Review of the Family Thanerocleridae (Coleoptera: Cleroidea) and the Description of Thanerosus gen. nov. from Cretaceous Amber Using Micro-CT Scanning
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
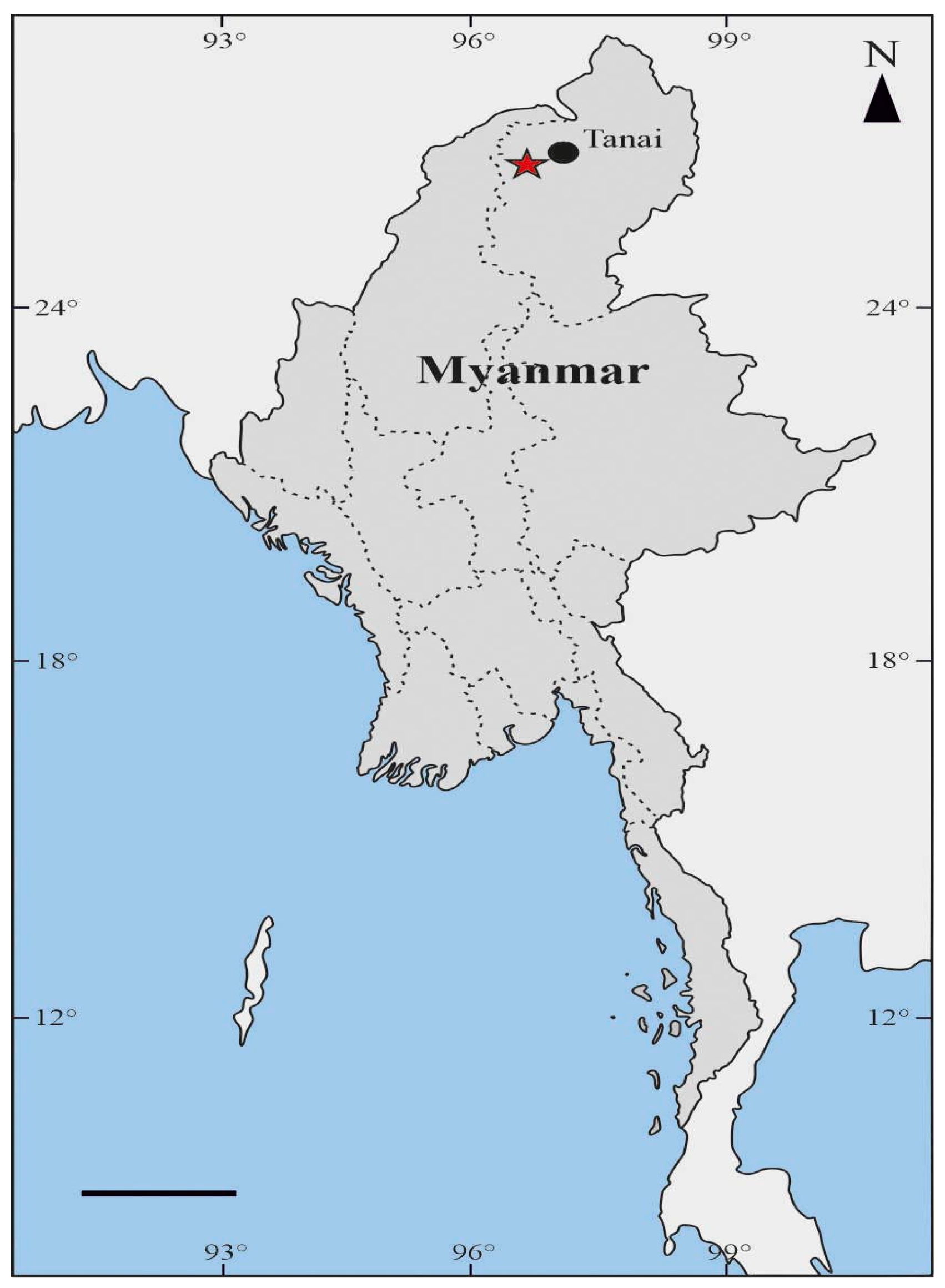
2.1. Geological Setting
2.2. Methods of Observation
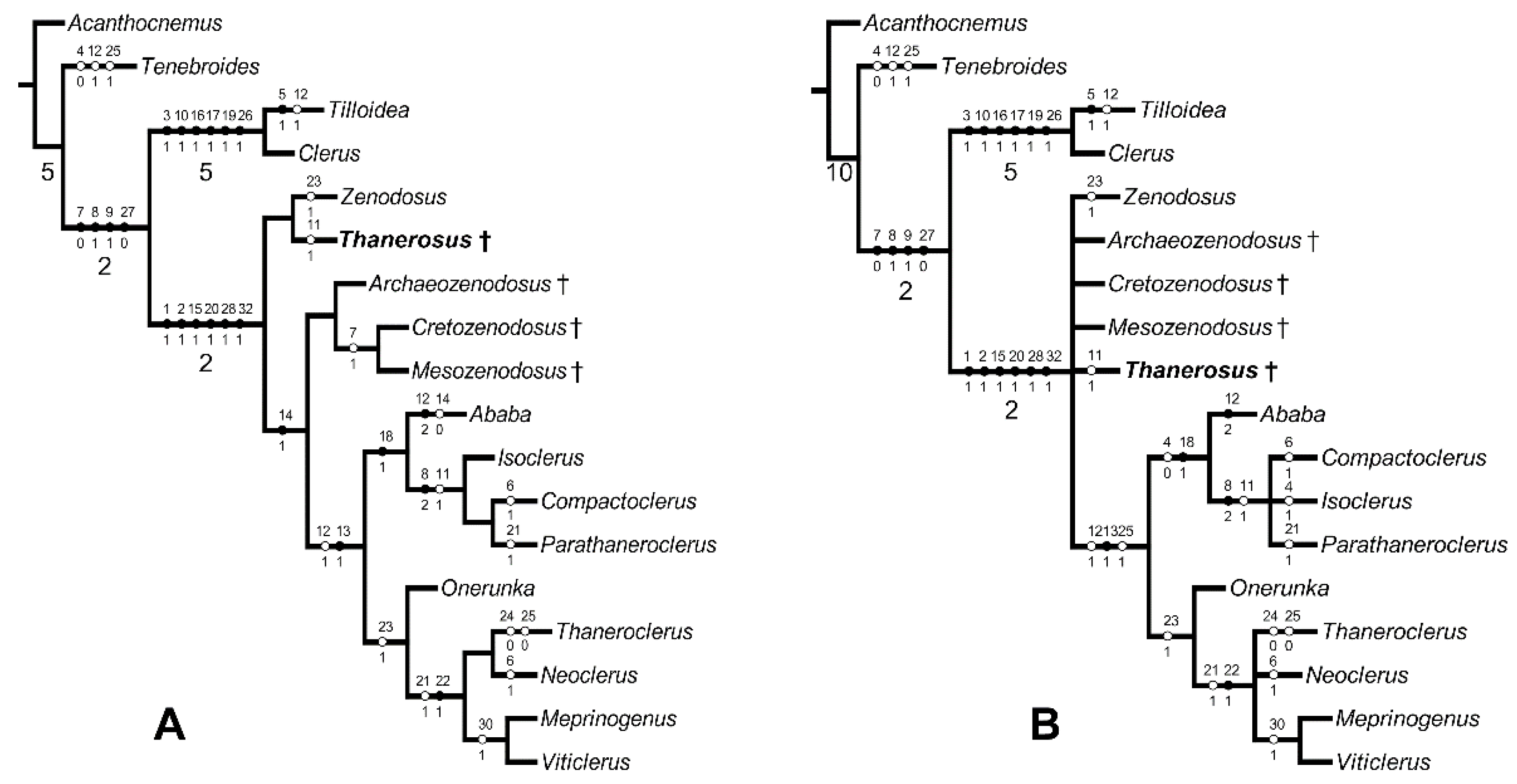
2.3. Phylogenetic Analysis
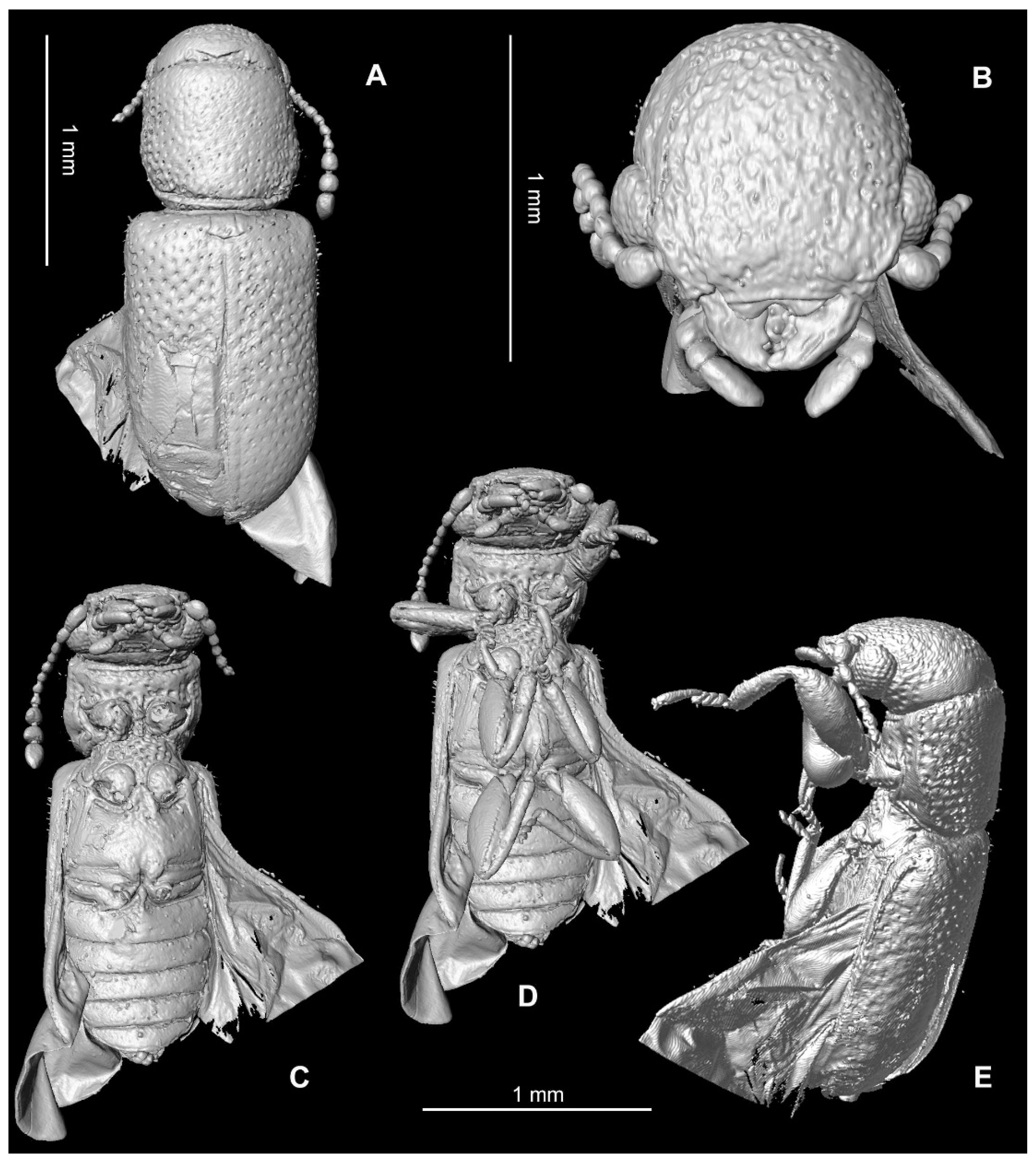
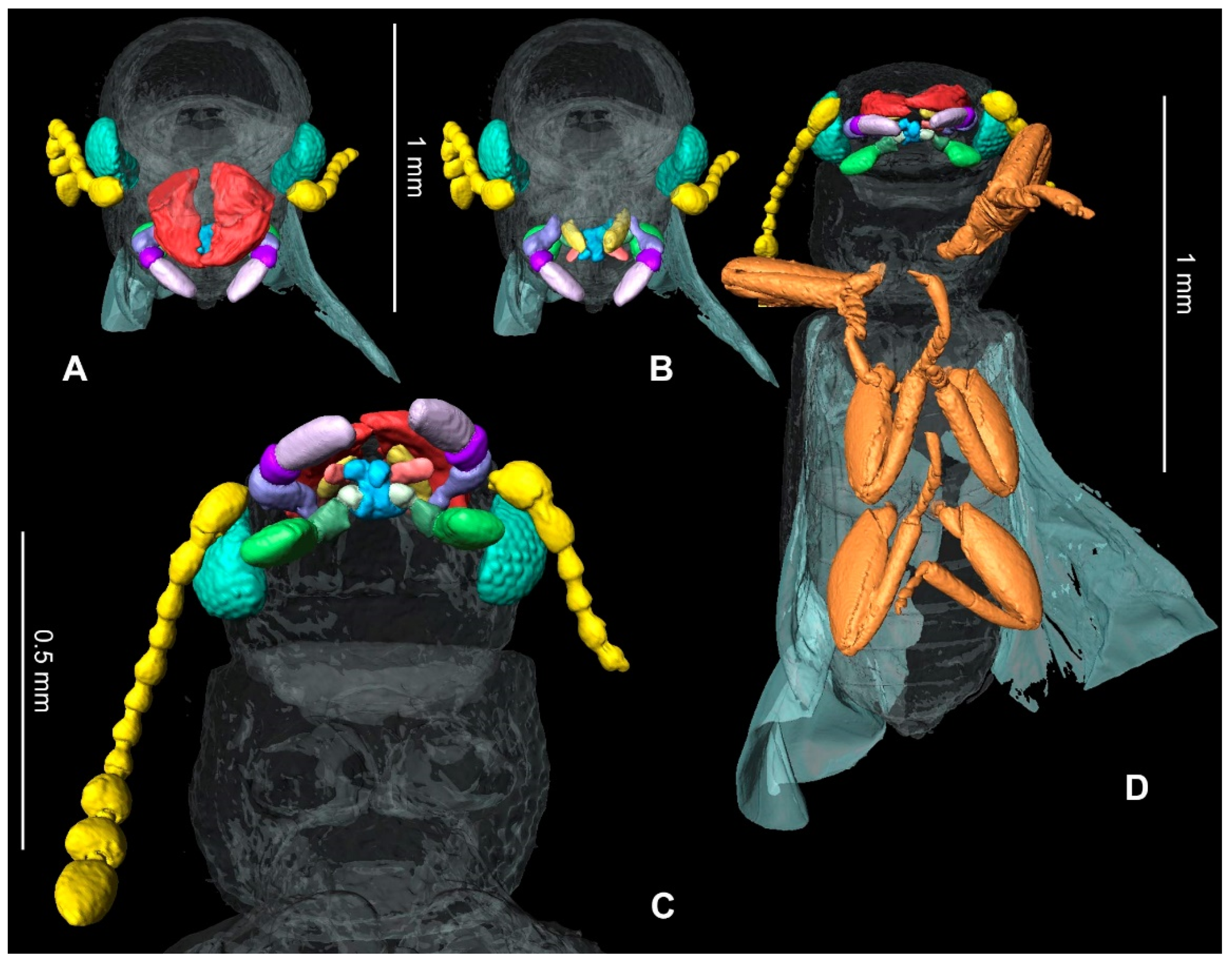
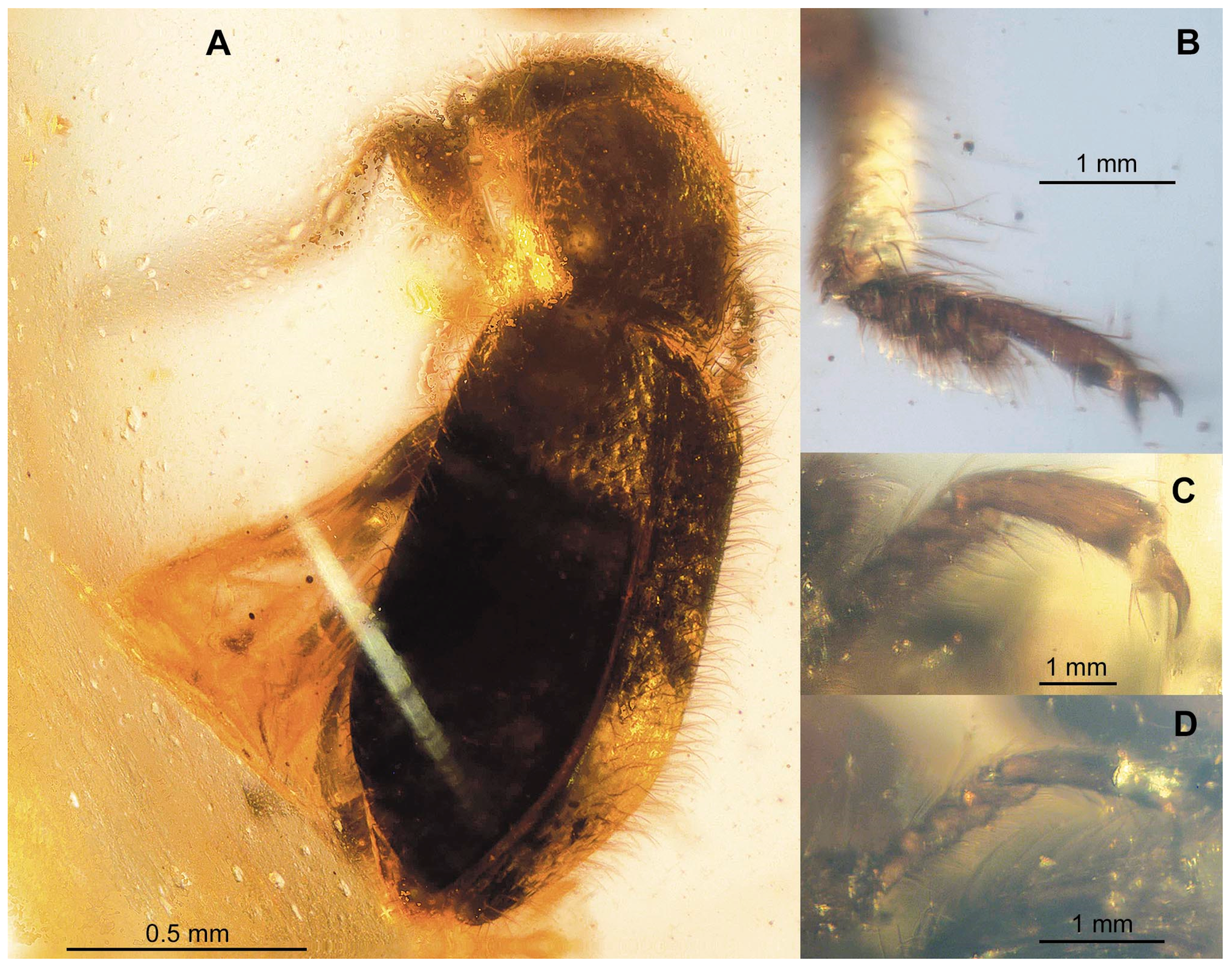
3. Results
Systematic Palaeontology
4. Discussion and Conclusions
4.1. Classification of Thanerosus antiquus gen. and sp. nov.
4.2. Phylogeny and Palaeontology of Thanerocleridae
4.3. Key to the Extinct and Extant Genera of Thanerocleridae
- 1.
