Abstract
The Glossina hytrosavirus (family Hytrosaviridae) is a double-stranded DNA virus with rod-shaped, enveloped virions. Its 190 kbp genome encodes 160 putative open reading frames. The virus replicates in the nucleus, and acquires a fragile envelope in the cell cytoplasm. Glossina hytrosavirus was first isolated from hypertrophied salivary glands of the tsetse fly, Glossina pallidipes Austen (Diptera; Glossinidae) collected in Kenya in 1986. A certain proportion of laboratory G. pallidipes flies infected by Glossina hytrosavirus develop hypertrophied salivary glands and midgut epithelial cells, gonadal anomalies and distorted sex-ratios associated with reduced insemination rates, fecundity and lifespan. These symptoms are rare in wild tsetse populations. In East Africa, G. pallidipes is one of the most important vectors of African trypanosomosis, a debilitating zoonotic disease that afflicts 37 sub-Saharan African countries. There is a large arsenal of control tactics available to manage tsetse flies and the disease they transmit. The sterile insect technique (SIT) is a robust control tactic that has shown to be effective in eradicating tsetse populations when integrated with other control tactics in an area-wide integrated approach. The SIT requires production of sterile male flies in large production facilities. To supply sufficient numbers of sterile males for the SIT component against G. pallidipes, strategies have to be developed that enable the management of the Glossina hytrosavirus in the colonies. This review provides a historic chronology of the emergence and biogeography of Glossina hytrosavirus, and includes researches on the infectomics (defined here as the functional and structural genomics and proteomics) and pathobiology of the virus. Standard operation procedures for viral management in tsetse mass-rearing facilities are proposed and a future outlook is sketched.
Keywords:
Glossina; Musca; trypanosomosis; hytrosavirus; sterile insect technique; SIT; salivary gland hypertrophy; SGH 1. Introduction
Tsetse flies (Glossinidae: Diptera [1]) are important vectors of two debilitating diseases; the human African trypanosomosis (HAT or sleeping sickness), and African animal trypanosomosis (AAT or nagana) [2]. Tsetse flies and trypanosomoses render vast areas of agricultural land uninhabitable, especially during the rainy seasons [3]. Although over 30 species and sub-species of tsetse are described in the genus Glossina and most of which can transmit trypanosomoses, only 8–10 tsetse species are of medical and agricultural importance. The most important tsetse vectors are the riverine species (G. palpalis, G. fuscipes, and G. tachinoides) in Western and Central Africa and the savannah species (G. morsitans, G. austeni and G. pallidipes) in Eastern and Southern Africa [4]. Although tsetse fly fossils have been found in the 26-million-year-old shales of Florissant, Colorado, USA [5], to-date, tsetse flies are confined to Africa and in isolated populations on the Arabian Peninsula [6].
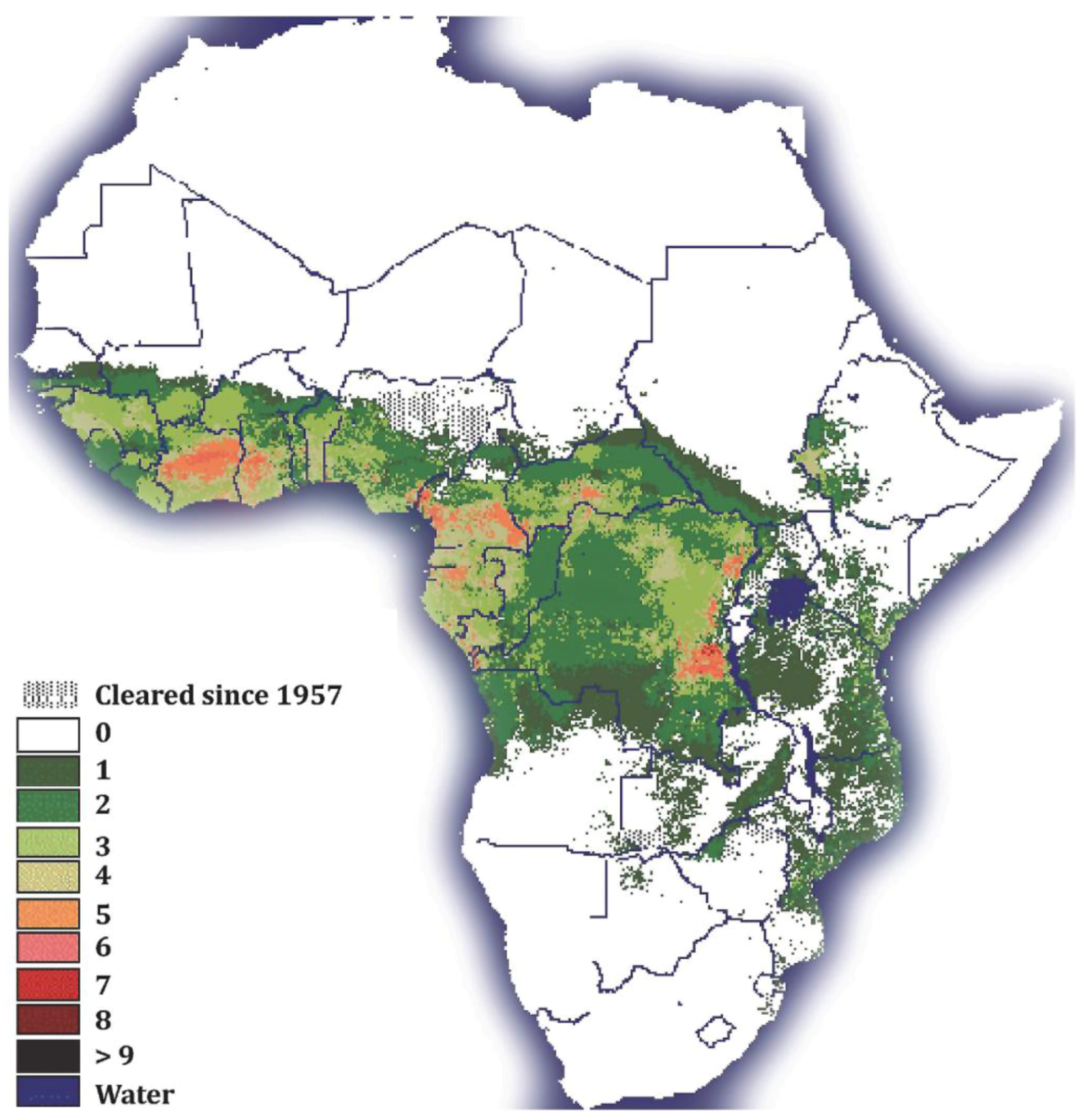
HAT is one of the most serious of the so-called “neglected tropical diseases” (NTDs [7]). NTDs are a group of chronic diseases endemic in low-income populations in Africa, Asia and the Americas [8]. Although trypanosomosis is restricted to 37 sub-Saharan African countries, its distribution extends to more than 10 million square kilometers of the African continent [9] (Figure 1).
The people at the highest risk of tsetse bites, and of contracting HAT are the rural populations that primarily depend on small-scale agriculture, fishing, animal husbandry and hunting. Resurgence and epidemics of HAT are often associated with economic decline, civil disturbance/wars, population movements and refugees [4]. The presence of tsetse and trypanosomosis is considered as one of the “roots of hunger and poverty” in sub-Saharan Africa [10]. It is estimated that approximately 90% of Africa’s livestock consists of herds in small villages [11]. This implies that maintaining healthy animals can be the difference between subsistence misery and a tolerable life for the herders and their families. The FAO estimates that ~US$ 4.75 billion worth of agricultural products are lost annually due to AAT (including ~3 million cattle deaths), and ≥100 human lives are lost daily due to HAT [12].
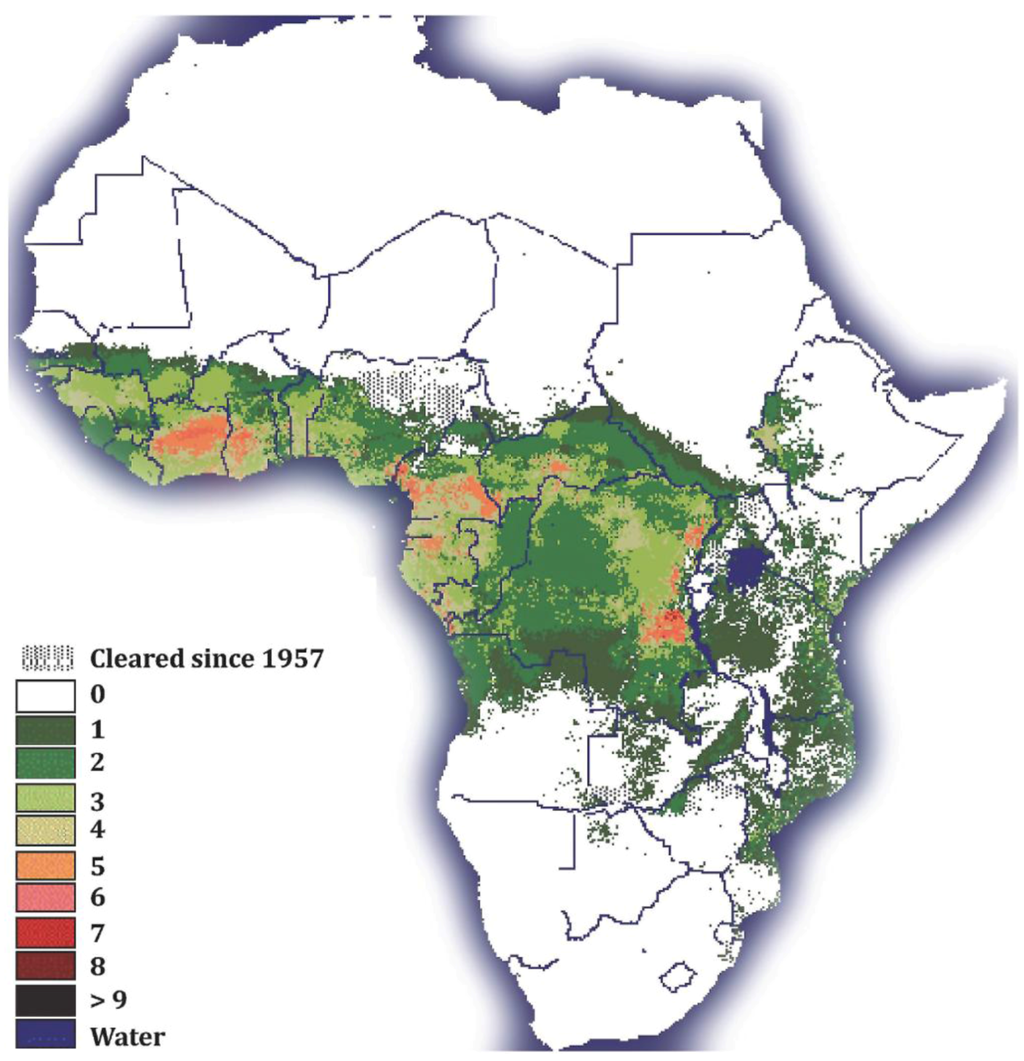
Figure 1.
Tsetse fly distribution in sub-Saharan Africa: The Figure legend shows the numbers of different tsetse species present in sub-Saharan African countries. Note that the colors in the figure legend correspond to the colors in the figure. (Map courtesy of the FAO).
Figure 1.
Tsetse fly distribution in sub-Saharan Africa: The Figure legend shows the numbers of different tsetse species present in sub-Saharan African countries. Note that the colors in the figure legend correspond to the colors in the figure. (Map courtesy of the FAO).
HAT is difficult to treat, and there are no effective vaccines are available against either AAT or HAT. None of the available trypanocidal drugs for HAT is ideal; their treatment schedules are prolonged, excruciatingly painful (often described by patients as “fire in the veins”), and require continuous hospitalization [13]. The tsetse-transmitted trypanosomes initiate their lifecycle by first colonizing the tsetse hosts’ midguts, then migrate into the ectoperitrophic space, and via the alimentary canal to the salivary glands or the mouth parts [14]. The parasites differentiate into the final mammalian-infective form (trypomastigocyte) in the tsetse salivary glands, and are then transmitted to the mammalian host by an infected tsetse bite [15]. It should however, be noted that some steps in the lifecycle of trypanosomes are group-specific. For instance, members of the T. vivax group only stays in the proboscis, the T. congolense group has a lifecycle involving the proboscis and the midguts, while only the T. brucei group has a cycle involving the salivary glands [15].
Without treatment, HAT can be fatal. However, fatalities of trypanosomes differ from one group to another. For instance, in West Africa T. b. gambiensis cause a chronic HAT that can take many years to kill a patient, while in East Africa, T. b. rhodiensis cause an acute HAT that can kill a patient within weeks [16,17]. The most widely used drug, melarsoprol, which was developed in 1949 [18], is lethal for up to 10% of the treated patients [19,20]. It is also important to note that, one of the biggest problems in the treatment of HAT is that the patients are usually so weak that they are more likely to die from the treatment rather than from the disease. In addition, patients need to be properly fed for several weeks to regain strength before commencement of treatments. This presents a very serious problem considering that there are hardly any available funds to properly feed or purchase drugs for HAT treatment. The available drugs for AAT are overly expensive for African peasant farmers and there are reports of increasing drug-resistance [21] and drug-counterfeiting [22]. For all the above-mentioned reasons, control of the disease vector (tsetse) is of critical importance, and likely represents the most sustainable method to manage trypanosomoses [23].
In this review, we describe the “evolution” of tsetse control methods: from traditional methods used in the early 20th Century, to modern control methods. The sterile insect technology (SIT) is considered an effective control method to eradicate tsetse populations within the frame of area-wide integrated pest management (AW-IPM) approaches [24]. Successful application of SIT to manage tsetse fly populations heavily depends on the maintenance of large tsetse colonies in mass-rearing facilities to supply the required numbers of sterile males. However, laboratory colonies of some tsetse species such as G. pallidipes are infected by Glossina hytrosavirus, a double-stranded (ds) DNA virus recently classified into the Hytrosaviridae family of insect viruses [25,26,27]. Virus infection often causes collapse of G. pallidipes colonies [28]. In this regard, we also present a historical overview of the knowledge gained about Glossina hytrosavirus, and consider the virology, epidemiology, pathology, and prospects for control of viral infections in laboratory colonies of G. pallidipes.
2. An Overview of Tsetse Fly Control Methods
Two main characteristics of tsetse render them suitable for eradication. Firstly, compared to other insects of medical and agricultural importance, tsetse flies have a very low reproduction rate (k-strategists) [29,30]. Therefore, unlike many insect vectors which produce large numbers of eggs (r-strategist) [30], tsetse flies have limited capacity to rebound in areas where their populations have been reduced. Secondly, tsetse flies are adapted for efficient exploitation of stable habitats offered by vertebrate nests or human dwellings with low levels of cross-breeding. This means that tsetse flies have reduced genetic variability within each vector population and therefore, have limited capacity to respond through selection pressures to various control interventions [31].
Tsetse control methods have evolved from discriminate bush clearing and wild game culling at the beginning of the 20th Century, to broadcast insecticide applications after the Second World War [32], traps, insecticide-impregnated targets [33] and live bait technologies [34]. Although these methods have been successfully used to locally reduce tsetse population sizes [29], each of these methods has limitations. Firstly, the methods do not protect the cleared areas from re-invasion by tsetse flies from residual pockets and from neighboring territories [35]. Secondly, the methods are applied in administratively-defined regions and run for an administratively-specified time [23], which mostly depend on how long external donor funds are available for the projects. Since the methods cannot be sustained beyond the time of these external donor-funded projects, the risk of the cleared areas being re-infested by tsetse flies increases. In the light of these developments, there was a need to explore other tsetse control methods.
In 1937, Knipling developed the theory of controlling insect pest population by manipulating their reproductive capacity. He likewise modeled that a target population could be eradicated when the release of sterile males was applied on an area-wide basis against an entire insect pest populations in a delineated area [36,37]. This method, commonly known as the sterile insect technique (SIT), involves large-scale production of insects in laboratory colonies in mass-rearing facilities. The male flies are then sexually-sterilized by exposure to a precise and specific dose of ionizing radiation, usually from a 60Co or 137Ce source [38,39]. The sterile males are then sequentially released into the target insect population in numbers that allows them to out-compete wild type males for wild virgin females [40]. After the virgin females mate with the sterile males, embryogenesis is arrested and consequently no viable offspring is produced. When the release of the sterile males is sustained, the size of the target insect population declines and can become extinct. The SIT is a robust control tactic that has been used very successfully against insect pests that are important in agriculture and trade. For instance, SIT was used to control Mediterranean fruit fly Ceratitis capitata (Diptera: Tephritidae) populations in Chile, Argentina, Mexico, Central America, South Africa, Israel etc. [41,42]. Lately, the SIT has also been used with great success against several lepidopteran pests such as the codling moth Cydia pomonella (L.) (Lepidoptera: Tortricidae) in the Okanagan Valley of Canada [43], the false codling moth Thaumatotibia leucotreta (Meyrick) (Lepidoptera: Tortricidae) in South Africa [44], the Australian painted apple moth Teia anartoides Walker (Lepidoptera: Lymantriidae) in New Zealand [45], and the pink bollworm Pectinophora gossypiella (Saunders) (Lepidoptera: Gelechiidae) in Texas, New Mexico, Arizona, California (US) and in Sonora and Chihuahua of northern Mexico [41,46].
