Perspectives for Synergic Blends of Attractive Sources in South American Palm Weevil Mass Trapping: Waiting for the Red Palm Weevil Brazil Invasion
Simple Summary
Abstract
1. Introduction
2. The Genus Rhynchophorus spp.
2.1. South American Palm Weevil
Biology
3. Red Palm Weevil
Biology
4. Damage to the Host Plant
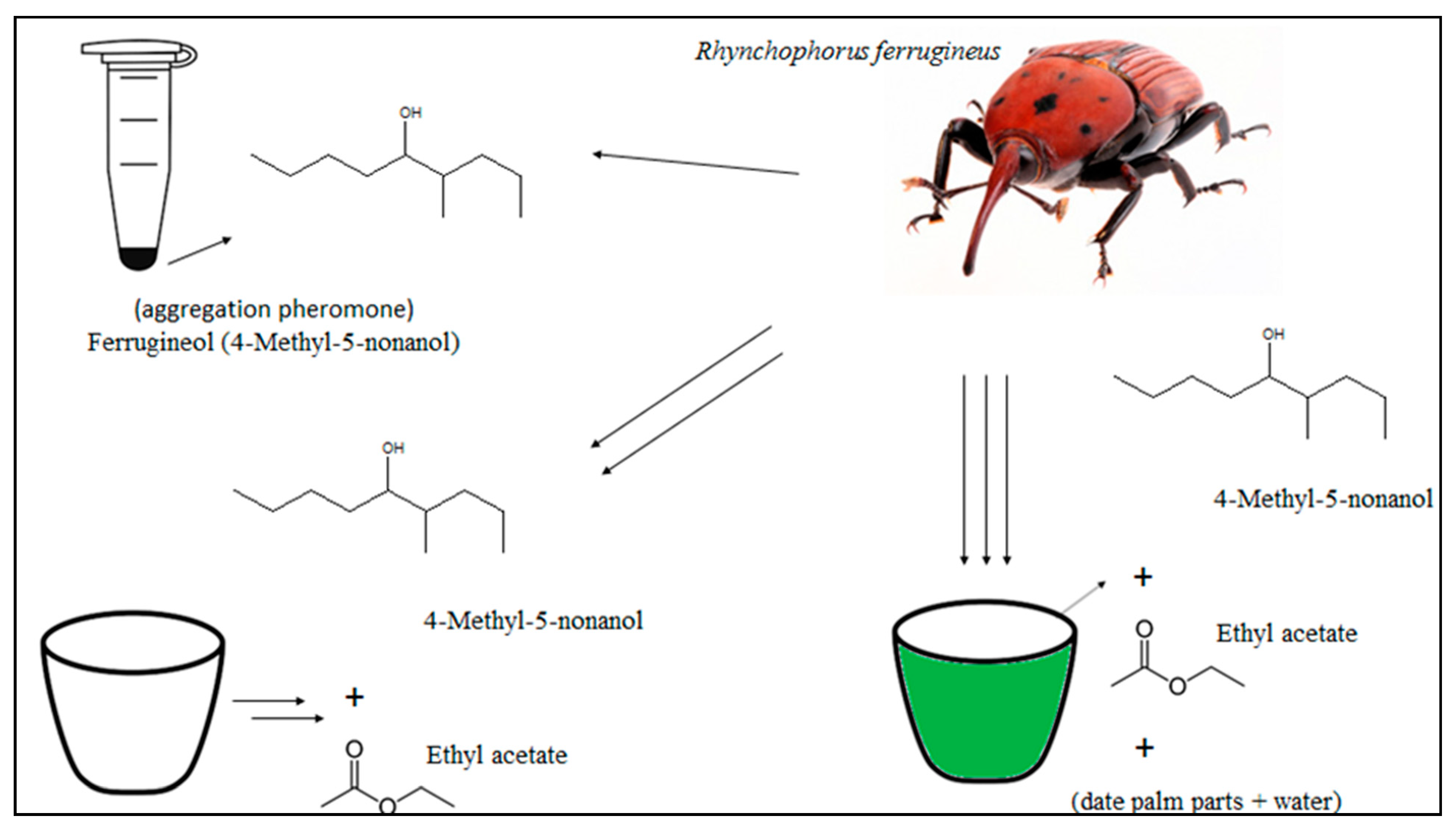
5. Rhynchophorus spp. Pheromone and Other Attractive Sources
5.1. Attractive Source for South American Palm Weevil
5.2. Attractive Source for Red Palm Weevil
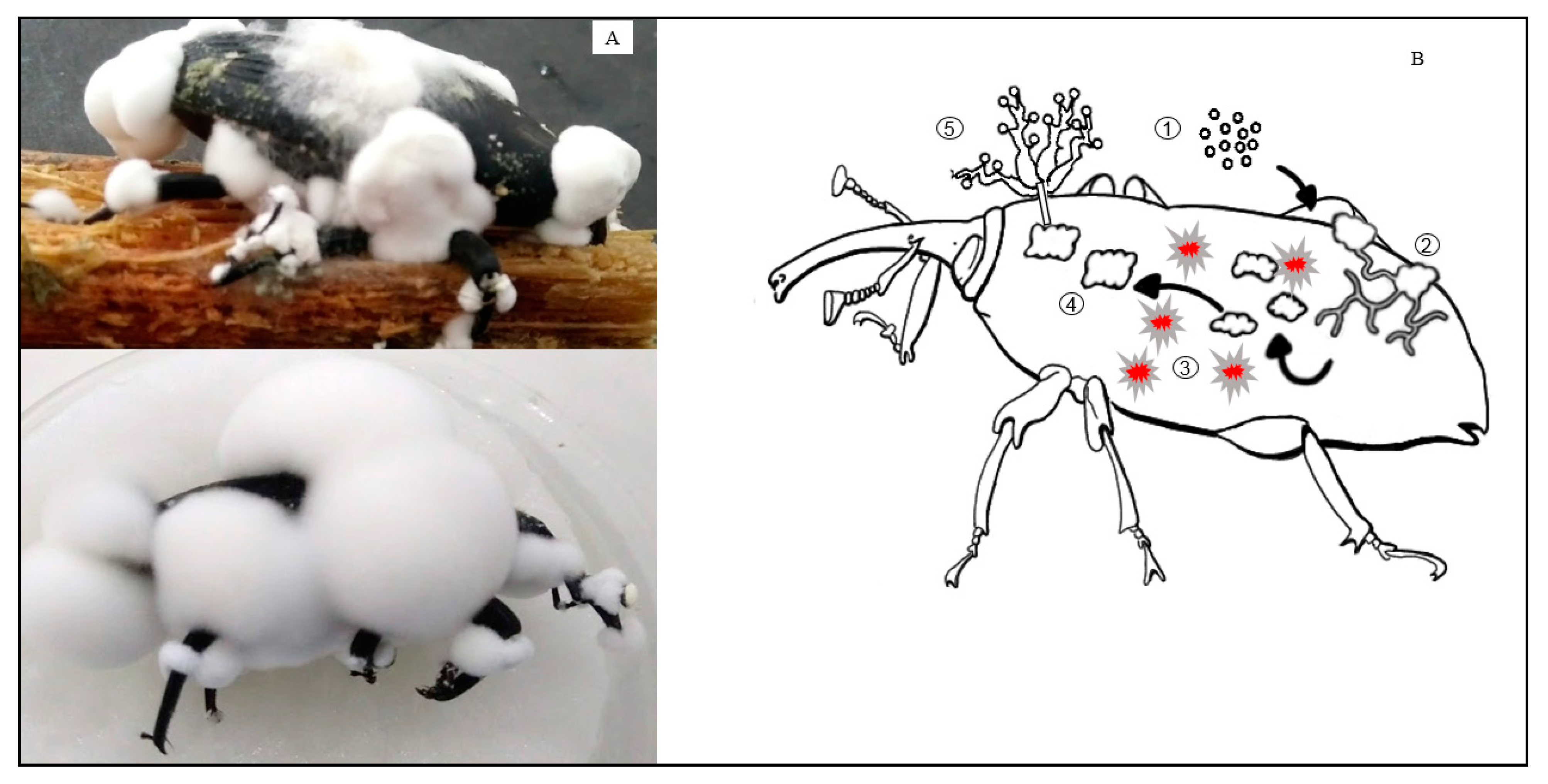
6. Alternative for Rhynchophorus palmarum and Rhynchophorus ferrugineus Control
7. Future Challenges and Prospects
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bondar, G. Insectos Nocivos e Moléstias do Coqueiro (Cocos nucifera L.) No Brasil; Tipografia Naval: Salvador, Brazil, 1940; 156p. [Google Scholar]
- Dalbon, V.; Acevedo, J.P.M.; Santana, A.E.G.; Goulart, H.F.; Laterza, I.; Riffel, A.; Negrisoli, A., Jr.; Lohr, B.; Porcelli, F. Early detection and preventive control of Rhynchophorus ferrugineus (Coleóptera Curculionidae): A quarantine pest in Brasil. Arab J. Plant Prot. 2019, 37, 130–135. [Google Scholar] [CrossRef]
- Dionisio, L.F.S.; Lima, A.C.S.; de Morais, E.G.F.; Farias, P.R.S.; Correia, R.G.; Martins, W.B.R.; dos Santos, A.V.F. Spatial distribution of Rhynchophorus palmarum L. (Coleoptera: Curculionidae) in oil palm, Roraima state, Brazil. Rev. Bras. Cienc. Agrar. 2020, 15, e5683. [Google Scholar] [CrossRef]
- Faleiro, J.R. A review on the issues and management of red palm weevil Rhynchophorus ferrugineus (Coleoptera: Rhynchophoridae) in coconut and date palm during the last one hundred years. Int. J. Trop. Insect Sci. 2006, 26, 135–154. [Google Scholar] [CrossRef]
- Fiaboe, K.K.M.; Peterson, A.T.; Kairo, M.T.K.; Roda, A.L. Predicting the potential worldwide distribution of the red palm weevil Rhynchophorus ferrugineus (Olivier) (Coleóptera: Curculionidae) using ecological niche modeling. Fla. Entomol. 2012, 95, 659–673. [Google Scholar] [CrossRef]
- Gonzalez, F.; Kharrat, S.; Rodríguez, C.; Calvo, C.; Oehlschlager, A.C. Red palm weevil (Rhynchophorus ferrugineus Olivier): Recent advances. Arab J. Plant Prot. 2019, 37, 178–187. [Google Scholar] [CrossRef]
- Hoddle, M.S.; Hoddle, C.D.; Milosavljević, I. How far can Rhynchophorus palmarum (Coleoptera: Curculionidae) fly? J. Econ. Entomol. 2020, 113, 1786–1795. [Google Scholar] [CrossRef] [PubMed]
- Hoddle, M.S.; Hoddle, C.D.; Milosavljević, I. Quantification of the life time flight capabilities of the south American palm weevil, Rhynchophorus palmarum (L.) (Coleoptera: Curculionidae). Insects 2021, 12, 126. [Google Scholar] [CrossRef] [PubMed]
