Kairomonal Effect of Aphid Alarm Pheromones and Analogs on the Parasitoid Diaeretiella rapae
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Culture of Parasitoids
2.2. Chemicals
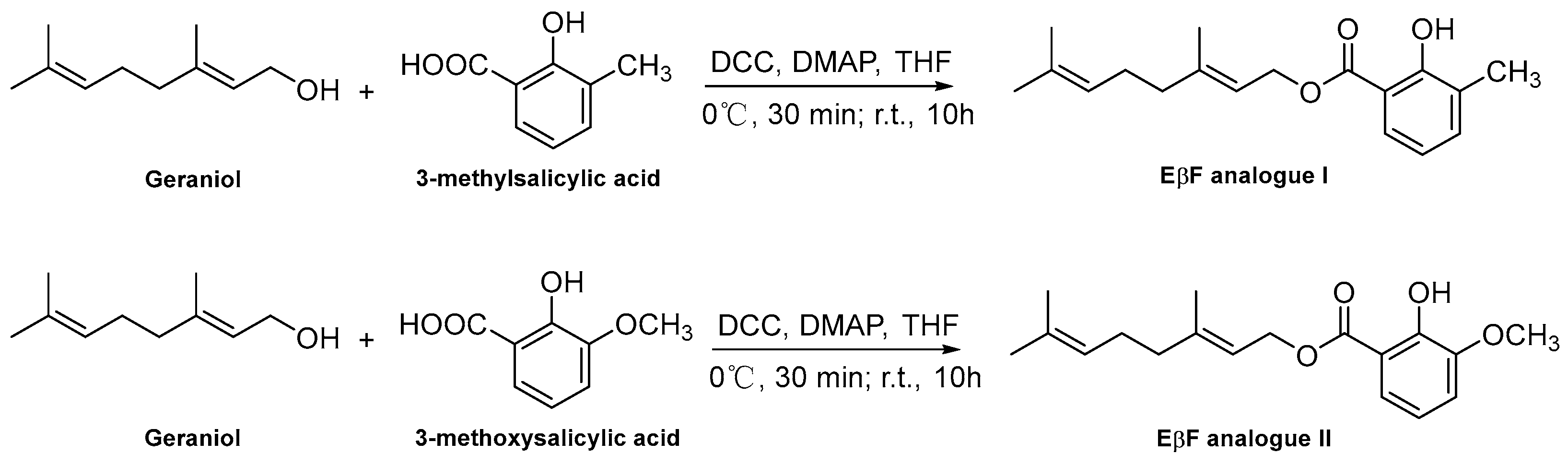
2.3. Synthesis Procedure of EβF Analogs
2.4. Olfactometer Bioassays
2.5. Behavioral Response Time Bioassay
2.6. Statistical Analysis
3. Results
3.1. Synthesis of EβF Analogs
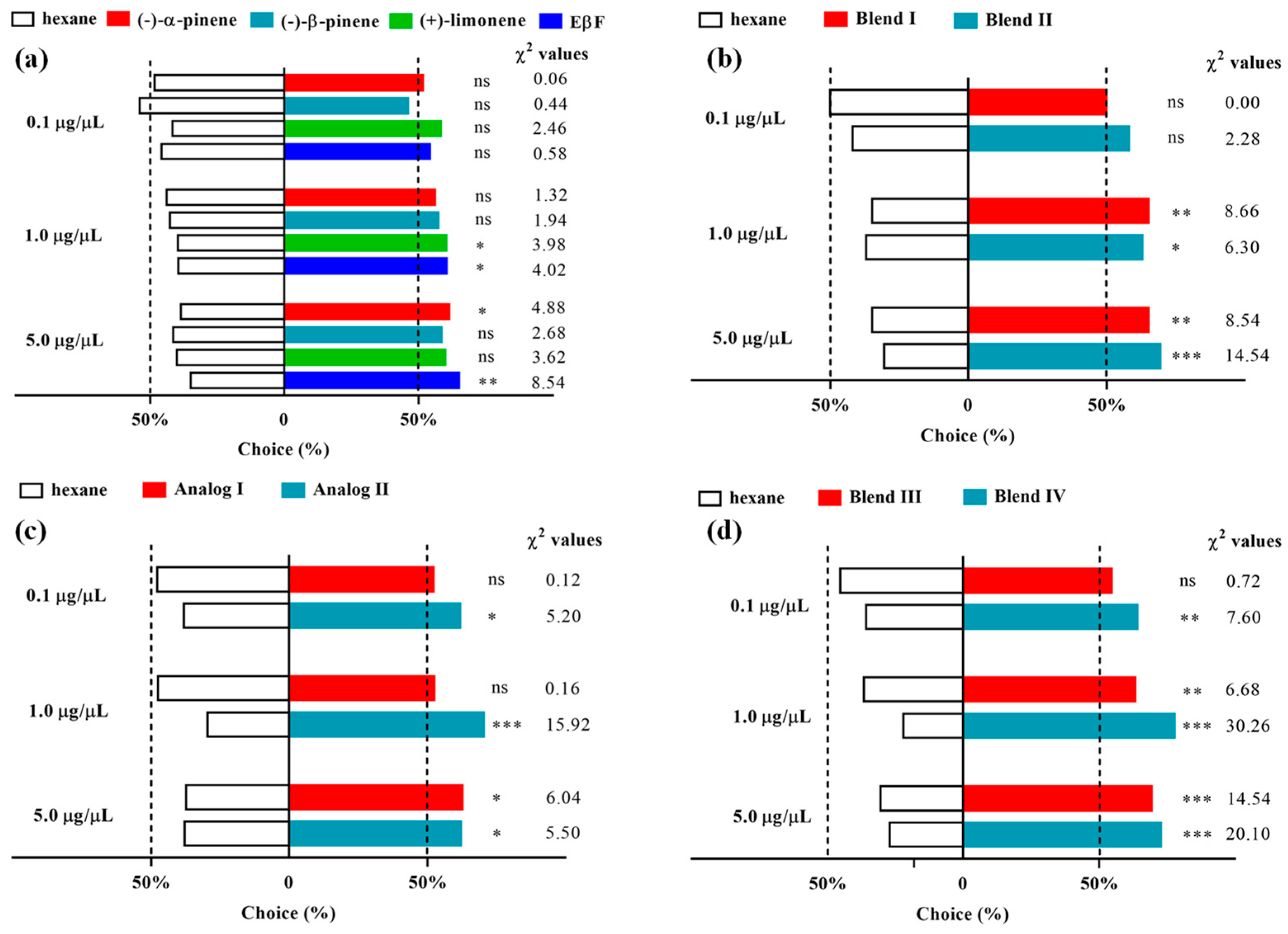
3.2. Olfactometer Bioassay: Response of D. rapae to Single Components of Alarm Pheromones
3.3. Olfactory Response of D. rapae to the Blend of Alarm Pheromones
3.4. Olfactory Response of D. rapae to Single EβF Analogs
3.5. Olfactory Response of D. rapae to the Blend of EβF Analogs
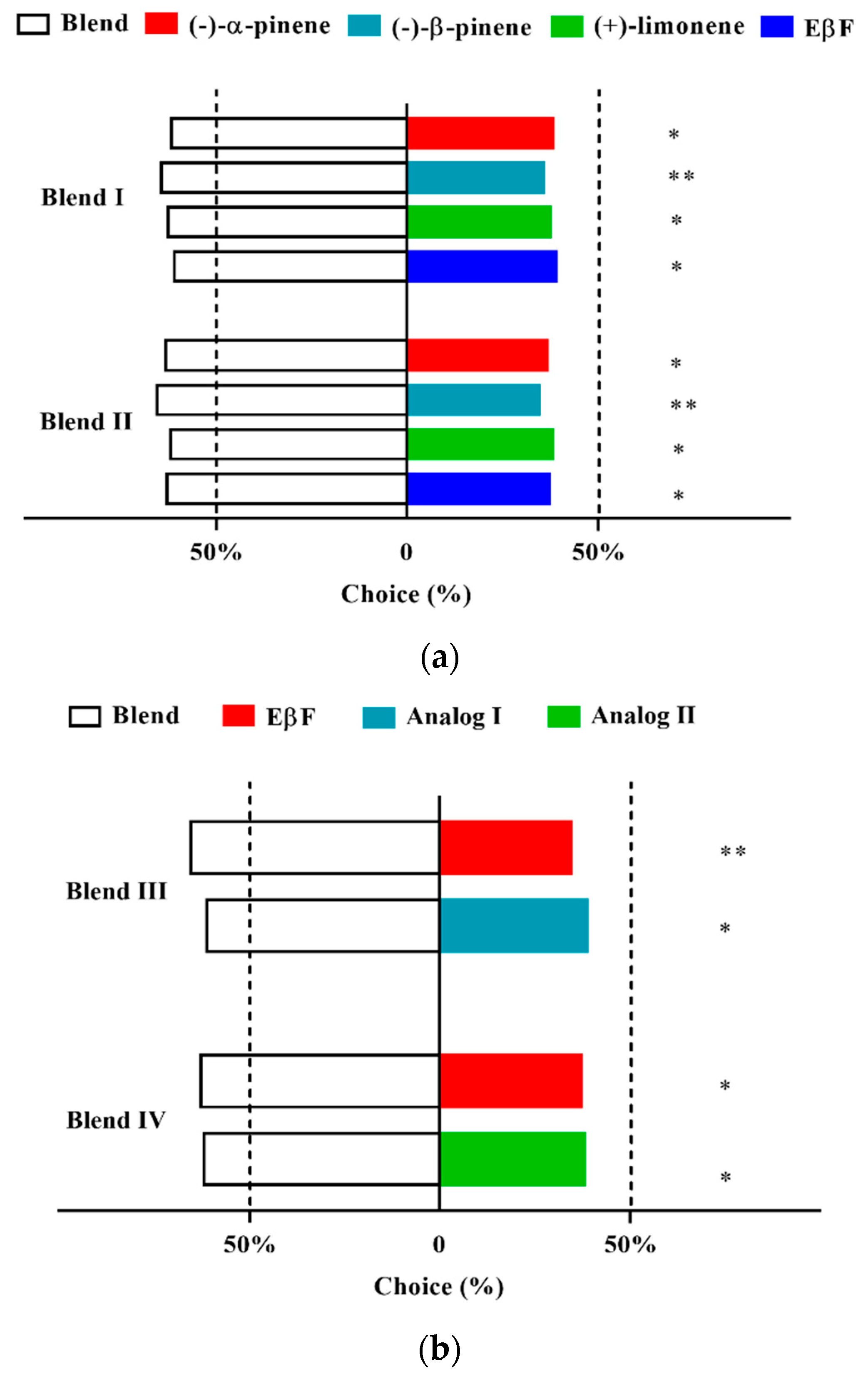
3.6. Two-Way Choice of D. rapae for Aphid Alarm Pheromones and Analogs
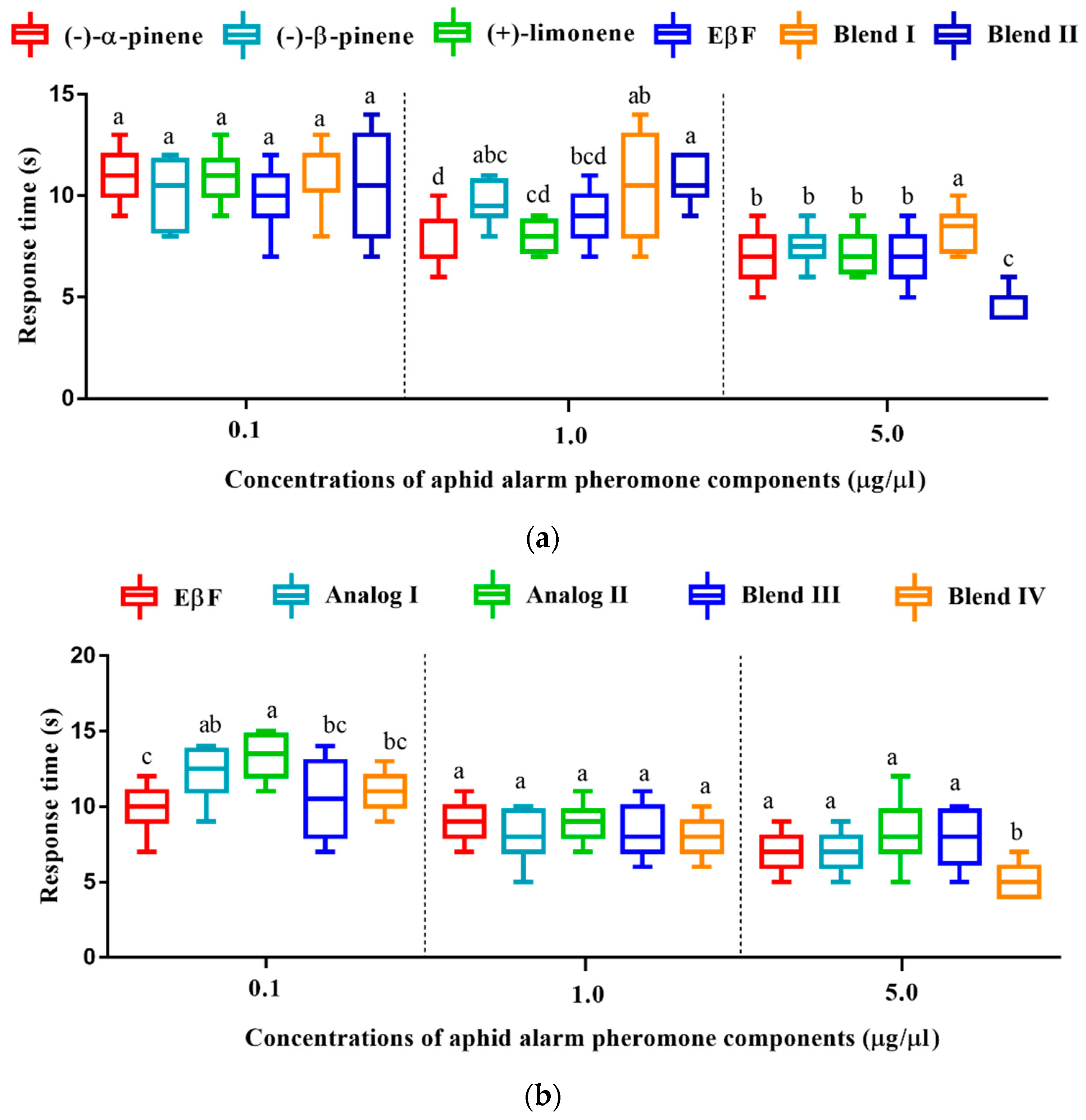
3.7. Olfactory Response Time of D. rapae to Aphid Alarm Pheromones and Analogs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pickett, J.A.; Allemann, R.K.; Birkett, M.A. The semiochemistry of aphids. Nat. Prod. Rep. 2013, 30, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, J.T.; Zhang, Y.J.; Chen, J.L.; Li, X.C.; Liang, P.; Gao, X.W.; Zhou, J.-J.; Gu, S.H. Coordinative mediation of the response to alarm pheromones by three odorant binding proteins in the green peach aphid Myzus persicae. Insect Biochem. Molec. Biol. 2021, 103, 130528. [Google Scholar] [CrossRef] [PubMed]
- Al-Jalely, B.H.; Xu, W. Olfactory sensilla and olfactory genes in the parasitoid wasp Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae). Insects 2021, 12, 11. [Google Scholar] [CrossRef]
