Potato (Solanum tuberosum) as a New Host for Pentastiridius leporinus (Hemiptera: Cixiidae) and Candidatus Arsenophonus Phytopathogenicus
Abstract
Simple Summary
Abstract
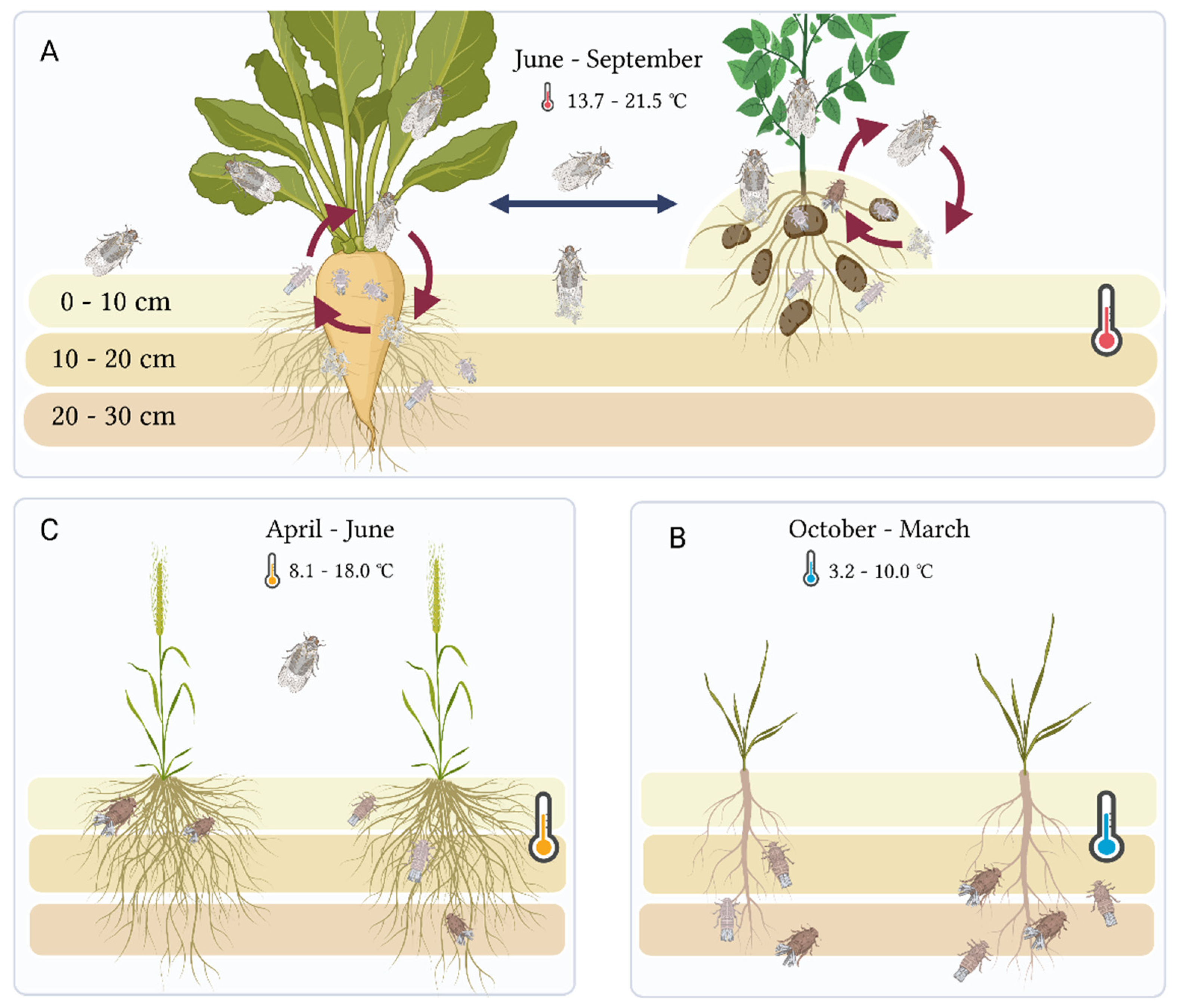
1. Introduction
2. Materials and Methods
2.1. Field Collection of P. leporinus from Potato Tubers and Sugar Beet Roots
2.2. Identification of Nymphs and Adults
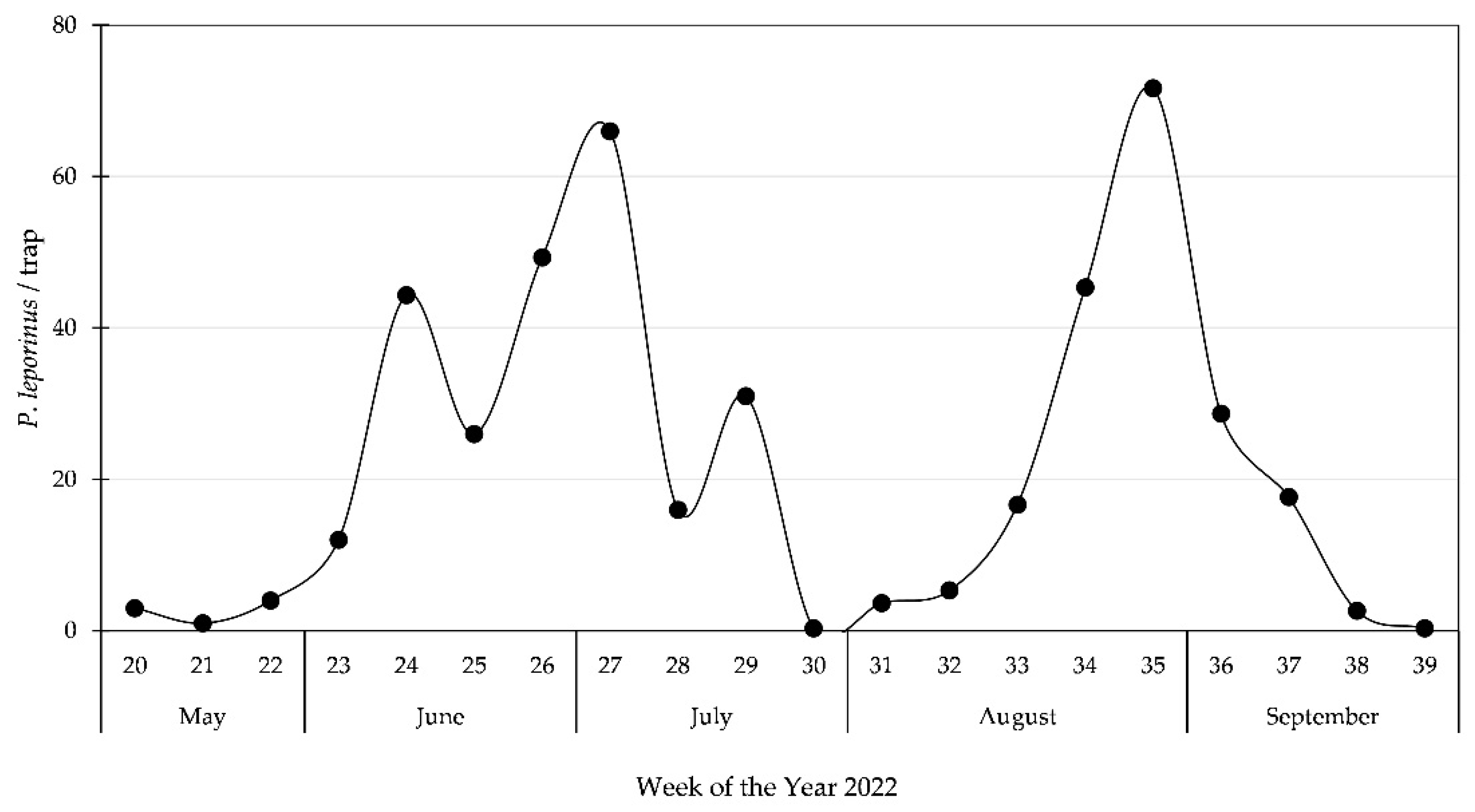
2.3. Abundance of P. leporinus in 2022
2.4. Transmission Assays with Nymphs
2.5. Comparison of Arsenophonus Isolated from Potato Tubers, Beet Roots, and P. leporinus
2.6. Analysis of Arsenophonus and Stolbur Infections
3. Results
3.1. Identification and Morphometric Analysis of P. leporinus on Potato Plants
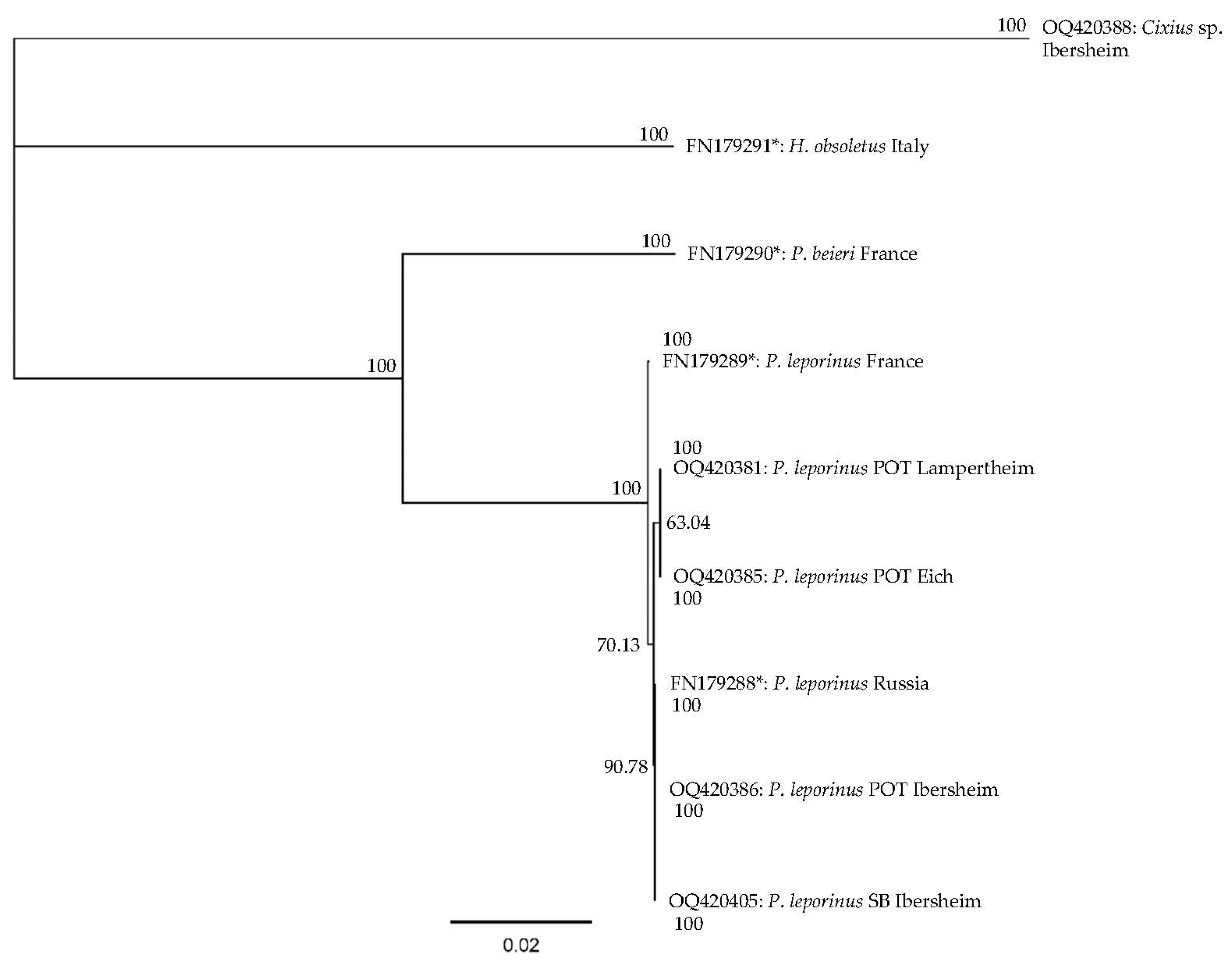
3.2. Molecular Identification of P. leporinus
3.3. Second Generation of Adults in Bickenbach, 2022
3.4. Transmission Assay
3.5. Similarity of Arsenophonus in All Organisms
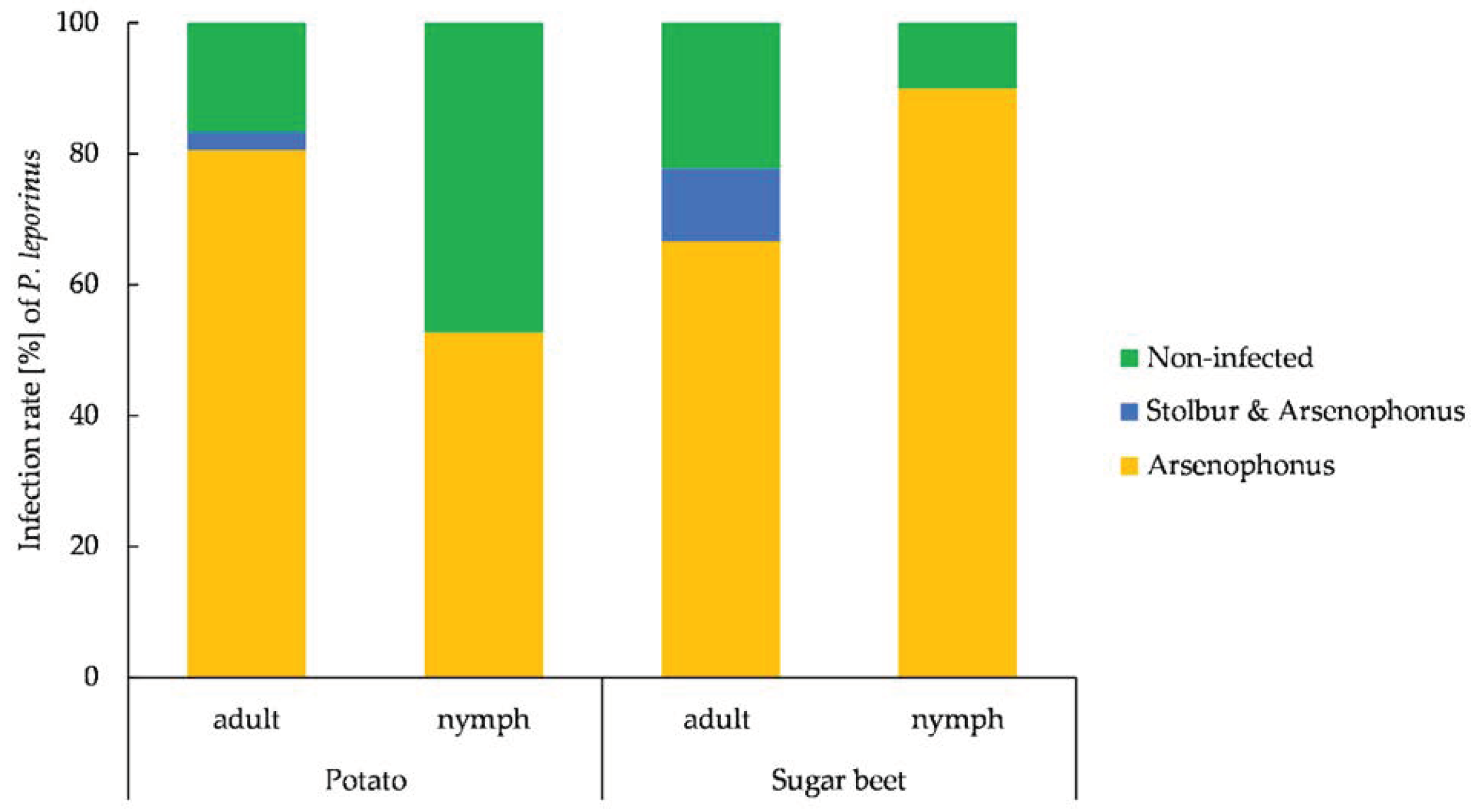
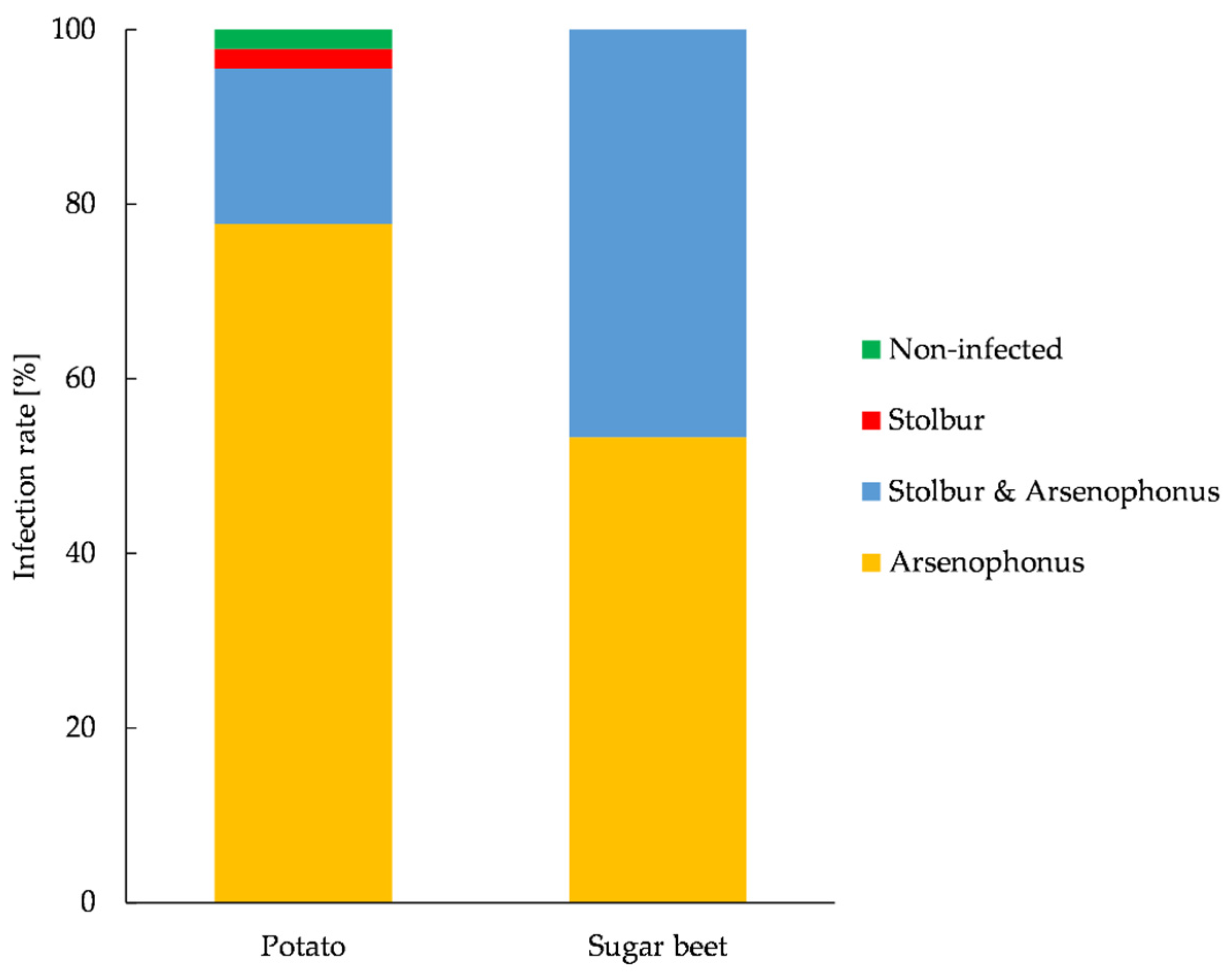
3.6. Prevalence of Arsenophonus and Stolbur in P. leporinus and Plant Material
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gatineau, F.; Larrue, J.; Clair, D.; Lorton, F.; Richard-Molard, M.; Boudon-Padieu, E. A New Natural Planthopper Vector of Stolbur Phytoplasma in the Genus Pentastiridius (Hemiptera: Cixiidae). Eur. J. Plant Pathol. 2001, 107, 263–271. [Google Scholar] [CrossRef]