- Rochat, D.; Dembillo, O.; Jaques, J.A.; Suma, P.; Pergola, A.; Hamidi, R.; Kontodimas, D.; Soroker, V. Rhynchophorus ferrugineus: Taxonomy, distribution, biology and life cycle. In Handbook of Major Palm Pests; Colazza, S., Soroker, V., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2017; pp. 69–104. [Google Scholar] [CrossRef]
- Roda, A.; Kairo, M.; Damian, T.; Franken, F.; Heidweiller, K.; Johanns, C.; Mankin, R. Red palm weevil (Rhynchophorus ferrugineus), an invasive pest recently found in the Caribbean that threatens the region. EPPO Bull. 2011, 41, 116–121. [Google Scholar] [CrossRef]
- Thomas, M.C. Giant Palm Weevils of the Genus Rhynchophorus (Coleoptera: Curculionidae) and Their Threat to Florida Palms. Florida Department of Agriculture and Consumer Services, Division of Plant Industry. DACS-P-01682: 1-2. 2010. Available online: https://www.fdacs.gov/content/download/66344/file/pest_alert_-_giant_palm_weevils_of_the_genus_rhynchophorus.pdf (accessed on 6 January 2020).
- Bertone, C.; Michalak, P.S.; Roda, A. New Pest Response Guidelines. Red Palm Weevil. USDA-APHIS-PPQ-Emergency and Domestic Programs-Emergency Planning, Riverdale, Maryland. 2011. Available online: https://assets.ippc.int/static/media/uploads/resources/new_pest_response_guidelines_red_palm_weevil.pdf (accessed on 18 August 2021).
- Esteban-Durán, J.; Yela, J.L.; Beitia-Crespo, F.; Jiménez-Álvarez, A. Curculiónidos exóticos susceptibles de ser introducidos en España y otros países de la Unión Europea a través de vegetales importados (Coleóptera: Curculionidae: Rhynchophorinae). Boletín Sanid. Veg. 1998, 24, 23–40. Available online: http://hdl.handle.net/20.500.11939/7308 (accessed on 19 August 2021).
- Murphy, S.T.; Briscoe, B.R. The red palm weevil as an alien invasive: Biology and the prospects for biological control as a component of IPM. Biocontrol News Inf. 1999, 20, 34n–46n. [Google Scholar]
- Wattanapongsiri, A. A Revision of the Genera Rhynchophorus and Dynamis (Coleoptera: Curculionidae). PhD Thesis, Oregon State University, Corvallis, OR, USA, 1966; 431p. Available online: https://ir.library.oregonstate.edu/concern/graduate_thesis_or_dissertations/6d570047d (accessed on 19 August 2021).
- NAPPO First U.S. Detection of Red Palm Weevil, Rhychophorus Ferrugineus, in California. Letter to State Plant Regulatory Officials. 2010. Available online: https://www.pestalerts.org/official-pest-report/first-u-s-detection-red-palm-weevil-rhynchophorus-ferrugineus-california (accessed on 6 January 2020).
