Pharmacological Properties and Function of the PxOctβ3 Octopamine Receptor in Plutella xylostella (L.)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Total RNA Extraction and cDNA Synthesis
2.3. Cloning of the Full-Length PxOctβ3 ORF Sequence
2.4. Bioinformatic Analysis of the PxOctβ3
2.5. Analysis of the Pattern of Expression of PxOctβ3
2.6. Construction of a Cell Line That Stably Expresses PxOctβ3
2.7. Cyclic AMP (cAMP) Assays
2.8. Synthesis and Microinjection of dsRNA
2.9. Data Analysis
3. Results
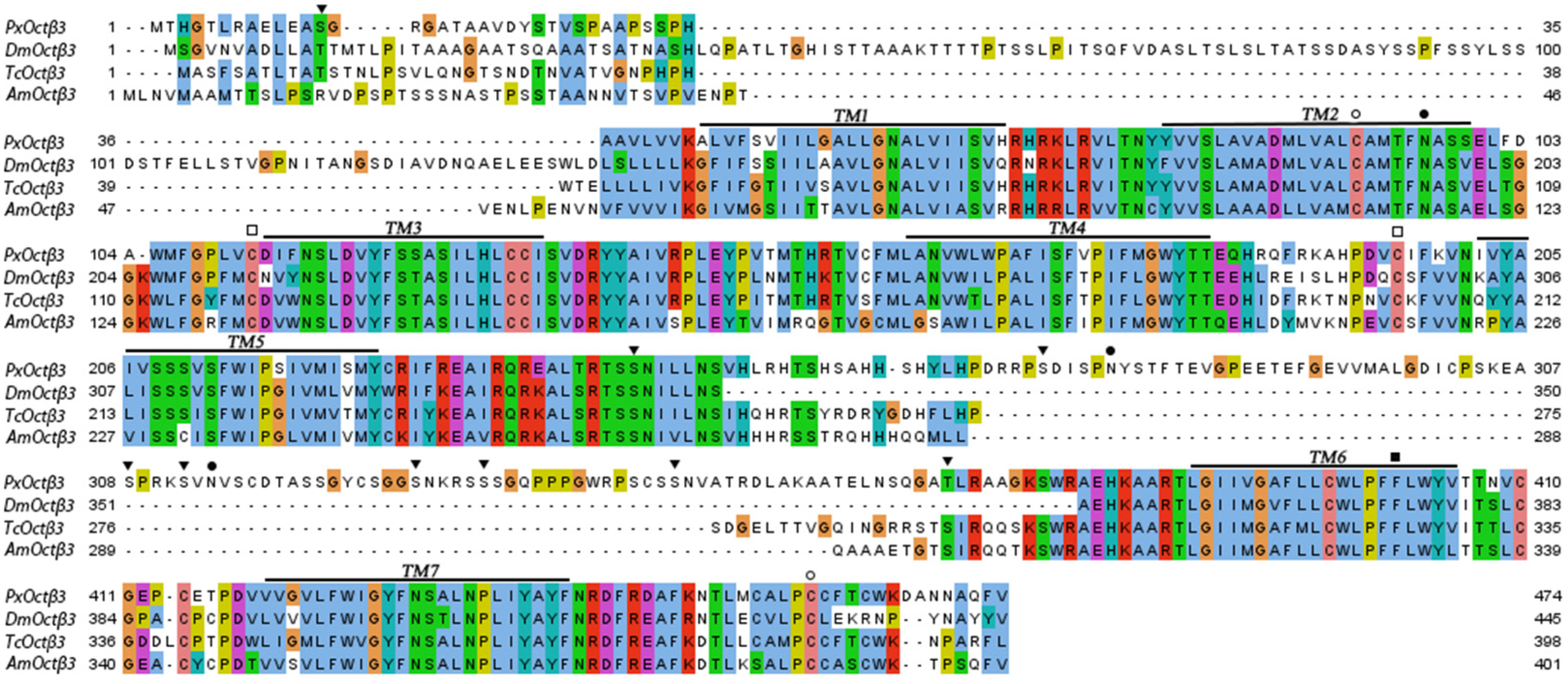
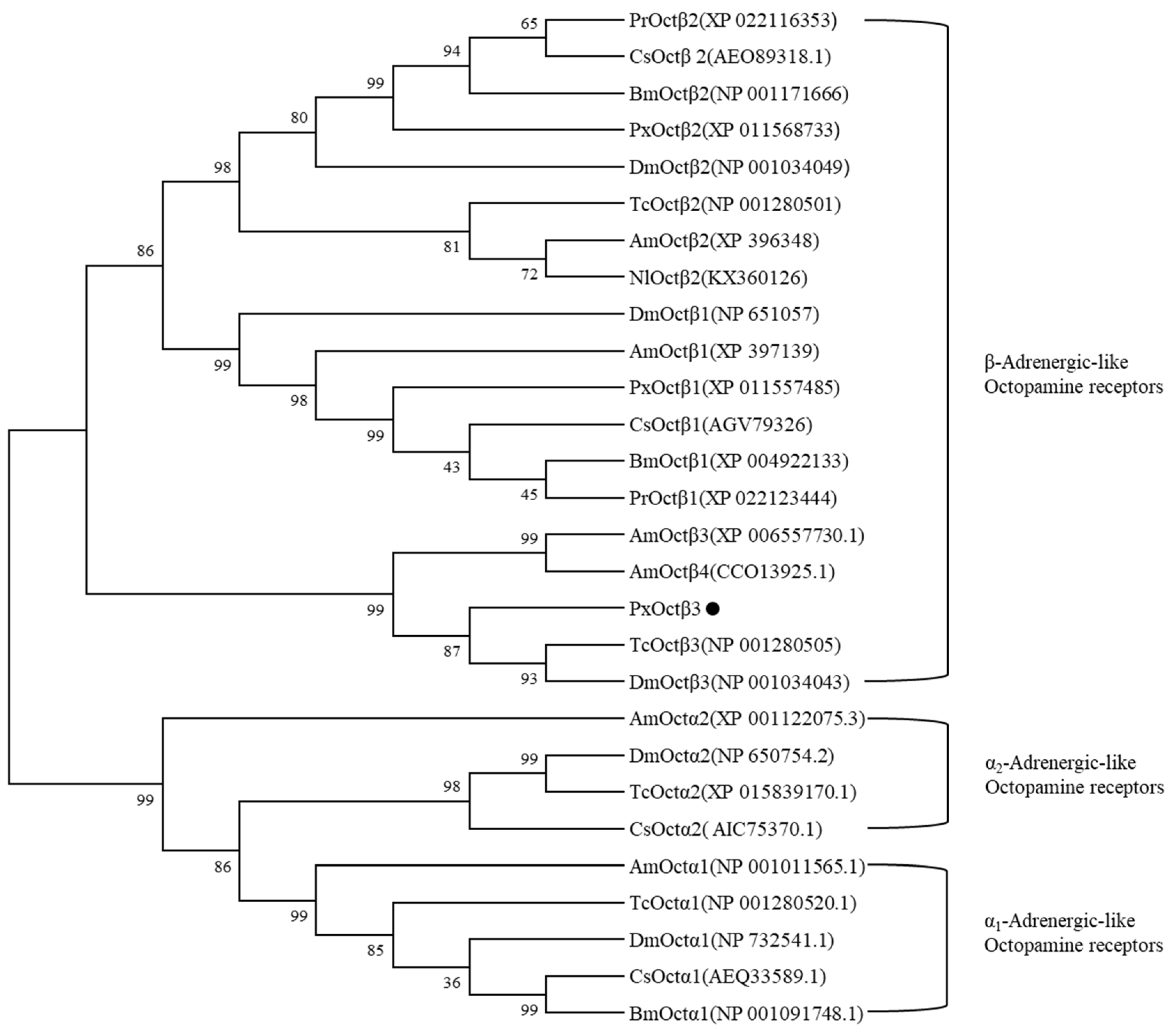
3.1. Molecular Cloning and Sequence Analysis
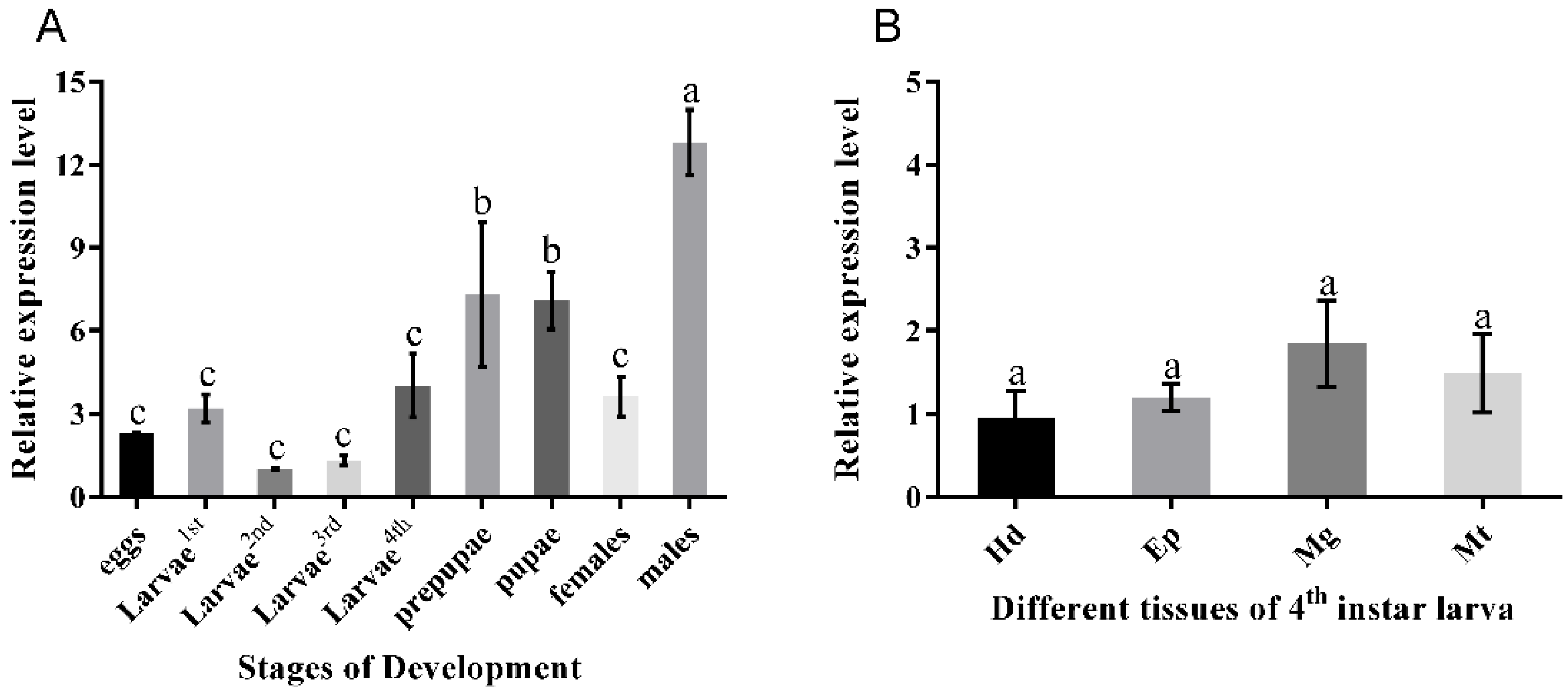
3.2. Pattern of Expression of PxOctβ3
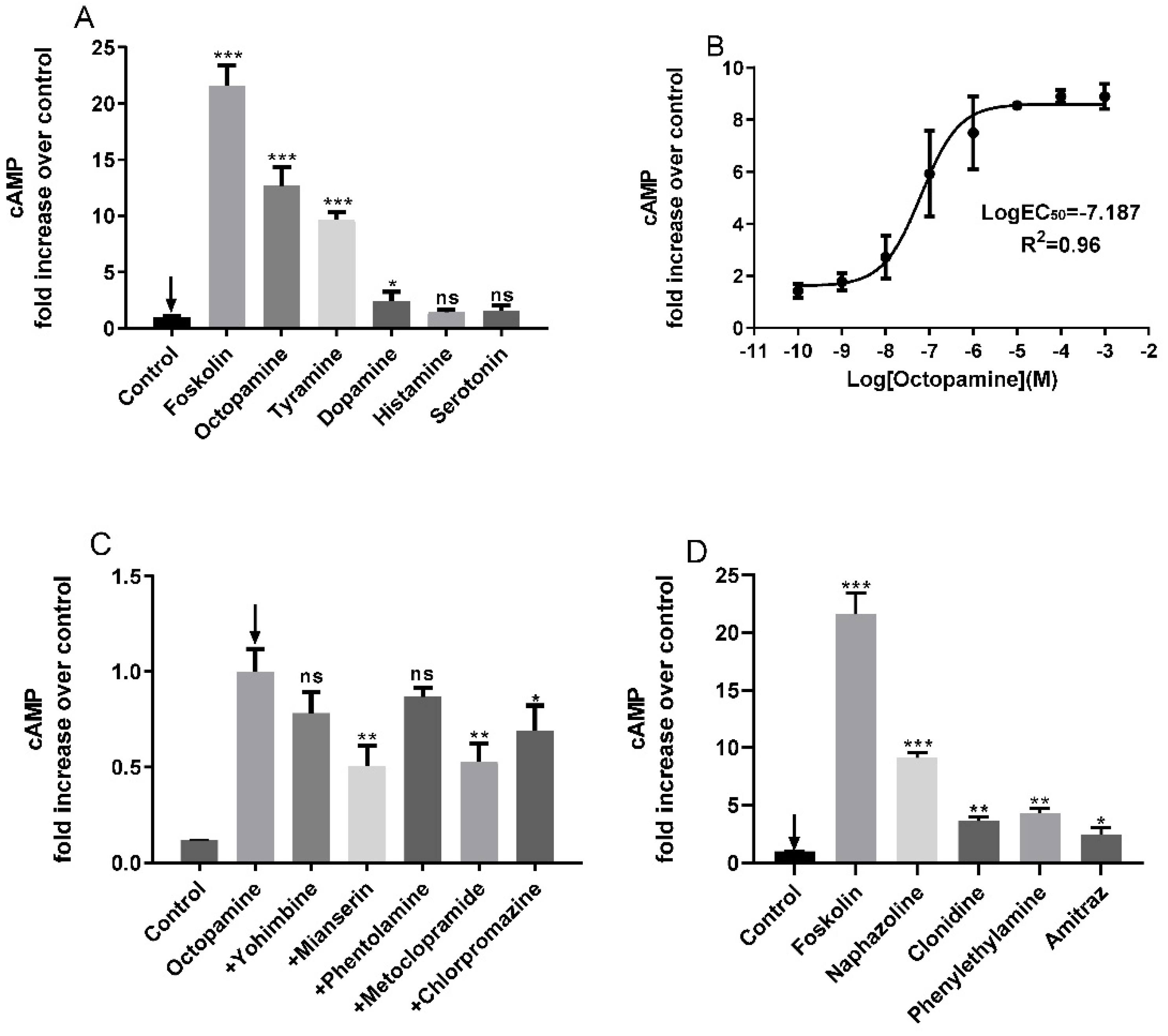
3.3. Pharmacological Property Analysis of PxOctβ3
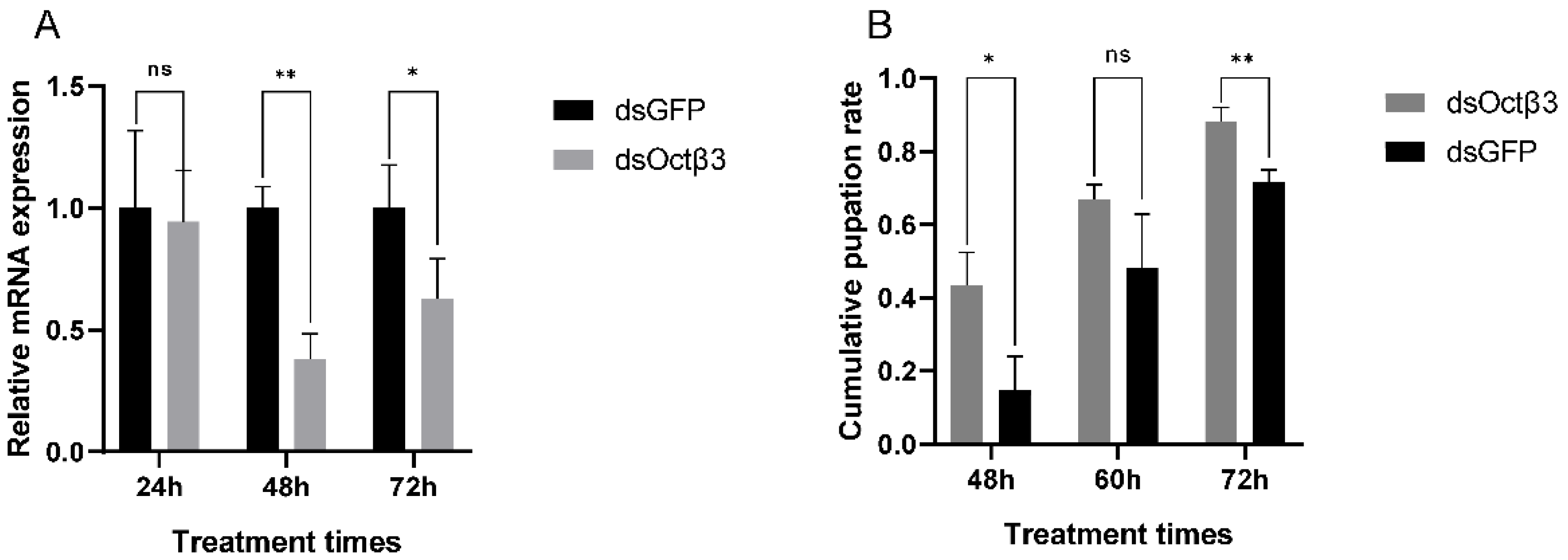
3.4. RNA Interference and Pupation Rate Determination after RNAi
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, S.; Zhang, X.; Shen, J.; Li, D.; Wan, H.; You, H.; Li, J. Cross-resistance and biochemical mechanisms of resistance to indoxacarb in the diamondback moth Plutella xylostella. Pestic. Biochem. Physiol. 2017, 140, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Li, D.; Zhang, S.; Zhu, X.; Wan, H.; Li, J. Fitness and inheritance of metaflumizone resistance in Plutella xylostella. Pestic. Biochem. Physiol. 2017, 139, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.A.; Garratt, M.P.; Wickens, J.B.; Wickens, V.J.; Potts, S.G.; Raine, N.E. Neonicotinoid pesticide exposure impairs crop pollination services provided by bumblebees. Nature 2015, 528, 548–550. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.R.; Muscedere, M.L.; Seid, M.A.; Traniello, J.F.A.; Hughes, W.O.H. Biogenic amines are associated with worker task but not patriline in the leaf-cutting ant Acromyrmex echinatior. J. Comp. Physiol. A 2013, 199, 1117–1127. [Google Scholar] [CrossRef]