- Gatineau, F.; Jacob, N.; Vautrin, S.; Larrue, J.; Lherminier, J.; Richard-Molard, M.; Boudon-Padieu, E. Association with the Syndrome “Basses Richesses” of Sugar Beet of a Phytoplasma and a Bacterium-Like Organism Transmitted by a Pentastiridius sp. Phytopathology 2002, 92, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Sémétey, O.; Bressan, A.; Gatineau, F.; Boudon-Padieu, E. Development of a specific assay using RISA for detection of the bacterial agent of “basses richesses” syndrome of sugar beet and confirmation of a Pentastiridius sp. (Fulgoromopha, Cixiidae) as the economic vector. Plant Pathol. 2007, 56, 797–804. [Google Scholar] [CrossRef]
- Biedermann, R.; Niedringhaus, R. Die Zikaden Deutschlands: Bestimmungstafeln für alle Arten; Wissenschaftlich-Akademischer-Buchvertrieb: Scheeßel, Germany, 2004; ISBN 978-3-939202-00-4. [Google Scholar]
- Bressan, A. Emergence and evolution of Arsenophonus bacteria as insect-vectored plant pathogens. Infect. Genet. Evol. 2014, 22, 81–90. [Google Scholar] [CrossRef]
- Behrmann, S.-C.; Witczak, N.; Lang, C.; Schieler, M.; Dettweiler, A.; Kleinhenz, B.; Schwind, M.; Vilcinskas, A.; Lee, K.-Z. Biology and Rearing of an Emerging Sugar Beet Pest: The Planthopper Pentastiridius leporinus. Insects 2022, 13, 656. [Google Scholar] [CrossRef]
