Verlag: Berlin, Germany, 2004; Volume 3, pp. 301–318. [Google Scholar]
- Tanaka, N.; Matsuda, Y.; Kato, E.; Kokabe, K.; Furukawa, T.; Nonomura, T.; Honda, K.; Kusakari, S.; Imura, T.; Kimbara, J.; et al. An electric dipolar screen with oppositely polarized insulators for excluding whiteflies from greenhouses. Crop Prot. 2008, 27, 215–221. [Google Scholar] [CrossRef]
- Nonomura, T.; Matsuda, Y.; Kakutani, K.; Kimbara, J.; Osamura, K.; Kusakari, S.; Toyoda, H. An electric field strongly deters whiteflies from entering window-open greenhouses in an electrostatic insect exclusion strategy. Eur. J. Plant Pathol. 2012, 134, 661–670. [Google Scholar] [CrossRef]
- Matsuda, Y.; Nonomura, T.; Kakutani, K.; Takikawa, Y.; Kimbara, J.; Kasaishi, Y.; Kusakari, S.; Toyoda, H. A newly devised electric field screen for avoidance and capture of cigarette beetles and vinegar flies. Crop Prot. 2011, 30, 155–162. [Google Scholar] [CrossRef]
- Murai, T. Rearing method for clones of some aphids on tick bean, Vicia faba. Bull. Shimane Agric. Exp. Stat. 1991, 25, 78–82. [Google Scholar]
- Murai, T.; Loomans, A.J.M. Evaluation of an improved method for mass-rearing of thrips and a thrips parasitoid. Entomol. Exp. Appl. 2001, 101, 281–289. [Google Scholar] [CrossRef]
- Nonomura, T.; Matsuda, Y.; Bingo, M.; Onishi, M.; Matsuda, K.; Harada, S.; Toyoda, H. Algicidal effect of 3-(3-indolyl)butanoic acid, a control agent of the bacterial wilt pathogen, Ralstonia solanacearum. Crop Prot. 2001, 20, 935–939. [Google Scholar] [CrossRef]
- Griffith, W.T. Electrostatic phenomena. In The Physics of Everyday Phenomena, A Conceptual Introduction to Physics; Bruflodt, D., Loehr, B.S., Eds.; McGraw-Hill: New York, NY, USA, 2004; pp. 232–252. [Google Scholar]
- Hsieh, C.H.; Wang, H.Y.; Chen, Y.F.; Ko, C.C. Loop-mediated isothermal amplification for rapid identification of biotypes B and Q of the globally invasive pest Bemisia tabaci, and studying population dynamics. Pest Manag. Sci. 2012, 68, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, S.; Kato, S.; Yoshida, K.; Mizukami, Y.; Ishida, A.; Ueda, J.; Kanbe, M.; Ishimoto, Y. Detection of tomato yellow leaf curl virus by loop-mediated isothermal amplification reaction. J. Virol. Methods 2003, 112, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, D.R.; Menzies, J.G. Fungus gnats vector Fusarium oxysporum f. sp. radicis-lycopersici. Ann. Appl. Biol. 1993, 123, 539–544. [Google Scholar] [CrossRef]
- El-Hamalawi, Z.A. Attraction, acquisition, retention and spatiotemporal distribution of soilborne plant pathogenic fungi by shore flies. Ann. Appl. Biol. 2008, 152, 169–177. [Google Scholar] [CrossRef]
- Nonomura, T.; Matsuda, Y.; Kakutani, K.; Takikawa, Y.; Toyoda, H. Physical control of powdery mildew (Oidium neolycopersici) on tomato leaves by exposure to corona discharge. Can. J. Plant Pathol. 2008, 30, 517–524. [Google Scholar] [CrossRef]
- Matsuda, Y.; Ikeda, H.; Moriura, N.; Tanaka, N.; Shimizu, K.; Oichi, W.; Nonomura, T.; Kakutani, K.; Kusakari, S.; Higashi, K.; et al. A new spore precipitator with polarized dielectric insulators for physical control of tomato powdery mildew. Phytopathology 2006, 96, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Matsuda, Y.; Nonomura, T.; Ikeda, H.; Tamura, N.; Kusakari, S.; Kimbara, J.; Toyoda, H. Dual protection of hydroponic tomatoes from rhizosphere pathogens Ralstonia solanacearum and Fusarium oxysporum f. sp. radicis-lycopersici and airborne conidia of Oidium neolycopersici with an ozone-generative electrostatic spore precipitator. Plant Pathol. 2007, 56, 987–997. [Google Scholar]
