Simple Summary
While little is known about their phylogenetic relationship, the species Lispe are predators inhabiting semi-aquatic environments. Here, we undertook the first comprehensive study to establish the phylogeny of the genus Lispe, and to elucidate the intrageneric relationships of the known species group system. The monophyly of the genus Lispe is well supported, and the validity of the 11 species group is clarified, while the evolutionary causes remains unclear. This study will provide a better understanding of the phylogenetic relationships and evolutionary history of the genus Lispe.
Abstract
Lispe Latreille (Diptera: Muscidae) are a widespread group of predatory flies that inhabit semi-aquatic environments. Previous studies on this genus have mainly focused on morphological classification, so molecular data are entirely lacking, and there has been no attempt at a phylogenetic placement of the genus or the resolution of intragenic relationships. To address the phylogenetic placement of Lispe and to fill its gap in the Tree of Life Web Project, 58 Lispe spp. (covering 11 out of 13 acknowledged Lispe species groups) were selected to reconstruct a phylogeny using Maximum likelihood (ML) estimates, Maximum Parsimony (MP) analyses, and Bayesian inference (BI) based on two mitochondrial protein-coding genes (cytochrome c oxidase subunit I (COXI) and cytochrome b gene (CYTB)) and one nuclear gene (a fragment of the carbamoyl phosphate synthetase region of the CAD gene). The phylogenetic analyses indicated that the monophyletic Lispe is the sister group of the monophyletic Limnophora, together forming the tribe Limnophorini under the subfamily Coenosiinae. Three generic categories are proven obsolete: Chaetolispa Malloch, Lispacoenosia Snyder, and Xenolispa Malloch. Within the genus, the validity of 11 species groups is clarified by both molecular and morphological data. This study provides a sound basis for continuing intergeneric and intrageneric research into this fascinating and widespread genus.
1. Introduction
Muscidae (Diptera: Calyptratae) is by far the most species-rich family in the muscoid grade of the Calyptratae, with more than 5000 species from around 180 genera recorded in all biogeographic regions [1,2,3,4]. Remarkably, Muscid flies have conquered both terrestrial and aquatic habitats and have evolved various feeding habits including saprophagy, coprophagy, herbivory, and predation [4,5]. The evolutionary history of the Muscidae has therefore been the focal subject of continuous re-evaluation and reinterpretation at different taxonomic levels (subfamily, tribe, and genus) [3,4,5,6,7,8]. This research has made it possible to establish stable classifications and to begin to understand the evolutionary history of the family as a whole. However, there are still some groups of muscoids that have been largely neglected in this regard; for example, the taxonomic relationships, morphological evolution, and natural history of many aquatic muscoids, such as the genus Lispe, remains poorly understood compared to other taxa.
Lispe Latreille 1796 is widely distributed in all biogeographic regions with the exception of Antarctica and New Zealand, and includes approximately 200 described species [9,10]. Adult Lispe are active predators of small arthropods and also frequently scavenge on arthropod remains [11,12,13,14]. In both larval and adult stages, Lispe flies inhabit semi-aquatic environments, ranging from flowing or standing, to fresh or salty, and have thus evolved to tolerate exceptionally variable environmental conditions [15]. Adults are commonly found close to the water bodies, resting or running quickly on mud or sand, in search of mating partners [16,17] and prey. Importantly, Lispe play vital ecological roles in the functioning of littoral ecosystems, which makes them useful for monitoring water quality [18]. Yet, despite their ecological, evolutionary, and phylogenetic importance, no comprehensive phylogeny based on modern cladistic analysis has been proposed for Lispe as a whole.
The characteristic diagnoses of Lispe are as follows: broad frons in both sexes, two upper orbital setae, palpi enlarged from slight to strong, almost spoon-like, setulae present on the anepimeron, katepisternal bristles 1:1 or 1:2 [11,19,20,21]. Within the genus, three sets of morphological traits are particularly useful for specific classification, including thoracic and leg chaetotaxy, leg external modification, and male terminalia [9,11,21,22,23,24,25]. The division of the genus Lispe into species groups based on these three sets of characters were offered in several papers [9,11,21,22,24,26,27,28], although all of these studies were only based on morphological approaches.
The first division of Lispe into species groups was proposed by Snyder [11], which outlined three Nearctic groups including the L. tentaculata-, L. palposa-, and L. uliginosa-groups [11]. In the work on Palaearctic Muscidae, Hennig [21] agreed with Snyder [11] and expanded Snyder’s species groups with several Palaearctic species, offering three new species groups for the Palaearctic fauna (the L. caesia-, L. longicollis-, and L. scalaris-groups) [21]. Much effort has since been contributed to these six typical Lispe groups: L. tentaculata-group [21,22,26,28], L. palposa-group [9,21], L. uliginosa-group [9,11,24], L. caesia-group [21,23,24,25], L. longicollis-group [21,22,27], and L. scalaris-group [21,22]. These six morphological groups are now well acknowledged by taxonomists [9,22,24,26,27,28]. The definitions and developments of these species groups are summarized as follows.
The initial L. tentaculata-group is composed of three species (L. patellata, L. sociabilis and L. tentaculata) [21]. More species were eventually added to this group, and then Vikhrev [22] merged the L. nana complex with the L. tentaculata-group as the extended L. tentaculata-supergroup [22]. Although Vikhrev [22] had considered the L. nana-group as an independent group with a unique knob-like process on the ventral margin of tergite 3 in males and the absence of pd seta on the third tibia, it shares many characters with the L. tentaculata-group, so Vikhrev [22] also stated: “It is not clear why Hennig [21] had not included L. nana Macquart, 1851 in the L. tentaculata-group”. The extended L. tentaculata-group is characterised by remarkably wide palpi, fore tibia without p, mid tibia with 1 p, hind tibia with ad and weak pd, similar abdomen patterns, and sternite 5 with lateral and median processes. All described members of this group seem to inhabit fresh water. Based on two morphological characteristics (first fore tarsomere modification and phallus shape) and molecular data, Ge et al. divided the group into two subgroups (the L. tentaculata-subgroup and the L. orientalis-subgroup) [28].