- Behrmann, S.-C.; Schwind, M.; Schieler, M.; Vilcinskas, A.; Martinez, O.; Lee, K.-Z.; Lang, C. Spread of bacterial and virus yellowing diseases of sugar beet in South and Central Germany from 2017–2020. Sugar Ind. 2021, 146, 476–485. [Google Scholar] [CrossRef]
- Bressan, A.; Holzinger, W.-E.; Nusillard, B.; Sémétey, O.; Gatineau, F.; Simonato, M.; Boudon-Padieu, E. Identification and biological traits of a planthopper from the genus Pentastiridius (Hemiptera: Cixiidae) adapted to an annual cropping rotation. Eur. J. Entomol. 2009, 106, 405–413. [Google Scholar] [CrossRef]
- Mahillon, M.; Groux, R.; Bussereau, F.; Brodard, J.; Debonneville, C.; Demal, S.; Kellenberger, I.; Peter, M.; Steinger, T.; Schumpp, O. Virus Yellows and Syndrome “Basses Richesses” in Western Switzerland: A Dramatic 2020 Season Calls for Urgent Control Measures. Pathogens 2022, 11, 885. [Google Scholar] [CrossRef]
- Bressan, A.; Sémétey, O.; Nusillard, B.; Clair, D.; Boudon-Padieu, E. Insect Vectors (Hemiptera: Cixiidae) and Pathogens Associated with the Disease Syndrome “Basses Richesses” of Sugar Beet in France. Plant Dis. 2008, 92, 113–119. [Google Scholar] [CrossRef]
- Pfitzer, R.; Schrameyer, K.; Voegele, R.-T.; Maier, J.; Lang, C.; Varrelmann, M. Causes and effects of the occurrence of “Syndrome Basses Richesses” in German sugar beet growing areas. Sugar Ind. 2019, 145, 70–80. [Google Scholar]
