Unraveling the Role of Lac Insects in Providing Natural Industrial Products
Abstract
Simple Summary
Abstract
1. Introduction
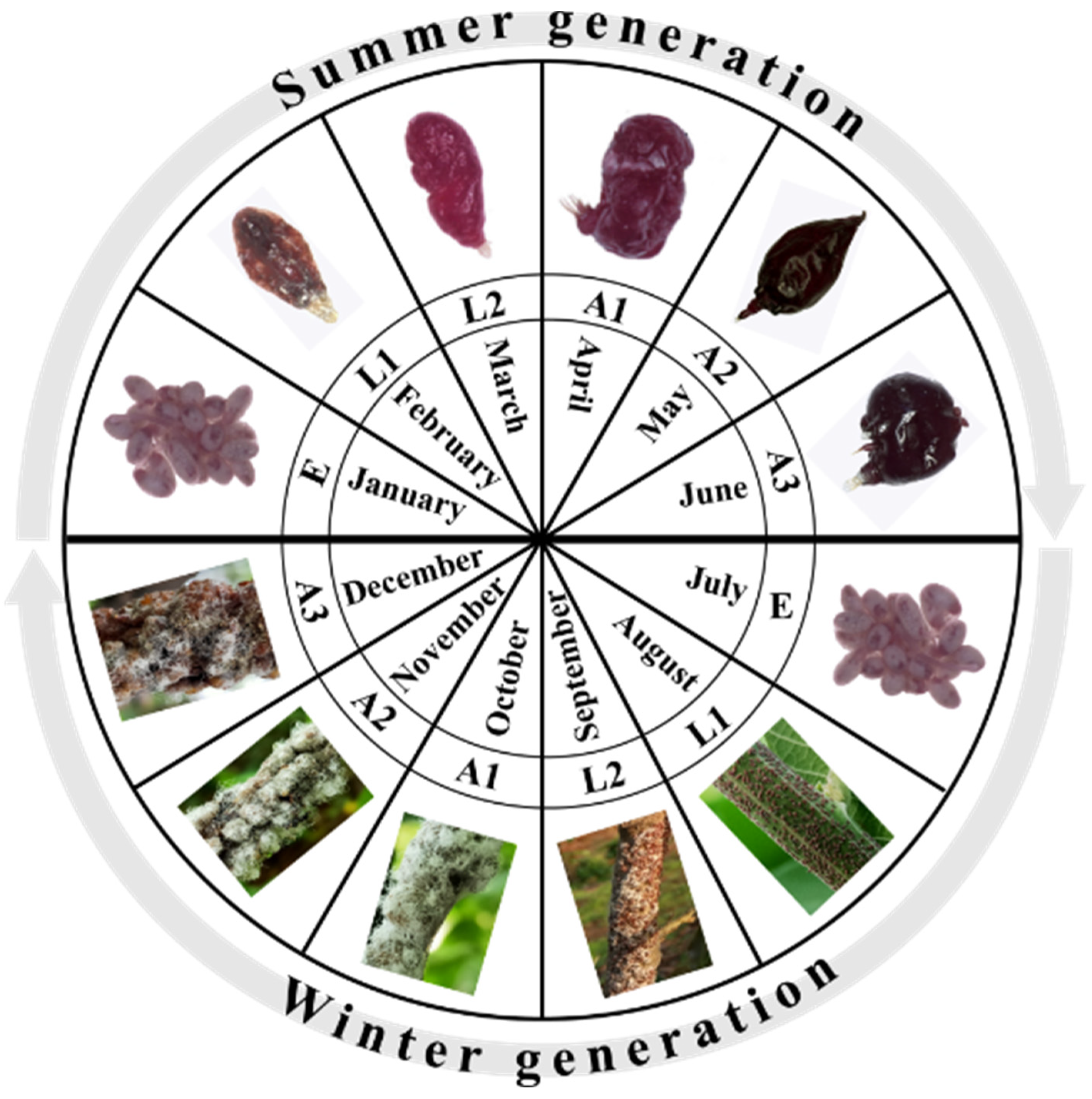
2. Lac Insect Interaction
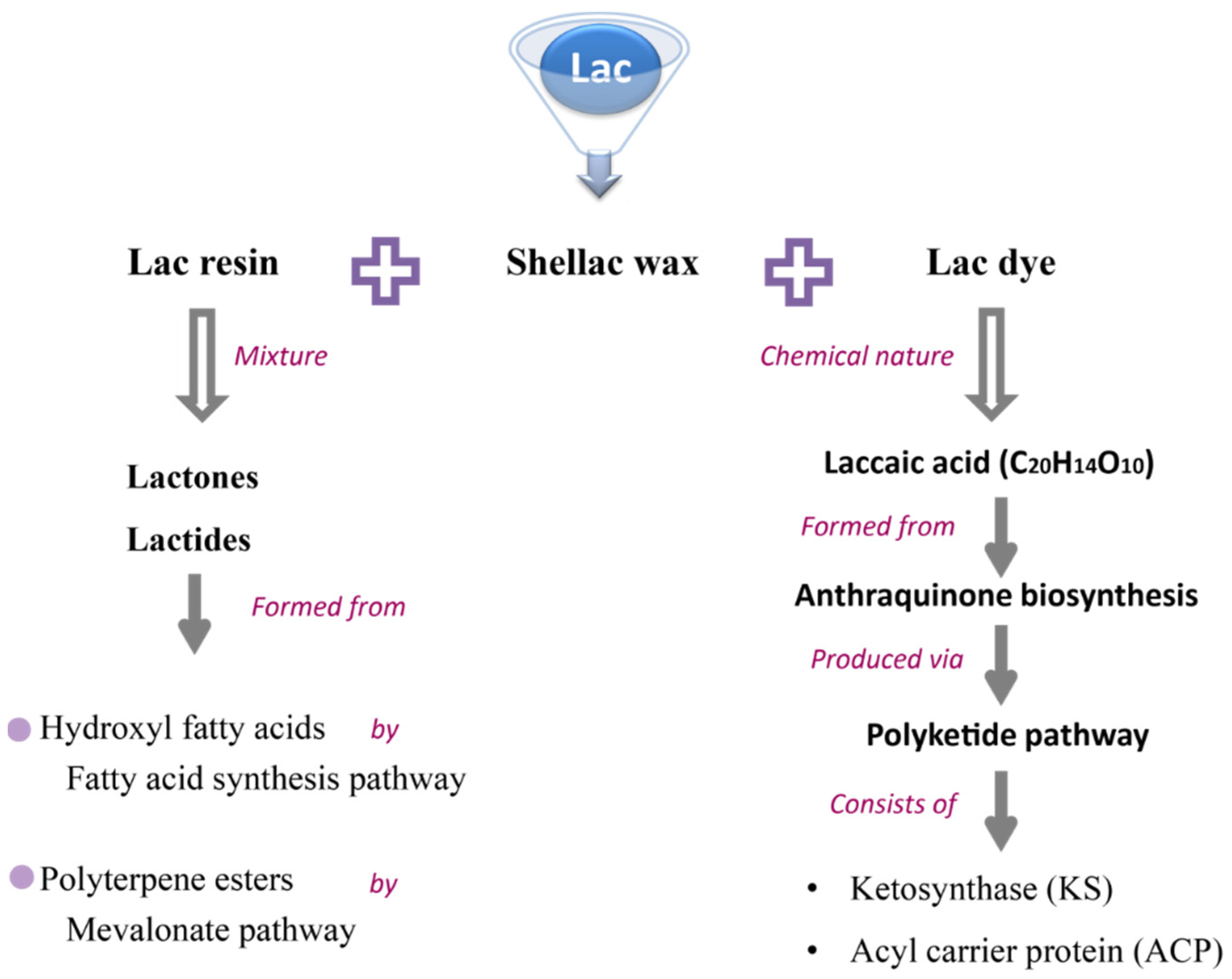
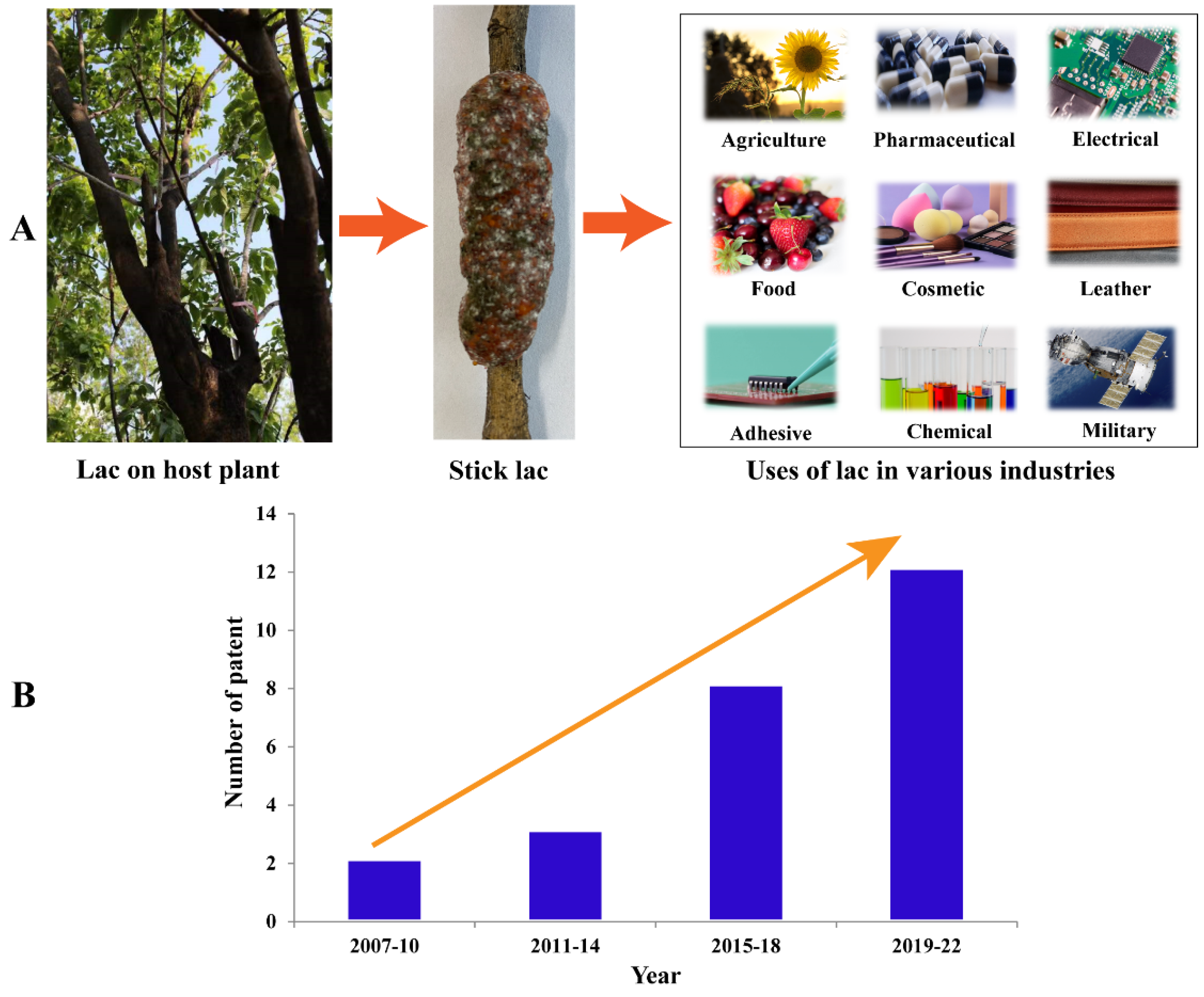
3. Lac Composition and Their Industrial Application
4. Important Lac Genes Involved in Production
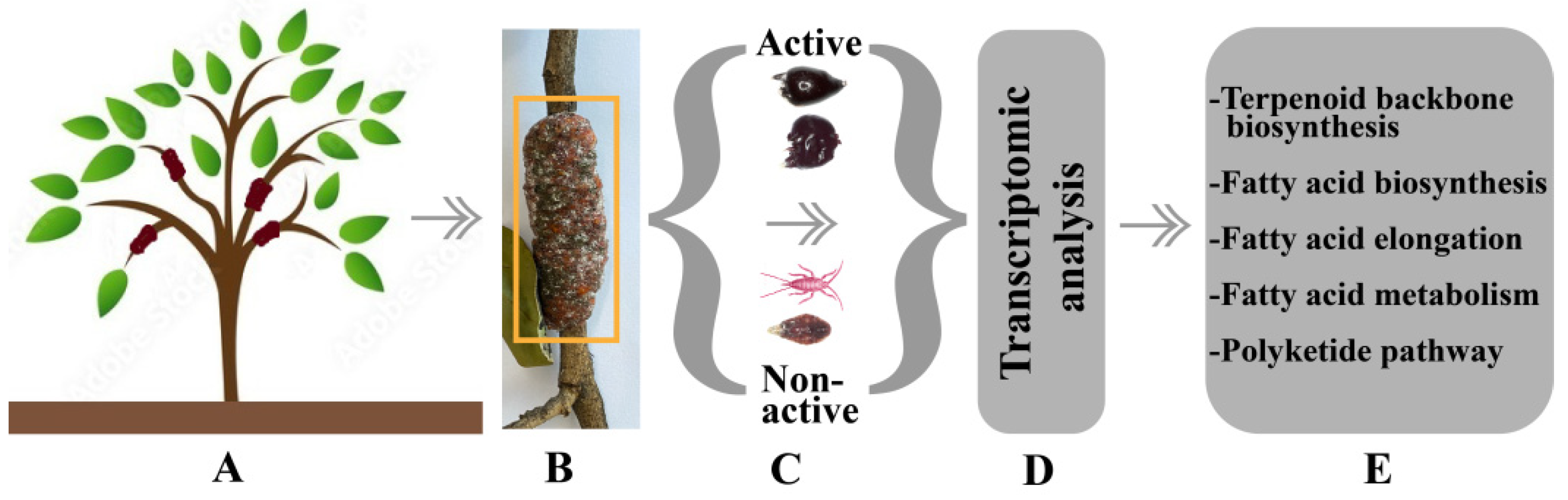
5. Deciphering the Transcriptomic of Lac Secretion Mechanism
6. Role of Native Microbial Diversity of Lac Insects
7. Important Host Plants for Lac
8. Natural Enemies of Lac Insects and Their Management
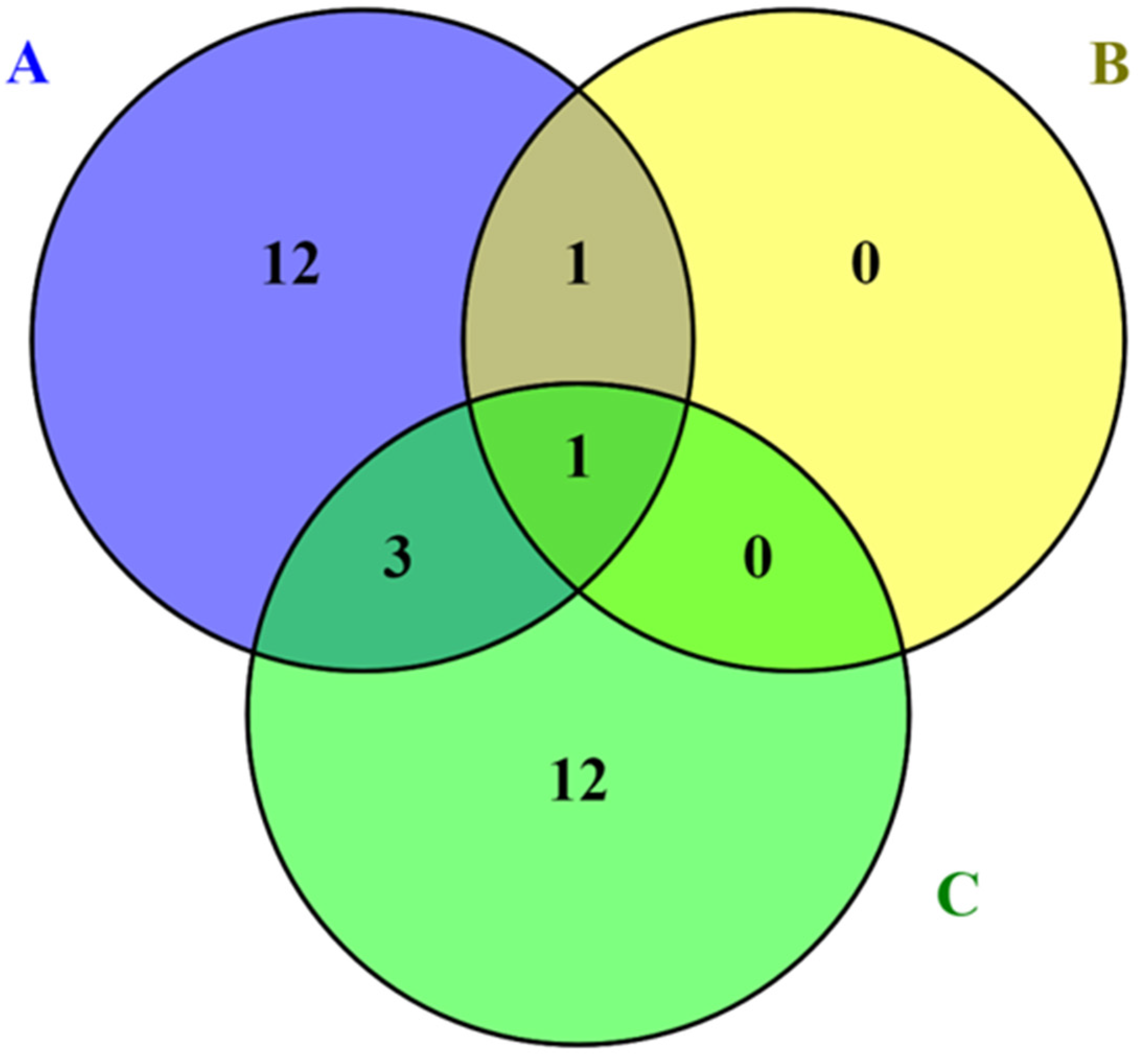
9. Conclusions and Future Research Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mohanasundaram, A.; Kaprakkaden, A.; Nebapure, S.M.; Sharma, K.K.; Naaz, N.; Mishra, R.; Shree, U. Electroantennography and behavioral studies of Eublemma amabilis [Moore] and Pseudohypatopa pulverea [Meyr] in relation to volatiles of lac insect (Kerria lacca Kerr.) and its associated products. Int. J. Trop. Insect Sci. 2022, 42, 2313–2324. [Google Scholar] [CrossRef]
- Chen, X.M.; Chen, Y.Q.; Zhang, H.; Shi, L. Lac Insects Breeding and Lac Processing; Chinese Forestry Press: Beijing, China, 2008. [Google Scholar]
- Chen, X.M.; Chen, H.; Ying, F.; Rui, H.; Yang, Z.X. Status of two species of lac insects in the genus Kerria from China based on morphological, cellular, and molecular evidence. J. Insect Sci. 2011, 11, 106. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.W.; Liu, P.F.; Lu, Q.; Ling, X.F.; Zhang, J.W.; Chen, M.S.; Chen, H.; Chen, X.M. Potential Pathways and Genes Involved in Lac Synthesis and Secretion in Kerria chinensis (Hemiptera: Kerriidae) Based on Transcriptomic Analyses. Insects 2019, 10, 430. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.M. Biodiversity of Lac Insects; Yunnan Science and Technology Press: Kunming, China, 2005; p. 96. [Google Scholar]
- Chen, Y.Q.; Li, Q.; Chen, Y.L.; Lu, Z.X.; Zhou, X.Y. Ant diversity and bio-indicators in land management of lac insect agroecosystem in Southwestern China. Biodivers. Conserv. 2011, 20, 3017–3038. [Google Scholar] [CrossRef]
- Wang, W.W.; Ling, X.F.; Bashir, N.H.; Lu, Q.; Zhang, J.W.; Li, T.C.; Chen, H. Selection and evaluation of reference genes for quantitative real-time PCR analysis in lac insect (Kerria lacca). Entomol. Res. 2022, 52, 57–67. [Google Scholar] [CrossRef]
- Chen, X.M.; Feng, Y. Chapter 1—Lac insects. In An Introduction to Resource Entomology; Science Press: Beijing, China, 2009; pp. 12–28. [Google Scholar]
- Thombare, N.; Kumar, S.; Kumari, U.; Sakare, P.; Yogi, R.K.; Prasad, N.; Sharma, K.K. Shellac as a multifunctional biopolymer: A review on properties, applications and future potential. Int. J. Biol. Macromol. 2022, 215, 203–223. [Google Scholar] [CrossRef]
- Sharma, K.K. Understanding the Diversity of Lac Insects of Kerria spp. in India and the Nature of Insect-Host Plant Interaction. In Natural Resource Management: Ecological Perspectives; Peshin, R., Dhawan, A.K., Eds.; Springer Nature: Cham, Switzerland, 2018; pp. 219–233. [Google Scholar] [CrossRef]
