Comparison of Biological Activity of Field Isolates of Steinernema feltiae with a Commercial S. feltiae Biopesticide Product
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
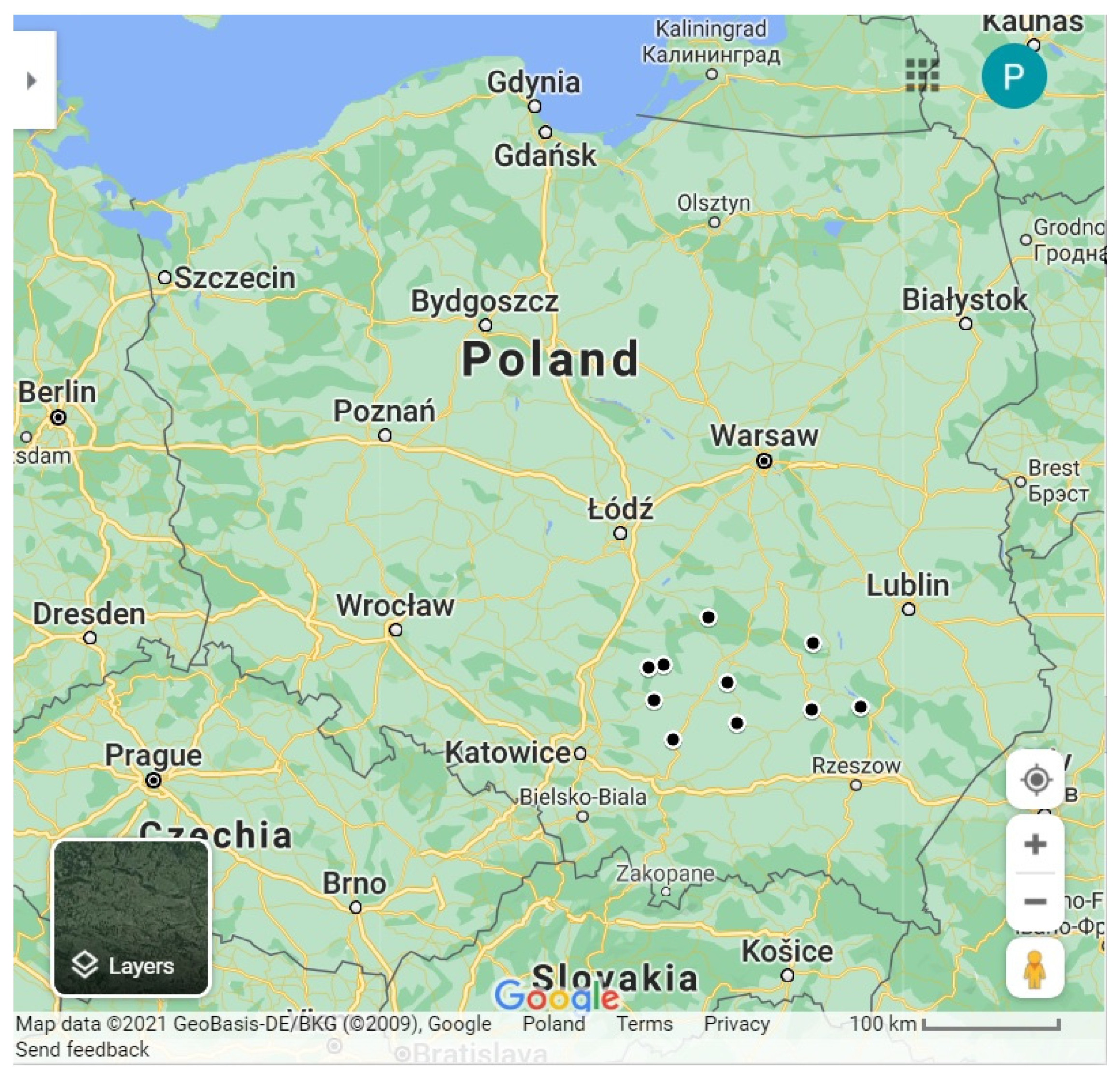
2.1. Soil Samples’ Collection and Nematodes’ Isolation
2.2. Reproduction of the Collected EPN
2.3. Identification of Nematode Species
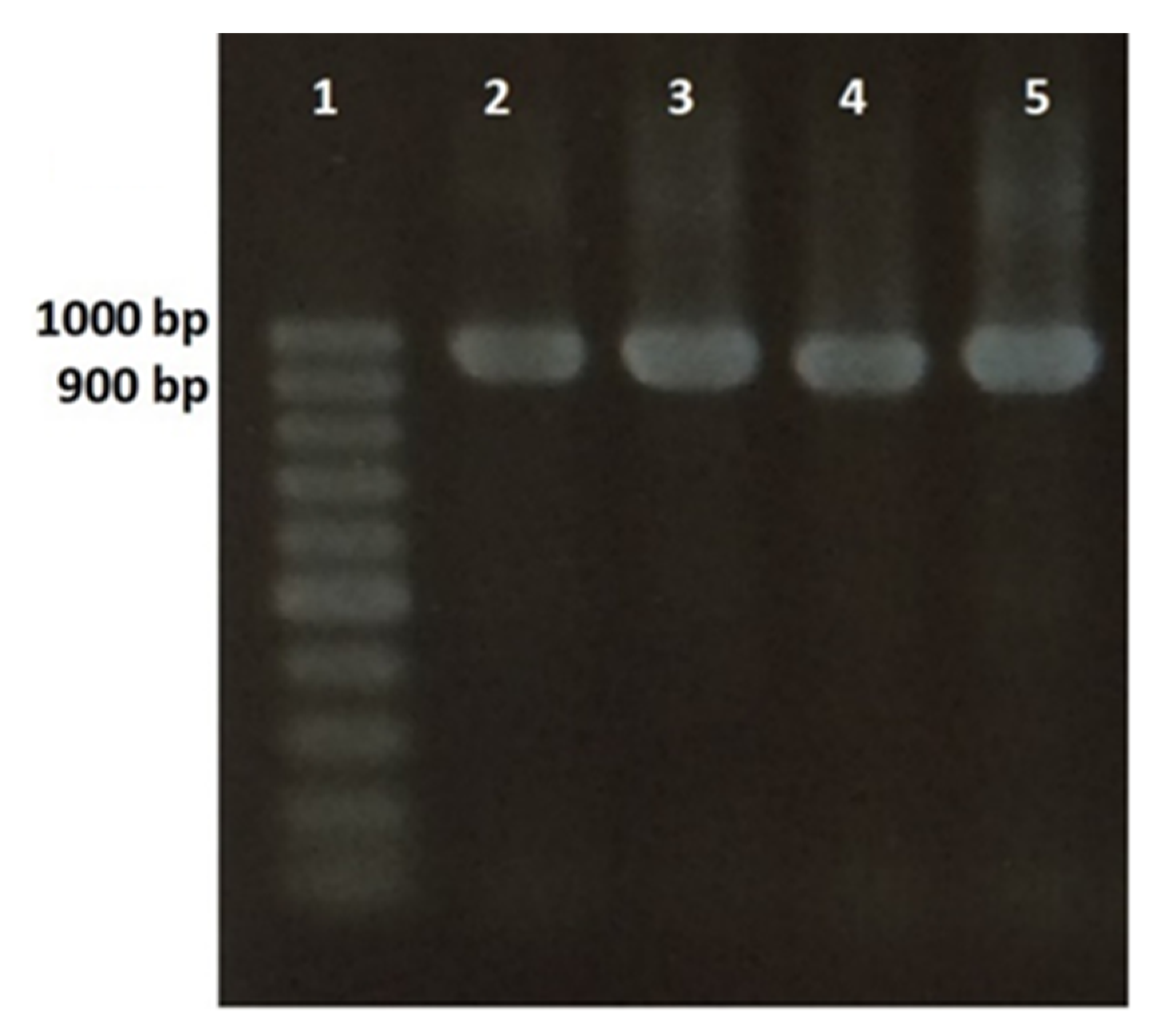
2.4. Gene Sequencing
2.5. Phylogenetic Analysis
2.6. Quantification of Biological Activity of EPN
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Characteristics of the Areas of S. feltiae Isolation
| Locality | Latitude (N) | Longitude (E) | Soil Type | pH |
|---|---|---|---|---|
| Bałtów | 51°01′02.6″ | 21°32′10.7″ | loamy sand | 5.2 |
| Busko-Zdrój | 50°28′06.5″ | 20°41′47.3″ | sandy loam | 5.2 |
| Danków Duży | 50°52′26.5″ | 19°55′29.6″ | sandy loam | 5.2 |
| Końskie | 51°11′38.9″ | 20°23′17.3″ | loamy sand | 5.4 |
| Łoniów | 50°33′31.1″ | 21°31′37.2″ | sandy loam | 5.2 |
| Miechów | 50°21′04.5″ | 20°03′07.4″ | loamy sand | 5.6 |
| Morawica | 50°44′48.4″ | 20°36′52.5″ | loamy sand | 5.4 |
| Oblasy | 50°50′20.1″ | 19°46′31.5″ | sandy loam | 5.4 |
| Stalowa Wola | 50°36′15.2″ | 22°02′32.3″ | loamy sand | 5.2 |
| Szczekociny | 50°37′14.5″ | 19°48′01.1″ | loamy sand | 5.6 |
References
- Abd-Alla, A.M.M.; Meki, I.K.; Demirbas-Uzel, G. Insect Viruses as Biocontrol Agents: Challenges and Opportunities. In Cottage Industry of Biocontrol Agents and Their Applications; El-Wakeil, N., Saleh, M., Abu-hashim, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 277–295. [Google Scholar]
- Ehler, L.E. Some Contemporary Issues in Biological Control of Insects and Their Relevance to the Use of Entomopathogenic Nematodes. In Entomopathogenic Nematodes in Biological Control; Gaugler, R., Kaya, H.K., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 1–20. ISBN 9781351071741. [Google Scholar]
- Gonzalez, F.; Tkaczuk, C.; Dinu, M.M.; Fiedler, Ż.; Vidal, S.; Zchori-Fein, E.; Messelink, G.J. New opportunities for the integration of microorganisms into biological pest control systems in greenhouse crops. J. Pest Sci. 2016, 89, 295–311. [Google Scholar] [CrossRef]
- Lahiri, S.; Orr, D. Biological Control in Tomato Production Systems: Theory and Practice. In Sustainable Management of Arthropod Pests of Tomato; Wakil, W., Brust, G.E., Perring, T.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 253–267. ISBN 9780128135082. [Google Scholar]
- Dillman, A.R.; Sternberg, P.W. Entomopathogenic Nematodes. Curr. Biol. 2012, 22, 430–431. [Google Scholar] [CrossRef] [PubMed]
- Butt, T.M.; Coates, C.J.; Dubovskiy, I.; Ratcliffe, N.A. Entomopathogenic Fungi: New Insights into Host-Pathogen Interactions. In Genetics and Molecular Biology of Entomopathogenic Fungi; Lovett, B., St Leger, R.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; Volume 94, p. 364. ISBN 9780128046944. [Google Scholar]