- Qin, Y.G.; Yang, Z.K.; Song, D.L.; Wang, Q.; Gu, S.H.; Li, W.H.; Duan, H.X.; Zhou, J.J.; Yang, X.L. Bioactivities of synthetic salicylate-substituted carboxyl (E)-β-Farnesene derivatives as ecofriendly agrochemicals and their binding mechanism with potential targets in aphid olfactory system. Pest Manag. Sci. 2020, 76, 2465–2472. [Google Scholar] [CrossRef]
- Wang, B.; Dong, W.Y.; Li, H.M.; D’Onofrio, C.; Bai, P.H.; Chen, R.P.; Yang, L.L.; Wu, J.N.; Wang, X.Q.; Wang, B.; et al. Molecular basis of (E)-β-farnesene-mediated aphid location in the predator Eupeodes corollae. Curr. Biol. 2022, 32, 951–962.e7. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.L.; Francis, F.; Heuskin, S.; Lognay, G.; Liu, Y.J.; Dong, J.; Chen, J.L.; Song, X.M.; Liu, Y. The functional significance of E-β-Farnesene: Does it influence the populations of aphid natural enemies in the fields? Biol. Control 2012, 60, 108–112. [Google Scholar] [CrossRef]
- Dewhirst, S.Y.; Pickett, J.A.; Hardie, J. Chapter Twenty-two—Aphid pheromones. In Vitamins & Hormones; Litwack, G., Ed.; Pheromone; Academic Press: Cambridge, MA, USA, 2010; Volume 83, pp. 551–574. [Google Scholar]
- Pickett, J.A.; Griffiths, D.C. Composition of aphid alarm pheromones. J. Chem. Ecol. 1980, 6, 349–360. [Google Scholar] [CrossRef]
- Francis, F.; Vandermoten, S.; Verheggen, F.; Lognay, G.; Haubruge, E. Is the (E)-β-farnesene only volatile terpenoid in aphids? J. Appl. Entomol. 2005, 129, 6–11. [Google Scholar] [CrossRef]
- Song, X.; Qin, Y.G.; Yin, Y.; Li, Z.X. Identification and behavioral assays of alarm pheromone in the vetch aphid Megoura viciae. J. Chem. Ecol. 2021, 47, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Pickett, J.A.; Wadhams, L.J.; Woodcock, C.M.; Hardie, J. The chemical ecology of aphids. Annu. Rev. Entomol. 1992, 37, 67–90. [Google Scholar] [CrossRef]
