Monarchs Reared in Winter in California Are Not Large Enough to Be Migrants. Comment on James et al. First Population Study on Winter Breeding Monarch Butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the Urban South Bay of San Francisco, California. Insects 2021, 12, 946
1. Synopsis
2. Background
3. Alternative Interpretation
4. Implications for Conservation
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- James, D.G.; Schaefer, M.C.; Krimmer Easton, K.; Carl, A. First population study on winter breeding monarch butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the urban south bay of San Francisco, California. Insects 2021, 12, 946. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.B.; Brown, L.M.; Pelton, E.; Crone, E.E. Citizen science monitoring demonstrates dramatic declines of monarch butterflies in western North America. Biol. Conserv. 2017, 214, 343–346. [Google Scholar] [CrossRef]
- Thogmartin, W.E.; Wiederholt, R.; Oberhauser, K.; Drum, R.G.; Diffendorfer, J.E.; Altizer, S.; Taylor, O.R.; Pleasants, J.; Semmens, D.; Semmens, B.; et al. Monarch butterfly population decline in North America: Identifying the threatening processes. R. Soc. Open Sci. 2017, 4, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crone, E.E.; Schultz, C.B. Resilience or Catastrophe? A possible state change for monarch butterflies in western North America. Ecol. Lett. 2021, 6, 1533–1538. [Google Scholar] [CrossRef]
- James, D.G. Western North American Monarchs: Spiraling into Oblivion or Adapting to a Changing Environment? Anim. Migr. 2021, 8, 19–26. [Google Scholar] [CrossRef]
- Dilts, T.E.; Steele, M.O.; Engler, J.D.; Pelton, E.M.; Jepsen, S.J.; McKnight, S.J.; Taylor, A.R.; Fallon, C.E.; Black, S.H.; Cruz, E.E.; et al. Host plants and climate structure habitat associations of the western monarch butterfly. Front. Ecol. Evol. 2019, 7, 188. [Google Scholar] [CrossRef] [Green Version]
- Satterfield, D.A.; Maerz, J.C.; Hunter, M.D.; Flockhart, D.T.T.; Hobson, K.A.; Norris, D.R.; Streit, H.; de Roode, J.C.; Altizer, S. Migratory monarchs that encounter resident monarchs show life-history differences and higher rates of parasite infection. Ecol. Lett. 2018, 21, 1670–1680. [Google Scholar] [CrossRef]
- Majewska, A.A.; Altizer, S. Exposure to non-native tropical milkweed promotes reproductive development in migratory monarch butterflies. Insects 2019, 10, 253. [Google Scholar] [CrossRef] [Green Version]
- James, D.G.; Kappen, L. Further insights on the migration biology of monarch butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) from the Pacific Northwest. Insects 2021, 12, 161. [Google Scholar] [CrossRef]
