A New Genus of Spittlebugs (Hemiptera, Cercopidae) from the Eocene of Central Tibetan Plateau †
Abstract
Simple Summary
Abstract
1. Introduction
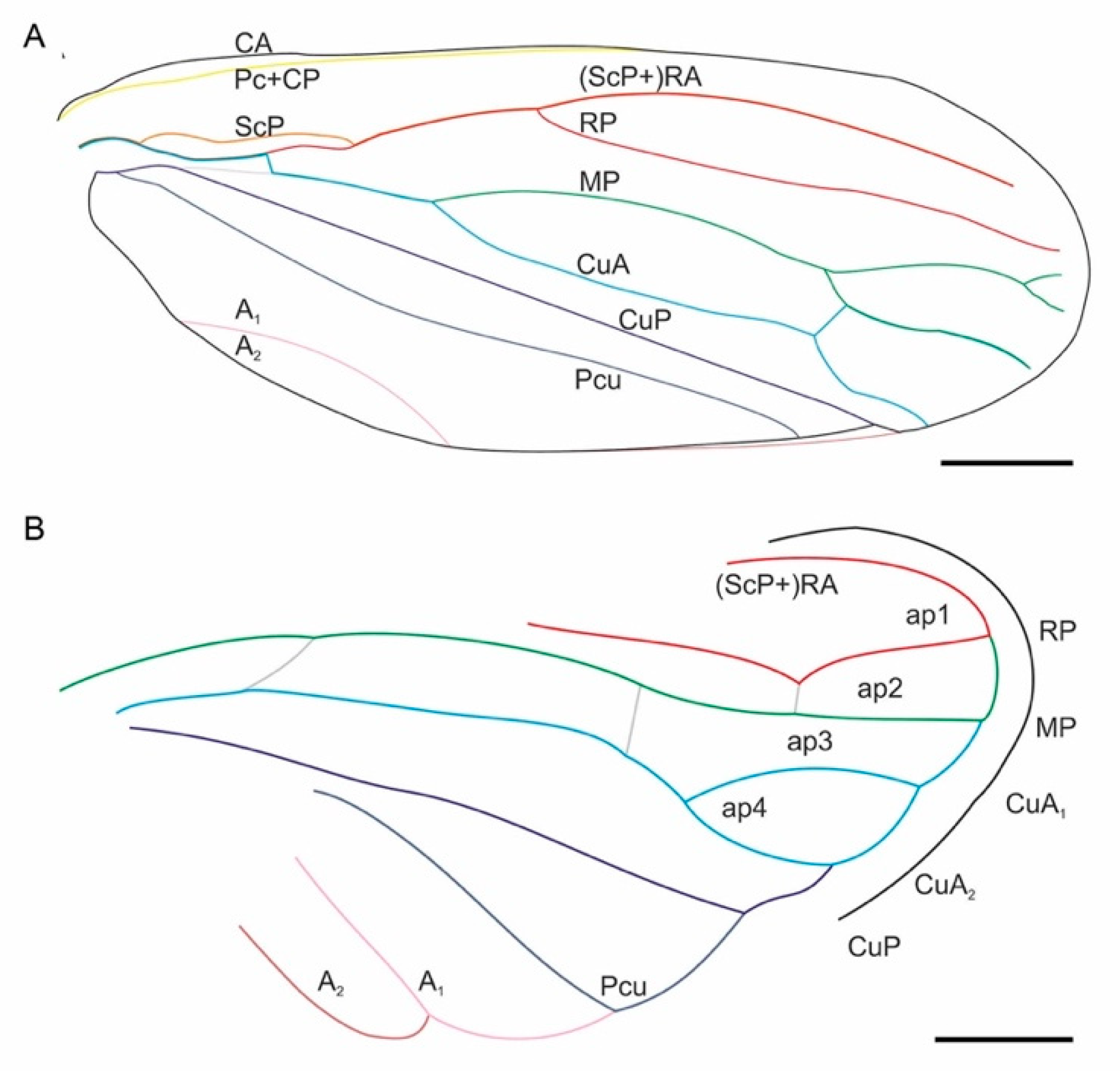
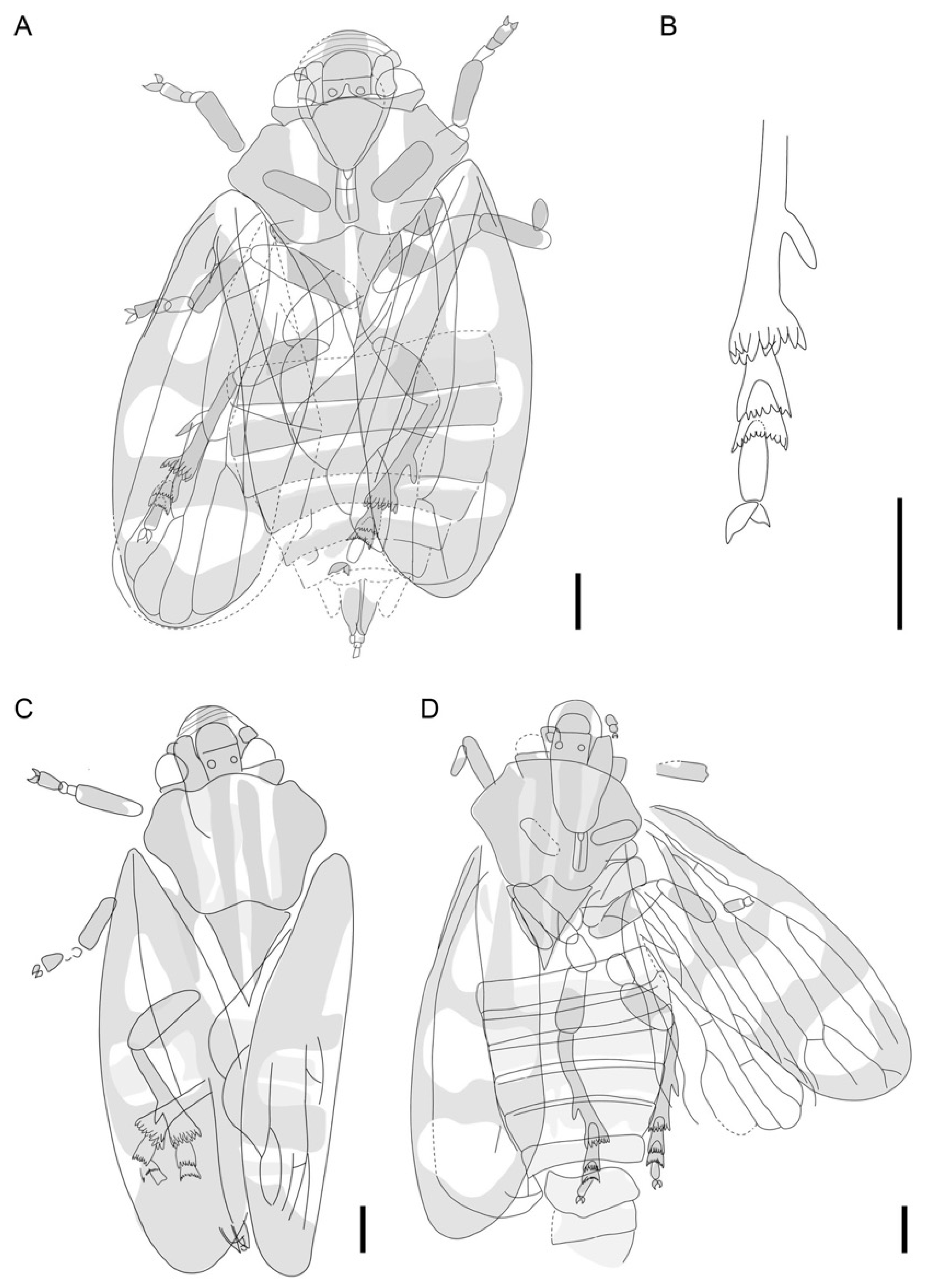
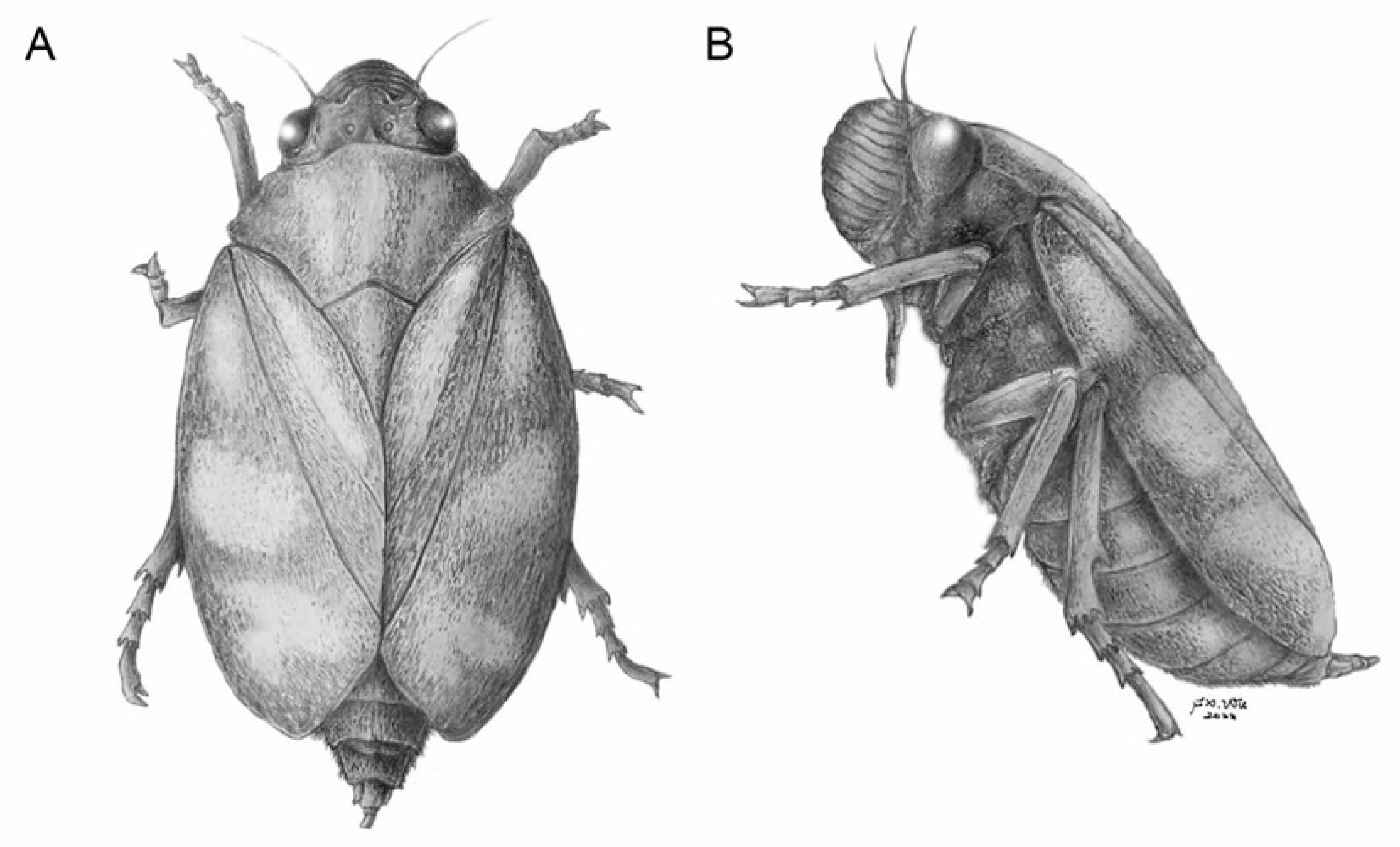
2. Materials and Methods
3. Results
4. Discussion
4.1. Taxonomic Remarks
4.2. Biogeographic Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paladini, A.; Carvalho, G.S.; Cavichioli, R.R. Taxonomic review of Sphenoclypeana and cladistic analysis of Ischnorhinini (Hemiptera, Cercopidae, Tomaspidinae). Zootaxa 2010, 2502, 24–36. [Google Scholar] [CrossRef]