- Roeder, T. The control of metabolic traits by octopamine and tyramine in invertebrates. J. Exp. Biol. 2020, 223, jeb194282. [Google Scholar] [CrossRef]
- Rosenbaum, D.M.; Rasmussen, S.G.F.; Kobilka, B.K. The structure and function of G-protein-coupled receptors. Nature 2009, 459, 356–363. [Google Scholar] [CrossRef]
- Evans, P.D.; Maqueira, B. Insect octopamine receptors: A new classification scheme based on studies of cloned Drosophila G-protein coupled receptors. Invertebr. Neurosci. 2005, 5, 111–118. [Google Scholar] [CrossRef]
- Farooqui, T.; Robinson, K.; Vaessin, H.; Smith, B.H. Modulation of early olfactory processing by an octopaminergic reinforcement pathway in the honeybee. J. Neurosci. 2003, 23, 5370–5380. [Google Scholar] [CrossRef]
- Schwaerzel, M.; Monastirioti, M.; Scholz, H.; Friggi-Grelin, F.; Birman, S.; Heisenberg, M. Dopamine and octopamine differentiate between aversive and appetitive olfactory memories in Drosophila. J. Neurosci. 2003, 23, 10495–10502. [Google Scholar] [CrossRef]
- Ohta, H.; Ozoe, Y. Chapter two-molecular signalling, pharmacology, and physiology of octopamine and tyramine receptors as potential insect pest control targets. Adv. Insect Physiol. 2014, 46, 73–166. [Google Scholar]
- Wong, J.Y.H.; Wan, B.A.; Bland, T.; Montagnese, M.; McLachlan, A.D.; O’Kane, C.J.; Zhang, S.W.; Masuda-Nakagawa, L.M. Octopaminergic neurons have multiple targets in Drosophila larval mushroom body calyx and can modulate behavioral odor discrimination. Learn. Mem. 2021, 28, 53–71. [Google Scholar] [CrossRef] [PubMed]
