Blinded by the Light: Artificial Light Lowers Mate Attraction Success in Female Glow-Worms (Lampyris noctiluca L.)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
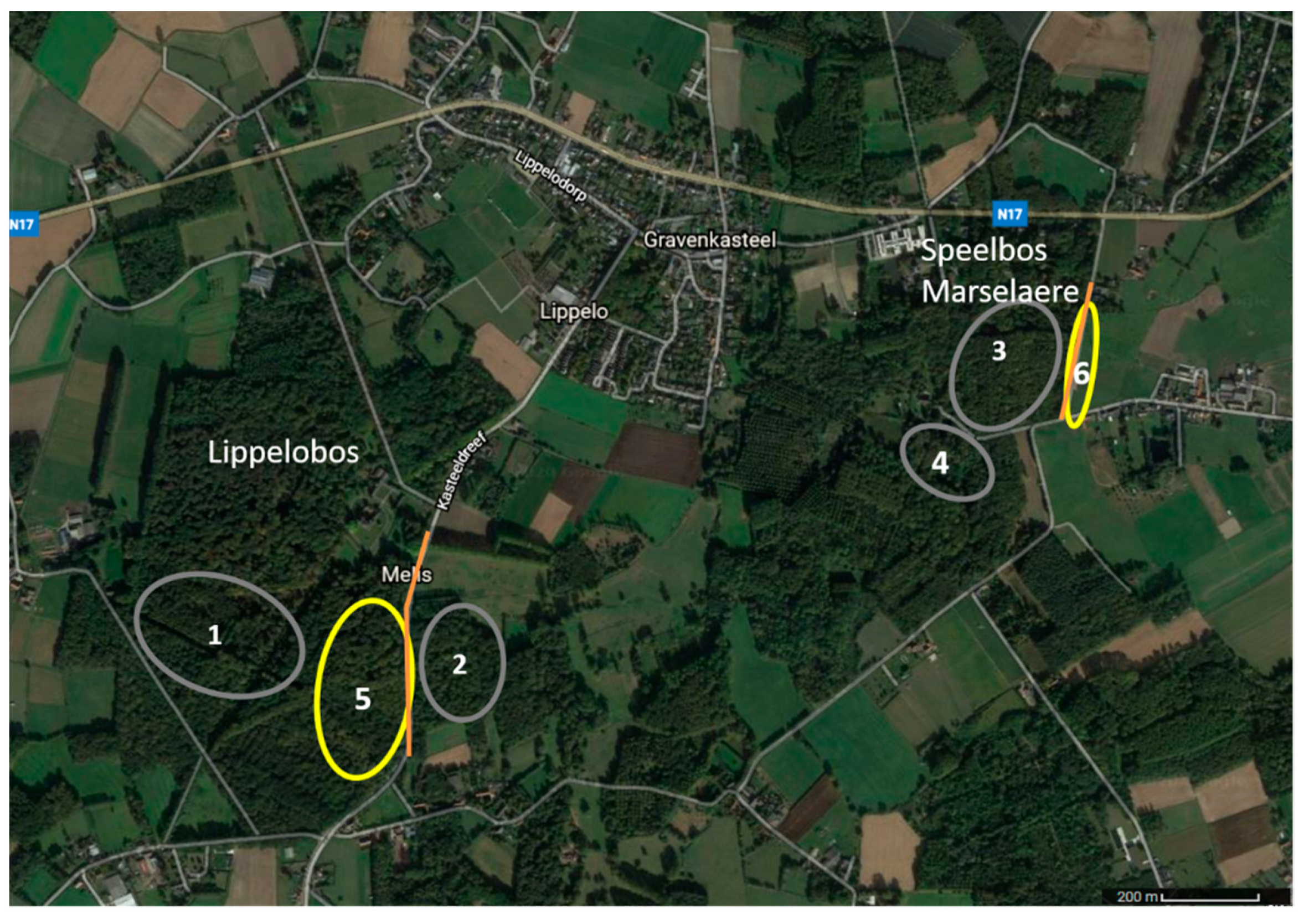
2.1. Study Area
2.2. Light Environments
2.3. Study Species
2.4. Monitoring
2.5. Statistical Analysis
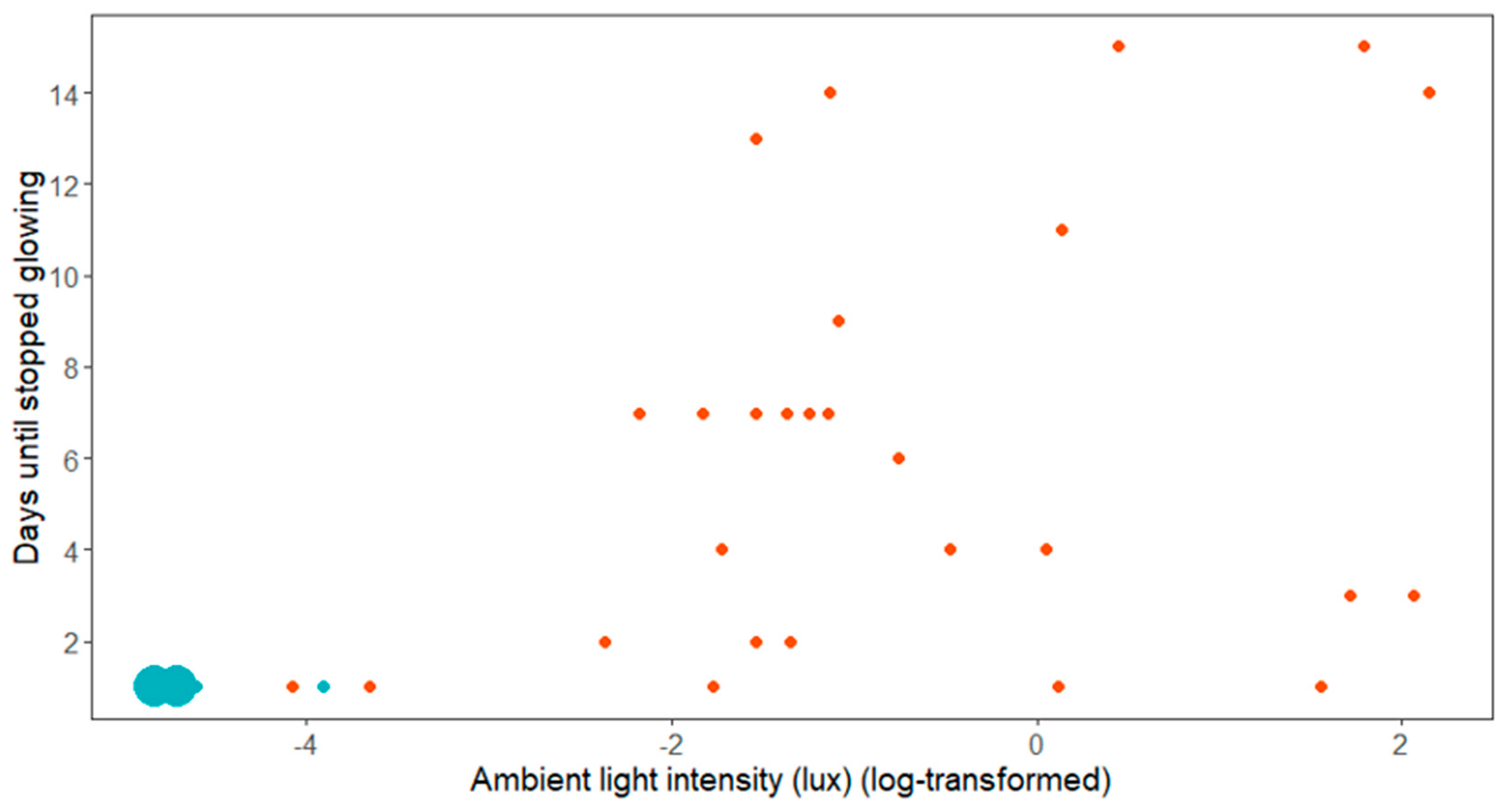
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

References
- Owens, A.; Cochard, P.; Durrant, J.; Farnworth, B.; Perkin, E.; Seymoure, B. Light pollution is a driver of insect declines. Biol. Conserv. 2020, 241, 108259. [Google Scholar] [CrossRef]
- Owens, A.; Lewis, S.M. The impact of artificial light at night on nocturnal insects: A review and synthesis. Ecol. Evol. 2018, 8, 11337–11358. [Google Scholar] [CrossRef] [Green Version]
- Davies, T.W.; Bennie, J.; Inger, R.; De Ibarra, N.H.; Gaston, K.J. Artificial light pollution: Are shifting spectral signatures changing the balance of species interactions? Glob. Chang. Biol. 2013, 19, 1417–1423. [Google Scholar] [CrossRef] [Green Version]
- Longcore, T.; Rich, C. Ecological light pollution. Front. Ecol. Environ. 2004, 2, 191–198. [Google Scholar] [CrossRef]
- Hölker, F.; Moss, T.; Griefahn, B.; Kloas, W.; Voigt, C.C.; Henckel, D.; Hänel, A.; Kappeler, P.M.; Völker, S.; Schwope, A. The dark side of light: A transdisciplinary research agenda for light pollution policy. Ecol. Soc. 2010, 15, 13. [Google Scholar] [CrossRef]
- Coulthard, E.; Norrey, J.; Shortall, C.; Harris, W.E. Ecological traits predict population changes in moths. Biol. Conserv. 2019, 233, 213–219. [Google Scholar] [CrossRef]