The L. palposa-group contains more than 20 species in the world [9,21] and most of them live in brackish or salt-water habitats. The main characters of this group are well-defined, such as mid tibia with one or more ad, hind tibia without av and pd, sternite 5 in one piece and either with a shallow median incision or almost concave apically, cerci nearing closely each other along most of their length, but not fused, and the apices rather slender and pointed.
The L. uliginosa-group was proposed by Snyder [11] for seven species sharing characters such as the first tibia with p, second tibia with 1 ad and 1 pd, third tibia with 1 av, and sternite 5 reduced to a pair of membranous sclerites [9,11,24].
The L. caesia-group [21,23,24,25] was first proposed by Hennig [21] and includes five valid Palaearctic taxa, i.e., L. caesia, L. candicans, L. halophora, L. leucocephala, and L. odessae. Hennig [21] called the L. caesia-group one of the most clearly defined, and pointed out the following group characters: a frontal triangle broad with convex margins, femora with ventral rows of short spines, and an abdomen with a characteristic pattern. However, there is an evident discrepancy in Hennig’s approach to the L. caesia-group; for example, he included L. leucocephala, which has neither a spine on the femora, nor the typical abdominal pattern. Then, some authors extended the volume of the L. caesia-group with several Palaearctic and Oriental species [23,24,25].
For the L. Longicollis-group [21,22,27], members of the old L. longicollis-group were divided into two subgroups (subgroup I = L. longicollis-subgroup, subgroup II = L. assimilis-subgroup) based on morphological differences. According to Vikhrev [27], the subgroup II is further divided into two clades [27]. The first clade consists of L. glabra and L. manicata, large species with scutum mostly subshining, dc 0+2, and prst intraalar setae absent. The second clade is L. assimilis, L. nuba, and L. pacifica, all of which are medium sized species with scutum densely dusted, dc 2+4, and prst intraalar setae present.
The L. scalaris-group is characterized by ac setulae in two rows distinctly separated from scutal setulae, the lower katepisternal setae weak and hairlike [21]. Three species are proposed to form this species-group: L. persica, L. nubilipennis, and L. elegantissima.
In more recent publications, either the volumes of Hennig’s groups have been extended or new groups have been proposed [9,22,23,24,25,27,29,30], such as the L. bivittata-group [24], L. nivalis-group [22,29], L. kowarzi species complex [22], the L. leucospila-group [9,22], L. nicobarensis-group [9], L. rigida-group [9,29], and the L. pygmaea-group [30]. It is important to note that Vikhrev [22,30] used a separate term—“complex”—when raising several small groups. We choose “group” and “subgroup” hereinafter to maintain consistent terminology.
Considering the complex radiation of Lispe throughout the world and into a variety of semi-aquatic habitats, as well as their remarkable behaviours and ecology, there is much to be gained from a molecular phylogeny that can be used as a foundation for evolutionary questions. To address this, we sample and sequence 58 representative species from the genus Lispe (covering 11 out of all 13 proposed species groups), as well as other muscids, aiming to (1) construct the first phylogeny of the genus Lispe, and (2) clarify and supplement intrageneric relationships under the current species group system.
2. Materials and Methods
2.1. Sampling and Molecular Protocols
To investigate the phylogenetic position of Lispe in Muscidae and the validity of the currently proposed species group system, a total of 84 calyptrate species were sampled (Supplementary Table S1), covering four species from Fanniidae and Sarcophagidae as outgroups, and 80 muscid species representing all subfamilies. Among these were 58 representative species from the genus Lispe, covering 11 out of all 13 proposed Lispe species groups. Of all the taxa above, data for 54 Lispe species were newly sequenced and documented, although some sequenced data are discarded due to the low DNA quality of specimens (Supplementary Table S1).
For the newly retrieved species, DNA was extracted from part or all of the specimen using different DNA extracting kits following the manufacturer’s instructions and stored at −20 °C until use (Supplementary Table S2). Three genes have been amplified using either published [4,31,32] or self-designed primers (Table 1).

Table 1.
Primers sequences used for PCR amplication and sequencing.
PCR amplification was carried out with 1 μL genomic DNA, 1 μL of each primer (10 μmol/L), 9.5 μL double distilled water, and 12.5 μL 2 × Es Taq MasterMix (Dye) (Beijing Cowin Biosciencee Co., Ltd., Beijing, China). The PCR protocols were just as described before [31]. To ensure the correct PCR amplification, the PCR products were visualized on 1% agarose gels (dyed with Goldeview), then purified and sequenced in both directions by BGI (Beijing, China), as described before [31].
2.2. Assembling, Aligning and Nucleotide Substitution Saturation Analysis
Raw sequences were edited and trimmed with BioEdit version 7.0.9.0 [35]. After bidirectional assembling of each fragment using SeqMan (DNAStar, Steve ShearDown, 1998–2001 version, DNASTAR Inc., Madison, WI, USA), assemblies were compared with sequences from GenBank through a blast search to identify the accuracy of data. Sequentially, all genes were aligned individually using MAFFT version 7.3.1 [36] as described before [4]. Each alignment was subsequently examined manually using MEGA version 7.0.26 [37].
We measured whether the sequences were saturated (Index of Substitution Saturation, Iss) and thus useful for the phylogenetic reconstruction based on Xia’s test [38] implemented in DAMBE program version 6.4.81 [39]. In addition, nucleotide substitution saturation of each gene and codon position was evaluated by plotting pair-wise transitions and transversions against divergence for all alignments using the DAMBE following the F84 model. Since third codon positions of protein-coding genes (PCGs) are always the most variable [38], substitution saturation was estimated at the third and combined first and second codon positions of PCGs.
2.3. Phylogenetic Analyses
All genes were concatenated together by SequenceMatrix version 1.9.0 [40]. Then, phylogenetic construction was conducted using Maximum Likelihood (ML), Maximum Parsimony (MP), and Bayesian Inference (BI) analyses. For ML analyses, each matrix was subjected to analyses with IQTREE version 1.6.7.1 [41] on its web server [42]. By using the edge-unlinked partition type, the best-fit substitution models for different genes were assigned automatically (Table 2). After tree searching, a standard bootstrap analysis was performed. To estimate the best partitioning scheme and nucleotide substitution models for a BI analysis of each dataset, we used PartitionFinder version 2.1.1 [43] implemented in CIPRES (Cyberinfrastructure for Phylogenetic Research) Science Gateway [44], with “branchlenths” set to unlinked, “models” as mrbayes, “model selection” using aicc, and search for greedy. Then, Bayesian inference was performed with MrBayes v3.2.6 on CIPRES, as described before [31]. The MP analyses were carried out in TNT version 1.5 [45] with the same parameters from a previous study, except for initial addseqs = 13 [4].