- Procoxal cavities externally widely open, procoxae oval or transverse; abdominal segment 9 fully developed; discriminal line present; tarsal formula 5-5-5; mesocoxal cavities externally open or closed. Subfamily Zenodosinae.................................2
- –
- Procoxal cavities externally perfectly, rarely imperfectly (Ababa, Parathaneroclerus) closed; procoxa more or less spherical; abdominal segment 9 reduced to spicular fork (tergite and sternite VIII inconspicuous); discriminal line scarcely perceptible; tarsal formula 5-5-5 or 5-4-4; mesocoxal cavities externally closed, rarely open (Ababa). Subfamily Thaneroclerinae........................................................................6
- 2.
- Mesocoxal cavities externally open...............................................................3
- –
- Mesocoxal cavities externally closed; if the latter feature unknown, a body under 2.5 mm; terminal antennomere with concavity......................................................4
- 3.
- Antennal club weak and indistinct; scutellum transverse; North America.........................................................................................................Zenodosus Wolcott
- –
- Antennae with a loose but distinct club; scutellum quadrate................................................................................................† Thanerosus gen. nov. Peris & Kolibáč
- 4.
- Eyes without interfacetal setae; pronotal carina well-developed; terminal antennomere without concavity; body > 2.5 mm...............................................5
- –
- Eyes with interfacetal setae; pronotal carina weakly developed; terminal antennomere with concavity; body size < 2.5 mm.................................................................................† Mesozenodosus Tihelka, Cheng, Huang, Perrichot & Cai
- 5.
- Pronotum widest anteriorly; procoxal cavity oval; elytral bases about as broad as the base of pronotum; more than 5 mm in length.......† Archaeozenodosus Yu & Kolibáč
- –
- Pronotum widest at 1/3 from the base; procoxal cavity transverse; the base of pronotum half the width of elytral bases; less than 5 mm in length.............................................................................................† Cretozenodosus Cai & Huang
- 6.
- Tarsal formula 5-5-5; depression along notosternal suture absent; procoxae subspherical and moderately narrowly separated. Tribe Thaneroclerini...............7
- –
- Tarsal formula 5-4-4, depression along notosternal suture present; procoxae spherical and extremely narrowly separated. Tribe Isoclerini.......................................11
- 7.
- Tarsomere 4 of protarsi distinctly smaller than 3; pronotal depressions present; unicolorous black or brown species...............................................................8
- –
- Tarsomeres 2, 3 and 4 of protarsi subequal in size; pronotal depressions are absent......................................................................................................9
- 8.