- Van Langevelde, F.; Braamburg-Annegarn, M.; Huigens, M.E.; Groendijk, R.; Poitevin, O.; van Deijk, J.R.; Ellis, W.N.; van Grunsven, R.H.; de Vos, R.; Vos, R.A. Declines in moth populations stress the need for conserving dark nights. Glob. Chang. Biol. 2018, 24, 925–932. [Google Scholar] [CrossRef]
- Giavi, S.; Fontaine, C.; Knop, E. Impact of artificial light at night on diurnal plant-pollinator interactions. Nat. Commun. 2021, 12, 1690. [Google Scholar] [CrossRef] [PubMed]
- Vaz, S.; Manes, S.; Gama-Maia, D.; Silveira, L.; Mattos, G.; Paiva, P.C.; Figueiredo, M.; Lorini, M.L. Light pollution is the fastest growing potential threat to firefly conservation in the Atlantic Forest hotspot. Insect Conserv. Divers. 2021, 14, 211–224. [Google Scholar] [CrossRef]
- Lewis, S.M.; Wong, C.H.; Owens, A.; Fallon, C.; Jepsen, S.; Thancharoen, A.; Wu, C.; De Cock, R.; Novák, M.; López-Palafox, T. A Global Perspective on Firefly Extinction Threats. BioScience 2020, 70, 157–167. [Google Scholar] [CrossRef] [Green Version]
- Yiu, V. Effects of artificial light on firefly flashing activity. Insect News 2012, 4, 5–9. [Google Scholar]
- Costin, K.J.; Boulton, A.M. A field experiment on the effect of introduced light pollution on fireflies (Coleoptera: Lampyridae) in the Piedmont Region of Maryland. Coleopt. Bull. 2016, 70, 84–86. [Google Scholar] [CrossRef]
- Firebaugh, A.; Haynes, K.J. Experimental tests of light-pollution impacts on nocturnal insect courtship and dispersal. Oecologia 2016, 182, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Hagen, O.; Santos, R.M.; Schlindwein, M.N.; Viviani, V.R. Artificial night lighting reduces firefly (Coleoptera: Lampyridae) occurrence in Sorocaba, Brazil. Adv. Entomol. 2015, 3, 24. [Google Scholar] [CrossRef] [Green Version]