- Altizer, S.M.; Oberhauser, K. Effects of the protozoan parasite Ophryocystis elektroscirrha on the fitness of monarch butterflies (Danaus plexippus). J. Invertebr. Pathol. 1999, 74, 76–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wise, K.A.J. Monarch butterfly dispersal in New Zealand. Rec. Auckl. Inst. Mus. 1980, 17, 157–173. [Google Scholar]
- Zalucki, M.P.; Kitching, R.L. The dynamics of adult Danaus plexippus L. (Danaidae) within patches of its food plant, Asclepias spp. J. Lepid. Soc. 1984, 38, 209–219. [Google Scholar]
- Zalucki, M.P.; Parry, H.R.; Zalucki, J.M. Movement and egg laying in Monarchs: To move or not to move, that is the equation. Austral Ecol. 2016, 41, 154–167. [Google Scholar] [CrossRef] [Green Version]
- Marchetti, K.; Price, T.; Richman, A. Correlates of wing morphology with foraging behavior and migration distance in the genus Phylloscopus. J. Avian Biol. 1995, 26, 177–181. [Google Scholar] [CrossRef]
- Norberg, U.M.; Rayner, J.M.V. Ecological morphology and flight in bats (Mammalia: Chiroptera): Wing adaptations, flight performance, foraging strategy and echolocation. Philos. Trans. R. Soc. Lond. Ser. B-Biol. Sci. 1987, 316, 337–419. [Google Scholar]
- Suarez-Tovar, C.M.; Sarmiento, C.E. Beyond the wing planform: Morphological differentiation between migratory and nonmigratory dragonfly species. J. Evol. Biol. 2016, 29, 690–703. [Google Scholar] [CrossRef] [Green Version]
- Zhan, S.; Zhang, W.; Niitepold, K.; Hsu, J.; Haeger, J.F.; Zalucki, M.P.; Altizer, S.; de Roode, J.C.; Reppert, S.M.; Kronforst, M.R. The genetics of monarch butterfly migration and warning colouration. Nature 2014, 514, 317–321. [Google Scholar] [CrossRef] [Green Version]
- Altizer, S.; Davis, A.K. Populations of monarch butterflies with different migratory behaviors show divergence in wing morphology. Evolution 2010, 64, 1018–1028. [Google Scholar] [CrossRef]
- Li, Y.; Pierce, A.A.; de Roode, J.C. Variation in forewing size linked to migratory status in monarch butterflies. Anim. Migr. 2016, 3, 27–34. [Google Scholar] [CrossRef]
- Freedman, M.G.; Dingle, H.; Strauss, S.Y.; Ramirez, S.R. Two centuries of monarch butterfly collections reveal contrasting effects of range expansion and migration loss on wing traits. Proc. Natl. Acad. Sci. USA 2020, 117, 28887–28893. [Google Scholar] [CrossRef]
- Leong, K.L.H.; Frey, D.; Hamaoka, D.; Honma, K. Wing damage in overwintering populations of monarch butterfly at two California sites. Ann. Entomol. Soc. Am. 1993, 86, 728–733. [Google Scholar] [CrossRef]
- Leong, K.L.H.; O’Brien, E.; Lowerisen, K.; Colleran, M. Mating activity and status of overwintering monarch butterflies (Lepidoptera: Danaidae) in central California. Ann. Entomol. Soc. Am. 1995, 88, 45–50. [Google Scholar] [CrossRef]
- Dockx, C. Directional and stabilizing selection on wing size and shape in migrant and resident monarch butterflies, Danaus plexippus (L.), in Cuba. Biol. J. Linn. Soc. 2007, 92, 605–616. [Google Scholar] [CrossRef] [Green Version]