The SIT played a pivotal role in the sustainable eradication of the tsetse fly Glossina austeni from the Unguja Island (Zanzibar) [47,48]. This program was preceded by successful applications of the technique against G. palpalis gambiensis and G. tachinoides in the Sideradougou area in Burkina Faso, and against G. palpalis palpalis in the Lafia area of Nigeria [49,50]. The programs in Burkina Faso and Nigeria were, however, not implemented according to AW-IPM principles and the tsetse-cleared area was re-invaded after the programs were completed. Following the area-wide eradication of G. austeni the island was declared tsetse-free in 1997, it still is to-date.
The success of SIT in eradicating G. austeni and trypanosomosis from Unguja Island inspired African Governments to call for increased efforts to manage the tsetse fly and trypanosomosis on mainland Africa. Consequently, an AW-IMP program with an SIT component was initiated in 1997 to eradicate G. pallidipes from a 25,000 square kilometers of under-utilized fertile land in the Southern Rift Valley of Ethiopia [51]. For this Ethiopian SIT program, a laboratory colony of G. pallidipes was established in 1997 at the Insect Pest Control Laboratories (IPCL) of the Joint FAO/IAEA Seibersdorf Laboratories, Austria. This colony was initiated from pupae originating from Arba Minch, Ethiopia. However, the colony collapsed in 2002. After the collapse of the colony, more pupae were shipped from Arba Minch to IPCL, Seibersdorf in an attempt to re-establish the colony. However, these attempts have been largely unsuccessful: currently, only a few of these flies (n = 25) are surviving, and their fecundity is very low. Further investigations revealed that the colony collapse was due to infection by a virus that caused salivary gland hypertrophy (SGH) syndrome [52,53,54]. A chronology of the emergence of the SGH syndrome and the discovery of the virus that causes the syndrome is discussed in Section 3: a summary is presented in in Table 1.

Table 1.
Chronological history of the discovery and distribution of hytrosaviruses.
| Investigator(s) | Year | Major contribution(s) | Ref. |
|---|---|---|---|
| Whitnall | 1932, 34 | First published record of SGH Glossina spp. | [55,56] |
| Burtt | 1945 | Suggested that SGH is sex-linked | [57] |
| Jenni et al. | 1973, 74, 76 | Described virus particles in G. morsitans and G. fuscipes fuscipes; suggested Golgi-ER viral assembly | [58,59,60,61] |
| Lyon | 1973 | First published record of SGH in M. equestris | [62] |
| Jaenson | 1978 | First clear association of viral particles with SGH | [63] |
| Amargier et al. | 1979 | Reported SGH in M. equestris | [64] |
| Otieno et al. | 1980 | Reported SGH as common feature in wild G. pallidipes | [65] |
| Opiyo | 1983 | Reported poor productivity of G. pallidipes colony at Kenya Trypanosomosis Research Institute (KETRI), Kenya | [66] |
| Odindo et al. | 1981, 83, 86 | Demonstrated that viral particles are infectious per os; First report that Glossina virus has dsDNA genome | [67,68,69,70] |
| Jaenson | 1986 | First report on reduced insemination rates, fecundity and lifespan in laboratory colonies of G. pallidipes | [71] |
| Ellis et al. | 1987 | Reported SGH in Zimbabwe and Ivory Coast | [72,73] |
| International Atomic Energy Agency | 1987, 89 | Reported poor productivity of G. pallidipes colonies at IPCL, Seibersdorf, Austria | |
| Odindo | 1988 | Proposed Glossina virus as a bio-control agent | [74] |
| Jura et al. | 1988, 89, 92, 93 | Demonstrated transmission of Glossina virus after artificial infection | [75,76,77,78] |
| Kokwaro et al. | 1990–1991 | Cytopathology of virus particles in tsetse salivary glands | [79,80] |
| Shaw | 1993 | Reported SGH in G. m. swyenatoni and G. brevipalpis | [81] |
| Coler et al. | 1993 | First published record of SGH in M. domestica | [82] |
| Sang | 1996–1999 | Reported SGHV in tsetse milk glands, mid-gut and male accessory reproductive glands | [83,84,85,86] |
| International Atomic Energy Agency | 2002 | Collapse of an Ethiopian-derived G. pallidipes colony at IPCL, Seibersdorf, Austria | |
| Kokwaro | 2006 | Reported viral particles in male accessory reproductive glands of G. m. morsitans Westwood | [87] |
| Abd-Alla et al.; Garcia-Maruniak et al. | 2008 | G. pallidipes and M. domestica SGHVs genome sequenced | [26,27] |
| Abd-Alla et al. | 2009 | Establishment Hytrosaviridae family | [27] |
| Salem et al. | 2009 | Transcription analysis of M. domestica SGHV | [88] |
| Kariithi et al. | 2010–2013 | Described proteome and morphogenesis of Glossina SGHV | [89,90] |
| Prompiboon et al. | 2010 | Reported wild-wide distribution of SGHV in M. domestica | [91] |
| Luo and Zheng | 2010 | SGHV-like virus described in accessory gland filaments of the parasitic braconid wasp, D. longicuadata | [92] |
| Boucias et al. | 2013 | Described the role of endosymbionts on trans generational trans mission of SGHV in G. pallidipes | [93] |
| Abd-Alla et al. | 2013 | Reported successful management of Glossina hytrosavirus and eradication of SGH in G. pallidipes colonies at IPCL, Seibersdorf | [94] |
3. A Historic Chronology of the Discovery and Biogeography of SGH
3.1. SGH in Dipteran Insects
In the 1930s, Whitnall reported that some G. pallidipes individuals collected in the Umfolosi Game Reserve, Zululand, South Africa, had grossly enlarged salivary glands [55]. In the 1970s, the enlargement of salivary glands was shown to be associated with a virus found in cytoplasmic vacuoles of the salivary gland and midgut epithelial cells of G. fuscipes and G. morsitans [56,57,58,59]. The virus was at that time described as “virus-like particles” (VLPs), morphologically resembling the VLPs previously described in Drosophila, mosquitoes, and nematodes [58]. The virus detected in tsetse was erroneously suggested to be an arbovirus because other hematophagous insects (mosquitoes, ticks, sandflies and gnats) had been widely known to transmit arboviruses [57].
There were several notable features that supported the suggestion of the tsetse virus as an arbovirus. For instance, the rod-shaped viral particles detected in tsetse were thought to resemble one non-typical arbovirus group, the vesicular stomatitis Indiana virus, which was found to be rod-shaped with distinctive helical symmetry [95,96]. Other notable features included the size of the viral particles, replication in the host salivary glands, and secretion of mature virions via saliva [97,98,99,100]. In addition to resemblance to arboviruses, it was also suggested that the tsetse virus was morphologically similar to baculoviruses [63].
In the 1980s and 90s, several researchers suggested that the tsetse virus is maintained in nature via transmission from the mother to her progeny and not from the male parent [71,75,83,85]. However, father-to-progeny viral transmission cannot be totally ruled out. Possibly, the lack of detectable father-to-progeny viral transmission may result from reduced transmission rates, rather than a total failure of transmission. The phenomenon of father-to-progeny virus transmission has been demonstrated in other insect viruses. For instance, in the sigma viruses (Rhabdoviridae), the apparent failure of father-to-progeny transmission of the sigma virus in Drosophila was attributed to transfer of very low viral titers to the developing embryo via the sperm [101]. In the case of sigma viruses, the virus fails to infect early germ cells and thus prevents gamete-infection, even though viral titers in the somatic cells may increase later when the fly reaches adulthood [102]. Although the Glossina hytrosavirus is transferred from mother to her progeny, father-to-progeny transmission cannot be totally ruled out. This revelation implies that, under undefined conditions, the virus is reactivated from a “latent” state to a symptomatic state [68]. It was proposed that the virus-induced abnormalities were contributing to the natural regulation of tsetse populations in the field [68]. This suggestion appears to be supported by reports that related viruses such as baculoviruses regulate the population dynamics of their insect hosts in a density-dependent manner, some of which do not induce disease symptoms [103].
SGH symptoms have been reported in two other dipteran insects: in adult populations of the narcissus bulb fly, Merodon equestris (Diptera; Syrphidae) [62,64], and in infections in the house fly, Musca domestica L. (Diptera; Muscidae) collected at a dairy in Florida, USA [82]. Notably, the main disease symptoms (SGH) of the virus in the bulb fly and in the house fly were similar to those observed in the tsetse [62]. To date, there has been no further research performed on the Merodon virus. Although the Glossina and Musca viruses share pathological impacts on their respective hosts such as seasonal fluctuations in the incidence of SGH symptoms [68,91] and suppression of reproductive fitness in the host [75,82,85,86], the two hytrosaviruses differ in several aspects. For instance, unlike the tsetse virus, the house fly virus appears not to be maintained in nature by mother-to-progeny transmission [104,105]. Notably, whereas there is no documented evidence that the tsetse virus has potential to infect heterologous hosts, the house fly virus can infect, reduce egg production and the lifespan in the stable fly, Stomoxys calcitrans (Diptera; Muscidae) [106], but it did not induce expression of SGH symptoms in S. calcitrans [106].
3.2. Possible Hytrosaviruses in other Insect Species
Recently, a virus was fortuitously detected in hypertrophied accessory gland filaments (AGFs) of the parasitic wasp, Diachasmimorpha longicuadata (Ashmead) (Hymenoptera; Braconidae) [92]. The virus, presumed to cause the hypertrophy of the AGFs had ultra-structural features similar to those causing SGH in dipteran hosts [92]. The wasp virus was detected in a D. longicuadata that originated from Hawaii and released in Thailand and subsequently introduced to southern China as a bio-control agent for the oriental fruit fly, Bactrocera dorsalis (Hendel) (Diptera; Tephritidae) [92]. Inferring from other insect-viral systems, this discovery of the virus in the wasp may be significant. B. dorsalis is distributed throughout Southeast Asia and the Pacific, and is considered to be one of the most prominent agricultural pests in this part of the globe [107]. D. longicuadata is known to introduce D. longicuadata entomopoxvirus (DlEPV) into Caribbean fruit fly Anastrepha suspensa (Diptera: Tephritidae) during oviposition [108]. DlEPV, the first reported endosymbiotic entomopoxvirus, protects the wasp’s eggs by inhibiting encapsulation by the host’s hemocytes [109]. Consequently, one might argue that, similar to the DlEPV, the virus detected in D. longicuadata may potentially be an endosymbiont to the wasp. Unfortunately, similar to the Merodon virus, the virus detected in the AGFs of D. longicuadata has not been further investigated.
4. Pathology of Hytrosaviruses
As indicated in Section 3, dipteran adults infected by hytrosaviruses can exhibit overt SGH symptoms. Flies with SGH are recognizable to the naked eye by the swollen opaque-white appearance of the abdomen, and sac-like course textures [55,60] (Figure 2).
Notably, whereas the SGH syndrome in Glossina mainly results from cellular hypertrophy (enlargement), SGH in Musca reflects a combined effect of both cellular and nuclear hypertrophy. Pathological effects of the Glossina hytrosavirus in colonized G. pallidipes were first documented the early 1980s. In 1979, a G. pallidipes colony was initiated at the Kenya Trypanosomosis Research Institute (KETRI) using flies caught from the Kibwezi forest, Kenya. This colony collapsed within two years of its establishment due to poor productivity [66]. Investigations were done to identify possible causes of the poor performance and eventual collapse of the colony. The study parameters included dissections of female spermathecae (to check insemination rates) and male testes (to check presence of motile spermatozoa as an indicator of male efficiency) [66]. The results demonstrated deterioration of insemination in females, aspermia, reversed ovariole development, and distortion of sex ratio. Other hytrosavirus-induced collapses of G. pallidipes colonies have been reported over the last two decades [28,52]. The colony collapses are a direct consequence of testicular degeneration (in males) and ovarian abnormalities (in females).
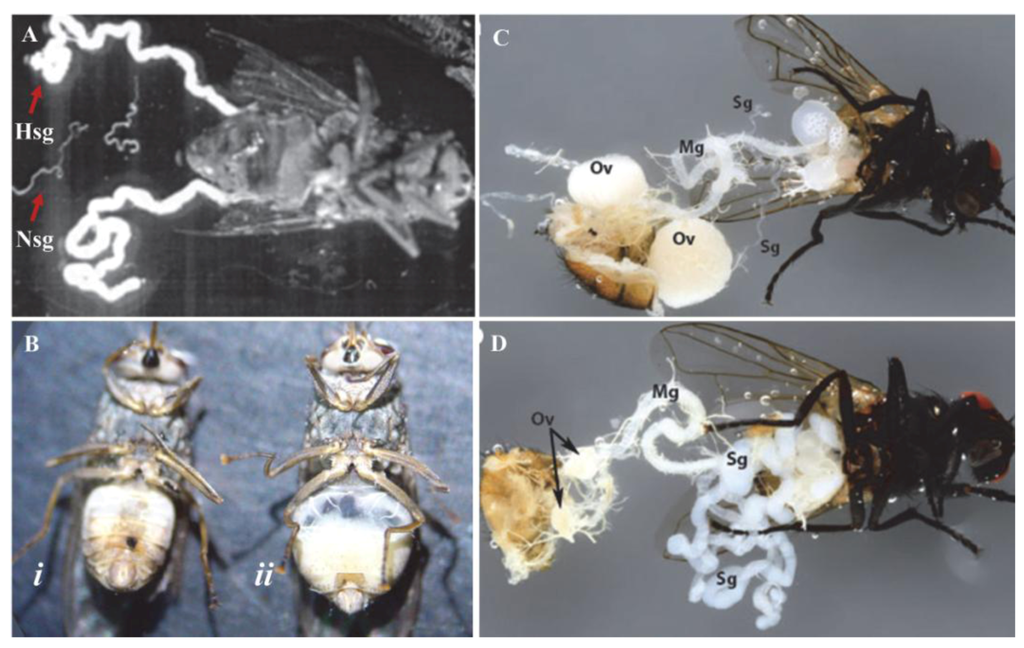
Figure 2.
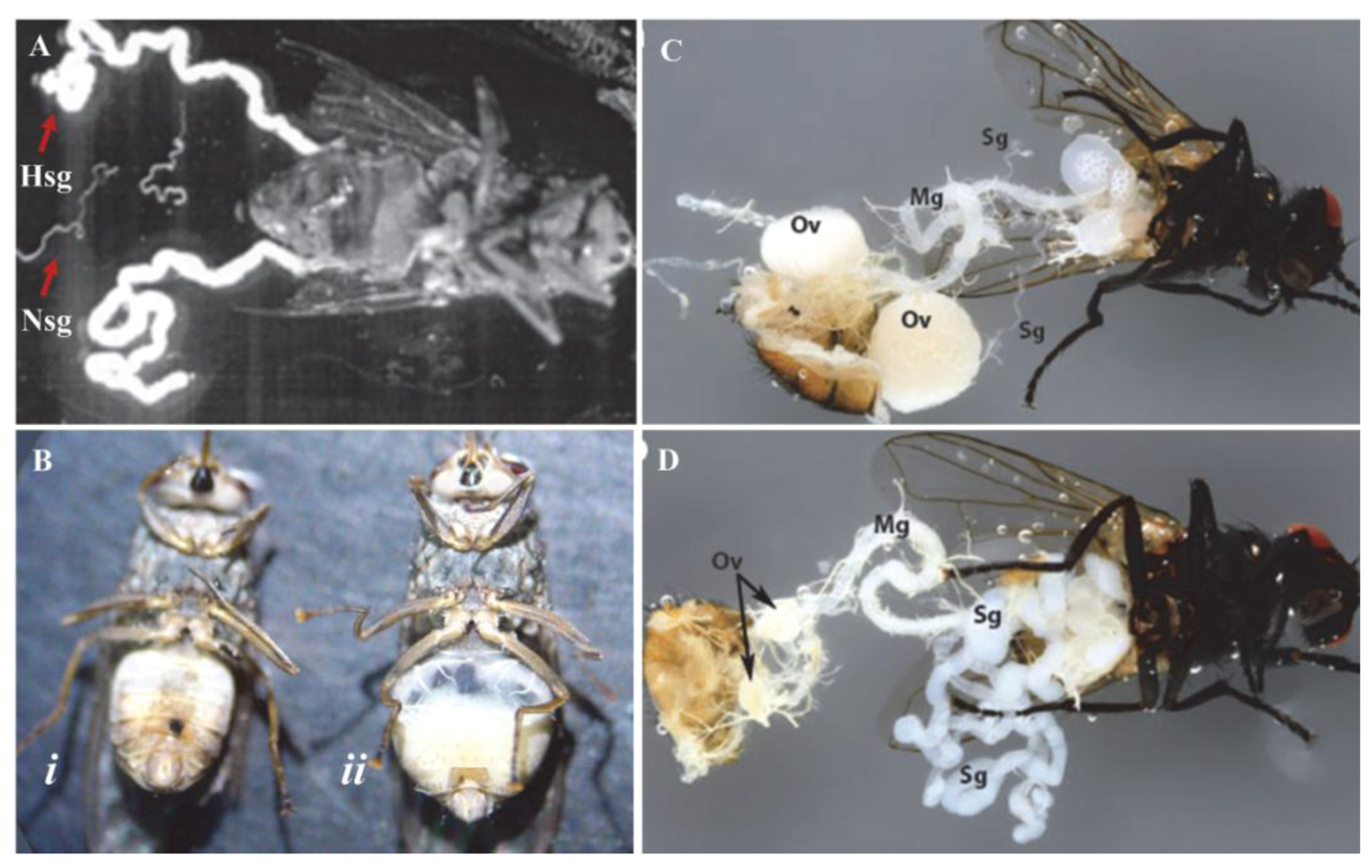
Pathology of hytrosaviruses: (A) Normal (Nsg) and hypertrophied (Hsg) salivary glands dissected from G. pallidipes. It should be noted that the pair of Nsg are dissected from a different fly for comparison with the Hsg. Notice that the glands exhibiting salivary gland hypertrophy (SGH) symptoms are enlarged ≥5 times the size of normal glands; (B) Male G. pallidipes with asymptomatic (i) and symptomatic (ii) salivary glands; (C) Female M. domestica with healthy and (D) hypertrophied salivary glands showing lack of ovarian development in the virus-infected fly (D). Abbreviations: Mg, midgut; Ov, ovary; Sg, salivary gland. (Figure sources: Panel A [28]; panel B [110]; panels C and D [111]; used with permission).