- Mbugua, S.W.; Wong, C.H.; Ratnayeke, S. Effects of artificial light on the larvae of the firefly Lamprigera sp. in an urban city park, Peninsular Malaysia. J. Asia-Pac. Entomol. 2020, 23, 82–85. [Google Scholar] [CrossRef]
- Ineichen, S.; Rüttimann, B. Impact of artificial light on the distribution of the common European glow-worm, Lampyris noctiluca (Coleoptera: Lampyridae). Lampyrid 2012, 2, 31–36. [Google Scholar]
- Bird, S.; Parker, J. Low levels of light pollution may block the ability of male glow-worms (Lampyris noctiluca L.) to locate females. J. Insect Conserv. 2014, 18, 737–743. [Google Scholar] [CrossRef]
- Stewart, A.J.; Perl, C.D.; Niven, J.E. Artificial lighting impairs mate attraction in a nocturnal capital breeder. J. Exp. Biol. 2020, 223, jeb229146. [Google Scholar] [CrossRef]
- Elgert, C.; Hopkins, J.; Kaitala, A.; Candolin, U. Reproduction under light pollution: Maladaptive response to spatial variation in artificial light in a glow-worm. Proc. R. Soc. B 2020, 287, 20200806. [Google Scholar] [CrossRef]
- Van den Broeck, M.; De Cock, R.; Van Dongen, S.; Matthysen, E. White LED light intensity, but not colour temperature, interferes with mate-finding by glow-worm (Lampyris noctiluca L.) males. J. Insect Conserv. 2021, 25, 339–347. [Google Scholar] [CrossRef]
- Dreisig, H. The circadian rhythm of bioluminescence in the glowworm, Lampyris noctiluca L. (Coleoptera, Lampyridae). Behav. Ecol. Sociobiol. 1978, 3, 1–18. [Google Scholar] [CrossRef]