- Talla, V.; Pierce, A.A.; Adams, K.L.; de Man, T.J.B.; Nallu, S.; Villablanca, F.X.; Kronforst, M.R.; de Roode, J.C. Genomic evidence for gene flow between monarchs with divergent migratory phenotypes and flight performance. Mol. Ecol. 2020, 29, 2567–2582. [Google Scholar] [CrossRef]
- Freedman, M.G.; Roode, J.C.; Forister, M.L.; Kronforst, M.R.; Pierce, A.A.; Schultz, C.B.; Taylor, O.R.; Crone, E.E. Are eastern and western monarch butterflies distinct populations? A review of evidence for ecological, phenotypic, and genetic differentiation and implications for conservation. Conserv. Sci. Pract. 2021, 3, e432. [Google Scholar] [CrossRef]
- Yang, L.H.; Ostrovsky, D.; Rogers, M.C.; Welker, J.M. Intra-population variation in the natal origins and wing morphology of overwintering western monarch butterflies Danaus plexippus. Ecography 2016, 39, 998–1007. [Google Scholar] [CrossRef]
- Freedman, M.G.; Dingle, H. Wing morphology in migratory North American monarchs: Characterizing sources of variation and understanding changes through time. Anim. Migr. 2018, 5, 61–73. [Google Scholar] [CrossRef] [Green Version]
- Fric, Z.; Klimova, M.; Konvicka, M. Mechanical design indicates differences in mobility among butterfly generations. Evol. Ecol. Res. 2006, 8, 1511–1522. [Google Scholar]
- Steffy, G. Trends observed in fall migrant monarch butterflies (Lepidoptera: Nymphalidae) east of the Appalachian Mountains at an inland stopover in southern Pennsylvania over an eighteen year period. Ann. Entomol. Soc. Am. 2015, 108, 718–728. [Google Scholar] [CrossRef]
- Satterfield, D.A.; Villablanca, F.X.; Maerz, J.C.; Altizer, S. Migratory monarchs wintering in California experience low infection risk compared to monarchs breeding year-round on non-native milkweed. Integr. Comp. Biol. 2016, 56, 343–352. [Google Scholar] [CrossRef] [Green Version]
- Bradley, C.A.; Altizer, S. Parasites hinder monarch butterfly flight: Implications for disease spread in migratory hosts. Ecol. Lett. 2005, 8, 290–300. [Google Scholar] [CrossRef]
- Davis, A.K.; de Roode, J.C. Effects of the parasite, Ophryocystis elektroscirrha, on wing characteristics important for migration in the monarch butterfly. Anim. Migr. 2018, 5, 84–93. [Google Scholar] [CrossRef]
- Decker, L.E.; Soule, A.J.; de Roode, J.C.; Hunter, M.D. Phytochemical changes in milkweed induced by elevated CO2 alter wing morphology but not toxin sequestration in monarch butterflies. Funct. Ecol. 2019, 33, 411–421. [Google Scholar] [CrossRef]
- Freedman, M.G.; Dingle, H.; Tabuloc, C.A.; Chiu, J.C.; Yang, L.H.; Zalucki, M.P. Non-migratory monarch butterflies, Danaus plexippus (L.), retain developmental plasticity and a navigational mechanism associated with migration. Biol. J. Linn. Soc. 2018, 123, 265–278. [Google Scholar] [CrossRef]