- Dalbon, V.A.; Oliveira, E.R.L.; Silva, A.F.; Santana, A.E.G.; Acevedo, J.P.M.; Lisboa-Junior, K.A.; Goulart, H.F.; Oliveria, J.F.T.; Pitta, G.B.B. O uso de una nova mistura de conposição química, de fontes atrativas, para controlar o inseto-praga Rhynchophorus ferrugineus (Coleóptera: Curculionidae) em arecáceas: Un estudo prospectivo. Cad. Prospecção 2019, 12, 825–834. [Google Scholar] [CrossRef]
- Hoddle, M.S.; Hoddle, C.D.; Alzubaidy, M.; Kabashima, J.; Nisson, J.N.; Millar, J.; Dimson, M. The palm weevil Rhynchophorus vulneratus is eradicated from Laguna Beach. Calif. Agric. 2016, 71, 23–29. [Google Scholar] [CrossRef]
- Liebhold, A.M.; Tobin, P.C. Population ecology of insect invasions and their management. Ann. Rev. Entomol. 2008, 387–408. [Google Scholar] [CrossRef] [PubMed]
- Tobin, P.C.; Kean, J.M.; Suckling, D.M.; McCullough, D.G.; Herms, D.A.; Stringer, L.D. Determinants of successful arthropod eradication programs. Invas. Biol. 2014, 16, 401–414. [Google Scholar] [CrossRef]
- Vásquez-Ordóñez, A.A.; Löhr, B.L.; Marvaldi, A.E. Comparative morphology of the larvae of the palm weevils Dynamis borassi (Fabricius) and Rhynchophorus palmarum (Linnaeus) (Curculionidae: Dryophthorinae): Two major pests of peach palms in the Neotropics. Papéis Avulsos Zool. 2020, 60, e202060-27. [Google Scholar] [CrossRef]
- Hallett, R.H.; Gries, G.; Gries, R.; Borden, J.H. Aggregation pheromones of two Asian palm weevils, Rhynchophorus ferrugineus and R. vulneratus. Naturwissenschaften 1993, 80, 328–331. [Google Scholar] [CrossRef]
- Oehlschlager, A.C.; Chinchilla, C.M.; González, L.M. Manejo del picudo de la palma (Rhynchophorus palmarum) y la enfermedad de anillo rojo, mediante un sistema de trampeo basado en la feromona de agregación. ASD Oil Palm 1992, 5, 24–30. [Google Scholar]
- Perez, A.L.; Gries, G.; Gries, R.; Giblin-Davis, R.M.; Oehlschlager, C. Pheromone chirality of african palm weevil, Rhynchophorus phoenicis (F.) and palmetto weevil, Rhynchophorus cruentatus (F.) (Coleóptera: Curculionidae). J. Chem. Ecol. 1994, 20, 2653–2671. [Google Scholar] [CrossRef]
- Rochat, D.; Malosse, C.; Lettere, M.; Ducrot, P.-H.; Zagatti, P.; Renou, M.; Descoins, C. Male-produced aggregation pheromone of the American palm weevil, Rhynchophorus palmarum (L.) (Coleóptera: Curculionidae), collection, identification, electrophysiological activity and laboratory bioassay. J. Chem. Ecol. 1991, 17, 2127–2141. [Google Scholar] [CrossRef] [PubMed]
- Franco, E. Estudo Sobre o Anel Vermelho do Coqueiro; Publisher Ministry of Agriculture, Department of Defense and Agricultural Inspection, Plant Health Defense Service, Plant Health Defense Inspectorate in Sergipe: Aracaju, Brazil, 1964; 236p. [Google Scholar]
- Mexzón, R.G.; Chinchilla, C.M.; Castrillo, G.; Salamanca, D. Biología y hábitos de Rhynchophorus palmarum L. asociado a la palma aceitera en Costa Rica. ASD Oil Palm 1994, 8, 14–21. [Google Scholar]
- Prabhu, S.T.; Patil, R.S. Studies on the biological aspects of red palm weevil, Rhynchophorus ferrugineus (Oliv.). J. Agric. Sci. 2009, 22, 732–733. [Google Scholar]
- Restrepo, L.G.; Rivera, F.; Raigosa, J.D. Ciclo de vida, hábitos y morfometria de Metamasius hemipterus Olivier y Rhynchophorus palmarum L. (Coleoptera: Curculionidae) en caña de azucar (Saccharum officinarum L.). Acta Agronómica 1982, 32, 33–44. [Google Scholar]
- Arun Kumar, S.; Payal, J.; Satyendra, P. Taxonomic redescription of the red palm weevil (Rhynchophorus ferrugineus). J. Pharmacogn. Phytochem. 2017, 6, 1054–1060. [Google Scholar]
- Löhr, B.; Vásquez-Ordóñez, A.A.; Lopez- Lavalle, L.A.B. Rhynchophorus palmarum in disguise: Undescribed polymorphism in the “black” palm weevil. PLoS ONE 2015, 10, e0143210. [Google Scholar] [CrossRef]
- Sukirno, S.; Tufail, M.; Rasool, K.G.; Aldawood, A.S. Undescribed color polymorphism of the Asiatic palm weevil, Rhynchophorus vulneratus Panzer (Coleoptera: Curculionidae) in Indonesia: Biodiversity study based on COI gene. Fla. Entomol. 2018, 101, 642–648. [Google Scholar] [CrossRef]
- EL-Mergawy, R.A.A.M. Genetic comparison among Rhynchophorus ferrugineus and four Rhynchophorus species. J. Agric. Sci. Technol. B 2011, 1, 610–615. [Google Scholar]
- Faleiro, J.R.; Satarkar, V.R. Diurnal activity of red palm weevil Rhynchophorus ferrugineus (Olivier) in coconut plantation. Insect Environ. 2003, 9, 63–64. [Google Scholar]
- Hagley, E.A.C. On the life history and habits of the palm weevil Rhynchophorus palmarum (L.). Ann. Entomol. Soc. Am. 1965, 5, 22–28. [Google Scholar] [CrossRef]
- Milosavljević, I.; El-Shafie, H.A.F.; Faleiro, J.R.; Hoddle, C.D.; Lewis, M.; Mark, S.; Hoddle, M.S. Palmageddon: The wasting of ornamental palms by invasive palm weevils, Rhynchophorus sp. J. Pest Sci. 2019, 92, 143–156. [Google Scholar] [CrossRef]
- Al-Ansi, A.; Aldryhim, Y.; Al-Janobi, A. First use of radio telemetry to assess behavior of red palm weevil, Rhynchophorus ferrugineus (Olivier) (Coleoptera: Dryophthoridae) in the presence and absence of pheromone traps. Comput. Electron. Agric. 2020, 170, 105252. [Google Scholar] [CrossRef]
