Semiochemical and Vibrational Cues and Signals Mediating Mate Finding and Courtship in Psylloidea (Hemiptera): A Synthesis
Abstract
:1. Signals and Strategies of Insect Sexual Selection
2. Introduction to the Psylloidea
3. Psyllid Reproductive Biology and Mating Systems

4. Male Mating Strategies

5. Semiochemical Signalling in Psyllids
| Family | Species | Semiochemical (s) | Vibrational Signal | Nature of Vibrational Signal | Mechanism of Vibrational Signal Production | Ref. | |
|---|---|---|---|---|---|---|---|
| Substrate Borne Vibrations | Air Borne Vibrations | ||||||
| Aphalaridae | Anoeconeossa sp. Anoeconeossa unicornuta | ● | ■ | RWV, TO [?] | [96,97,98] | ||
| Aphalara affinis, Aphalara polygoni | ● | ■ | RWV | [60,96] | |||
| Australopsylla sp. | ● | ■ | RWV/TO [?] | [96,97,98] | |||
| Blastopsylla sp. | ●? | TO [?] | [97] | ||||
| Cardiaspina albitextura, C. retator, C. tenuitela, C. densitexta | ● | ■ | Faint whirring sound | RWV | [67,98] | ||
| Cardiaspina maniformis, C. fiscella | ● | ■ | RWV | [96,99] | |||
| Craspedolepta gloriosa, C. campestrella, C. nervosa, C. flavipennis, C. nebulosa | ● | ■ | RWV | [60] | |||
| Creiis spp. | ● | High pitched buzzing calls | RWV | [76,98] | |||
| Ctenarytaina sp. | ●? | TO [?] | [97] | ||||
| Glycaspis spp. G. brimblecombei, G. johnsoni, G. neureta | ● | ■ | RWV/TO [?] | [97,98] | |||
| Lasiopsylla rotundipennis | ● | ■ | RWV | [96,98] | |||
| Spondyliaspis sp. | ● | ■ | RWV | [96,98] | |||
| Phellopsylla sp. | ● | ■ | RWV | [96,98] | |||
| Carsidaridae | Protyora sterculiae | ●? | TO [?] | [97] | |||
| Liviidae | Diaphorina citri | ▲ (A) | ● | ■ | RWV | [78,92,100] | |
| Eremopsylloides amirabilis | ● | RWV | [63] | ||||
| Livia juncorum | ● | Short buzzing sounds | RWV | [60,101] | |||
| Pachypsylloides citreus | ● | ■ | RWV | [63] | |||
| Psyllidae | Cacopsylla bidens | ▲ | ? | [82] | |||
| Cacopylla pyri | ● | ■ | RWV | [77] | |||
| Cacopsylla pyricola | ▲ (B) | RWV | [88,91,94] | ||||
| Colposcenia aliena | ● | ■ | RWV | [63] | |||
| Livilla ulicis | ● | ■ | RWV | [60,96] | |||
| Triozidae | Aacanthocnema dobsoni | ● | ■ | RWV | [59] | ||
| Bactericera cockerelli | ▲ | ? | [90] | ||||
| Bactericera perrisii, B. kratochvili, B. calcarata, Eryngiofaga deserta | ● | ■ | RWV | [63] | |||
| Schedotrioza apicobystra, S. cornuta, S. distorta, S. marginata, S. multitudinea, Schedotrioza sp. | ● | ■ | RWV | [59] | |||
| Trioza tricornuta, T. percyae | ● | RWV | [59] | ||||
| Trioza spp. Trioza acutipennis, T. nigricornis, Trioza urticae | ● | ■ | Short buzzing sounds | RWV | [59,63,101] | ||
6. Vibrational Signalling in Psyllids
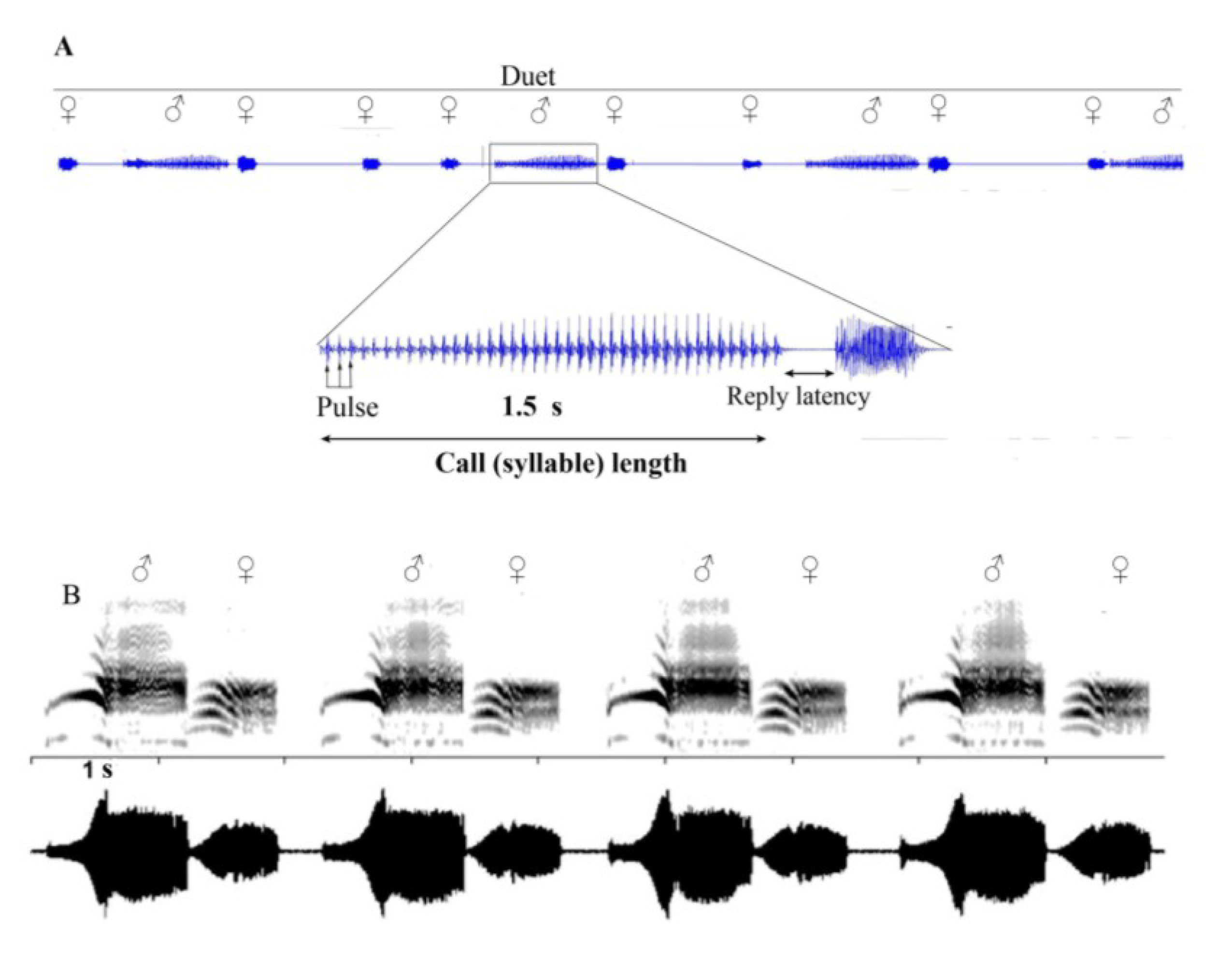
7. Multimodal Signalling
8. Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bradbury, J.W.; Vehrencamp, S.L. Mate attraction and courtship. In Principles of Animal Communication, 2nd ed.; Sinauer Associate Inc.: Massacusets, MA, USA, 1998; Volume 12. [Google Scholar]
- Thornhill, R. Male and female sexual selection and the evolution of mating strategies in insects. In Sexual Selection and Reproductive Competition in Insects; Academic Press: New York, NY, USA, 1979; pp. 81–121. [Google Scholar]