- Kita, T.; Hayashi, T.; Ohtani, T.; Takao, H.; Takasu, H.; Liu, G.; Ohta, H.; Ozoe, F.; Ozoe, Y. Amitraz and its metabolite differentially activate α- and β-adrenergic-like octopamine receptors. Pest Manag. Sci. 2017, 73, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Ozoe, Y. Ion channels and G protein-coupled receptors as targets for invertebrate pest control: From past challenges to practical insecticides. Biosci. Biotechnol. Biochem. 2021, 85, 1563–1571. [Google Scholar] [CrossRef]
- Blenau, W.; Wilms, J.A.; Balfanz, S.; Baumann, A. AmOctα2R: Functional Characterization of a Honeybee Octopamine Receptor Inhibiting Adenylyl Cyclase Activity. Int. J. Mol. Sci. 2020, 21, 9334. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.X.; Xu, G.; Gu, G.X.; Mao, F.; Ye, G.Y.; Liu, W.; Huang, J. A new Drosophila octopamine receptor respondsto serotonin. Insect Biochem. Mol. Biol. 2017, 90, 61–70. [Google Scholar] [CrossRef]
- Wu, S.F.; Xu, G.; Qi, Y.X.; Xia, R.Y.; Huang, J.; Ye, G.Y. Two splicing variants of a novel family of octopamine receptors with different signaling properties. J. Neurochem. 2014, 129, 37–47. [Google Scholar] [CrossRef]
- Blenau, W.; Baumann, A. Octopaminergic and Tyraminergic Signaling in the Honeybee (Apis mellifera) Brain. In Trace Amines and Neurological Disorders; Academic Press: Cambridge, MA, USA, 2016; pp. 203–219. [Google Scholar]
- Wu, S.F.; Yao, Y.; Huang, J.; Ye, G.Y. Characterization of a β-adrenergic-like octopamine receptor from the rice stem borer (Chilo suppressalis). J. Exp. Biol. 2012, 215 Pt 15, 2646–2652. [Google Scholar] [CrossRef]
- Evans, P.D.; Robb, S. Octopamine receptor subtypes and their modes of action. Neurochem. Res. 1993, 18, 869–874. [Google Scholar] [CrossRef]