- Borah, N.; Garkoti, S.C. Indigenous lac culture and local livelihood: A case study of Karbi community of Assam, North-Eastern India. Indian J. Tradit. Knowl. 2020, 19, 197–207. [Google Scholar]
- Gullan, P.J.; Kondo, T. The morphology of lac insects (Hemiptera: Coccoidea: Kerriidae). In Proceedings of the XI International Symposium of Scale Insect Studies, Oeiras, Portugal, 24–27 September 2007; pp. 63–70. [Google Scholar]
- Rajgopal, N.N.; Mohanasundaram, A.; Sharma, K.K. A new species of lac insect in the genus Kerria Targioni Tozzetti (Hemiptera: Coccomorpha: Tachardiidae) on Samanea saman (Fabaceae) from India. Zootaxa 2021, 4938, 60–68. [Google Scholar] [CrossRef]
- Rajgopal, N.N. Taxonomic aspects of Lac Insect. In Training Manual: ICAR-NAHEP-CAAST Short Training on Natural Resins and Gums: Production, Processing, Value Addition and Marketing; Prasad, N., Thombare, N., Singh, R., Sakare, P., Sharma, K.K., Eds.; ICAR-IINRG: Ranchi, India, 2020; pp. 34–39. [Google Scholar]
- Varshney, R.K.; Sharma, K.K. Lac Insects of the World—An Updated Catalogue and Bibliography; Indian Council of Agricultural Research, Indian Institute of Natural Resins and Gums: Ranchi, India, 2020; p. 84. [Google Scholar]
- Mohanasundaram, A.; Sharma, K.K.; Naaz, N. Lac insect, natural enemies and their management. In Commercial Entomology; Kumar, A., Kumar, N., Chand, H., Eds.; New India Publishing Agency: New Delhi, India, 2019; pp. 111–128. [Google Scholar]
- Mishra, Y.D.; Yadav, S.K.; Singh, R.K. Lac host plants. In Beneficial Insect Farming-Benefits Livelihood Generation; Sharma, K.K., Monobrullah, M., Mohanasundaram, A., Ramani, R., Eds.; ICAR-Indian Institute of Natural Resins & Gums: Ranchi, India, 2016; pp. 66–78. [Google Scholar]
- Ramani, R.; Sharma, K.K. Lac insects. In Beneficial Insects Farming; Sharma, K.K., Monobrullah, M., Mohanasundaram, A., Ramani, R., Eds.; ICAR-Indian Institute of Natural Resins and Gums: Ranchi, India, 2016; pp. 53–65. [Google Scholar]
- Bashir, N.H.; Wang, W.W.; Ling, X.F.; Zhang, J.W.; Lu, Q.; He, R.; Chen, H. Characterization of Potential Molecular Markers in Lac Insect Kerria lacca (Kerr) Responsible for Lac Production. Insects 2022, 13, 545. [Google Scholar] [CrossRef]
- Shah, T.H.; Thomas, M.; Bhandari, R. Lac production, constraints and management: A review. Int. J. Curr. Res. 2015, 7, 13652–13659. [Google Scholar]
- Sharma, K.K. Lac insects and host plants. In Industrial Entomology; Omkar, Ed.; Springer Nature: Singapore, 2017; pp. 157–180. [Google Scholar]
- Sharma, K.K.; Jaiswal, A.K.; Kumar, K.K. Role of lac culture in biodiversity conservation: Issues at stake and conservation strategy. Curr. Sci. 2006, 91, 894–898. [Google Scholar]
- Shamim, G.; Pandey, D.M.; Sharma, K.K.; Ramani, R. Genes Involved in the Biosynthesis of Lac Dye Constituents in Indian Lac Insect, Kerria lacca (Kerr). J. Entomol. Res. Soc. 2016, 18, 7–17. [Google Scholar]
- Meena, S.C.; Sharma, K.K. Species composition and richness of entomo-fauna associated with lac insect, Kerria lacca Kerr in western plains of India. J. Entomol. Zool. Stud. 2018, 6, 684–690. [Google Scholar]