- Parker, G.A. Sperm competition and the evolution of animal mating strategies. In Sperm Competition and the Evolution of Animal Mating Systems; Academic Press: New York, NY, USA, 1984; pp. 1–60. [Google Scholar]
- Johansson, B.G.; Jones, T.M. The role of chemical communication in mate choice. Biol. Rev. 2007, 82, 265–289. [Google Scholar] [CrossRef]
- Chapman, T.; Arnqvist, G.; Bangham, J.; Rowe, L. Sexual conflict. Trends Ecol. Evol. 2003, 18, 41–47. [Google Scholar] [CrossRef]
- Schärer, L.; Rowe, L.; Arnqvist, G. Anisogamy, chance and the evolution of sex roles. Trends Ecol. Evol. 2012, 27, 260–264. [Google Scholar] [CrossRef]
- Pashler, H.; Mozer, M.; Harris, C.R. Mating strategies in a Darwinian microworld: Simulating the consequences of female reproductive refractoriness. Adapt. Behav. 2001, 9, 5–15. [Google Scholar] [CrossRef]
- Kaitala, A.; Wiklund, C. Female mate choice and mating costs in the polyandrous butterfly Pieris napi (Lepidoptera: Pieridae). J. Insect Behav. 1994, 8, 355–363. [Google Scholar] [CrossRef]
- Karlström, A. Female mate choice and modulation of oviposition preference in the moth Spodoptera littoralis. BSc. Thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2013. [Google Scholar]
- Crean, C.S.; Dunn, D.W.; Day, T.H.; Gilburn, A.S. Female mate choice for large males in several species of seaweed fly (Diptera: Coelopidae). Anim. Behav. 2000, 59, 121–126. [Google Scholar] [CrossRef]
- Bonduriansky, R. The evolution of male mate choice in insects: A synthesis of ideas and evidence. Biol. Rev. 2001, 76, 305–339. [Google Scholar] [CrossRef]
- Rutowski, R.L. Courtship solicitation by females of the checkered white butterfly, Pieris protodice. Behav. Ecol. Sociobiol. 1980, 7, 113–117. [Google Scholar] [CrossRef]
- Gwynne, D.T.; Jamieson, I. Sexual selection and sexual dimorphism in a harem-polygynous insect, the Alpine weta Hemideina maori (Orthoptera Stenopelmatidae). Ethol. Ecol. Evol. 1998, 10, 393–402. [Google Scholar] [CrossRef]
- Helinski, M.E.H.; Valerio, L.; Facchinelli, L.; Scott, T.W.; Ramsey, J.; Harrington, L.C. Evidence of polyandry for Aedes aegypti in semifield enclosures. Am. J. Trop. Med. Hyg. 2012, 86, 635–641. [Google Scholar] [CrossRef]
- Franco, K.; Jauset, A.; Castañé, C. Monogamy and polygamy in two species of mirid bugs: A functional-based approach. J. Insect Physiol. 2011, 57, 307–315. [Google Scholar] [CrossRef]
- Thornhill, R.; Alcock, J. The Evolution of Insect Mating Systems; Harvard University Press: Cambridge, MA, USA, 1983. [Google Scholar]
- Emlen, S.T.; Oring, L.W. Ecology, sexual selection, and the evolution of mating systems. Science 1977, 197, 215–223. [Google Scholar]
- Shuster, S.M.; Wade, M.J. Mating Systems and Strategies; Princeton University Press: Princeton, NJ, USA, 2003; pp. 262–443. [Google Scholar]
- Greenfield, M.D. Signalers and receivers. In Mechanisms and Evolution of Arthropod Communication; Oxford University Press: New York, NY, USA, 2002; p. 414. [Google Scholar]