Table 2.
The loci information, substitution saturation analysis and partitioning scheme for the dataset based on the genes of a basic tree and the respective best models. A: phylogenetic tree for muscoids.
2.4. Specimen Deposition and Abbreviations
The specimens examined in this study are deposited in the following two museums: MBFU—Museum of Beijing Forestry University, Beijing, China; ZMUM—Zoological Museum of Moscow University, Russia. Abbreviations used for external characters include: ac = acrostichal setae; dc = dorsocentral setae; a, p, d, v = anterior, posterior, dorsal, ventral setae; prst = presutural, post = postsutural; tar = tarsi.
3. Results
We present the first comprehensive phylogenetic analysis for the genus Lispe, which includes 11 out of 13 currently acknowledged Lispe species groups. Four species were used as outgroups (two from Fanniidae and two from Sarcophagidae). Among them, Sarcophaga forma and S. arizonica were used to root the tree. In total, 315 molecular sequences were submitted to GenBank (see Supplementary Table S1).
3.1. Estimation of Substitution Saturation
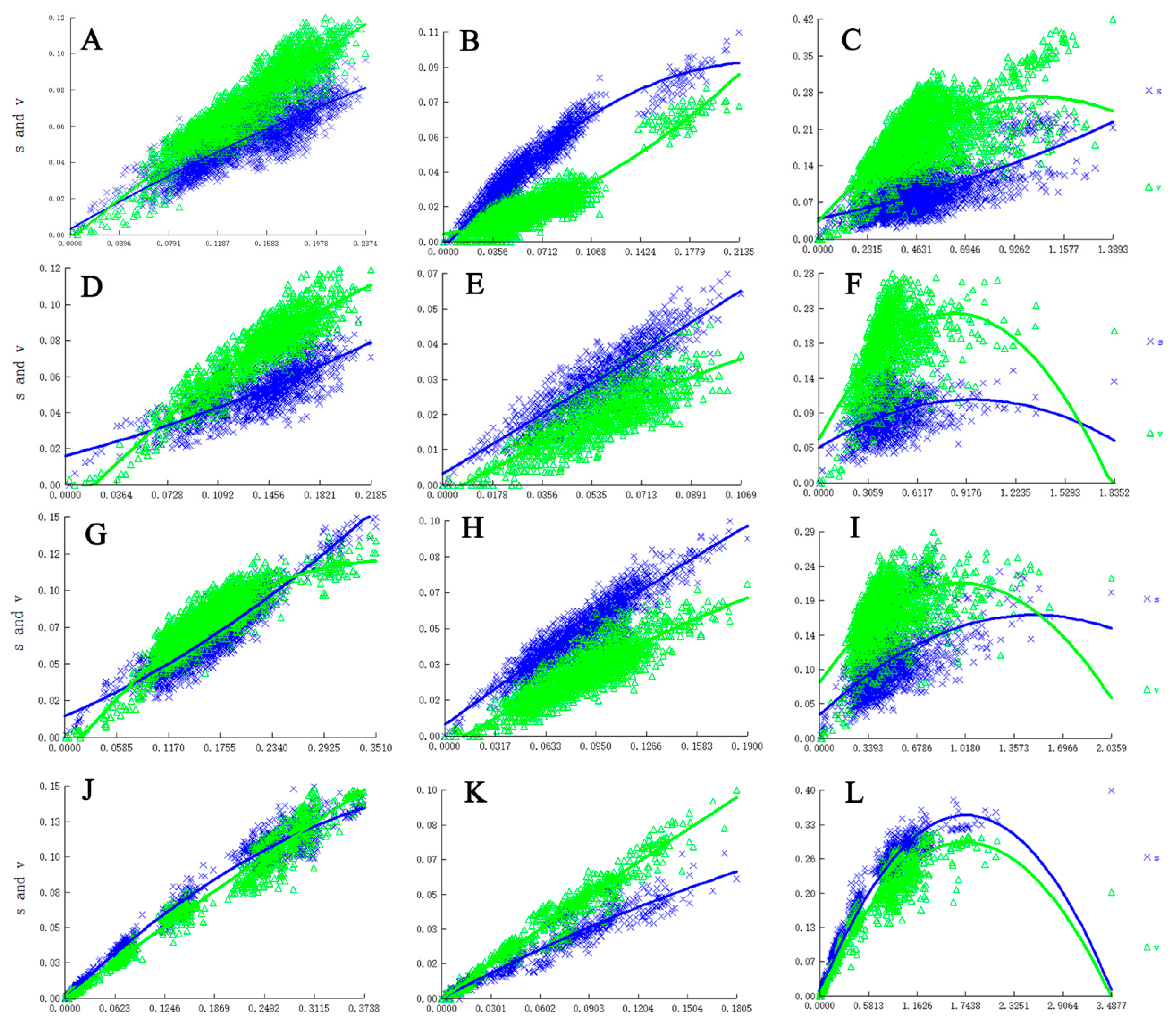
Our results suggest no or limited saturation of all genes with all codons and all PCGs excluding third codon positions, but with oversaturation at third codon positions of all PCGs (Figure 1). In our analyses, IssSym was selected for assuming a symmetrical topology, and Iss is significantly lower than Iss.c (Table 2). Therefore, all sequences were suitable for phylogenetic analyses.
