Plant-Rich Field Margins Influence Natural Predators of Aphids More Than Intercropping in Common Bean
Abstract
:Simple Summary
Abstract
1. Introduction
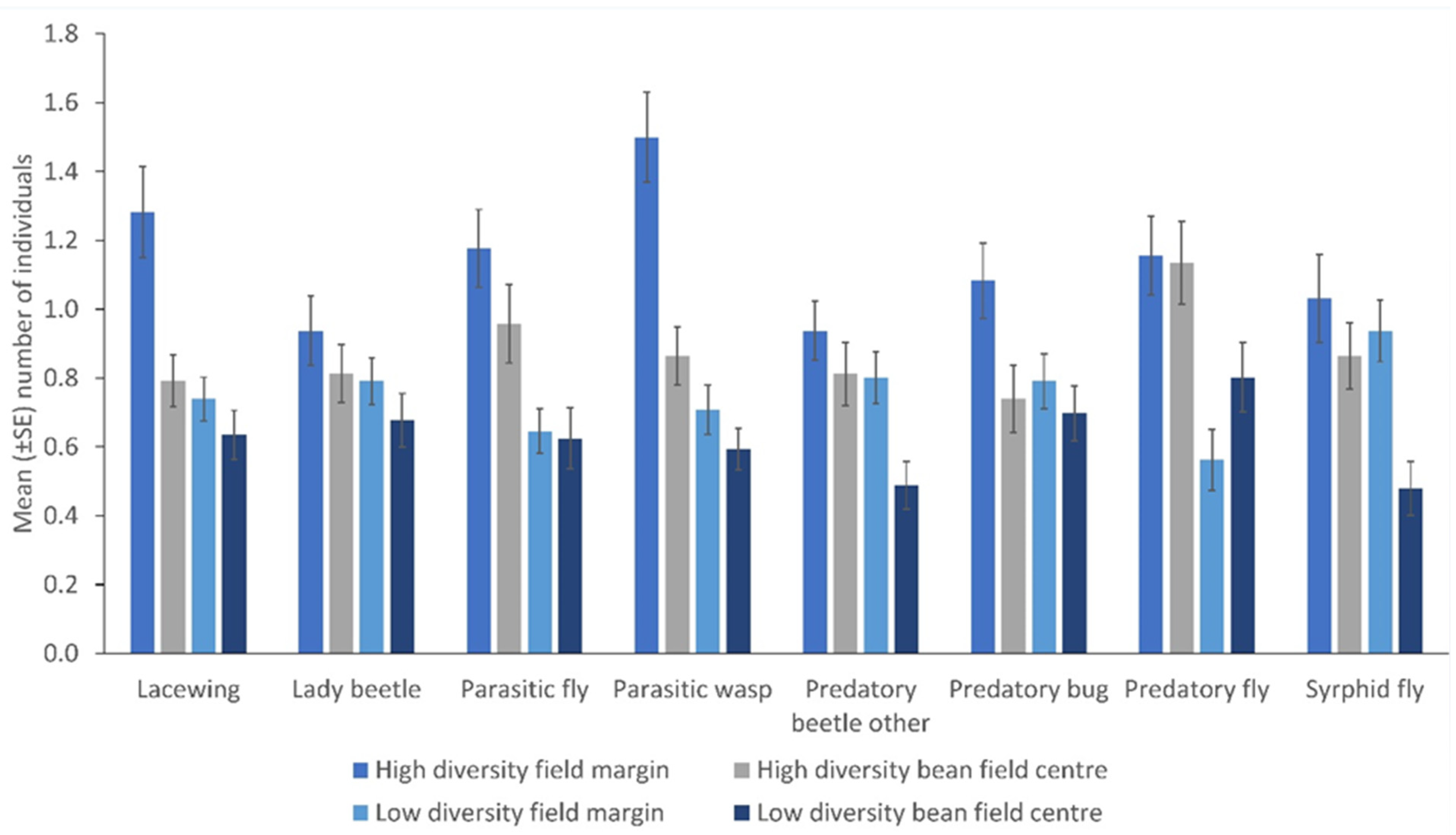
- Plant-rich field margins influence the number of NEs in bean fields
- An increase in NEs assemblage in field margins influence their numbers within the crop.