- Jacobson, M. Occurrence of sex pheromones in the female. In Insect Sex Pheromones; Academic Press, Inc.: New York, NY, USA, 2012; pp. 5–27. [Google Scholar]
- KonDo, Y.; Naka, H.; Tsuchida, K. Pheromones and body coloration affect mate recognition in the Japanese nine-spotted moth Amata fortunei (Lepidoptera: Arctiidae). J. Ethol. 2012, 30, 301–308. [Google Scholar] [CrossRef]
- Benelli, G.; Giunti, G.; Messing, R.H.; Wright, M.G. Visual and olfactory female-borne cues evoke male courtship in the aphid parasitoid Aphidius colemani Viereck (Hymenoptera: Braconidae). J. Insect Behav. 2013, 26, 695–707. [Google Scholar] [CrossRef]
- Itadani, H.; Ueno, T. Chemically mediated mate finding of the polyphagous politary parasitoid Itoplectis naranyae (Hymenoptera: Ichneumonidae). Ann. Entomol. Soc. Am. 2014, 107, 288–294. [Google Scholar] [CrossRef]
- Cooley, J.R. Long-range acoustical signals, phonotaxis, and risk in the sexual pair-forming behaviors of Okanagana canadensis and O. rimosa (Hemiptera: Cicadidae). Ann. Entomol. Soc. Am. 2001, 94, 755–760. [Google Scholar] [CrossRef]
- Virant-Doberlet, M.; Cokl, A. Vibrational communication in insects. Neotrop. Entomol. 2004, 33, 121–134. [Google Scholar] [CrossRef]
- Čokl, A.; Virant-Doberlet, M. Communication with substrate-borne signals in small plant-dwelling insects 1. Annu. Rev. Entomol. 2003, 48, 29–50. [Google Scholar] [CrossRef]
- Hansson, B.S.; Stensmyr, M.C. Evolution of insect olfaction. Neuron 2011, 72, 698–711. [Google Scholar] [CrossRef]
- Bennet-Clark, H.C. Songs and the physics of sound production. Cricket Behav. Neurobiol. 1989, 227, 261. [Google Scholar]
- Young, D.; Bennet-Clark, H.C. The role of the tymbal in cicada sound production. J. Exp. Biol. 1995, 198, 1001–1020. [Google Scholar]
- Martin, J.P.; Beyerlein, A.; Dacks, A.M.; Reisenman, C.E.; Riffell, J.A.; Lei, H.; Hildebrand, J.G. The neurobiology of insect olfaction: Sensory processing in a comparative context. Prog. Neurobiol. 2011, 95, 427–447. [Google Scholar] [CrossRef]
- Ringo, J. Sexual receptivity in insects. Annu. Rev. Entomol. 1996, 41, 473–494. [Google Scholar] [CrossRef]
- Singer, T.L. Roles of hydrocarbons in the recognition systems of insects. Am. Zool. 1998, 38, 394–405. [Google Scholar]
- Martin, S.; Drijfhout, F. A review of ant cuticular hydrocarbons. J. Chem. Ecol. 2009, 35, 1151–1161. [Google Scholar] [CrossRef]
- Cocroft, R.B.; Rodríguez, R.L. The behavioral ecology of insect vibrational communication. Bioscience 2005, 55, 323–334. [Google Scholar] [CrossRef]
- Higham, J.P.; Hebets, E.A. An introduction to multimodal communication. Behav. Ecol. Sociobiol. 2013, 67, 1381–1388. [Google Scholar] [CrossRef]
- Miklas, N.; Čokl, A.; Renou, M.; Virant-Doberlet, M. Variability of vibratory signals and mate choice selectivity in the southern green stink bug. Behav. Process. 2003, 61, 131–142. [Google Scholar] [CrossRef]
- Miklas, N.; Lasnier, T.; Renou, M. Male bugs modulate pheromone emission in response to vibratory signals from conspecifics. J. Chem. Ecol. 2003, 29, 561–574. [Google Scholar] [CrossRef]
- Paterson, H.E.H. The recognition concept of species. In Species and Speciation; Transvaal Museum: Pretoria, South Africa, 1985; pp. 21–29. [Google Scholar]