Figure 2.
Pathology of hytrosaviruses: (A) Normal (Nsg) and hypertrophied (Hsg) salivary glands dissected from G. pallidipes. It should be noted that the pair of Nsg are dissected from a different fly for comparison with the Hsg. Notice that the glands exhibiting salivary gland hypertrophy (SGH) symptoms are enlarged ≥5 times the size of normal glands; (B) Male G. pallidipes with asymptomatic (i) and symptomatic (ii) salivary glands; (C) Female M. domestica with healthy and (D) hypertrophied salivary glands showing lack of ovarian development in the virus-infected fly (D). Abbreviations: Mg, midgut; Ov, ovary; Sg, salivary gland. (Figure sources: Panel A [28]; panel B [110]; panels C and D [111]; used with permission).
One of the outstanding questions to be answered is: why does the Glossina hytrosavirus cause such serious problems in colonized G. pallidipes? It should be noted that such serious impacts are yet to be reported in other tsetse species. Further, other insect cultures (e.g., Drosophila) do not seem to experience such virus-induced problems. Although this question needs more research, inferences can be made from other viral-insect systems to understand the case of Glossina hytrosavirus pathology. It is important to keep in mind that insect viruses hardly “walk alone”: viruses co-infect (simultaneously) their hosts with other micro-organisms such as the vertically-transmitted bacterial endosymbionts [112,113,114]. Population dynamics and roles of endosymbionts on viral infections have been documented in the infection of Drosophila by sigma viruses. Boucias et al., have recently suggested that the pathological effects of Glossina hytrosavirus on colonized G. pallidipes flies are modulated by interplay between the virus and tsetse endosymbionts [93]. Specifically, the researchers highlighted the absence of Wolbachia in the laboratory stock of G. pallidipes that was used in the study. Although Glossina hytrosavirus has been reported in other Wolbachia-harboring tsetse species [115], so far, no harmful impacts have been reported on the virus-infected tsetse fly species that harbor Wolbachia. It has been reported that the levels of PCR-detectable Glossina hytrosavirus in G. fuscipes fuscipes populations are influenced by the tsetse genotype, and inversely correlate with Wolbachia prevalence [116]. Notably, Wolbachia is also absent from some of the major economically and medically important species of mosquitoes such as Anopheles spp. [117]. Studies have also demonstrated that while none of the Wolbachia-infected Ae. aegypti tested positive for Dengue virus after oral infection, 30%–100% of Wolbachia-free mosquitoes were virus-infected [118]. It can therefore, be hypothesized that the absence of Wolbachia in the G. pallidipes colonies explains the severe negative impact of Glossina hytrosavirus on large-scale colonies. Considering the shared intracellular locations of Glossina hytrosavirus and Wolbachia, this endosymbiont may influence the outcome of viral infection in tsetse hosts.
5. Virology of the Hytrosaviruses
5.1. Genomics of Hytrosaviruses
To understand the pathobiology of Glossina hytrosavirus, the virus was purified from hypertrophied salivary glands dissected from G. pallidipes flies originating from Tororo, Uganda in 1975. This colony was initially maintained at Leiden University, The Netherlands, and subsequently transferred to IPCL, Seibersdorf, Austria in 1982. Twenty-six years later, the genome of the virus was fully sequenced (NC_010356.1) [26]. The 190 kbp-long viral genome is a circular dsDNA molecule [26] (Figure 3), and represents an entirely new group of insect viruses. A total of 322 non-overlapping open reading frames (ORFs) were identified, of which 160 ORFs were presumed to encode viral proteins. One hundred thirteen (70.6%) of these ORFs did not match to any of the sequences available in the various databases [26]. Thirty-seven ORFs (23.1%) were homologues to genes of other viruses, while ten (6.3%) were homologues to non-viral/cellular genes. Most notable of the Glossina virus ORFs was the presence of homologues to five of the so-called per os infectivity factor genes (pifs) (p74, pif-1, pif-2, pif-3, and odv-e66) encoded by baculoviruses, nudiviruses and whispoviruses [119]. Other notable homologies included homologues to sixteen (entomo-) poxvirus genes, three iridovirus and nimavirus genes each, two ascovirus genes and one herpesvirus gene. Homologues to cellular genes included chitinase, DNA helicases, thymidylate synthases, and several bacterial endosymbionts genes [26]. Approximately 3% of the Glossina virus genome is characterized by an inverted repeat (ir) sequence, and fourteen direct repeat sequences (drs) composed of 51–246 bp.
The Musca virus genome has also been sequenced [120]. The viral genome is a ~124 kbp-long circular dsDNA molecule [120], with a total of 108 putative ORFs. Similar to the Glossina virus, over 70% (76/108) of the putative Musca virus ORFs had no significant homologies to proteins available in various databases. Notably, the two viruses share at least eight homologs of baculovirus core genes, including homologues to the pif genes, and to cellular genes [120]. Eighteen tandem drs were distributed throughout the viral genome [120]. The size of the repeated sequences ranged from 149 bp (dr15) to only 9 bp long (dr18), and the number of copies of the drs ranged from 1.9 to 7.4, making the total sizes of the drs from 30 bp–380 bp-long. The G + C content of Musca virus is 43.5%, a ratio similar to that found in several nudiviruses: Heliothis zea nudivirus-1 (HzNV-1) and Oryctes rhinoceros nudivirus (OrNV), but significantly higher than that of Gryllus bimaculatus nudivirus (GbNV; 28%) and the Glossina virus (28%) [121,122,123].
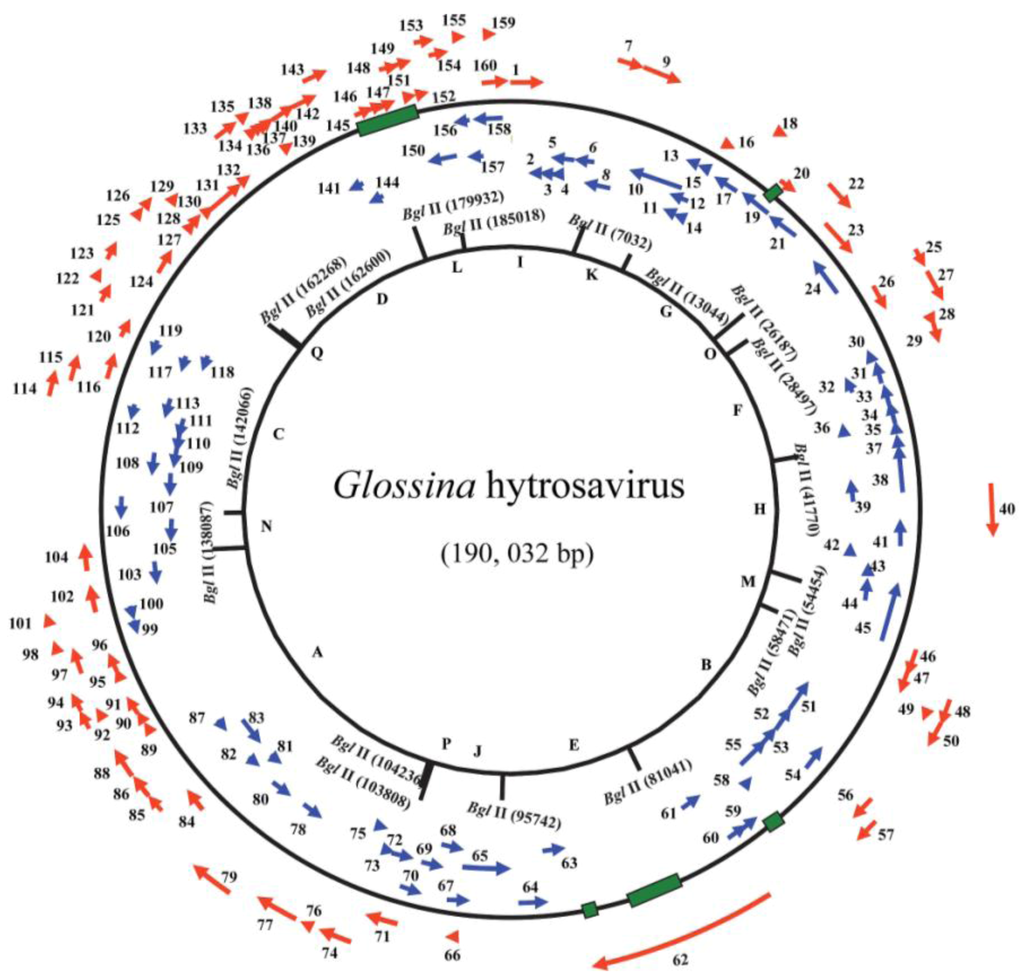
Figure 3.
Circular representation of the Glossina hytrosavirus genome: Arrows indicate positions and directions of transcription for the putative open reading frames (ORFs). The ORF numbers and putative genes are shown. The alphabetical numbers represent restriction fragments generated by BglII enzyme during the electrophoretic profiling of the viral genome.
Figure 3.
Circular representation of the Glossina hytrosavirus genome: Arrows indicate positions and directions of transcription for the putative open reading frames (ORFs). The ORF numbers and putative genes are shown. The alphabetical numbers represent restriction fragments generated by BglII enzyme during the electrophoretic profiling of the viral genome.
The above-mentioned hytrosavirus gene homologies to other insect viral genes have several implications. Firstly, the conserved nature of the pifs among all the baculoviruses that have so far been sequenced [124], and their importance in early oral infection [125], may reflect a common ancestry or similar modes of transmission and infection of the hytrosaviruses, baculoviruses, nudiviruses and nimaviruses. Secondly, the presence of repeat regions in the genomes of the these hytrosaviruses possibly reflects a common mode of transcriptional regulation and DNA replication [126]. Thirdly, the presence of the ORFs potentially encoding orthologues of cellular proteins may be advantageous for the viral infection. For instance, it has been suggested that large DNA viruses gain independence of the transcription and replication machineries of their hosts by encoding their own functional cellular orthologues of cellular proteins, mostly enzymes [127]. It remains to be ascertained whether the Glossina and Musca hytrosavirus ORFs do encode functional cellular orthologues, and what their functional roles are.
5.2. Classification and Phylogeny of Hytrosaviruses
Initially, the Glossina virus could not be assigned to any of the families of insect DNA viruses described at that time [69]. Accumulated data from various studies on the viral pathobiology singled-out signature of hytrosaviruses are shown in Table 2. Notably, in contrast to many other invertebrate large dsDNA viruses such as baculoviruses and entomopoxviruses, virions of hytrosavirus are not “packaged” in occlusion bodies. Based on these characteristics, the virus was proposed to be accommodated into a new virus family, Hytrosaviridae [27], a name derived from “Hypertrophia sialoadenitis”, the Greek word for “salivary gland inflammation”. The Glossina virus is now a member of the newly-established Hytrosaviridae family, genus Glossinavirus, as the species Glossina hytrosavirus [27,128]. Similarly, based on the shared characteristics between Glossina and Musca viruses, the Musca virus is the second member of Hytrosaviridae family, but accommodated in the genus Muscavirus, and species Musca hytrosavirus. Hereafter, the Glossina and Musca hytrosaviruses are abbreviated as GpSGHV and MdSGHV, respectively.

Table 2.
Signature characteristics of hytrosaviruses: The table summarizes the principal biological, structural and molecular characteristics of the GpSGHV and MdSGHV. Table modified from [129]; used with permission.
| Key characteristics | GpSGHV | MdSGHV | Ref. | |
|---|---|---|---|---|
| Biological | Replication site(s) | Salivary glands, milk glands | Salivary glands | [75,82,83,104,130] |
| Infection phenotype | Symptomatic; asymptomatic | symptomatic | [28,93] | |
| Symptoms besides SGH | Male/female gonadal abnormities | Under-developed ovaries | [75,82,93,105] | |
| Vertical (trans-generational) transmission | Milk glands, trans-ovarian | No evid ence available to-date | [52,75,82,93,105] | |
| Horizontal transmission | Oral (salivary) secretions | Oral (salivary) secretions and excreta | [25,52,131] | |
| Sterilizing agent | Male and female infertility | Female infertility | [86,105] | |
| Impact on host behavior | Impaired feeding | Mating disruption | [84,105,132] | |
| Morphogenesis | Cytoplasmic envelopment, egress by disintegration or rapture of the plasma membrane | Cytoplasmic, egress via budding on the plasma membrane | [93,133,134] | |
| Structural | Virion size | 50 × 1000 nm | 65 × 550 nm | [120,134] |
| Ultra-structure | Nucleocapsid, tegument, envelop, outer surface projections | Nucleocapsid, envelop | [89] | |
| Virion topography | Helical surface projections | Braided, bead-like surface | [89,120] | |
| Molecular | Genome size | 190,032 bp | 124,279 bp | [26,120] |
| G + C content (%) | 28 | 44 | [26,120] | |
| No. of RFs | 160 | 108 | [26,120] | |
| Shared ORFs between GpSGHV and MdSGHV | 41 | 37 | [90,128] | |
| ORFs homologs in other large dsDNA viruses | Nudivirus (11), whispovirus (4), baculovirus (12) | Nudivirus (17), whispovirus (6), baculovirus (12) | [26,120] | |
Phylogenetic analysis of GpSGHV and MdSGHV based on the DNA polymerase gene (dna pol), which is present in all large dsDNA viruses, does not cluster these hytrosaviruses with other insect dsDNA viruses [26,129]. Instead, the dna pol of GpSGHV and MdSGHV clusters more closely to that of herpesviruses and other viruses with linear dsDNA genomes. On the other hand, the alignments free method using whole proteome phylogenetic analyses of dsDNA viruses shows close association of the hytrosaviruses and nimaviruses (specifically the white spot syndrome virus; WSSV) [135,136,137]. Despite the apparent ambiguities, these and other phylogenetic methods, such as super tree and super matrix methods [138,139], support the notion of a common ancestry of GpSGHV and MdSGHV with baculoviruses, nudiviruses and nimaviruses [124,139]. The hytrosaviruses share 12 out of the 31 baculovirus core genes identified to date [139], and are therefore, more distantly-related to baculoviruses than for instance the nudiviruses: Nudiviruses share 20 of baculovirus core genes [124].
5.3. Proteomics and Interactomics of Hytrosaviruses
The presence of protein-coding regions or ORFs in a viral genome does not necessarily imply the presence of functional proteins. For instance, frame shift mutations caused by “indels” (insertions/deletions) in viral genomes can alter the structure and functions of the encoded proteins resulting in decrease or complete loss in protein expression [140]. Further, some genes may not have functional promoters.
Proteomics can be used to validate presence of virally-encoded proteins. Although the repertoire of biological processes controlled by viral gene products is complex [141], proteomic analysis of the GpSGHV made it possible to reconstruct a dynamic view of the viral infection process [89,90]. A combined approach of proteomics and electron microscopy revealed that the GpSGHV virion consists of four morphologically distinct structures: nucleocapsid, tegument, envelope, and helical surface projections. Almost 50% of the virally encoded proteins were found to reside within the viral tegument, reflecting potential roles in cytoplasmic trafficking of the virions during infection [89]. A limited proteome of the MdSGHV proteome is also available [133]. Although GpSGHV and MdSGHV share more than thirty protein homologues [90], the ultra-structural features of the MdSGHV differ from GpSGHV, but have not been studied in detail. GpSGHV also incorporates cellular proteins derived from infected cells, which may significantly contribute to the viral morphogenesis [89]. It is not yet known whether MdSGHV incorporates any host-derived cellular proteins into its mature virion, but it is very likely.