- Tyler, J. The Glow-Worm; Lakeside Printing Ltd.: Sevenoaks, UK, 2002. [Google Scholar]
- Gardiner, T.; Didham, R.K. Glow-worm abundance declines with increasing proximity to artificial lighting. Entomol. Gaz. 2021, 72, 1. [Google Scholar] [CrossRef]
- Hickmott, W.; Tyler, J. Seasonal variation in the female display period of the glow-worm Lampyris noctiluca L. (Coleoptera: Lampyridae). Lampyrid 2011, 1, 14–21. [Google Scholar]
- Lehtonen, T.K.; Babic, N.L.; Piepponen, T.; Valkeeniemi, O.; Borshagovski, A.-M.; Kaitala, A. High road mortality during female-biased larval dispersal in an iconic beetle. Behav. Ecol. Sociobiol. 2021, 75, 26. [Google Scholar] [CrossRef]
- Baudry, G.; Hopkins, J.; Watts, P.C.; Kaitala, A. Female Sexual Signaling in a Capital Breeder, the European Glow-Worm Lampyris noctiluca. J. Insect Behav. 2021, 34, 16–25. [Google Scholar] [CrossRef]
- Tyler, J. What do glow-worms do on their day off? The diurnal behaviour of adult Lampyris noctiluca (L.) (Coleoptera: Lampyridae). Lampyrid 2013, 3, 23–34. [Google Scholar]
- Dreisig, H. Environmental control of the daily onset of luminescent activity in glowworms and fireflies (Coleoptera: Lampyridae). Oecologia 1975, 18, 85–99. [Google Scholar] [CrossRef]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Therneau, T.M. A Package for Survival Analysis in S. Version 2.38. Available online: https://CRAN.R-project.org/package=survival (accessed on 15 April 2020).
- Kassambara, A.; Kosinski, M.; Przemyslaw, B. Survminer: Drawing Survival Curves Using ‘ggplot2’. R Package Version 0.4.6. 2019. Available online: https://CRAN.R-project.org/package=survminer (accessed on 15 April 2020).
