Effect of Ageing in the Mating Behaviour Sequence of Osmia cornuta Latr. (Hymenoptera: Megachilidae)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Biological Samples
2.2. Behavioural Study
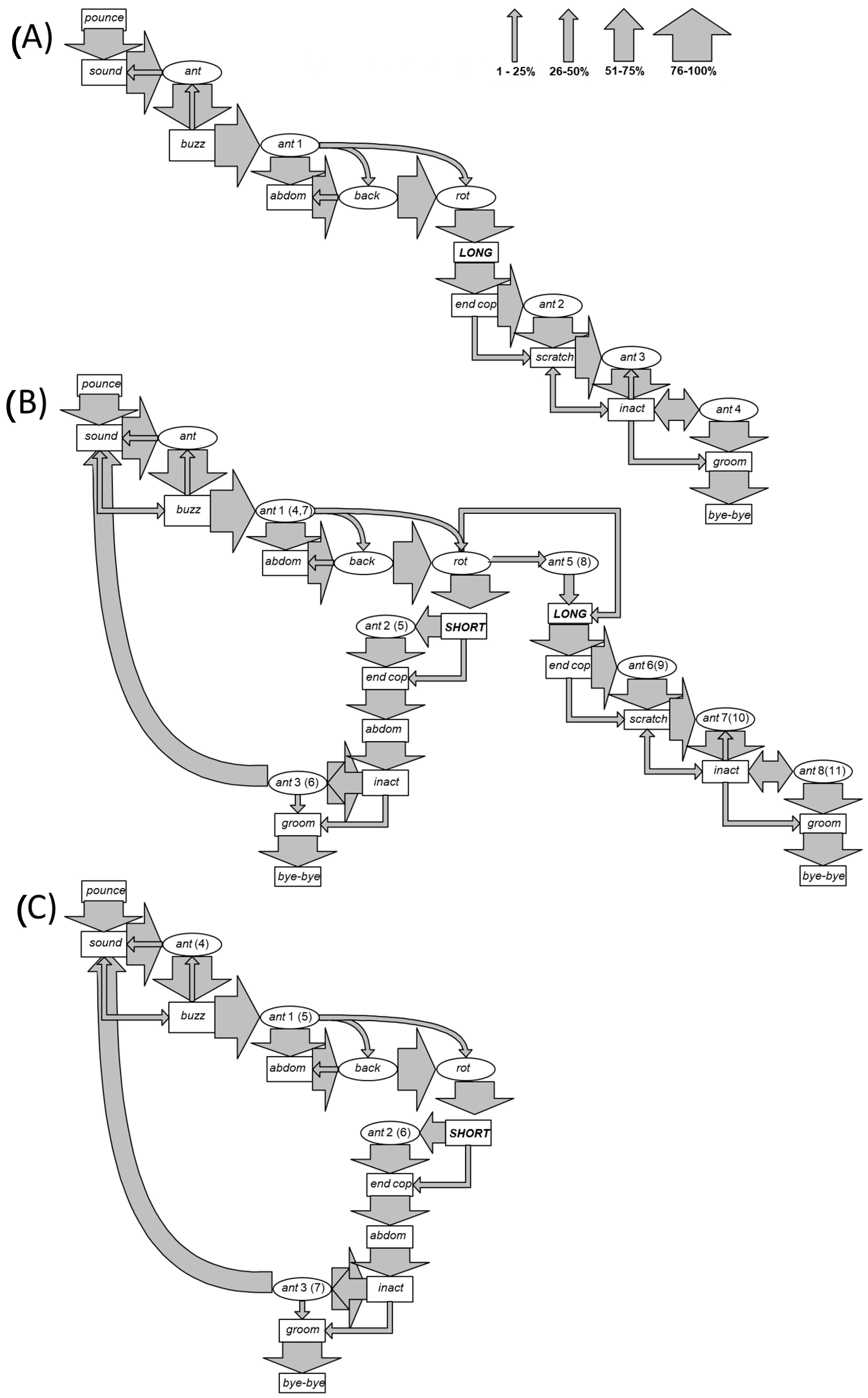
3. Results
- Pounce (pounce): the male flies and rapidly climbs onto the back of the female, encircling the lateroventral portion of her metasoma with both his mesothoracic legs. Duration: from 1 to 2 s.
