1. Introduction
The Bethylidae (Chrysidoidea) are a diverse and cosmopolitan family of aculeate wasps, with around 3000 extant species [
1] and more than 100 fossil species (see summary: [
2]; and other recent discoveries: [
3,
4,
5,
6,
7,
8]). As far as is known, all species for which the biology has been reported are parasitoids of lepidopteran and coleopteran larvae [
9]. Currently, nine subfamilies are recognized for the Bethylidae, i.e., the extant Bethylinae, Epyrinae, Mesitiinae, Pristocerinae, and Scleroderminae and the extinct Elektroepyrinae, Lancepyrinae, Protopristocerinae, and Holopsenellinae [
10]. The Holopsenellinae, however, are not considered a bethylid subfamily any longer since its type genus, i.e.,
Holopsenella, has been transferred into its own family (as Aculeata incertae sedis) [
11], leaving the remaining three genera formerly included in Holopsenellinae as orphans until the erection of a new subfamily for them, i.e., the Cretabythinae [
8]. The family dates back to the Lower Cretaceous [
12] and has a well-documented geological record with fossil representatives known for all but one subfamily, albeit with a larger proportion of fossils within the Epyrinae [
13].
The Epyrinae comprise 19 genera and about 1000 species [
14] and are considered the second most species-rich subfamily of Bethylidae, outnumbered only by the Pristocerinae [
1]. Epyrine representatives are common in upper Eocene Baltic and Rovno ambers, with 15 species recorded [
13], and two species are known in Miocene Dominican and Mexican ambers [
3]. The oldest epyrine wasps have been described from the lower Eocene amber of France [
15,
16]. However, while the Bethylidae are quite frequent in Upper Cretaceous ambers (although understudied; [
7,
13,
17,
18,
19], the Epyrinae were hitherto unknown from this period. The early Cenomanian amber from the Kachin State, Northern Myanmar, is one of the richest fossiliferous insect deposits in the world and a promising source to explore the diversity of fossil Bethylidae [
20]. The aim of the present study is to describe and illustrate the first Cretaceous Epyrinae preserved in a piece of mid-Cretaceous Kachin amber from Myanmar.
2. Materials and Methods
The study is based on a single specimen preserved in an amber piece that was collected from the deposit of Noije Bum, in the Hukawng Valley, Kachin State, Northern Myanmar [
21]. An Early Cenomanian age (98.79 ± 0.62 Ma) was suggested for Kachin amber, based on radiometric data of zircons from volcanic clasts found within the amber-bearing sediments [
22] and taphonomic analysis of some amber pieces [
23]. Some ammonites found in the amber-bearing bed and within amber corroborate a ate Albian–early Cenomanian age [
24,
25].
The amber piece is deposited in the amber collection of the Museum für Naturkunde, Berlin, Germany (MB.I.8638). It originates from the private collection of Jens-Wilhelm Janzen and was part of a collection of aculeate Hymenoptera kindly made available to one of us (V. Lohrmann) in 2013 and that was subsequently purchased by the Museum in 2018.