- Singh, J.P. Effect of broodlac treatment with insecticides on population reduction of predators and parasitoids of lac insect, Kerria lacca (Kerr). Natl. Acad. Sci. Lett. 2013, 36, 379–383. [Google Scholar] [CrossRef]
- Bashir, N.H.; Wang, W.W.; Liu, J.; Wang, W.; Chen, H. First record of the lac-producing species Kerria nepalensis Varshney (Hemiptera, Kerriidae) from China, with a key to Chinese species. ZooKeys 2021, 1061, 1–9. [Google Scholar] [CrossRef]
- Mishra, Y.D.; Kumar, P. Lac culture. In Industrial Entomology; Omkar, Ed.; Springer Nature: Singapore, 2017; pp. 109–155. [Google Scholar] [CrossRef]
- Talukder, B.; Das, B.K. A new invasive species of Kerria Targioni-Tozzetti (Hemiptera: Coccomorpha: Kerriidae) on rain tree, Albizia saman (Fabaceae) from India. Orient. Insects 2020, 54, 176–190. [Google Scholar] [CrossRef]
- Kaushik, S.; Sharma, K.K.; Ramani, R.; Lakhanpaul, S. Detection of Wolbachia Phage (WO) in Indian Lac Insect [Kerria lacca (Kerr.)] and Its Implications. Indian J. Microbiol. 2018, 59, 237–240. [Google Scholar] [CrossRef]
- Sharma, K.K.; Ramani, R. An update on synoptic catalogue of lac insects (Homoptera: Tachardiidae). J. Bombay Nat. Hist. Soc. 1999, 96, 438–443. [Google Scholar]
- Chen, X.M.; Chen, H.; Zhao, M.; Yang, Z.X.; Feng, Y. Insect industrialization and prospect in commerce: A case of China. Entomol. Res. 2022, 52, 178–194. [Google Scholar] [CrossRef]
- Yusuf, M.; Aijaz, M.; Keserwani, N.; Ansari, N.H.; Ahmad, S. Ethnomedicinal, Pharmacological and Commercial Perspectives of Laccifer lacca Body Exudate (LBE). Lett. Appl. NanoBioSci. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Barrie, W.E. Shellac Based Skin Care Lotion. U.S. Patent US20150258010A1, 17 September 2015. [Google Scholar]
- Schwarz, K.; Keppler, J.; Theismann, E.M.; Knipp, J.; Fangmann, D.; Laudes, M.; Schreiber, S.; Wätzig, G. Shellac Microcapsule Formulations and Compositions for Topical Intestinal Delivery of Vitamin b3. U.S. Patent US20210275513A1, 9 September 2021. [Google Scholar]
- Santos, S.A.; Barrie, W.E.; Murphy, K.M. Cannabis Delivery with Protective Glaze Coating. U.S. Patent US20200323791A1, 15 October 2020. [Google Scholar]
- Cho, I.G. Chocolate Confectionery Preparation Method and Chocolate Confectionery Prepared Thereby. U.S. Patent US10448655b2, 22 October 2019. [Google Scholar]
- Orlowski, M. Shellac and Paclitaxel Coated Catheter Balloons. U.S. Patent US10293085B2, 21 May 2019. [Google Scholar]
- Ghantous, A.; Most, C.; McVay, R. Metal Cans Coated with Shellac-Containing Coatings. U.S. Patent US10392515B2, 27 August 2019. [Google Scholar]
- Shyam, K.C.; Manish, K.B.; Tapan, K.R.; Sumitesh, D.; Ranjan, K.S.; Yashabanta, N.S.; Ashit, K.P.; Vikas, C.S. Process for Synthesizing Reduced Graphene Oxide on a Substrate from Seedlac. U.S. Patent US9617159B2, 11 April 2017. [Google Scholar]
- Seidl, W.; Wache, S.; Stohr, W.; Zürn, S.; Riedmiller, J.; Schwarz, V. Cementitious System Comprising Accelerator Particles Coated with Crosslinked Shellac. U.S. Patent US9428419B2, 30 August 2016. [Google Scholar]