- Sound (sound): a rhythmic sound is emitted by an oscillation of the anterior legs simultaneously with a slight movement and beating of the wings. Duration: from 30 to 35 s.
- Antenna motion (ant): downward movement of the antennae almost touching the female’s eyes, at first alternating and then simultaneous. Duration: from 30 to 50 s.
- Buzz (buzz): continuous emission of sound obtained by wings vibration. Duration: 2 s.
- Abdomen (abdom): the abdomen is first stretched upward and then toward the ventral part of the female’s abdomen. Duration: from 3 to 28 s.
- Antennae backward (back): the antennae are held forward and with a marked downwards curvature of the distal part of antenna for a duration of 1/24 s, then suddenly return to the starting position. Duration: from 40 s to 1 min.
- Antenna rotation (rot): rapid rotating movement of the antennae in front of the eyes of the female. Duration: from 1 to 2 min.
- Copulations:
- Long copulation (long): insertion of the male copulatory organ into the female genital aperture. This copula has duration of 1 or 2 min, during which the male lightly beats his abdomen on the female abdomen.
- Short copula (short): insertion of the male copulatory organ into the female genital aperture, for a duration of only few seconds.
- End of copulation (end cop): the male withdraws his aedeagus from the female genital aperture.
- Scratching (scratch): this behavioural unit is expressed by the male only following a long copulation, and it comprises three closely connected actions performed in sequence. Duration: from 2 to 6 min.
- Simultaneous downward motion of the antennae, which then becomes an alternating movement in an upward direction or an alternating motion both downward and upward.
- The insect shows the “ant back” movement concomitantly with the emission of a sharp and brief sound and a series of tapping motions performed by curving his abdomen on the urotergal portion of the female.
- Simultaneous contraction of the meso- and metathoracic legs of the male on the female’s abdomen.
- Inactivity (inact): the male remains immobile on the back of the female. Duration: from 10 to 20 min.
- Self-grooming (groom): rubbing of the head, the antennae, and the lower part of the thorax using the anterior legs and rubbing of the abdomen by means of the posterior legs, which, in turn, clean each other. Duration: from 2 to 3 min.
- End of mating (bye-bye): the male moves away from the female and this action signals the end of mating.
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cobelli, R. Note Biologiche Sugli Apidi Chalicodoma Muraria L., Chalicodoma Lefebvrei Gerst., Osmia Cornuta Latr., Xilocopa Violacea Poda; Museo Civico: Rovereto, Italia, 1888. [Google Scholar]
- Peters, D. Systemattik und Zoogeographie der west—Palaearktischen Arten von Osmia Penzer 1806 s. str., Monosmia Tkalcu 1974 und Orientosmia n. subgen. (Insecta: Hymenoptera: Megachilidae). Senckenberg. Biol. 1977, 58, 287–346. [Google Scholar]
- Bosch, J. Osmia cornuta Latr. (Hym., Megachilidae) as a potential pollinator in almond orchards. J. Appl. Entomol. 1994, 117, 151–157. [Google Scholar] [CrossRef]
- Bosch, J. The nesting behavior of the mason bee Osmia cornuta Latr. with special reference to its pollinating potential (Hymenoptera, Megachilidae). Apidologie 1994, 25, 84–93. [Google Scholar] [CrossRef]
- Bosch, J. Improvement of field management of Osmia cornuta Latr. (Hymenoptera, Megachilidae) to pollinate almond. Apidologie 1994, 25, 71–83. [Google Scholar] [CrossRef]