The amber piece has been ground and polished to optimize the observation of the specimen under different views, using wet silicon carbide papers on a grinder polisher (Buehler MetaServ 3000). Photographs were conducted with a Leica DMC4500 camera attached to a Leica M205C stereomicroscope. All images are digitally stacked photo-micrographic composites of several focal planes, which were obtained using Helicon Focus 6.7. Adobe Illustrator CC2019 and Photoshop CC2019 software were used to compose the figures and ImageJ 1.53 for measurements [
26]. The morphological examination of the specimen has been carried out using a Leica MZ APO and a Leica M205C stereomicroscope. The description of the characters largely follows the nomenclature of Lanes et al. [
27] with the exception of the forewing venation, which follows Azevedo et al. [
1]. Additionally, we prefer to use “metasomal segments” (with respective numbers) over “abdominal segments” and fore, mid, and hind legs over pro-, meso- and metalegs. The description of surface sculpturing follows Harris [
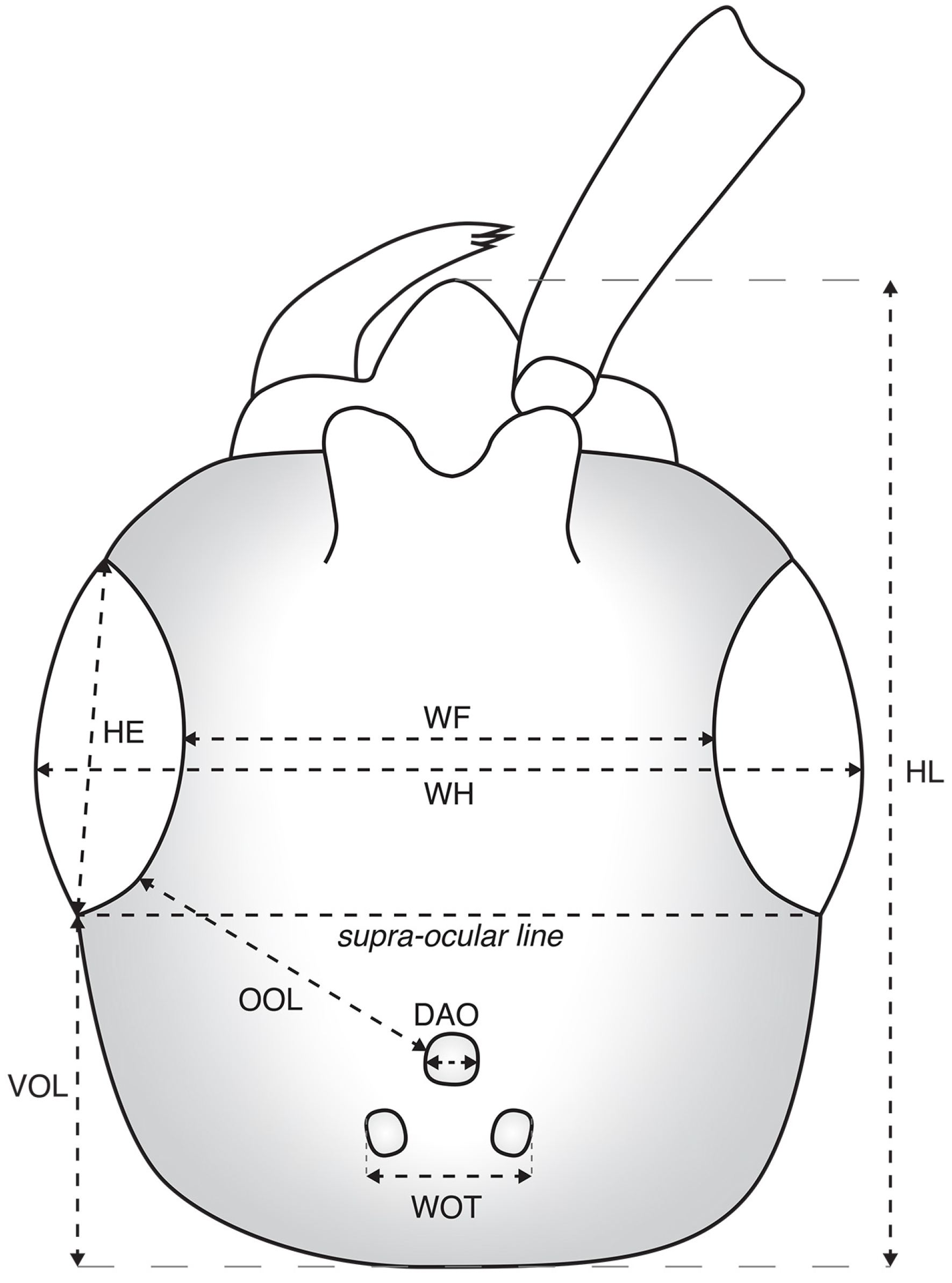
28]. Main measurements and indices used are as follows throughout the text and figures: LFW = length of forewing; LH = length of head, measured from vertex to apex of clypeus; WH = width of head above eyes; WF = width of frons; HE = height of eye; OOL = ocello-ocular line, the shortest distance between posterior margin of eye to the anterior margin of the anterior ocellus; WOT = width of ocellar triangle, the shortest distance between posterior ocelli; DAO = diameter of anterior ocellus; VOL = vertex-ocular line, the shortest distance between posterior margin of eye to the vertex crest (
Figure 1).