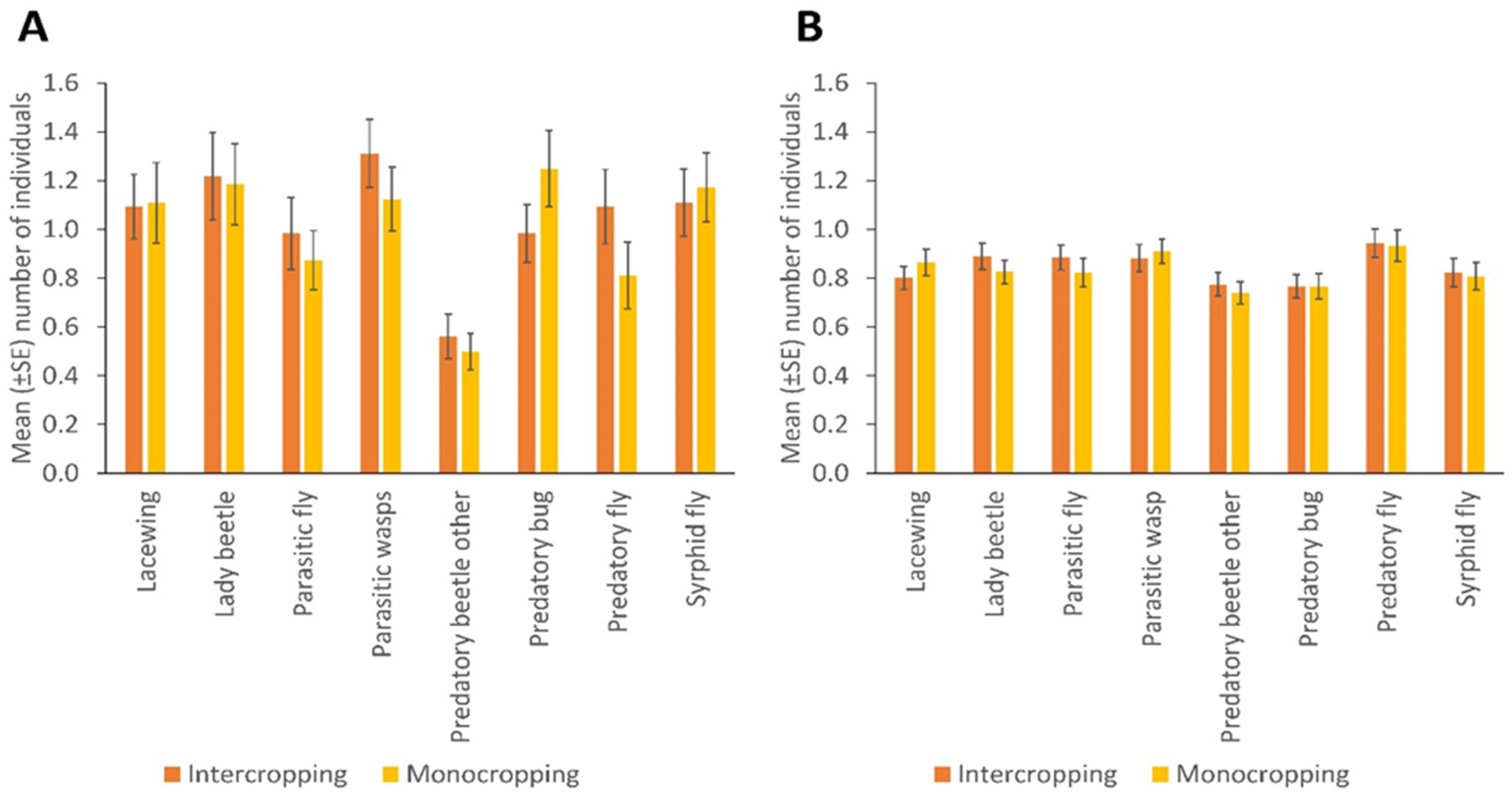
- Intercropping in bean fields is associated with high populations of NEs compared to monocropping.
2. Materials and Methods
2.1. Sampling Natural Enemies from the Fields
- pi—Proportion (n/N) of individuals of particular species in a whole community,
- n—individuals of a particular species,
- N—total number of individuals found,
- ∑—Sum symbol,
- ln = natural logarithm to base e.
2.2. Estimation of Aphid Severity
2.3. Statistical Analysis
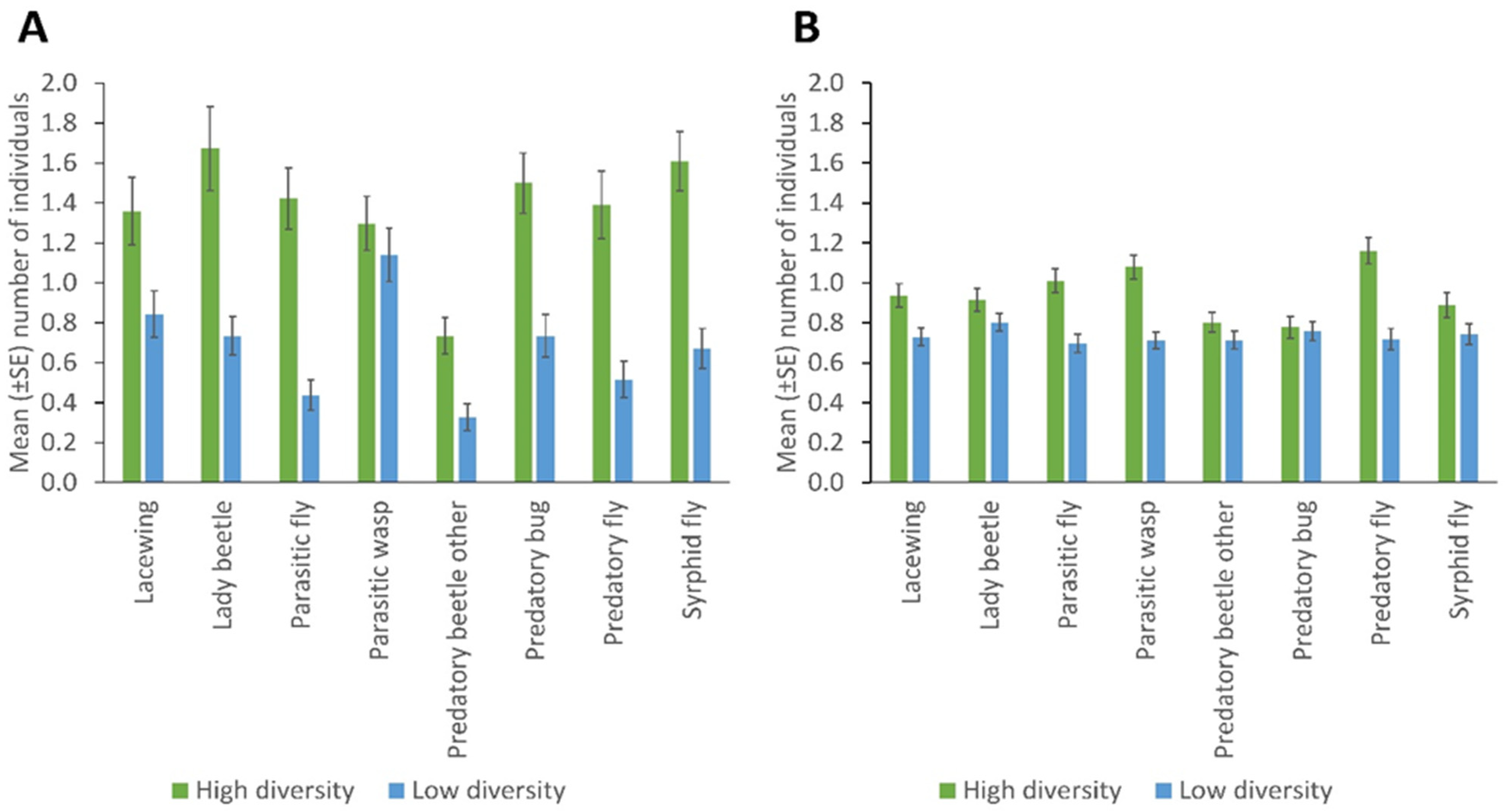
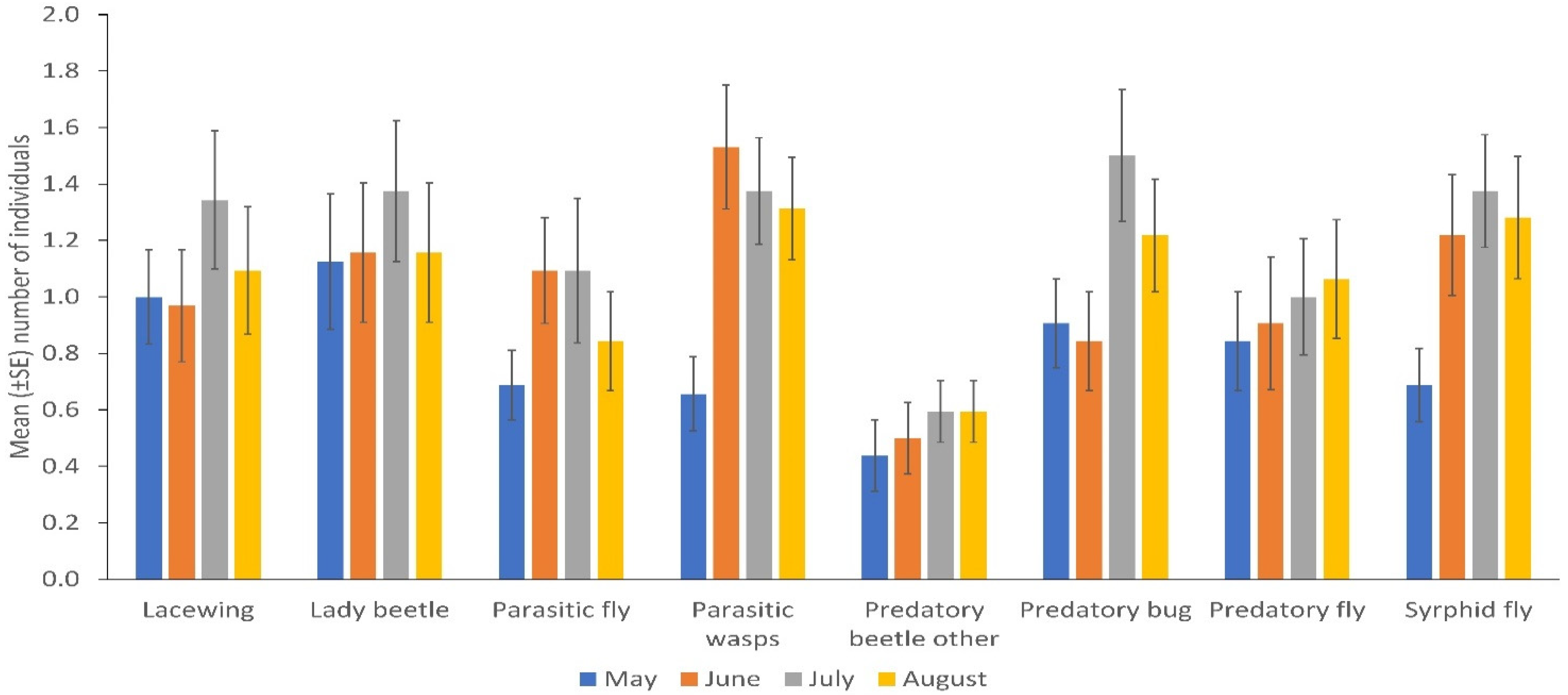
3. Results
Spatial and Temporal Distribution of Natural Enemies in Bean Fields
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Broughton, W.J.; Hernandez, G.; Blair, M.; Beebe, S.; Gepts, P.; Vanderleyden, J. Beans (Phaseolus spp.)–Model food legumes. Plant Soil 2003, 252, 55–128. [Google Scholar] [CrossRef]
- Nedumaran, S.; Abinaya, P.; Jyosthnaa, P.; Shraavya, B.; Rao, P.P.; Bantilan, M.C.S. Grain Legumes Production, Consumption and Trade Trends in Developing Countries; Working Paper Series No. 60; International Crops Research Institute for the Semi-Arid Tropics: Patancheru, India, 2015; p. 64. [Google Scholar]
- Hillocks, R.J.; Madata, C.S.; Chirwa, R.; Minja, E.M.; Msolla, S. Phaseolus bean improvement in Tanzania, 1959–2005. Euphytica 2006, 150, 215–231. [Google Scholar] [CrossRef]
- Abate, T.; Ampofo, J.K. Insect pests of beans in Africa: Their ecology and management. Annu. Rev. Entomol. 1996, 41, 45–73. [Google Scholar] [CrossRef] [PubMed]
- Karel, A.K.; Autrique, A. Insects and other pests in Africa. In Bean Production Problems in the Tropics; Schwartz, H.F., Pastor-Corrales, M.A., Eds.; Centro Internacional de Agricultura Tropical (CIAT): Cali, Colombia, 1989; pp. 455–504. [Google Scholar]
- Nyiira, Z.M. Pests of grain legumes and their control in Uganda. In Pests of Grain Legumes: Ecology and Control; Taylor, T.A., Singh, S.R., van Emden, H.F., Eds.; Academic Press: London, UK, 1978; pp. 118–121. [Google Scholar]
- Swaine, G. Studies on the biology and control of pests of seed beans (Phaseolus vulgaris) in northern Tanzania. Bull. Entomol. Res. 1969, 59, 323–338. [Google Scholar] [CrossRef]
- Wamonje, F.O.; Donnelly, R.; Tungadi, T.D.; Murphy, A.M.; Pate, A.E.; Woodcock, C.; Caulfield, J.; Mutuku, J.M.; Bruce, T.J.; Gilligan, C.A.; et al. Different plant viruses induce changes in feeding behavior of specialist and generalist aphids on common bean that are likely to enhance virus transmission. Front. Plant. Sci. 2020, 10, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Belmain, S.R.; Haggar, J.; Holt, J.; Stevenson, P.C. Managing Legume Pests in Sub-Saharan Africa: Challenges and Prospects for Improving Food Security and Nutrition through Agro-Ecological Intensification; Natural Resources Institute: London, UK, 2013; pp. 1–34. [Google Scholar]
- Otieno, M.; Steffan-Dewenter, I.; Potts, S.G.; Kinuthia, W.; Kasina, M.J.; Garratt, M.P. Enhancing legume crop pollination and natural pest regulation for improved food security in changing African landscapes. Glob. Food Secur. 2020, 26, 100394. [Google Scholar] [CrossRef]
- Mkenda, P.A.; Ndakidemi, P.A.; Stevenson, P.C.; Arnold, S.E.; Darbyshire, I.; Belmain, S.R.; Priebe, J.; Johnson, A.C.; Tumbo, J.; Gurr, G.M. Knowledge gaps among smallholder farmers hinder adoption of conservation biological control. Biocontrol. Sci. Technol. 2020, 30, 256–277. [Google Scholar] [CrossRef]
- El-Heneidy, A.H.; Khidr, A.A.; Taman, A.A. Side-effects of insecticides on non-target organisms: 1-In Egyptian cotton fields. Egypt. J. Biol. Pest Control 2015, 25, 685–690. [Google Scholar]
- Gill, H.K.; Garg, H. Pesticide: Environmental impacts and management strategies. Pestic.-Toxic Asp. 2014, 8, 187. [Google Scholar] [CrossRef] [Green Version]