- Fan, J.; Zhang, Y.; Francis, F.; Cheng, D.F.; Sun, J.R.; Chen, J.L. Orco mediates olfactory behaviors and winged morph differentiation induced by alarm pheromone in the grain aphid, Sitobion avenae. Insect Biochem. Molec. Biol. 2015, 64, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Wen, X.X.; Hong, X.D.; Jiang, X.; Zhang, Y.; Yu, W.J.; Jiang, S.S.; Sun, J.R.; Chen, J.L. Identification of an intraspecific alarm pheromone and two conserved odorant-binding proteins associated with (E)-β-farnesene perception in aphid Rhopalosiphum padi. J. Insect Physiol. 2017, 101, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.B.; Wang, B.; Grossi, G.; Falabella, P.; Liu, Y.; Yan, S.C.; Lu, J.; Xi, J.H.; Wang, G.R. Molecular basis of alarm pheromone detection in aphids. Curr. Biol. 2017, 27, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Nishino, C.; Bowers, W.S.; Montgomery, M.E.; Nault, L.R. Aphid alarm pheromone mimics: The nor-farnesenes. Appl. Entomol. Zool. 1976, 11, 340–343. [Google Scholar] [CrossRef]
- Sun, Y.F.; De Biasio, F.; Qiao, H.L.; Iovinella, I.; Yang, S.X.; Ling, Y.; Riviello, L.; Battaglia, D.; Falabella, P.; Yang, X.L.; et al. Two odorant-binding proteins mediate the behavioural response of aphids to the alarm pheromone (E)-β-farnesene and structural analogues. PLoS ONE 2012, 7, e32759. [Google Scholar] [CrossRef]
- Qin, Y.G.; Yang, Z.K.; Zhou, J.-J.; Zhang, S.Y.; Pan, S.X.; Liu, Y.; Gu, S.H.; Duan, H.X.; Yang, X.L. Effects of carboxyl and acylamino linkers in synthetic derivatives of aphid alarm pheromone (E)-β-farnesene on repellent, binding and aphicidal activity. J. Mol. Struct. 2022, 1268, 133658. [Google Scholar] [CrossRef]