- Bosch, J.; Blas, M. Effect of over-wintering and incubation temperatures on adult emergence in Osmia cornuta Latr. (Hymenoptera, Megachilidae). Apidologie 1994, 25, 265–277. [Google Scholar] [CrossRef]
- Krunic, M.; Stanisavljevic, L.; Pinzauti, M.; Felicioli, A. The accompanying fauna of Osmia cornuta and Osmia rufa and effective measures of protection. Bull. Insectol. 2005, 58, 141–152. [Google Scholar]
- Felicioli, A.; Pinzauti, M. Imenotteri del genere Osmia (Hymenoptera: Megachilidae) come potenziali impollinatori di piante di interesse agrario e forestale. In Apicoltura Moderna, 1st ed.; Regione Toscana: Firenze, Italy, 1994; pp. 116–136. [Google Scholar]
- Rossi, A.; Felicioli, A.; Pinzauti, M. Analisi etologica dell’accoppiamento in Osmia cornuta Latr. (Hymenoptera: Megachilidae). In Proceedings of the 17th Convegno Della Società Italiana Di Etologia, Pisa, Italia, 3–5 May 1996. [Google Scholar]
- Seidelmann, K. Double insurance of paternity by a novel type of mating plug in a monandrous solitary mason bee Osmia bicornis (Hymenoptera: Megachilidae). Biol. J. Linn. Soc. 2015, 115, 28–37. [Google Scholar] [CrossRef]
- Banaszak, J.; Romasenko, L. Megachilid Bees of Europe (Hymenoptera, Apoidea, Megachilidae), 1st ed.; Pedagogical University of Bydgoszcz: Bydgoszcz, Poland, 1998; pp. 192–213. [Google Scholar]
- Tasei, J. Le comportement de nidification chez Osmia (Osmia) cornuta Latr. et Osmia (Osmia) rufa L. (Hymenoptera Megachilidae). Apidologie 1973, 4, 195–225. [Google Scholar] [CrossRef]
- Tasei, J. Observations sur le developpement d’Osmia cornuta Latr. et Osmia rufa L. (Hymenoptera Megachilidae). Apidologie 1973, 4, 295–315. [Google Scholar] [CrossRef]
- Fliszkiewicz, M.; Giejdasz, K.; Wilkaniec, Z. The effect of diapause termination time on the successful insemination of Osmia rufa L. females. J. Apic. Sci. 2011, 55, 79–84. [Google Scholar]
- Wasielewski, O.; Wojciechowicz, T.; Giejdasz, K.; Krishnan, N. Influence of methoprene and temperature on diapause termination in adult females of the over-wintering solitary bee, Osmia rufa L. J. Insect Physiol. 2011, 57, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Felicioli, A.; Rossi, A.; Pinzauti, M. Nest-site preference and gregarious nesting in Osmia cornuta Latr. (Hymenoptera, Megachilidae). Frustula Entomol. 1996, 19, 132–138. [Google Scholar]
- Felicioli, A.; Ambroselli, S.; Pinzauti, M. Preliminary observations on usurping behaviour in Osmia cornuta Latr. (Hymenoptera, Megachilidae). Ins. Soc. Life 2000, 3, 101–105. [Google Scholar]
- Pinzauti, M.; Lazzarini, D.; Felicioli, A. Preliminary investigation of Osmia cornuta Latr. (Hymenoptera, Megachilidae) as a potential pollinator for blackberry (Rubus fruticosus L.) under confined environment. Acta Hortic. 1997, 437, 329–333. [Google Scholar] [CrossRef]
- Torchio, P.; Asensio, E. The indroduction of the European bee, Osmia cornuta Latr., into the US as a potential pollinator of orchard crops, and a comparison of its manageability with Osmia lignaria propinqua Cresson (Hymenoptera, Megachilidae). J. Kans. Entomol. Soc. 1985, 58, 42–52. [Google Scholar]
- Marquez, J.; Bosch, J.; Vicens, N. Pollens collected by wild and managed populations of the potential orchard pollinator Osmia cornuta Latr. (Hymenoptera, Megachilidae). J. Appl. Entomol. 1994, 117, 353–359. [Google Scholar] [CrossRef]
- Torchio, P.; Asensio, E.; Thorp, R. Introduction of the European Bee, Osmia cornuta, into California Almond Orchards (Hymenoptera, Megachilidae). Environ. Entomol. 1987, 16, 664–667. [Google Scholar] [CrossRef]