- Signorino, C.A.; Smith, T.L.; Levine, S. Shellac Enteric Coatings. U.S. Patent 20110269850A1, 3 November 2011. [Google Scholar]
- Durig, T.; Zong, Y. Stable Shellac Enteric Coating Formulation for Nutraceutical and Pharmaceutical Dosage Forms. U.S. Patent US20110002986A1, 6 June 2011. [Google Scholar]
- Diaz, M. Sprayable Hand Cleaner Made of Edible Shellac and Micellular Surfactants. U.S. Patent US20100144574A1, 10 June 2010. [Google Scholar]
- Rohmer, M. The mevalonate-independent methylerythritol 4-phosphate (MEP) pathway for isoprenoid biosynthesis, including carotenoids. Pure Appl. Chem. 1999, 71, 2279–2284. [Google Scholar] [CrossRef]
- Bede, J.C.; Teal, P.E.A.; Goodman, W.G.; Tobe, S.S. Biosynthetic Pathway of Insect Juvenile Hormone III in Cell Suspension Cultures of the Sedge Cyperus iria. Plant Physiol. 2001, 127, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.H.; Goodman, H.M. The Arabidopsis IspH Homolog Is Involved in the Plastid Nonmevalonate Pathway of Isoprenoid Biosynthesis. Plant Physiol. 2005, 138, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, T.; Ekbal, S.; Kumari, K. SSH reveals different functional categories of putative resin synthesizing genes in the industrial insect, Kerria lacca (Kerr). J. Asia-Pac. Entomol. 2021, 24, 1187–1193. [Google Scholar] [CrossRef]
- Shamim, G.; Pandey, D.M.; Ramani, R.; Sharma, K.K. Identification of genes related to resin biosynthesis in the Indian lac insect, Kerria lacca (Hemiptera: Tachardiidae). Int. J. Trop. Insect Sci. 2014, 34, 149–155. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Z.X. In vitro and in vivo characterization of a novel insect decaprenyl diphosphate synthase: A two-major step catalytic mechanism is proposed. Biochem. Biophys. Res. Commun. 2013, 442, 105–111. [Google Scholar] [CrossRef]
- Koyama, T. Molecular analysis of prenyl chain elongating enzymes. Biosci. Biotechnol. Biochem. 1999, 63, 1671–1676. [Google Scholar] [CrossRef]
- Ogura, K. Enzymatic aspects of isoprenoid chain elongation. Chem. Rev. 1998, 98, 1263–1276. [Google Scholar] [CrossRef]
- Sun, Z.J.; Li, Z.X. The terpenoid backbone biosynthesis pathway directly affects the biosynthesis of alarm pheromone in the aphid. Insect Mol. Biol. 2018, 27, 824–834. [Google Scholar] [CrossRef]
- Gu, L.H.; Han, J.M.; Wood, J.D.; Chang, C.Y.Y.; Sun, Y. Sun-induced Chl fluorescence and its importance for biophysical modeling of photosynthesis based on light reactions. New Phytol. 2019, 223, 1179–1191. [Google Scholar] [CrossRef]
- Sperling, P.; Ternes, P.; Zank, T.K.; Heinz, E. The evolution of desaturases. Prostaglandins Leukot. Essent. Fat. Acids 2003, 68, 73–95. [Google Scholar] [CrossRef] [PubMed]
- Dallerac, R.; Labeur, C.; Jallon, J.M.; Knipple, D.C.; Roelofs, W.L.; Wicker, T.C. A Δ9 desaturase gene with a different substrate specificity is responsible for the cuticular diene hydrocarbon polymorphism in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2000, 97, 9449–9454. [Google Scholar] [CrossRef] [PubMed]