- Qin, Y.G.; Zhang, J.P.; Song, D.L.; Duan, H.X.; Li, W.H.; Yang, X.L. Novel (E)-β-farnesene analogues containing 2-nitroimino-hexahydro-1,3,5-triazine: Synthesis and biological activities evaluation. Molecules 2016, 21, 825. [Google Scholar] [CrossRef]
- Zhang, J.P.; Qin, Y.G.; Dong, Y.W.; Song, D.L.; Duan, H.X.; Yang, X.L. Synthesis and biological activities of (E)-β-farnesene analogues containing 1,2,3-thiadiazole. Chinese Chem. Lett. 2017, 28, 372–376. [Google Scholar] [CrossRef]
- Heuskin, S.; Lorge, S.; Godin, B.; Leroy, P.; Frere, I.; Verheggen, F.J.; Haubruge, E.; Wathelet, J.P.; Mestdagh, M.; Hance, T.; et al. Optimisation of a semiochemical slow-release alginate formulation attractive towards Aphidius ervi Haliday parasitoids. Pest Manag. Sci. 2012, 68, 127–136. [Google Scholar] [CrossRef]
- Francis, F.; Lognay, G.; Haubruge, E. Olfactory responses to aphid and host plant volatile releases: (E)-β-farnesene an effective kairomone for the predator Adalia bipunctata. J. Chem. Ecol. 2004, 30, 741–755. [Google Scholar] [CrossRef]
- Micha, S.G.; Wyss, U. Aphid alarm pheromone (E)-β-farnesene: A host finding kairomone for the aphid primary parasitoid Aphidius uzbekistanicus (Hymenoptera: Aphidiinae). Chemoecology 1996, 7, 132–139. [Google Scholar] [CrossRef]
- Du, Y.J.; Poppy, G.M.; Powell, W.; Pickett, J.A.; Wadhams, L.J.; Woodcock, C.M. Identification of semiochemicals released during aphid feeding that attract parasitoid Aphidius ervi. J. Chem. Ecol. 1998, 24, 1355–1368. [Google Scholar] [CrossRef]