- Rice, P.J.; Arthur, E.L.; Barefoot, A.C. Advances on environmental fate and exposures assessments. J. Agric. Food Chem. 2007, 55, 5367–5376. [Google Scholar] [CrossRef]
- Tamò, M.; Srinivasan, R.; Dannon, E.; Agboton, C.; Datinon, B.; Dabiré, C.; Baoua, I.; Ba, M.; Haruna, B.; Pittendrigh, B. Biological control: A major component for the long-term cowpea pest management strategy. In Improving Livelihoods in the Cowpea Value Chain through Advancements in Science; Proceedings of the 5th World Cowpea Research Conference, Dakar, Senegal, 27 September–1 October 2010; Boukar, O., Coulibaly, C., Fatokun, C., Lopez, K., Tamò, M., Eds.; International Institute of Tropical Agriculture: Ibadan, Nigeria, 2012; pp. 249–259. [Google Scholar]
- Ballal, C.R.; Verghese, A. Role of parasitoids and predators in the management of insect pests. In New Horizons in Insect Science: Towards Sustainable Pest Management; Springer: New Delhi, India, 2015; pp. 307–326. [Google Scholar]
- Gurr, G.M.; Wratten, S.D.; Altieri, M.A. Ecological engineering: A new direction for agricultural pest management. AFBM J. 2004, 1, 28–35. [Google Scholar] [CrossRef]
- Amoabeng, B.W.; Gurr, G.M.; Gitau, C.W.; Stevenson, P.C. Cost: Benefit analysis of botanical insecticide use in cabbage: Implications for smallholder farmers in developing countries. Crop Prot. 2014, 57, 71–76. [Google Scholar] [CrossRef]
- Arnold, S.E.; Elisante, F.; Mkenda, P.A.; Tembo, Y.L.; Ndakidemi, P.A.; Gurr, G.M.; Darbyshire, I.A.; Belmain, S.R.; Stevenson, P.C. Beneficial insects are associated with botanically rich margins with trees on small farms. Sci. Rep. 2021, 11, 15190. [Google Scholar] [CrossRef] [PubMed]
- Balzan, M.V.; Bocci, G.; Moonen, A.C. Landscape complexity and field margin vegetation diversity enhance natural enemies and reduce herbivory by Lepidoptera pests on tomato crop. BioControl 2016, 61, 141–154. [Google Scholar] [CrossRef]
- Dyer, L.E.; Landis, D.A. Influence of noncrop habitats on the distribution of Eriborus terebrans (Hymenoptera: Ichneumonidae) in cornfields. Environ. Entomol. 1997, 26, 924–932. [Google Scholar] [CrossRef]
- Mkenda, P.A.; Ndakidemi, P.A.; Stevenson, P.C.; Arnold, S.E.; Belmain, S.R.; Chidege, M.; Gurr, G.M. Field margin vegetation in tropical African bean systems harbours diverse natural enemies for biological pest control in adjacent crops. Sustainability 2019, 11, 6399. [Google Scholar] [CrossRef] [Green Version]
- Tschumi, M.; Albrecht, M.; Entling, M.H.; Jacot, K. High effectiveness of tailored flower strips in reducing pests and crop plant damage. Proc. R. Soc. B Biol. Sci. 2015, 282, 1814. [Google Scholar] [CrossRef] [Green Version]
- Tscharntke, T.; Karp, D.S.; Chaplin-kramer, R.; Batáry, P.; Declerck, F.; Gratton, C.; Hunt, L.; Ives, A.R.; Jonsson, M.; Larsen, A.E.; et al. When natural habitat fails to enhance biological pest control–Five hypotheses. Biol. Conserv. 2002, 204, 449–458. [Google Scholar] [CrossRef] [Green Version]
- Chaplin-Kramer, R.; O’Rourke, M.E.; Blitzer, E.J.; Kremen, C. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecol. Lett. 2011, 14, 922–932. [Google Scholar] [CrossRef]
- González, E.; Štrobl, M.; Janšta, P.; Hovorka, T.; Kadlec, T.; Knapp, M. Artificial temporary non-crop habitats support parasitoids on arable land. Biol. Conserv. 2022, 265, 109409. [Google Scholar] [CrossRef]
- Baggen, L.R.; Gurr, G.M. The influence of food on Copidosoma koehleri (hymenoptera: Encyrtidae), and the use of flowering plants as a habitat management tool to enhance biological control of potato moth, Phthorimaea operculella (lepidoptera: Gelechiidae). Biol. Control 1998, 11, 9–17. [Google Scholar] [CrossRef]