- Horne, I.; Gibb, N.; Damcevski, K.; Glover, K.; Haritos, V.S. Two conserved Z9-octadecanoic acid desaturases in the red flour beetle, Tribolium castaneum. Gene 2010, 468, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Riddervold, M.H.; Tittiger, C.; Blomquist, G.J.; Borgeson, C.E. Biochemical and molecular characterizaton of house cricket (Acheta domesticus, Orthoptera: Gryllidae) Δ9 desaturase. Insect Biochem. Mol. Biol. 2002, 32, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Li, Z.; Ma, C.; Zhang, Z.; Hull, J.J.; Lei, C.; Jin, S.; Chen, L. Knockdown of a metathoracic scent gland desaturase enhances the production of (E)-4-oxo-2-hexenal and suppresses female sexual attractiveness in the plant bug Adelphocoris suturalis. Insect Mol. Biol. 2017, 26, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.M.; Ye, W.F.; Noman, A.; Machado, R.A.R.; Lou, Y.G. The desaturase gene family is crucially required for fatty acid metabolism and survival of the brown planthopper, Nilaparvata lugens. Int. J. Mol. Sci. 2019, 20, 1369. [Google Scholar] [CrossRef]
- Haritos, V.S.; Horne, I.; Damcevski, K.; Glover, K.; Gibb, N.; Okada, S.; Hamberg, M. The convergent evolution of defensive polyacetylenic fatty acid biosynthesis genes in soldier beetles. Nat. Commun. 2012, 3, 1150. [Google Scholar] [CrossRef]
- Kayukawa, T.; Chen, B.; Hoshizaki, S.; Ishikawa, Y. Upregulation of a desaturase is associated with the enhancement of cold hardiness in the onion maggot, Delia antiqua. Insect Biochem. Mol. Biol. 2007, 37, 1160–1167. [Google Scholar] [CrossRef]
- Wang, Y.; da Cruz, T.C.; Pulfemuller, A.; Grégoire, S.; Ferveur, J.F.; Moussian, B. Inhibition of fatty acid desaturases in Drosophila melanogaster larvae blocks feeding and developmental progression. Arch. Insect Biochem. Physiol. 2016, 92, 6–23. [Google Scholar] [CrossRef]
- Bousquet, F.; Nojima, T.; Houot, B.; Chauvel, I.; Chaudy, S.; Dupas, S.; Yamamoto, D.; Ferveur, J.F. Expression of a desaturase gene, desat1, in neural and nonneural tissues separately affects perception and emission of sex pheromones in Drosophila. Proc. Natl. Acad. Sci. USA 2012, 109, 249–254. [Google Scholar] [CrossRef]
- Köhler, K.; Brunner, E.; Guan, X.L.; Boucke, K.; Greber, U.F.; Mohanty, S.; Barth, J.M.; Wenk, M.R.; Hafen, E. A combined proteomic and genetic analysis identifies a role for the lipid desaturase Desat1 in starvation-induced autophagy in Drosophila. Autophagy 2009, 5, 980–990. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Shirley, N.; Bleackley, M.; Dolan, S.; Shafee, T. Transcriptomics technologies. PLoS Comput. Biol. 2017, 13, e1005457. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.C.; Chen, Y. Transcriptomics: Advances and approaches. Sci. China Life Sci. 2013, 56, 960–967. [Google Scholar] [CrossRef] [PubMed]
- Dubey, N.K.; Goel, R.; Ranjan, A.; Idris, A.; Singh, S.K.; Bag, S.K.; Chandrashekar, K.; Pandey, K.D.; Singh, P.K.; Sawant, S.V. Comparative transcriptome analysis of Gossypium hirsutum L. in response to sap sucking insects: Aphid and whitefly. BMC Genom. 2013, 14, 241. [Google Scholar] [CrossRef]
