Simple Summary
Paracoccus marginatus is a polyphagous pest that threatens more than 200 plant species. Currently, the most widely applied biological control methods predominantly utilize parasitoid natural enemies, while the application of predatory natural enemies remains relatively limited. This study investigates the predatory behavior of Diomus guilavoguii against Paracoccus marginatus by observing functional responses, search effects and interspecific interference effects under controlled laboratory conditions. The results indicate that Diomus guilavoguii shows promise as a biological control agent for managing Paracoccus marginatus infestations.
Abstract
Paracoccus marginatus (Hemiptera: Pseudococcidae) poses a significant threat to over 200 plant species, severely impacting agricultural productivity. Diomus guilavoguii (Coleoptera: Coccinellidae) is a natural predator of P. marginatus. To develop an effective and environmentally friendly management strategy against P. marginatus, this study investigates the predation relationship between D. guilavoguii and P. marginatus by focusing on functional response and mutual interference under controlled laboratory conditions (Petri dishes). The results indicated that D. guilavoguii exhibits a type II functional response toward P. marginatus, with adults of D. guilavoguii demonstrating superior efficiency in preying upon P. marginatus (the theoretical daily maximum predation rate for female adults of D. guilavoguii on young mealybugs is 416.667). However, a decrease in the predators’ search effect was observed with increasing prey density. Additionally, interspecific interference competition intensified as the number of predators increased, resulting in reduced predation efficiency. Consequently, D. guilavoguii shows promise biological control agent for the management of P. marginatus under laboratory conditions, although further studies in greenhouse and field environments are required to validate its potential in practical pest management.
1. Introduction
The papaya mealybug, Paracoccus marginatus Williams & Granara de Willink (Hemiptera: Pseudococcidae), is native to Mexico and Central America [1]. It has rapidly expanded its host range and invaded over 50 countries and regions, adversely affecting agricultural and forestry industries [2]. This species exhibits strong reproductive capacity and remarkable adaptability to diverse environments, making it a significant global invasive pest.
P. marginatus mainly congregates on the veins of leaves and fruits, inserting its stylets into plant tissues such as leaf epidermis, fruit skin, or stems to feed on cell sap [3,4,5,6,7]. Meanwhile, it inject toxic substances into plants, causing severe damage including chlorosis, distortion, stunting, premature leaf and fruit drop, honeydew production, sooty mold formation, and potentially plant death [8,9]. Moreover, this insect also acts as a vector of plant viruses such as Piper yellow mottle virus, Cocoa swollen shoot virus, and Papaya ringspot virus [8]. P. marginatus infestations can reduce crop yields by 10–60%, and in extreme cases up to 90% [10]. Therefore, it is imperative to implement suitable pest control measures to prevent further damage by P. marginatus.
Chemical control was once regarded as an effective method for preventing plant infestation by P. marginatus. Early pesticides commonly used to control P. marginatus included methyl demeton, quinalphos, fenthion, acephate, dimethoate, malathion, and white mineral oils [11]. However, due to the wax coating and cottony ovisac of the mealybug, it is challenging to develop effective strategies to ensure adequate chemical penetration [7,11]. Some newer insecticides, such as buprofezin, imidacloprid, thiomethoxam, spirodiclofen, pyridaben, chlorfenapyr, and plant essential oils (neem, citrus, garlic, and castor), have shown promise in controlling the papaya mealybug [11,12,13,14]. Nevertheless, chemical pesticides often have residual effects and can cause environmental pollution [7,11,12,14]. Additionally, frequent use of chemical pesticides can lead to pest resistance and may harm non-targeted insects and natural enemies, complicating pest management efforts [15].
Compared with chemical control, biological control aligns more closely with the principles of sustainable development and environmental protection. Commonly employed biological control agents for managing papaya mealybug include parasitoids such as Acerophagus papayae Noyes & Schauff, Anagyrus loecki Noyes & Menezes, and Pseudleptomastix mexicana Noyes & Schauff (Hymenoptera: Encyrtidae) [15,16,17,18,19,20]. These parasitoids have been shown to significantly reduce papaya mealybug populations in the field, thereby mitigating potential economic losses [16,18,19]. However, there are instances where the parasitoid populations fail to recover adequately after release [16,17].
As an important natural enemy of insects, predatory ladybird beetles have been utilized in biological control for nearly 120 years. Due to their broad prey spectrum and adaptability to new environments, they play a crucial role in the management of pest populations [21]. Xia et al. [22] demonstrated that Chilocorus kuwanae (Coleoptera: Coccinellidae), as a natural predator of various pests, can prey on 27 species across 5 families. Li et al. [23] reported that Coccinella septempunctata, Chilocorus kuwanae, Harmonia axyridis, Coccinula quatuordecimpustulata, Rodolia limbata and Synharmonia bissexnotata (Coleoptera: Coccinellidae) all exhibit biocontrol potential against Matsucoccus sinensis (Hemiptera: Margarodidae). Similarly, the feeding behavior of Cryptolaemus montrouzieri (Coleoptera: Coccinellidae) has been well studied, showing its strong efficiency in suppressing mealybug populations, which provides a valuable reference for evaluating other coccinellid predators [24,25]. Information regarding the host range of species within the genus Diomus remains limited; however, existing data suggest a clear preference for mealybugs [26]. Diomus guilavoguii Duverger, native to Conakry, Guinea [27], was first recorded in Guangzhou, China in 2022, where it was observed preying extensively on P. marginatus [28]. However, detailed studies on the predatory efficacy of D. guilavoguii against P. marginatus are unavailable to date. Therefore, this study aims to evaluate the predation rate, functional response, search effect, and mutual interference of D. guilavoguii when preying on P. marginatus.
2. Material and Methods
2.1. Paracoccus marginatus
P. marginatus was collected from infested Jatropha integerrima (Malpighiales: Euphorbiaceae) leaves on the campus of South China Agricultural University (SCAU). Subsequently, J. integerrima seedlings were inoculated with P. marginatus to propagate the pest for further study.
