Possible Regulation of Larval Juvenile Hormone Titers in Bombyx mori by BmFAMeT6
Abstract
Simple Summary
Abstract
1. Introduction
1.1. Synthesis Pathway of Juvenile Hormone
1.2. Role of FAMeT in Juvenile Hormone Synthesis
2. Materials and Methods
2.1. Experimental Insects and Feeding
2.2. Fluorescence Screening of Transgenic Silkworm and Insertion Site Detection of Overexpressed Genes
2.3. Molecular Detection of Transgenic Bombyx Mori
2.4. Detection of Farnesoic Acid (FA), Methyl Farnesoate (MF), Juvenile Hormone I and Ⅱ (JH Ⅰ and JH Ⅱ)
2.5. Construction of the Recombinant Expression Vector
2.6. Injection of Exogenous Farnesoic Acid
2.7. Data Processing
3. Results
3.1. Insertion Sites of Overexpressed Genes
3.2. Identification of the Overexpression Silkworm Strain
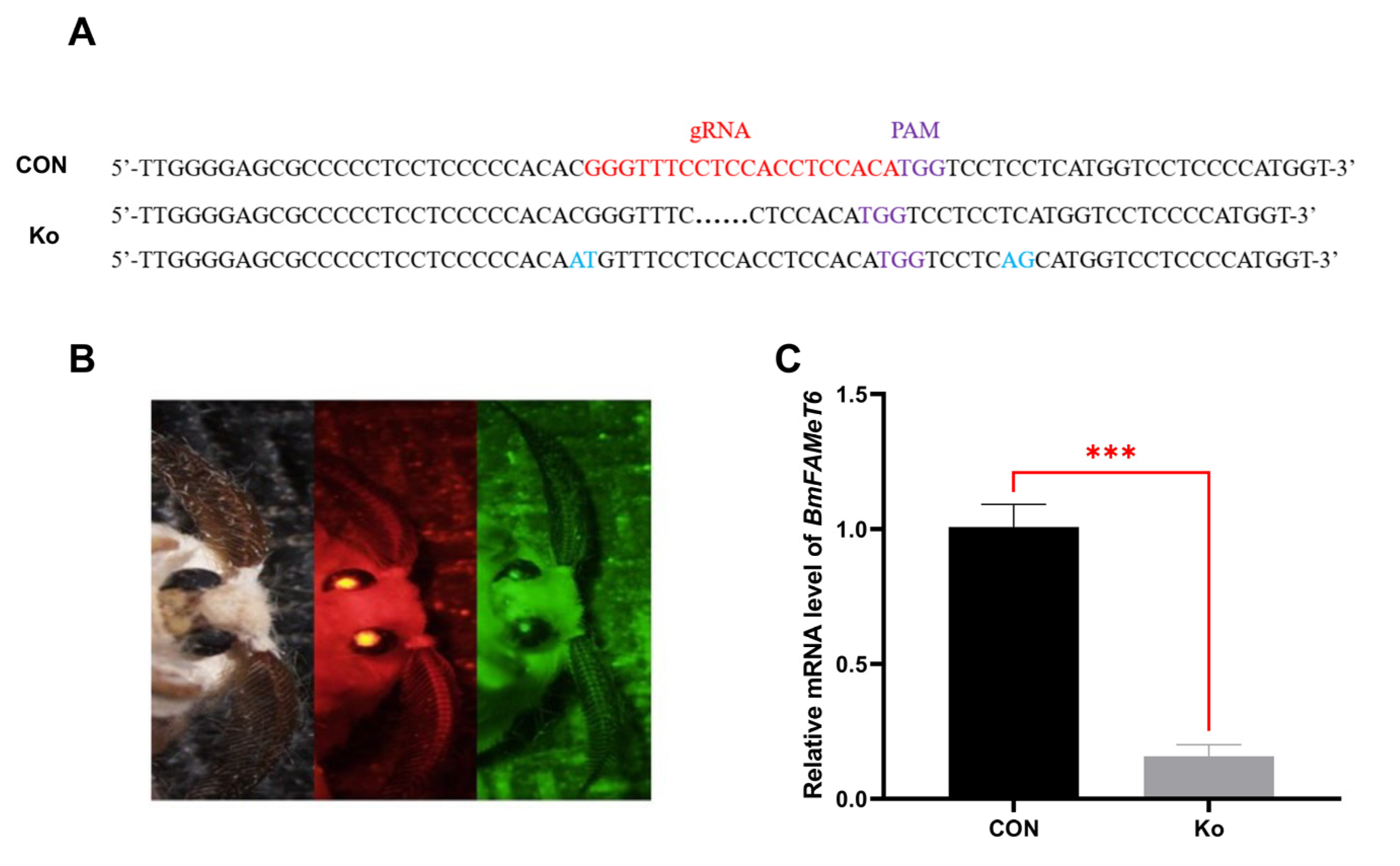
3.3. Construction and Verification of the Knockout Silkworm Strain
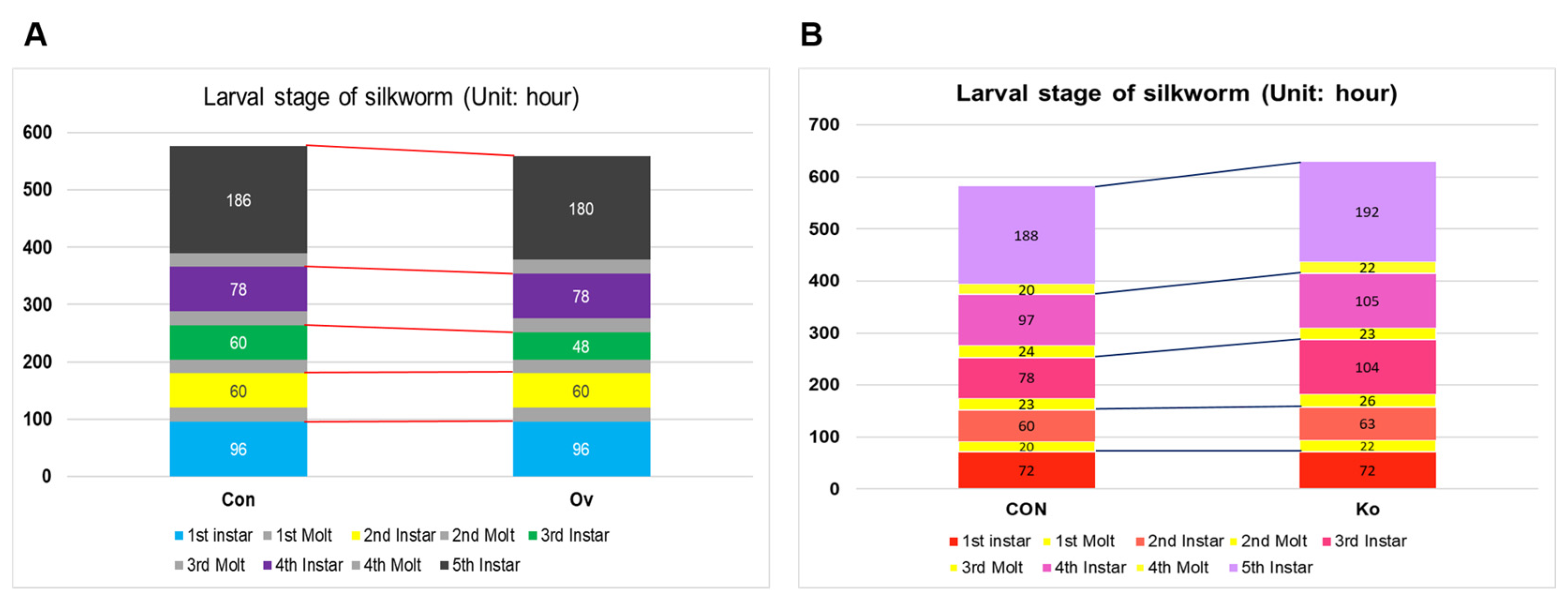
3.4. Observation of Phenotype in Transgenic Individuals
3.5. Analysis of FA and JH Related Indexes in Transgenic Individuals
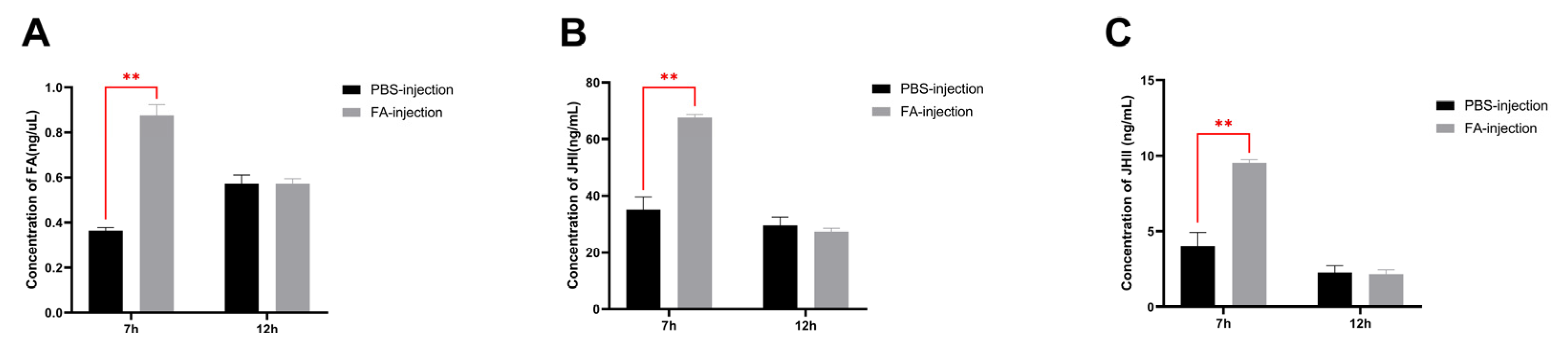
3.6. Effect of Exogenous Farnesoic Acid on JH Titer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Daimon, T.; Uchibori, M.; Nakao, H.; Sezutsu, H.; Shinoda, T. Knockout silkworms reveal a dispensable role for juvenile hormones in holometabolous life cycle. Proc. Natl. Acad. Sci. USA 2015, 112, E4226–E4235. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, G.R.; Davey, K.G. Cellular and Molecular Actions of Juvenile Hormone. II. Roles of Juvenile Hormone in Adult Insects. ADV Insect Physiol. 1996, 26, 1–155. [Google Scholar]
- Chieka, M.; Riddiford, L.M. Insect juvenile hormone action as a potential target of pest management. J. Pest. 2006, 31, 77–84. [Google Scholar]
- Daimon, T.; Shinoda, T. Function, diversity, and application of insect juvenile hormone epoxidases (CYP15). Biotechnol. Appl. Biochem. 2013, 60, 82–91. [Google Scholar] [CrossRef]
- Bellés, X.; Martín, D.; Piulachs, M.D. The mevalonate pathway and the synthesis of juvenile hormone in insects. Annu. Rev. Entomol. 2005, 50, 181–199. [Google Scholar] [CrossRef]