- Fan, X.B.; Pang, R.; Li, W.X.; Ojha, A.; Li, D.; Zhang, W.Q. An overview of embryogenesis: External morphology and transcriptome profiling in the Hemipteran Insect Nilaparvata lugens. Front. Physiol. 2020, 11, 106. [Google Scholar] [CrossRef]
- Hao, K.; Jarwar, A.R.; Ullah, H.; Tu, X.B.; Nong, X.Q.; Zhang, Z.H. Transcriptome sequencing reveals potential mechanisms of the maternal effect on egg diapause induction of Locusta migratoria. Int. J. Mol. Sci. 2019, 20, 1974. [Google Scholar] [CrossRef]
- Chen, Y.R.; Jiang, T.; Zhu, J.; Xie, Y.C.; Tan, Z.C.; Chen, Y.H.; Tang, S.M.; Hao, B.F.; Wang, S.P.; Huang, J.S. Transcriptome sequencing reveals potential mechanisms of diapause preparation in bivoltine silkworm Bombyx mori (Lepidoptera: Bombycidae). Comp. Biochem. Physiol.-D Genom. Proteom. 2017, 24, 68–78. [Google Scholar] [CrossRef]
- Chen, J.; Guo, Y.S.; Huang, S.M.; Zhan, H.R.; Zhang, M.F.; Wang, J.W.; Shu, Y.H. Integration of transcriptome and proteome reveals molecular mechanisms underlying stress responses of the cutworm, Spodoptera litura, exposed to different levels of lead (Pb). Chemosphere 2021, 283, 131205. [Google Scholar] [CrossRef]
- Liu, Y.C.; Su, H.; Li, R.Q.; Li, X.T.; Xu, Y.S.; Dai, X.P.; Zhou, Y.Y.; Wang, H.B. Comparative transcriptome analysis of Glyphodes pyloalis Walker (Lepidoptera: Pyralidae) reveals novel insights into heat stress tolerance in insects. BMC Genom. 2017, 18, 974. [Google Scholar] [CrossRef]
- Kandasamy, T.; Ekbal, S.; Kumari, K.; Lohot, V.D.; Mohanasundaram, A.; Sharma, K.K. Unraveling bacterial diversity of the Indian Lac Insect Kerria lacca (Kerr) using next generation sequencing. Int. J. Trop. Insect Sci. 2022, 42, 2365–2372. [Google Scholar] [CrossRef]
- Sharma, K.K.; Jaiswal, A.K. Microbial flora associated with lac insects. In Recent Advances in Lac Culture; Kumar, K.K., Ramani, R., Sharma, K.K., Eds.; Indian Lac Research Institute: Ranchi, India, 2011; pp. 22–33. [Google Scholar]
- Shamim, G.; Ranjan, S.K.; Kandasamy, T.; Sharma, K.K.; Ramani, R. Bacterial flora associated with Kerria lacca (Kerr). Indian J. Entomol. 2017, 79, 41–48. [Google Scholar] [CrossRef]
- Vashishtha, A.; Sharama, K.K.; Lakhanpaul, S. Co-existence, phylogeny and putative role of Wolbachia and yeast-like symbiont (YLS) in Kerria lacca (Kerr). Curr. Microbiol. 2011, 63, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, T.; Sharma, K.K. Interaction of lac insects with microbes. In Commercial Entomology; Kumar, A., Kumar, N., Chand, H., Eds.; New India Publishing Agency: New Delhi, India, 2019; pp. 129–139. [Google Scholar]
- Chen, D.Q.; Montllor, C.B.; Purcell, A.H. Fitness effects of two facultative endosymbiotic bacteria on the pea aphid, Acyrthosiphon pisum, and the blue alfalfa aphid, A. kondoi. Entomol. Exp. Appl. 2000, 95, 315–323. [Google Scholar] [CrossRef]
- Fukatsu, T.; Nikoh, N.; Kawai, R.; Koga, R. The secondary endosymbiotic bacterium of the pea aphid Acyrthosiphon pisum (Insecta: Homoptera). Appl. Environ. Microbiol. 2000, 66, 2748–2758. [Google Scholar] [CrossRef]