- Dietrich, C.H. Keys to the families of Cicadomorpha and subfamilies and tribes of Cicadellidae (Hemiptera: Auchenorrhyncha). Fla. Entomol. 2005, 88, 502–517. [Google Scholar] [CrossRef]
- Cryan, J.R.; Svenson, G.J. Family-level relationships of the spittlebugs and froghoppers (Hemiptera: Cicadomorpha: Cercopoidea). Syst. Entomol. 2010, 35, 393–415. [Google Scholar] [CrossRef]
- Szwedo, J. The unity, diversity and conformity of bugs (Hemiptera) through time. Earth Environ. Sci. Trans. R. Soc. Edinb. 2018, 107, 109–128. [Google Scholar] [CrossRef]
- Bartlett, C.R.; Deitz, L.L.; Dmitriev, D.A.; Sanborn, A.F.; Soulier-Perkins, A.; Wallace, M.S. The diversity of the true hoppers (Hemiptera: Auchenorrhyncha). In Insect Biodiversity. Science and Society; Foottit, R.G., Adler, P., Eds.; Wiley Blackwell: Chichester, UK, 2018; Volume 2, pp. 501–590. [Google Scholar] [CrossRef]
- Cryan, J.R. Molecular phylogeny of Cicadomorpha (Insecta: Hemiptera: Cicadoidea, Cercopoidea and Membracoidea): Adding evidence to the controversy. Syst. Entomol. 2005, 30, 563–574. [Google Scholar] [CrossRef]
- Soulier-Perkins, A. Cercopoidea Organised on Line. Available online: http://hemiptera-databases.org/Cool/ (accessed on 9 March 2022).