Equally important to the repertoire of viral proteins composing the viral proteome are the interactions encrypted by both the viral and host genomes. Analyses of the secretome of GpSGHV-infected G. pallidipes revealed that up to twenty of the secreted host proteins potentially interact with at least twenty-five viral proteins [142]. GpSGHV alters the protein expression patterns in the host salivary glands, and ≥40% of both virally-encoded and host proteins are specifically expressed in the symptomatic flies but not in asymptomatic flies [142]. The GpSGHV-host are important because, in the wild tsetse populations, the viral infections are primarily asymptomatic, whereas in a laboratory setting, at least for G. pallidipes colonies, the response to GpSGHV can be highly symptomatic. This suggests that in the wild tsetse populations, GpSGHV is mainly in covert (latent or persistent) infection state, and the virus is only reactivated to symptomatic state under certain specific ecological and/or environmental conditions. It is not clear which viral and/or host proteins may be expressed during viral latency. However, Lee et al. reported that latency can be caused by the infection of Spodoptera frugiperda (Sf) cells with Autographa californica multiple nucleopolyhedrovirus (AcMNPV) that lack the anti-apoptosis gene p35 [143]. Notably, even during the latency, infectious AcMNPV particles were continuously produced. Under the conditions used by these investigators, it appears that there is a balance between protein expression and the apoptotic pathway. Whereas GpSGHV does not have any homologue of any known anti-apoptotic gene, MdSGHV contains a homologue of Melanoplus sanguinipes entomopoxvirus anti-apoptotic gene, iap [120]. It should be noted that one of the major differences between GpSGHV and MdSGHV is that, MdSGHV does not have asymptomatic infection: instead, the virus induces symptomatic infection within 48 h post oral infection [144]. In addition, unlike GpSGHV, there is no evidence of vertical MdSGHV transmission. Since the issue of latency in hytrosaviruses is currently being debated among the Hytrosavirus Study Group of the ICTV, we briefly address it in Section 5.4 below.
5.4. Latency of Hytrosaviruses
Latency of insect virus infections is probably widespread. Latency can be defined as a viral infection that does not produce visible disease symptoms (e.g., SGH in the case of GpSGHV), but the virus may be transmitted, either vertically or horizontally [145]. This phenomenon raises a number of questions, particularly in the case of hytrosaviruses, which is the subject of this review.
The first question about latency is: is latency advantageous to the virus? Possibly, latency is an evolutionary viral strategy to utilize cues from the host and/or environment as opportunities for dispersal and transmission. In 1992, Fuxa et al. argued that in the adults of the armyworm S frugiperda (Lepidoptera: Noctuidae) latency may provide dispersal and reproductive advantages to S frugiperda nucleopolyhedrovirus (SfNPV) [145]. As the insect host metamorphoses to adulthood, vertical transmission becomes epizootiologically more important than horizontal transmission. The virus, therefore, takes advantage of the changes in the infected cells’ quality to produce nonlethal, vertically transferable virions. In 1993, Hughes et al. described a latent infection in a laboratory colony of the cabbage moth, Mamestra brassicae (Lepidoptera: Noctuidae) that was vertically transferred from one generation to another without disease symptoms [146]. Latency may also be influenced by virus-microbiome interplay as has been recently suggested [93]. Latency may also be stress-induced: for instance by crowding, food shortage, and superinfection in some baculoviruses [147,148]. Latency may also be connected to the antiviral response mechanisms of the hosts. Recently, it has been found that invertebrate DNA viruses, such as baculoviruses, iridoviruses and nudiviruses, provoke a specific RNA interference (RNAi) or microRNA response, which may prevent premature death of the host by preventing viral over-replication [149,150,151]. In case of the tsetse fly, there may be a further interaction between GpSGHV, the microbiome and the trypanosome parasite, and this tripartite interaction may be tailored to prevent trypanosome transmission [152].
The second question on viral latency is: how is viral latency established? One strategy in which viruses are vertically transmitted is by integrating into the host genome. For instance, the dsDNA polydnaviruses (PDVs) are vertically transmitted as proviruses stably integrated into the genomes of the parasitic wasps, ichneumonid (Hymenoptera; Ichneumonoidae) and braconid (Hymenoptera; Braconidae) [153]. Another strategy of vertical viral transmission is as infectious virions, such as the gypsy elements in D. melanogaster, which are transmitted as infectious particles from mother to progeny via oocytes [154]. Other insect viruses are maternally transmitted to the progeny flies, but also rely on horizontal transmission to be sustained in the host populations. An example of a virus utilizing this strategy is the DNA Leptopilina boulardi filamentous virus (LbFV) that infects the solitary parasitic wasp L. boulardi (Hymenoptera: Figitidae). When both an LbFV-infected and a non-infected female wasp lay eggs in Drosophila larvae during oviposition, LbFV is horizontally transmitted to the offspring of the non-infected wasp parent [155]. Consequently, the daughters of the non-infected females acquire the super-parasitizing phenotype (conferred by LbFV-infection) on emergence, with a 55% probability [156]. Other insect-infecting viruses, such as dengue virus (DENV) and GpSGHV, are primarily horizontally transmitted, but are also maternally transmitted [111,157]. Whether or not hytrosaviruses do integrate into the host genome is an issue currently under investigation.
5.5. Replication, Morphology and Morphogenesis of Hytrosaviruses
The GpSGHV replicates in the nucleus of host cells, where it induces formation of virogenic stroma (chromatin-like network of electron-dense filaments) [89]. After nuclear assembly the GpSGHV progeny nucleocapsids translocate to the cytoplasm where the envelopment is orchestrated, possibly via the ER-Golgi system, based on residual ER-Golgi proteins in the virion proteome. The possibility of the ER-Golgi assembly of the tsetse virus was proposed in the 1970s [59]. Based on the GpSGHV pathobiological data obtained to date (see summary in Table 1), cytoplasmic assembly of the virus particles induces cellular damage that possibly culminates into disintegration of the cell plasma membrane as the mature virions egress from the infected cell [89]. The virions are continuously shed via the saliva into the blood meals during membrane feeding in G. pallidipes colonies, and are infectious per os to healthy flies [52,142]. A remarkable difference between GpSGHV and MdSGHV is that the MdSGHV virions migrate to, and bud out of the plasma membrane of infected cells [133,134]. Unlike GpSGHV, MdSGHV does not have surface projections. Based on the available data, an infection model for GpSGHV can be hypothesized (Figure 4).
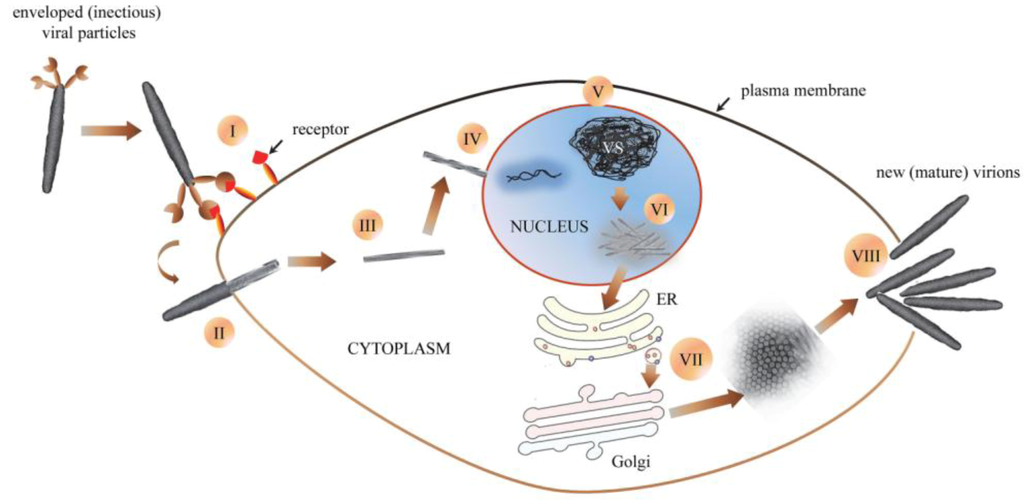
Figure 4.
Schematic (hypothetical) representation of GpSGHV morphogenesis: (I) an enveloped (infectious) viral particle binds to receptors on susceptible host cell. (II) Once bound, the virus is uncoated as it enters the host cell (III). Cytoplasmic trafficking of the viral nucleocapsid to the nucleus ensues (IV), followed by disassembly of viral nucleocapsids by partial degradation of capsid and tegument proteins and release of viral DNA into the host cell nucleus. (V) Once in the nucleus, the virus induces formation of virogenic stroma (VS), where viral nuclear replication occurs. (VI) After packaging of nascent viral DNA into capsids, nucleocapsids are assembled, after which they egress into the cell cytoplasm. (VII) The entire envelopment of nascent nucleocapsids is orchestrated in the cytoplasm, possibly via the ER-Golgi system. (VIII) Egress of the new mature virions from the infected cell possibly occurs via rupture or disintegration of the plasma membranes.
Figure 4.
Schematic (hypothetical) representation of GpSGHV morphogenesis: (I) an enveloped (infectious) viral particle binds to receptors on susceptible host cell. (II) Once bound, the virus is uncoated as it enters the host cell (III). Cytoplasmic trafficking of the viral nucleocapsid to the nucleus ensues (IV), followed by disassembly of viral nucleocapsids by partial degradation of capsid and tegument proteins and release of viral DNA into the host cell nucleus. (V) Once in the nucleus, the virus induces formation of virogenic stroma (VS), where viral nuclear replication occurs. (VI) After packaging of nascent viral DNA into capsids, nucleocapsids are assembled, after which they egress into the cell cytoplasm. (VII) The entire envelopment of nascent nucleocapsids is orchestrated in the cytoplasm, possibly via the ER-Golgi system. (VIII) Egress of the new mature virions from the infected cell possibly occurs via rupture or disintegration of the plasma membranes.
6. Epidemiology of Hytrosaviruses
6.1. Prevalence and Ecogeography of Hytrosaviruses
Early epidemiological surveys established that the prevalence of SGH symptoms in wild tsetse populations depends on the geographical location, seasonality, the distribution, age and species tsetse flies [65,68,72,73], and that the prevalence was generally low (0.9–15% in Kenya [65,68], 0.4–2% in Zimbabwe [72], 0.3–4.5% in Burkina Faso [73]). However, these surveys were based on the occurrence of SGH symptoms by fly dissections, which did not take into account that the majority of GpSGHV infections are asymptomatic [110]. Recently, PCR-based surveys were performed on G. pallidipes samples that were randomly collected from eleven geographical locations in six countries of eastern and central Africa [54]. The PCR-based surveys showed that an average of 34% of G. pallidipes samples were GpSGHV-infected: viral prevalence ranged from 2% to 100% [54]. Moreover, GpSGHV diversity was noted to be low. Twenty-three different viral haplotypes occurred in the same geographical locations. The GpSGHV haplotypes distribution patterns were somewhat confused: some of the viral haplotypes occurred only in certain geographical locations compared to others.
Unlike tsetse flies, house flies are among the most widely distributed insects, found in all inhabited areas of the world [158]. The global distribution of house flies allowed isolation of MdSGHV was isolated from the fly samples collected from various geographical locations around the globe i.e., from North America, Europe, Asia, the Caribbean, and the south-western Pacific [91,159]. The prevalence of MdSGHV infections in the field house fly populations can peak at ~30% at certain geographical sites, with a typical prevalence range of 0.5–10% at any given sampling period [104]. Further, the frequencies of MdSGHV infections positively correlate with the house fly population densities [104]. To date, it is unknown what MdSGHV haplotypes circulate among house fly populations. However, ecogeographic and behavioral attributes of the tsetse and house flies may influence selection pressure of GpSGHV and MdSGHV. Unlike in the case of GpSGHV, the opportunity to expose MdSGHV to the external environmental conditions such as UV radiation has probably led to the higher genetic variability of MdSGHV compared to the GpSGHV [54,91].
6.2. Transmission Dynamics of GpSGHV in the Laboratory Fly Colonies
Studies have revealed that GpSGHV-infected flies typically exhibit two infection phenotypes: a chronic non-debilitating asymptomatic state and an acute symptomatic state that causes reproductive dysfunction and colony collapse [93]. Various crosses were made between “healthy” (asymptomatic; PCR-negative) and symptomatic flies [52], whose outcomes are summarized in an infection model shown in Figure 5 [53]. Three key features of GpSGHV infection dynamics in laboratory colonies of G. pallidipes should be noted. Firstly, all males with SGH symptoms are fully sterile. Secondly, females with SGH symptoms do produce progeny flies; most (if not all) of these progeny flies exhibit SGH symptoms. Thirdly, asymptomatic females produce a small proportion of SGH-positive progeny flies; if such progeny flies are female, the F2 generation is sterile. The large numbers of virus particles released via saliva into the blood meals by the GpSGHV-infected flies during the in vitro membrane feeding (Figure 5B) are a source of per os transmission of GpSGHV to healthy flies [52].
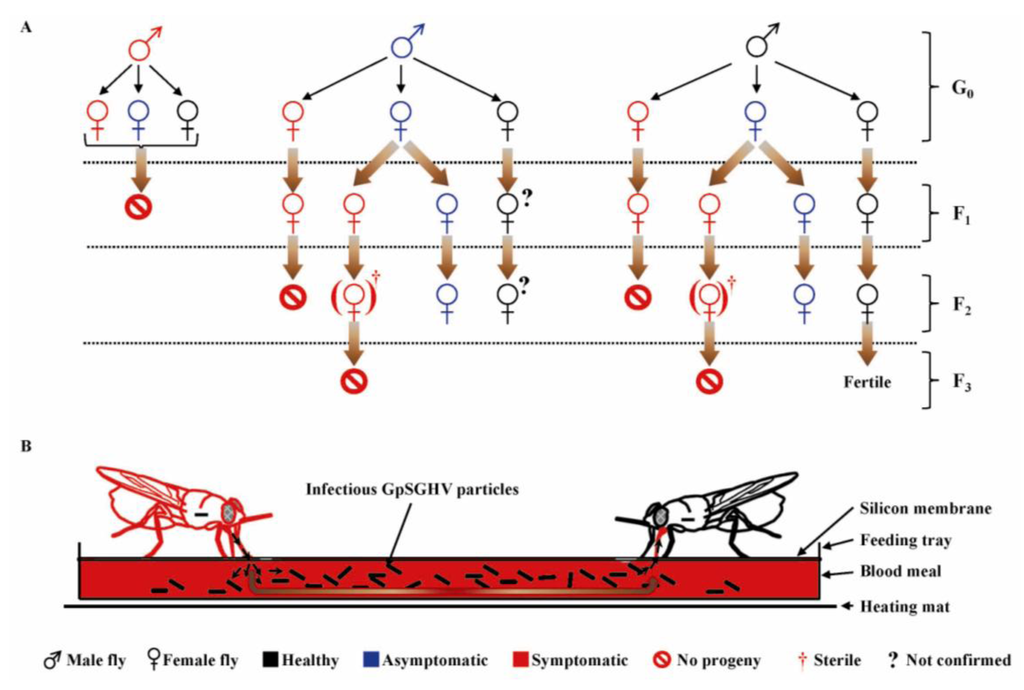
Figure 5.
A model of dynamics of vertical (A) and horizontal (B) GpSGHV transmission in laboratory colonies of G. pallidipes. The laboratory colony flies in the colony may either be “healthy” (PCR-negative; shown in black), asymptomatic—(in blue), or symptomatic—(in red). (A) Some symptomatic (SGH-positive) females do produce F1 progeny flies and never F2 (regardless of the status of the males that inseminate the females); (B) During membrane feeding, virus particles released by symptomatic flies via saliva into blood meals are infectious to other healthy flies. G0, F1 and F2 represent parental, 1st and 2nd fly generations, respectively. (?) Represent progeny flies with unknown infection status. (Figure modified from [53]; used with permission).
Figure 5.
A model of dynamics of vertical (A) and horizontal (B) GpSGHV transmission in laboratory colonies of G. pallidipes. The laboratory colony flies in the colony may either be “healthy” (PCR-negative; shown in black), asymptomatic—(in blue), or symptomatic—(in red). (A) Some symptomatic (SGH-positive) females do produce F1 progeny flies and never F2 (regardless of the status of the males that inseminate the females); (B) During membrane feeding, virus particles released by symptomatic flies via saliva into blood meals are infectious to other healthy flies. G0, F1 and F2 represent parental, 1st and 2nd fly generations, respectively. (?) Represent progeny flies with unknown infection status. (Figure modified from [53]; used with permission).