- Felicioli, A.; Krunic, M.; Pinzauti, M. Solitary bees conservation, rearing and management for pollination. A contribution to the International Workshop on Solitary Bees and Their Role in Pollination, Held in Beberibe, Ceará, Brazil, in April 2004. In Rearing and Using Osmia Bees for Crop Pollination: A Help from a Molecular Approach; Freitas, B.M., Pereira, J.O.P., Eds.; Imprensa Universitária: Maputo, República de Moçambique, 2004; pp. 161–174. [Google Scholar]
- Pekár, J.; Reiff, M.; Brezina, I. Location problem of Osmia cornuta nesting aids for optimum pollination. PLoS ONE 2020, 15, e0244610. [Google Scholar] [CrossRef]
- Belien, T.; Raymaekers, S.; Eeraerts, M.; Mommaerts, V.; Claus, G.; Bogen, C.; Piot, N.; Smagghe, G.; Spanoghe, P.; Bylemans, D. Towards integrated pest and pollinator management in intensive pear cultivation: A case study from Belgium. Insects 2021, 12, 901. [Google Scholar] [CrossRef]
- Laduerner, E.; Maccagnani, B.; Tesoriero, D.; Nepi, M.; Felicioli, A. Laboratory rearing of Osmia cornuta Latreille (Hymenoptera Megachilidae) on artificial diet. Boll. Ist. Entomol. G. Grandi Univ. Bologna 1999, 53, 133–146. [Google Scholar]
- Steen, J.V.D. Indoor rearing of the solitary bee Osmia rufa. Proc. Exp. Appl. Entomol. 1997, 8, 81–84. [Google Scholar]
- Krunić, M.D.; Stanisavljević, L.Ž. Supercooling points and diapause termination in overwintering adults of orchard bees Osmia cornuta and O. rufa (Hymenoptera: Megachilidae). Bull. Entomol. Res. 2006, 96, 323–326. [Google Scholar] [CrossRef]
- Yan, Z.; Wang, L.; Reddy, G.V.; Gu, S.; Men, X.; Xiao, Y.; Su, J.; Ge, F.; Ouyang, F. The Supercooling Responses of the Solitary Bee Osmia excavata (Hymenoptera: Megachilidae) under the Biological Stress of Its Brood Parasite, Sapyga coma (Hymenoptera: Sapygidae). Insects 2022, 13, 235. [Google Scholar] [CrossRef] [PubMed]
- Kemp, W.; Bosch, J. Development and emergence of the alfalfa pollinator Megachile rotundata (Hymenoptera: Megachilidae). Ann. Entomol. Soc. Am. 2000, 93, 904–911. [Google Scholar] [CrossRef]
- Krunic, M.; Pinzauti, M.; Felicioli, A.; Stanisavljevic, L.J. Further observations on Osmia cornuta Latr. and Osmia rufa L. as alternative fruit pollinators; Domestication and Utilization. Arch. Biol. Sci. 1995, 47, 59–66. [Google Scholar]
- Sgolastra, F.; Kemp, W.P.; Maini, S.; Bosh, J. Duration of prepupal summer dormancy regulates synchronization of adult diapause with winter temperatures in bees of the genus Osmia. J. Insect Physiol. 2012, 58, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Alcock, J. Natural selection and the mating systems of the solitary bees. Am. Sci. 1980, 68, 146–153. [Google Scholar]
- Alcock, J.; Barrows, E.; Gordh, G.; Hubbard, L.J.; Kirkendall, L.; Pyle, D.W.; Ponder, T.L.; Zalom, F.G. The ecology and evolution of male reproductive behaviour in the bees and wasps. Zool. J. Linn. Soc. 1978, 64, 293–326. [Google Scholar] [CrossRef]
- Paxton, R.; Giovanetti, M.; Andrietti, F.; Scamoni, E.; Scanni, B. Mating in a communal bee, Andrena agilissima (Hymenoptera Andrenidae). Ethol. Ecol. Evol. 1999, 11, 371–382. [Google Scholar] [CrossRef]
- Paxton, R.J. Male mating behaviour and mating systems of bees: An overview. Apidologie 2005, 36, 145–156. [Google Scholar] [CrossRef]
- Raw, A. The behaviour of males of the solitary bee Osmia rufa (Megachilidae) searching for females. Behaviour 1976, 56, 3–4. [Google Scholar] [CrossRef]
- Seidelmann, K. The race of females: The mating system of the red mason bee, Osmia rufa (L.) (Hymenoptera: Megachilidae). J. Insect Behav. 1999, 12, 13–25. [Google Scholar] [CrossRef]