- Long, R.; Corbett, A.; Lamb, C.; Reberg-Horton, C.; Chandler, J.; Stimmann, M. Beneficial insects move from flowering plants to nearby crops. Calif. Agric. 1998, 52, 23–26. [Google Scholar] [CrossRef] [Green Version]
- Thies, C.; Tscharntke, T. Landscape structure and biological control in agroecosystems. Science 1999, 285, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Landis, D.A.; Wratten, S.D.; Gurr, G.M. Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu. Rev. Entomol. 2000, 45, 175–201. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, C.I.; Parrella, M.; Altieri, M.A. The effects of a vegetational corridor on the abundance and dispersal of insect biodiversity within a northern California organic vineyard. Landsc. Ecol. 2001, 16, 133–146. [Google Scholar] [CrossRef]
- Tylianakis, J.; Veddeler, D.; Lozada, T.; Lopez, R.M.; Benítez, P.; Klein, A.M.; De Koning, G.H.J.; Olschewski, R.; Veldkamp, E.; Navarrete, H.; et al. Biodiversity of land-use systems in coastal Ecuador and bioindication using trap-nesting bees, wasps, and their natural enemies. Lyonia 2004, 6, 7–15. [Google Scholar]
- Pywell, R.F.; James, K.L.; Herbert, I.; Meek, W.R.; Carvell, C.; Bell, D.; Sparks, T.H. Determinants of overwintering habitat quality for beetles and spiders on arable farmland. Biol. Conserv. 2005, 123, 79–90. [Google Scholar] [CrossRef]
- Bianchi, F.J.; Booij, C.J.H.; Tscharntke, T. Sustainable pest regulation in agricultural landscapes: A review on landscape composition, biodiversity and natural pest control. Proc. R. Soc. B Biol. Sci. 2006, 273, 1715–1727. [Google Scholar] [CrossRef] [Green Version]
- Olson, D.M.; Wäckers, F.L. Management of field margins to maximize multiple ecological services. J. Appl. Ecol. 2007, 44, 13–21. [Google Scholar] [CrossRef]
- Thomson, L.J.; Hoffmann, A.A. Vegetation increases the abundance of natural enemies in vineyards. Biol. Control 2009, 49, 259–269. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, B.; Li, Q.; Sun, M.; Li, M.; Wyckhuys, K.A.; Wang, P.; Lu, Y. Perennial flowering plants sustain natural enemy populations in Gobi Desert Oases of Southern Xinjiang, China. Insects 2022, 13, 399. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, S.K.; Sigsgaard, L.; Johansen, A.B.; Thorup, K.; Per, K. The impact of reduced tillage and distance to field margin on predator functional diversity. J. Insect Conserv. 2022, 26, 491–501. [Google Scholar] [CrossRef]
- Rand, T.A.; Tylianakis, J.M.; Tscharntke, T. Spillover edge effects: The dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats. Ecol. Lett. 2006, 9, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Wäckers, F.L. Assessing the suitability of flowering herbs as parasitoid food sources: Flower attractiveness and nectar accessibility. Biol. Control 2004, 29, 307–314. [Google Scholar] [CrossRef]
- Knapp, M.; Milan, Ř. Even the smallest non-crop habitat islands could be beneficial: Distribution of carabid beetles and spiders in agricultural landscape. PLoS ONE 2015, 10, e0123052. [Google Scholar] [CrossRef]
- Aguilera, G.; Roslin, T.; Miller, K.; Tamburini, G.; Birkhofer, K.; Sandra, B.C.; Öckinger, E.; Rundlöf, M.; Rusch, A.; Smith, H.G.; et al. Crop diversity benefits carabid and pollinator communities in landscapes with semi-natural habitats. J. Appl. Ecol. 2020, 57, 2170–2179. [Google Scholar] [CrossRef]
- Bartual, A.M.; Sutter, L.; Bocci, G.; Moonen, A.C.; Cresswell, J.; Entling, M.; Giffard, B.; Jacot, K.; Jeanneret, P.; Holland, J.; et al. The potential of different semi-natural habitats to sustain pollinators and natural enemies in European agricultural landscapes. Agric. Ecosyst. Environ. 2019, 279, 43–52. [Google Scholar] [CrossRef]