- Dillon, R.J.; Dillon, V.M. The gut bacteria of insects: Nonpathogenic interactions. Annu. Rev. Entomol. 2004, 49, 71–92. [Google Scholar] [CrossRef]
- Wu, H.X.; Chen, X.M.; Chen, H.; Lu, Q.; Yang, Z.X.; Ren, W.B.; Liu, J.; Shao, S.X.; Wang, C.; King-Jones, K. Variation and diversification of the microbiome of Schlechtendalia chinensis on two alternate host plants. PLoS ONE 2018, 13, e0200049. [Google Scholar] [CrossRef]
- Ahmad, A.; Kaushik, S.; Ramamurthy, V.V.; Lakhanpaul, S.; Ramani, R.; Sharma, K.K.; Vidyarthi, A.S. Mouthparts and stylet penetration of the lac insect Kerria lacca (Kerr) (Hemiptera: Tachardiidae). Arthropod Struct. Dev. 2012, 41, 435–441. [Google Scholar] [CrossRef]
- Kerr, J. Natural history of the insect which produces the gum lacca. Philos. Trans. R. Soc. 1782, 71, 374–382. [Google Scholar]
- Sangha, K.S.; Shera, P.S.; Sharma, S.; Kaur, R.; Thakur, A.; Kaur, H. Studies on the population dynamics and host range of Lac insect in different agro-climatic zones of Punjab (India). J. Entomol. Zool. Stud. 2019, 7, 183–189. [Google Scholar]
- Roonwal, M.L.; Raizada, M.B.; Chatterji, R.N.; Singh, B. Descriptive Account of the Host-Plants of the Lac Insect, Laccifer lacca (Kerr) and the Allied Plants in the Indian Region—Part 2; Indian Lac Cess Committee: Ranchi, India, 1958; p. 101. [Google Scholar]
- Ben-Dov, Y. A Systematic Catalogue of Eight Scale Insect Families (Hemiptera: Coccoidea) of the World: Aclerdidae, Asterolecaniidae, Beesoniidae, Carayonemidae, Conchaspididae, Dactylopiidae, Kerriidae and Lecanodiaspididae; Elsevier: Amsterdam, The Netherlands, 2006; p. 368. [Google Scholar]
- Chen, H.; Chen, X.M.; Feng, Y.; Yang, H.; He, R.; Zhang, W.F.; Yang, Z.X. Molecular phylogeny and biogeography of lac insects (Hemiptera: Kerriidae) inferred from nuclear and mitochondrial gene sequences. Mol. Biol. Rep. 2013, 40, 5943–5952. [Google Scholar] [CrossRef]
- Bhatnagar, P.; Prajapati, S.; Lodhi, B.; Bhalawi, R.; Aarmo, B.S. Studies on lac host plant occurrence in different Agro-climatic zones of Madhya Pradesh. Int. J. Ecol. Environ. Sci. 2020, 2, 197–204. [Google Scholar]
- Vaibhav, D.L.; Jyotirmoy, G.; Thamilarasi, K.; Mohanasundaram, A.; Rajagopal, N.N.; Thakur, V.V.; Sharma, K.K. Exploring the utilization potential of Ficus species for cultivation of lac insects (Kerria species) in India. Agric. Food 2019, 1, 1–6. [Google Scholar]
- Ferdousee, N.; Julker, N.M.; Hoque, A.; Mohiuddin, M. Lac production and its economic return to rural economy in Rajshahi Division, Bangladesh. In Proceedings of the International Conference on Environmental Aspects of Bangladesh (ICEAB10), Kitakyushu, Japan, 4 September 2010; pp. 69–72. [Google Scholar]
- Chen, Y.Q.; Li, Q.; Chen, Y.L.; Wang, S.M.; Yang, Y.C. Lac-production, arthropod biodiversity and abundance, and pesticide use in Yunnan Province, China. Trop. Ecol. 2010, 51, 255–263. [Google Scholar]
- Singh, J.P.; Jaiswal, A.K.; Monobrullah, M.D. Green lacewing (Chrysoperla zastrowi arabica) incidence on Indian lac insect (Kerria lacca). Indian J. Agric. Sci. 2011, 81, 1087–1089. [Google Scholar]