- Radial cell present; elytra without tufts of hairs; mostly five abdominal ventrites; southeastern and eastern Asia, cosmopolitan..................Thaneroclerus Lefebvre
- –
- Radial cell absent; each elytron with a humeral tuft of black hairs and three tufts of white, stout, semierect or decumbent hairs; six abdominal ventrites; India........................................................................Meprinogenus Kolibáč
- 9.
- Pronotum laterally rounded; protibia without any apical spine at the apex.........10
- –
- Pronotum parallel-sided; protibia with one short, blunt, not hooked apical spine at apex; New Guinea...............................................................Onerunka Kolibáč
- 10.
- Antennomeres 10 and 11 coalescent (suture perceptible); elytron not depressed, without distinct humeral gibbae; abdominal ventrite 1 as long as 2 and 3 together, 2 to 5 subequal in length; winged species; southeastern and eastern Asia...........................................................................................................Neoclerus Lewis
- –
- Antenna 11-segmented, club loose; elytron strongly depressed in anterior half, with elevated humeral gibbae; abdominal ventrite 1 as long as 2 to 4 together, 2 to 6 successively shorter; single wingless species; Fiji.....................Viticlerus Miyatake
- 11.
- Tendency to a coalescence of antennomeres 10 and 11 (suture between them perceptible), club 2-segmented; procoxae spherical, very small, space between them minute; prosternum in front of procoxa very long; terminal palpomere of labial palpi coniform; Africa, Madagascar.............................................Compactoclerus Pic
- –
- Antennae 11-segmented with a more or less distinct 3-segmented club; terminal palpomere of labial palpi truncate; Americas, southeastern Asia........................12
- 12.
- Procoxal cavities externally perfectly closed; southeastern Asia......Isoclerus Lewis
- –
- Procoxal cavities externally imperfectly closed; Americas.................................13
- 13.
- Mesocoxal cavities open; pronotum with weak depressions or flat; Americas......................................................................................................Ababa Casey
- –
- Mesocoxal cavities closed; pronotum with three conspicuous depressions; Brazil.........................................................................................................Parathaneroclerus Pic
4.4. Checklist of the Family Thanerocleridae Chapin, 1924
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kolibáč, J.; Leschen, R.A.B. 9.5. Thanerocleridae Chapin, 1924. In Handbook of Zoology, Coleoptera, Volume 2: Morphology and Systematics (Elateroidea, Bostrichiformia, Cucujiformia partim); Leschen, R.A.B., Beutel, R.G., Lawrence, J.F., Eds.; Walter de Gruyter: Berlin, Germany, 2010; Volume 2, pp. 253–257. [Google Scholar]
- Kolibáč, J. Onerunka longi, a new genus and species of Thanerocleridae (Coleoptera) from Papua New Guinea, with systematic notes on the tribe Thaneroclerini. Zootaxa 2012, 3577, 71–79. [Google Scholar] [CrossRef] [Green Version]
- Murakami, H. A new species of the genus Thaneroclerus (Coleoptera, Cleridae, Thaneroclerinae) from Ishigaki-jima island, Japan. Elytra 2016, 6, 285–289. [Google Scholar]
- Murakami, H. New synonym of Thaneroclerus aino Lewis, 1892 (Thanerocleridae: Thaneroclerinae). Jpn. J. Syst. Entomol. 2021, 27, 202–205. [Google Scholar]
- Yu, Y.; Deng, C.; Kolibáč, J.; Ślipiński, S.A.; Ren, D.; Jin, J.; Pang, H. The first record of Cretaceous thaneroclerids (Insecta: Coleoptera) from the Burmese amber. Ann. Zool. 2017, 67, 549–554. [Google Scholar] [CrossRef]
- Cai, C.; Huang, D. First fossil thaneroclerid beetle from mid-Cretaceous Burmese amber (Coleoptera: Cleroidea: Thanerocleridae). Alcheringa Australas. J. Palaeontol. 2018, 42, 115–119. [Google Scholar] [CrossRef]