- Wagner, W.E.; Reiser, M.G. The importance of calling song and courtship song in female mate choice in the variable field cricket. Anim. Behav. 2000, 59, 1219–1226. [Google Scholar] [CrossRef]
- Fonseca, P.J. Cicada acoustic communication. In Insect Hearing and Acoustic Communication; Hedwig, B., Ed.; Berlin Heidelberg, Springer: Berlin, Heidelberg, Germany, 2014; pp. 101–121. [Google Scholar]
- Austin, A.D.; Yeates, D.K.; Cassis, G.; Fletcher, M.J.; La Salle, J.; Lawrence, J.F.; McQuillan, P.B.; Mound, L.A.; Bickel, D.J.; Gullan, P.J.; et al. Insects “Down Under”—Diversity, endemism and evolution of the Australian insect fauna: Examples from select orders. Aust. J. Entomol. 2004, 43, 216–234. [Google Scholar] [CrossRef]
- Ouvrard, D. Psyl’list. The World Psylloidea Database. Available online: http://www.hemiptera-databases.com/psyllist (accessed on 5 July 2014).
- Woodburn, T.L.; Lewis, E.E. A comparative histological study of the effects of feeding by nymphs of four psyllid species on the leaves of eucalypts. Aust. J. Entomol. 1973, 12, 134–138. [Google Scholar] [CrossRef]
- Hodkinson, I.D. The biology of the Psylloidea (Homoptera): A review. Bull. Entomol. Res. 1974, 64, 325–338. [Google Scholar] [CrossRef]
- Munyaneza, J.E.; Crosslin, J.M.; Upton, J.E. Association of Bactericera cockerelli (Homoptera: Psyllidae) with “zebra chip,” a new potato disease in southwestern United States and Mexico. J. Econ. Entomol. 2007, 100, 656–663. [Google Scholar] [CrossRef]
- Munyaneza, J.E.; Goolsby, J.A.; Crosslin, J.M.; Upton, J.E. Further evidence that zebra chip potato disease in the lower Rio Grande Valley of Texas is associated with Bactericera cockerelli. Subtrop. Plant Sci. 2007, 59, 30–37. [Google Scholar]
- Hung, T.H.; Hung, S.C.; Chen, C.N.; Hsu, M.H.; Su, H.J. Detection by PCR of Candidatus Liberibacter asiaticus, the bacterium causing citrus huanglongbing in vector psyllids: Application to the study of vector-pathogen relationships. Plant Pathol. 2004, 53, 96–102. [Google Scholar] [CrossRef]
- Halbert, S.E.; Manjunath, K.L. Asian citrus psyllids (Sternorrhyncha: Psyllidae) and greening disease of citrus: A literature review and assessment of risk in Florida. Fla. Entomol. 2004, 87, 330–353. [Google Scholar] [CrossRef]
- Mayer, C.J.; Vilcinskas, A.; Gross, J. Phytopathogen lures its insect vector by altering host plant odor. J. Chem. Ecol. 2008, 34, 1045–1049. [Google Scholar] [CrossRef]
- Mayer, C.J.; Vilcinskas, A.; Gross, J. Chemically mediated multitrophic interactions in a plant—insect vector-phytoplasma system compared with a partially nonvector species. Agric. For. Entomol. 2011, 13, 25–35. [Google Scholar] [CrossRef]
- Burckhardt, D.; Ouvrard, D.; Queiroz, D.; Percy, D. Psyllid host-plants (Hemiptera: Psylloidea): Resolving a semantic problem. Fla. Entomol. 2014, 97, 242–246. [Google Scholar]
- Yang, M.-M.; Raman, A. Diversity, richness, and patterns of radiation among gall-inducing psyllids (Hemiptera: Psylloidea) in the Orient and Eastern Palearctic. Oriental Insects. 2007, 41, 55–65. [Google Scholar] [CrossRef]