- Kakutani, K.; Matsuda, Y.; Nonomura, T.; Kimbara, J.; Kusakari, S.; Toyoda, H. Practical application of an electric field screen to an exclusion of flying insect pests and airborne conidia from greenhouses with a good air penetration. J. Agric. Sci. 2012, 4, 51–60. [Google Scholar]
- Kakutani, K.; Matsuda, Y.; Haneda, K.; Sekoguchi, D.; Nonomura, T.; Kimbara, J.; Osamura, K.; Kusakari, S.; Toyoda, H. An electric field screen prevents captured insects from escaping by depriving bioelectricity generated through insect movements. J. Electrost. 2012, 70, 207–211. [Google Scholar] [CrossRef]
- Matsuda, Y.; Kakutani, K.; Nonomura, T.; Kimbara, J.; Kusakari, S.; Osamura, K.; Toyoda, H. An oppositely charged insect exclusion screen with gap-free multiple electric fields. J. Appl. Phys. 2012. [Google Scholar] [CrossRef]
- Matsuda, Y.; Setomoto, M.; Yoshimoto, N.; Nonomura, T.; Kakutani, K.; Takikawa, Y.; Toyoda, H. Identification of Bemisia tabaci biotypes and detection of TYLCV from diseased tomato plants and viruliferous biotype Q whiteflies. Mem. Fac. Agr. Kinki Univ. 2013, 46, 1–5. [Google Scholar]
- Wegner, H.E. Electrical charging generators. In McGraw-Hill Encyclopedia of Science and Technology, 9th ed.; The Lakeside Press: New York, NY, USA, 2002; pp. 42–43. [Google Scholar]
- Halliday, D.; Resnick, R.; Walker, J. Electric fields. In Fundamentals of Physics; Johnson, S., Ford, E., Eds.; John Wiley & Sons: New York, NY, USA, 2005; pp. 580–604. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takikawa, Y.; Matsuda, Y.; Kakutani, K.; Nonomura, T.; Kusakari, S.-i.; Okada, K.; Kimbara, J.; Osamura, K.; Toyoda, H. Electrostatic Insect Sweeper for Eliminating Whiteflies Colonizing Host Plants: A Complementary Pest Control Device in An Electric Field Screen-Guarded Greenhouse. Insects 2015, 6, 442-454. https://doi.org/10.3390/insects6020442
Takikawa Y, Matsuda Y, Kakutani K, Nonomura T, Kusakari S-i, Okada K, Kimbara J, Osamura K, Toyoda H. Electrostatic Insect Sweeper for Eliminating Whiteflies Colonizing Host Plants: A Complementary Pest Control Device in An Electric Field Screen-Guarded Greenhouse. Insects. 2015; 6(2):442-454. https://doi.org/10.3390/insects6020442
Chicago/Turabian StyleTakikawa, Yoshihiro, Yoshinori Matsuda, Koji Kakutani, Teruo Nonomura, Shin-ichi Kusakari, Kiyotsugu Okada, Junji Kimbara, Kazumi Osamura, and Hideyoshi Toyoda. 2015. "Electrostatic Insect Sweeper for Eliminating Whiteflies Colonizing Host Plants: A Complementary Pest Control Device in An Electric Field Screen-Guarded Greenhouse" Insects 6, no. 2: 442-454. https://doi.org/10.3390/insects6020442
APA StyleTakikawa, Y., Matsuda, Y., Kakutani, K., Nonomura, T., Kusakari, S.-i., Okada, K., Kimbara, J., Osamura, K., & Toyoda, H. (2015). Electrostatic Insect Sweeper for Eliminating Whiteflies Colonizing Host Plants: A Complementary Pest Control Device in An Electric Field Screen-Guarded Greenhouse. Insects, 6(2), 442-454. https://doi.org/10.3390/insects6020442