3. Systematic Paleontology
Superfamily Chrysidoidea Latreille, 1802
Family Bethylidae Haliday, 1839
Subfamily Epyrinae Kieffer, 1914
Genus Hukawngepyris gen. nov.
Etymology. The name is a combination of Hukawng, as a reference to the Hukawng Valley where Kachin amber is widely found, and Epyris, the type genus of the Epyrinae. Gender masculine.
Type species. Hukawngepyris setosus sp. nov., by monotypy.
Diagnosis. Females of the new genus stand out of all known bethylid genera by the following combination of characters: Vertex, gena, pronotum, mesopleuron, and mesoscutellum foveolate; head and anterior mesosoma with scattered long erect setae (
Figure 2 and
Figure 3A); compound eyes without setation, 0.6 × HL; malar space convergent, longer than VOL; median clypeal lobe triangular, projecting; mandible elongate, quadridentate; antenna filiform (
Figure 3B); scape and pedicel bearing long thick setae; pedicel shorter than each of the flagellomeres; anterior ocellus crossing supra-ocular line; distance between anterior and posterior ocelli shorter than width of ocellar triangle; occipital carina complete. Pronotum longer than wide, with anterior angle rounded; without longitudinal sulcus (
Figure 3B); notaulus and parapsidal signum present; sulcus of the mesoscuto-mesoscutellar suture present; mesoscutellum not contacting metapectal–propodeal complex (
Figure 3C); metapostnotal median carina complete, straight, extending on propodeal declivity (
Figure 4A). Forewing with C, Sc + R, M + Cu, A, Rs&M, cu-a, and 2r-rs&Rs tubular; 2r-rs&Rs tubular and curved, distally reaching anterior wing margin. Hind wing with three straight proximal and three hook-like distal hamuli (
Figure 2 and
Figure 4C). Protibial spur long and stout (
Figure 3C); tarsal claw bifid (
Figure 4B). Metasomal tergum 2 entirely smooth (
Figure 4B).
Hukawngepyris setosus sp. nov.
Material. Holotype number MB.I.8638, a nearly complete female, with right side of the first and second metasomal terga damaged and parts of the right midleg and right fore and hind wings missing.
Etymology. This species epithet refers to the conspicuous setation of the new species. The species epithet is to be treated as an adjective.
Diagnosis. As for genus.
Type locality and age. Noije Bum, Hukawng Valley, Kachin State, Northern Myanmar; late Albian–early Cenomanian, ca. 99 Ma, mid-Cretaceous.
Description. As for the genus diagnosis, with the following emendation: Body stout, length 4.39 mm, with some metallic reflections. Head rugulose; anteromesoscutum minutely rugulose; metasoma smooth. Head and anterior mesosoma with setae longer than the distance between neighboring setae.
Head prognathous, ovoid in lateral view, slightly longer than wide; LH: 0.58 mm, WH: 0.46 mm; WF: 0.34 mm, HE: 0.31 mm; OOL: 0.19 mm; WOT: 0.18 mm; DAO: 0.06 mm; VOL: 0.13 mm; frons convex in lateral view; compound eye elliptical, protruding, located laterally on head, covering 0.6 × LH; median clypeal lobe slightly projecting forward, lateral lobe reduced, median clypeal carina not particularly developed, not continuing on frons; mandible 3 × longer than basal width, 4 short teeth of similar size; palpal formula 6:3; antenna inserted close to median clypeal carina; scape cylindrical, length 0.24 mm, 1.7 × longer than pedicel; pedicel pyriform, length 0.14 mm, base narrower than apex, shorter than flagellomere 1; flagellum with 11 flagellomeres, flagellomeres elongate, cylindrical, distinctly longer than wide (length 0.10–0.17 mm; width 0.04–0.05 mm), no conspicuous setation; flagellomere 11 longest, tapering at apex; ocellar triangle posteriad on head.