- Brivio, M.F.; Mastore, M. Nematobacterial Complexes and Insect Hosts: Different Weapons for the Same War. Insects 2018, 9, 117. [Google Scholar] [CrossRef]
- Melo, A.L.D.A.; Soccol, V.T.; Soccol, C.R. Bacillus thuringiensis: Mechanism of action, resistance, and new applications: A review. Crit. Rev. Biotechnol. 2014, 36, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Strand, M.R.; Burke, G.R. Polydnaviruses: Evolution and Function. Curr. Issues Mol. Biol. 2020, 34, 163–181. [Google Scholar] [CrossRef] [PubMed]
- Matuska-Łyżwa, J. Ecological and morphological characteristics of Steinernema kraussei (Rhabditida: Steinernematidae): Comparison of nematodes isolated from the natural environments and originated from the commercial pesticide. Ecol. Quest. 2014, 19, 51–55. [Google Scholar] [CrossRef][Green Version]
- Mráček, Z.; Bečvář, S.; Kindlmann, P.; Webster, J.M. Infectivity and specificity of Canadian and Czech isolates of Steinernema kraussei (Steiner, 1923) to some insect pests at low temperatures in the laboratory. Nematologica 1998, 44, 437–448. [Google Scholar] [CrossRef]
- Nguyen, K.B.; Hunt, D.J.; Mráček, Z. Steinernematidae: Species description. In Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts; Nguyen, K.B., Hunt, D., Eds.; Brill: Leiden, The Netherland, 2007; pp. 270–282. [Google Scholar]
- Kagimu, N.; Ferreira, T.; Malan, A.P. The Attributes of Survival in the Formulation of Entomopathogenic Nematodes Utilised as Insect Biocontrol Agents. Afr. Entomol. 2017, 25, 275–291. [Google Scholar] [CrossRef]
- Makirita, W.E.; Zhang, F.; Mbega, E.R.; He, N.; Li, X.; Chacha, M.; Liu, T. Influence of Metal Oxides Nanoparticles on Pathogenicity of Steinernema carpocapsae Nematodes Against Lepidopteran Galleria mellonella. J. Nanosci. Nanotechnol. 2019, 20, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Santhi, V.S.; Salame, L.; Muklada, H.; Azaizeh, H.; Haj-Zaroubi, M.; Awwad, S.; Landau, S.Y.; Glazer, I. Toxicity of phenolic compounds to entomopathogenic nematodes: A case study with Heterorhabditis bacteriophora exposed to lentisk (Pistacia lentiscus) extracts and their chemical components. J. Invertebr. Pathol. 2019, 160, 43–53. [Google Scholar] [CrossRef]
- Widiyaningrum, P.; Fauziyah, L.; Indriyanti, D.R. Biological activity of local entomopathogenic nematodes from two different origins based on various temperatures. Pak. J. Biol. Sci. 2018, 21, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Dzięgielewska, M.; Berdzik, M.; Myśków, B. The first molecular characterisation of Steinernema silvaticum recorded in Poland and ITS differentiation from Steinernema kraussei using ribosomal DNA (rDNA) sequences. Folia Pomer. Univ. Technol. Stetin. Agric. Aliment. Pisc. Zootech. 2015, 316, 41–48. [Google Scholar]
- Grewal, P.S.; De Nardo, E.A.B.; Aguillera, M.M. Entomopathogenic nematodes: Potential for exploration and use in south America. Neotrop. Entomol. 2001, 30, 191–205. [Google Scholar]