- Roeder, T.; Seifert, M.; Kähler, C.; Gewecke, M. Tyramine and octopamine: Antagonistic modulators of behaviour and metabolism. Arch. Insect Biochem. Physiol. 2003, 54, 1–13. [Google Scholar] [CrossRef]
- Maqueira, B.; Chatwin, H.; Evans, P.D. Identification and characterization of a novel family of Drosophila β-adrenergic-like octopamine G-protein coupled receptors. J. Neurochem. 2005, 94, 547–560. [Google Scholar] [CrossRef]
- Balfanz, S.; Strünker, T.; Frings, S.; Baumann, A. A family of octopamine [corrected] receptors that specifically induce cyclic AMP production or Ca2+ release in Drosophila melanogaster. J. Neurochem. 2005, 93, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Balfanz, S.; Jordan, N.; Langenstück, T.; Breuer, J.; Bergmeier, V.; Baumann, A. Molecular, pharmacological, and signaling properties of octopamine receptors from honeybee (Apis mellifera) brain. J. Neurochem. 2014, 129, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fink, C.; El-Kholy, S.; Roeder, T. The octopamine receptor octβ2R is essential for ovulation and fertilization in the fruit fly Drosophila melanogaster. Arch. Insect Biochem. Physiol. 2015, 88, 168–178. [Google Scholar] [CrossRef]
- Wu, S.F.; Jv, X.M.; Li, J.; Xu, G.J.; Cai, X.Y.; Gao, C.F. Pharmacological characterization and functional roles for egg-laying of a β-adrenergic-like octopamine receptor in the brown planthopper Nilaparvata lugens. Insect Biochem. Mol. Biol. 2017, 87, 55–64. [Google Scholar] [CrossRef]
- Sujkowski, A.; Gretzinger, A.; Soave, N.; Todi, S.V.; Wessells, R. Alpha-and beta-adrenergic octopamine receptors in muscle and heart are required for Drosophila exercise adaptations. PLoS Genet. 2020, 16, e1008778. [Google Scholar] [CrossRef]