- Bressan, A.; Sémétey, O.; Arneodo, J.; Lherminier, J.; Boudon-Padieu, E. Vector transmission of a plant-pathogenic bacterium in the Arsenophonus clade sharing ecological traits with facultative insect endosymbionts. Phytopathology 2009, 99, 1289–1296. [Google Scholar] [CrossRef]
- Ćurčić, Ž.; Stepanović, J.; Zübert, C.; Taški-Ajduković, K.; Kosovac, A.; Rekanović, E.; Kube, M.; Duduk, B. Rubbery Taproot Disease of Sugar Beet in Serbia Associated with ‘Candidatus Phytoplasma solani’. Plant Dis. 2021, 105, 255–263. [Google Scholar] [CrossRef]
- Panjan, M. Reserches sur stolbur des Solanaceae et le mode de lutte. Plant Prot. 1950, 2, 49–58. [Google Scholar]
- Maixner, M.; Ahrens, U.; Seemüller, E. Detection of the German grapevine yellows (Vergilbungskrankheit) MLO in grapevine, alternative hosts and a vector by a specific PCR procedure. Eur. J. Plant Pathol. 1995, 101, 241–250. [Google Scholar] [CrossRef]
- Johannesen, J.; Lux, B.; Michel, K.; Seitz, A.; Maixner, M. Invasion biology and host specificity of the grapevine yellows disease vector Hyalesthes obsoletus in Europe. Entomol. Exp. Appl. 2008, 126, 217–227. [Google Scholar] [CrossRef]
- Carraro, L.; Ferrini, F.; Martini, M.; Ermacora, P.; Loi, N. A serious epidemic of stolbur on celery. J. Plant Pathol. 2008, 90, 131–135. [Google Scholar]