- Prophetou-Athanasiadou, D.A.; Tzanakakis, M.E. Diapause termination in the olive psyllid Euphyllura phillyreae, in the field and in the laboratory. Entomol. Exp. Appl. 1986, 40, 263–272. [Google Scholar] [CrossRef]
- Krysan, J.L.; Higbee, B.S. Seasonality of mating and ovarian development overwintering Cacopsylla pyricola (Homoptera: Psyllidae). Environ. Entomol. 1990, 19, 544–550. [Google Scholar]
- Purvis, G.; Chauzar, M.P.; Segonds. Pichon, A.; Dunne, R. Life history and phenology of the eucalyptus psyllid, Ctenarytaina eucalypti in Ireland. Ann. Appl. Biol. 2002, 141, 283–292. [Google Scholar] [CrossRef]
- Wenninger, E.J.; Hall, D.G. Daily timing of mating and age at reproductive maturity in Diaphorina citri (Hemiptera: Psyllidae). Fla. Entomol. 2007, 90, 715–722. [Google Scholar] [CrossRef]
- Guédot, C.; Horton, D.R.; Landolt, P.J. Age at reproductive maturity and effect of age and time of day on sex attraction in the potato psyllid Bactericera cockerelli. Insect Sci. 2012, 19, 585–594. [Google Scholar] [CrossRef]
- Van den Berg, M.A.; Deacon, V.E.; Thomas, C.D. Ecology of the citrus psylla, Trioza erytreae (Hemiptera: Triozidae). 3. Mating, fertility and oviposition. Phytophylactica 1991, 23, 195–200. [Google Scholar]
- Percy, D.M.; Taylor, G.S.; Kennedy, M. Psyllid communication: Acoustic diversity, mate recognition and phylogenetic signal. Invertebr. Syst. 2006, 20, 431–445. [Google Scholar] [CrossRef]
- Tishechkin, D.Y.; Drosopoulos, S.; Claridge, M.F. Vibratory communication in Psylloidea (Hemiptera). In Insect Sounds and Communication; Drosopoulos, S., Claridge, M.F., Eds.; CRS Press: New York, NY, USA, 2005; pp. 357–363. [Google Scholar]
- Wenninger, E.J.; Hall, D.G. Importance of multiple mating to female reproductive output in Diaphorina citri. Physiol. Entomol. 2008, 33, 316–321. [Google Scholar] [CrossRef]
- Burts, E.C.; Fischer, W.R. Mating behavior, egg production, and egg fertility in the Pear psylla. J. Econ. Entomol. 1967, 60, 1297–1300. [Google Scholar]
- Tishechkin, D.Y. New data on vibratory communication in jumping plant lice of the families Aphalaridae and Triozidae (Homoptera, Psyllinea). Entomol. Rev. 2007, 87, 394–400. [Google Scholar] [CrossRef]
- Guédot, C.; Horton, D.R.; Landolt, P.J.; Munyaneza, J.E. Effect of mating on sex attraction in Bactericera cockerelli with evidence of refractoriness. Entomol. Exp. Appl. 2013, 149, 27–35. [Google Scholar] [CrossRef]
- Horton, D.R.; Lewis, T.M. Interplant movement by pear psylla (Homoptera: Psyllidae): Effects of sex ratio and reproductive status. J. Insect Behav. 1995, 8, 687–700. [Google Scholar] [CrossRef]
- Andersson, J.; Borg-Karlson, A.-K.; Wiklund, C. Sexual cooperation and conflict in butterflies: A male-transferred anti-aphrodisiac reduces harassment of recently mated females. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2000, 267, 1271–1275. [Google Scholar] [CrossRef]
- White, T.C.R. Some aspects of the life history, host selection, dispersal and ovipostion of adult Cardiaspina densitexta (Homoptera: Psyllidae). Aust. J. Zool. 1970, 18, 105–117. [Google Scholar] [CrossRef]