Mesosoma shorter than metasoma, length 1.12 mm; propleuron barely visible in dorsal view; prosternum reduced; dorsal pronotal area slender (length 0.28 mm), without carina, lateral pronotal margins slightly incurved, posterior margin almost straight; anteromesoscutum slightly shorter than dorsal pronotal area (length 0.24 mm), notauli deeply impressed, converging posterad, posteriorly separated by width of sulcus of mesoscuto-mesoscutellar suture; sulcus of mesoscuto-mesoscutellar suture reniform; metanotum almost not visible; metapectal–propodeal complex wider than long, dorsally carinate, metapostnotal-propodeal suture present, short; propodeal declivity concave, long. Forewing developed (LFW: 1.62 mm), hyaline, homogeneously micro-pubescent; pterostigma elongate, 3.9 × longer than wide, narrow; 2r-rs&Rs originating from distal half of pterostigma and widely arched; costal [C], radial [R] and cubital [Cu] cells closed; 1Rs slightly angled at junction with 1M (both veins of similar length); 1M evenly incurved; 1Rs straight; no other tubular veins present but patterns of flexion lines visible in the distal section of the wing. Hind wing hyaline, homogeneously micro-pubescent, 1.01 mm long; Sc + R and A veins tubular, Sc + R reaching half the distance to distal hamuli, anal vein length about 0.25 × length of clavus of hind wing. Legs particularly pubescent, slender; tibial spurs formula 1–1–2; protibial spur with bifurcate apex; tarsal claws slightly curved, at least metatarsal claw with one subapical tooth.
Metasoma smooth, fusiform, length 1.23 mm (indicative), posterior segments partly retracted; six terga visible; petiole short; length of measurable terga: T1 = 0.24 mm; T2 = 0.35 mm; T3 = 0.15); metasomal sterna with few isolated erect bristles; apex of sting exserted.
4. Key to the Genera of Epyrinae (Modified from Colombo et al., [14])
1. Mesoscuto-mesoscutellar suture with foveae connected by sulcus…………….…….… 2
- Mesoscuto-mesoscutellar suture with foveae not connected by sulcus………………..... 10
2. Eye densely setose………….………………………………………………...……. Anisepyris
- Eye glabrous or at most slightly setose………………………….…………………………… 3
3. Dorsal pronotal area much wider than long………….…………………...……. †Gloxinius
- Dorsal pronotal area as long as or longer than wider…………………………………….... 4
4. Mesoscuto-mesoscutellar sulcus usually smooth, posterior margin incurved; clypeus with inconspicuous lateral lobes……………………………………………….………………. 5
- Mesoscuto-mesoscutellar sulcus slightly scrobiculate, posterior margin straight; clypeus with conspicuous lateral lobes………………………………………...……………………….. 8
5. 2r-rs&Rs vein curved towards and reaching anterior wing margin.……..†Hukawngepyris
- 2r-rs&Rs vein curved towards wing tip, not reaching margin or absent…………...….… 6
6. Body and wings with thick black setae……………………………………………… Laelius
- Body and wings with thin light castaneous setae………………………………………….. 7
7. Forewing with 2r-rs&Rs vein absent or faintly visible; protarsus and mesotibiae with spine-shaped setae (♀); genitalia with cuspis undivided (♂)……………………... Austrepyris
- Forewing with developed 2r-rs&Rs; protarsus and mesotibiae without spine-shaped setae (♀); genitalia with cuspis divided (♂)………….………………………………. Chlorepyris
8. Eye medium-sized, occupying at most 2/3 of side of head; median clypeal lobe as long as lateral ones or reduced; dorsal pronotal area usually carinate………..….……... Rysepyris
- Eye remarkably large, occupying almost all sides of the head; median clypeal lobe shorter than lateral ones; dorsal pronotal area usually ecarinate……………...…………………….. 9
9. Protarsus with long spatulate spine-shaped filament (♀); forewing with 2r-rs&Rs vein short; anteromesoscutum without foveolate transverse groove; posterior corner of metapectal-propodeal complex one-toothed……………………………………………. Disepyris
- Protarsus without spatulate spine-shaped filament (♀); forewing with 2r-rs&Rs vein long; anteromesoscutum with foveolate transverse groove; posterior corner of metapectal-propodeal complex two-toothed…………………………..…………………………. Holepyris
10. Distance between posterior ocelli much less than their own diameter……... Trissepyris
- Distance between posterior ocelli more than their own diameter………………………. 11
11. Dorsal pronotal area with wider anterior elevation…….……. Aspidepyris (type pattern)
- Dorsal pronotal area without such elevation………………………..……………………... 12
12. Dorsal pronotal area anterior margin incurved; humeral angle angulate…... Calyozina