- Kaspi, R.; Ross, A.; Hodson, A.K.; Stevens, G.N.; Kaya, H.K.; Lewis, E.E. Foraging efficacy of the entomopathogenic nematode Steinernema riobrave in different soil types from California citrus groves. Appl. Soil Ecol. 2010, 45, 243–253. [Google Scholar] [CrossRef]
- Kaya, H.K.; Aguillera, M.M.; Alumai, A.; Choo, H.Y.; de la Torre, M.; Fodor, A.; Ganguly, S.; Hazir, S.; Lakatos, T.; Pye, A.; et al. Status of entomopathogenic nematodes and their symbiotic bacteria from selected countries or regions of the world. Biol. Control 2006, 38, 134–155. [Google Scholar] [CrossRef]
- San-Blas, E.; Luzardo, M.; Larreal, J.; Portillo, E.; Bastidas, B. Biological control of the fungus gnats Bradysia difformis (Diptera, Mycetophilidae) in mushrooms with Heterorhabditis amazonensis in tropical conditions. Sci. Hortic. 2017, 216, 120–125. [Google Scholar] [CrossRef]
- Burnell, A.M.; Stock, S.P. Heterorhabditis, Steinernema and their bacterial symbionts—Lethal pathogens of insects. Nematology 2000, 2, 31–42. [Google Scholar] [CrossRef]
- Lechowicz, L.; Chrapek, M.; Czerwonka, G.; Korzeniowska-Kowal, A.; Tobiasz, A.; Urbaniak, M.; Matuska-Lyzwa, J.; Kaca, W. Detection of ureolytic activity of bacterial strains isolated from entomopathogenic nematodes using infrared spectroscopy. J. Basic Microbiol. 2016, 56, 922–928. [Google Scholar] [CrossRef]
- Somvanshi, V.S.; Lang, E.; Stäubler, B.; Spröer, C.; Schumann, P.; Ganguly, S.; Saxena, A.K.; Stackebrandt, E. Providencia vermicola sp. nov., isolated from infective juveniles of the entomopathogenic nematode Steinernema thermophilum. Int. J. Syst. Evol. Microbiol. 2006, 56, 629–633. [Google Scholar] [CrossRef]
- Ogier, J.C.; Pagès, S.; Frayssinet, M.; Gaudriault, S. Entomopathogenic nematode-associated microbiota: From monoxenic paradigm to pathobiome. Microbiome 2020, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Grewal, P.S.; Selvan, S.; Gaugler, R. Thermal adaptation of entomopathogenic nematodes: Niche breadth for infection, establishment, and reproduction. J. Therm. Biol. 1994, 19, 245–253. [Google Scholar] [CrossRef]
- Trdan, S.; Valič, N.; Urek, G.; Milevoj, L. Concentration of suspension and temperature as factors of pathogenicity of entomopathogenic nematodes for the control of granary weevil, Sitophilus granarius (L.) (Coleoptera: Curculionidae). Acta Agric. Slov. 2005, 1, 117–124. [Google Scholar]
- Mastore, M.; Quadroni, S.; Toscano, A.; Mottadelli, N.; Brivio, M.F. Susceptibility to entomopathogens and modulation of basal immunity in two insect models at different temperatures. J. Therm. Biol. 2019, 79, 15–23. [Google Scholar] [CrossRef]
- Fodor, A.; Vecseri, G.; Farkas, T. Caenorhabditis elegans as a model for the study of entomopathogenic nematodes. In Entomopathogenic Nematodes in Biological Control; Gaugler, R., Kaya, H.K., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 247–270. ISBN 9781351071741. [Google Scholar]