- Hamilton, K.G.A. Neotropical spittlebugs related to Neaenini (Hemiptera, Cercopidae) and the origins of subfamily Cercopinae. Zootaxa 2016, 4169, 201. [Google Scholar] [CrossRef]
- Behrensmeyer, A.K.; Turner, A. Taxonomic Occurrences of Cercopoidea Recorded in the Paleobiology Database. Fossilworks. Available online: http://fossilworks.org. (accessed on 14 April 2022).
- Paleobiology Database. Cercopoidea; PaleoBioDB. Available online: https://paleobiodb.org/navigator/ (accessed on 14 April 2022).
- Lin, Q.B.; Lee, C.M. Early Tertiary insects from Peng Chau, Hong Kong. Palaeontol. Stratigr. Hong Kong 1997, 1, 185–199. [Google Scholar]
- Brodie, P.B. A History of the Fossil Insects in the Secondary Rocks of England. Accompanied by a Particular Account of the Strata in Which They Occur, and of the Circumstances Connected with Their Preservation; J. van Voorst: London, UK, 1845; pp. 399–402. [Google Scholar]
- Coram, R.A.; Jepson, J.E. Fossil Insects of the Purbeck Limestone Group of Southern England: Palaeoentomology from the Dawn of the Cretaceous; Siri Scientific Press: Manchester, UK, 2012; Volume 3, p. 144. [Google Scholar]
- Carvalho, G.S.; Webb, M.D. Cercopid Spittle Bugs of the New World (Hemiptera, Auchenorrhyncha, Cercopidae); Pensoft: Sofia, Bulgaria, 2005; Volume 271. [Google Scholar]
- Peck, D.C. Reflex bleeding in froghoppers (Homoptera: Cercopidae): Variation in behavior and taxonomic distribution. Ann. Entomol. Soc. Am. 2000, 93, 1186–1194. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Casson, D. A lesser predilection for bugs: Hemiptera (Insecta) diversity in tropical rain forests. Biol. J. Linn. Soc. 1991, 43, 101–109. [Google Scholar] [CrossRef]
- Thompson, V. Spittlebug indicators of nitrogen-fixing plants. Los salivazos como indicadores de plantas fijadoras de nitrógeno. Ecol. Entomol. 1994, 19, 391–398. [Google Scholar] [CrossRef]
- Jarvis, A.; Reuter, H.; Nelson, A.; Guevara, E. Hole-Filled Seamless SRTM Data V4: International Centre For Tropical Agriculture (CIAT), 2008. Available online: https://srtm.csi.cgiar.org (accessed on 27 March 2021).