- Dorsal pronotal area anterior margin straight or outcurved; humeral angle rounded... 13
13. Anterior area of the S2 with flap scale-shaped (♂♀); antenna usually pectinate; flagellomere 1 very reduced (♂)………………………….……………………………………. Calyoza
- Anterior area of the S2 without flap; antenna filiform; flagellomere 1 not reduced…... 14
14. Foveae of mesoscuto-mesoscutellar suture distant by less than their half own….….. 15
- Foveae of mesoscuto-mesoscutellar suture distant by more than their half own….…... 17
15. Metapectal-propodeal disc with indistinct metapostnotal carinae…………... Idatepyris
- Metapectal-propodeal disc with distinct metapostnotal carinae………………………... 16
16. Dorsal pronotal area usually carinate (♂♀); metapostnotal-propodeal carina sinuous (♀); apical aedeagal lobe with conspicuous projection ventrad (♂)………………. Bakeriella
- Dorsal pronotal area ecarinate (♂♀); metapostnotal-propodeal carina never sinuous (♀); apical aedeagal lobe without such projection (♂)……………………………………….. Epyris
17. Mesoscuto-mesoscutellar foveae distant by less than 4 × own width………………... 18
- Mesoscuto-mesoscutellar foveae distant by more than 4× own width…………………. 19
18. Mesoscuto-mesoscutellar fovea drop-shaped; cuspis undivided…………….. Psilepyris
- Mesoscuto-mesoscutellar fovea circular/oval-shaped; cuspis divided partially……. Dolus
19. Antennal scape with spine pecten-shaped; ventral margin of mandible with laminar expansion (♀); cuspis and harpe very long (♂)……………………………………. Muellerella
- Antennal scape without spine pecten-shaped; ventral margin of mandible without such expansion; cuspis and harpe short (♂)…………………………………………….…………. 20
20. Eye flat; notaulus faint (♂♀); dorsal mandibular tooth curved dorsad (♂)….. Gracilepyris
- Eye protuberant; notaulus well-impressed (♂♀); mandible with dorsal tooth mesad (♂) …………………………………………………………………… Aspidepyris (non-type pattern)
5. Discussion
Four of the nine subfamilies currently assigned to the Bethylidae are exclusively known from the fossil record but
Hukawngepyris gen. nov. does not fit in any of them. The new fossil can be distinguished from the Cretabythinae by displaying a reduced forewing venation with only three cells fully surrounded by tubular veins and the metanotum much obscured by the mesoscutellum [
29,
30]; from the Lancepyrinae by the absence of the forewing vein Rs + M, and the pronotum not anteriorly strongly narrowing [
8]; from the Protopristocerinae by the metanotum much obscured by the mesoscutellum [
30]; and from the Elektroepyrinae by the absence of the forewing vein 3Cu [
10,
15].
Of the five extant subfamilies, three have a fossil record that dates back to the mid-Cretaceous, i.e., the Pristocerinae [
31], the Scleroderminae [
32,
33], and the Bethylinae [
1,
34]. Whereas the first two are known from specimens included in Kachin amber (although Cockerell’s attribution of the Scleroderminae specimen to this subfamily is difficult to rely on [
33]), the Bethylinae are known from the Lower Cretaceous of Transbaikala (Russia). Females of the Pristocerinae are apterous, even in the fossil record, excluding this subfamily [
1,
31]. The Scleroderminae are generally characterized by a polished cuticle, a reduced forewing venation with C vein often absent (most complete venation found in Nothepyris Evans, 1973), the anterior wing margin angularly incurved, and the pedicel longer than the first flagellomere [
1,
35], precluding an assignment of our specimen to this subfamily. Our specimen displays some characteristics reminiscent of the Bethylinae, namely the 2r-rs&Rs vein reaching the anterior margin of the wing and the somewhat angled 1Rs&1M vein. However, it lacks the tubular Rs + M vein, the clypeal carina extending on the frons, and the angled tarsal claws, diagnostic for this subfamily [
1]. The only bethylid subfamily that has no documented fossil record at all are the Mesitiinae, members of which have a conspicuously long second metasomal segment that is longer than all subsequent segments. The metasoma of our specimen superficially resembles that of Mesitiinae as it is contracted, with the posterior segments retracted and not entirely visible, giving the illusion that the metasomal segment 2 is much longer than the subsequent segments. This contraction, however, may be due to a muscle contraction during its embedment in amber or death, so it is likely that the metasomal segment 2 is actually not longer than the posterior segments at all.