- Kovács, G.; Kaasik, R.; Lof, M.E.; van der Werf, W.; Kaart, T.; Holland, J.M.; Luik, A.; Veromann, E. Effects of land use on infestation and parasitism rates of cabbage seed weevil in oilseed rape. Pest Manag. Sci. 2019, 75, 658–666. [Google Scholar] [CrossRef]
- Tschumi, M.; Albrecht, M.; Collatz, J.; Dubsky, V.; Entling, M.H.; Najar-Rodriguez, A.J.; Jacot, K. Tailored flower strips promote natural enemy biodiversity and pest control in potato crops. J. Appl. Ecol. 2016, 53, 1169–1176. [Google Scholar] [CrossRef] [Green Version]
- Bohinc, T.; Trdan, S. Sowing mixtures of Brassica trap crops is recommended to reduce Phyllotreta beetles injury to cabbage. Acta Agric. Scand.-B Soil Plant Sci. 2013, 63, 297–303. [Google Scholar] [CrossRef]
- Girma, H.; Rao, M.R.; Sithanantham, S. Insect pests and beneficial hedgerow intercropping systems in semiarid Kenya. Agrofor. Syst. 2000, 50, 279–292. [Google Scholar] [CrossRef]
- Tingey, W.M.; Lamont, W.J. Insect abundance in field beans altered by intercropping. Bull. Entomol. Res. 1988, 78, 527–535. [Google Scholar] [CrossRef]
- Munyuli, M.B.T.; Luther, G.C.; Kyamanywa, S. Effects of cowpea cropping systems and insecticides on arthropod predators in Uganda and the Democratic Republic of the Congo. Crop Prot. 2007, 26, 114–126. [Google Scholar] [CrossRef]
- Ndakidemi, B.J.; Mbega, E.R.; Ndakidemi, P.A.; Belmain, S.R.; Arnold, S.E.J.; Woolley, V.C.; Stevenson, P.C. Field margin plants support natural enemies in sub-Saharan Africa smallholder common bean farming systems. Plants 2022, 11, 898. [Google Scholar] [CrossRef]
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; University of Illinois Press: Urbana, IL, USA, 1963; p. 117. [Google Scholar]
- Rebek, E.J.; Sadof, C.S.; Hanks, L.M. Manipulating the abundance of natural enemies in ornamental landscapes with floral resource plants. Biol. Control 2005, 33, 203–216. [Google Scholar] [CrossRef]
- Sutherland, J.P.; Sullivan, M.S.; Poppy, G.M. Distribution and abundance of aphidophagous hoverflies (Diptera: Syrphidae) in wildflower patches and field margin habitats. Agric. For. Entomol. 2001, 3, 57–64. [Google Scholar] [CrossRef]
- Nabirye, J.; Nampala, P.; Ogenga-Latigo, M.W.; Kyamanywa, S.; Wilson, H.; Odeke, V.; Iceduna, C.; Adipala, E. Farmer-participatory evaluation of cowpea integrated pest management (IPM) technologies in Eastern Uganda. Crop Prot. 2003, 22, 31–38. [Google Scholar] [CrossRef]
- Tena, A.; Pekas, A.; Cano, D.; Wäckers, F.L.; Urbaneja, A. Sugar provisioning maximizes the biocontrol service of parasitoids. J. Appl. Ecol. 2015, 52, 795–804. [Google Scholar] [CrossRef]
- Crawley, M.J. The R Book; John Wiley: New York, NY, USA, 2007; pp. 627–685. [Google Scholar]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2018. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 17 June 2022).
- Woolley, V.C.; Tembo, Y.L.B.; Ndakidemi, B.; Obanyi, J.N.; Sarah, E.J.; Belmain, S.R.; Ndakidemi, P.A.; Ogendo, J.O.; Stevenson, P.C. The diversity of aphid parasitoids in East Africa and implications for biological control. Pest Manag. Sci. 2022, 78, 1109–1116. [Google Scholar] [CrossRef]
- Mccravy, K.W. A Review of Sampling and Monitoring Methods for Beneficial Arthropods in Agroecosystems. Insects 2018, 9, 170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakker, L.; van der Werf, W.; Bianchi, F.J.J.A. Sweep netting samples, but not sticky trap samples, indicate beneficial arthropod abundance is negatively associated with landscape wide insecticide use. J. Appl. Ecol. 2022, 59, 942–952. [Google Scholar] [CrossRef]