2.2. Diomus guilavoguii
D. guilavoguii was collected from J. integerrima leaves infested by P. marginatus on the campus of South China Agricultural University (SCAU). D. guilavoguii were kept in a square cage (60 cm × 60 cm × 60 cm) and fed with P. marginatus on fresh J. integerrima leaves. After oviposition, the eggs on the leaves were transferred to disposable polystyrene Petri dishes (3.5 cm in diameter, 1 cm in height; Guangzhou Qianhui Reagent Instrument Co., Ltd., Guangzhou, China) containing moderately sized P. marginatus leaves and moistened cotton wool. The Petri dishes were wrapped with cling film to prevent desiccation and larval escape. The leaves with prey were changed daily to maintain cleanliness and ensure proper feeding of the predators. Larvae were observed regularly until they emerged as adults. Adults were then transferred using a fine brush into new cages for mating and subsequent oviposition.
2.3. Functional Response
Third-instar and fourth-instar larvae, male and female adults of D. guilavoguii were selected and kept without food for 24 h in separate disposable polystyrene Petri dishes. The selected individuals included larvae within two hours after molting and adults within 24 to 48 h after eclosion. Subsequently, all individuals were exposed to varying densities of 3rd- to 4th-instars nymphs and adults (hereafter referred to as “late-instar nymphs and adults”) of P. marginatus (1, 3, 5, 7, 9, 11, 13, 15, 17 and 19). Third- and fourth-instar larvae were also exposed to different densities of 1st- to 2nd-instar nymphs (hereafter referred to as “young nymphs”) of P. marginatus (10, 20, 30, 40, 50, 60, 70, 80, 90 and 100). Male and female adults of D. guilavoguii were also exposed to different densities of young nymphs of P. marginatus (80, 120, 160, 200, 240, 280, 320, 360, 400 and 440). Leaves (approximately 6 cm in length after being trimmed into square-shaped sections) of J. integerrima were placed in disposable polystyrene Petri dishes (9 cm in diameter; Guangzhou Qianhui Reagent Instrument Co., Ltd., Guangzhou, China), and predators were transferred to these dishes using a fine brush. Each dish contained one predator, a damp cotton ball, and was covered with plastic wrap pierced with needles. All dishes were placed in an artificial climate chamber at a temperature of 26 ± 1 °C with a 14 h photophase. After 24 h of exposure, the remaining number of P. marginatus was counted. Each treatment was repeated five times.
2.4. Mutual Interference
The predators, which had been starved for 24 h, were divided into five groups, and placed in separate disposable polystyrene Petri dishes (9 cm in diameter), each containing fresh J. integerrima leaves. Each dish contained 1, 2, 3, 4, or 5 predators, respectively. The 3rd and 4th instar larvae of D. guilavoguii were exposed to either 200 young nymphs of P. marginatus or 30 late-instar nymphs and adults of P. marginatus, while adult females and males were exposed to either 500 young nymphs of P. marginatus or 30 late-instar nymphs and adults of P. marginatus. Each dish had a moist cotton plug and was covered with plastic wrap punctured with needles to allow ventilation. All dishes were placed in an artificial climate chamber (RXZ-500A, Ningbo Jiangnan Instrument Factory, Ningbo, China) maintained at 26 ± 1 °C with a 14 h photoperiod. After 24 h of exposure, the remaining number of P. marginatus was recorded. Each treatment was repeated five times.
2.5. Analysis of Data
Data on predation rate and the amount of D. guilavoguii feeding on P. marginatus were analyzed using one-way analysis of variance (ANOVA) to test for differences among treatments. When significant differences were detected (p < 0.05), means were compared using Tukey’s Honestly Significant Difference (HSD) test. Statistical analyses were conducted using SPSS Statistics 25.0 software (IBM Corp., Armonk, NY, USA).
The type of functional response was determined by nonlinear logistic regression using the “frair” package within the “R” software version 4.0.2 (R Foundation for Statistical Computing, Vienna, Austria) [29]. The polynomial function that describes the relationship between and was derived using the following equation:
In this equation, presents the number of prey consumed, denotes the initial number of prey provided, and P0, P1, P2, and P3, are the constant, linear, quadratic, and cubic coefficients (maximum likelihood estimates), respectively, related to the slope of the curve. A positive and a negative in polynomial logistic regression indicate a type III functional response. Conversely, when is negative, the number of prey consumed initially decreases with increasing prey availability, which characterizes a type II functional response [30].
The selection of the functional response model entails the application of polynomial logistic regression. For a clear type II response, both the disk equation [31] and the random attack equation [32] can be used to estimate handling time (Th) and attack rate (a). Given that the disk equation requires constant prey density during experiments, we opted for the random attack equation as our model.
Here, is the number of prey consumed, is the initial number of prey provided, a represents the attack rate, refers to the time required for capturing, subduing, and consuming prey (digestion may continue after ingestion but does not necessarily prevent subsequent predation) [33,34], T indicates the total exposure time of the predator to prey, typically set at 24 h. The attack rate (a) and handling time () were estimated using nonlinear regression, following the method of Rogers [32], and compared using the “frair_compare” function from the “frair” package in “R” software [29]. Additionally, the maximum predation rate was also calculated.
The search effect (S) is related to the prey density (N), and is defined as the proportion of the available prey population that a predator effectively encounters and attacks per unit time [35,36]. It can be expressed by the formula , where S represents the searching effect, while the parameters a and are derived from the type II functional response.
The predation efficiency (E) is affected by the predator’s own density (P). As the predator density increases, the interference effect also intensifies. The formula for predation efficiency is , where Na represents the number of prey captured [37]. The mutual interference equation is , where Q denotes the search constant and m presents the interference coefficient. The apportionment competition intensity (I) fits the formula , where is the predation efficiency of a single predator, and is the predation efficiency of a group of p predator. The mutual interference coefficient (m) among predators was measured using the Hassell and Varley [38] model: . This was achieved by fitting the data to the equation using a linear regression procedure in IBM SPSS Statistics 25.
3. Results
3.1. Predation Rate of Diomus guilavoguii Feeding on Paracoccus marginatus
Through analysis of variance, there was a statistically significant difference in predation rates observed for D. guilavoguii at different densities of P. marginatus. The results showed that the overall consumption percentage of mealybug by D. guilavoguii decreased as prey density increased. Specifically, at the highest prey density tested in the experiment, the predation rate for adult D. guilavoguii was more than 0.22, while the predation rate for larvae was less than 0.28 (Table 1 and Table 2).

Table 1.
Predation rate and amount of Diomus guilavoguii feeding on young nymphs of Paracoccus marginatus.

Table 2.