- Tihelka, E.; Cheng, G.; Huang, D.; Perrichot, V.; Cai, C. First thaneroclerid beetle from Cretaceous Charentese amber (Coleoptera: Cleroidea: Thanerocleridae). Palaeoentomology 2020, 3, 390–398. [Google Scholar] [CrossRef]
- Kolibáč, J.; Huang, D.Y. Mathesius liaoningensis gen. et sp. nov. of Jehol Biota, a presumptive relative of the clerid or thaneroclerid branches of Cleroidea (Coleoptera). Zootaxa 2014, 2872, 1–17. [Google Scholar] [CrossRef]
- Kolibáč, J. Revision of Thanerocleridae n. stat. (Coleoptera, Cleroidea). Mitt. Schweiz. Entomol. Ges. 1992, 65, 303–340. [Google Scholar]
- Kolibáč, J. Metaxinidae, fam. nov., a new family of Cleroidea (Coleoptera). Entomol. Basiliensia 2004, 26, 239–268. [Google Scholar]
- Bouchard, P.; Bousquet, Y.; Davies, A.E.; Alonso-Zarazaga, M.A.; Lawrence, J.F.; Lyal, C.H.; Newton, A.F.; Reid, C.A.M.; Schmitt, M.; Ślipiński, S.A.; et al. Family group names in Coleoptera (Insecta). Zookeys 2011, 88, 1–972. [Google Scholar] [CrossRef] [Green Version]
- Gimmel, M.L.; Bocakova, M.; Gunter, N.L.; Leschen, R.A.B. Comprehensive phylogeny of the Cleroidea (Coleoptera: Cucujiformia). Syst. Entomol. 2019, 44, 527–558. [Google Scholar] [CrossRef]
- Opitz, W. Classification, natural history, phylogeny, and subfamily composition of the Cleridae and generic content of the subfamilies (Coleoptera: Cleroidea). Entomol. Basiliensia Collect. Frey 2010, 32, 31–128. [Google Scholar]
- Gunter, N.L.; Leavengood, J.M.; Bartlett, J.S.; Chapman, E.G.; Cameron, S.L. A molecular phylogeny of the checkered beetles and a description of Epiclininae a new subfamily (Coleoptera: Cleroidea: Cleridae). Syst. Entomol. 2013, 38, 626–636. [Google Scholar] [CrossRef] [Green Version]
- Kolibáč, J.; Huang, D.Y. The oldest known clerid fossils from the Middle Jurassic of China, with a review of Cleridae systematics (Coleoptera). Syst. Entomol. 2016, 41, 808–823. [Google Scholar] [CrossRef]
- Zhang, S.-Q.; Che, L.-H.; Li, Y.; Liang, D.; Pang, H.; Ślipiński, S.A.; Zhang, P. Evolutionary history of Coleoptera revealed by extensive sampling of genes and species. Nat. Commun. 2018, 9, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKenna, D.D.; Shin, S.; Ahrens, D.; Balke, M.; Beza-Beza, C.; Clarke, D.J.; Donath, A.; Escalona, H.E.; Friedrich, F.; Letsch, H.; et al. The evolution and genomic basis of beetle diversity. Proc. Natl. Acad. Sci. USA 2019, 116, 24729–24737. [Google Scholar] [CrossRef] [Green Version]
- Cai, C.; Tihelka, E.; Giacomelli, M.; Lawrence, J.F.; Slipinski, A.; Kundrata, R.; Yamamoto, S.; Thayer, M.K.; Newton, A.F.; Leschen, R.A.B.; et al. Integrated phylogenomics and fossil data illuminate the evolution of beetles. R. Soc. Open Sci. 2022, 9, 211771. [Google Scholar] [CrossRef]
- Kolibáč, J.; Bocakova, M.; Liebherr, J.K.; Ramage, T.; Porch, N. Extinct and extant Pacific Trogossitidae and the evolution of Cleroidea (Coleoptera) after the Late Triassic biotic crisis. Zool. J. Linn. Soc. 2021, 191, 846–882. [Google Scholar] [CrossRef]
- Kolibáč, J. New Australian Thanerocleridae, with notes on the biogeography of the subtribe Isoclerinae Kolibáč (Coleoptera: Cleroidea). Invertebr. Taxon. 1998, 12, 951–975. [Google Scholar] [CrossRef]
- Melnik, I.V. Isoclerus succedaneus sp. n.—A new species of the Thanerocleridae (Coleopera) from Southern India. Russ. Entomol. J. 2005, 14, 281–282. [Google Scholar]
- Opitz, W. Taxonomic revision of the Western Hemisphere checkered beetle genus Ababa Casey (Coleoptera: Cleridae: Isoclerinae). Entomol. Am. 2018, 124, 1–18. [Google Scholar] [CrossRef]
- Zheng, D.; Chang, S.-C.; Perrichot, V.; Dutta, S.; Rudra, A.; Mu, L.; Kelly, R.S.; Li, S.; Zhang, Q.; Zhang, Q.; et al. A Late Cretaceous amber biota from central Myanmar. Nat. Commun. 2018, 9, 3170. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.; Qiu, L. Zircon U-Pb age constraints on the mid-Cretaceous Hkamti amber biota in northern Myanmar. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2020, 558, 109960. [Google Scholar] [CrossRef]