- Turlings, T.C.J.; Erb, M. Tritrophic interactions mediated by herbivore-induced plant volatiles: Mechanisms, ecological relevance, and application potential. Annu. Rev. Entomol. 2018, 63, 433–452. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.J.; Cividanes, F.J.; Pedroso, E.C.; Sala, S.R.D. Host quality of different aphid species for rearing Diaeretiella rapae (McIntosh) (Hymenoptera: Braconidae). Neotrop. Entomol. 2011, 40, 477–482. [Google Scholar] [PubMed]
- Kant, R.; Minor, M.A. Parasitoid Diaeretiella rapae (Hymenoptera: Braconidae) adjusts reproductive strategy when competing for hosts. Environ. Entomol. 2017, 46, 521–527. [Google Scholar] [CrossRef]
- Soni, S.; Kumar, S.; Singh, R.; Badiyala, A.; Chandel, R.S. Aphid parasitoid, Diaeretiella rapae (McIntosh) (Hymenoptera: Braconidae): Opportunities for its use in integrated management of aphids infesting rapeseed-mustard in north-western Indian Himalayas. Crop. Prot. 2022, 151, 105819. [Google Scholar] [CrossRef]
- Beale, M.H.; Birkett, M.A.; Bruce, T.J.A.; Chamberlain, K.; Field, L.M.; Huttly, A.K.; Martin, J.L.; Parker, R.; Phillips, A.L.; Pickett, J.A.; et al. Aphid alarm pheromone produced by transgenic plants affects aphid and parasitoid behavior. Proc. Natl. Acad. Sci. USA 2006, 103, 10509–10513. [Google Scholar] [CrossRef]
- Wang, G.P.; Yu, X.D.; Fan, J.; Wang, C.S.; Xia, L.Q. Expressing an (E)-β-farnesene synthase in the chloroplast of tobacco affects the preference of green peach aphid and its parasitoid. J. Integr. Plant Biol. 2015, 57, 770–782. [Google Scholar] [CrossRef]
- Foster, S.P.; Denholm, I.; Thompson, R.; Poppy, G.M.; Powell, W. Reduced response of insecticide-resistant aphids and attraction of parasitoids to aphid alarm pheromone; a potential fitness trade-off. B. Entomol. Res. 2005, 95, 37–46. [Google Scholar] [CrossRef]