Predation rate and amount of Diomus guilavoguii feeding on late-instar nymphs and adults of Paracoccus marginatus.
3.2. Functional Response of D. guilavoguii Feeding on P. marginatus
In these experiments, D. guilavoguii showed predatory capability against both nymph and adult stages of P. marginatus (Table 3). The results of the logistic regression analysis were significant (p < 0.05), with a negative linear coefficient (), indicating that D. guilavoguii displays a type II functional response.

Table 3.
Maximum likelihood estimates for logistic regression of functional response of Diomus guilavoguii on Paracoccus marginatus.
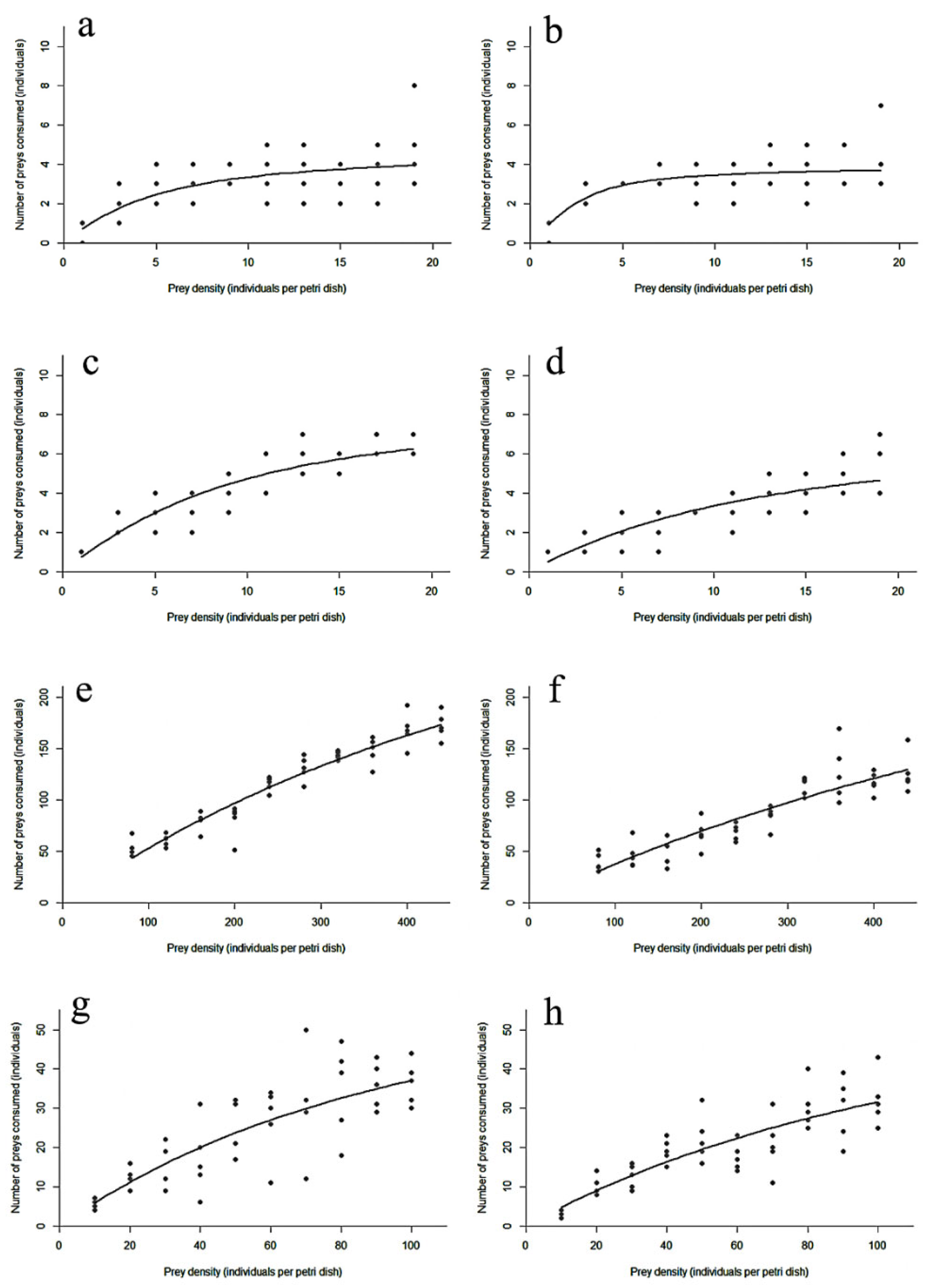
The functional response curves for larvae and adults of D. guilavoguii feeding on P. marginatus at different ages and densities are shown in Figure 1. Initially, the consumption rate increased sharply with prey density before eventually plateauing.
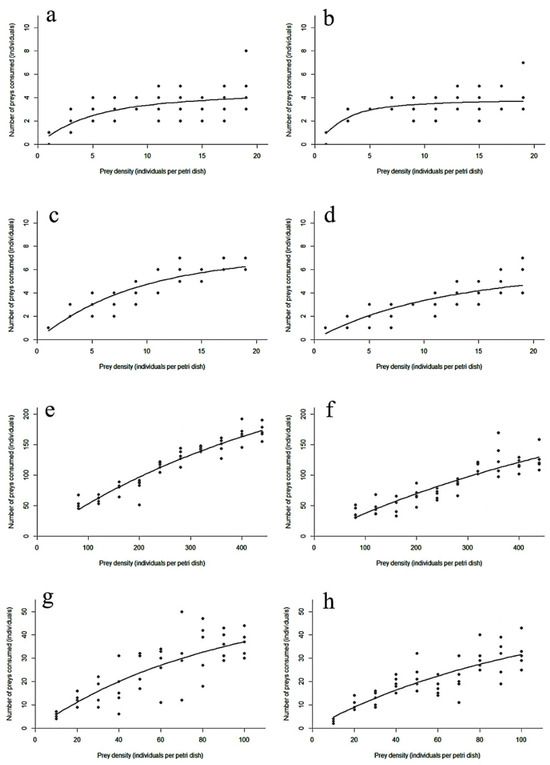
Figure 1.