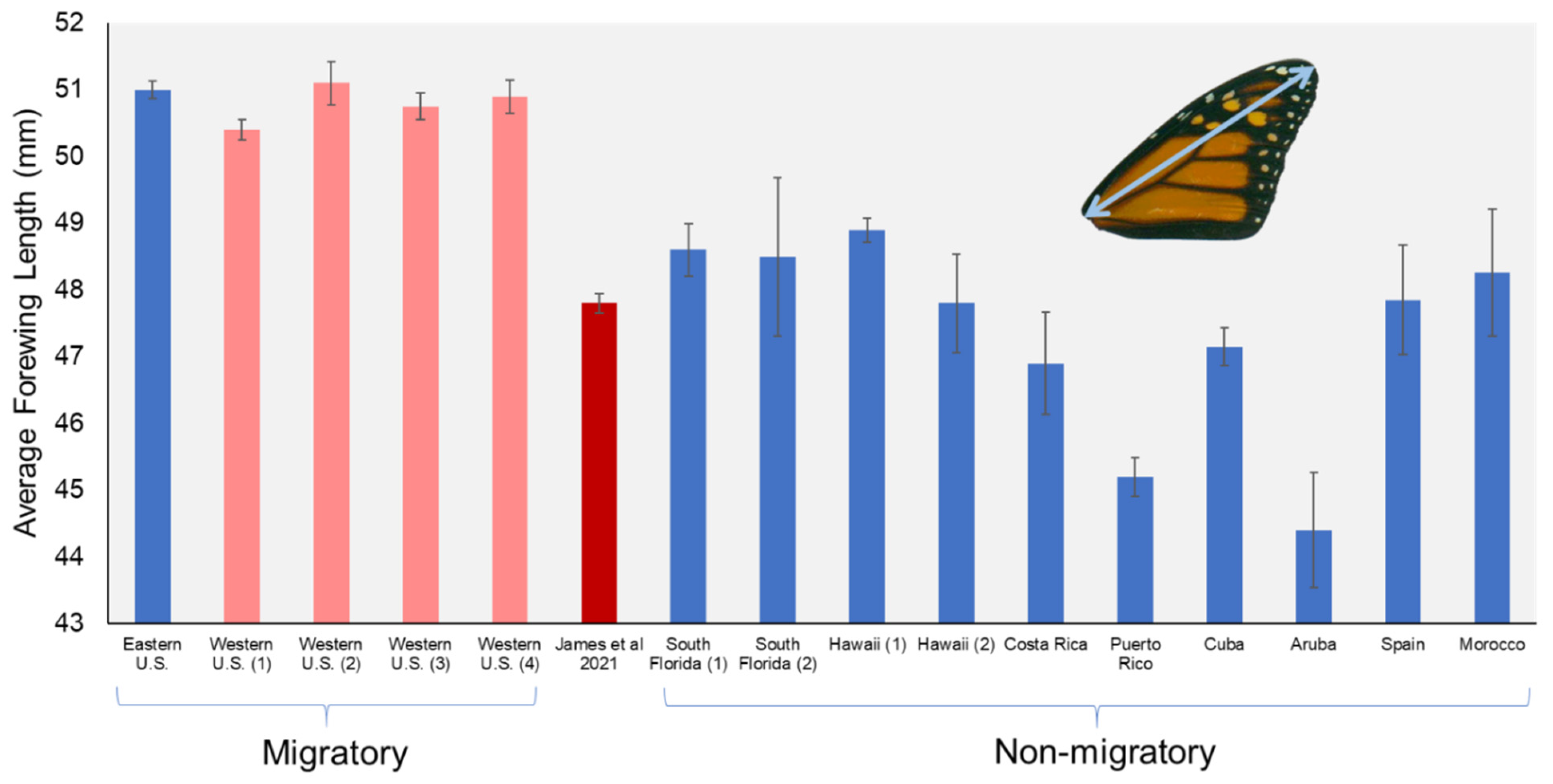
| Location | Status | N | Forewing Length | SE | Source |
|---|---|---|---|---|---|
| Eastern U.S. | Migratory | 389 | 51.0 | 0.13 | [18] |
| Western U.S. (1) | Migratory | 251 | 50.4 | 0.15 | [18] |
| Western U.S. (2) | Migratory | 59 | 51.1 | 0.32 | [19] |
| Western U.S. (3) | Migratory | 242 | 50.8 | 0.20 | [21] |
| Western U.S. (4) | Migratory | 728 | 50.9 | 0.25 | [22] |
| Winter-reared in California | 499 | 47.8 | 0.15 | [1] | |
| South Florida (1) | Nonmigratory | 54 | 48.6 | 0.40 | [18] |
| South Florida (2) | Nonmigratory | 20 | 48.5 | 1.19 | [19] |
| Hawaii (1) | Nonmigratory | 125 | 48.9 | 0.18 | [18] |
| Hawaii (2) | Nonmigratory | 24 | 47.8 | 0.74 | [19] |
| Costa Rica | Nonmigratory | 20 | 46.9 | 0.77 | [18] |
| Puerto Rico | Nonmigratory | 58 | 45.2 | 0.29 | [18] |
| Cuba | Nonmigratory | 135 | 47.2 | 0.28 | [23] |
| Aruba | Nonmigratory | 26 | 44.4 | 0.86 | [19] |
| Spain | Nonmigratory | 16 | 47.9 | 0.82 | [19] |
| Morocco | Nonmigratory | 24 | 48.3 | 0.96 | [19] |
| Average Migratory | 1669 | 50.8 | 0.21 | ||
| Average Nonmigratory | 502 | 47.4 | 0.60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davis, A.K. Monarchs Reared in Winter in California Are Not Large Enough to Be Migrants. Comment on James et al. First Population Study on Winter Breeding Monarch Butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the Urban South Bay of San Francisco, California. Insects 2021, 12, 946. Insects 2022, 13, 63. https://doi.org/10.3390/insects13010063
Davis AK. Monarchs Reared in Winter in California Are Not Large Enough to Be Migrants. Comment on James et al. First Population Study on Winter Breeding Monarch Butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the Urban South Bay of San Francisco, California. Insects 2021, 12, 946. Insects. 2022; 13(1):63. https://doi.org/10.3390/insects13010063
Chicago/Turabian StyleDavis, Andrew K. 2022. "Monarchs Reared in Winter in California Are Not Large Enough to Be Migrants. Comment on James et al. First Population Study on Winter Breeding Monarch Butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the Urban South Bay of San Francisco, California. Insects 2021, 12, 946" Insects 13, no. 1: 63. https://doi.org/10.3390/insects13010063
APA StyleDavis, A. K. (2022). Monarchs Reared in Winter in California Are Not Large Enough to Be Migrants. Comment on James et al. First Population Study on Winter Breeding Monarch Butterflies, Danaus plexippus (Lepidoptera: Nymphalidae) in the Urban South Bay of San Francisco, California. Insects 2021, 12, 946. Insects, 13(1), 63. https://doi.org/10.3390/insects13010063