- Moayeri, H.R.S.; Rasekh, A.; Enkegaard, A. Influence of cornicle droplet secretions of the cabbage aphid, Brevicoryne brassicae, on parasitism behavior of naïve and experienced Diaeretiella rapae. Insect Sci. 2014, 21, 56–64. [Google Scholar] [CrossRef]
- Goelen, T.; Vuts, J.; Sobhy, I.S.; Wackers, F.; Caulfield, J.C.; Birkett, M.A.; Rediers, H.; Jacquemyn, H.; Lievens, B. Identification and application of bacterial volatiles to attract a generalist aphid parasitoid: From laboratory to greenhouse assays. Pest Manag. Sci. 2021, 77, 930–938. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Clark, R.E.; Fu, Z.; Lee, B.W.; Crowder, D.W. Insect alarm pheromones in response to predators: Ecological trade-offs and molecular mechanisms. Insect Biochem. Molec. Biol. 2021, 128, 103514. [Google Scholar] [CrossRef] [PubMed]
- Ameixal, O.M.C.C.; Kindlmann, P. Effect of synthetic and plant-extracted aphid pheromones on the behaviour of Aphidius colemani. J. Appl. Entomol. 2012, 136, 292–301. [Google Scholar] [CrossRef]
- Alhmedi, A.; Haubruge, E.; Francis, F. Identification of limonene as a potential kairomone of the harlequin ladybird Harmonia axyridis (Coleoptera: Coccinellidae). Eur. J. Entomol. 2010, 107, 541–548. [Google Scholar] [CrossRef]
- Li, Z.Q.; Zhang, S.; Cai, X.M.; Luo, J.Y.; Dong, S.L.; Cui, J.J.; Chen, Z.M. Three odorant binding proteins may regulate the behavioural response of Chrysopa pallens to plant volatiles and the aphid alarm pheromone (E)-β-farnesene. Insect Mol. Biol. 2017, 26, 255–265. [Google Scholar] [CrossRef]
- Park, K.C.; Hardie, J. Electroantennogram responses of aphid nymphs to plant volatiles. Physiol. Entomol. 2003, 28, 215–220. [Google Scholar] [CrossRef]