- Peris, D.; Bao, T.; Mähler, B.; Philips, T.K. A morphologically unique species of Ptinidae (Coleoptera) and the first found in mid-Cretaceous Kachin amber (Myanmar). J. Syst. Palaeontol. 2020, 18, 873–883. [Google Scholar] [CrossRef]
- Shi, G.; Grimaldi, D.A.; Harlow, G.E.; Wang, J.; Wang, J.; Yang, M.; Lei, W.; Li, Q.; Li, X. Age constraint on Burmese amber based on U–Pb dating of zircons. Cretac. Res. 2012, 37, 155–163. [Google Scholar] [CrossRef]
- Nixon, K.C. WinClada; Version 1.0000; Published by the Author: Ithaca, NY, USA, 1999. [Google Scholar]
- Goloboff, P.A.; Farris, J.S.; Nixon, K.C. TNT, a free program for phylogenetic analysis. Cladistics 2008, 24, 774–786. [Google Scholar] [CrossRef]
- Goloboff, P.A.; Catalano, S.A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 2016, 32, 221–238. [Google Scholar] [CrossRef]
- Goloboff, P.A.; Torres, A.; Arias, J.S. Weighted parsimony outperforms other methods of phylogenetic inference under models appropriate for morphology. Cladistics 2018, 34, 407–437. [Google Scholar] [CrossRef]
- Smith, M.R. Bayesian and parsimony approaches reconstruct informative trees from simulated morphological datasets. Biol. Lett. 2019, 15, 20180632. [Google Scholar] [CrossRef] [Green Version]
- Kolibáč, J.; Huang, D.Y. New cleroid beetles from the Middle–Late Jurassic of China. Acta Palaeontol. Pol. 2019, 64, 143–155. [Google Scholar] [CrossRef] [Green Version]
- Peris, D.; Rust, J. Cretaceous beetles (Insecta: Coleoptera) in amber: The palaeoecology of this most diverse group of insects. Zool. J. Linn. Soc. 2020, 189, 1085–1104. [Google Scholar] [CrossRef]
- Solórzano-Kraemer, M.M.; Delclòs, X.; Clapham, M.E.; Arillo, A.; Peris, D.; Jäger, P.; Stebner, F.; Peñalver, E. Arthropods in modern resins reveal if amber accurately recorded forest arthropod communities. Proc. Natl. Acad. Sci. USA 2018, 115, 6739–6744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leschen, R.A.B.; Buckley, T.R. Multistate characters and diet shifts: Evolution of Erotylidae (Coleoptera). Syst. Biol. 2007, 56, 97–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Champion, C. III. Notes on various Central American Coleoptera, with descriptions of new genera and species. Trans. Entomol. Soc. Lond. 1913, 61, 58–169. [Google Scholar] [CrossRef]
- Corporaal, J.B. Revision of Thaneroclerinae. Bijdr. Dierkd. 1939, 27, 347–360. [Google Scholar] [CrossRef]
- Crowson, R.A. A review of the classification of Cleroidea (Coleoptera) with descriptions of two new genera of Peltidae and of several new larval types. Trans. R. Entomol. Soc. Lond. 1964, 116, 275–327. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peris, D.; Mähler, B.; Kolibáč, J. Review of the Family Thanerocleridae (Coleoptera: Cleroidea) and the Description of Thanerosus gen. nov. from Cretaceous Amber Using Micro-CT Scanning. Insects 2022, 13, 438. https://doi.org/10.3390/insects13050438
Peris D, Mähler B, Kolibáč J. Review of the Family Thanerocleridae (Coleoptera: Cleroidea) and the Description of Thanerosus gen. nov. from Cretaceous Amber Using Micro-CT Scanning. Insects. 2022; 13(5):438. https://doi.org/10.3390/insects13050438
Chicago/Turabian StylePeris, David, Bastian Mähler, and Jiří Kolibáč. 2022. "Review of the Family Thanerocleridae (Coleoptera: Cleroidea) and the Description of Thanerosus gen. nov. from Cretaceous Amber Using Micro-CT Scanning" Insects 13, no. 5: 438. https://doi.org/10.3390/insects13050438
APA StylePeris, D., Mähler, B., & Kolibáč, J. (2022). Review of the Family Thanerocleridae (Coleoptera: Cleroidea) and the Description of Thanerosus gen. nov. from Cretaceous Amber Using Micro-CT Scanning. Insects, 13(5), 438. https://doi.org/10.3390/insects13050438