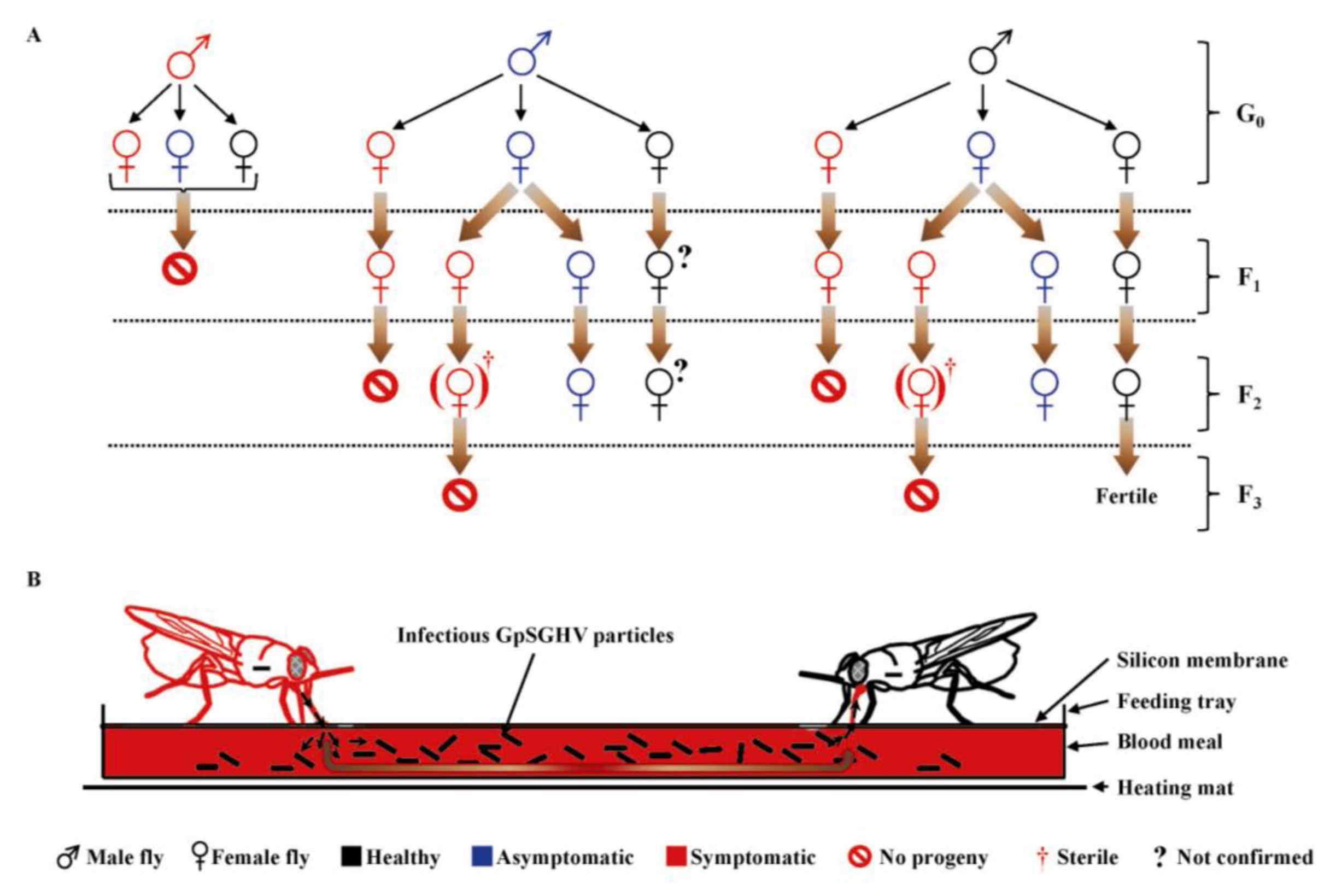
Taken together, the following implications are apparent. Firstly, as hypothesized by Jaenson in 1986 [71], survival of G. pallidipes colonies may depend on the infection status of the mothers: all F2 progeny flies produced by symptomatic mothers are sterile. Males with SGH symptoms are fully sterile and do release GpSGHV particles into blood via saliva during membrane feeding, and thus have a considerable impact on colony survival. Secondly, the observations that symptomatic flies die-off from the colony early in their life-span, and that asymptomatic flies have a relatively higher efficiency in salivary virus secretion compared to the symptomatic flies (Kariithi, unpublished) imply that asymptomatic flies may have more influence on the dynamics of GpSGHV transmission in the colonies compared to symptomatic flies [93]. The higher efficiency in viral transmission by asymptomatic compared to symptomatic flies results from the latter having impaired salivary secretions [89]. These studies revealed two noteworthy features. Firstly, when G. pallidipes flies were orally-infected with GpSGHV, the viral titers increased with the number of contaminated blood meals taken by the flies [52]. In marked contrast to tsetse flies, house flies are resistant to oral MdSGHV infections after 24 h post emergence (hpe) [91]. It can therefore be concluded that it is not the feeding history, but rather the age (hpe) of the tsetse fly when it takes the first (infective) meal that determines the fly’s susceptibility to viral infection [160]. Notably, the proportion of orally-infected G. pallidipes flies that secreted viral particles via saliva during the feeding events increased from 20% in the first blood meal to 100% in the seventh meal [52]. So, what hypotheses can explain why the efficiency of virus secretion increased with number of blood meals?
The peritrophic membrane (PM) of the intestinal tissue constitutes a physical barrier for viral diffusion into the salivary glands via the hemolymph. There is still conflicting information about the function of the PM in insects. Possible functions include protection of the stomach epithelium [161], ultrafiltration [162] and preventing the entry of microbes [163]. It has been suggested that the PM may limit the vectorial capacity of some hematophagous insects such as the sandflies [164]. In other insects such as lepidopterans, the PM is an age-dependent major barrier to baculovirus infection per os [165]. Further, Blackburn et al., [166] suggested that the PM is an impenetrable barrier for the diffusion of Leishmania or Trypanosoma parasites at certain stages after the blood meal. In mosquito-transmitted viruses, only 0.1% of Zika virus was shown to penetrate to the hemocoel of Aedes aegypti because of the barrier effect of the PM [167]. Subsequently, it has been suggested that the PM is a possible limiting factor for virus susceptibility in mosquitoes [168]. Likewise, the barrier function of the PM might have prevented GpSGHV from reaching the salivary glands of experimentally-infected G. pallidipes for at least 48 h-post-blood meal. Thereafter, the PM starts to decay and its disintegration may be completed a few days after, depending on availability of blood meal as has been suggested in sandflies [169]. It has been demonstrated that viruses enhance infection by disintegration of the PM [170]. There is strong evidence that GpSGHV virions contain chitinase [89,90], probably to facilitate the virus to penetrate the PM, which is essentially composed of chitin and proteins (mucins) [171]. This function has been demonstrated in the infection of mosquitoes by Plasmodium. The Plasmodium secretes chitinase to penetrate the PM in Ae. aegypti (inhibition of chitinase blocks transmission of Plasmodium) [172]. The important physiological functions of the PM suggest that it can be considered a significant structural target for the control of GpSGHV in the laboratory colonies of G. pallidipes. The potency of anti-chitinase antibodies to block transmission of pathogens has been demonstrated with the creation of Plasmodium-refractory mosquitoes [173]. A similar approach could be applied in GpSGHV management in G. pallidipes colonies.
Orally-infected G. pallidipes flies secrete infectious GpSGHV particles via saliva [52]. In contrast, when micro-injected with GpSGHV suspension, the flies did not secrete any detectable viral particles during the same duration of membrane feeding events [93]. Notably, expression of SGH symptoms in the F1 progeny produced by virus-injected mothers increased exponentially from 0–3% in the first larviposition cycle to 100% in the fourth cycle [93]. So, what hypotheses explain the apparent delayed expression of SGH symptoms in the F1 progeny produced in the early larviposition cycles? Possibly, if oocyte GpSGHV infection did not occur early in oogenesis, the developing egg membrane (chorion) might protect eggs from infection. Since the mothers ovipositioning later were blood-fed a few days post-infection, their ovaries probably became infected before egg maturation. It should be noted that in tsetse flies, larviposition is influenced by blood feeding, and is correlated to the digestion stages of the blood meal [174]. Unlike in the oral-infection, the virus injections were done on flies that had been fed (non-teneral). This implies that for the virus-injected flies, the egg chorion, possibly already formed at the time when the virus can reach the reproductive organs. This could be a further barrier for GpSGHV infection of eggs and thus of the F1 progeny.
Taken together, the invasion of G. pallidipes ovaries by GpSGHV could take place only during the later larviposition cycles. Since maturation of eggs begins shortly after blood meal ingestion [174], it is unlikely that GpSGHV in the blood meal would have sufficient time to infect the midgut, disseminate through the hemocoel to the ovaries and infect the developing oocytes immediately following the initial virus-contaminated blood meals. Therefore, trans-ovarial transmission of GpSGHV would not occur until the subsequent larviposition cycles after the infectious blood meal advance.
7. Potential of Hytrosaviruses as Bio-Pesticides
Many insect-pathogenic viruses such as baculoviruses are effective bio-control agents against insect pests [175,176]. The potential of GpSGHV as a male sterility factor in tsetse control was first proposed in 1988 [74]. After micro-injection of the virus into laboratory-bred G. pallidipes, infection was observed in 13.3% and 30.0% of treated male and female parent insects, respectively. The prevalence of SGH in the F1 progeny adults was much higher than in the parents (80% in males and 58.3% females). Whereas all infected females were fertile, all infected males had SGH syndrome and were sterile. Maternal larviposition, F1 pupae weight, and F1 pupae incubation periods were normal regardless of treatments. Two other studies reported that, although GpSGHV-infected males had reduced reproductive potential [86], such males did not lose their mating efficiency [77]. Although all the studies were performed under laboratory conditions, which probably differ from field conditions, it was hypothesized that Glossina hytrosavirus could be applied as a tsetse bio-control: the sterile male parents would compete with normal wild males in mating, and the fertile but infected females would transmit the virus trans-ovarially to subsequent generations, since such females produce only infected progeny [71], where males are sterile from eclosion. However, as discussed in previous sections, males with SGH are not sexually competitive. Further, SGH females produce sterile progeny flies, so the line quickly dies out.
Application of GpSGHV as a bio-pesticide for tsetse control however, is technically challenging for several reasons. Firstly, recent findings show that neither micro-injection nor per os infection of the virus in G. pallidipes result in SGH syndrome in the same (parental) generation, rather, the syndrome is only detectable in the third (~65%) and fourth (~100%) larviposition cycles of the F1 generation [93]. So, the effects of the viral infection are not immediately apparent. Secondly, high SGH prevalence in G. pallidipes colonies reduces the mating propensity and competitiveness of males thus affecting the stability and performance of tsetse colonies [133]. The colony instability would hinder production of large numbers of infected insects. Thirdly, in vitro mass production of GpSGHV for field applications is currently impossible due to limitations such as the absence of a cell culture system permissive to the virus. Currently, the only alternative method to multiply GpSGHV is by intra hemocoelic injection of the virus into colony flies, or by feeding the flies with virus-contaminated blood meals. Even if it were feasible to multiply GpSGHV by the injection method, it is a laborious process, and would require maintenance of huge number of flies. Fourthly, there is no available evidence for horizontal transmission of GpSGHV through contact between flies, mating, or fecal contamination, thus limiting the modes of how the virus would be dispersed in the field. Finally, GpSGHV does not produce occlusion bodies, as for instance baculoviruses do to achieve prolonged stability in the environment [177]. Besides, there is evidence that GpSGHV is highly unstable outside of the host [89], with more than 80% of purified virus suspension losing infectivity after 3 days at 4 °C [53], possibly due to the loss of the fragile viral envelope. Formulation of GpSGHV suspensions to allow retention of the viral infectivity under both laboratory and field conditions appears insurmountable at the moment. Therefore, the use of the GpSGHV as tsetse bio-control agent appears currently impractical.
8. Strategies to Control GpSGHV in Laboratory Colonies of G. pallidipes
8.1. Immune-Intervention Strategies
In view of the current understanding of GpSGHV epidemiology under laboratory conditions, two potential strategies to manage the viral infections in the colonies were considered. The first strategy was to reduce or inhibit horizontal GpSGHV transmission by either (a) modifying the feeding system currently used in the colony rearing [94], or (b) neutralizing the virus released via saliva into blood meals during membrane feeding by supplementing the blood meals with GpSGHV-specific antibodies, and/or (c) targeting virus ligands on host midgut cells with a phage display peptide library to block viral attachment to the midgut receptors [178,179]. The second strategy was to reduce or inhibit viral replication by: (d) oral administration of antiviral drugs to inhibit viral DNA polymerase [180], and (e) silencing essential GpSGHV genes by bacterially-expressed double-stranded RNA (dsRNA)-RNAi. Preliminary data so far obtained from the immune-intervention strategies, i.e., neutralizing antibodies, phage display technologies, and RNAi have shown potential to significantly reduce GpSGHV infections. Currently, these immune-intervention strategies are being optimized.
8.2. Modifications of in vitro Membrane Feeding Regime
The objective of various research reviewed in this paper was the development of a cost-effective strategy to manage the GpSGHV infections in G. pallidipes colonies. This was finally achieved mainly by modification of the in vitro membrane feeding regime routinely practiced in tsetse mass-rearing facilities. Since the membrane feeding favored horizontal viral transmission, it was conceptualized that modifications of the colony maintenance protocol(s) would significantly reduce GpSGHV transmission. The routine in vitro feeding regime involved feeding up to 10 sets of fly cages in succession [181], thus significantly augmenting horizontal GpSGHV transmission in the colonies. In the modified feeding regime, each fly-holding cage was provided with fresh blood at each meal to prevent flies from picking up any virus deposited via saliva into the during feeding of earlier fly cages [94]. Within 2 years of implementation of the modified feeding regime, GpSGHV loads in the fly colonies were significantly reduced and maintained at levels not detrimental to the survival and productivity of the colonies. More importantly, the SGH syndrome that causes colony collapse [28], was completely eliminated [94]. Additionally, the modified feeding regime is applicable in combination with other management strategies, for instance oral administration of the antiviral drugs in an integrated approach [180], or supplementing blood meals with GpSGHV-specific antibodies. The implications of the successful GpSGHV management in G. pallidipes colonies are discussed in sub-Section 8.3.
8.3. Implications of Successful Control of GpSGHV Infections in Colonized G. pallidipes
The membrane feeding system used in the tsetse mass-production facility located in Tanga, Tanzania greatly contributed to the production and release of ~8.5 million sterile males between 1994 to 1997, resulting in a crash of the G. austeni population in the Unguja Island [47]. However, attempts to mass-produce G. pallidipes for the SIT component of AW-IMP programs on mainland Africa were faced with difficulties because this tsetse species is susceptible to infections by GpSGHV. The virus has caused collapse of G. pallidipes colonies that were initiated in Ethiopia and Seibersdorf, Austria, and thus prevented full implementation of the release component. Now that strategies are available to successfully manage GpSGHV infections that can result in complete elimination of SGH from G. pallidipes colonies, production of sufficient numbers of sterile males is within reach. This success provides opportunity to revive the SIT component of the program to eradicate G. pallidipes from the fertile Rift Valley lands of Ethiopia [182]. This would translate into availability of more animals for plowing the fertile land, more milk, and manures to plant crops—in short, a permanent eradication of poverty and improvement of living standards. Similarly, other sub-Sahara African countries infested with trypanosome-transmitting tsetse species would benefit from this success. It should be noted that, although GpSGHV has not been shown to cause serious problems in other tsetse species, the virus infects G. fuscipes fuscipes, G. morsitans and G. swynnertoni, irrespective of their ages, sex, and season of the year [183]. Therefore, if GpSGHV (or a similar virus) will be problematic in the future, the research reviewed in this paper provides a solid basis to deal with the problem. The current knowledge and experiences in large-scale production of G. pallidipes can be used to make recommendations on standard operational procedures (SOPs) for the management of GpSGHV in large-scale tsetse fly production facilities. The proposed SOP is detailed in Supplementary File 1.
9. Concluding Remarks
The aim of this review was to highlight the intricacies associated with the occurrence of a newly described group of viruses, the hytrosaviruses, and the interaction with their dipteran hosts. The common denominators are the structure and genome content of these viruses and their potential to induce the SGH syndrome. Despite the knowledge of the genome and proteome, very little is known about the regulation of viral gene expression and the interaction of viral proteins with the dipteran host and its microbiome and parasites. Nevertheless, the wealth of information available in a relatively short period through the concerted efforts of many research groups [184] allowed the mitigation and control of SGH in tsetse fly colonies and set the stage for the development of more advanced strategies for tsetse fly control to eliminate trypanosomosis. A future challenge for virologists is to understand the natural role of hytrosaviruses in dipteran fly ecology and evolution.
Supplementary Files
Supplementary File 1Acknowledgments
Many of the studies reviewed in this paper were pioneered and fostered by the Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture, Seibersdorf, Austria. Most of the researches were done under the auspice of a Coordinated Research Project (No. D42012). The authors are indebted to The Netherlands Fellowship Program for a PhD Grant (No. CF7548/2011) awarded to Henry M. Kariithi for a Sandwich PhD program at the Laboratory of Virology, Wageningen University, The Netherlands.