- Kapp, P.; DeCelles, P.G.; Gehrels, G.E.; Heizler, M.; Ding, L. Geological records of the Lhasa-Qiangtang and Indo-Asian collisions in the Nima area of central Tibet. Geol. Soc. Am. Bull. 2007, 119, 917–933. [Google Scholar] [CrossRef]
- Kapp, P.; Yin, A.; Harrison, T.M.; Ding, L. Cretaceous-Tertiary shortening, basin development, and volcanism in central Tibet. Geol. Soc. Am. Bull. 2005, 117, 865–878. [Google Scholar] [CrossRef]
- DeCelles, P.G.; Kapp, P.; Ding, L.; Gehrels, G.E. Late Cretaceous to middle Tertiary basin evolution in the central Tibetan Plateau: Changing environments in response to tectonic partitioning, aridification, and regional elevation gain. Geol. Soc. Am. Bull. 2007, 119, 654–680. [Google Scholar] [CrossRef]
- Fang, X.; Dupont-Nivet, G.; Wang, C.; Song, C.; Meng, Q.; Zhang, W.; Nie, J.; Zhang, T.; Mao, Z.; Chen, Y. Revised chronology of central Tibet uplift (Lunpola Basin). Sci. Adv. 2020, 6, eaba7298. [Google Scholar] [CrossRef]
- Xiong, Z.; Liu, X.; Ding, L.; Farnsworth, A.; Spicer, R.A.; Xu, Q.; Valdes, P.; He, S.; Zeng, D.; Wang, C. The rise and demise of the Paleogene central Tibetan valley. Sci. Adv. 2022, 8, eabj0944. [Google Scholar] [CrossRef]
- Wu, F.; Miao, D.; Chang, M.; Shi, G.; Wang, N. Fossil climbing perch and associated plant megafossils indicate a warm and wet central Tibet during the late Oligocene. Sci. Rep. 2017, 7, 878. [Google Scholar] [CrossRef]
- Jia, L.-B.; Su, T.; Huang, Y.; Wu, F.; Deng, T.; Zhou, Z.-K. First fossil record of Cedrelospermum (Ulmaceae) from the Qinghai-Tibetan Plateau: Implications for morphological evolution and biogeography. J. Syst. Evol. 2018, 57, 94–104. [Google Scholar] [CrossRef]
- Cai, C.; Huang, D.; Wu, F.; Zhao, M.; Wang, N. Tertiary water striders (Hemiptera, Gerromorpha, Gerridae) from the central Tibetan Plateau and their palaeobiogeographic implications. J. Asian Earth Sci. 2019, 175, 121–127. [Google Scholar] [CrossRef]
- Su, T.; Farnsworth, A.; Spicer, R.A.; Huang, J.; Wu, F.; Liu, J.; Li, S.; Xing, Y.; Huang, Y.; Deng, W. No high Tibetan Plateau until the Neogene. Sci. Adv. 2019, 5, eaav2189. [Google Scholar] [CrossRef]
- Nel, A.; Prokop, J.; Nel, P.; Grandcolas, P.; Huang, D.-Y.; Roques, P.; Guilbert, E.; Dostál, O.; Szwedo, J. Traits and evolution of wing venation pattern in paraneopteran insects. J. Morphol. 2012, 273, 480–506. [Google Scholar] [CrossRef]
- Wang, T.; Pan, L.; Zhang, Y.; Dai, W. Morphology of the mouthparts of the spittlebug Philagra albinotata Uhler (Hemiptera: Cercopoidea: Aphrophoridae). Arthropod Struct. Dev. 2015, 44, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Lallemand, V. Révision des Cercopinae: (Hemiptera Homoptera), Première partie; Memoires, Institut royal des sciences naturelles de Belgique: Bruxelles, Belgium, 1949; 2ème serie; Volume 32, pp. 1–193+IV plates. [Google Scholar]
- Lallemand, V.; Synave, H. Révision des Cercopinae: (Hemiptera Homoptera), Deuxième partie; Memoires, Institut royal des sciences naturelles de Belgique: Bruxelles, Belgium, 1961; Volume 66, pp. 1–153+XL plates. [Google Scholar]
- Schöbel, C.; Carvalho, G.S. The “State of Art” of Mahanarva (Hemiptera: Cercopidae) research. An economically important New World spittlebug genus. Appl. Entomol. Zool. 2021, 56, 299–309. [Google Scholar] [CrossRef]
- Fennah, R.G. Revisionary notes on the New World genera of cercopid froghoppers (Homoptera: Cercopoidea). Bull. Entomol. Res. 1968, 58, 165–190. [Google Scholar] [CrossRef]