- McGraw, B.A.; Schlossberg, M.J. Fine-scale spatial analysis of soil moisture and entomopathogenic nematode distribution following release in wetting agent-treated turf. Appl. Soil Ecol. 2017, 114, 52–61. [Google Scholar] [CrossRef]
- Radová, Š.; Trnková, Z. Effect of soil temperature and moisture on the pathogenicity of two species of entomopathogenic nematodes (Rhabditida: Steinernematidae). J. Agrobiol. 2010, 27, 1–7. [Google Scholar] [CrossRef]
- Grant, J.A.; Villani, M.G. Soil moisture effects on entomopathogenic nematodes. Environ. Entomol. 2003, 32, 80–87. [Google Scholar] [CrossRef]
- Jaffuel, G.; Blanco-Pérez, R.; Büchi, L.; Mäder, P.; Fließbach, A.; Charles, R.; Degen, T.; Turlings, T.C.J.; Campos-Herrera, R. Effects of cover crops on the overwintering success of entomopathogenic nematodes and their antagonists. Appl. Soil Ecol. 2017, 114, 62–73. [Google Scholar] [CrossRef]
- Wu, S.Y.; El-Borai, F.E.; Graham, J.H.; Duncan, L.W. Geospatial relationships between native entomopathogenic nematodes and Fusarium solani in a Florida citrus orchard. Appl. Soil Ecol. 2019, 140, 108–114. [Google Scholar] [CrossRef]
- Boemare, N.; Laumond, C.; Mauleon, H. The entomopathogenic nematode-bacterium complex: Biology, life cycle and vertebrate safety. Biocontrol Sci. Technol. 1996, 6, 333–346. [Google Scholar] [CrossRef]
- Ehlers, R.U. Current and future use of nematodes in biocontrol: Practice and commercial aspects with regard to regulatory policy issues. Biocontrol Sci. Technol. 1996, 6, 303–316. [Google Scholar] [CrossRef]
- Gaugler, R. Ecological considerations in the biological control of soil-inhabiting insects with entomopathogenic nematodes. Agric. Ecosyst. Environ. 1988, 24, 351–360. [Google Scholar] [CrossRef]
- Agazadeh, M.; Mohammadi, D.; Kary, N.E. Distribution of entomopathogenic nematodes of the families Steinernematidae and Heterorhabditidae in potato fields in North-West Iran (Nematoda: Rhabditida). Munis Entomol. Zool. 2010, 5, 758–763. [Google Scholar]
- Iraki, N.; Salah, N.; Sansour, M.A.; Segal, D.; Glazer, I.; Johnigk, S.A.; Hussein, M.A.; Ehlers, R.U. Isolation and characterization of two entomopathogenic nematode strains, Heterorhabditis indica (Nematoda, Rhabditida), from the West Bank, Palestinian territories. J. Appl. Entomol. 2000, 124, 375–380. [Google Scholar] [CrossRef]
- Lakatos, T.; Toth, T. Biological control of European cockchafer larvae [Melolontha melolontha L.]—Preliminary results. J. Fruit Ornam. Plant Res. 2006, 14, 73–78. [Google Scholar]
- Toth, T. Collection of entomopathogenic nematodes for the biological control of insect pests. J. Fruit Ornam. Plant Res. 2006, 14, 225–230. [Google Scholar]
- Matuska-Łyzwa, J. Effectiveness of selected strains of entomopathogenic nematodes in peat substrate in container breeding. Ecol. Quest. 2013, 18, 63–67. [Google Scholar] [CrossRef][Green Version]