- Antony, B.; Jibin, J.; Aldosari Saleh, A. Silencing the odorant binding protein RferOBP1768 reduces the strong preference of palm weevil for the major aggregation pheromone compound ferrugineol. Front. Physiol. 2018, 9, 1–17. [Google Scholar] [CrossRef]
- Gonzalez, F.; Johny, J.; Walker, W.B., III; Guan, Q.; Mfarrej, S.; Jakše, J.; Montagné, N.; Jacquin-Joly, E.; Alquarni, A.A.; Al-Saleh, M.A.; et al. Antennal transcriptome sequencing and identification of candidate chemoreceptor proteins from an invasive pest, the American palm weevil, Rhynchophorus palmarum. Sci. Rep. 2021, 11, 8334. [Google Scholar] [CrossRef]
- Soffan, A.; Antony, B.; Abdelazim, M.; Shukla, P.; Witjaksono, W.; Aldosari, S.A.; Aldawood, A.S. Silencing the olfactory co-receptor RferOrco reduces the response to pheromones in the Red Palm Weevil, Rhynchophorus ferrugineus. PLoS ONE 2016, 11, e0162203. [Google Scholar] [CrossRef]
- Zhao, Y.Y.; Liu, F.; Yang, G.; You, M.S. PsOr1, a potential target for RNA interference-based pest management. Insect Mol. Biol. 2011, 20, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Dalbon, V.A.; Lisboa-Ribeiro, T.F.; Molina-Acevedp, J.P.; Silva, J.M.; Anacleto-Andrade, A.B.; Granja, B.S.; Ribeiro-Junior, K.A.L.; Fonseca-Goulart, H.; Goulart-Santana, A.E. Respuesta comportamental y electrofisiológica de Rhynchophorus palmarum (L.; 1764) (Coleoptera: Curculionidae) a compuestos volátiles de hongos entomopatógenos nativos. An. Biol. 2021, 43, 65–77. [Google Scholar] [CrossRef]
- Duarte, A.G.; De Lima, I.S.; Ferraz, D.M.A.; Santana, A.E.G. Captura de Rhynchophorus palmarum L. (Coleóptera: Curculionidae) em armadilhas iscadas con o feromônio de agregação e compostos voláteis de frutos do abacaxi. Rev. Bras. Frutic. 2003, 25, 81–84. [Google Scholar] [CrossRef]
- Abd El-Wahab, A.S.; Abd El-Fattah, A.Y.; El-Shafei, W.K.M.; El Helaly, A.A. Efficacy of aggregation nano gel pheromone traps on the catchability of Rhynchophorus ferrugineus (Olivier) in Egypt. Braz. J. Biol. 2020, 81, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Holighaus, G.; Rohlfs, M. Fungal allelochemicals in insect pest management. Appl. Microbiol. Biotechnol. 2016, 100, 5681–5689. [Google Scholar] [CrossRef]
- Leon-Martinez, G.A.; Campos-Pinzon, J.C.; Arguelles-Cardenas, J.H. Patogenicidad y autodiseminación de cepas promisorias de hongos entomopatógenos sobre Rhynchophorus palmarum L. (Coleoptera: Dryophthoridae). Agron. Mesoam. 2019, 30, 631–646. [Google Scholar] [CrossRef]
- Lopez-Llorca, L.V.; Jalinas, J.; Marhuenda Egea, F.C. Compuestos Orgánicos Volátiles del Hongo Entomopatógeno Beauveria bassiana Como Repelentes de Insectos. España Patente de Invención P201631534, 9 February 2017. [Google Scholar]
- Lozano-Soria, A.; Picciotti, U.; Lopez-Moya, F.; Lopez-Cepero, J.; Porcelli, F.; Lopez-Llorca, L.V. Volatile organic compounds from entomopathogenic and nematophagous fungi, repel banana black weevil (Cosmopolites sordidus). Insects 2020, 11, 509. [Google Scholar] [CrossRef]
- Xu, Y.-J.; Luo, F.; Gao, Q.; Shang, Y.; Wang, C. Metabolomics reveals insect metabolic responses associated with fungal infection. Anal. Bioanal. Chem. 2015, 407, 4815–4821. [Google Scholar] [CrossRef] [PubMed]
- Harith-Fadzilah, N.; Haris-Hussain, M.; Abd Ghani, I.; Zakaria, A.; Amit, S.; Zainal, Z.; Azmi, W.A.; Jalinas, J.; Hassan, M. Physical and physiological monitoring on red palm weevil-infested oil palms. Insects 2020, 11, 407. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Rizwan-ul-haq, M.; AlJabr, A.M.; Al-Ayedh, H. Lethality of sesquiterpenes reprogramming red palm weevil detoxification mechanism for natural novel biopesticide development. Molecules 2019, 24, 1648. [Google Scholar] [CrossRef] [PubMed]
- Reyad, N.F.; Al-Ghamdi, H.A.; Abdel-Raheem, M.A.; Al-Shaeri, M.A. The effects of botanical oils on the red palm weevil, Rhynchophorus ferrugineus Olivier (Coleoptera: Curculionidae). Appl. Ecol. Environ. Res. 2020, 2909–2919. [Google Scholar] [CrossRef]
- Scrascia, M.; Pazzani, C.; Valentini, F.; Oliva, M.; Russo, V.; D’Addabbo, P.; Porcelli, F. Identification of pigmented Serratia marcescens symbiotically associated with Rhynchophorus ferrugineus Olivier (Coleoptera: Curculionidae). Microbiol. Open 2016, 5, 883–890. [Google Scholar] [CrossRef]
- Scrascia, M.; D’Addabbo, P.; Roberto, R.; Porcelli, F.; Oliva, M.; Calia, C.; Dionisi, A.M.; Pazzani, C. Characterization of CRISPR-cas systems in Serratia marcescens isolated from Rhynchophorus ferrugineus (Olivier, 1790) (Coleoptera: Curculionidae). Microorganisms 2019, 7, 368. [Google Scholar] [CrossRef]
- Löhr, B.; Negrisoli, A.; Molina, J.P. Billaea rhynchophorae, a palm weevil parasitoid with global potential. J. Arab Plant Prot. 2019, 37, 101–108. [Google Scholar] [CrossRef]
- Moura, J.I.L.; Toma, R.; Sgrillo, R.B.; Delabie, J.H.C. Natural efficiency of parasitism by Billaea rhynchophorae (Blanchard) (Diptera: Tachinidae) for the Control of Rhynchophorus palmarum (L.) (Coleoptera: Curculionidae). Neotrop. Entomol. 2006, 35, 273–274. [Google Scholar] [CrossRef]
- CABI CPC Datasheet Rhynchophorus palmarum. Available online: https://www.cabi.org/cpc/search/?q=Rhynchophorus+palmarum (accessed on 21 February 2021).