- Visser, J.H.; Yan, F.S. Electroantennogram responses of the grain aphids Sitobion avenae (F.) and Metopolophium dirhodum (Walk.) (Hom., Aphididae) to plant odour components. J. Appl. Entomol. 1995, 119, 539–542. [Google Scholar] [CrossRef]
- Leroy, P.D.; Schillings, T.; Farmakidis, J.; Heuskin, S.; Lognay, G.; Verheggen, F.J.; Brostaux, Y.; Haubruge, E.; Francis, F. Testing semiochemicals from aphid, plant and conspecific: Attraction of Harmonia axyridis. Insect Sci. 2011, 19, 372–382. [Google Scholar] [CrossRef]
- Jiang, H.; Kong, J.J.; Chen, H.C.; Xiang, Z.Y.; Zhang, W.P.; Han, Z.D.; Liao, P.C.; Lee, Y.I. Cypripedium subtropicum (Orchidaceae) employs aphid colony mimicry to attract hoverfly (Syrphidae) pollinators. New Phytol. 2020, 227, 1213–1221. [Google Scholar] [CrossRef]
- Ayelo, P.M.; Pirk, C.W.W.; Yusuf, A.A.; Chailleux, A.; Mohamed, S.A.; Deletre, E. Exploring the kairomone-based foraging behaviour of natural enemies to enhance biological control: A review. Front. Ecol. Evol. 2021, 9, 641974. [Google Scholar] [CrossRef]
- Vaello, T.; Casas, J.L.; Pineda, A.; de Alfonso, I.; Marcos-Garcia, M.A. Olfactory response of the predatory bug Orius laevigatus (Hemiptera: Anthocoridae) to the aggregation pheromone of its prey, Frankliniella occidentalis (Thysanoptera: Thripidae). Environ. Entomol. 2017, 46, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Maeda, T.; Kishimoto, H.; Wright, L.C.; James, D.G. Mixture of synthetic herbivore-induced plant volatiles attracts more Stethorus punctum picipes (Casey) (Coleoptera: Coccinellidae) than a single volatile. J. Insect Behav. 2015, 28, 126–137. [Google Scholar] [CrossRef]
- Jones, V.P.; Horton, D.R.; Mills, N.J.; Unruh, T.R.; Baker, C.C.; Melton, T.D.; Miliczky, E.; Steffan, S.A.; Shearer, P.W.; Amarasekare, K.G. Evaluating plant volatiles for monitoring natural enemies in apple, pear and walnut orchards. Biol. Control 2016, 102, 53–65. [Google Scholar] [CrossRef]