- Matuska-Łyżwa, J. Invasiveness and the morphometry of Steinernema feltiae from different agrocoenoses of Poland. Ecol. Chem. Eng. A 2013, 20, 565–572. [Google Scholar]
- Stock, S.P. Molecular approaches and the taxonomy of insect-parasitic and pathogenic nematodes. In Insect Pathogens: Molecular Approaches and Techniques; Stock, S.P., Vandenberg, J., Glazer, I., Boemare, N., Eds.; CABI International: Oxfordshire, UK, 2009; pp. 71–100. ISBN 9781845934781. [Google Scholar]
- Matuska-Łyżwa, J.; Kaca, W.; Żarnowiec, P. Biological activity of wild isolates of entomopathogenic nematodes to horse-chestnut leaf miner (Cameraria ohridella). Pol. J. Environ. Stud. 2015, 24, 1181–1184. [Google Scholar] [CrossRef]
- Fan, X.; Hominick, W.M. Efficiency of the Galleria (wax moth) baiting technique for recovering infective stages of entomopathogenic rhabditids (Steinernematidae and Heterorhabditidae) from Sand and soil. Rev. Nématol. 1991, 14, 381–387. [Google Scholar]
- Matuska-Łyżwa, J. Method of Multiplication of Entomopathogenic Nematodes for the Plant Protection Research Purposes. WUP, Patent PL 212617 B1, 31 October 2012. [Google Scholar]
- Koppenhöfer, A.M. Nematodes. In Field Manual of Techniques in Invertebrate Pathology; Lacey, L., Kaya, H.K., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 249–264. [Google Scholar]
- Kaya, H.K.; Stock, S.P. Techniques in insect nematology. In Field Manual of Techniques in Invertebrate Pathology; Lacey, L.A., Ed.; Academic Press: Cambridge, MA, USA, 1997; pp. 281–324. [Google Scholar]
- Filipjev, I.N. On the classification of the Tylenchinae. Proc. Helminthol. Soc. Wash. 1936, 3, 80–82. [Google Scholar]
- Nguyen, K.B. Methodology, morphology and identification. In Entomopathogenic Nematodes: Systematics, Phylogeny and Bacterial Symbionts; Nguyen, K.B., Hunt, D., Eds.; Brill: Leiden, The Netherland, 2007; pp. 59–119. [Google Scholar]
- Dereeper, A.; Guignon, V.; Blanc, G.; Audic, S.; Buffet, S.; Chevenet, F.; Dufayard, J.F.; Guindon, S.; Lefort, V.; Lescot, M.; et al. Phylogeny.fr: Robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 2008, 36, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.B.; Mráček, Z.; Webster, J.M. Morphological and molecular characterization of a new isolate of Steinernema feltiae (Filipjev, 1934) from Vancouver, Canada, with morphometrical comparison with the topotype population from Russia. Zootaxa 2006, 1132, 51–61. [Google Scholar] [CrossRef]
- Helmberger, M.S.; Shields, E.J.; Wickings, K.G. Ecology of belowground biological control: Entomopathogenic nematode interactions with soil biota. Appl. Soil Ecol. 2017, 121, 201–213. [Google Scholar] [CrossRef]
- Keshari, A.K.; Hari, B.K.C.; Bhat, A.H.; Shah, M.M. Prospects and Present Status and of Entomopathogenic Nematodes (Steinernematidae and Heterorhabditidae) in Nepal. J. Appl. Adv. Res. 2019, 4, 31–35. [Google Scholar] [CrossRef]