- Gaudin, J.; Sémétey, O.; Foissac, X.; Eveillard, S. Phytoplasma titer in diseased lavender is not correlated to lavender tolerance to stolbur phytoplasma. Bull. Insectology 2011, 64, 179–180. [Google Scholar]
- Mori, N.; Mitrović, J.; Smiljković, M.; Duduk, N.; Paltrinieri, S.; Bertaccini, A.; Duduk, B. Hyalesthes obsoletus in Serbia and its role in the epidemiology of corn reddening. Bull. Insectology 2013, 66, 245–250. [Google Scholar]
- Chuche, J.; Danet, J.-L.; Rivoal, J.-B.; Arricau-Bouvery, N.; Thiéry, D. Minor cultures as hosts for vectors of extensive crop diseases: Does Salvia sclarea act as a pathogen and vector reservoir for lavender decline? J. Pest Sci. 2018, 91, 145–155. [Google Scholar] [CrossRef]
- Jović, J.; Ember, I.; Mitrović, M.; Cvrković, T.; Krstić, O.; Krnjajić, S.; Acs, Z.; Kölber, M.; Toševski, I. Molecular detection of potato stolbur phytoplasma in Serbia. Bull. Insectology 2011, 64, 83–84. [Google Scholar]
- Mitrović, M.; Trivellone, V.; Jović, J.; Cvrković, T.; Jakovljevic, M.; Kosovac, A.; Krstić, O.; Toševski, I. Potential Hemipteran vectors of “stolbur” phytoplasma in potato fields in Serbia. Phyt. Moll. 2015, 5, 49. [Google Scholar] [CrossRef]