- Hamilton, K.G.A. A new family of froghoppers from the American tropics (Hemiptera: Cercopoidea: Epipygidae). Biodiversity 2001, 2, 15–21. [Google Scholar] [CrossRef]
- Hamilton, K.G.A. Revision of Neotropical aphrophorine spittlebugs, part 1: Ptyelini (Hemiptera, Cercopoidea). Zootaxa 2012, 3497, 41. [Google Scholar] [CrossRef]
- Liang, A.; Jiang, G.; Webb, M.D. Revision of the Oriental spittlebug genera Baibarana Matsumura and Telogmometopius Jacobi (Hemiptera: Cercopidae: Rhinaulacini). J. Nat. Hist. 2006, 40, 2071–2099. [Google Scholar] [CrossRef]
- Distant, W.L. Rhynchota. Homoptera: Membracidae, Cercopidae, Jassidae & Heteroptera: Appendix. In Fauna Br. India Incl. Ceylon Burma; Lt.-Col., C.T., Ed.; Secretary of State for India in Council: London, UK; Taylor and Francis: London, UK, 1908; Volume 4, pp. i–xv+1–501. [Google Scholar]
- Schmidt, E. Neue Gattungen und Arten der Subfamilie Cercopinae Stål, ein Beitrag zur Kenntnis der Cercopiden (Hemiptera-Homoptera). Arch. Naturgesch. 1910, 76, 53–112. [Google Scholar]
- Lallemand, V. Nouveaux cercopides de la presqu’île Malaise. J. Fed. Malay States Mus. 1930, 16, 98–107. [Google Scholar]
- Lallemand, V. Descriptions de quatre nouveaux Cercopidae (Rhynch., Homoptera). Treubia 1953, 22, 501–504. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Dai, R.-H. Spittlebug genus Kanozata Matsumura (Hemiptera, Cercopoidea: Cercopidae) with a new distribution record of K. contermina (Distant) to China. Entomol. Fenn. 2018, 29, 191–200. [Google Scholar] [CrossRef]
- Schmidt, E. Neue und bekannte Gattungen und Arten der Subfamilie Cercopinae Stål des Indoaustralischen Faunengebietes, ein Beitrag zur Kenntnis der Cercopiden. Hemiptera-Homoptera III. Stettin. Entomol. Ztg. 1911, 72, 52–129. [Google Scholar]
- Stål, C. Hemiptera insularum Philippinarum. Bidrag till Philippinska öarnes Hemipter-Fauna. Öfvers. Kongl. Vetensk.-Akad. Förh. 1870, 27, 607–776+IX plates. [Google Scholar]
- Metcalf, Z.P.; Wade, V. A catalogue of the fossil Homoptera (Homoptera: Auchenorhyncha). In General Catalogue of the Homoptera. A supplement to Fascicle I, Paper N° 2049; North Carolina State University: Raleigh, NC, USA, 1966; pp. 1–245. [Google Scholar]
- Wang, L.L.; Zhao, L.L.; Tian, M.Z.; Wu, F.D.; Zhang, X.J. Late Jurassic to Early Cretaceous volcanism of Hong Kong: Insights from the Ping Chau Formation. Geosci. Front. 2019, 10, 553–568. [Google Scholar] [CrossRef]
- Zeuner, F.E. V.—The Eocene insects of the Ardtun Beds, Isle of Mull, Scotland. Ann. Mag. Nat. Hist. 1941, 7, 82–100. [Google Scholar] [CrossRef]
- Germar, E.F.; Berendt, G.C. Die im Bernstein befindlichen Hemipteren und Orthopteren der Vorwelt. In Die im Bernstein befindlichen organischen Resten der Vorwelt; Nicolai Buchhandlung: Berlin, Germany, 1856; Volume 2, pp. 1–40. [Google Scholar]
- Szwedo, J.; Drohojowska, J.; Popov, Y.A.; Simon, E.; Wegierek, P. Aphids, true hoppers, jumping plant-lice, scale insects, true bugs and whiteflies (Insecta: Hemiptera) from the Insect Limestone (latest Eocene) of the Isle of Wight, UK. Earth Environ. Sci. Trans. R. Soc. Edinb. 2019, 110, 331–396. [Google Scholar] [CrossRef]
- Paladini, A.; Takiya, D.M.; Cavichioli, R.R.; Carvalho, G.S. Phylogeny and biogeography of Neotropical spittlebugs (Hemiptera: Cercopidae: Ischnorhininae): Revised tribal classification based on morphological data. Syst. Entomol. 2015, 40, 82–108. [Google Scholar] [CrossRef]