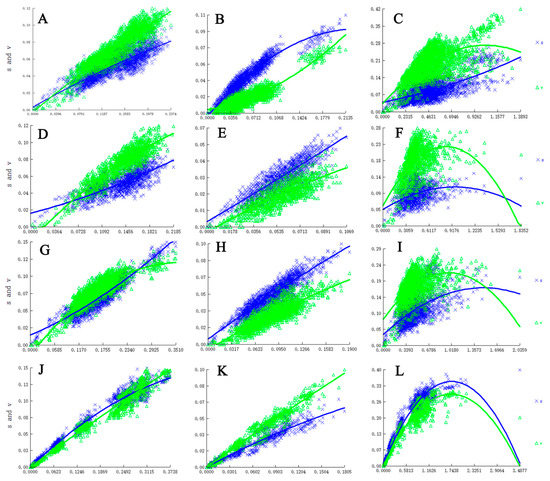
Figure 1.
Nucleotide substitution saturation plots of all genes. (A). All codon positions of COXIa gene; (B). First and second codon positions of COXIa; (C). Third codon positions of COXIa; (D). All codon positions of COXIb; (E). First and second codon positions of the COXIb gene; (F). Third codon positions of COXIb; (G). All codon positions of CYTB; (H). First and second codon positions of CYTB; (I). Third codon positions of CYTB; (J). All codon positions of CAD; (K). First and second codon positions of CAD; (L). Third codon positions of CAD.
3.2. Phylogenetic Analysis
Although topologies resulting from Bayesian inference were overall not well supported (Supplementary Figure S1), we found that both the ML and MP analyses yielded similar results (Figure 2). Coenosiinae form a well-supported monophyletic group (Figure 2) with most subfamilies recovered as para- or polyphyletic. The branch support of Azeliini was weak by bootstrap values (Bootstrap < 50) and denied in MP trees. The Atherigoninae was recovered as monophyletic in consistency with previous research [4]. Lispe is inferred as a strongly supported monophyletic group (Bootstrap = 100, Jackknife = 99), and the Limnophora was recovered as a sister group to Lispe with moderate support (Bootstrap = 72, Jackknife = 3). Thus, with the Limnophora as a sister group, Lispe are divided into the same 11 clades in accordance with the traditional species group divisions based on taxonomy (Figure 2) with four clades.
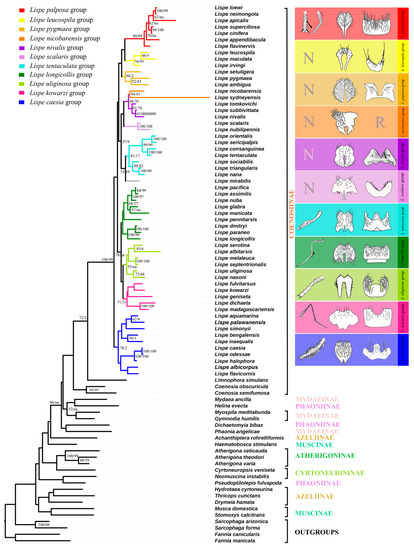
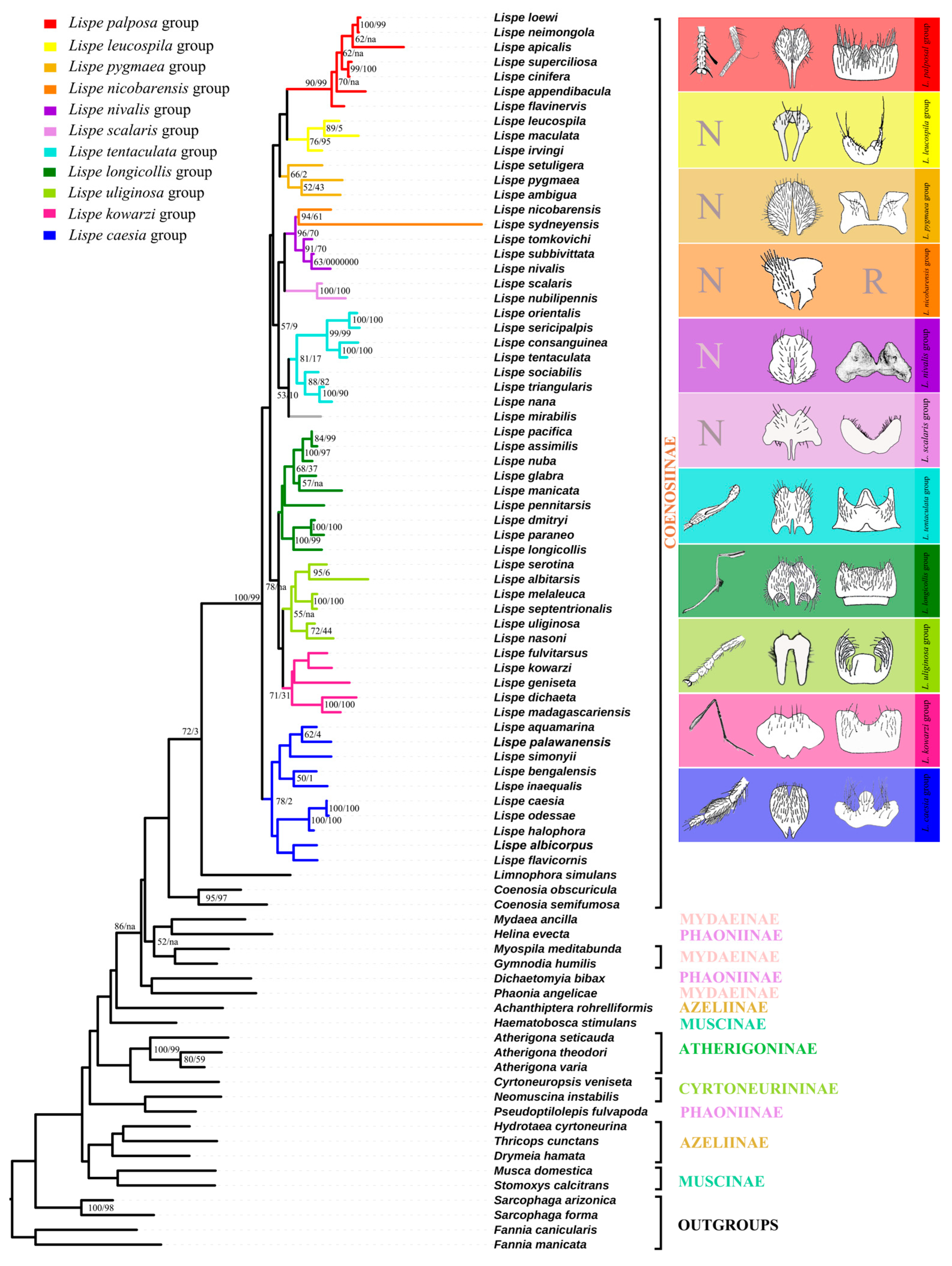
Figure 2.