- Hopkins, J.; Baudry, G.; Candolin, U.; Kaitala, A. I’m sexy and I glow it: Female ornamentation in a nocturnal capital breeder. Biol. Lett. 2015, 11, 20150599. [Google Scholar] [CrossRef] [Green Version]
- Borshagovski, A.-M.; Baudry, G.; Hopkins, J.; Kaitala, A. Pale by comparison: Competitive interactions between signaling female glow-worms. Behav. Ecol. 2019, 30, 20–26. [Google Scholar] [CrossRef]
- Tyler, J. A study of the male flight season in the glow-worm Lampyris noctiluca (L.) (Coleoptera: Lampyridae). Lampyrid 2011, 1, 32–38. [Google Scholar]
- Desouhant, E.; Gomes, E.; Mondy, N.; Amat, I. Mechanistic, ecological, and evolutionary consequences of artificial light at night for insects: Review and prospective. Entomol. Exp. Appl. 2019, 167, 37–58. [Google Scholar] [CrossRef] [Green Version]
- Schwalb, H.H. Beiträge zur Biologie der Einheimischen Lampyriden Lampyris noctiluca Geoffr. und Phausis splendidula Lec. und Experimentelle Analyse ihres Beuteumfang-und Sexualverhaltens. Ph.D. Thesis, Johannes Gutenberg-Universität Mainz, Mainz, Germany, 1960. [Google Scholar]
- Bek, R. Investigating the Impact of Artificial Night Lighting on the Common European Glow-Worm, Lampyris noctiluca (L.) (Coleoptera: Lampyridae). Bachelor’s Thesis, University of Leeds, Leeds, UK, 2015. [Google Scholar]
- Van den Broeck, M. Impact of Artificial Light on Mate-Seeking Behaviour of Glow-Worms (Lampyris noctiluca): An Experimental Study. Master’s Thesis, Universiteit Antwerpen, Antwerp, Belgium, 2020. [Google Scholar]
- Lall, A.B.; Worthy, K.M. Action spectra of the female’s response in the firefly Photinus pyralis (Coleoptera: Lampyridae): Evidence for an achromatic detection of the bioluminescent optical signal. J. Insect Physiol. 2000, 46, 965–968. [Google Scholar] [CrossRef]
- Eguchi, E.; Nemoto, A.; Meyer-Rochow, V.B.; Ohba, N. A comparative study of spectral sensitivity curves in three diurnal and eight nocturnal species of Japanese fireflies. J. Insect Physiol. 1984, 30, 607–612. [Google Scholar] [CrossRef]
- Lall, A.B.; Strother, G.; Cronin, T.W.; Seliger, H.H. Modification of spectral sensitivities by screening pigments in the compound eyes of twilight-active fireflies (Coleoptera: Lampyridae). J. Comp. Physiol. A 1988, 162, 23–33. [Google Scholar] [CrossRef]
- Martin, G.J.; Lord, N.P.; Branham, M.A.; Bybee, S.M. Review of the firefly visual system (Coleoptera: Lampyridae) and evolution of the opsin genes underlying color vision. Org. Divers. Evol. 2015, 15, 513–526. [Google Scholar] [CrossRef]
- Sander, S.E.; Hall, D.W. Variation in opsin genes correlates with signalling ecology in North American fireflies. Mol. Ecol. 2015, 24, 4679–4696. [Google Scholar] [CrossRef] [Green Version]
- Booth, D.; Stewart, A.J.; Osorio, D. Colour vision in the glow-worm Lampyris noctiluca (L.) (Coleoptera: Lampyridae): Evidence for a green-blue chromatic mechanism. J. Exp. Biol. 2004, 207, 2373–2378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wing, S.R. Energetic costs of mating in a flightless female firefly, Photinus collustrans (Coleoptera: Lampyridae). J. Insect Behav. 1989, 2, 841–847. [Google Scholar] [CrossRef]
- Horne, J.; Horne, A.; Tyler, J. The influence of female body weight on egg production in the glow-worm Lampyris noctiluca (Coleoptera: Lampyridae). Lampyrid 2017, 4, 36–39. [Google Scholar]
- Jönsson, K.I. Capital and income breeding as alternative tactics of resource use in reproduction. Oikos 1997, 78, 57–66. [Google Scholar] [CrossRef]