- Bannerman, J.A.; Costamagna, A.C.; Mccornack, B.P.; Ragsdale, D.W. Comparison of Relative Bias, Precision, and Efficiency of Sampling Methods for Natural Enemies of Soybean Aphid (Hemiptera: Aphididae). J. Econ. Entomol. 2015, 108, 1381–1397. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.D.; Zhao, H.Y.; Fu, B.L.; Han, Y.; Liu, K.; Wu, J.H. Coloured sticky traps to selectively survey thrips in cowpea ecosystem. Neotrop. Entomol. 2016, 45, 96–101. [Google Scholar] [CrossRef]
- Pobozniak, M.; Tokarz, K.; Musynov, K. Evaluation of sticky trap colour for thrips (Thysanoptera) monitoring in pea crops (Pisum sativum L.). J. Plant Dis. Prot. 2020, 127, 307–321. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, P.A. Conservation Biological Control; Academic Press: San Diego, CA, USA, 1998; p. 396. [Google Scholar]
- Martin, E.A.; Dainese, M.; Clough, Y.; Báldi, A.; Bommarco, R.; Gagic, V.; Garratt, M.P.; Holzschuh, A.; Kleijn, D.; Kovács-Hostyánszki, A.; et al. The interplay of landscape composition and configuration: New pathways to manage functional biodiversity and agroecosystem services across Europe. Ecol. Lett. 2019, 22, 1083–1094. [Google Scholar] [CrossRef] [Green Version]
- Vandereycken, A.; Durieux, D.; Joie, E.; Francis, F.; Haubruge, E.; Verheggen, F.J. Aphid species and associated natural enemies in field crops: What about the invasive ladybird Harmonia axyridis (Coleoptera: Coccinellidae)? Entomol. Faun. 2015, 68, 3–15. [Google Scholar]
- Khodeir, I.A.; Khattab, M.A.; Rakha, O.M.; Sharabash, A.S.; Ueno, T.; Mousa, K.M. Population densities of pest aphids and their associated natural enemies on faba bean in Kafr el–Sheikh, Egypt. J. Fac. Agric. Kyushu Univ. 2020, 65, 97–102. [Google Scholar] [CrossRef]
- Pinheiro, F.; Diniz, I.R.; Coelho, D.; Bandeira, M.P.S. Seasonal pattern of insect abundance in the Brazilian cerrado. Austral Ecol. 2002, 27, 132–136. [Google Scholar] [CrossRef]
- Kataria, R.; Kumar, D. Population dynamics of Aphis craccivora (Koch) and its natural enemies on bean crop in relation to weather parameters in Vadodara, Gujarat, India. Legume Res. 2016, 40, 571–579. [Google Scholar] [CrossRef] [Green Version]
- Bommarco, R.; Marini, L.; Vaissière, B.E. Insect pollination enhances seed yield, quality, and market value in oilseed rap. Oecologia 2012, 169, 1025–1032. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, J.G. Nutritional aspects of non-prey foods in the life histories of predaceous Coccinellidae. Biol. Control 2009, 51, 294–305. [Google Scholar] [CrossRef]
- Narbona, E.; Dirzo, R. A reassessment of the function of floral nectar in Croton suberosus (Euphorbiaceae): A reward for plant defenders and pollinators. Am. J. Bot. 2010, 97, 672–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramsden, M.W.; Menéndez, R.; Leather, S.R.; Wäckers, F. Optimizing field margins for biocontrol services: The relative role of aphid abundance, annual floral resources, and overwinter habitat in enhancing aphid natural enemies. Agric. Ecosyst. Environ. 2014, 199, 94–104. [Google Scholar] [CrossRef]
- Tallamy, D.W.; Walsh, E.; Peck, D.C. Revisiting paternal care in the assassin bug, Atopozelus pallens (Heteroptera: Reduviidae). J. Insect Behav. 2004, 17, 431–436. [Google Scholar] [CrossRef]
- Parolin, P.; Bresch, C.; Desneux, N.; Brun, R.; Bout, A.; Boll, R.; Poncet, C. Secondary plants used in biological control: A review. Int. J. Pest Manag. 2012, 58, 91–100. [Google Scholar] [CrossRef]
- Redlich, S.; Martin, E.A.; Wende, B.; Steffan-dewenter, I. Landscape heterogeneity rather than crop diversity mediates bird diversity in agricultural landscapes. PLoS ONE 2018, 13, e0200438. [Google Scholar] [CrossRef] [Green Version]
- Muneret, L.; Auriol, A.; Bonnard, O.; Richart-Cervera, S.; Thiéry, D.; Rusch, A. Organic farming expansion drives natural enemy abundance but not diversity in vineyard- dominated landscapes. Ecol. Evol. 2019, 9, 13532–13542. [Google Scholar] [CrossRef]