- Krysan, J.L. Laboratory study of mating behavior as related to diapause in overwintering Cacopsylla pyricola (Homoptera: Psyllidae). Environ. Entomol. 1990, 19, 551–557. [Google Scholar]
- Cook, P.P. Mating behaviour of Psylla pyricola Forster (Hom. Psyllidae). Pan-Pac. Entomol. 1963, 39, 175. [Google Scholar]
- Brown., R.L. Chemical and Behavioral Ecology of the Pear Psylla, Cacopsylla pyricola Förster (Hemiptera: Psyllidae). M.Sc. Thesis, Washington State University, Washington, DC, USA, 2008. [Google Scholar]
- Kozlowski, M.W.; Aoxiang, S. Ritual behaviors associated with spermatophore transfer in Deuterosminthurus bicinctus (Collembola: Bourletiellidae). J. Ethol. 2006, 24, 103–109. [Google Scholar] [CrossRef]
- Kessel, E.L. The mating activities of balloon flies. Syst. Biol. 1955, 4, 97–104. [Google Scholar]
- Nelson, C.M.; Nolen, T.G. Courtship song, male agonistic encounters, and female mate choice in the house cricket, Acheta domesticus (Orthoptera: Gryllidae). J. Insect Behav. 1997, 10, 557–570. [Google Scholar] [CrossRef]
- Knowlton, G.F.; Janes, M.J. Studies on the biology of Paratrioza cockerelli (Sulc). Ann. Entomol. Soc. Am. 1931, 24, 283–292. [Google Scholar]
- Yang, M.M.; Yang, C.T.; Chao, J.T. Reproductive isolation and taxonomy of two Taiwanese Paurocephala species (Homoptera: Psylloidea). Monogr. Taiwan Musuem 1986, 6, 177–203. [Google Scholar]
- Taylor, K.L. A possible stridulatory organ in some Psylloidea (Homoptera). Aust. J. Entomol. 1985, 24, 77–80. [Google Scholar] [CrossRef]
- Eben, A.; Mühlethaler, R.; Gross, J.; Hoch, H. First evidence of acoustic communication in the pear psyllid Cacopsylla pyri L. (Hemiptera: Psyllidae). J. Pest Sci. 2014, in press. [Google Scholar]
- Wenninger, E.J.; Hall, D.G.; Mankin, R.W. Vibrational communication between the sexes in Diaphorina citri (Hemiptera: Psyllidae). Ann. Entomol. Soc. Am. 2009, 102, 547–555. [Google Scholar] [CrossRef]
- Kristoffersen, L.; Hallberg, E.; Wallén, R.; Anderbrant, O. Sparse sensillar array on Trioza apicalis (Homoptera, Triozidae) antennae-an adaptation to high stimulus levels? Arthropod Struct. Dev. 2006, 35, 85–92. [Google Scholar] [CrossRef]
- Onagbola, E.O.; Meyer, W.L.; Boina, D.R.; Stelinski, L.L. Morphological characterization of the antennal sensilla of the Asian citrus psyllid, Diaphorina citri Kuwayama (Hemiptera: Psyllidae), with reference to their probable functions. Micron 2008, 39, 1184–1191. [Google Scholar] [CrossRef]
- Arras, J.; Hunter, W.; Bextine, B. Comparative analysis of antennae sensory arrays in asian citrus psyllid, Diaphorina citri, and potato psyllid, Bactericera cockerelli (Hemiptera). Southwest. Entomol. 2012, 37, 1–12. [Google Scholar] [CrossRef]
- Soroker, V.; Talebaev, S.; Harari, A.R.; Wesley, S.D. The role of chemical cues in host and mate location in the pear psylla Cacopsylla bidens (Homoptera: Psyllidae). J. Insect Behav. 2004, 17, 613–626. [Google Scholar] [CrossRef]
- Moran, V.C.; Brown, R.P. The antennae, host plant chemoreception and probing activity of the citrus psylla, Trioza erytreae (Del Guercio) (Homoptera: Psyllidae). J. Entomol. Soc. South. Afr. 1973, 36, 191–202. [Google Scholar]