- Tarasco, E.; Clausi, M.; Rappazzo, G.; Panzavolta, T.; Curto, G.; Sorino, R.; Oreste, M.; Longo, A.; Leone, D.; Tiberi, R.; et al. Biodiversity of entomopathogenic nematodes in Italy Biodiversity of entomopathogenic nematodes in Italy. J. Helminthol. 2015, 89, 359–366. [Google Scholar] [CrossRef]
- Malan, A.P.; Knoetze, R.; Tiedt, L.R. Steinernema jeffreyense n. sp. (Rhabditida: Steinernematidae), a new entomopathogenic nematode from South Africa. J. Helminthol. 2016, 90, 262–278. [Google Scholar] [CrossRef] [PubMed]
- Nikdel, M.; Niknam, G.; Kary, N.E. First report of an entomopathogenic nematode, Steinernema kraussei (Rhabditida, Steinernematidae) from Iran. J. Entomol. Soc. Iran 2011, 30, 39–51. [Google Scholar]
- Campos-Herrera, R.; Escuer, M.; Robertson, L.; Gutiérrez, C. Morphological and ecological characterization of Steinernema feltiae (Rhabditida: Steinernematidae) rioja strain isolated from Bibio hortulanus (Diptera: Bibionidae) in Spain. J. Nematol. 2006, 38, 68–75. [Google Scholar] [PubMed]
- Çimen, H.; Půža, V.; Nermuť, J.; Hatting, J.; Ramakuwela, T.; Faktorová, L.; Hazir, S. Steinernema beitlechemi n. sp., a new entomopathogenic nematode (Nematoda: Steinernematidae) from South Africa. Nematology 2016, 18, 439–453. [Google Scholar] [CrossRef]
- De Brida, A.L.; Rosa, J.M.O.; De Oliveira, C.M.G.; De Castro E Castro, B.M.; Serrão, J.E.; Zanuncio, J.C.; Leite, L.G.; Wilcken, S.R.S. Entomopathogenic nematodes in agricultural areas in Brazil. Sci. Rep. 2017, 7, 45254. [Google Scholar] [CrossRef] [PubMed]
- Dzięgielewska, M.; Skwiercz, A. The Influence of Selected Abiotic Factors on the Occurrence of Entomopathogenic Nematodes (Steinernematidae, Heterorhabditidae) in Soil. Pol. J. Soil Sci. 2018, 51, 11–21. [Google Scholar] [CrossRef]
- Hominick, W.M.; Reid, A.P.; Bohan, D.A.; Briscoe, B.R. Entomopathogenic nematodes: Biodiversity, geographical distribution and the convention on biological diversity. Biocontrol Sci. Technol. 1996, 6, 317–332. [Google Scholar] [CrossRef]
- El Khoury, Y.; Oreste, M.; Noujeim, E.; Nemer, N.M.; Tarasco, E. Effect of temperature on the pathogenicity of mediterranean native entomopathogenic nematodes (steinernematidae and heterorhabditidae) from natural ecosystems effect of temperature on the pathogenicity of mediterranean native entomopathogenic nematodes. Redia 2018, 101, 123–127. [Google Scholar] [CrossRef]
- Matuska-Łyżwa, J. Invasiveness and reproduction of the Steinernema feltiae from selected agrocoenose located in Wielun. Ecol. Chem. Eng. A 2015, 22, 327–334. [Google Scholar] [CrossRef]
- Lortkipanidze, M.; Gorgadze, O.; Kokhia, M.; Melashvili, N.; Kuchava, M. Effectiveness of entomopathogenic nematodes (Steinernema carpocapsae) against the Melolontha hippocastani (Coleoptera: Scarabaeidae). Bull. Georgian Natl. Acad. Sci. 2011, 5, 155–157. [Google Scholar]