- Bakker, L.; van der Werf, W.; Bianchi, F.J. No significant effects of insecticide use indicators and landscape variables on biocontrol in field margins. Agric. Ecosyst. Environ. 2021, 308, 107253. [Google Scholar] [CrossRef]
- Mkenda, P.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.; Mtei, K.; Belmain, S.R. Extracts from field margin weeds provide economically viable and environmentally benign pest control compared to synthetic pesticides. PLoS ONE 2015, 10, e0143530. [Google Scholar] [CrossRef]
- Mkindi, A.; Mpumi, N.; Tembo, Y.; Stevenson, P.C.; Ndakidemi, P.A.; Mtei, K.; Machunda, R.; Belmain, S.R. Invasive weeds with pesticidal properties as potential new crops. Ind. Crop. Prod. 2017, 110, 113–122. [Google Scholar] [CrossRef]
- Tembo, Y.; Mkindi, A.G.; Mkenda, P.A.; Mpumi, N.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.A.; Belmain, S.R. Pesticidal plant extracts improve yield and reduce insect pests on legume crops without harming beneficial arthropods. Front. Plant Sci. 2018, 9, 1425. [Google Scholar] [CrossRef] [PubMed]
- Araya, J.E.; Araya, M.; Guerrero, M.A. Effects of some insecticides applied in sublethal concentrations on the survival and longevity of Aphidius ervi (Haliday) (Hymenoptera: Aphidiidae) adults. Chil. J. Agric. Res. 2010, 70, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.X.; Chen, T.-Y. Effects of the insect growth regulator fenoxycarb on immature Chrysoperla rufilabris (Neuroptera: Chrysopidae). Fla. Entomol. 2001, 84, 628–633. [Google Scholar] [CrossRef]
- Schneider, M.; Smagghe, G.; Pineda, S.; Vinuela, E. Action of insect growth regulator insecticides and spinosad on life-history parameters and absorption in third-instar larvae of the endoparasitoid Hyposoter didymator. Biol. Control 2004, 31, 189–198. [Google Scholar] [CrossRef]
- Shoeb, M.A. Effect of some insecticides on the immature stages of the egg parasitoid Trichogramma evanescens. Egypt. Acad. J. Biol. Sci. 2010, 3, 31–38. [Google Scholar] [CrossRef]
- Thomson, L.; Glenn, D.; Hoffmann, A. Effects of Sulfur on Trichogramma egg parasitoids in vineyards: Measuring toxic effects and establishing release windows. Anim. Prod. Sci. 2001, 40, 1165–1171. [Google Scholar] [CrossRef]
- Golmohammadi, G.; Torshizi, H.R.R.; Vafaei-Shooshtari, R.; Faravardeh, L.; Rafei-Karehroudi, Z. Lethal and sublethal effects of three insecticides on green lacewing, Chrysoperla carnea (Neuroptera: Chrysopidae) under laboratory conditions. J. Entomol. Soc. Iran 2021, 41, 105–121. [Google Scholar] [CrossRef]
- Azimi, S.; Amini, R. Population density of Aphis fabae Scopoli (Hemiptera, Aphididae) and its natural enemies in intercropping of faba bean (Vicia faba L.) and dragonhead (Dracocephalum moldavica L.). J. Biodivers. Environ. Sci. 2015, 6, 380–388. [Google Scholar]
- Birch, N. Field evaluation of resistance to black bean aphid, Aphis fabae, in close relatives of the Faba bean, Vicia faba. Ann. Appl. Biol. 1985, 106, 561–569. [Google Scholar] [CrossRef]
- Karley, A.J.; Parker, W.E.; Pitchford, J.W.; Douglas, A.E. The mid-season crash in aphid populations: Why and how does it occur? Ecol. Entomol. 2004, 29, 383–388. [Google Scholar] [CrossRef]
- Caballero-López, B.; Bommarco, R.; Blanco-Moreno, J.M.; Sans, F.X.; Pujade-Villar, J.; Rundlöf, M.; Smith, H.G. Aphids and their natural enemies are differently affected by habitat features at local and landscape scales. Biol. Control 2012, 63, 222–229. [Google Scholar] [CrossRef]
- Clem, C.S.; Harmon-Threatt, A.N. Field borders provide winter refuge for beneficial predators and parasitoids: A case study on organic farms. J. Insect Sci. 2021, 21, 2. [Google Scholar] [CrossRef] [PubMed]