- Holzinger, W.-E.; Kammerlander, I.; Nickel, H. The Auchenorrhyncha of Central Europe. Die Zikaden Mitteleuropas: Volume 1: Fulgoromorpha, Cicadomorpha excl. Cicadellidae; BRILL: Leiden, The Netherlands, 2003; ISBN 9789004128958. [Google Scholar]
- Simon, C.; Buckley, T.R.; Frati, F.; Stewart, J.-B.; Beckenbach, A.-T. Incorporating Molecular Evolution into Phylogenetic Analysis, and a New Compilation of Conserved Polymerase Chain Reaction Primers for Animal Mitochondrial DNA. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 545–579. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Munyaneza, J.-E.; Henne, D.-C. Leafhopper and psyllid pests of potato. In Insect Pests of Potato; San Diego, A.P., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 65–102. ISBN 9780123868954. [Google Scholar]
- Pfitzer, R. Untersuchung zur Verbreitung und Kontrolle von SBR in Zuckerrüben. Master’s Thesis, University of Hohenheim, Stuttgart, Germany, 1 February 2019. [Google Scholar]
- Deutsch, C.A.; Tewksbury, J.J.; Tigchelaar, M.; Battisti, D.S.; Merrill, S.C.; Huey, R.B.; Naylor, R.L. Increase in crop losses to insect pests in a warming climate. Science 2018, 361, 916–919. [Google Scholar] [CrossRef]
- Danet, J.-L.; Foissac, X.; Zreik, L.; Salar, P.; Verdin, E.; Nourrisseau, J.-G.; Garnier, M. “Candidatus Phlomobacter fragariae” Is the Prevalent Agent of Marginal Chlorosis of Strawberry in French Production Fields and Is Transmitted by the Planthopper Cixius wagneri (China). Phytopathology 2003, 93, 644–649. [Google Scholar] [CrossRef]