We attribute our specimen to the Epyrinae, as it shows more affinities with this subfamily than with any of the other bethylid subfamilies. The Epyrinae are challenging to define based on their morphology due to their large phenotypic plasticity [
36]. Alencar and Azevedo [
37] proposed a diagnosis for the subfamily they just re-established, later followed by Azevedo et al. [
1]. Among the diagnostic characters, our specimen displays the antennae with 11 flagellomeres, the 6:3 palpal formula, the clypeus divided in three lobes with the lateral lobe reduced (present in most genera but not all), the anteromesoscutum with notauli, the metapectal–propodeal complex dorsally carinate with metapostnotal median carina complete, the forewing developed with only three closed cells. These characters are more or less shared with other bethylid subfamilies but Alencar and Azevedo [
37] additionally identified three synapomorphies: the propleural epicoxal sulcus circular, the anterior area of the sternite 1 (=petiolar root) outcurved with medial emargination, and fused to the posterior area of the sternite 1 (=body root). In our specimen the propleural epicoxal sulcus is circular (
Figure 3C) rather than elongate [
37]. The configuration of the sternite 1 is difficult to assess on the specimen but based on the characters described above, it is likely that it belongs to the Epyrinae.
Within the Epyrinae, following the key of Colombo et al. [
14], our specimen keys in
Chlorepyris Kieffer, 1913 because of the following couplets: (1) mesoscuto-mesoscutellar suture with foveae connected by sulcus; (2) eye glabrous; (3) dorsal pronotal area distinctly longer than wide; (4) sulcus of the mesoscuto-mesoscutellar suture incurved, clypeus without lateral lobe; (5) body without thick black setae (see below for details on that character); (6) 2r-rs&Rs tubular, pigmented. However, it differs from that genus in having quadridentate mandibles. On the basis of the Colombo et al. key, the other epyrine genera are excluded as follows:
Trissepyris Kieffer, 1905,
Aspidepyris Evans, 1964,
Caliozyna Enderlein, 1912,
Calyoza Hope, 1837,
Idatepyris Colombo, Tribull and Azevedo, 2022,
Bakeriella Kieffer, 1910,
Epyris Westwood, 1832,
Psilepyris Kieffer, 1913,
Dolus Motschulsky, 1863,
Muellerella Saussure, 1892 and
Gracilepyris Colombo, Tribull and Azevedo, 2022 have the mesoscuto-mesoscutellar suture with foveae not connected by a sulcus (couplet 1);
Anysepyris Kieffer, 1905 has strongly setose eyes (couplet 2); the extinct
Gloxinius Colombo and Azevedo, 2021 has the dorsal pronotal area wider than long (couplet 3);
Rysepyris Kieffer, 1906,
Disepyris Kieffer, 1905 and
Holepyris Kieffer, 1904 have the sulcus of the mesoscuto-mesocutellar suture straight and a large lateral clypeal lobe (couplet 4); and
Austrepyris Colombo, Tribull and Azevedo, 2022 has the 2r-rs&Rs vein spectral (couplet 6).
Laelius Ashmead, 1893 is mainly characterized by possessing thick long black setae on the body and forewing [
38] and our specimen displays some long erect setae, superficially similar to that of the aforementioned genus. Actually,
Laelius have sparser and longer setae, sometimes being as long as HE, a shorter pronotum, and the 2r-rs&Rs vein projected straightly toward the wing tip (when present) (couplet 5). Therefore, we propose the new genus
Hukawngepyris gen. nov. to accommodate this specimen. This genus is composed of only one species,
Hukawngepyris setosus sp. nov.