- Lavandero, B.; Wratten, S.D.; Didham, R.K.; Gurr, G. Increasing floral diversity for selective enhancement of biological control agents: A double-edged sward? Basic Appl. Ecol. 2006, 7, 236–243. [Google Scholar] [CrossRef]
- Nilsson, L.; Klatt, B.K.; Smith, H.G.; Campbell, A.J. Effects of flower-enriched ecological focus areas on functional diversity across scales. Front. Ecol. Evol. 2021, 9, 1–12. [Google Scholar] [CrossRef]
- Samaranayake, K.G.; Costamagna, A.C. Adjacent habitat type affects the movement of predators suppressing soybean aphids. PLoS ONE 2019, 14, e0218522. [Google Scholar] [CrossRef]
- Tiroesele, B.; Obopile, M.; Karabo, O. Insect diversity and population dynamics of natural enemies under sorghum–legume intercrops. Trans. R. Soc. S. Afr. 2019, 74, 258–267. [Google Scholar] [CrossRef]
- Blaauw, B.R.; Isaacs, R. Wildflower plantings enhance the abundance of natural enemies and their services in adjacent blueberry fields. Biol. Control 2015, 91, 94–103. [Google Scholar] [CrossRef] [Green Version]
- Dover, J.W. The Ecology of Hedgerows and Field Margins; Routledge: Abingdon, UK, 2019. [Google Scholar] [CrossRef]
- Li, P.; Kleijn, D.; Badenhausser, I.; Zaragoza-Trello, C.; Gross, N.; Raemakers, I.; Scheper, J. The relative importance of green infrastructure as refuge habitat for pollinators increases with local land-use intensity. J. Appl. Ecol. 2020, 57, 1494–1503. [Google Scholar] [CrossRef]
- Marshall, E.J.; Moonen, A.C. Field margins in northern Europe: Their functions and interactions with agriculture. Agric. Ecosyst. Environ. 2002, 89, 5–21. [Google Scholar] [CrossRef]
- Pywell, R.F.; Heard, M.S.; Woodcock, B.A.; Hinsley, S.; Ridding., L.; Nowakowski, M.; Bullock, J.M. Wildlife-friendly farming increases crop yield: Evidence for ecological intensification. Proc. R. Soc. B Biol. Sci. 2015, 282, 1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Field Location | Mean Number of Natural Enemies (±SEM) | |||||||
|---|---|---|---|---|---|---|---|---|
| Lady Beetle | Syrphid Fly | Lacewing | Parasitic Wasp | Predatory Fly | Parasitic Fly | Other Predatory Beetles | Predatory Bug | |
| Field margin | 0.86 ± 0.06 a | 0.99 ± 0.08 a | 1.01 ±0.08 a | 1.10 ± 0.08 a | 0.86 ± 0.08 a | 0.91± 0.07 a | 0.87 ± 0.06 a | 0.94 ± 0.07 a |
| Crop edge | 0.96 ± 0.07 a | 0.79 ± 0.07 b | 0.78 ±0.06 b | 0.85 ± 0.06 b | 0.99 ± 0.07 a | 0.86± 0.06 a | 0.75 ± 0.06 a | 0.65 ± 0.05 b |
| Field centre | 0.74 ± 0.06 a | 0.67 ± 0.06 b | 1.71 ±0.05 b | 0.73 ± 0.05 b | 0.97 ± 0.08 a | 0.79± 0.07 a | 0.65 ± 0.06 b | 0.72 ± 0.06 ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ndakidemi, B.J.; Mbega, E.R.; Ndakidemi, P.A.; Belmain, S.R.; Arnold, S.E.J.; Woolley, V.C.; Stevenson, P.C. Plant-Rich Field Margins Influence Natural Predators of Aphids More Than Intercropping in Common Bean. Insects 2022, 13, 569. https://doi.org/10.3390/insects13070569
Ndakidemi BJ, Mbega ER, Ndakidemi PA, Belmain SR, Arnold SEJ, Woolley VC, Stevenson PC. Plant-Rich Field Margins Influence Natural Predators of Aphids More Than Intercropping in Common Bean. Insects. 2022; 13(7):569. https://doi.org/10.3390/insects13070569
Chicago/Turabian StyleNdakidemi, Baltazar J., Ernest R. Mbega, Patrick A. Ndakidemi, Steven R. Belmain, Sarah E. J. Arnold, Victoria C. Woolley, and Philip C. Stevenson. 2022. "Plant-Rich Field Margins Influence Natural Predators of Aphids More Than Intercropping in Common Bean" Insects 13, no. 7: 569. https://doi.org/10.3390/insects13070569
APA StyleNdakidemi, B. J., Mbega, E. R., Ndakidemi, P. A., Belmain, S. R., Arnold, S. E. J., Woolley, V. C., & Stevenson, P. C. (2022). Plant-Rich Field Margins Influence Natural Predators of Aphids More Than Intercropping in Common Bean. Insects, 13(7), 569. https://doi.org/10.3390/insects13070569