- Valterová, I.; Nehlin, G.; Borg-Karlson, A.-K. Host plant chemistry and preferences in egg-laying Trioza apicalis (Homoptera, Psylloidea). Biochem. Syst. Ecol. 1997, 25, 477–491. [Google Scholar] [CrossRef]
- Horton, D.R.; Krysan, J.L. Host acceptance behavior of pear psylla (Homoptera: Psyllidae) affected by plant species, host deprivation, habituation, and eggload. Ann. Entomol. Soc. Am. 1991, 84, 612–627. [Google Scholar]
- Horton, D.R.; Landolt, P.J. Attraction of male pear psylla, Cacopsylla pyricola, to female infested pear shoots. Entomol. Exp. Appl. 2007, 123, 177–183. [Google Scholar] [CrossRef]
- Horton, D.R.; Guédot, C.; Landolt, P.J. Attraction of male summerform pear psylla to volatiles from female pear psylla: Effects of female age, mating status, and presence of host plant. Can. Entomol. 2008, 140, 184–191. [Google Scholar] [CrossRef]
- Guédot, C.; Horton, D.R.; Landolt, P.J. Attraction of male winterform pear psylla to female produced volatiles and to female extracts and evidence of male-male repellency. Entomol. Exp. Appl. 2009, 130, 191–197. [Google Scholar] [CrossRef]
- Brown, R.L.; Landolt, P.J.; Horton, D.R.; Zack, R.S. Attraction of Cacopsylla pyricola (Hemiptera: Psyllidae) to female psylla in pear orchards. Environ. Entomol. 2009, 38, 815–822. [Google Scholar] [CrossRef]
- Guédot, C.; Horton, D.R.; Landolt, P.J. Sex Attraction in Bactericera cockerelli (Hemiptera: Triozidae). Environ. Entomol. 2010, 39, 1302–1308. [Google Scholar] [CrossRef]
- Guédot, C.; Horton, D.R.; Landolt, P.J. Response of summerform pear psylla (Hemiptera: Psyllidae) to male-and female-produced odors. Can. Entomol. 2011, 143, 245. [Google Scholar] [CrossRef]
- Mann, R.S.; Rouseff, R.L.; Smoot, J.; Rao, N.; Meyer, W.L.; Lapointe, S.L.; Robbins, P.S.; Cha, D.; Linn, C.E.; Webster, F.X. Chemical and behavioral analysis of the cuticular hydrocarbons from Asian citrus psyllid, Diaphorina citri. Insect Sci. 2012, 20, 367–378. [Google Scholar]
- Horton, D.R.; Guédot, C.; Landolt, P.J. Diapause status of females affects attraction of male pear psylla, Cacopsylla pyricola, to volatiles from female infested pear shoots. Entomol. Exp. Appl. 2007, 123, 185–192. [Google Scholar] [CrossRef]
- Guédot, C.; Millar, J.G.; Horton, D.R.; Landolt, P.J. Identification of a sex attractant pheromone for male winterform pear psylla, Cacopsylla pyricola. J. Chem. Ecol. 2009, 35, 1437–1447. [Google Scholar] [CrossRef]
- Bradbury, J.W.; Vehrencamp, S.L. Chemical signals. In Principles of Animal Communication, 2nd ed.; Sinauer Associate Inc.: Massacusets, MA, USA, 1998; Volume 12, pp. 193–228. [Google Scholar]
- Taylor, K.L. The Australian Genera Cardiaspina Crawford and Hyalinaspis Taylor, (Homoptera: Psyllidae). Aust. J. Zool. 1962, 10, 307–348. [Google Scholar] [CrossRef]
- Carver, M. Distinctive motory behaviour in some adult psyllids (Homoptera: Psylloidea). Aust. J. Entomol. 1987, 26, 369–372. [Google Scholar] [CrossRef]
- Percy, D.M. Psyllid communication. Available online: http://www.psyllids.org/ (accessed on 5 July 2014).