- Ohhara, Y.; Shimada-Niwa, Y.; Niwa, R.; Kayashima, Y.; Hayashi, Y.; Akagi, K.; Ueda, H.; Yamakawa-Kobayashi, K.; Kobayashi, S. Autocrine regulation of ecdysone synthesis by β3-octopamine receptor in the prothoracic gland is essential for Drosophila metamorphosis. Proc. Natl. Acad. Sci. USA 2015, 112, 1452–1457. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.T.; Ma, H.H.; Deng, X.L.; Zhu, H.; Liu, J.; Zhou, Y.; Zhou, X.M. Pharmacological characterization of a β-adrenergic-like octopamine receptor in Plutella xylostella. Arch. Insect Biochem. Physiol. 2018, 98, e21466. [Google Scholar] [CrossRef]
- Zheng, L.S.; Liu, X.Q.; Liu, G.G.; Huang, Q.Q.; Wang, J.J.; Jiang, H.B. Knockdown of a β-Adrenergic-Like Octopamine Receptor Affects Locomotion and Reproduction of Tribolium castaneum. Int. J. Mol. Sci. 2021, 22, 7252. [Google Scholar] [CrossRef]
- Deng, X.L.; Guo, L.; Ma, H.H.; Hu, X.P.; Zhou, X.M. Phenyl imidazolidin-2-ones antagonize a β-adrenergic-like octopamine receptor in diamondback moth (Plutella xylostella). Pest Manag. Sci. 2021, 77, 3224–3232. [Google Scholar] [CrossRef]
- Blenau, W.; Bremer, A.S.; Schwietz, Y.; Friedrich, D.; Ragionieri, L.; Predel, R.; Balfanz, S.; Baumann, A. PaOctβ2R: Identification and Functional Characterization of an Octopamine Receptor Activating Adenylyl Cyclase Activity in the American Cockroach Periplaneta americana. Int. J. Mol. Sci. 2022, 23, 1677. [Google Scholar] [CrossRef]
- Audsley, N.; Down, R.E. G protein coupled receptors as targets for next generation pesticides. Insect Biochem. Mol. Biol. 2015, 67, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Vogel, C.F. The role of octopamine receptor agonists in the synergistic toxicity of certain insect growth regulators (IGRs) in controlling Dengue vector Aedes aegypti (Diptera: Culicidae) mosquito. Acta Trop. 2016, 155, 1–5. [Google Scholar] [CrossRef]