- Noosidum, A.; Hodson, A.K.; Lewis, E.E.; Chandrapatva, A. Characterization of new entomopathogenic nematodes from thailand: Foraging behavior and virulence to the greater wax moth, Galleria mellonella L. (Lepidoptera: Pyralidae). J. Nematol. 2010, 42, 281–291. [Google Scholar]
- McMullen, J.G.; Patricia Stock, S. In vivo and in vitro rearing of entomopathogenic nematodes (Steinernematidae and Heterorhabditidae). J. Vis. Exp. 2014, 52096. [Google Scholar] [CrossRef]
- Rahoo, A.M.; Mukhtar, T.; Gowen, S.R.; Rahoo, R.K.; Abro, S.I. Reproductive potential and Host Searching Ability of Entomopathogenic nematode, Steinernema feltiae. Pak. J. Zool. 2017, 49, 229–234. [Google Scholar] [CrossRef]

| Character | Infective Juvenile | |||||
|---|---|---|---|---|---|---|
| Control (S. feltiae from Biopreparation) | S. feltiae (iso1Lon) | S. feltiae (iso1Dan) | S. feltiae (iso1Obl) | S. feltiae [53] | F (df1, df2), p | |
| n | 25 | 25 | 25 | 25 | 20 | |
| L | 861.2 ± 47 | 875 ± 54 | 816 ± 46 | 749 ± 54 | 879 ± 49 | F = 29.9666 (3, 96), p < 0.00001 |
| (760–950) | (768–927) | (720–890) | (640–850) | (766–928) | ||
| W | 27 ± 1.7 | 28 ± 1.9 | 28 ± 2 | 26 ± 1.4 | 29 ± 1.9 | F = 9.5558 (3, 96), p = 0.00001 |
| (25–32) | (24–31) | (25–32) | (23–29) | (26–32) | ||
| EP | 59 ± 1.9 | 61 ± 2.5 | 58 ± 1.5 | 56 ± 1.9 | 63 ± 2.3 | F = 26.0012 (3, 96), p < 0.00001 |
| (57–63) | (57–65) | (56–61) | (53–59) | (58–67) | ||
| NR | 111 ± 3.2 | 112 ± 2.8 | 111 ± 1.7 | 106 ± 2 | 113 ± 5.1 | F = 29.1955 (3, 96), p < 0.00001 |
| (107–116) | (108–116) | (108–115) | (102–110) | (108–117) | ||
| ES | 135 ± 3.6 | 135 ± 3.4 | 134 ± 3 | 128 ± 3.4 | 136 ± 3.5 | F = 24.7273 (3, 96), p < 0.00001 |
| (130–142) | (128–141) | (127–141) | (120–136) | (130–143) | ||
| T | 82 ± 1.7 | 83 ± 1.9 | 82 ± 2.4 | 79 ± 2 | 86 ± 2.6 | F = 14.944 (3, 96), p < 0.00001 |
| (80–86) | (79–87) | (77–87) | (75–83) | (81–89) | ||
| ABW | 16 ± 1.1 | 17 ± 0.7 | 16 ± 0.8 | 15 ± 0.8 | 18 ± 0.8 | F = 20.6043 (3, 96), p < 0.00001 |
| (15–18) | (15–18) | (14–18) | (13–17) | (16–19) | ||
| a | 31.3 | 31.2 | 28.9 | 29 | 30 | - |
| b | 6.4 | 6.5 | 6.1 | 5.9 | 6.4 | - |
| c | 10.4 | 10.5 | 9.9 | 9.4 | 10 | - |
| D% | 43.8 | 45.1 | 43.4 | 43.7 | 46 | - |
| E% | 71.8 | 73.4 | 71 | 70.4 | 74 | - |
| Character | Male First Generation | |||||
|---|---|---|---|---|---|---|
| Control (S. feltiae from Biopreparation) | S. feltiae (iso1Lon) | S. feltiae (iso1Dan) | S. feltiae (iso1Obl) | S. feltiae [53] | F (df1, df2), p | |
| n | 25 | 25 | 25 | 25 | 20 | |
| L | 1559 ± 93 | 1582 ± 91 | 1513 ± 88 | 1546 ± 96 | 1612 ± 88 | F = 2.3369 (3, 96), p = 0.07851 |
| (1360–1720) | (1410–1795) | (1370–1690) | (1330–1769) | (1414–1817) | ||
| W | 134 ± 10.7 | 138 ± 9 | 124 ± 10 | 133 ± 10 | 140 ± 10 | F = 8.759 (3, 96), p = 0.00003 |
| (118–157) | (118–160) | (105–140) | (108–158) | (121–162) | ||