The topologies of maximum likelihood trees based on all selected genes with third codon positions, mapped with morphological and biological characters. N represents the unmodified normal legs. R represents that sternite 5 is reduced. Bootstrap/Jackknife node support values for the ML and MP trees are indicated with on the branches of the tree respectively, bootstrap values less than 50 are not indicated on the tree. Branches indicated with na are not supported on MP trees.
Clade 1 contains three species groups: the L. palposa-group, L. leucospila-group, and L. pygmaea-group. The L. palposa-group is strongly supported in both the ML and MP analyses (Bootstrap = 90, Jackknife = 99), which is in line with the previous morphological taxonomy (Zhang, 2005). The monophyly of the L. leucospila-group [22] is moderately recovered (Bootstrap = 76, Jackknife = 95). The polyphyly of the L. pygmaea-group [30] was also proven based on molecular data. Vikhrev [30] divided the old L. pygmaea-group into five smaller groups. Among these five groups, the relationships of L. dichaeta- and L. geniseta-groups are discussed below. Importantly, the L. pumila-group is removed from our paper due to its instability and we thus regard it as a rogue taxa. The molecular data indicate a relationship between the two remaining members, the L. pygmaea- and L. ambigua-groups (Bootstrap = 52, Jackknife = 43). Morphologically, they share the reduced chaetotaxy of a third tibia (1 ad seta only). L. pygmaea is widespread in the Old World, with L. ambigua in the Afrotropical region, while L. setuligera is in the Neotropical region. Considering that neither L. pygmaea nor L. setuligera are distributed in North America, the occurrence of L. setuligera in South America is particularly interesting. We tend to integrate the L. pygmaea- and L. ambigua-groups into the new L. pygmaea-group based on the ML tree.
Clade 2 is formed by the L. nicobarensis-group (Bootstrap = 96, Jackknife = 70), the L. nivalis-group (Bootstrap = 91, Jackknife = 70), the L. scalaris-group (Bootstrap = 100, Jackknife = 100), the extended L. tentaculata-group (Bootstrap = 81, Jackknife < 50) and the unique species L. mirabilis. The topology of these groups in the ML and MP trees is well supported by morphological traits. The molecular data also indicate a close relationship between the L. nivalis- and L. nicobarensis-groups, which is a new hypothesis that has not been previously suggested. It is, however, not without morphological basis, as the two groups share a sunshining thorax with reduced pollinosity, dc reduced to 0+2 or 0+1, reduced leg chaetotaxy, a similar shape of cercal plates (Figure 2) and freshwater habitats. The extended L. tentaculata-group consists of the previous L. tentaculata-group and the L. nana-group (Vikhrev, 2014), which is moderately supported by bootstrap values (Bootstrap = 81, Jackknife < 50) and previously supported by morphological traits [22].
Clade 3 contains three groups, the L. longicollis-group, the L. uliginosa-group and the L. kowarzi-group. For the L. longicollis-group, our data showed relatively weak support for its separation into the two former proposed subgroups (the L. longicollis-subgroup and the L. assimilis-subgroup) (Bootstrap < 50, Jackknife < 50). L. pennitarsis, the only genetically examined species from the L. desjardinsii-group, is placed in an intermediate position between two subgroups of the L. longicollis-group. This result is not unexpected, as the similarity of L. desjardinsii- and L. longicollis-groups had been proposed by Vikhrev (2014) [22]. For example, both groups have a set of submedian av, ad and pd setae on t3 and very similar body shapes. Although the validity of the L. desjardinsii-group as a whole cannot be verified in this tree, it is suggested that the L. desjardinsii-group should be regarded as a subgroup of the L. longicollis-group, making up the extended L. longicollis-group. The monophyly of the L. uliginosa-group is also supported by molecular data (Bootstrap = 55, Jackknife < 50), and the former hypotheses of the L. uliginosa- and L. melaleuca-subgroups within this group are verified [9]. The relationship of the L. kowarzi-group appeared to be enigmatic based on taxonomic research, but the molecular data offer a very interesting hypothesis which combines the old L. kowarzi-, L. dichaeta- and L. geniseta-groups into the new L. kowarzi-group (Bootstrap = 71, Jackknife < 50). Such a relationship is also supported by their identical leg chaetotaxy: first tibia with 1 p, second tibia with 1 ad and 1 pd, third tibia with 1 pd, 1 ad and 1 av.
Clade 4 is the L. caesia-group [21,23,24,25], which is placed at the base of the Lispe genus. The molecular data support the monophyly of the L. caesia-group and infer it as the sister group of all other Lispe species. However, most species within the L. caesia-group are lacking at least one or two of Hennig’s three diagnositic characters. Thus, we offer the following morphological substantiations to redefine the L. caesia-group: the presence of at least one of the Hennig’s characters is enough for including species in the L. caesia-group, i.e., either L. leucocephala with the characteristic frons or L. bengalensis with the strong v spines on femora.
4. Discussion
4.1. Generic Relationships within Muscidae
The genus Limnophora is proven to be the sister group of Lispe, together forming the tribe Limnophorini within the subfamily Coenosiinae in our sampling data. Three genera were proven obsolete: Chaetolispa Malloch, Xenolispa Malloch, and Lispacoenosia Snyder.