- Birgül Iyison, N.; Shahraki, A.; Kahveci, K.; Düzgün, M.B.; Gün, G. Are insect GPCRs ideal next-generation pesticides: Opportunities and challenges. FEBS J. 2021, 288, 2727–2745. [Google Scholar] [CrossRef] [PubMed]
- Grohmann, L.; Blenau, W.; Erber, J.; Ebert, P.R.; Strünker, T.; Baumann, A. Molecular and functional characterization of an octopamine receptor from honeybee (Apis mellifera) brain. J. Neurochem. 2003, 86, 725–735. [Google Scholar] [CrossRef]
- Chen, X.; Ohta, H.; Ozoe, F.; Miyazawa, K.; Huang, J.; Ozoe, Y. Functional and pharmacological characterization of a β-adrenergic-like octopamine receptor from the silkworm Bombyx mori. Insect Biochem. Mol. Biol. 2010, 40, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Fan, X.Y.; Qiao, X.; Montell, C.; Huang, J. An octopamine receptor confers selective toxicity of amitraz on honeybees and Varroa mites. Elife 2021, 10, e68268. [Google Scholar] [CrossRef]
- Huang, J.; Hamasaki, T.; Ozoe, Y. Pharmacological characterization of a Bombyx mori alpha-adrenergic-like octopamine receptor stably expressed in a mammalian cell line. Arch Insect Biochem Physiol. 2010, 73, 74–86. [Google Scholar] [CrossRef]
- Liu, X.; Jiang, H.; Li, H.; Xiong, Y.; Wang, J. The cDNA Cloning, Expression Profiling and Functional Characterization of Octopamine Receptor 3(TcOctβR3) in Tribolium castaneum. Sci. Agric. Sin. 2018, 51, 1315–1324. [Google Scholar]
- El-Kholy, S.; Stephano, F.; Li, Y.; Bhandari, A.; Fink, C.; Roeder, T. Expression analysis of octopamine and tyramine receptors in Drosophila. Cell Tissue Res. 2015, 361, 669–684. [Google Scholar] [CrossRef]