Paracoccus marginatus consumption by Diomus guilavoguii at different prey densities. Note: (a) female adults of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (b) male adults of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (c) 4th larvae of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (d) 3rd larvae of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (e) female adults of Diomus guilavoguii on young nymphs of Paracoccus marginatus, (f) male adults of Diomus guilavoguii on young nymphs of Paracoccus marginatus, (g) 4th larvae of Diomus guilavoguii on young nymphs of Paracoccus marginatus, (h) 3rd larvae of Diomus guilavoguii on young nymphs of Paracoccus marginatus.
The instantaneous attack rate (a) of D. guilavoguii preying upon young nymphs of P. marginatus did not differ significantly between developmental stages, except for male adults, which exhibited a significantly lower (a) value. However, when preying on late-instar nymphs and adult mealybugs, the attack rate showed statistically different (a) values, with female adults (1.4124), male adults (3.4485), 4th instar larvae (1.3898), and 3rd instar larvae (0.7280).
At the same prey age, the predation rate of 4th instar larvae was significantly higher than that of 3rd instar larvae, and female adults also had a significantly higher predation rate than male adults. As the age of the mealybug increased, the handling time of D. guilavoguii decreased. For 1st to 2nd instar mealybugs, female adults spent the shortest time capturing, ingesting each prey item compared to other stages, while larvae required more time. For 3rd to 4th instar mealybugs, 4th instar larvae of D. guilavoguii performed better in terms of the time spent capturing, ingesting each prey item compared to adults and 3rd instar larvae. The maximum predation rate () to 1st to 2nd instar mealybugs was significantly higher in adults (female: 416.67, male: 400) compared to larval stages (3rd instar: 64.52, 4th instar: 70.93). Conversely, for 3rd to 4th instar mealybugs, the maximum predation rate of adults (female: 4.699, male: 3.925) was significantly lower than that of larval stages (3rd instar: 8.772, 4th instar: 7.576) (Table 4).

Table 4.
Predatory functional responses of Diomus guilavoguii feeding on Paracoccus marginatus.
3.3. Search Effects of D. guilavoguii Feeding on P. marginatus
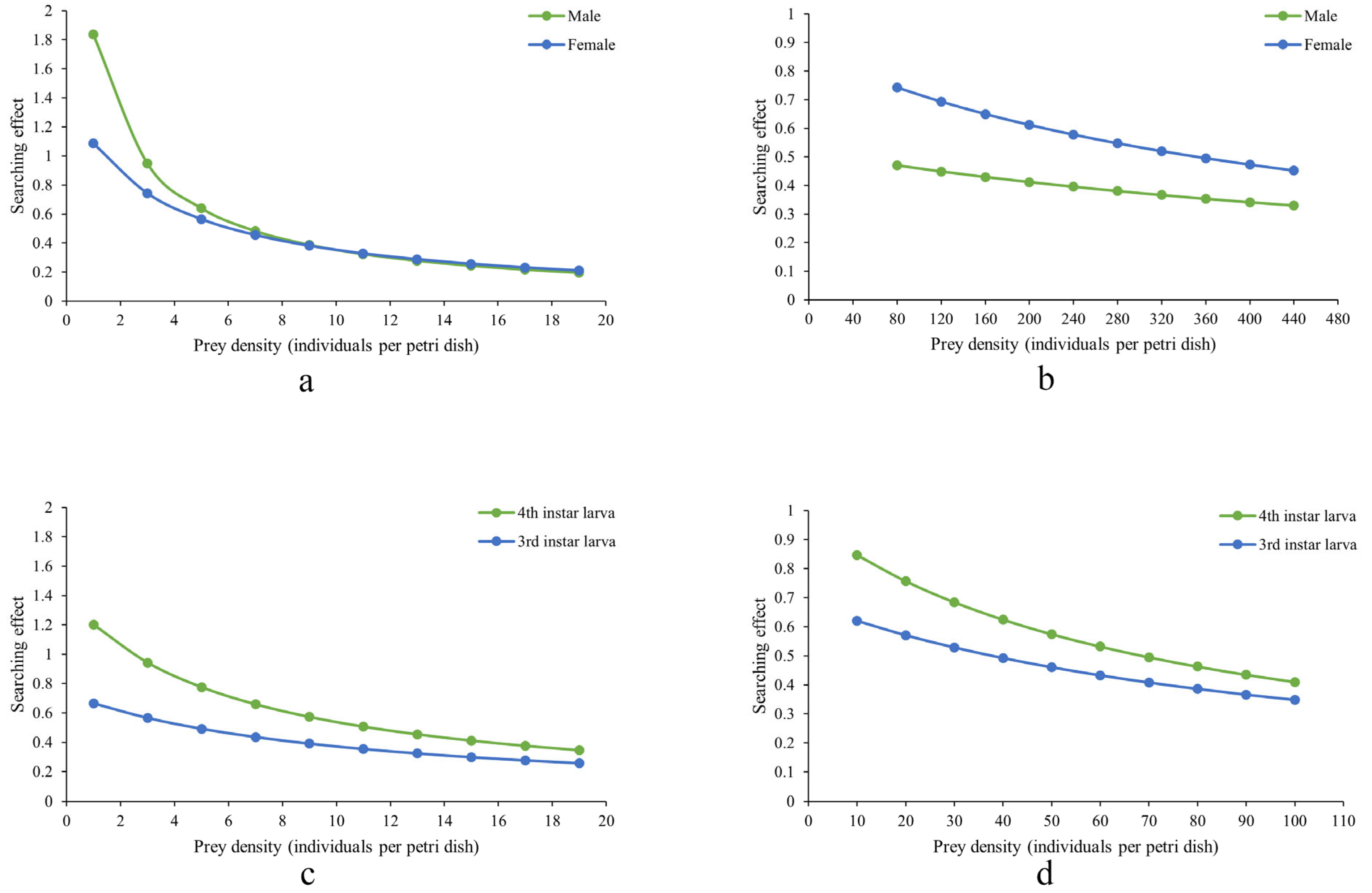
Figure 2 shows that the search effects of D. guilavoguii feeding on P. marginatus decreased with increasing prey density. In addition, the reduction in search effects was more pronounced for late-instar nymphs and adults of P. marginatus compared to younger nymphs, suggesting that higher prey density had a more significant impact on the predatory behavior of ladybirds targeting older stages of the prey. In the case of young nymphs of P. marginatus, female adults of D. guilavoguii consistently exhibit a higher search effect than male adults. Meanwhile, at the same instar stage and prey density, fourth instar larvae of D. guilavoguii demonstrate a higher search effect compared to third instar larvae. These differences can be attributed to variations in handling time, as shorter handling times in female adults and fourth-instar larvae contribute to higher searching efficiency. Since search effect values are derived from the type II functional response model (based on attack rate (a) and handling time ), statistical differences among developmental stages are reflected in the comparisons of these parameters. Female adults and fourth-instar larvae exhibited significantly higher attack rates than third-instar larvae (p < 0.05), which accounts for their consistently higher search effects across all prey densities.