- Lloyd, J.E. Firefly parasites and predators. Coleopt. Bull. 1973, 27, 91–106. [Google Scholar]
- Day, J.C. Parasites, predators and defence of fireflies and glow-worms. Lampyrid 2011, 1, 70–102. [Google Scholar]
- Tyler, J.; Mckinnon, W.; Lord, G.A.; Hilton, P.J. A defensive steroidal pyrone in the Glow-worm Lampyris noctiluca L. (Coleoptera: Lampyridae). Physiol. Entomol. 2008, 33, 167–170. [Google Scholar] [CrossRef]
- De Cock, R.; Matthysen, E. Do glow-worm larvae (Coleoptera: Lampyridae) use warning coloration? Ethology 2001, 107, 1019–1033. [Google Scholar] [CrossRef]
- Pulliam, H.R. Sources, sinks, and population regulation. Am. Nat. 1988, 132, 652–661. [Google Scholar] [CrossRef]
- Elgert, C.; Lehtonen, T.K.; Kaitala, A.; Candolin, U. Sexual selection for bright females prevails under light pollution. Curr. Zool. 2020, 67, 329–331. [Google Scholar] [CrossRef]
- Atkins, V.; Bell, D.; Bowker, A.; Charig, M.; Crew, J.; Dale, M.; Hickmott, B.; Payne, B.; Pendleton, D.; Pendleton, T.; et al. The status of the glow-worm Lampyris noctiluca L. (Coleoptera: Lampyridae) in England. Lampyrid 2016, 4, 20–35. [Google Scholar]
- Rieger, I. Why are glowworm females sitting under a street light?-Habitat Use of Lampyris noctiluca-Facts and Open Questions. In Proceedings of the Firefly Meeting, Gaia, Portugal, 21–25 June 2007. [Google Scholar] [CrossRef]
- Lewis, S. Silent Sparks: The Wondrous World of Fireflies; Princeton University Press: Princeton, NJ, USA, 2016. [Google Scholar]
- Pimputkar, S.; Speck, J.S.; DenBaars, S.P.; Nakamura, S. Prospects for LED lighting. Nat. Photonics 2009, 3, 180–182. [Google Scholar] [CrossRef]
- Fallon, C.; Hoyle, S.; Lewis, S.; Owens, A.; Lee-Mäder, E.; Black, S.H.; Jepsen, S. Conserving the Jewels of the Night: Guidelines for Protecting Fireflies in the United States and Canada; The Xerces Society For Invertebrate Conservation: Portland, OR, USA, 2019. [Google Scholar]
- Owens, A.; Meyer-Rochow, V.B.; Yang, E.-C. Short-and mid-wavelength artificial light influences the flash signals of Aquatica ficta fireflies (Coleoptera: Lampyridae). PLoS ONE 2018, 13, e0191576. [Google Scholar] [CrossRef] [Green Version]
- Owens, A.C.S.; Lewis, S.M. Narrow-spectrum artificial light silences female fireflies Coleoptera: Lampyridae). Insect Conserv. Divers. 2021, 14, 199–210. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van den Broeck, M.; De Cock, R.; Van Dongen, S.; Matthysen, E. Blinded by the Light: Artificial Light Lowers Mate Attraction Success in Female Glow-Worms (Lampyris noctiluca L.). Insects 2021, 12, 734. https://doi.org/10.3390/insects12080734
Van den Broeck M, De Cock R, Van Dongen S, Matthysen E. Blinded by the Light: Artificial Light Lowers Mate Attraction Success in Female Glow-Worms (Lampyris noctiluca L.). Insects. 2021; 12(8):734. https://doi.org/10.3390/insects12080734
Chicago/Turabian StyleVan den Broeck, Mira, Raphaël De Cock, Stefan Van Dongen, and Erik Matthysen. 2021. "Blinded by the Light: Artificial Light Lowers Mate Attraction Success in Female Glow-Worms (Lampyris noctiluca L.)" Insects 12, no. 8: 734. https://doi.org/10.3390/insects12080734
APA StyleVan den Broeck, M., De Cock, R., Van Dongen, S., & Matthysen, E. (2021). Blinded by the Light: Artificial Light Lowers Mate Attraction Success in Female Glow-Worms (Lampyris noctiluca L.). Insects, 12(8), 734. https://doi.org/10.3390/insects12080734