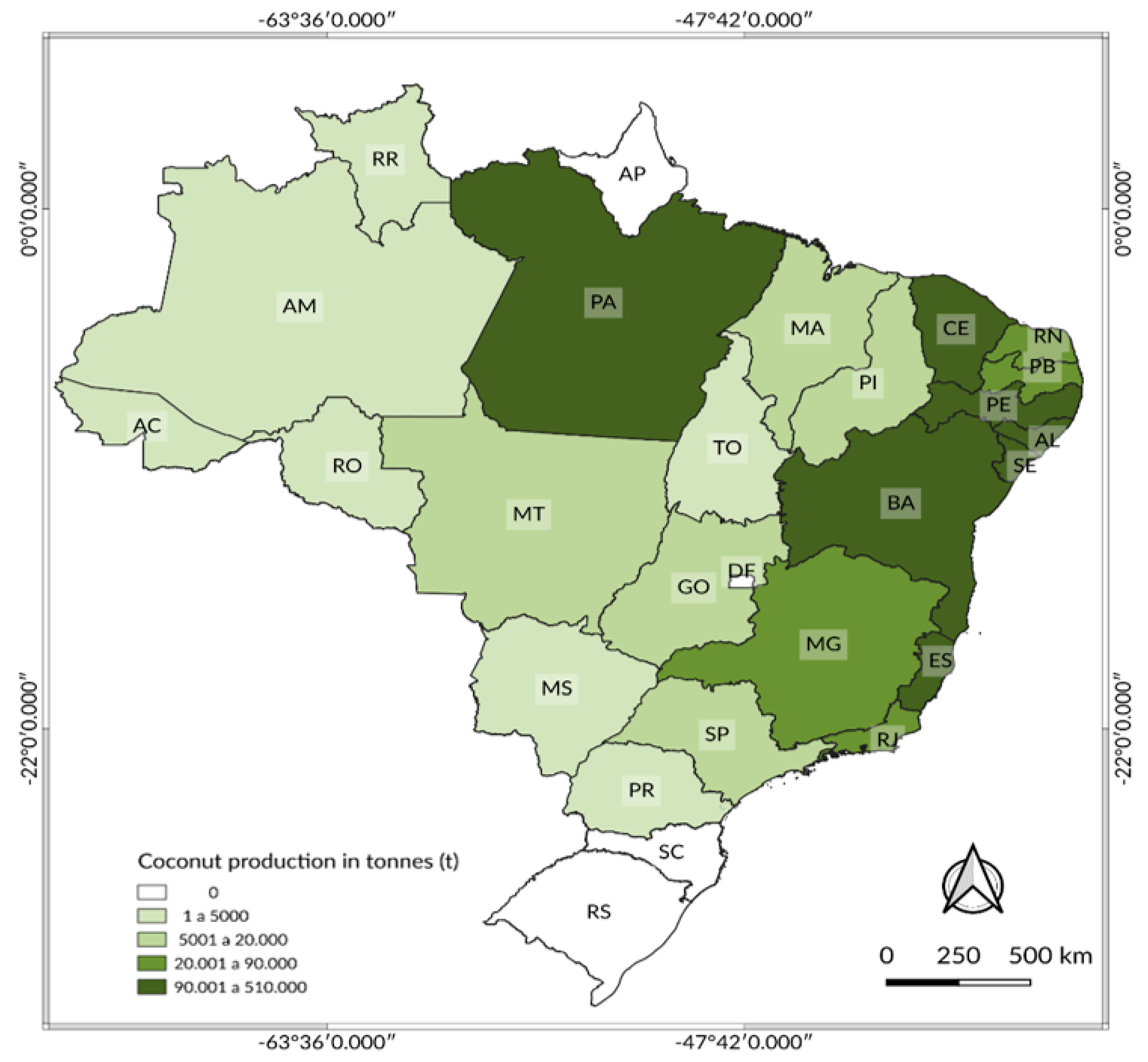
- IBGE—Brazilian Institute of Geography and Statistics. Agricultural Census, Permanent Coconut Crop Production. 2019. Available online: https://sidra.ibge.gov.br/tabela/5457#resultado (accessed on 26 February 2021).
- Takada, H.M.; Batista Filho, A.; Hojo, H. Aspectos biológicos de Rhynchophorus palmarum Linnaeus, 1764 (Coleoptera: Curculionidae) em banana-prata (Musa sapientum). Biológico 2014, 76, 35–39. [Google Scholar]
- Sánchez, P.A.; Cerda, H. El complejo Rhynchophorus palmarum L. (Coleóptera: Curculionidae)—Bursaphelenchus cocophilus Cobb. (Tylenchida: Aphelenchoididae) en palmas. Bol. Entom. Venez. 1993, 8, 1–18. [Google Scholar]
- Ferreira, J.M.S. Controle biológico do agente transmissor do nematoide causador do anel vermelho-do-coqueiro. EMBRAPA Tech. Bull. 2002, 31, 1–4. [Google Scholar]
- Griffith, R. The relationship between the red ring nematode and the palm weevil. J. Agric. Soc. Trinidad Tobago 1968, 68, 342–356. [Google Scholar]
- Moura, J.I.L.; Vilela, E.F. Pragas do Coqueiro e Dendezeiro, 2nd ed.; Aprenda Fácil: Viçosa, Brazil, 1998; 126p. [Google Scholar]
- Cysne, A.Q.; Cruz, B.A.; Cunha, R.N.V.; Rocha, R.N.C. Flutuação populacional de Rhynchophorus palmarum (L.) (Coleoptera: Curculionidae) em palmeiras oleíferas no Amazonas. Acta Amaz. 2013, 197–202. [Google Scholar] [CrossRef]
- Landero-Torres, I.; Presa-Parra, E.; Galindo-Tovar, M.E.; Leyva-Ovalle, O.R.; Murguía-González., J.; Valenzuela-González, J.E.; García-Martínez, M.A. Variación temporal y espacial de la abundancia del picudo negro (Rynchophorus palmarum L.; Coleóptera: Curculionidae) en cultivos de palmas ornamentales del centro de Veracruz, México. Southwest Entomol. 2015, 40, 179–188. [Google Scholar] [CrossRef]
- Schlickmann-Tank, J.A.; Enciso-Maldonado, G.A.; Haupenthal, D.I.; Luna-Alejandro, G.; Badillo-López, S.E. Detección y variación temporal de Rhynchophorus palmarum (Linnaeus) (Coleoptera: Dryophthoridae) en cultivos de Acrocomia aculeata (Jacq.) Lodd. ex Mart. en Itapúa, Paraguay. Rev. Chil. Entomol. 2020, 46, 163–169. [Google Scholar] [CrossRef]
- Chang, M.L.C.; Delgado, H.V. Estudio de trampa com atrayentes: Feromona de agregacion y frutas vegetales para capturas de adultos de gualpas (Rhynchophorus palmarum) y picudos rallados (Metamasius sericeus) em cocotero. Rev. Técnica 2012, 8, 12–19. [Google Scholar] [CrossRef]
- Landero-Torres, I.; Galindo-Tovar, M.E.; Leyva-Ovalle, O.R.; Murguía-González, J.; Presa-Parra, E.; García-Martínez, M.A. Evaluación de cebos para el control de Rhynchophorus palmarum (Coleóptera: Curculionidae) en cultivos de palmas ornamentales. Entomol. Mex. 2015, 2, 112–118. [Google Scholar]
- Milosavljević, I.; Hoddle, C.D.; Mafra-Neto, A.; Gómez-Marco, F.; Hoddle, M.S. Effects of food bait and trap type on captures of Rhynchophorus palmarum (Coleoptera: Curculionidae) and trap bycatch in Southern California. J. Econ. Entomol. 2020, 113, 2407–2417. [Google Scholar] [CrossRef]
- Moura, J.I.L.; Resende, M.L.V.; Sgrillo, R.; Nascimento, L.A.; Romano, R. Diferente tipos de armadilhas de iscas no controle de Rhynchophorus palmarum L. (Coleóptera: Curculionidae). J. Agrotrópica 1990, 2, 165–169. [Google Scholar]
- Murguía-González, J.; Landero-Torres, I.; Leyva-Ovalle, O.R.; Galindo-Tovar, M.E.; Llarena-Hernández, R.C.; Presa-Parra, E.; García-Martínez, M.A. Efficacy and cost of trap–bait combinations for capturing Rhynchophorus palmarum L. (Coleóptera: Curculionidae) in ornamental palm polycultures. Neotrop. Entomol. 2018, 47, 302–310. [Google Scholar] [CrossRef]
- Navarro, D.M.A.F.; Murta, M.M.; Duarte, A.G.; De Lima, I.S.; Nascimento, R.R.; Santana, A.E.G. Aspectos práticos relacionados ao uso do rincoforol, o feromônio de agregação da broca-do-olho-do-coqueiro Rhynchophorus palmarum (Coleópterate: Curculionidae) no controle de pragas do coqueiro. Análise de sua eficiencia em campo. Quim. Nova 2002, 25, 32–36. [Google Scholar] [CrossRef]
- Ovando-Cruz, M.E.; Serrano-Altamirano, V.; Galvez-Marroquin, L.A.; Ariza-Flores, R.; Martinez-Bolanos, M.; Ovando-Barroso, E. Evaluacion de trampas para Rhynchophorus palmarum L. (Coleoptera: Curculionidae) en la costa de Oaxaca, Mexico. Agroproductividad 2019, 12, 3–8. [Google Scholar] [CrossRef]
- Silva, A.B.; Múller, A.A.; Souza, L.A.; Ohashi, O.S.; Paulo, R.B. Avaliação de Iscas e Armadilhas para Captura de Rhynchophorus palmarum em Dendezais; Technical Bulletin Number 200; Brazilian Agricultural Research Corporation (EMBRAPA) Eastern Amazon Agroforestry Research Center (CPATU): Belém, Brazil, 1998; 18p. [Google Scholar]