The genus Chaetolispa Malloch 1922 with type species C. geniseta Stein, 1909 and the genus Xenolispa Malloch 1922 with type species X. atrifrontata Malloch, 1922 (= Lispe sydneyensis Schiner, 1868) were both isolated from Lispe by Malloch [43,44]. These two genera have not been well recognized by subsequent dipterists [21,45,46]. Our molecular data support the notion that both Chaetolispa and Xenolispa are groundless and should be merged back into Lispe. This result is reinforced by morphology—the diagnostic strong bristle on parafacial close to the lower anterior margin of eyes for Chaetolispa is also found in Lispe species (L. dichaeta, L. madagascariensis, and L. stuckenbergi). The diagnostic reduced dc setae of Xenolispa are also shared by some Lispe clades (L. kowarzi- and L. nivalis-groups). Snyder (1949) described Lispacoenosia fulvitarsus Snyder, 1949 as a Lispe-like Muscidae without the characteristic hairs on the anepimeron [20]. Vikhrev noted that Snyder’s species is very similar to L. kowarzi, which also has greatly reduced setulae on the anepimeron occurring as only 1–3 fine setulae, and subsequently transferred it into the genus Lispe [22]. Our molecular data confirm that Lispacoenosia fulvitarsus belongs to the L. kowarzi-group, so the name Lispacoenosia should be correctly synonymized to Lispe.
4.2. Intrageneric Relationships within Lispe
For the Lispe uliginosa-group, Vikhrev [9] included one Neotropical species (L. serotina) and two Palaearctic species (L. melaleuca and L. septentrionalis). Both L. melaleuca and L. septentrionalis were proposed to be sister clades to the other species of the L. uliginosa-group [9,24]. Vikhrev [9] also supposed that all American species of the group are descendants of a common ancestor of L. uliginosa which colonized America via the Bering land bridge. Molecular data support all of these hypotheses, and the L. uliginosa-group can thus be divided into the L. uliginosa- and L. melaleuca-subgroups.
L. mirabilis is an interesting species characterized by strongly reduced chaetotaxy of the scutum and legs (like that in the new L. nivalis-group), as well as a modified male tar 1-1 (Figure 3) reminiscent of the finger-like protuberance on male tar 1-1 in the L. tentaculata-group. Although the molecular data supports the sister group relationship of the L. mirabilis- and L. tentaculata-groups, the true relationship requires further resolution because the former group is only represented by a single species.

Figure 3.
(A). L. mirabilis Stein, 1918, male, Thailand; (B). L. kowarzi kowarzi Becker, 1903, female, Turkey (photo: N. Vikhrev).
4.3. Evolution of Morphological Characteristics
The morphological evolution of Lispe in terms of three primary traits (external modifications, the shape of cerci, and the shape of sternite) can be traced using the ML tree.
Male terminalia present helpful and reliable information for species identification in numerous dipteran taxa, including Lispe [11,21,25,28]. As for flies of small size such as Lispe, the dissection of the phallosome into clear divided sub-structures can be difficult. Fortunately, the shape of the cerci and sternite 5 are generally sufficient for group and species level separation (Figure 2). Generally, the gap between the cerci plate increases in size in subsequent descendants of Lispe (e.g., L. uliginosa-, L. tentaculata-, L. longicollis-, and L. pygmaea-groups).
Various external modifications have evolved throughout Lispe. Examples include the swollen hind first tarsomere in the L. caesia-group, the merged sternite 4 in the L. nicobarensis-group, the flattened fore tarsomeres in the L. melaleuca clade of the L. uliginosa-group; the finger-like protuberance on the fore tarsomere in the L. tentaculata clade of the L. tentaculata-group; and the shining black body with limited pollinosity in the L. nicobarensis-, and L. kowarzi-groups). Interestingly, many of these morphological traits may have evolved in response to sexual selection [17]. This is particularly so for leg morphology and chaetotaxy, because in many Lispe species, the leg structures play important roles in courtship behaviour. Males of many species will grasp the wings of females [16] and stridulate their legs on the head of the female, and certain other species will use their legs to position their entire body atop the female (i.e., L. sydneyensis) [17].
4.4. Origin of Lispe
The origin of the genus Lispe is still ambiguous. Vikhrev [22] hypothesized that Lispe might have originated from the southern part of the Palearctic region, since the most impressive diversity of Lispe is presented in the warmer regions of Asia and Africa. This hypothesis has excluded North America or Australia (both with significant diversity also) as the possible origin of Lispe because it is clear that all American or Australian species are descendants of few invaders via the Bering land bridge or dried Torres Strait bridge, respectively [22]. There is, however, a need for more representative samples of Australasian species in future phylogenies, as the region has a rich diversity of at least 39 species [21,45,46,47], including many that inhabit environments ranging from freshwater to salt water. The diversity in the Palearctic region is more impressive than in the Afrotropical region, but it is not so obvious that Africa can be ruled out as the origin of Lispe.
Our molecular data support this hypothesis. Among the Lispe species-groups proposed by morphological taxonomy, there are several small clades represented only in the Afrotropical region. However, according to our molecular phylogeny, the old L. desjardinsii-group is a subgroup of the extended L. longicollis-group; the old L. dichaeta-group is a subgroup of the new L. kowarzi-group; and the old L. ambigua-group is a subgroup of the new L. pygmaea-group. So, there are no exclusively African clades any longer, and all three new groups include species of both African and Asian origin. At the same time, the large L. uliginosa and L. palposa-groups are well supported by the molecular data and are represented in the Palearctic region only. In conclusion, the place of origin of Lispe is most likely Eurasia, possibly Central Eurasia; specifically, the arid regions between the Caspian lowland and the west part of India.
5. Conclusions
This study represents the first comprehensive analysis of the phylogenetic relationships of Lispe based on molecular and morphological data and elucidates the validity of 11 species groups of Lispe. Although the origin of the genus Lispe is still ambiguous, we considered that the origin of Lispe is most likely to be Eurasia, possibly in Central Eurasia, and specifically the arid regions between the Caspian lowland and the west part of India. However, it remains unclear as to the causes and consequences of such evolutionary transitions between different habitat types, which itself may require further research into the basic biology and ecology of global Lispe species in the future.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects13111015/s1, Figure S1: Phylogenetic tree of Muscidae based on molecular data by Bayesian inference; Table S1: Species used in present analyses with GenBank accession numbers; Table S2: Collection and DNA isolation information of species.