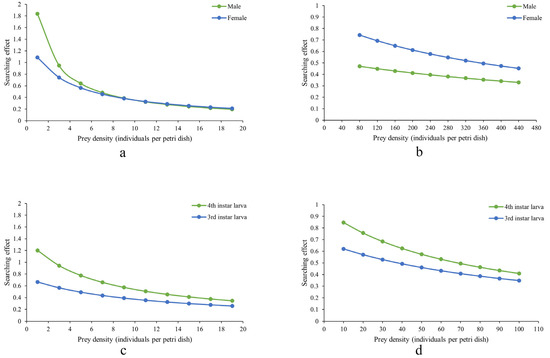
Figure 2.
Search effects of Diomus guilavoguii feeding on Paracoccus marginatus. Note: (a) adults of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (b) adults of Diomus guilavoguii on young nymphs of Paracoccus marginatus, (c) larvaes of Diomus guilavoguii on late-instar nymphs and adults of Paracoccus marginatus, (d) larvae of Diomus guilavoguii on young nymphs of Paracoccus marginatus.
3.4. Mutual Interference of D. guilavoguii Feeding on P. marginatus
From the perspective of prey consumed per predator, when there is only one predator, the predation rate per predator reached its peak. When five predators are present in a group, the predation rate per predator drops to its lowest point. As predator density increases, both the daily predation and average predation efficiency of D. guilavoguii against P. marginatus decrease, while competition intensifies. At the same predator density, the average predation efficiency of D. guilavoguii against elder P. marginatus is lower than that against younger nymphs, and the competition intensity from elder mealybug is also lower than that from younger nymphs.
As shown in Table 5 and Table 6, the maximum daily predation of female adult D. guilavoguii on older nymphs was 7.6 per day (), while on younger nymphs it was 132.6 per day (). For male adults, the maximum daily predation on older nymphs was 5.25 per day (), and on younger nymphs it was 92.6 per day (). The 4th instar larvae had a maximum daily predation of 5 on older nymphs () and 47 on younger nymphs (). Third instar larvae showed a maximum daily predation of 3.8 on older nymphs () and 40.6 on younger nymphs ().

Table 5.
Mutual interference of Diomus guilavoguii feeding on late-instar nymphs and adults of Paracoccus marginatus.

Table 6.
Mutual interference of Diomus guilavoguii feeding on young nymphs of Paracoccus marginatus.
The mutual interference parameter (m) for female adults preying on late-instar nymphs and adults of mealybugs was the highest, while that for 3rd instar larvae preying on 1st to 2nd instar mealybugs was the lowest.
4. Discussion
Functional response has been widely used to evaluate the predation ability of natural enemies. However, no studies have yet investigated the functional response of D. guilavoguii toward P. marginatus. Our study revealed that both larvae and adults of D. guilavoguii exhibit a type II functional response when feeding on P. marginatus under laboratory conditions, a pattern also observed in Harmonia axyridis preying on various prey species [39,40].
In a type II functional response, the attack rate and handling time are critical determinants of predation efficiency. The attack rate of fourth-instar larvae and female adults is higher than that of other developmental stages of D. guilavoguii, indicating a stronger predation capacity during these stages. This pattern is consistent with the feeding behavior of Cryptolaemus montrouzieri when preying on P. marginatus [25]. The increased predation ability in these stages may be attributed to the higher nutritional demands of fourth-instar larvae preparing for pupation and female adults undergoing reproductive development. The theoretical maximum daily predation rate, which is inversely related to handling time, further reflects the predation efficiency of D. guilavoguii. Notably, adults consumed significantly more young nymphs than larvae, demonstrating a higher predatory capability in the adult stage. This observation corresponds to the predation behavior of Xylocoris sordidus (Hemiptera: Anthocoridae) feeding on Enneothrips enigmaticus (Thysanoptera: Thripidae) [41]. Moreover, D. guilavoguii exhibited a preference for younger nymphs over older mealybugs, a trend also reported in Arma chinensis (Hemiptera: Pentatomidae) preying on Henosepilachna vigintioctopunctata (Coleoptera: Coccinellidae) [42]. Additionally, as prey density increases, the predation rate per individual decreases and the total number of prey consumed reaches a plateau, a phenomenon similarly observed in Mallada basalis (Neuroptera: Chrysopidae) feeding on Spodoptera frugiperda (Lepidoptera: Noctuidae) [43]. These findings suggest that for optimal biological control using D. guilavoguii, they should be released early during a P. marginatus outbreak.
Moreover, female adults of D. guilavoguii consistently exhibit a higher search effect than males when preying on young nymphs of P. marginatus. Additionally, 4th-instar larvae show greater search effect than 3rd-instar larvae on the same prey stage, a pattern also observed in Harmonia axyridis preying on Rhopalosiphum nymphaeae (Hemiptera: Aphididae) [44]. A higher searching effect suggests that natural enemies can regulate prey populations even at low densities, highlighting their potential effectiveness in controlling pest outbreaks during the early stages of population growth. In other words, female adults are more effective than males in preying on young mealybugs, and under the same prey stage and density, 4th-instar larvae exhibit stronger predatory capacity compared to 3rd-instar larvae.
The mutual interference experiments revealed that increased predator density intensifies intraspecific competition, leading to a reduced per capita predation rate. Comparable effects have been documented in other predator-prey systems, such as Orius tantillus (Hemiptera: Anthocoridae) preying on Thrips hawaiiensis (Thysanoptera: Thripidae) [45]. Regarding young nymphs of P. marginatus, when the density of D. guilavoguii reached the maximum level (5 individuals per Petri dish), the intensity of intraspecific competition per ladybird was highest among female adults, which may be attributed to variations in predation capacity across developmental stages.