- Campbell, K.G. Sound production by Psyllidae (Hemiptera). Aust. J. Entomol. 1964, 1, 3–4. [Google Scholar]
- Wenninger, E.J.; Stelinski, L.L.; Hall, D.G. Behavioral evidence for a female-produced sex attractant in Diaphorina citri. Entomol. Exp. Appl. 2008, 128, 450–459. [Google Scholar] [CrossRef]
- Ossiannilsson, F. Sound production in psyllids (Hem. Hom.). Opusc. Entomol. 1950, 15, 202. [Google Scholar]
- Niño, E.L.; Malka, O.; Hefetz, A.; Tarpy, D.R.; Grozinger, C.M. Chemical profiles of two pheromone glands are differentially regulated by distinct mating factors in honey bee queens (Apis mellifera L.). PLoS One 2013, 8, e78637. [Google Scholar]
- Ginzel, M.D.; Blomquist, G.J.; Millar, J.G.; Hanks, L.M. Role of contact pheromones in mate recognition in Xylotrechus colonus. J. Chem. Ecol. 2003, 29, 533–545. [Google Scholar] [CrossRef]
- Lockey, K.H. Insect cuticular hydrocarbons. Comp. Biochem. Physiol. 1980, 65, 457–462. [Google Scholar]
- Everaerts, C.; Farine, J.-P.; Cobb, M.; Ferveur, J.-F. Drosophila cuticular hydrocarbons revisited: Mating status alters cuticular profiles. PLoS One. 2010, 5, e9607. [Google Scholar]
- Monnin, T. Chemical recognition of reproductive status in social insects. Ann. Zool. Fenn. 2006, 43, 515–530. [Google Scholar]
- Kuo, T.-H.; Yew, J.Y.; Fedina, T.Y.; Dreisewerd, K.; Dierick, H.A.; Pletcher, S.D. Aging modulates cuticular hydrocarbons and sexual attractiveness in Drosophila melanogaster. J. Exp. Biol. 2012, 215, 814–821. [Google Scholar] [CrossRef]
- Heslop-Harrison, G. Sound production in the Homoptera with special reference to sound producing mechanisms in the Psyllidae. Ann. Mag. Nat. Hist. 1960, 3, 633–640. [Google Scholar] [CrossRef]
- Bailey, W.J. Insect duets: Underlying mechanisms and their evolution. Physiol. Entomol. 2003, 28, 157–174. [Google Scholar] [CrossRef]
- Claridge, M.F. Acoustic recognition signals: Barriers to hybridization in Homoptera Auchenorrhyncha. Can. J. Zool. 1990, 68, 1741–1746. [Google Scholar] [CrossRef]
- Webb, J.C.; Sivinski, J.; Litzkow, C. Acoustical behavior and sexual success in the Caribbean fruit fly, Anastrepha suspensa (Loew) (Diptera: Tephritidae). Environ. Entomol. 1984, 13, 650–656. [Google Scholar]
- Choe, D.-H.; Ramírez, S.R.; Tsutsui, N.D. A silica gel based method for extracting insect surface hydrocarbons. J. Chem. Ecol. 2012, 38, 176–187. [Google Scholar] [CrossRef]
- Ruther, J.; Döring, M.; Steiner, S. Cuticular hydrocarbons as contact sex pheromone in the parasitoid Dibrachys cavus. Entomol. Exp. Appl. 2011, 140, 59–68. [Google Scholar] [CrossRef]
- Olaniran, O.A.; Sudhakar, A.V.S.; Drijfhout, F.P.; Dublon, I.A.N.; Hall, D.R.; Hamilton, J.G.C.; Kirk, W.D.J. A male-predominant cuticular hydrocarbon, 7-methyltricosane, is used as a contact pheromone in the western flower thrips Frankliniella occidentalis. J. Chem. Ecol. 2013, 39, 559–568. [Google Scholar] [CrossRef]
- Guarino, S.; de Paswuale, C.; Peri, E.; Alonzo, G.; Colazza, S. Role of volatile and contact pheromones in the mating behaviour of Bagrada hilaris (Heteroptera: Pentatomidae). Eur. J. Entomol. 2008, 105, 613–617. [Google Scholar] [CrossRef]
- Taylor, G.S.; Austin, A.D.; Jennings, J.T.; Purcell, M.F.; Wheeler, G.S. Casuarinicola, a new genus of jumping plant lice (Hemiptera: Triozidae) from Casuarina (Casuarinaceae). Zootaxa 2010, 2601, 1–27. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lubanga, U.K.; Guédot, C.; Percy, D.M.; Steinbauer, M.J. Semiochemical and Vibrational Cues and Signals Mediating Mate Finding and Courtship in Psylloidea (Hemiptera): A Synthesis. Insects 2014, 5, 577-595. https://doi.org/10.3390/insects5030577
Lubanga UK, Guédot C, Percy DM, Steinbauer MJ. Semiochemical and Vibrational Cues and Signals Mediating Mate Finding and Courtship in Psylloidea (Hemiptera): A Synthesis. Insects. 2014; 5(3):577-595. https://doi.org/10.3390/insects5030577
Chicago/Turabian StyleLubanga, Umar K., Christelle Guédot, Diana M. Percy, and Martin J. Steinbauer. 2014. "Semiochemical and Vibrational Cues and Signals Mediating Mate Finding and Courtship in Psylloidea (Hemiptera): A Synthesis" Insects 5, no. 3: 577-595. https://doi.org/10.3390/insects5030577
APA StyleLubanga, U. K., Guédot, C., Percy, D. M., & Steinbauer, M. J. (2014). Semiochemical and Vibrational Cues and Signals Mediating Mate Finding and Courtship in Psylloidea (Hemiptera): A Synthesis. Insects, 5(3), 577-595. https://doi.org/10.3390/insects5030577