- Hunter, J.S.; Baggott, D.; Everett, W.R.; Fourie, J.J.; Cramer, L.G.; Yoon, S.S.; Collidor, N.; Mallouk, Y.; Lee, L.; Blair, J.; et al. Efficacy of a novel topical combination of fipronil, amitraz and (S)-methoprene for treatment and control of induced infestations of brown dog ticks (Rhipicephalus sanguineus) on dogs. Vet. Parasitol. 2011, 179, 318–323. [Google Scholar] [CrossRef]
- Martel, A.C.; Zeggane, S.; Aurières, C.; Drajnudel, P.; Faucon, J.P.; Aubert, M. Acaricide residues in honey and wax after treatment of honeybee colonies with Apivar or Asuntol50. Apidologie 2007, 38, 534–544. [Google Scholar] [CrossRef]
- Hellmann, K.; Adler, K.; Parker, L.; Pfister, K.; Delay, R.L.; Rugg, D. Evaluation of the efficacy and safety of a novel formulation of metaflumizone plus amitraz in dogs naturally infested with fleas and ticks in Europe. Vet. Parasitol. 2007, 150, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Pophof, B. Octopamine enhances moth olfactory responses to pheromones, but not those to general odorants. J. Comp. Physiol. A 2002, 188, 659–662. [Google Scholar]
- Zhang, Y.J.; Jiang, L.; Ahamd, S.; Chen, Y.; Zhang, J.Y.; Stanley, D.; Miao, H.; Ge, L.Q. The octopamine receptor, OA2B2, modulates stress resistance and reproduction in Nilaparvata lugens Stål (Hemiptera: Delphacidae). Insect Mol. Biol. 2022, 31, 33–48. [Google Scholar] [CrossRef] [PubMed]
- Terenius, O.; Papanicolaou, A.; Garbutt, J.S.; Eleftherianos, I.; Huvenne, H.; Kanginakudru, S.; Albrechtsen, M.; An, C.; Aymeric, J.L.; Barthel, A.; et al. RNA interference in Lepidoptera: An overview of successful and unsuccessful studies and implications for experimental design. J. Insect Physiol. 2011, 57, 231–245. [Google Scholar] [CrossRef]