According to Hassell and Varley, mutual interference can play an important role in regulating host population dynamics [38]. In this context, the observed interference in D. guilavoguii suggests that although increased predator density can suppress P. marginatus populations, excessive intraspecific competition may diminish individual predation efficiency, thereby preventing uncontrolled host depletion. This dynamic indicates a potential mechanism through which predator interference may contribute to long-term pest population regulation. While our results indicate that increased predator density reduces the individual efficiency of D. guilavoguii due to mutual interference, predator density can be effectively managed in practical orchard settings. In practice, augmentative releases of coccinellid predators are a widely adopted strategy in integrated pest management programs, offering a means to optimize predator numbers, minimize interference, and ensure effective pest suppression [46,47].
However, this study was conducted under controlled indoor conditions using Petri dishes with fixed prey density, which may artificially enhance predation efficiency. In natural environments, preys are typically dispersed across various parts of host plants, thereby increasing the time and effort required for predators to locate them [36]. Consequently, the actual predation rate in the field is likely lower than that observed in laboratory conditions, which often deviates from the results of functional response studies conducted in controlled chambers [48]. Moreover, the predation capacity of natural enemies in the field is also affected by multiple factors. For example, higher pest densities can reduce predator dispersal and intensify both intra- and interspecific competition, thereby lowering individual predation efficiency [49]. Additionally, environmental factors such as weather conditions, temperature, host plants and the presence of multiple prey species also play significant roles [50]. Therefore, future studies should take into account various natural factors, such as abiotic conditions (including temperature, humidity, and photoperiod), prey density, prey species diversity, and interspecific competition among predators, in order to gain a more comprehensive understanding of the interactions that affect predation dynamics. In this regard, we intend to investigate higher prey densities under greenhouse or semi-field conditions to confirm whether the asymptotic predation rate observed in these settings differs from that estimated in laboratory environments. Greenhouse or semi-field experiments involving potted plants or field cages can effectively simulate natural prey distribution patterns and plant structures, thereby facilitating a more accurate assessment of predator searching efficiency and handling time. The integration of these experimental approaches with field release trials will be essential for verifying whether the parameters derived from laboratory studies can be successfully applied to achieve practical pest suppression. Ultimately, combining laboratory-based models with greenhouse and field evaluations will establish a more robust basis for incorporating D. guilavoguii into integrated pest management (IPM) programs.
In conclusion, this study demonstrates the potential of that D. guilavoguii as a biological control agent against P. marginatus under laboratory conditions. However, further investigation is required to evaluate its efficacy in practical field applications.
Author Contributions
Methodology, Q.L., G.R. and M.T.; Software and Data Analysis, Q.L.; Investigation, Q.L., G.R., M.T., X.G. and M.Y.; Resources, X.W. and X.C.; Writing—Original Draft Preparation, Q.L.; Writing—Review and Editing, X.C. and X.W.; Supervision, X.C.; Funding Acquisition, X.C. All authors have read and agreed to the published version of the manuscript.
Funding
The present study was supported by the Guangzhou Collaborative Innovation Center on Science-Tech of Ecology and Landscape (202206010058) and the Guangdong Provincial Key Laboratory of Silviculture, Protection and Utilization, under the project titled “Investigation and Application of Predatory Coccinellidae Resources in the Urban Forest Ecosystems within the Guangdong-Hong Kong-Macao Greater Bay Area” (No. SPU 2024-10).
Data Availability Statement
All data is contained within the article.
Acknowledgments
We sincerely thank Shaukat Ali from South China Agricultural University for his guidance in the data analysis of this study, as well as his comments on an earlier draft of the manuscript. We also extend our sincere gratitude to the editors and reviewers for their critical comments and suggestions regarding this manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Miller, D.R.; Williams, D.J.; Hamon, A.B. Notes on a new mealybug (Hemiptera: Coccoidea: Pseudococcidae) pest in Florida and the Caribbean: The papaya mealybug, Paracoccus marginatus Williams and Granara de Willink. Insecta Mundi 1999, 13, 179–181. [Google Scholar]
- Zhao, Q.; Li, H.; Chen, C.; Fan, S.; Wei, J.; Cai, B.; Zhang, H. Potential global distribution of Paracoccus marginatus, under climate change conditions, using MaxEnt. Insects 2024, 15, 98. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.Z.; He, R.R.; Wu, M.T.; Gu, Y.J.; Ren, J.M.; Liang, F.; Li, H.L.; Hu, X.N.; Qiu, B.L.; Mannion, C.M.; et al. First report of the papaya mealybug, Paracoccus marginatus (Hemiptera: Pseudococcidae), in China and genetic record for its recent invasion in Asia and Africa. Fla. Entomol. 2015, 98, 1157–1162. [Google Scholar] [CrossRef]
- Heya, H.M.; Khamis, F.M.; Onyambu, G.K.; Akutse, K.S.; Mohamed, S.A.; Kimathi, E.K.; Ombura, F.L.O.; Ekesi, S.; Dubois, T.; Subramanian, S.; et al. Characterization and risk assessment of the invasive papaya mealybug, Paracoccus marginatus, in Kenya under changing climate. J. Appl. Entomol. 2020, 144, 442–458. [Google Scholar] [CrossRef]
- Chuai, H.Y.; Shi, M.Z.; Li, J.Y.; Zheng, L.Z.; Fu, J.W. Fitness of the papaya mealybug, Paracoccus marginatus (Hemiptera: Pseudococcidae), after transferring from Solanum tuberosum to Carica papaya, Ipomoea batatas, and Alternanthera philoxeroides. Insects 2022, 13, 804. [Google Scholar] [CrossRef]
- Mendel, Z.; Watson, G.W.; Protasov, A.; Spodek, M. First record of the papaya mealybug, Paracoccus marginatus Williams & Granara de Willink (Hemiptera: Coccomorpha: Pseudococcidae), in the Western Palaearctic. EPPO Bull. 2016, 46, 580–582. [Google Scholar] [CrossRef]
- Mwanauta, R.W.; Ndakidemi, P.A.; Venkataramana, P.B. Biopesticide efficacy of four plant essential oils against papaya mealybug, Paracoccus marginatus Williams and Granara de Willink (Hemiptera: Pseudococcidae). Heliyon 2023, 9, e14162. [Google Scholar] [CrossRef]
- Lu, H.; Tang, J.H.; Lu, B.Q.; Lu, F.P.; He, X.; Chen, Q. Risk analysis of Paracoccus marginatus in China. China Plant Prot. 2020, 40, 73–75. [Google Scholar]
- Song, Z.J.; Zhao, Q.Y.; Ma, C.; Chen, R.R.; Ma, T.B.; Li, Z.H.; Guo, P.Z. Quarantine disinfestation of papaya mealybug, Paracoccus marginatus (Hemiptera: Pseudococcidae) using Gamma and X-rays irradiation. Insects 2023, 14, 682. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Liu, X.Q.; Liang, X.; Liu, Y.; Wu, C.L.; Xu, X.L.; Wu, Y.; Chen, Q.; Yao, X.W.; Qiao, Y.; et al. Resistant cassava cultivars inhibit the papaya mealybug Paracoccus marginatus population based on their interaction: From physiological and biochemical perspectives. J. Pest Sci. 2023, 96, 555–572. [Google Scholar] [CrossRef]
- Laneesha, M.; Suroshe, S.S.; Babasaheb, B.F.; Shankarganesh, K. Papaya mealybug (Paracoccus marginatus) (Hemiptera: Pseudococcidae): A new threat to agri-horticulture ecosystem. Indian J. Agric. Sci. 2020, 90, 455–462. [Google Scholar] [CrossRef]
- Yang, S.Y.; Zhang, R.; Zhang, H.; Wang, H.G.; Wang, H.X.; Chen, Y.H. Synergistic effect of five spray adjuvants on spirodiclofen 240 g/L suspension concentrate and pyridaben 15% emulsifiable concentrate for thecontrol of Paracoccus marginatus. Chin. J. Pestic. Sci. 2019, 21, 531–537. Available online: http://nyxxb.alljournal.cn (accessed on 24 May 2025).