| Compounds | Concentrations (µg/µL) | ||
|---|---|---|---|
| 0.1 | 1.0 | 5.0 | |
| (–)-α-pinene | 5.6 ± 4.8 lmn | 10.0 ± 4.4 i–n | 21.4 ± 2.9 e–i |
| (–)-β-pinene | 1.1 ± 1.9 n | 14.7 ± 1.3 h–n | 18.9 ± 4.8 g–m |
| (+)-limonene | 16.9 ± 7.0 h–n | 21.1 ± 3.4 e–i | 19.2 ± 9.5 f–m |
| EβF | 6.1 ± 1.9 k–n | 21.9 ± 1.3 e–k | 33.6 ± 0.5 b–g |
| Analog I | 4.4 ± 4.2 mn | 4.4 ± 1.0 mn | 25.8 ± 3.8 d–i |
| Analog II | 25.0 ± 3.0 d–i | 39.4 ± 2.5 bcd | 23.1 ± 2.4 e–j |
| Blend I | 9.7 ± 5.5 i–n | 34.2 ± 6.5 b–g | 36.7 ± 6.0 bcde |
| Blend II | 6.9 ± 8.7 j–n | 30.8 ± 4.3 c–h | 42.2 ± 4.7 bc |
| Blend III | 8.3 ± 3.8 j–n | 35.3 ± 13.5 b–f | 40.8 ± 3.0 bcd |
| Blend IV | 25.6 ± 7.9 d–i | 56.9 ± 1.7 a | 47.8 ± 9.6 ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, Y.; Zhang, S.; Li, Z. Kairomonal Effect of Aphid Alarm Pheromones and Analogs on the Parasitoid Diaeretiella rapae. Insects 2022, 13, 1055. https://doi.org/10.3390/insects13111055
Qin Y, Zhang S, Li Z. Kairomonal Effect of Aphid Alarm Pheromones and Analogs on the Parasitoid Diaeretiella rapae. Insects. 2022; 13(11):1055. https://doi.org/10.3390/insects13111055
Chicago/Turabian StyleQin, Yaoguo, Shangyang Zhang, and Zhengxi Li. 2022. "Kairomonal Effect of Aphid Alarm Pheromones and Analogs on the Parasitoid Diaeretiella rapae" Insects 13, no. 11: 1055. https://doi.org/10.3390/insects13111055
APA StyleQin, Y., Zhang, S., & Li, Z. (2022). Kairomonal Effect of Aphid Alarm Pheromones and Analogs on the Parasitoid Diaeretiella rapae. Insects, 13(11), 1055. https://doi.org/10.3390/insects13111055