- Vera, H.D.; Orellana, F.M. Combate de la gualpa (Rhynchophorus palmarum L.) en plantaciones de cocotero y palma africana mediante la captura con trampa del insecto adulto. INIAP Estac. Exp. St. Domingo Dep. Entomol. Tech. Bull. 1988, 198, 12. [Google Scholar]
- Milosavljević, I.; Hoddle, C.D.; Mafra-Neto, A.; Gómez-Marco, F.; Hoddle, M.S. Use of digital video cameras to determine the efficacy of two trap types for capturing Rhynchophorus palmarum (Coleoptera: Curculionidae). J. Econ. Entomol. 2020, 113, 3028–3031. [Google Scholar] [CrossRef]
- Ali-Bob, M. Management of the red palm weevil Rhynchophorus ferrugineus (Olivier) using sustainable options in Saudi Arabia. Arab J. Plant Prot. 2019, 37, 163–169. [Google Scholar] [CrossRef]
- Sardaro, R.; Grittani, R.; Scrascia, M.; Pazzani, C.; Russo, V.; Garganese, F.; Porfido, C.; Diana, L.; Porc, F. Red palm weevil city Bari: A first damage assessment. Forests 2018, 9, 452. [Google Scholar] [CrossRef]
- Rugman-Jones, P.F.; Hoddle, C.D.; Hoddle, M.S.; Stouthamer, R. The lesser of two Weevils: Molecular-genetics of Pest Palm Weevil populations confirm Rhynchophorus vulneratus (Panzer 1798) as a valid species distinct from R. ferrugineus (Olivier 1790), and reveal the global extent of both. PLoS ONE 2013, 8, e78379. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture, Livestock and Supply of Brazil (MAPA). Pragas quarentenárias present and absent in Brazil. Normative Instruction n°39. Off. J. União 2018, 190, 11. [Google Scholar]
- Zhao, M.; Rui-Ting, J. Effects of temperature on the development and fecundity of experimental population of Rhychophorus ferrugineus. Acta Phytophylacica Sin. 2010, 37, 517–521. Available online: https://www.cabi.org/ISC/abstract/20113039019 (accessed on 19 August 2021).
- Giblin-Davis, R.M.; Faleiro, J.R.; Jacas, J.A.; Peña, J.E.; Vidyasagar, P.S.P.V. Biology and management of the red palm weevil, Rhynchophorus ferrugineus. In Potential Invasive Pests of Agricultural Crops; Peña, J.E., Ed.; CABI International: Wallingford, UK, 2012; pp. 1–34. [Google Scholar]
- Faleiro, J.R.; Ferry, M.; Yaseen, T.; Al-Dobai, S. Overview of the gaps, challenges and prospects of red palm weevil management. Arab J. Plant Prot. 2019, 37, 170–177. [Google Scholar] [CrossRef]
- Porcelli, F.; Scrascia, M.; Pazzani, C.; Pollastro, S.; Di Palma, A.; Russo, V.; Roberto, R.; Salerno, M.; Valentini, F.; Djelouah, K. The RPW as Vector of Bacteria, Fungi and Acari. 2017. Available online: http://www.fao.org/fileadmin/templates/fcc/documents/01_The_RPW_as_vector.pdf (accessed on 19 August 2021).
- Troccoli, A.; Oreste, M.; Fanelli, E.; De Luca, F.; Tarasco, E. Mononchoides macrospiculumn. sp. (Nematoda: Neodiplogastridae) and Teratorhabditis synpapillata Sudhaus, 1985 (Nematoda: Rhabditidae): Nematode associates of Rhynchophorus ferrugineus (Oliver) (Coleoptera: Curculionidae) in Italy. Nematology 2015, 17, 953–966. [Google Scholar] [CrossRef]
- De Luca, F.; Fanelli, E.; Oreste, M.; Scarcia, G.; Troccoli, A.; Vovlas, A.; Tarasco, E. Molecular profiling of nematode associates with Rhynchophorus ferrugineus in southern Italy. Ecol. Evol. 2019, 9, 14286–14294. [Google Scholar] [CrossRef] [PubMed]
- Potamitis, I.; Rigakis, I.; Tatlas, N.-A.; Potirakis, S. In-vivo vibroacoustic surveillance of trees in the context of the IoT. Sensors 2019, 19, 1366. [Google Scholar] [CrossRef]
- Dembilio, O.; Llácer, E.; Martínez de Altube del Mar, M.; Jacas, J.A. Field efficacy of imidacloprid and Steinernema carpocapsae in a chitosan formulation against the red palm weevil Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in Phoenix canariensis. Pest Manag. Sci. 2010, 66, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.M.S.; Araújo, R.P.C.; Sarro, F.B. Táticas de manejo das pragas. In Coco, Fitossanidade; Ferreira, J.M.S., Ed.; Technical Bulletin Brazilian Agricultural Research Corporation (EMBRAPA) Tabuleiros Costeiros: Aracaju, Brazil, 2002; Volume 83, p. 1. [Google Scholar]
- Oliveira, C.A.; Alves, V.M.C.; Gomes, E.A.; Lanna, U.G.P.; Sá, N.M.H.; Marriel, I.E. Otimizacao da Metodologia de Extracao e Amplificacao do DNA de Fungos do Solo. Technical Bulletin of Embrapa Corn and Sorghum. 2005. Available online: https://ainfo.cnptia.embrapa.br/digital/bitstream/CNPMS/18425/1/Circ_69.pdf (accessed on 19 August 2021).