Conflict of Interest
The authors declare no conflict of interest.
References and Notes
- Gooding, R.H.; Krafsur, E.S. Tsetse genetics: Contributions to biology, systematics, and control of tsetse flies. Annu. Rev. Entomol. 2005, 50, 101–123. [Google Scholar] [CrossRef]
- Mattioli, R.C.; Feldmann, U.; Hendrickx, G.; Wint, W.; Jannin, J.; Slingenbergh, J. Tsetse and trypanosomiasis intervention policies supporting sustainable animal-agricultural development. J. Food Agric. Environ. 2004, 2, 310–314. [Google Scholar]
- Mamoudou, A.; Zoli, A.; Hamadama, H.; Abah, S.; Geerts, S.; Clausen, P.H.; Zessin, K.H.; Kyule, M.; van den Bossche, P. Seasonal distribution and abundance of tsetse flies (Glossina spp.) in the Faro and Deo Division of the Adamaoua Plateau in Cameroon. Med. Vet. Entomol. 2008, 22, 32–36. [Google Scholar] [CrossRef]
- Smith, D.H.; Pepin, J.; Stich, A.H. Human African trypanosomiasis: An emerging public health crisis. Br. Med. Bull. 1998, 54, 341–355. [Google Scholar] [CrossRef]
- Cockerell, T.D.A. A fossil tsetse-fly in Colorado. Nature 1907, 76, 414. [Google Scholar] [CrossRef]
- Elsen, P.; Amoudi, M.A.; Leclercq, M. First record of Glossina fuscipes fuscipes Newstead, 1910 and Glossina morsitans submorsitans Newstead, 1910 in southwestern Saudi Arabia. Ann. Soc. Bel. Med. Trop. 1990, 70, 281–287. [Google Scholar]
- Hotez, P.J.; Kamath, A. Neglected tropical diseases in sub-saharan Africa: Review of their prevalence, distribution, and disease burden. PLoS Negl. Trop. Dis. 2009, 3, e412. [Google Scholar] [CrossRef]
- Hotez, P.J.; Molyneux, D.H.; Fenwick, A.; Kumaresan, J.; Sachs, S.E.; Sachs, J.D.; Savioli, L. Control of neglected tropical diseases. N. Engl. J. Med. 2007, 357, 1018–1027. [Google Scholar] [CrossRef]
- Cecchi, G.; Mattioli, R.C.; Slingenbergh, J.; de la Rocque, S. Land cover and tsetse fly distributions in sub-Saharan Africa. Med. Vet. Entomol. 2008, 22, 364–373. [Google Scholar] [CrossRef]
- Vreysen, M.J.B. Prospects for area-wide integrated control of tsetse flies (Diptera: Glossinidae) and trypanosomosis in sub-Saharan Africa. Rev. Soc. Entomol. Argent 2006, 65, 1–21. [Google Scholar]
- Otte, M.J.; Chilonda, P. Classification of cattle and small ruminant production systems in sub-Saharan Africa. Outlook Agr. 2003, 32, 183–190. [Google Scholar] [CrossRef]
- Budd, L.T. DFID-funded Tsetse and Trypanosomosis Research and Development Since 1980; Department of International Development: Aylesford, UK, 1999; Volume 2. [Google Scholar]
- Matovu, E.; Seebeck, T.; Enyaru, J.C.K.; Kaminsky, R. Drug resistance in Trypanosoma brucei spp., the causative agents of sleeping sickness in man and nagana in cattle. Microb. Infect. 2001, 3, 763–770. [Google Scholar] [CrossRef]
- Oberholzer, M.; Lopez, M.A.; McLelland, B.T.; Hill, K.L. Social motility in african trypanosomes. PLoS Pathog. 2010, 6, e1000739. [Google Scholar]
- Vickerman, K. Developmental cycles and biology of pathogenic trypanosomes. Br. Med. Bull. 1985, 41, 105–114. [Google Scholar]
- Brun, R.; Blum, J.; Chappuis, F.; Burri, C. Human African trypanosomiasis. Lancet 2010, 375, 148–159. [Google Scholar] [CrossRef]
- Chappuis, F.; Loutan, L.; Simarro, P.; Lejon, V.; Büscher, P. Options for field diagnosis of human african trypanosomiasis. Clin. Microbiol. Rev. 2005, 18, 133–146. [Google Scholar] [CrossRef]
- Friedheim, E.A.H. Mel B in the treatment of human trypanosomiasis. Am. J. Trop. Med. Hyg. 1949, 29, 173–180. [Google Scholar]
- Burri, C.; Nkunku, S.; Merolle, A.; Smith, T.; Blum, J.; Brun, R. Efficacy of new, concise schedule for melarsoprol in treatment of sleeping sickness caused by Trypanosoma brucei gambiense: A randomised trial. Lancet 2000, 355, 1419–1425. [Google Scholar] [CrossRef]
- Burri, C. Chemotherapy against human African trypanosomiasis: Is there a road to success? Parasitology 2010, 137, 1987–1994. [Google Scholar] [CrossRef]
- Barrett, M.P.; Vincent, I.M.; Burchmore, R.J.; Kazibwe, A.J.; Matovu, E. Drug resistance in human African trypanosomiasis. Future Microbiol. 2011, 6, 1037–1047. [Google Scholar] [CrossRef]
- Geerts, S.; Holmes, P.H. Drug Management and Parasite Resistance in Bovine Trypanosomiasis in Africa; FAO: Rome, Italy, 1998. [Google Scholar]
- Schofield, C.J.; Kabayo, J.P. Trypanosomiasis vector control in Africa and Latin America. Parasit Vectors 2008, 1, 24. [Google Scholar] [CrossRef]
- Vreysen, M.J.B.; Seck, M.T.; Sall, B.; Bouyer, J. Tsetse flies: Their biology and control using area-wide integrated pest management approaches. J. Invertebr. Pathol. 2013, 112, S15–S25. [Google Scholar] [CrossRef]
- Abd-Alla, A.; Cousserans, F.; Parker, A.; Parker, N.; Robinson, A.; Bergoin, M. Characterization of a Novel virus from the Mass-reared Tsetse Fly Glossina pallidipes. In Proceedings of 11th meeting of the Working Group Arthropod Mass Rearing and Quality Control, Montreal, Canada, 28 October–1 November 2007; pp. 1–4.
- Abd-Alla, A.M.M.; Cousserans, F.; Parker, A.G.; Jehle, J.A.; Parker, N.J.; Vlak, J.M.; Robinson, A.S.; Bergoin, M. Genome analysis of a Glossina pallidipes salivary gland hypertrophy virus reveals a novel large double-stranded circular DNA virus. J. Virol. 2008, 82, 4595–4611. [Google Scholar] [CrossRef]
- Abd-Alla, A.M.M.; Vlak, J.M.; Bergoin, M.; Maruniak, J.E.; Parker, A.G.; Burand, J.P.; Jehle, J.A.; Boucias, D.G. Hytrosavirus Study Group of the ICTV Hytrosaviridae: A proposal for classification and nomenclature of a new insect virus family. Arch. Virol. 2009, 154, 909–918. [Google Scholar] [CrossRef]
- Abd-Alla, A.; Bossin, H.; Cousserans, F.; Parker, A.; Bergoin, M.; Robinson, A. Development of a non-destructive PCR method for detection of the salivary gland hypertrophy virus (SGHV) in tsetse flies. J. Virol. Methods 2007, 139, 143–149. [Google Scholar] [CrossRef]
- Leak, S.G.A. Tsetse Biology and Ecology: Their Role in the Epidemiology and Control of Trypanosomosis; CABI Publishing: Wallingford, CT, USA, 1998. [Google Scholar]
- Matthews, E.G.; Clyne, D.; Kitching, R.L. Insect Ecology, 2nd ed.; University of Queensland Press: St. Lucia, Qld, Australia, 1984; Volume 6. [Google Scholar]
- Dujardin, J.-P.; Schofield, C.J. Triatominae: Systematics, morphology and population biology. In The Trypanosomiases; Maudlin, I., Holmes, P.H., Miles, M.A., Eds.; CABI Publishing: Wallingford, CT, USA, 2004; pp. 181–201. [Google Scholar]
- Allsopp, R. Options for vector control against trypanonomiasis in Africa. Trends Parasitol. 2001, 17, 15–19. [Google Scholar] [CrossRef]
- Brightwell, B.; Dransfield, R. Odour attractants for tsetse: Glossina austeni, G. brevipalpis and G. swynnertoni. Med. Vet. Entomol. 1997, 11, 297–299. [Google Scholar] [CrossRef]
- Thomson, J.W.; Wilson, A. The Control of Tsetse Flies and Trypanosomiasis by the Application of Deltamethrin to Cattle. In proceedings of the 20th Meeting of the International Scientific Coubcil for the Trypanosomiasis Research and Control (ISCTRC), Mombasa, Kenya, 10–14 April 1989; pp. 450–454.
- Brightwell, B.; Dransfield, R.D.; Stevenson, P.; Williams, B. Changes over twelve years in populations of Glossina pallidipes and Glossina longipennis (Diptera: Glossinidae) subject to varying trapping pressure at Nguruman, sourth-west Kenya. Bull. Entomol. Res. 1997, 87, 349–370. [Google Scholar] [CrossRef]
- Knipling, E.F. Possibilities of insect control or eradication through the use of sexually sterile males. J. Econ. Entomol. 1955, 48, 459–469. [Google Scholar]
- Knipling, E.F. Sterile-male method of population control. Science 1959, 130, 902–904. [Google Scholar]
- Robinson, A.S. Mutations and their use in insect control. Mutat. Res. 2002, 511, 113–132. [Google Scholar] [CrossRef]
- Robinson, A.S. Genetic Basis of the Sterile Insect Technique. In Sterile Insect Technique. Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 95–114. [Google Scholar]
- Abila, P.P.; Kiendrebeogo, M.; Mutika, G.N.; Parker, A.G.; Robinson, A.S. The effect of age on the mating competitiveness of male Glossina fuscipes fuscipes and G. palpalis palpalis. J. Insect Sci. 2003, 3, 13. [Google Scholar]
- Enkerlin, W.R. Impact of Fruit Fly Control Programmes Using the Sterile Insect Technique. In Sterile Insect Technique. Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 651–676. [Google Scholar]
- Franz, G. Genetic Sexing Strains in Mediterranean Fruit Fly, An Example for Other Species Amenable to Large-scale Rearing as Required for the Sterile Insect Technique. In Sterile Insect Technique. Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 427–451. [Google Scholar]
- Dyck, V.A.; Graham, S.H.; Bloem, K.A. Implementation of the Sterile Insect Release Programme to Eradicate the Codling Moth, Cydia pomonella (L.) (Lepidoptera: Olethreutidae), in British Columbia, Canada. In Proceedings of Management of Insect Pests: Nuclear and Related Molecular and Genetic Techniques, Vienna, Austria, 19–23 October 1992; pp. 285–297.
- Carpenter, J.; Bloem, S.; Hofmeyr, H. Area-wide Control Tactics for the False Codling Moth Thaumatotibia leucotreta in South Africa: A Potential Invasive Species. In Area-wide Control of Insect Pests: From Research to Field Implementation, 1st ed.; Vreysen, M.J.B., Robinson, A.S., Hendrichs, J., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 351–359. [Google Scholar]
- Daly, J.M.; Barrington, A.M.; Hackett, J.K.; Suckling, D.M. Sterilisation of painted apple moth Teia anartoides (Lepidoptera: Lymantridae) by irradiation. N. Z. Plant Prot. 2002, 55, 7–11. [Google Scholar]
- Koyama, J.; Kakinohana, H.; Miyatake, T. Eradication of the melon fly, Bactrocera cucurbitae, in Japan: Importance of behaviour, ecology, genetics and evolution. Ann. Rev. Entomol. 2004, 49, 331–349. [Google Scholar] [CrossRef]
- Vreysen, M.J.; Saleh, K.M.; Ali, M.Y.; Abdulla, A.M.; Zhu, Z.R.; Juma, K.G.; Dyck, V.A.; Msangi, A.R.; Mkonyi, P.A.; Feldmann, H.U. Glossina austeni (Diptera: Glossinidae) eradicated on the island of Unguja, Zanzibar, using the sterile insect technique. J. Econ. Entomol. 2000, 93, 123–135. [Google Scholar] [CrossRef]
- Vreysen, M.J.; Saleh, K.M.; Lancelot, R.; Bouyer, J. Factory tsetse flies must behave like wild flies: A prerequisite for the sterile insect technique. PLoS Negl. Trop. Dis. 2011, 5, e907. [Google Scholar] [CrossRef]
- Politzar, H.; Merot, P.; Brandl, F.E. Experimental aerial release of sterile males of Glossina palpalis gambiensis and Glossina tachinoides in a biological control operation. Rev. Élev. Méd. Vét. Pays Trop. 1984, 37, 198–202. [Google Scholar]
- Olandunmade, M.A.; Feldmann, U.; Takken, W.; Tanabe, S.O.; Hamann, H.J.; Onah, J.; Dengwat, L.; Vloedt, A.M.V.V.D.; Gingrich, R.E. Eradication of Glossina palpalis palpalis (Robineau-Desvoidy) (Diptera: Glossinidae) from agropastoral land in central Nigeria by means of the sterile insect technique. In Proceedings of the final research co-ordination meeting, Vom, Plateau State, Nigeria, 6–10 June 1988; pp. 5–23.