| EP | 113 ± 3.6 | 115 ± 3.2 | 110 ± 2.9 | 112 ± 2.4 | 115 ± 3.4 | F = 12.2043 (3, 96), p < 0.00001 |
| (108–121) | (109–123) | (105–115) | (108–116) | (110–126) | ||
| ES | 169 ± 4.5 | 169 ± 3.5 | 163 ± 4 | 166 ± 4.8 | 170 ± 3.4 | F = 12.0016 (3, 96), p < 0.00001 |
| (161–176) | (163–177) | (155–170) | (159–175) | (164–180) | ||
| T | 36 ± 1.6 | 37 ± 1.4 | 35 ± 1.3 | 36 ± 1.4 | 39 ± 1.2 | F = 6.3343 (3, 96), p = 0.00058 |
| (34–41) | (35–41) | (33–39) | (34–40) | (37–43) | ||
| ABW | 44 ± 2.4 | 47 ± 2.3 | 42 ± 2.4 | 43 ± 2.1 | 48 ± 1.7 | F = 20.1168 (3, 96), p < 0.00001 |
| (42–51) | (42–52) | (36–48) | (37–49) | (43–53) | ||
| SL | 65 ± 1.7 | 64 ± 1.7 | 63 ± 1.8 | 64 ± 1.6 | 66 ± 1.5 | F = 6.0791 (3, 96), p = 0.00078 |
| (61–68) | (61–68) | (58–67) | (60–66) | (62–68) | ||
| GL | 51 ± 2.2 | 51 ± 2.2 | 47 ± 2.3 | 49 ± 2.1 | 52 ± 1.9 | F = 13.4403 (3, 96), p < 0.00001 |
| (47–57) | (46–55) | (42–52) | (44–54) | (48–56) | ||
| a | 11.67 | 11.43 | 12.22 | 11.63 | 11.5 | - |
| b | 9.3 | 9.3 | 9.3 | 9.3 | 9.5 | - |
| c | 43.1 | 42.5 | 42.7 | 42.8 | 41.3 | - |
| D% | 67 | 68 | 67.6 | 67.5 | 68 | - |
| E% | - | - | - | - | - | - |
| Parameter | Control S. feltiae (from Biopreparation) | S. feltiae (iso1Lon) | S. feltiae (iso1Dan) | S. feltiae (iso1Obl) |
|---|---|---|---|---|
| Insect mortality (%) | 100 a | 100 a | 100 a | 95 b |
| Infectivity (%) | 98 a | 97 a | 100 a | 92 a |
| Number of nematodes invading per insect (no. of ind.) | 10 a | 27 c | 12 b | 12 b |
| Time to kill insects (days) | 2 a | 3 a | 2 a | 3 a |
| Time from nematode infection to emergence of infective juveniles (days) | 10 a | 11 a | 10 a | 11 a |
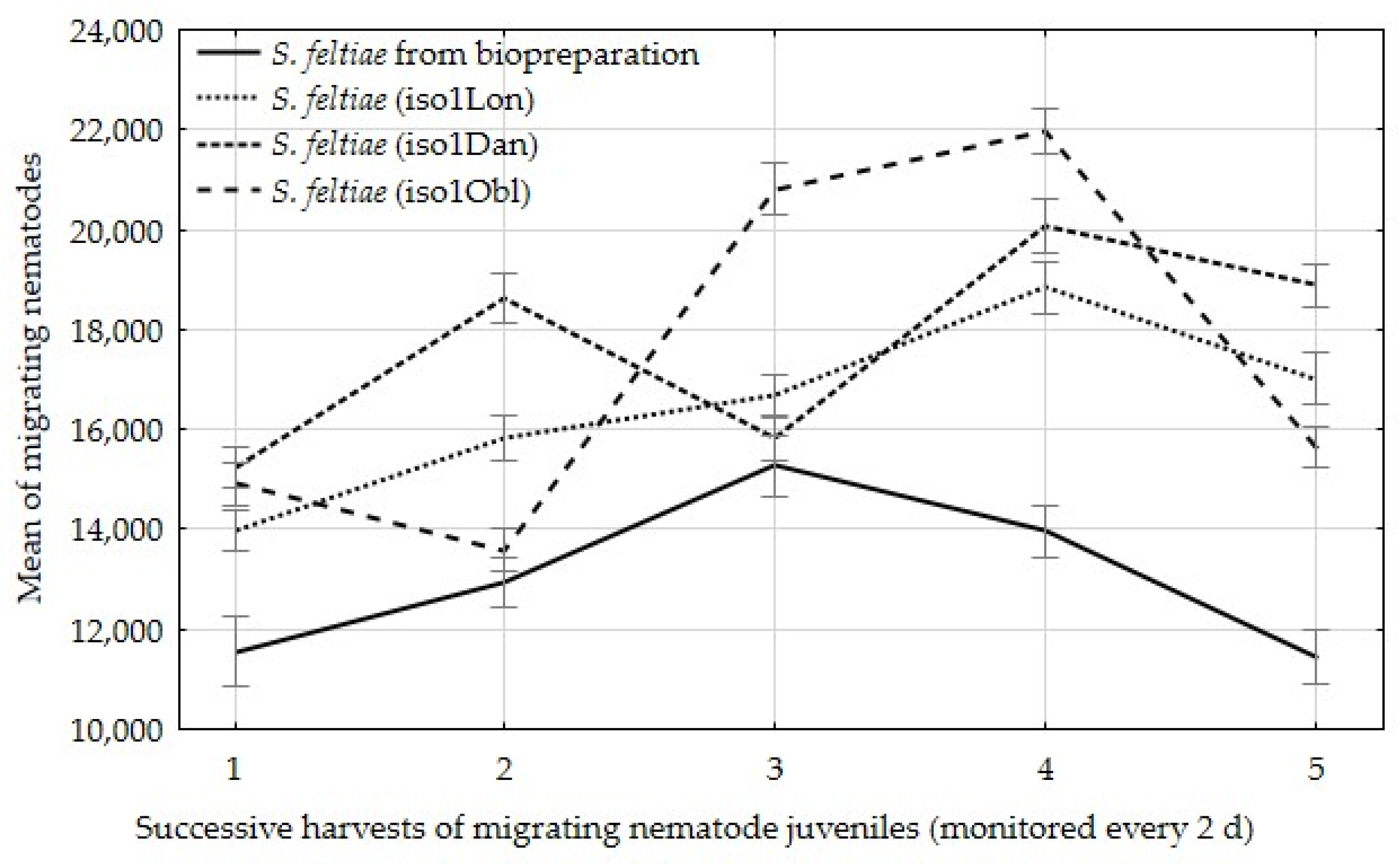
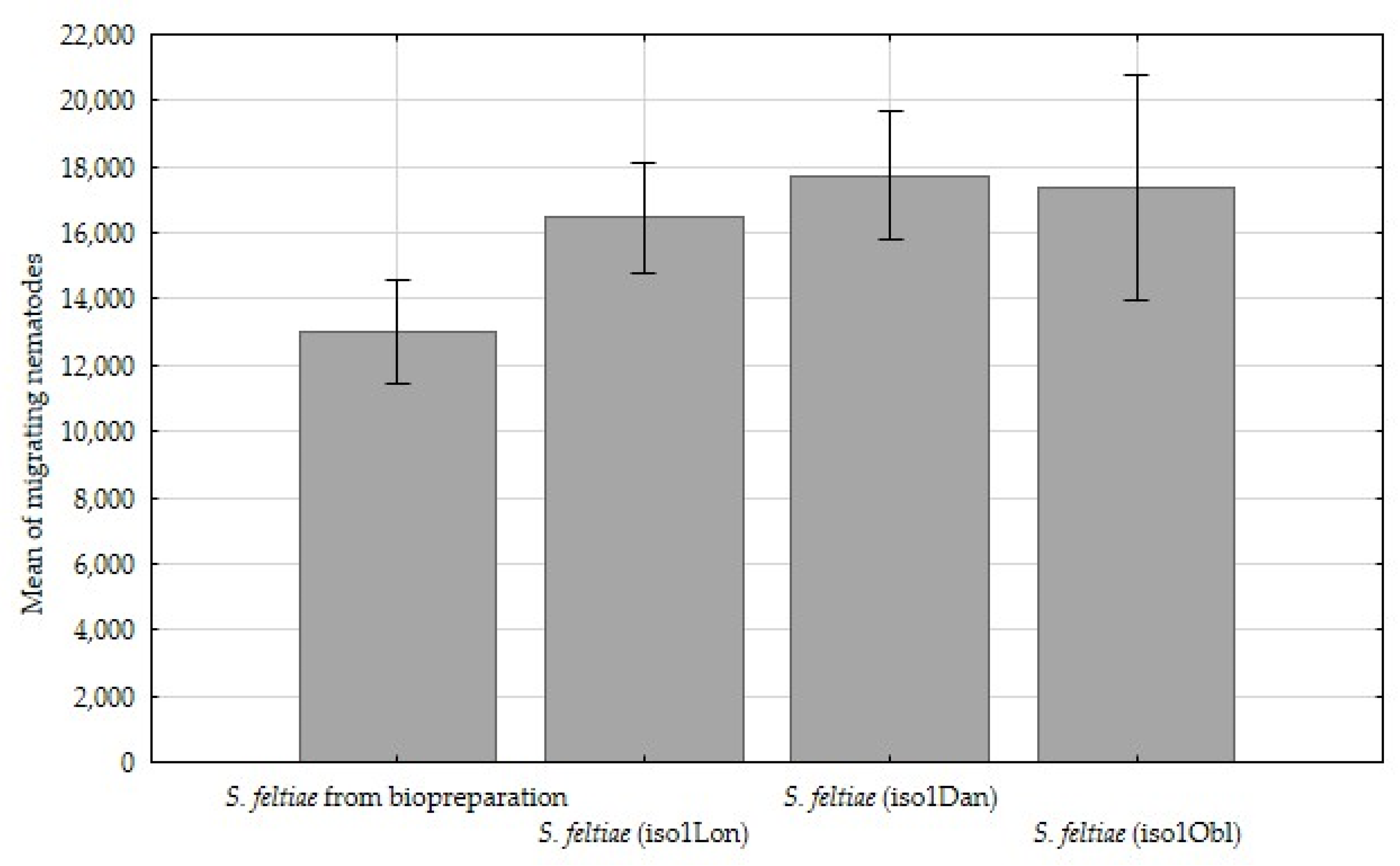
| Number of infective juveniles emerging per insect (no. of ind.) | 65,150 a | 82,320 b | 88,640 b | 86,910 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matuska-Łyżwa, J.; Żarnowiec, P.; Kaca, W. Comparison of Biological Activity of Field Isolates of Steinernema feltiae with a Commercial S. feltiae Biopesticide Product. Insects 2021, 12, 816. https://doi.org/10.3390/insects12090816
Matuska-Łyżwa J, Żarnowiec P, Kaca W. Comparison of Biological Activity of Field Isolates of Steinernema feltiae with a Commercial S. feltiae Biopesticide Product. Insects. 2021; 12(9):816. https://doi.org/10.3390/insects12090816
Chicago/Turabian StyleMatuska-Łyżwa, Joanna, Paulina Żarnowiec, and Wiesław Kaca. 2021. "Comparison of Biological Activity of Field Isolates of Steinernema feltiae with a Commercial S. feltiae Biopesticide Product" Insects 12, no. 9: 816. https://doi.org/10.3390/insects12090816
APA StyleMatuska-Łyżwa, J., Żarnowiec, P., & Kaca, W. (2021). Comparison of Biological Activity of Field Isolates of Steinernema feltiae with a Commercial S. feltiae Biopesticide Product. Insects, 12(9), 816. https://doi.org/10.3390/insects12090816