- Navarro-Llopis, V.; Primo, J.; Vacas, S. Improvements in Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) trapping systems. J. Econ. Entomol. 2018, 111, 1298–1305. [Google Scholar] [CrossRef]
- Abbass, M.K.; Nasser, S.A. Ecological studies and evaluation of some aggregation pheromone types with measuring the potential of female reproductive system in red palm weevil, Rhynchophorus ferrugineus (Olivier). Nat. Sci. 2012, 10, 188–193. Available online: http://www.sciencepub.net/nature (accessed on 18 August 2021).
- Abuagla, A.M.; Al-Deeb, M.A. Effect of bait quality and trap color on the trapping efficacy of the pheromone trap for the red palm weevil, Rhynchophorus ferrugineus. J. Insect Sci. 2012, 12, 1–6. [Google Scholar] [CrossRef]
- Arafa, O.E. Field evaluation of synthetic pheromone, allomone, palm kairomone and ester in capturing adult red palm weevils, Rhynchophorus ferrugineus (Olivier) by aggregation pheromone traps in date palm plantations. Plant Arch. 2020, 20, 1857–1862. [Google Scholar]
- Vacas, S.; Melita, O.; Michaelakis, A.; Milonas, P.; Minuz, R.; Riolo, P.; Abbass, M.K.; lo Bue, P.; Colazza, S.; Peri, E.; et al. Lures for red palm weevil trapping systems: Aggregation pheromone and synthetic kairomone. Pest Manag. Sci. 2017, 23, 223–231. [Google Scholar] [CrossRef]
- Vacas, S.; Abab-Payá, M.; Primo, J.; Navarro-Llopis, V. Identification of pheromone synergists for Rhynchophorus ferrugineus trapping systems from Phoenix canariensis palm volatiles. J. Agric. Food Chem. 2014, 62, 6053–6064. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Suckling, D.; Wearing, C.; Byers, J. Potential of mass trapping for long-term pest management and eradication of invasive species. J. Econ. Entomol. 2006, 99, 1550–1564. [Google Scholar] [CrossRef]
- Abdel-Azim, M.M.; Aldosari, S.A.; Mumtaz, R.; Vidyasagar, P.S.P.V. Pheromone trapping system for Rhynchophorus ferrugineus in Saudi Arabia: Optimization of trap contents and placement. Emir. J. Food Agric. 2017, 29, 936–948. [Google Scholar] [CrossRef]
- Abbas, M.K.A.; El-Deeb, M.A.; El-Zohairy, M.M.; Arafa, O.E. Impact of the aggregation pheromone traps baited with fermented food materials on the attraction of the red palm weevil, Rhynchophorus ferrugineus (Olivier) in Egypt. Egypt. J. Agric. Res. 2019, 97, 67–75. [Google Scholar] [CrossRef]
- Ishak, I.; Ng, L.C.; Haris-Hussain, M.; Jalinas, J.; Idris, A.B.; Azlina, Z.; Samsudin, A.; Wahizatul, A.A. Pathogenicity of an indigenous strain of the entomopathogenic fungus Metarhizium anisopliae (Hypocreales: Clavicipitaceae) (MET-GRA4 Strain) as a potential biological control agent against the red palm weevil (Coleoptera: Dryophthoridae). J. Econ. Entomol. 2020, 113, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Lima, H.M.A.; de Rodrigues, V.M.; Sabino, A.R.; dos Santos, M.Q.C.; de Lima, I.S.; Duarte, A.G.; Negrisoli, A.S., Jr.; da Silva, J.M. Effect of isolates of entomopathogenic fungi in the coconut eye borer. Comun. Sci. 2020, 11, e3300. [Google Scholar] [CrossRef]
- Negrisoli, A.S., Jr.; Barbosa, C.R.C.; Moino, J.R.A. Avaliação da compatibilidade de produtos fitossanitários com nematoides entomopatogênicos (Rhabditida: Steinernematidae: Heterorhabditidae) utilizando o protocolo da IOBC/WPRS. Nematol. Bras. 2008, 32, 111–116. [Google Scholar]
- Yasin., M.; Wakil, W.; Ghazanfar, M.U.; Qayyum, M.A.; Tahir, M.; Bedford, G.O. Virulence of entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae against red palm weevil, Rhynchophorus ferrugineus (Olivier). Entomol. Res. 2019, 49, 3–12. [Google Scholar] [CrossRef]
- Lo Verde, G.; Torta, L.; Mondello, V.; Caldarella, C.G.; Burruano, S.; Caleca, V. Pathogenicity bioassays of isolates of Beauveria bassiana on Rhynchophorus ferrugineus. Pest Manag. Sci. 2014, 71, 323–328. [Google Scholar] [CrossRef]
- Sanjuan, T.; Tabima, J.; Restrepo, S.; Læssøe, T.; Spatafora, J.W.; Franco-Molano, A.E. Entomopathogens of Amazonian stick insects and locusts are members of the Beauveria species complex (Cordyceps sensu stricto). Mycologia 2014, 106, 260–275. [Google Scholar] [CrossRef]
- Alves, S.B. Quantificação de inóculo de patógenos de inseto. In Controle Microbiano de Insetos, 2nd ed.; Alves, S.B., Ed.; FEALQ: Piracicaba, Brazil, 1998; pp. 765–777. [Google Scholar]
- Gallo, D.; Nakano, O.; Silveira Neto, S.; Carvalho, R.P.L.; de Baptista, G.C.; Berti Filho, E.; Parra, J.R.; Zucchi, R.A.; Alves, S.B.; Vendramim, J.D.; et al. Entomologia Agricola; FEALQ: Piracicaba, Brazil, 2002; 920p. [Google Scholar]
- Fernandes, E.K.K.; Angelo, I.C.; Rangel, D.E.N.; Bahiense, T.C.; Moraes, A.M.L.; Roberts, D.W.; Bittencourt, V.R.E.P. An intensive search for promising fungal biological control agents of ticks, particularly Rhipicephalus microplus. Vet. Parasitol. 2011, 182, 307–318. [Google Scholar] [CrossRef]
- Wang, C.; Wang, S. Insect pathogenic fungi: Genomics. Molecular interactions and genetic improvements. Ann. Rev. Ent. 2017, 62, 73–90. [Google Scholar] [CrossRef]
- Kandasamy, D.; Gershenzon, J.; Hammerbacher, A. Volatile organic compounds emitted by fungal associates of conifer bark beetles and their potential in bark beetle control. J. Chem. Ecol. 2016, 42, 952–969. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Polle, A.; Brinkmann, N. Belowground communication: Impacts of volatile organic compounds (VOCs) from soil fungi on other soil-inhabiting organisms. Appl. Microbiol. Biotechnol. 2016, 100, 8651–8665. [Google Scholar] [CrossRef] [PubMed]
- Ormond, E.L.; Thomas, A.P.M.; Pell, J.K.; Freeman, S.N.; Roy, H.E. Avoidance of a generalist entomopathogenic fungus by the ladybird, Coccinella septempunctata. FEMS Microbiol. Ecol. 2011, 77, 229–237. [Google Scholar] [CrossRef] [PubMed]