- Zhang, R.; Yang, S.Y.; Mao, X.X.; Lin, S.Y.; Wang, W.J.; Zhang, Z.X.; Cheng, D.M. Toxicities and synergism of different pesticides against Paracoccus marginatus. J. Environ. Entomol. 2020, 42, 221–226. Available online: http://hjkcxb.alljournals.net (accessed on 24 May 2025).
- Li, J.Y.; Chen, Y.T.; Wang, Q.Y.; Zheng, L.Z.; Fu, J.W.; Shi, M.Z. Sublethal and transgenerational toxicities of chlorfenapyr on biological traits and enzyme activities of Paracoccus marginatus (Hemiptera: Pseudococcidae). Insects 2022, 13, 874. [Google Scholar] [CrossRef]
- Sakthivel, P.; Karuppuchamy, P.; Kalyanasundaram, M.; Srinivasan, T. Toxicity of insecticides to papaya mealybug parasitoid, Acerophagus papayae (Noyes and Schauff) (Hymenoptera: Encyrtidae). J. Biol. Control 2012, 26, 274–278. [Google Scholar]
- Muniappan, R.; Meyerdirk, D.E.; Sengebau, F.M.; Berringer, D.D.; Reddy, G.V.P. Classical biological control of the papaya mealybug Paracocus marginatus (Hemiptera: Pseudococidae) in the Republic of Palau. Fla. Entomol. 2006, 89, 212–217. [Google Scholar] [CrossRef]
- Amarasekare, K.G.; Mannion, C.M.; Epsky, N.D. Efficiency and establishment of three introduced parasitoids of the mealybug Paracocus marginatus (Hemiptera: Pseudococcidae). Biol. Control 2009, 51, 91–95. [Google Scholar] [CrossRef]
- Myrick, S.; Norton, G.W.; Selvaraj, K.N.; Natarajan, K.; Muniappan, R. Economic impact of classical biological control of papaya mealybug in India. Crop Prot. 2014, 56, 82–86. [Google Scholar] [CrossRef]
- Hartmann, L.; Grandgirard, J.; Germain, J.F.; Hostachy, B.; Wong, M. First report of the papaya mealybug, Paracoccus marginatus (Coccomorpha: Pseudococcidae), in Tahiti, French Polynesia. EPPO Bull. 2021, 51, 229–232. [Google Scholar] [CrossRef]
- Le, K.H.; Tran, T.H.D.; Tran, D.H.; Nguyen, T.D.; Van Doan, C. Parasitoid wasp Acerophagus papayae: A promising solution for the control of papaya mealybug Paracoccus marginatus in cassava fields in Vietnam. Insects 2023, 14, 528. [Google Scholar] [CrossRef]
- Duan, J.C. Study on Artificial Rearing Technology of Propylaea quatuordecimpunctata and Evaluation of Its Pest Control Function. Master’s Thesis, Xinjiang Agricultural University, Urumqi, China, 2022. [Google Scholar]
- Xia, B.C.; Zhang, Y.; Shen, B.Y. Biology of Chilocorus kuwanae and its field applications. Chin. J. Biol. Control. 1986, 2, 70–74. [Google Scholar]
- Li, D.C.; Fu, Z.L.; Liu, J.Q.; Zhang, W.Y.; Yang, J. Investigation and study on the types and occurrence patterns of Matsucoccus sinensis and its natural enemies in the southern region of Gansu Province. J. Gansu Agric. Univ. 2024, 59, 180–187. [Google Scholar]
- Kairo, M.T.K.; Paraiso, O.; Gautam, R.D.; Peterkin, D.D. Cryptolaemus montrouzieri (Mulsant) (Coccinellidae: Scymninae): A review of biology, ecology, and use in biological control with particular reference to potential impact on non-target organisms. CAB Rev. 2013, 8, 1–20. [Google Scholar] [CrossRef]
- Rostami, E.; Huang, D.L.; Shi, M.Z.; Zheng, L.Z.; Li, J.Y.; Madadi, H.; Fu, J.W. Functional response and predation rate of Cryptolaemus montrouzieri (Coleoptera: Coccinellidae) to Paracoccus marginatus (Hemiptera: Pseudococcidae) at different temperatures. J. Econ. Entomol. 2024, 117, 1406–1417. [Google Scholar] [CrossRef]
- Gordon, R.D. South American Coccinellidae (Coleoptera), Part VI: A systematic revision of the South American Diomini, new tribe (Scymninae). Ann. Zool. 1999, 49 (Suppl. 1), 1–219. [Google Scholar]
- Duverger, C. Un Scymninae africain de Guinée Conakry Diomus guilavoguii n. sp. (Coleoptera Coccinellidae) récolté sur Manihot esculenta Cralntz. Bull. Soc. Linn. Bordx. 1994, 22, 121–125. [Google Scholar]
- Zhuang, J.; Huo, L.Z.; Tang, M.J.; Xie, X.F.; Chen, X.S. First report of Diomus guilavoguii Duverger, 1994 (Coleoptera, Coccinellidae, Diomini) predating on papaya mealybug Paracoccus marginatus from China. Biodivers. Data J. 2023, 11, e113291. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, D.W.; Paterson, R.A.; Bovy, H.C.; Barrios-O’Neill, D. Frair: An R package for fitting and comparing consumer functional responses. Methods Ecol. Evol. 2017, 8, 1528–1534. [Google Scholar] [CrossRef]
- Juliano, S.A. Nonlinear curve fitting: Predation and functional response curves. In Design and Analysis of Ecological Experiments; Scheiner, S.M., Gurevitch, J., Eds.; Oxford University Press: New York, NY, USA, 2001; pp. 178–196. [Google Scholar] [CrossRef]