- Goodman, W.G.; Cusson, M. The Juvenile Hormones. Insect Endocrinology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 310–365. [Google Scholar]
- Cheng, D.; Meng, M.; Peng, J.; Qian, W.; Kang, L.; Xia, Q. Genome-wide comparison of genes involved in the biosynthesis, metabolism, and signaling of juvenile hormone between silkworm and other insects. Genet. Mol. Biol. 2014, 37, 444–459. [Google Scholar] [CrossRef]
- Hammock, B.D. NADPH dependent epoxidation of methyl farnesoate to juvenile hormone in the cockroach Blaberus giganteus L. Life Sci. 1975, 17, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, C.V.; Maestro, J.L. Expression of juvenile hormone acid O-methyltransferase and juvenile hormone synthesis in Blattella germanica. Insect Sci. 2018, 25, 787–796. [Google Scholar] [CrossRef]
- Guo, P.; Zhang, Y.; Zhang, L.; Xu, H.; Zhang, H.; Wang, Z.; Jiang, Y.; Molloy, D.; Zhao, P.; Xia, Q. Structural basis for juvenile hormone biosynthesis by the juvenile hormone acid methyltransferase. J. Biol. Chem. 2021, 297, 101234. [Google Scholar] [CrossRef]
- Minakuchi, C.; Namiki, T.; Yoshiyama, M.; Shinoda, T. RNAi-mediated knockdown of juvenile hormone acid O-methyltransferase gene causes precocious metamorphosis in the red flour beetle Tribolium castaneum. FEBS J. 2008, 275, 2919–2931. [Google Scholar] [CrossRef]
- Nouzova, M.; Edwards, M.J.; Michalkova, V.; Ramirez, C.E.; Ruiz, M.; Areiza, M.; DeGennaro, M.; Fernandez-Lima, F.; Feyereisen, R.; Jindra, M.; et al. Epoxidation of juvenile hormone was a key innovation improving insect reproductive fitness. Proc. Natl. Acad. Sci. USA 2021, 118, e2109381118. [Google Scholar] [CrossRef]
- Yang, Z.M.; Wu, Y.; Li, F.F.; Zhou, Z.J.; Yu, N.; Liu, Z.W. Genomic Identification and Functional Analysis of JHAMTs in the Pond Wolf Spider, Pardosa pseudoannulata. Int. J. Mol. Sci. 2021, 22, 11721. [Google Scholar] [CrossRef]
- Silva Gunawardene, Y.I.; Chow, B.K.; He, J.G.; Chan, S.M. The shrimp FAMeT cDNA is encoded for a putative enzyme involved in the methylfarnesoate (MF) biosynthetic pathway and is temporally expressed in the eyestalk of different sexes. Insect Biochem. Mol. Biol. 2001, 31, 1115–1124. [Google Scholar] [CrossRef]