- Alemu, T.; Kapitano, B.; Mekonnen, S.; Aboset, G.; Kiflom, M.; Bancha, B.; Woldeyes, G.; Bekele, K.; Feldmann, U. Area-wide Control of Tsetse and Trypanosomosis: Ethiopian Experience in the Southern Rift Valley. In Area-Wide Control of Insect Pests: From Research to Field Implementation; Vreysen, M.J.B., Robinson, A.S., Hendrichs, J., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 325–335. [Google Scholar]
- Abd-Alla, A.M.M.; Kariithi, H.M.; Parker, A.G.; Robinson, A.S.; Kiflom, M.; Bergoin, M.; Vreysen, M.J.B. Dynamics of the salivary gland hypertrophy virus in laboratory colonies of Glossina pallidipes (Diptera: Glossinidae). Virus Res. 2010, 150, 103–110. [Google Scholar] [CrossRef]
- Abd-Alla, A.M.M.; Parker, A.G.; Vreysen, M.J.B.; Bergoin, M. Tsetse salivary gland hypertrophy virus: Hope or hindrance for tsetse control? PLoS Negl. Trop. Dis. 2011, 5, e1220. [Google Scholar] [CrossRef]
- Kariithi, H.M.; Ahmadi, M.; Parker, A.G.; Franz, G.; Ros, V.I.D.; Haq, I.; Elashry, A.M.; Vlak, J.M.; Bergoin, M.; Vreysen, M.J.B.; et al. Prevalence and genetic variation of salivary gland hypertrophy virus in wild populations of the tsetse fly Glossina pallidipes from southern and eastern Africa. J. Invertebr. Pathol. 2013, 112, S123–S132. [Google Scholar] [CrossRef]
- Whitnall, A.B.M. The trypanosome infections of Glossina pallidipes in the Umfolosi Game Reserve, Zululand. Onderstepoort J. Vet. Sci. Anim. Ind. 1934, 2, 2–21. [Google Scholar]
- Whitnall, A.B.M. The trypanosome infections of Glossina pallidipes in the Umfolosi Game Reserve, Zululand. (Preliminary Report); In 18th Report of the Director of Veterinary Services and Animal Industry; Union of South Africa: Onderstepoort, South Africa, 1932; pp. 7–21. [Google Scholar]
- Burtt, E. Hypertrophied salivary glands in Glossina: Evidence that G. pallidipes with this abnormality is particularly suited to trypanosome infection. Ann. Trop. Med. Parasitol. 1945, 39, 11–13. [Google Scholar]
- Jenni, L. Virus like particles in a strain of G. morsitans centralis, Machado 1970. Trans. R. Soc. Trop. Med. Hyg. 1973, 67, 295. [Google Scholar] [CrossRef]
- Jenni, L.; Steiger, R.F. Virus like particles in the tsetse fly, Glossina morsitans sspp. Preliminary results. Rev. Suisse Zool. 1974, 81, 663–666. [Google Scholar]
- Jenni, L.; Steiger, R. Virus like particles of Glossina fuscipes fuscipes Newst. 1910. Acta Trop. 1974, 31, 177–180. [Google Scholar]
- Jenni, L.; Böhringer, S. Nuclear coat and virus like particles in the midgut epithelium of Glossina morsitans sspp. Acta Trop. 1976, 33, 380–389. [Google Scholar]
- Lyon, J.P. La mouche des Narcisses (Merodon equestris F., Diptere Syrphidae). I. Identification de l'insecte et de ses degats et biologie dans le sud-est de la France. Rev. Zool. Agr. Pathol. Veg. 1973, 72, 65–92. [Google Scholar]
- Jaenson, T.G.T. Virus-like rods associated with salivary gland hyperplasia in tsetse, Glossina pallidipes. Trans. R. Soc. Trop. Med. Hyg. 1978, 72, 234–238. [Google Scholar] [CrossRef]
- Amargier, A.; Lyon, J.P.; Vago, C.; Meynadier, G.; Veyrunes, J.C. Mise en evidence et purification d'un virus dans la proliferation monstrueuse glandulaire d'insectes. Etude sur Merodon equestris F. (Diptere: Syrphidae). C. R. Acad. Sci. D 1979, 289, 481–484. [Google Scholar]
- Otieno, L.H.; Kokwaro, E.D.; Chimtawi, M.; Onyango, P. Prevalence of enlarged salivary glands in wild populations of Glossina pallidipes in Kenya, with a note on the ultrastructure of the affected organ. J. Invertebr. Pathol. 1980, 36, 113–118. [Google Scholar] [CrossRef]
- Opiyo, E.A.; Okumu, I. The KETRI Glossina pallidipes Colony: Further Observations. In Diseases of the Tropics; Tukei, P.M., Njogu, A.R., Eds.; Africa Book Services: Nairobi, Kenya, 1983; pp. 52–54. [Google Scholar]
- Odindo, M.O.; Sabwa, D.M.; Amutalla, P.A.; Otieno, W.A. Preliminary tests on the transmission of virus-like particles to the tsetse Glossina pallidipes. Insect Sci. Appl. 1981, 2, 219–221. [Google Scholar]
- Odindo, M.O. Incidence of salivary gland hypertrophy in field populations of the tsetse Glossina pallidipes on the South Kenya coast. Insect Sci. Appl. 1982, 3, 59–64. [Google Scholar]
- Odindo, M.O.; Payne, C.C.; Crook, N.E.; Jarret, P. Properties of a novel DNA virus from the tsetse fly, Glossina pallidipes. J. Gen. Virol. 1986, 67, 527–536. [Google Scholar] [CrossRef]
- Odindo, M.O.; Amutalla, P.A. Distribution pattern of the virus of Glossina pallidipes Austen in a forest ecosystem. Insect Sci. Appl. 1986, 7, 79–84. [Google Scholar]
- Jaenson, T.G.T. Sex ratio distortion and reduced lifespan of Glossina pallidipes infected with the virus causing salivary gland hyperplasia. Entomol. Exp. Appl. 1986, 41, 256–271. [Google Scholar]
- Ellis, D.S.; Maudlin, I. Salivary gland hyperplasia in wild caught tsetse from Zimbabwe. Entomol. Exp. Appl. 1987, 45, 167–173. [Google Scholar] [CrossRef]
- Gouteux, J.P. Prevalence of enlarged salivary glands in Glossina palpalis, G. pallicera, and G. nigrofusca (Diptera: Glossinidae) from the Vavoua area, Ivory Coast. J. Med. Entomol. 1987, 24, 268. [Google Scholar]
- Odindo, M.O. Glossina pallidipes virus: Its potential for use in biological control of tsetse. Insect Sci. Appl. 1988, 9, 399–403. [Google Scholar]
- Jura, W.G.Z.O.; Odhiambo, T.R.; Otieno, L.H.; Tabu, N.O. Gonadal lesions in virus-infected male and female tsetse, Glossina pallidipes (Diptera: Glossinidae). J. Invertebr. Pathol. 1988, 52, 1–8. [Google Scholar] [CrossRef]
- Jura, W.G.Z.O.; Zdarek, J.; Otieno, L.H. A simple method for artificial infection of tsetse, Glossina morsitans morsitans larvae with the DNA virus of G. pallidipes. Int. J. Trop. Insect Sci. 1993, 14, 383–387. [Google Scholar] [CrossRef]
- Jura, W.G.Z.O.; Davies-Cole, J.O.A. Some aspects of mating behavior of Glossina morsitans morsitans males infected with a DNA virus. Biol. Control 1992, 2, 188–192. [Google Scholar] [CrossRef]
- Jura, W.; Otieno, L.; Chimtawi, M. Ultrastructural evidence for trans-ovum transmission of the DNA virus of tsetse, Glossina pallidipes (Diptera: Glossinidae). Curr. Microbiol. 1989, 18, 1–4. [Google Scholar] [CrossRef]
- Kokwaro, E.D.; Nyindo, M.; Chimtawi, M. Ultrastructural changes in salivary glands of tsetse, Glossina morsitans morsitans, infected with virus and rickettsia-like organisms. J. Invertebr. Pathol. 1990, 56, 337–346. [Google Scholar] [CrossRef]
- Kokwaro, E.D.; Otieno, L.H.; Chimtawi, M. Salivary glands of the tsetse Glossina pallidipes Austen infected with Trypanosoma brucei and virus particles: Ultrastructural study. Int. J. Trop. Insect Sci. 1991, 12, 661–669. [Google Scholar]
- Shaw, M.K.; Moloo, S.K. Virus-like particles in Rickettsia within the midgut epithelial cells of Glossina morsitans centralis and Glossina brevipalpis. J. Invertebr. Pathol. 1993, 61, 162–166. [Google Scholar] [CrossRef]
- Coler, R.R.; Boucias, D.G.; Frank, J.H.; Maruniak, J.E.; Garcia-Canedo, A.; Pendland, J.C. Characterization and description of a virus causing salivary gland hyperplasia in the housefly, Musca domestica. Med. Vet. Entomol. 1993, 7, 275–282. [Google Scholar] [CrossRef]
- Sang, R.C.; Jura, W.G.Z.O.; Otieno, L.H.; Ogaja, P. Ultrastructural changes in the milk gland of tsetse Glossina morsitans centralis (Diptera; Glissinidae) female infected by a DNA virus. J. Invertebr. Pathol. 1996, 68, 253–259. [Google Scholar] [CrossRef]
- Sang, R.C.; Jura, W.G.Z.O.; Otieno, L.H.; Tukei, P.M.; Mwangi, R.W. Effects of tsetse DNA virus infection on the survival of a host fly Glossina morsitans centralis (Diptera: Glossinidae). J. Invertebr. Pathol. 1997, 69, 253–260. [Google Scholar] [CrossRef]
- Sang, R.C.; Jura, W.G.Z.O.; Otieno, L.H.; Mwangi, R.W. The effects of a DNA virus infection on the reproductive potential of female tsetse flies, Glossina morsitans centralis and Glossina morsitans morsitans (Diptera: Glossinidae). Mem. Inst. Oswaldo Cruz. 1998, 93, 861–864. [Google Scholar] [CrossRef]
- Sang, R.C.; Jura, W.G.Z.O.; Otieno, L.H.; Mwangi, R.W.; Ogaja, P. The effects of a tsetse DNA virus infection on the functions of the male accessory reproductive gland in the host fly Glossina morsitans centralis (Diptera; Glossinidae). Curr. Microbiol. 1999, 38, 349–354. [Google Scholar]
- Kokwaro, E.D. Virus particles in male accessory reproductive glands of tsetse, Glossina morsitans morsitans (Diptera: Glossinidae) and associated tissue changes. Int. J. Trop. Insect Sci. 2006, 26, 266–272. [Google Scholar] [CrossRef]
- Salem, T.Z.; Garcia-Maruniak, A.; Lietze, V.U.; Maruniak, J.E.; Boucias, D.G. Analysis of transcripts from predicted open reading frames of Musca domestica salivary gland hypertrophy virus. J. Gen. Virol. 2009, 90, 1270–1280. [Google Scholar] [CrossRef]
- Kariithi, H.M.; van Lent, J.W.; Boeren, S.; Abd-Alla, A.M.; Ince, I.A.; van Oers, M.M.; Vlak, J.M. Correlation between structure, protein composition, morphogenesis and cytopathology of Glossina pallidipes salivary gland hypertrophy virus. J Gen. Virol. 2013, 94, 193–208. [Google Scholar] [CrossRef]
- Kariithi, H.M.; Ince, I.A.; Boeren, S.; Vervoort, J.; Bergoin, M.; van Oers, M.M.; Abd-Alla, A.M.M.; Vlak, J.M. Proteomic analysis of Glossina pallidipes salivary gland hypertrophy virus virions for immune intervention in tsetse fly colonies. J. Gen. Virol. 2010, 91, 3065–3074. [Google Scholar] [CrossRef]
- Prompiboon, P.; Lietze, V.-U.; Denton, J.S.S.; Geden, C.J.; Steenberg, T.; Boucias, D.G. Musca domestica salivary gland hypertrophy virus, a globally distributed insect virus that infects and sterilizes female houseflies. Appl. Environ. Microbiol. 2010, 76, 994–998. [Google Scholar] [CrossRef]
- Luo, L.; Zeng, L. A new rod-shaped virus from parasitic wasp Diachasmimorpha longicaudata (Hymenoptera: Braconidae). J. Invertebr. Pathol. 2010, 103, 165–169. [Google Scholar] [CrossRef]
- Boucias, D.G.; Kariithi, H.M.; Bourtzis, K.; Schneider, D.I.; Kelley, K.; Miller, W.J.; Parker, A.G.; Abd-Alla, A.M.M. Trans-generational transmission of the Glossina pallidipes hytrosavirus depends on the presence of a functional symbiome. PLoS One 2013, 8, e61150. [Google Scholar]
- Abd-Alla, A.M.M.; Kariithi, H.M.; Mohamed, A.H.; Lapiz, E.; Parker, A.G.; Vreysen, M.J.B. Managing hytrosavirus infections in Glossina pallidipes colonies: Feeding regime affects the prevalence of salivary gland hypertrophy syndrome. PLoS One 2013, 8, e61875. [Google Scholar]
- Howatson, A.; Whitmore, G. The development and structure of vesicular stomatitis virus. Virology 1962, 16, 466–478. [Google Scholar] [CrossRef]
- Mussgay, M.; Weibel, J. Electron microscopic studies on the development oof vesicular stomatis virus in KB cells. J. Cell Biol. 1963, 16, 119–129. [Google Scholar] [CrossRef]
- Chamberlain, R.W. Arboviruses, the arthropod-borne animal viruses. Curr. Top. Microbiol. Immunol. 1968, 42, 38–58. [Google Scholar] [CrossRef]
- Janzen, H.G.; Rhodes, A.J.; Doane, F.W. Chikungunya virus in salivary glands of Aedes aegypti (L.): An electron microscope study. Can. J. Microbiol. 1970, 16, 581–586. [Google Scholar] [CrossRef]
- Lamotte, L.C., Jr. Japanese B encephalitis virus in the organs of infected mosquitoes. Am. J. Epidemiol. 1960, 72, 73–87. [Google Scholar]
- Mims, C.A.; Day, M.F.; Marshall, I.D. Cytopathic effect of Semliki Forest virus in the mosquito, Aedes aegypti. Am. J. Trop. Med. Hyg. 1966, 15, 775–784. [Google Scholar]
- Longdon, B.; Wilfert, L.; Obbard, D.J.; Jiggins, F.M. Rhabdoviruses in two species of Drosophila: Vertical transmission and a recent sweep. Genetics 2011, 188, 141–150. [Google Scholar] [CrossRef]
- L'Heritier, P.H. The Hereditary virus of Drosophila. In Advances in Virus Research; Kenneth, M.S., Lauffer, M.A., Eds.; Academic Press Inc.: New York, NY, USA, 1958; Volume 5, pp. 195–245. [Google Scholar]
- Anderson, R.M.; May, R.M. Infectious diseases and population cycles of forest insects. Science 1980, 210, 658–661. [Google Scholar]
- Geden, C.J.; Lietze, V.U.; Boucias, D.G. Seasonal prevalence and transmission of salivary gland hypertrophy virus of house flies (Diptera: Muscidae). J. Med. Entomol. 2008, 45, 42–51. [Google Scholar] [CrossRef]
- Lietze, V.U.; Geden, C.J.; Blackburn, P.; Boucias, D.G. Effects of salivary gland hypertrophy virus on the reproductive behavior of the housefly, Musca domestica. Appl. Environ. Microbiol. 2007, 73, 6811–6818. [Google Scholar] [CrossRef]
- Geden, C.; Garcia-Maruniak, A.; Lietze, V.U.; Maruniak, J.; Boucias, D.G. Impact of house fly salivary gland hypertrophy virus (MdSGHV) on a heterologous host, Stomoxys calcitrans. J. Med. Entomol. 2011, 48, 1128–1135. [Google Scholar] [CrossRef]
- Chou, M.-Y.; Haymer, D.S.; Feng, H.-Y.; Mau, R.F.L.; Hsu, J.-C. Potential for insecticide resistance in populations of Bactrocera dorsalis in Hawaii: Spinosad susceptibility and molecular characterization of a gene associated with organophosphate resistance. Entomol. Exp. Appl. 2010, 134, 296–303. [Google Scholar]
- Lawrence, P.O.; Akin, D. Virus-like particles from the poison glands of the parasitic wasp Biosteres longicaudatus (Hymenoptera: Braconidae). Can. J. Zool. 1990, 68, 539–546. [Google Scholar] [CrossRef]
- Lawrence, P.O. Morphogenesis and cytopathic effects of the Diachasmimorpha longicaudata entomopoxvirus in host haemocytes. J. Insect Physiol. 2005, 51, 221–233. [Google Scholar] [CrossRef]
- Abd-Alla, A.; Cousserans, F.; Parker, A.; Bergoin, M.; Chiraz, J.; Robinson, A. Quantitative PCR analysis of the salivary gland hypertrophy virus (GpSGHV) in a laboratory colony of Glossina pallidipes. Virus Res. 2009, 139, 48–53. [Google Scholar] [CrossRef]
- Lietze, V.-U.; Abd-Alla, A.; Vreysen, M.; Geden, C.C.; Boucias, D.G. Salivary gland hypertrophy viruses: A novel group of insect pathogenic viruses. Annu. Rev. Entomol. 2011, 56, 63–80. [Google Scholar] [CrossRef]
- Duron, O.; Bouchon, D.; Boutin, S.; Bellamy, L.; Zhou, L.; Engelstadter, J.; Hurst, G.D. The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol. 2008, 6, 27. [Google Scholar] [CrossRef]
- Engelstadter, J.; Hurst, G.D.D. The ecology and evolution of microbes that manipulate host reproduction. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 127–149. [Google Scholar] [CrossRef]
- Hilgenboecker, K.; Hammerstein, P.; Schlattmann, P.; Telschow, A.; Werren, J.H. How many species are infected with Wolbachia?—A statistical analysis of current data. FEMS Microbiol. Lett. 2008, 281, 215–220. [Google Scholar] [CrossRef]
- Doudoumis, V.; Tsiamis, G.; Wamwiri, F.; Brelsfoard, C.; Alam, U.; Aksoy, E.; Dalaperas, S.; bd-Alla, A.; Ouma, J.; Takac, P.; et al. Detection and characterization of Wolbachia infections in laboratory and natural populations of different species of tsetse flies (genus Glossina). BMC Microbiol. 2012, 12, S3. [Google Scholar] [CrossRef]
- Alam, U.; Hyseni, C.; Symula, R.E.; Brelsfoard, C.; Wu, Y.; Kruglov, O.; Wang, J.; Echodu, R.; Alioni, V.; Okedi, L.M.; et al. Implications of microfauna-host interactions for trypanosome transmission dynamics in Glossina fuscipes fuscipes in Uganda. Appl. Environ. Microbiol. 2012, 78, 4627–4637. [Google Scholar] [CrossRef]
- Hughes, G.L.; Ren, X.; Ramirez, J.L.; Sakamoto, J.M.; Bailey, J.A.; Jedlicka, A.E.; Rasgon, J.L. Wolbachia infections in Anopheles gambiae cells: Transcriptomic characterization of a novel host-symbiont interaction. PLoS Pathog. 2011, 7, e1001296. [Google Scholar] [CrossRef]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A Wolbachia symbiont in Aedes aegypti limits infection with Dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef]
- Song, J.; Wang, R.; Deng, F.; Wang, H.; Hu, Z. Functional studies of per os infectivity factors of Helicoverpa armigera single nucleocapsid nucleopolyhedrovirus. J. Gen. Virol. 2008, 89, 2331–2338. [Google Scholar] [CrossRef]
- Garcia-Maruniak, A.; Maruniak, J.E.; Farmerie, W.; Boucias, D.G. Sequence analysis of a non-classified, non-occluded DNA virus that causes salivary gland hypertrophy of Musca domestica, MdSGHV. Virology 2008, 377, 184–196. [Google Scholar] [CrossRef]
- Cheng, C.H.; Liu, S.M.; Chow, T.Y.; Hsiao, Y.Y.; Wang, D.P.; Huang, J.J.; Chen, H.H. Analysis of the complete genome sequence of the Hz-1 virus suggests that it is related to members of the Baculoviridae. J. Virol. 2002, 76, 9024–9034. [Google Scholar] [CrossRef]
- Wang, Y.; Kleespies, R.G.; Huger, A.M.; Jehle, J.A. The genome of Gryllus bimaculatus nudivirus indicates an ancient diversification of baculovirus-related nonoccluded nudiviruses of insects. J. Virol. 2007, 81, 5395–5406. [Google Scholar] [CrossRef]
- Wang, Y.; van Oers, M.M.; Crawford, A.M.; Vlak, J.M.; Jehle, J.A. Genomic analysis of Oryctes rhinoceros virus reveals genetic relatedness to Heliothis zea virus 1. Arch. Virol. 2006, 152, 519–531. [Google Scholar]
- Wang, Y.; Bininda-Emonds, O.; van Oers, M.; Vlak, J.; Jehle, J. The genome of Oryctes rhinoceros nudivirus provides novel insight into the evolution of nuclear arthropod-specific large circular double-stranded DNA viruses. Virus Genes 2011, 42, 444–456. [Google Scholar] [CrossRef]
- Ohkawa, T.; Washburn, J.O.; Sitapara, R.; Sid, E.; Volkman, L.E. Specific binding of Autographa californica M nucleopolyhedrovirus occlusion-derived virus to midgut cells of Heliothis virescens larvae is mediated by products of pif genes Ac119 and Ac022 but not by Ac115. J. Virol. 2005, 79, 15258–15264. [Google Scholar] [CrossRef]
- Blissard, G.W.; Rohrmann, G.F. Baculovirus diversity and molecular biology. Ann. Rev. Entomol. 1990, 35, 127–155. [Google Scholar] [CrossRef]
- Tidona, C.A.; Darai, G. Iridovirus homologues of cellular genes—Implications for the molecular evolution of large DNA viruses. Virus Genes 2000, 21, 77–81. [Google Scholar] [CrossRef]
- Invertebrate Virus Subcommittee, Hytrosavirus Study Group. Available online: http://www.ictvonline.org/taxonomyHistory.asp?taxnode_id=20125369&taxa_name=Glossinavirus/ (accessed on 19 June 2013).