- El-Shafie, H.A.F.; Faleiro, J.R. Optimizing components of pheromone-baited trap for the management of red palm weevil, Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in date palm agro-ecosystem. J. Plant Dis. Prot. 2017, 124, 279–287. [Google Scholar] [CrossRef]
- Faleiro, J.R.; Al-Shawaf, A.M.; El-Shafie, H.A.F.; Raikar, S.P. Studies on service free semiochemical mediated technologies to control red palm weevil Rhynchophorus ferrugineus Olivier based on trials in Saudi Arabia and India. Arab J. Plant Prot. 2019, 37, 136–142. [Google Scholar] [CrossRef]
- Pluess, T.; Jarošík, V.; Pyšek, P.; Cannon, R.; Pergl, J.; Breukers, A.; Bacher, S. Which factors affect the success or failure of eradication campaigns against alien species? PLoS ONE 2012, 7, 48157. [Google Scholar] [CrossRef]
- Porcelli, F.; Cornara, D. Mezzi e metodi di controllo integrato del Punteruolo Rosso delle Palme. Atti Accad. Naz. Ital. Entomol. 2013, LXI, 233–238. [Google Scholar]
- Bautista-Giraldo, M.A.; Armbrecht, I.; Vásquez-Ordoñez, A.A. The weevil Dynamis borassi (Coleoptera: Curculionidae: Dryophthorinae) associated with native palms in forests and disturbed areas in Buenaventura, Colombia. Rev. Colomb. Entomol. 2020, 46, e7721. [Google Scholar] [CrossRef]
- Gaviria, J.; Montoya-Lerma, J.; Armbrecht, I.; Löhr, B.; Vásquez-Ordóñez, A.A. Dynamis borassi (Coleoptera: Curculionidae), a new potential pest to the palms (Arecaceae): An early warning for the palm producers. Fla. Entomol. 2021, 104, 107–116. [Google Scholar] [CrossRef]
- Picciotti, U.; Lahbib, N.; Sefa, V.; Porcelli, F.; Garganese, F. Aphrophoridae role in Xylella fastidiosa subsp. pauca ST53 invasion in southern Italy. Pathogens 2021, 10, 1035. [Google Scholar] [CrossRef]
- Fierro, A.; Liccardo, A.; Porcelli, F. A lattice model to manage the vector and the infection of the Xylella fastidiosa on olive trees. Sci. Rep. 2019, 9, 8723. [Google Scholar] [CrossRef] [PubMed]
- Liccardo, A.; Fierro, A.; Garganese, F.; Picciotti, U.; Porcelli, F. A biological control model to manage the vector and the infection of Xylella fastidiosa on olive trees. PLoS ONE 2020, 15, e0232363. [Google Scholar] [CrossRef]
- Abbas, M.S.T.; Hanounik, S.B.; Shahdad, A.S. Aggregation pheromone traps, a major component of IPM strategy for the red palm weevil, Rhynchophorus ferrugineus in date palms (Coleoptera: Curculionidae). J. Pest Sci. 2006, 79, 69–73. [Google Scholar] [CrossRef]
- Negrisoli, A.S., Jr.; Silva, E.S.; Barbosa Negrisoli, C.R.C.; Santos, N.L.; Guzzo, E.C. Criação em Laboratório da Broca-do-Olho-do-Coqueiro Rhynchophorus palmarum L. (Coleoptera: Curculionidae) Visando Pesquisas para o Controle das Suas Larvas. Aracaju: Embrapa Tabuleiros Costeiros Comunicado Técnico, 116. 2011. Available online: http://www.cpatc.embrapa.br/publicacoes_2011/cot_116.pdf (accessed on 19 August 2021).
- Kassem, H.S.; Alotaibi, B.A.; Ahmed, A.; Aldosri, F.O. Sustainable Management of the Red Palm Weevil: The Nexus between Farmers’ Adoption of Integrated Pest Management and Their Knowledge of Symptoms. Sustainability 2020, 12, 9647. [Google Scholar] [CrossRef]
- Leefmans, S. De Palmsnuitkever (Rhynchophorus ferrugineus Oliv.). Meded. Van Het Inst. Voor Plantenziekten 1920, 43, 1–87. [Google Scholar]
| Rhynchophorus palmarum | Rhynchophorus ferrugineus | ||||||
|---|---|---|---|---|---|---|---|
| Phase | Measure (mm) | Duration (Days) | Colour | Measure (mm) | Duration (Days) | Colour | Source |
| Eggs | 25 × 10 | 2–5 | White | 9 × 29 | 3–5 | white | [15,21] |
| Larve | 75 | 45–70 | Yellowish | 36–46 | 40–60 | yellowish | [15,21] |
| Pupae | 80–100 | 25–45 | Yellowish | 50–95 | 20–25 | yellowish | [15,21] |
| Adults | 35–60 | 60–95 | black and reddish atypical chromatic natural polymorphism | (♂): 19–42 | 60–95 | rust red, typical chromatic natural polymorphism | [9,15,31] |
| (♀): 26–40 | |||||||
| Total cycle | 132 to 215 days | 82 days | [15,21] | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalbon, V.A.; Acevedo, J.P.M.; Ribeiro Junior, K.A.L.; Ribeiro, T.F.L.; da Silva, J.M.; Fonseca, H.G.; Santana, A.E.G.; Porcelli, F. Perspectives for Synergic Blends of Attractive Sources in South American Palm Weevil Mass Trapping: Waiting for the Red Palm Weevil Brazil Invasion. Insects 2021, 12, 828. https://doi.org/10.3390/insects12090828
Dalbon VA, Acevedo JPM, Ribeiro Junior KAL, Ribeiro TFL, da Silva JM, Fonseca HG, Santana AEG, Porcelli F. Perspectives for Synergic Blends of Attractive Sources in South American Palm Weevil Mass Trapping: Waiting for the Red Palm Weevil Brazil Invasion. Insects. 2021; 12(9):828. https://doi.org/10.3390/insects12090828
Chicago/Turabian StyleDalbon, Viviane Araujo, Juan Pablo Molina Acevedo, Karlos Antônio Lisboa Ribeiro Junior, Thyago Fernando Lisboa Ribeiro, Joao Manoel da Silva, Henrique Goulart Fonseca, Antônio Euzébio Goulart Santana, and Francesco Porcelli. 2021. "Perspectives for Synergic Blends of Attractive Sources in South American Palm Weevil Mass Trapping: Waiting for the Red Palm Weevil Brazil Invasion" Insects 12, no. 9: 828. https://doi.org/10.3390/insects12090828
APA StyleDalbon, V. A., Acevedo, J. P. M., Ribeiro Junior, K. A. L., Ribeiro, T. F. L., da Silva, J. M., Fonseca, H. G., Santana, A. E. G., & Porcelli, F. (2021). Perspectives for Synergic Blends of Attractive Sources in South American Palm Weevil Mass Trapping: Waiting for the Red Palm Weevil Brazil Invasion. Insects, 12(9), 828. https://doi.org/10.3390/insects12090828