- Xie, X.; Zhu, D.; Li, Y.; Qiu, X.; Cui, X.; Tang, J. Hemolymph Levels of Methyl Farnesoate During Ovarian Development of the Swimming Crab Portunus trituberculatus, and Its Relation to Transcript Levels of HMG-CoA Reductase and Farnesoic Acid O-Methyltransferase. Biol. Bull. 2015, 228, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Burtenshaw, S.M.; Su, P.P.; Zhang, J.R.; Tobe, S.S.; Dayton, L.; Bendena, W.G. A putative farnesoic acid O-methyltransferase (FAMeT) orthologue in Drosophila melanogaster (CG10527): Relationship to juvenile hormone biosynthesis? Peptides 2008, 29, 242–251. [Google Scholar] [CrossRef]
- Zhang, H.; Tian, L.; Tobe, S.; Xiong, Y.; Wang, S.; Lin, X.; Liu, Y.; Bendena, W.; Li, S.; Zhang, Y.Q. Drosophila CG10527 mutants are resistant to juvenile hormone and its analog methoprene. Biochem. Biophys. Res. Commun. 2010, 401, 182–187. [Google Scholar] [CrossRef]
- Vannini, L.; Ciolfi, S.; Spinsanti, G.; Panti, C.; Frati, F.; Dallai, R. The putative-farnesoic acid O-methyl transferase (FAMeT) gene of Ceratitis capitata: Characterization and pre-imaginal life expression. Arch. Insect Biochem. Physiol. 2010, 73, 106–117. [Google Scholar] [PubMed]
- Vieira, C.U.; Bonetti, A.M.; Simões, Z.L.; Maranhão, A.Q.; Costa, C.S.; Costa, M.C.; Siquieroli, A.C.; Nunes, F.M. Farnesoic acid O-methyl transferase (FAMeT) isoforms: Conserved traits and gene expression patterns related to caste differentiation in the stingless bee, Melipona scutellaris. Arch. Insect Biochem. Physiol. 2008, 67, 97–106. [Google Scholar] [CrossRef]
- Minna, J.; Yun, Y.; Bing, S.; Bo, W.; Xinda, L. Expression of FAMeT and JHE Influenced by Kruppel homolog-1 Gene from Brown Planthopper. Plant Dis. Pests 2013, 4, 23. [Google Scholar]
- Meng, M. Identification and Expression Profiling of Farnesoic Acid O-methyltrans-ferase Gene in Silkworm, Bombyx mori. Sci. Seric. 2013, 39, 680–688. [Google Scholar]