- Shimron, O.; Hefetz, A. Mating behavior and sex attraction of Eucera palestinae Friese (Hymenoptera: Anthophoridae). J. Kans. Entomol. Soc. 1985, 58, 526–531. [Google Scholar]
- Wcislo, W. Attraction and learning in mate-finding by solitary bees, Lasioglossum (Dialictus) figuresi Wcsilo and Nomia triangulifera Vachal (Hymenoptera: Halicidae). Behav. Ecol. Sociobiol. 1992, 31, 139–148. [Google Scholar] [CrossRef]
- Alcock, J.; Eickwort, G.C.; Eickwort, K.R. The reproductive behavior of Anthidium maculosum (hymenoptera: Megachilidae) and evolutionary significance of multiple copulations by females. Behav. Ecol. Sociobiol. 1977, 2, 385–396. [Google Scholar] [CrossRef]
- Splitt, A.; Schulz, M.; Skórka, P. Current state of knowledge on the biology and breeding of the solitary bee–Osmia bicornis. J. Apic. Res. 2022, 61, 163–179. [Google Scholar] [CrossRef]
- Strohm, E.; Daniels, H.; Warmers, C.; Stoll, C. Nest provisioning and a possible cost of reproduction in the megachilid bee Osmia rufa studied by a new observation method. Ethol. Ecol. Evol. 2002, 14, 255–268. [Google Scholar] [CrossRef]
- Strobl, V.; Straub, L.; Bruckner, S.; Albrecht, M.; Maitip, J.; Kolari, E.; Chantawannakul, P.; Neumann, P. Not every sperm counts: Male fertility in solitary bees, Osmia cornuta. PLoS ONE 2019, 14, e0214597. [Google Scholar] [CrossRef]
- Alcock, J. Postinsemination associations between males in insects: The mate-guarding hypothesis. Annu. Rev. Entomol. 1994, 39, 1–21. [Google Scholar] [CrossRef]
- Maccagnani, B. Bombus terrestris. Infect. Fitopatol. 1996, 5, 25–31. [Google Scholar]
- O’Neill, K.; Bjostad, L. The amel mating strategy of the bee Nomia nevadensis (Hymenoptera: Halictidae): Leg structure and mate guarding. Pan-Pac. Entomol. 1987, 63, 207–217. [Google Scholar]
- Barrows, E. Mating behavior in Halictine bees (Hymenoptera: Halictidae): III. Copulatory behavior and olfactory communication. Insect Sociaux 1975, 22, 307–332. [Google Scholar] [CrossRef]
- Lee, K.Y.; Yoon, H.J.; Lee, K.S.; Jin, B.R. Development and mating behaviour of Osmia cornifrons (Hymenoptera: Megachilidae) in the constant temperature. J. Asia-Pac. Entomol. 2016, 19, 281–287. [Google Scholar] [CrossRef]
- Villalobos, E.; Schelly, T. Correlates of male mating success in two species of Anthidium bees (Hymenoptera: Megachilidae). Behav. Ecol. Sociobiol. 1991, 29, 47–53. [Google Scholar] [CrossRef]
- Felicioli, A.; Isidoro, N.; Romani, R.; Bin, F.; Pinzauti, M. Ethological and morphological analysis of mating behaviour in Osmia cornuta Latr. (Hymenoptera, Megachilidae). Ins. Soc. Life 1998, 2, 137–144. [Google Scholar]
- Wittmann, D.; Blochtein, B. Why males of leafcutter bees hold the females’ antennae with their front legs during mating? Apidologie 1995, 26, 181–195. [Google Scholar] [CrossRef]
- Eberhard, W. Copulatory courtship and cryptic female choice in insects. Biol. Rev. 1991, 66, 1–31. [Google Scholar] [CrossRef]
- Wcislo, W.; Minckley, R.; Spangler, H.C. Pre-copulatory courtship behavior in a solitary bee, Nomia triangulifera Vachal (Hymenoptera: Halictidae). Apidologie 1992, 23, 431–442. [Google Scholar] [CrossRef]
- Conrad, T.; Ayasse, M. The role of vibrations in population divergence in the red mason bee, Osmia bicornis. Curr. Biol. 2015, 25, 2819–2822. [Google Scholar] [CrossRef]
- Turrel, M. Observations on the mating behavior of Anthidiellum notatum and Anthidiellum perplexum. Fla. Entomol. 1976, 59, 55–62. [Google Scholar] [CrossRef]
- Batra, S. Aggression, territoriality, mating and nest aggregation of some solitary bees (Hymenoptera: Halictidae, Megachilidae, Colletidae, Anthophoridae). J. Kans. Entomol. Soc. 1978, 51, 547–559. [Google Scholar]