- Garcia-Maruniak, A.; Abd-Alla, A.M.M.; Salem, T.Z.; Parker, A.G.; van Oers, M.M.; Maruniak, J.E.; Kim, W.; Burand, J.P.; Cousserans, F.; Robinson, A.S.; et al. Two viruses that cause salivary gland hypertrophy in Glossina pallidipes and Musca domestica are related and form a distinct phylogenetic clade. J. Gen. Virol. 2009, 90, 334–346. [Google Scholar] [CrossRef]
- Jaenson, T.G.T. Reproductive Biology of the Tsetse Glossina pallidipes Austen (Diptera, Glossinidae) with Special Reference to Mating Behaviour; Department of entomology, University of Uppsala: Uppsala, Sweden, 1978; p. 39. [Google Scholar]
- Lietze, V.U.; Sims, K.R.; Salem, T.Z.; Geden, C.J.; Boucias, D.G. Transmission of MdSGHV among adult house flies, Musca domestica (Diptera: Muscidae), occurs via oral secretions and excreta. J. Invertebr. Pathol. 2009, 101, 49–55. [Google Scholar] [CrossRef]
- Mutika, G.N.; Marin, C.; Parker, A.G.; Boucias, D.G.; Vreysen, M.J.; Abd-Alla, A.M. Impact of salivary gland hypertrophy virus infection on the mating success of male Glossina pallidipes: Consequences for the sterile insect technique. PLoS One 2012, 7, e42188. [Google Scholar]
- Boucias, D.G.; Deng, F.; Hu, Z.; Garcia-Maruniak, A.; Lietze, V.U. Analysis of the structural proteins from the Musca domestica hytrosavirus with an emphasis on the major envelope protein. J. Invertebr. Pathol. 2013, 112, S44–S52. [Google Scholar] [CrossRef]
- Lietze, V.U.; Abd-Alla, A.M.M.; Boucias, D.G. Two hytrosaviruses, MdSGHV and GpSGHV, induce distinct cytopathologies in their respective host insects. J. Invertebr. Pathol. 2011, 107, 161–163. [Google Scholar] [CrossRef]
- Gao, Y.; Luo, L. Genome-based phylogeny of dsDNA viruses by a novel alignment-free method. Gene 2012, 492, 309–314. [Google Scholar] [CrossRef]
- Wu, G.A.; Jun, S.R.; Sims, G.E.; Kim, S.H. Whole-proteome phylogeny of large dsDNA virus families by an alignment-free method. Proc. Natl. Acad. Sci. USA 2009, 106, 12826–12831. [Google Scholar] [CrossRef]
- Yu, Z.G.; Chu, K.H.; Li, C.P.; Anh, V.; Zhou, L.Q.; Wang, R.W. Whole-proteome phylogeny of large dsDNA viruses and parvoviruses through a composition vector method related to dynamical language model. BMC Evol. Biol. 2010, 10, 192. [Google Scholar]
- Wang, Y.; Jehle, J.A. Nudiviruses and other large, double-stranded circular DNA viruses of invertebrates: New insights on an old topic. J. Invertebr. Pathol. 2009, 101, 187–193. [Google Scholar] [CrossRef]
- Jehle, J.A.; Abd-Alla, A.M.; Wang, Y. Phylogeny and evolution of Hytrosaviridae. J. Invertebr. Pathol. 2013, 112, S62–S67. [Google Scholar] [CrossRef]
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Mutations: Types and Causes. In Molecular Cell Biology, 4th ed.; W. H. Freeman and Company: New York, NY, USA, 2000. [Google Scholar]
- Joyce, A.R.; Palsson, B.O. The model organism as a system: Integrating “omics” data sets. Nat. Rev. Mol. Cell Biol. 2006, 7, 198–210. [Google Scholar] [CrossRef]
- Kariithi, H.M.; Ince, I.A.; Boeren, S.; Abd-Alla, A.M.M.; Parker, A.G.; Aksoy, S.; Vlak, J.M.; van Oers, M.M. The salivary secretome of the tsetse fly, Glossina pallidipes (Diptera: Glossinidae) infected by salivary gland hypertrophy virus. PLoS Negl. Trop. Dis. 2011, 5, e1371. [Google Scholar] [CrossRef]
- Lee, J.C.; Chen, H.H.; Chao, Y.C. Persistent baculovirus infection results from deletion of the apoptotic suppressor gene p35. J. Virol. 1998, 72, 9157–9165. [Google Scholar]
- Geden, C.J.; Steenberg, T.; Lietze, V.U.; Boucias, D.G. Salivary gland hypertrophy virus of house flies in Denmark: Prevalence, host range, and comparison with a Florida isolate. J. Vector. Ecol. 2011, 36, 231–238. [Google Scholar] [CrossRef]
- Fuxa, J.R.; Weidner, E.H.; Richter, A.R. Polyhedra without virions in a vertically transmitted nuclear polyhedrosis virus. J. Invertebr. Pathol. 1992, 60, 53–58. [Google Scholar]
- Hughes, D.S.; Possee, R.D.; King, L.A. Activation and detection of a latent baculovirus resembling Mamestra brassicae nuclear polyhedrosis virus in M. brassicae insects. Virology 1993, 194, 608–615. [Google Scholar] [CrossRef]
- Hughes, D.S.; Possee, R.D.; King, L.A. Evidence for the presence of a low-level, persistent baculovirus infection of Mamestra brassicae insects. J. Gen. Virol. 1997, 78, 1801–1805. [Google Scholar]
- Murillo, R.; Hussey, M.S.; Possee, R.D. Evidence for covert baculovirus infections in a Spodoptera exigua laboratory culture. J. Gen. Virol. 2011, 92, 1061–1070. [Google Scholar] [CrossRef]
- Bronkhorst, A.W.; van Cleef, K.W.; Vodovar, N.; Ince, I.A.; Blanc, H.; Vlak, J.M.; Saleh, M.C.; van Rij, R.P. The DNA virus invertebrate iridescent virus 6 is a target of the Drosophila RNAi machinery. Proc. Natl. Acad. Sci. USA. 2012, 109, E3604–E3613. [Google Scholar]
- Jayachandran, B.; Hussain, M.; Asgari, S. RNA interference as a cellular defense mechanism against the DNA virus baculovirus. J. Virol. 2012, 86, 13729–13734. [Google Scholar] [CrossRef]
- Wu, Y.L.; Wu, C.P.; Liu, C.Y.; Hsu, P.W.; Wu, E.C.; Chao, Y.C. A non-coding RNA of insect HzNV-1 virus establishes latent viral infection through microRNA. Sci. Rep. 2011, 1, 1–10. [Google Scholar]
- Van Den Abbeele, J.; Bourtzis, K.; Weiss, B.; Cordön-Rosales, C.; Miller, W.; Abd-Alla, A.M.M.; Parker, A. Enhancing tsetse fly refractoriness to trypanosome infection—A new IAEA coordinated research project. J. Invertebr. Pathol. 2013, 112, S142–S147. [Google Scholar] [CrossRef]
- Strand, M.R. Polydnaviruses. In Insect Virology; Asgari, S., Johnson, K.N., Eds.; Caister Academic Press: Norfolk, UK, 2010; pp. 171–197. [Google Scholar]
- Terzian, C.; Pelisson, A.; Bucheton, A. When Drosophila Meets Retrovirology: The Gypsy Case. In Transposons and the Dynamic Genome; Lankenau, D.H., Volff, J.N., Eds.; Springer-Verlag Berlin Heidelberg: London, UK, 2009; pp. 95–107. [Google Scholar]
- Varaldi, J.; Ravallec, M.; Labrosse, C.; Lopez-Ferber, M.; Bouletreau, M.; Fleury, F. Artifical transfer and morphological description of virus particles associated with superparasitism behaviour in a parasitoid wasp. J. Insect Physiol. 2006, 52, 1202–1212. [Google Scholar] [CrossRef]
- Varaldi, J.; Fouillet, P.; Ravallec, M.; Lopez-Ferber, M.; Bouletreau, M.; Fleury, F. Infectious behavior in a parasitoid. Science 2003, 302, 1930. [Google Scholar] [CrossRef]
- Rosen, L. Mechanism of vertical transmission of the dengue virus in mosquitoes. C. R. Acad. Sci. III 1987, 304, 347–350. [Google Scholar]
- Capinera, J.L. Habita. In Encyclopedia of Entomology, 2nd ed.; Capinera, J.L., Ed.; Springer Science: Dordrecht, The Netherlands, 2008; pp. 1761–1906. [Google Scholar]
- Lietze, V.U.; Geden, C.J.; Doyle, M.A.; Boucias, D.G. Disease dynamics and persistence of Musca domestica salivary gland hypertrophy virus infections in laboratory house fly (Musca domestica) populations. Appl. Environ. Microbiol. 2012, 78, 311–317. [Google Scholar] [CrossRef]
- Walshe, D.P.; Lehane, M.J.; Haines, L.R. Post eclosion age predicts the prevalence of midgut trypanosome infections in Glossina. PLoS One 2011, 6, e26984. [Google Scholar] [CrossRef]
- Bignell, D.E. The Arthropod Gut as an Environment for Microorganisms. In Microbial Ecology of the Gut; Clarke, R.T.J., Bauchop, T., Eds.; Academic Press Inc.: New York, NY, USA, 1983; pp. 205–227. [Google Scholar]
- Zhuzhikov, D.P. Function of the peritrophic membrane in Musca domestica L. and Calliphora erythrocephala Meig. J. Insect Physiol. 1964, 10, 273–278. [Google Scholar] [CrossRef]
- Chapman, R.F. Circulatory System, Blood and Immune Systems. In The Insects: Structure and Functions, 4th ed.; Simpson, S.J., Douglas, A.E., Eds.; Cambridge University Press: Cambridge, NY, USA, 1998; pp. 94–127. [Google Scholar]
- Feng, L.C. The role of the peritrophic membrane in Leishmania and Trypanosome infections of sandflies. Peking Nat. Hist. Bull. 1951, 19, 327–334. [Google Scholar]
- Hegedus, D.; Erlandson, M.; Gillott, C.; Toprak, U. New insights into peritrophic matrix synthesis, architecture, and function. Annu. Rev. Entomol. 2009, 54, 285–302. [Google Scholar] [CrossRef]
- Blackburn, K.; Wallbanks, K.R.; Molyneux, D.H.; Lavin, D.R.; Winstanley, S.L. The peritrophic membrane of the female sandfly Phlebotomus papatasi. Ann. Trop. Med. Parasitol. 1988, 82, 613–619. [Google Scholar]
- Bertram, D.S.; Bird, R.G. Studies on mosquito-borne viruses in their vector. I. The normal fine structure of the midgut epithelium of the adult female Aedes aegypti (L). and the functional significance of its modifications following a bloodmeal. Trans. R. Soc. Trop. Med. Hyg. 1961, 55, 404–423. [Google Scholar] [CrossRef]
- Chamberlain, R.W.; Sudia, W.D. Mechanism of transmission of viruses by mosquitoes. Annu. Rev. Entomol. 1961, 6, 371–390. [Google Scholar] [CrossRef]
- Gemetchu, T. The morphology and fine structure of the midgut and peritrophic membrane of the adult female, Phlebotomus longipes Parrot and Martin (Diptera: Psychodidae). Ann. Trop. Med. Parasitol. 1974, 68, 111–124. [Google Scholar]
- Mitsuhashi, W.; Kawakita, H.; Murakami, R.; Takemoto, Y.; Saiki, T.; Miyamoto, K.; Wada, S. Spindles of an entomopoxvirus facilitate its infection of the host insect by disrupting the peritrophic membrane. J. Virol. 2007, 81, 4235–4243. [Google Scholar] [CrossRef]
- Wang, P.; Granados, R.R. Molecular structure of the peritrophic membrane (PM): Identification of potential PM target sites for insect control. Arch. Insect Biochem. Physiol. 2001, 47, 110–118. [Google Scholar] [CrossRef]
- Tsai, Y.L.; Hayward, R.E.; Langer, R.C.; Fidock, D.A.; Vinetz, J.M. Disruption of Plasmodium falciparum chitinase markedly impairs parasite invasion of mosquito midgut. Infect. Immun. 2001, 69, 4048–4054. [Google Scholar] [CrossRef]
- Li, F.; Patra, K.P.; Vinetz, J.M. An anti-Chitinase malaria transmission-blocking single-chain antibody as an effector molecule for creating a Plasmodium falciparum-refractory mosquito. J. Infect. Dis. 2005, 192, 878–887. [Google Scholar] [CrossRef]
- Ejezie, G.C.; Davey, K.G. Some effects of mating in female tsetse, Glossina austeni Newst. J. Exp. Zool. 1977, 200, 303–310. [Google Scholar] [CrossRef]
- Harrison, R.; Hoover, K. Baculoviruses and other occluded viruses. In Insect Pathology, 2nd ed.; Vega, E.F., Kaya, H.K., Eds.; Elsevier: New York, NY, USA, 2012; pp. 73–131. [Google Scholar]
- Moscardi, F. Assessment of the application of baculoviruses for control of Lepidoptera. Annu. Rev. Entomol. 1999, 44, 257–289. [Google Scholar] [CrossRef]
- Horton, H.M.; Burand, J.P. Saturable attachment sites for polyhedron-derived baculovirus on insect cells and evidence for entry via direct membrane fusion. J. Virol. 1993, 67, 1860–1868. [Google Scholar]
- Sparks, W.O.; Rohlfing, A.; Bonning, B.C. A peptide with similarity to baculovirus ODV-E66 binds the gut epithelium of Heliothis virescens and impedes infection with Autographa californica multiple nucleopolyhedrovirus. J. Gen. Virol. 2011, 92, 1051–1060. [Google Scholar] [CrossRef]
- Sparks, W.O.; Harrison, R.L.; Bonning, B.C. Autographa californica multiple nucleopolyhedrovirus ODV-E56 is a per os infectivity factor, but is not essential for binding and fusion of occlusion-derived virus to the host midgut. Virology 2011, 409, 69–76. [Google Scholar] [CrossRef]
- Abd-Alla, A.M.; Adun, H.; Parker, A.G.; Vreysen, M.J.; Bergoin, M. The antiviral drug valacyclovir successfully suppresses salivary gland hypertrophy virus (SGHV) in laboratory colonies of Glossina pallidipes. PLoS One 2012, 7, e38417. [Google Scholar]
- Feldmann, U. Guidelines for the rearing of tsetse flies using the membrane feeding technique. In Techniques of Insect Rearing for the Development of Integrated Pest and Vector Management Strategies; Ochieng'-Odero, J.P.R., Ed.; ICIPE Science Press: Nairobi, Kenya, 1994; pp. 449–471. [Google Scholar]
- Enserink, M. Welcome to Ethiopia’s fly factory. Science 2007, 317, 310–313. [Google Scholar] [CrossRef]
- Malele, I.I.; Manangwa, O.; Nyingilili, H.H.; Kitwika, W.A.; Lyaruu, E.A.; Msangi, A.R.; Ouma, J.O.; Nkwangulila, G.; Abd-Alla, A.M.M. Prevalence of SGHV among tsetse species of economic importance in Tanzania and their implication for SIT application. J. Invertebr. Pathol. 2013, 112, S133–S137. [Google Scholar] [CrossRef]
- Abd-Alla, A.M.M.; Arif, B. Foreword. J. Invertebr. Pathol. 2013, 112, S1. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