Author Contributions
Conceptualization, project administration, funding acquisition, D.Z.; formal analysis, Y.G. (Yunyun Gao), Y.G. (Yingqiang Ge) and L.Y.; investigation, Y.G. (Yingqiang Ge) and N.E.V.; resources, N.E.V. and D.Z.; data curation, Y.G. (Yunyun Gao), L.Y. and D.Z.; writing—original draft preparation, Y.G. (Yunyun Gao), Y.G. (Yingqiang Ge), L.Y., N.E.V. (except for Section 4.3 and Section 4.4), Q.W., N.J.B. and D.Z.; writing—review and editing, visualization, Y.G. (Yunyun Gao), Y.G. (Yingqiang Ge), L.Y., N.E.V., Q.W. and N.J.B. All authors have read and agreed to the published version of the manuscript.
Funding
The research was supported by the Fundamental Research Funds for National Science Foundation of China (No. 31872964, 31201741 to D.Z.), and the Beijing Forestry University Outstanding Young Talent Cultivation Project (2019JQ0318 to D.Z.).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
DNA sequences are open available in National Center for Biotechnology Information (https://www.ncbi.nlm.nih.gov, accessed on 26 September 2022), accession numbers are attached in Table S1.
Acknowledgments
We thank Adrian Pont for his help editing the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Carvalho, C.J.B.; Couri, M.S.; Pont, A.C.; Pamplona, D.; Lopes, S.M. A Catalogue of the Muscidae (Diptera) of the Neotropical Region. Zootaxa 2005, 860, 1–282. [Google Scholar] [CrossRef]
- Pape, T.; Blagoderov, V.; Mostovski, M.B. Order Diptera Linnaeus, 1758. In Animal Biodiversity: An Outline of Higher-Level Classification and Survey of Taxonomic Richness; Magnolia: Auckland, New Zealand, 2011. [Google Scholar]
- Kutty, S.N.; Pape, T.; Wiegmann, B.M.; Meier, R. Molecular Phylogeny of the Calyptratae (Diptera: Cyclorrhapha) with an Emphasis on the Superfamily Oestroidea and the Position of Mystacinobiidae and McAlpine’s Fly. Syst. Entomol. 2010, 35, 614–635. [Google Scholar] [CrossRef]
- Kutty, S.N.; Pont, A.C.; Meier, R.; Pape, T. Complete Tribal Sampling Reveals Basal Split in Muscidae (Diptera), Confirms Saprophagy as Ancestral Feeding Mode, and Reveals an Evolutionary Correlation between Instar Numbers and Carnivory. Mol. Phylogenet. Evol. 2014, 78, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Kutty, S.N.; Meusemann, K.; Bayless, K.M.; Marinho, M.A.T.; Pont, A.C.; Zhou, X.; Misof, B.; Wiegmann, B.M.; Yeates, D.; Cerretti, P.; et al. Phylogenomic Analysis of Calyptratae: Resolving the Phylogenetic Relationships within a Major Radiation of Diptera. Cladistics 2019, 35, 605–622. [Google Scholar] [CrossRef]
- Kutty, S.N.; Pape, T.; Pont, A.; Wiegmann, B.M.; Meier, R. The Muscoidea (Diptera: Calyptratae) Are Paraphyletic: Evidence from Four Mitochondrial and Four Nuclear Genes. Mol. Phylogenet. Evol. 2008, 49, 639–652. [Google Scholar] [CrossRef]
- Ding, S.; Li, X.; Wang, N.; Cameron, S.L.; Mao, M.; Wang, Y.; Xi, Y.; Yang, D. The Phylogeny and Evolutionary Timescale of Muscoidea (Diptera: Brachycera: Calyptratae) Inferred from Mitochondrial Genomes. PLoS ONE 2015, 10, e0134170. [Google Scholar] [CrossRef]
- Haseyama, K.L.F.; Wiegmann, B.M.; Almeida, E.A.B.; de Carvalho, C.J.B. Say Goodbye to Tribes in the New House Fly Classification: A New Molecular Phylogenetic Analysis and an Updated Biogeographical Narrative for the Muscidae (Diptera). Mol. Phylogenet. Evol. 2015, 89, 1–12. [Google Scholar] [CrossRef]
- Vikhrev, N.E. Taxonomic Notes on Lispe (Diptera, Muscidae), Parts 10–12. Amurian Zool. J. 2015, VII, 228–247. [Google Scholar]
- Fogaça, J.M.; de Carvalho, C.J.B. Neotropical Lispe (Diptera: Muscidae): Notes, Redescriptions and Key to Species. J. Nat. Hist. 2018, 52, 2147–2184. [Google Scholar] [CrossRef]
- Snyder, F.M. A Review of Nearctic Lispe Latreille (Diptera, Muscidae). Am. Mus. Novit. 1954, 1675, 1–40. [Google Scholar]
- Steidle, J.L.M.; Dettner, K.; Huebner, G.; Koepf, A.; Reinhard, J. The Predaceous Fly Lispe Candicans (Diptera: Muscidae) Andits Chemically Protected Prey, the Rove Beetle Bledius Furcatus (Coleoptera: Staphylinidae). Entomol. Gen. 1995, 20, 11–19. [Google Scholar] [CrossRef]
- Frantsevich, L.; Gorb, S. Courtship Dances in the Flies of the Genus Lispe (Diptera: Muscidae): From the Flys Viewpoint. Arch. Insect Biochem. Physiol. 2006, 62, 26–42. [Google Scholar] [CrossRef] [PubMed]
- Pont, A.C.; Harutyunova, K.; Harutyunova, M.; Werner, D. The Hunter-Flies of Armenia. II. The Genus Lispe Latreille, 1796 (Insecta: Diptera: Muscidae). Zool. Middle East 2012, 55, 79–84. [Google Scholar] [CrossRef]
- Bradley, T.J. Saline-Water Insects: Ecology, Physiology and Evolution. In Aquatic Insects: Challenges to Populations; Lancaster, J., Briers, R., Eds.; CAB International: London, UK, 2008. [Google Scholar]
- White, T.E.; Vogel-Ghibely, N.; Butterworth, N.J. Flies Exploit Predictable Perspectives and Backgrounds to Enhance Iridescent Signal Salience and Mating Success. Am. Nat. 2020, 195, 733–742. [Google Scholar] [CrossRef]