| Primer Name | Primer Sequences | Purpose of Primers |
|---|---|---|
| Octβ3R_XhoI_F | CTCGAGCCACCATGACCCATGGCACGCTGC | DNA cloning |
| Octβ3R_PstI_R | TCTCTATATACTGCAGCACAAACTGTGC | |
| Octβ3_qF | CGTCTAGCGTATCGTTTTGGAT | Quantitative RT-PCR |
| Octβ3_qR | GTGGAGGTAGTGTGAATGGTGT | |
| Rpl32_qF | TGCCCAACATTGGTTACGG | |
| Rpl32_qR | ACGATGGCCTTGCGCTTC | |
| dsOctβ3_F | TAATACGACTCACTATAGGGCCATCGTCAGACCCTTAG | RNAi |
| dsOctβ3_R | TAATACGACTCACTATAGGCGTGCTGTAGTTCGGACTTAT | |
| Octβ3_ygF | CTGCCCTTCTTCCTGTGG | |
| Octβ3_ygR | GTGCGTTATTAGCGTCCTT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, H.; Liu, Z.; Ma, H.; Zheng, W.; Liu, J.; Zhou, Y.; Man, Y.; Zhou, X.; Zeng, A. Pharmacological Properties and Function of the PxOctβ3 Octopamine Receptor in Plutella xylostella (L.). Insects 2022, 13, 735. https://doi.org/10.3390/insects13080735
Zhu H, Liu Z, Ma H, Zheng W, Liu J, Zhou Y, Man Y, Zhou X, Zeng A. Pharmacological Properties and Function of the PxOctβ3 Octopamine Receptor in Plutella xylostella (L.). Insects. 2022; 13(8):735. https://doi.org/10.3390/insects13080735
Chicago/Turabian StyleZhu, Hang, Zheming Liu, Haihao Ma, Wei Zheng, Jia Liu, Yong Zhou, Yilong Man, Xiaomao Zhou, and Aiping Zeng. 2022. "Pharmacological Properties and Function of the PxOctβ3 Octopamine Receptor in Plutella xylostella (L.)" Insects 13, no. 8: 735. https://doi.org/10.3390/insects13080735
APA StyleZhu, H., Liu, Z., Ma, H., Zheng, W., Liu, J., Zhou, Y., Man, Y., Zhou, X., & Zeng, A. (2022). Pharmacological Properties and Function of the PxOctβ3 Octopamine Receptor in Plutella xylostella (L.). Insects, 13(8), 735. https://doi.org/10.3390/insects13080735