- Weintraub, P.G.; Beanland, L. Insect vectors of phytoplasmas. Annu. Rev. Entomol. 2006, 51, 91–111. [Google Scholar] [CrossRef]
- Hogenhout, S.A.; Oshima, K.; Ammar, E.-D.; Kakizawa, S.; Kingdom, H.N.; Namba, S. Phytoplasmas: Bacteria that manipulate plants and insects. Mol. Plant Pathol. 2008, 9, 403–423. [Google Scholar] [CrossRef]
- Crosslin, J.-M.; Hamlin, L.-L.; Buchman, J.-L.; Munyaneza, J.-E. Transmission of Potato Purple Top Phytoplasma to Potato Tubers and Daughter Plants. Am. J. Potato Res. 2011, 88, 339–345. [Google Scholar] [CrossRef]
- Statistical Office of the Federal State of Rhineland-Palatinate. Utilised Agricultural Area from 1960 until 2022 for Selected Crops. Available online: https://www.statistik.rlp.de/de/wirtschaftsbereiche/landwirtschaft/zeitreihen-land/tabelle-2/ (accessed on 5 February 2023).

| Location | State | Crops | Coordinates |
|---|---|---|---|
| Eich | Rhineland-Palatinate | Potato, sugar beet | 49.765428, 8.393707 |
| Ibersheim | Rhineland-Palatinate | Potato, sugar beet | 49.726559, 8.393205 |
| Lampertheim | Hesse | Potato | 49.610846, 8.465391 |
| Bickenbach | Hesse | Sugar beet | 49.758758, 8.577285 |
| Plants | Group of Nymphs | |||
|---|---|---|---|---|