- Crispolon, E.S., Jr.; Soulier-Perkins, A. A first attempt on the morphological phylogeny of Cercopinae (Hemiptera, Cercopidae). In Abstracts and Posters, Proceedings of the 16th International Auchenorrhyncha Congress and 12th International Workshop on Leafhoppers and Planthoppers of Economic Significance, Nho Quan, Vietnam, 2–8 July 2019; Vietnam National Museum of Nature: Hanoi, Vietnam, 2019; p. 114. [Google Scholar]
- Cockerell, T.D.A. Tertiary insects from Kudia River, Maritime Province, Siberia. Proc. U. S. Natl. Mus. 1925, 68, 1–16, plates 1, 2. [Google Scholar] [CrossRef][Green Version]
- Cockerell, T.D.A. LXXV.—Fossil insects in the British Museum. Ann. Mag. Nat. Hist. 1927, 20, 585–594. [Google Scholar] [CrossRef]
- Zhang, X.; Gélin, U.; Spicer, R.A.; Wu, F.X.; Farnsworth, A.; Chen, P.R.; Del Rio, C.; Li, S.F.; Liu, J.; Huang, J.; et al. Rapid Eocene diversification of spiny plants in subtropical woodlands of central Tibet. Nat. Commun. 2022, 13, 3787. [Google Scholar] [CrossRef]
- Xia, G.; Zheng, D.; Krieg-Jacquier, R.; Fan, Q.; Chen, Y.; Nel, A. The oldest-known Lestidae (Odonata) from the late Eocene of Tibet: Palaeoclimatic implications. Geol. Mag. 2022, 159, 511–518. [Google Scholar] [CrossRef]
- Xue, W.; Najman, Y.; Hu, X.; Persano, C.; Stuart, F.M.; Li, W.; Ma, A.; Wang, Y. Late Cretaceous to Late Eocene exhumation in the Nima area, central Tibet: Implications for development of low relief topography of the Tibetan Plateau. Tectonics 2022, 41, e2021TC006989. [Google Scholar] [CrossRef]
- Su, T.; Spicer, R.A.; Li, S.H.; Huang, J.; Sherlock, S.; Huang, Y.J.; Li, S.F.; Wang, L.; Jia, L.B.; Deng, W.Y.D.; et al. Uplift, climate and biotic changes at the Eocene–Oligocene transition in south-eastern Tibet. Nat. Sci. Rev. 2019, 6, 495–504. [Google Scholar] [CrossRef]
- DeCelles, P.; Quade, J.; Kapp, P.; Fan, M.J.; Dettman, D.L.; Ding, L. High and dry in central Tibet during the Late Oligocene. Earth Planet. Sci. Lett. 2007, 253, 389–401. [Google Scholar] [CrossRef]
- Outhwaite, C.L.; McCann, P.; Newbold, T. Agriculture and climate change are reshaping insect biodiversity worldwide. Nature 2022, 605, 97–102. [Google Scholar] [CrossRef]
- Robinet, C.; Roques, A. Direct impacts of recent climate warming on insect populations. Integr. Zool. 2010, 5, 132–142. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.-T.; Szwedo, J.; Huang, D.-Y.; Deng, W.-Y.-D.; Obroślak, M.; Wu, F.-X.; Su, T. A New Genus of Spittlebugs (Hemiptera, Cercopidae) from the Eocene of Central Tibetan Plateau. Insects 2022, 13, 770. https://doi.org/10.3390/insects13090770
Xu X-T, Szwedo J, Huang D-Y, Deng W-Y-D, Obroślak M, Wu F-X, Su T. A New Genus of Spittlebugs (Hemiptera, Cercopidae) from the Eocene of Central Tibetan Plateau. Insects. 2022; 13(9):770. https://doi.org/10.3390/insects13090770
Chicago/Turabian StyleXu, Xiao-Ting, Jacek Szwedo, Di-Ying Huang, Wei-Yu-Dong Deng, Martyna Obroślak, Fei-Xiang Wu, and Tao Su. 2022. "A New Genus of Spittlebugs (Hemiptera, Cercopidae) from the Eocene of Central Tibetan Plateau" Insects 13, no. 9: 770. https://doi.org/10.3390/insects13090770
APA StyleXu, X.-T., Szwedo, J., Huang, D.-Y., Deng, W.-Y.-D., Obroślak, M., Wu, F.-X., & Su, T. (2022). A New Genus of Spittlebugs (Hemiptera, Cercopidae) from the Eocene of Central Tibetan Plateau. Insects, 13(9), 770. https://doi.org/10.3390/insects13090770