- Zhang, L.; Li, X.Z.; Li, T.; Xiong, R.; Li, Y.; Yan, D.S.; Chen, P. Farnesoic acid methyltransferase 6 (BmFAMeT6) interrelates with moltinism of dominant trimolter in silkworm, Bombyx mori. Biologia 2021, 76, 2231–2240. [Google Scholar] [CrossRef]
- Ma, L.; Xu, H.; Zhu, J.; Ma, S.; Liu, Y.; Jiang, R.J.; Xia, Q.; Li, S. Ras1(CA) overexpression in the posterior silk gland improves silk yield. Cell Res. 2011, 21, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Hu, H.; Tong, X.; Han, M.; Gai, T.; Lou, J.; Yan, Z.; Xiong, G.; Lu, C.; Dai, F. Mutation of a lepidopteran-specific PMP-like protein, BmLSPMP-like, induces a stick body shape in silkworm, Bombyx mori. Pest. Manag. Sci. 2022, 78, 5334–5346. [Google Scholar] [CrossRef] [PubMed]
- Giniger, E.; Varnum, S.M.; Ptashne, M. Specific DNA binding of GAL4, a positive regulatory protein of yeast. Cell 1985, 40, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Imamura, M.; Nakai, J.; Inoue, S.; Quan, G.X.; Kanda, T.; Tamura, T. Targeted gene expression using the GAL4/UAS system in the silkworm Bombyx mori. Genetics 2003, 165, 1329–1340. [Google Scholar] [CrossRef]
- Duffy, J.B. GAL4 system in Drosophila: A fly geneticist’s Swiss army knife. Genesis 2002, 34, 1–15. [Google Scholar] [CrossRef]
- Li, J.J.; Shi, Y.; Wu, J.N.; Li, H.; Smagghe, G.; Liu, T.X. CRISPR/Cas9 in lepidopteran insects: Progress, application and prospects. J. Insect Physiol. 2021, 135, 104325. [Google Scholar] [CrossRef]
- Zhang, T.; Song, W.; Li, Z.; Qian, W.; Wei, L.; Yang, Y.; Wang, W.; Zhou, X.; Meng, M.; Peng, J.; et al. Krüppel homolog 1 represses insect ecdysone biosynthesis by directly inhibiting the transcription of steroidogenic enzymes. Proc. Natl. Acad. Sci. USA 2018, 115, 3960–3965. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Sui, Y.; Xu, J.; Zhu, F.; Palli, S.R. Juvenile hormone regulates Aedes aegypti Krüppel homolog 1 through a conserved E box motif. Insect Biochem. Mol. Biol. 2014, 52, 23–32. [Google Scholar] [CrossRef]
- Shinoda, T.; Itoyama, K. Juvenile hormone acid methyltransferase: A key regulatory enzyme for insect metamorphosis. Proc. Natl. Acad. Sci. USA 2003, 100, 11986–11991. [Google Scholar] [CrossRef]