- Butterworth, N.J.; Wallman, J.F. Flies Getting Filthy: The Precopulatory Mating Behaviours of Three Mud-Dwelling Species of Australian Lispe (Diptera: Muscidae). Ethology 2022, 128, 369–377. [Google Scholar] [CrossRef]
- Morse, J.C.; Yang, L.F.; Tian, L.X. Aquatic Insects of China Useful for Monitoring Water Quality; Hohai University Press: Nanjing, China, 1994. [Google Scholar]
- Aldrich, J.M. The North American Species of Lispa (Diptera; Anthomyidae). J. N. Y. Entomol. Soc. 1913, 21, 126–146. [Google Scholar]
- Snyder, F.M. New Genera and Species of Lispinae (Diptera, Muscidae). Am. Mus. Novit. 1949, 1403, 1–9. [Google Scholar]
- Hennig, W. Muscidae [Part, Lieferung 209 and 213]; Lindner, E., Ed.; Schweizerbart: Stuttgart, Germany, 1960. [Google Scholar]
- Vikhrev, N.E. Taxonomic Notes on Lispe (Diptera, Muscidae), Parts 1–9. Amur. Zool. J. 2014, VI, 147–170. [Google Scholar]
- Vikhrev, N.E.; Ge, Y.Q.; Zhang, D. On Taxonomy of the Lispe Caesia-Group (Diptera: Muscidae). Russ. Entomol. J. 2016, 25, 407–410. [Google Scholar] [CrossRef]
- Xue, W.Q.; Zhang, D. A Review of the Genus Lispe Latreille (Diptera: Muscidae) from China, with Descriptions of New Species. Orient. Insects 2005, 39, 117–139. [Google Scholar] [CrossRef]
- Zhang, D.; Ge, Y.Q.; Li, X.Y.; Liu, X.H.; Zhang, M.; Wang, R.R. Review of the Lispe Caesia-Group (Diptera: Muscidae) from Palaearctic and Adjacent Regions, with Redescriptions and One New Synonymy. Zootaxa 2016, 4098, 43–72. [Google Scholar] [CrossRef] [PubMed]
- Vikhrev, N. Review of the Palaearctic Members of the Lispe Tentaculata Species-Group (Diptera, Muscidae): Revised Key, Synonymy and Notes on Ecology. Zookeys 2011, 84, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Vikhrev, N.E. Revision of the Lispe Longicollis-Group (Diptera, Muscidae). Zookeys 2012, 235, 23–39. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Gao, Y.; Yan, L.; Liu, X.; Zhang, D. Review of the Lispe Tentaculata-Group (Diptera: Muscidae) in China, with One New Synonym. Zoosystema 2016, 38, 339–352. [Google Scholar] [CrossRef]
- Vikhrev, N.E. Four New Species of Lispe Latreille, 1796 (Diptera, Muscidae) with Taxonomic Notes on Related Species. Russ. Entomol. J. 2012, 21, 423–433. [Google Scholar] [CrossRef]
- Vikhrev, N.E. Taxonomic Notes on Lispe (Diptera, Muscidae), Part 13. Amur. Zool. J. 2016, VIII, 171–185. [Google Scholar]
- Zhang, D.; Yan, L.; Zhang, M.; Chu, H.; Cao, J.; Li, K.; Hu, D.; Pape, T. Phylogenetic Inference of Calyptrates, with the First Mitogenomes for Gasterophilinae (Diptera: Oestridae) and Paramacronychiinae (Diptera: Sarcophagidae). Int. J. Biol. Sci. 2016, 12, 489–504. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA Primers for Amplification of Mitochondrial Cytochrome c Oxidase Subunit I from Diverse Metazoan Invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Moulton, J.K.; Wiegmann, B.M. Evolution and Phylogenetic Utility of CAD (Rudimentary) among Mesozoic-Aged Eremoneuran Diptera (Insecta). Mol. Phylogenet. Evol. 2004, 31, 363–378. [Google Scholar] [CrossRef]
- Lalitha, S. Primer Premier 5. Biotech. Softw. Internet. Rep. Comput. Softw. J. Scient. 2000, 1, 270–272. [Google Scholar] [CrossRef]
- Hall, T.; Biosciences, I.; Carlsbad, C. BioEdit: An Important Software for Molecular Biology. GERF Bull. Biosci. 2011, 2, 60–61. [Google Scholar]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Xie, Z.; Salemi, M.; Chen, L.; Wang, Y. An Index of Substitution Saturation and Its Application. Mol. Phylogenet. Evol. 2003, 26, 1–7. [Google Scholar] [CrossRef]
- Xia, X. DAMBE6: New Tools for Microbial Genomics, Phylogenetics, and Molecular Evolution. J. Hered. 2017, 108, 431–437. [Google Scholar] [CrossRef]
- Meier, R.; Shiyang, K.; Vaidya, G.; Ng, P.K.L. DNA Barcoding and Taxonomy in Diptera: A Tale of High Intraspecific Variability and Low Identification Success. Syst. Biol. 2006, 55, 715–728. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A Fast and Effective Stochastic Algorithm for Estimating Maximum-Likelihood Phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A Fast Online Phylogenetic Tool for Maximum Likelihood Analysis. Nucleic Acids Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef]
- Drake, M. The Importance of Temporary Waters for Diptera (True-Flies). Freshw. Forum 2001, 17, 26–39. [Google Scholar]
- Malloch, J.R. Exotic Muscaridae (Diptera). VII. Ann. Mag. Nat. 1922, 9, 379–391. [Google Scholar] [CrossRef][Green Version]
- Malloch, J.R. Exotic Muscaridae (Diptera). V. Ann. Mag. Nat. 1992, 9, 271–280. [Google Scholar] [CrossRef][Green Version]
- van Emden, F.I. Keys to the Muscidae of the Ethiopian Region: Scatophaginae, Anthomyiinae, Lispinae, Fanniinae. Bull. Entomol. Res. 1941, 32, 251–275. [Google Scholar] [CrossRef]
- Pont, A.C. Family Muscidae. Catalog of the Diptera of the Australasian and Oceanian Regions; Evenhuis, N.L., Ed.; Bishop Museum Press: Honolulu, HI, USA, 1989; p. 804. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).