Hukawngepyris setosus gen. et sp. nov. is particular within the Epyrinae for possessing the following combination of characters: long setae scattered on the body, the 2r-rs&Rs vein fully pigmented, curved towards the anterior wing margin, the 1Rs&1M vein angled, the antennae long, four toothed-mandibles, the mesoscuto-mesoscutellar sulcus present and the metapostnotum with median and lateral carinae present.
A conspicuous characteristic feature of
Hukawngepyris setosus gen. et sp. nov. is the configuration of the hamuli, with three hamuli grouped proximally and three hamuli grouped distally. The presence of three distal hamuli is common within the Bethylidae (e.g., Scleroderminae: [
35];
Laelius: [
39];
Afrobethylus Ramos and Azevedo, 2016: [
40]; Mesitiinae: [
41]). The presence of proximal hamuli, and moreover in a group of three, is less frequent [
42,
43], reported only for three species, one in the Mesitiinae, i.e.,
Hadesmesitius simplicitus (Barbosa and Azevedo, 2011), and two in the Pristocerinae, i.e.,
Apenesia sahyadrica Azevedo and Waichert, 2006 [
41] and
Pseudisobrachium somaliense Benoit, 1957 (M. Brazidec pers. obs.). All three species, however, have a different number of distal hamuli compared to
Hukawngepyris setosus gen. et sp. nov. Therefore, this unique 3:3 configuration of the hamuli is a good additional character to define
Hukawngepyris gen. nov. as a new genus. The number of hamuli present in
Trissepyris was briefly discussed, highlighting an asymmetry between both hind wings in the type species and a different number between the two species, leading to the conclusion that it is not a good character for this genus [
44]. However, the evolution of the number and position of hamuli in the Bethylidae is unclear and may require a broader study to understand their diagnostic value at the subfamilial and generic levels.
With 32 species, the Epyrinae are the most diverse Bethylidae subfamily in the fossil record, followed by the Bethylinae, mostly known from Cenozoic ambers [
45], and the Pristocerinae and the Scleroderminae, of which several occurrences have been reported throughout the Cretaceous [
7,
18,
31]. All but two fossils are attributed to the extant
Epyris,
Anisepyris,
Bakeriella,
Chlorepyris,
Holepyris, and
Laelius; most of them have been described from the upper Eocene Baltic amber [
13]. The subfossil Tanzanian copal, Miocene Dominican amber, Miocene Mexican amber, Oligocene Rott Formation, Eocene Bembridge marls, Eocene Rovno amber, Eocene Florissant Formation, and Eocene French amber yielded between one to five species [
3,
13,
16,
46,
47,
48,
49,
50]. So far, the only extinct epyrine genus was
Gloxinius, with one species from Baltic amber [
51], making
Hukawngepyris setosus gen. et sp. nov. the second species belonging to a fossil genus. The description of the new species also results in a 47-million-year gap in the Epyrinae fossil record, between the early Cenomanian and early Eocene. Therefore, additional specimens may be discovered after investigation of Upper Cretaceous deposits such as amber from France, Russia, or Canada.
Additionally, this discovery emphasizes a little more on the diversity of the Bethylidae during the mid-Cretaceous.
Hukawngepyris setosus gen. et sp. nov. represents the earliest known Epyrinae, which is now the sixth bethylid subfamily to be recorded from mid-Cretaceous Burmese ambers (from Hkamti and Kachin), after the Cretabythinae [
30], the Protopristocerinae [
30], the Lancepyrinae [
8], the Pristocerinae [
31] and the Scleroderminae [
32]. The Elektroepyrinae are known only from Eocene French amber [
10] and the Mesitiinae have no fossil record at all. Considering the Epyrinae diversity and their abundance in Cenozoic ambers (most notably from the Eocene), it was surprising that the family had not yet been recorded from the Cretaceous. The discovery of
Hukawngepyris setosus gen. et sp. nov. pushes back the estimated divergence times of most of the bethylid subfamilies to the Lower Cretaceous and it could be interesting to include this species in a broader phylogenetic context to see how it affects the tree topologies.