- Li, K.; Jia, Q.Q.; Li, S. Juvenile hormone signaling—A mini review. Insect Sci. 2019, 26, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Furuta, K.; Ichikawa, A.; Murata, M.; Kuwano, E.; Shinoda, T.; Shiotsuki, T. Determination by LC-MS of juvenile hormone titers in hemolymph of the silkworm, Bombyx mori. Biosci. Biotechnol. Biochem. 2013, 77, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Minakuchi, C.; Ishii, F.; Washidu, Y.; Ichikawa, A.; Tanaka, T.; Miura, K.; Shinoda, T. Expressional and functional analysis of CYP15A1, a juvenile hormone epoxidase, in the red flour beetle Tribolium castaneum. J. Insect Physiol. 2015, 80, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Hui, J.H.; Hayward, A.; Bendena, W.G.; Takahashi, T.; Tobe, S.S. Evolution and functional divergence of enzymes involved in sesquiterpenoid hormone biosynthesis in crustaceans and insects. Peptides 2010, 31, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, H.; Zhang, Y.; Zhang, H.; Wang, Z.; Guo, P.; Zhao, P. Structural characterization and functional analysis of juvenile hormone acid methyltransferase JHAMT3 from the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2022, 151, 103863. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Li, T.; Guo, M.; Xiong, R.; Yan, D.; Chen, P. Possible Regulation of Larval Juvenile Hormone Titers in Bombyx mori by BmFAMeT6. Insects 2023, 14, 644. https://doi.org/10.3390/insects14070644
Yu Y, Li T, Guo M, Xiong R, Yan D, Chen P. Possible Regulation of Larval Juvenile Hormone Titers in Bombyx mori by BmFAMeT6. Insects. 2023; 14(7):644. https://doi.org/10.3390/insects14070644
Chicago/Turabian StyleYu, Yang, Tian Li, Meiwei Guo, Rong Xiong, Dongshen Yan, and Ping Chen. 2023. "Possible Regulation of Larval Juvenile Hormone Titers in Bombyx mori by BmFAMeT6" Insects 14, no. 7: 644. https://doi.org/10.3390/insects14070644
APA StyleYu, Y., Li, T., Guo, M., Xiong, R., Yan, D., & Chen, P. (2023). Possible Regulation of Larval Juvenile Hormone Titers in Bombyx mori by BmFAMeT6. Insects, 14(7), 644. https://doi.org/10.3390/insects14070644