- Holling, C.S. Some characteristics of simple types of predation and parasitism. Can. Entomol. 1959, 91, 385–398. [Google Scholar] [CrossRef]
- Rogers, D. Random search and insect population models. J. Anim. Ecol. 1972, 41, 369–383. [Google Scholar] [CrossRef]
- Holling, C.S. The components of predation as revealed by a study of small-mammal predation of the European pine sawfly. Can. Entomol. 1959, 91, 293–320. [Google Scholar] [CrossRef]
- Jeschke, J.M.; Kopp, M.; Tollrian, R. Predator functional responses: Discriminating between handling and digesting prey. Ecol. Monogr. 2002, 72, 95–112. [Google Scholar] [CrossRef]
- Trexler, J.C.; McCulloch, C.E.; Travis, J. How can the functional reponse best be determined? Oecologia 1988, 76, 206–214. [Google Scholar] [CrossRef]
- Hassell, M.P. The Dynamics of Arthropod Predator-Prey Systems; Princeton University Press: Princeton, NJ, USA, 1978. [Google Scholar]
- Yu, W.; Liu, J. Predation and control of Harmonia axyridis against Tinocallis kahawaluokalani. J. Zhejiang For. Sci. Technol. 2019, 39, 55–59. [Google Scholar] [CrossRef]
- Hassell, M.P.; Varley, G.C. New inductive population model for insect parasites and its bearing on biological control. Nature 1969, 223, 1133–1137. [Google Scholar] [CrossRef]
- Wu, P.; Zhang, J.; Haseeb, M.; Yan, S.; Kanga, L.; Zhang, R. Functional responses and intraspecifc competition in the ladybird Harmonia axyridis (Coleoptera: Coccinellidae) provided with Melanaphis sacchari (Homoptera: Aphididae) as prey. Eur. J. Entomol. 2018, 115, 232–241. [Google Scholar] [CrossRef]
- Islam, Y.; Shah, F.M.; Güncan, A.; DeLong, J.P.; Zhou, X. Functional response of Harmonia axyridis to the larvae of Spodoptera litura: The combined effect of temperatures and prey instars. Front. Plant Sci. 2022, 13, 849574. [Google Scholar] [CrossRef]
- De Oliveira, S.J.; Nascimento, V.F.; De Lacerda, L.B.; De Souza, J.M.; Ramalho, D.G.; Izidro, Y.E.; De Bortoli, S.A. Predator-prey interaction between Xylocoris sordidus (Hemiptera: Anthocoridae) and Enneothrips enigmaticus (Thysanoptera: Thripidae). Neotrop. Entomol. 2024, 53, 391–399. [Google Scholar] [CrossRef]
- Liao, J.H.; He, H.F.; Zhang, J. Study on the predatory functional response of Arma chinensis (Fallou) to Henosepilachna vigintioctopunctata (Fabricius). J. Biosaf. 2024, 33, 177–181. [Google Scholar]
- Shi, L.L.; Li, Z.Y.; Lin, M.D.; Lu, Y.Y.; Chen, K.W. Predatory capability of Mallada basalis (Neuroptera: Chrysopidae) larvae on the eggs and early instar larvae of Spodoptera frugiperda (Lepidoptera: Noctuidae). Acta Entomol. Sin. 2022, 65, 1324–1333. [Google Scholar]
- Li, C.; Yu, J.; Mao, R.; Kang, K.; Xu, L.; Wu, M. Functional and Numerical Responses of Harmonia axyridis (Coleoptera: Coccinellidae) to Rhopalosiphum nymphaeae (Hemiptera: Aphididae) and Their Potential for Biological Control. Insects 2024, 15, 633. [Google Scholar] [CrossRef]
- Qiu, H.Y.; Fu, B.L.; Ma, X.T.; Li, M.J.; Xun, T.; Tang, L.D.; Liu, K. Functional response and searching efficiency of Orius tantillus against Thrips hawaiiensis. Plant Prot. 2020, 46, 84–89. [Google Scholar]
- Obrycki, J.J.; Kring, T.J. Predaceous Coccinellidae in Biological Control. Annu. Rev. Entomol. 1998, 43, 295–321. [Google Scholar] [CrossRef]
- van Lenteren, J.C. The State of Commercial Augmentative Biological Control: Plenty of Natural Enemies, but a Frustrating Lack of Uptake. BioControl 2012, 57, 1–20. [Google Scholar] [CrossRef]
- Mahzoum, A.M.; Villa, M.; Benhadi-Marín, J.; Pereira, J.A. Functional response of Chrysoperla carnea (Neuroptera: Chrysopidae) larvae on Saissetia oleae (Olivier) (Hemiptera: Coccidae): Implications for biological control. Agronomy 2020, 10, 1511. [Google Scholar] [CrossRef]
- Lester, P.J.; Harmsen, R. Functional and numerical responses do not always indicate the most effective predator for biological control: An analysis of two predators in a two-prey system. J. Appl. Ecol. 2002, 39, 455–468. [Google Scholar] [CrossRef]
- Benhadi-Marín, J.; Pereira, J.A.; Sousa, J.P.; Santos, S.A.P. Functional responses of three guilds of spiders: Comparing single- and multiprey approaches. Ann. Appl. Biol. 2019, 175, 202–214. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).