- Singh, J.P.; Jaiswal, A.K.; Monobrullah, M.D. Safety evaluation of some newer pesticides against lac insect (Kerria lacca) for managing predators. Indian J. Agric. Sci. 2011, 81, 465–469. [Google Scholar]
- Sarvade, S.; Panse, R.K.; Rajak, S.K.; Upadhyay, V.B. Impact of biotic and abiotic factors on lac production and peoples’ livelihood improvement in India-An overview. J. Appl. Nat. Sci. 2018, 10, 894–904. [Google Scholar] [CrossRef]
- Jaiswal, A.K.; Bhattacharya, A.; Kumar, S.; Singh, J.P. Evaluation of Bacillus thuringiensis Berliner subsp. kurstaki for management of lepidopteran pests of lac insect. Entomon 2008, 33, 65–69. [Google Scholar]
- Sharma, K.K.; Kumari, K.; Lakhanpaul, S. Super parasitism in Indian lac insect, Kerria lacca (Kerr) and its implication on fecundity and resin producing efficiency of its two strains. Entomon 2007, 32, 33. [Google Scholar]


| Character | Female | Male |
|---|---|---|
| Metamorphosis | paurometabolous | holometabolous |
| Body | larger and degenerated | smaller and not degenerated as female |
| Head, thorax, and abdomen | indistinct | distinct |
| Eyes | absent | present |
| Antennae | vestigial | well-develop |
| Legs | absent | present |
| Wings | absent | present/absent |
| Mouthparts | present | absent |
| Movement | enclosed in lac secretion and neotenic | not enclosed in lac secretion and move freely |
| Species | Host plant | References |
|---|---|---|
| K. chinensis | Cajanus indicus, Schleichera sp., and some other host plants | [86] |
| K. lacca | Ziziphus mauritiana, Butea monosperma, Ficus religiosa, F. indica, and some other host plants | [15,87] |
| K. nepalensis | Litchi chinensis, Dalbergia cochinchinensis, Ficus sp. | [26] |
| K. pusana | Ziziphus jujube, Butea frondosa, B. monosperma, Schleichera oleosa, Samanea saman, Acacia montana | [3,15,86,87] |
| K. ruralis | Mallotus philippinensis, Pueraria tonkinensis | [3,86] |
| K. sindica | Acacia arabica, Albizia lebbeck, Ziziphus jujube, Ficus racemosa, F. glabella | [86,87] |
| K. yunnanensis | Dalbergia obtusifolia, D. assamica, D. mimosoides, Schleichera oleosa, Ficus microcarpa | [86,87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bashir, N.H.; Chen, H.; Munir, S.; Wang, W.; Chen, H.; Sima, Y.-K.; An, J. Unraveling the Role of Lac Insects in Providing Natural Industrial Products. Insects 2022, 13, 1117. https://doi.org/10.3390/insects13121117
Bashir NH, Chen H, Munir S, Wang W, Chen H, Sima Y-K, An J. Unraveling the Role of Lac Insects in Providing Natural Industrial Products. Insects. 2022; 13(12):1117. https://doi.org/10.3390/insects13121117
Chicago/Turabian StyleBashir, Nawaz Haider, Huanhuan Chen, Shahzad Munir, Weiwei Wang, Hang Chen, Yong-Kang Sima, and Jiandong An. 2022. "Unraveling the Role of Lac Insects in Providing Natural Industrial Products" Insects 13, no. 12: 1117. https://doi.org/10.3390/insects13121117
APA StyleBashir, N. H., Chen, H., Munir, S., Wang, W., Chen, H., Sima, Y.-K., & An, J. (2022). Unraveling the Role of Lac Insects in Providing Natural Industrial Products. Insects, 13(12), 1117. https://doi.org/10.3390/insects13121117