- Triplet, D.; Gittings, A. Nesting, mating and foraging habits of Mellisodes (Mellissodes) tepida tepida cresson in Idaho (Hymenoptera: Anthophoridae). Proc. Entomol. Soc. Wash. 1998, 90, 462–470. [Google Scholar]
- Davies, N.B. Mating systems. In Behavioural Ecology; Krebs, J.R., Davies, N.B., Eds.; Blackwell: Oxford, UK, 1991; pp. 263–294. [Google Scholar]
- Conrad, T.; Paxton, R.J.; Barth, F.G.; Francke, W.; Ayasse, M. Female choice in the red mason bee, Osmia rufa (L.) (Megachilidae). J. Exp. Biol. 2010, 213, 4065–4073. [Google Scholar] [CrossRef] [PubMed]
- Conrad, T.; Vidkjær, N.H.; Ayasse, M. The origin of the compounds found on males’ antennae of the red mason bee, Osmia bicornis (L.). Chemoecology 2017, 27, 207–216. [Google Scholar] [CrossRef]
- Conrad, T.; Stöcker, C.; Ayasse, M. The effect of temperature on male mating signals and female choice in the red mason bee, Osmia bicornis (L.). Ecol. Evol. 2017, 7, 8966–8975. [Google Scholar] [CrossRef]
- Felicioli, A. Studi Bio-Etologici e Applicati Su Osmia Cornuta Latreille (Hymenoptera, Megachilidae), Potenziale Impolinatore di Piante di Interesse Agrario. Ph.D. Thesis, Università di Pisa, Pisa, Italy, 1995. [Google Scholar]
- Haccou, P.; Meelis, E. Statistical Analysis of Behavioural Data: An Approach Based on Time-Structured Models; Oxford University Press: Oxford, UK, 1994. [Google Scholar]
- Seidelmann, K. Untersuchungen zur Reproduktionsbiologie der Roten Mauerbiene, Osmia Rufa (L., 1758). Ph.D. Thesis, University of Halle-Wittenberg, Halle, Germany, 1995. [Google Scholar]
- Bartlet, E.; Isidoro, N.; Williams, I.H. Antennal glands in Psylliodes chrysocephala, and their possible role in reproductive behaviour. Physiol. Entomol. 1994, 19, 241–250. [Google Scholar] [CrossRef]
- Cowan, D. Sexual behavior of eumenid wasps (Hymenoptera: Eumenidae). Proc. Entomol. Soc. Wash. 1986, 88, 531–541. [Google Scholar]
- Medvedev, L.; Pavlov, S. Mating behaviour of the Chrysomelidae (Coleoptera). Entomol. Rev. 1988, 67, 100–109. [Google Scholar]
- Yin, X.W.; Iovinella, I.; Marangoni, R.; Cattonaro, F.; Flamini, G.; Sagona, S.; Zhang, L.; Pelosi, P.; Felicioli, A. Odorant-binding proteins nad olfactory coding in the solitary bee Osmia cornuta. Cell. Mol. Life Sci. 2011, 70, 3029–3039. [Google Scholar] [CrossRef]
- Ayasse, M.; Paxton, R.J.; Tengö, J. Mating behavior and chemical communication in the order Hymenoptera. Annu. Rev. Entomol. 2001, 46, 31–78. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Felicioli, A.; Sagona, S.; Coppola, F.; Boni, C.B.; Pinzauti, M. Effect of Ageing in the Mating Behaviour Sequence of Osmia cornuta Latr. (Hymenoptera: Megachilidae). Insects 2023, 14, 335. https://doi.org/10.3390/insects14040335
Felicioli A, Sagona S, Coppola F, Boni CB, Pinzauti M. Effect of Ageing in the Mating Behaviour Sequence of Osmia cornuta Latr. (Hymenoptera: Megachilidae). Insects. 2023; 14(4):335. https://doi.org/10.3390/insects14040335
Chicago/Turabian StyleFelicioli, Antonio, Simona Sagona, Francesca Coppola, Chiara Benedetta Boni, and Mauro Pinzauti. 2023. "Effect of Ageing in the Mating Behaviour Sequence of Osmia cornuta Latr. (Hymenoptera: Megachilidae)" Insects 14, no. 4: 335. https://doi.org/10.3390/insects14040335
APA StyleFelicioli, A., Sagona, S., Coppola, F., Boni, C. B., & Pinzauti, M. (2023). Effect of Ageing in the Mating Behaviour Sequence of Osmia cornuta Latr. (Hymenoptera: Megachilidae). Insects, 14(4), 335. https://doi.org/10.3390/insects14040335