| Plant Species | Arsenophonus | Stolbur | Arsenophonus | Stolbur |
| Potato | 3/12 | 0/12 | 12/12 | 0/12 |
| Sugar beet | 1/3 | 1/3 | 3/3 | 1/3 |
| Potato (Control) | 0/3 | 0/3 | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behrmann, S.C.; Rinklef, A.; Lang, C.; Vilcinskas, A.; Lee, K.-Z. Potato (Solanum tuberosum) as a New Host for Pentastiridius leporinus (Hemiptera: Cixiidae) and Candidatus Arsenophonus Phytopathogenicus. Insects 2023, 14, 281. https://doi.org/10.3390/insects14030281
Behrmann SC, Rinklef A, Lang C, Vilcinskas A, Lee K-Z. Potato (Solanum tuberosum) as a New Host for Pentastiridius leporinus (Hemiptera: Cixiidae) and Candidatus Arsenophonus Phytopathogenicus. Insects. 2023; 14(3):281. https://doi.org/10.3390/insects14030281
Chicago/Turabian StyleBehrmann, Sarah Christin, André Rinklef, Christian Lang, Andreas Vilcinskas, and Kwang-Zin Lee. 2023. "Potato (Solanum tuberosum) as a New Host for Pentastiridius leporinus (Hemiptera: Cixiidae) and Candidatus Arsenophonus Phytopathogenicus" Insects 14, no. 3: 281. https://doi.org/10.3390/insects14030281
APA StyleBehrmann, S. C., Rinklef, A., Lang, C., Vilcinskas, A., & Lee, K.-Z. (2023). Potato (Solanum tuberosum) as a New Host for Pentastiridius leporinus (Hemiptera: Cixiidae) and Candidatus Arsenophonus Phytopathogenicus. Insects, 14(3), 281. https://doi.org/10.3390/insects14030281





