Combined Effects of Pesticides and Electromagnetic-Fields on Honeybees: Multi-Stress Exposure
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Sites
2.2. Population Parameters and Health Status of the Colonies
2.2.1. Comb Inspection and Analysis
2.2.2. Mortality in Underbaskets
2.2.3. Varroa Mite Monitoring
2.2.4. Virus Monitoring
2.2.5. American and European Foulbrood Monitoring
2.2.6. Biomarker Sampling and Analyses
2.3. Electric and Magnetic Field Measurements
2.4. Exposure to Pesticides
2.5. Meteorological Data
2.6. Statistical Analyses
3. Results
3.1. Exposure to Pesticides
3.2. Magnetic and Electric Field Measurements
3.3. Meteorological Conditions in the Experimental Sites
3.4. Health Status of the Colonies
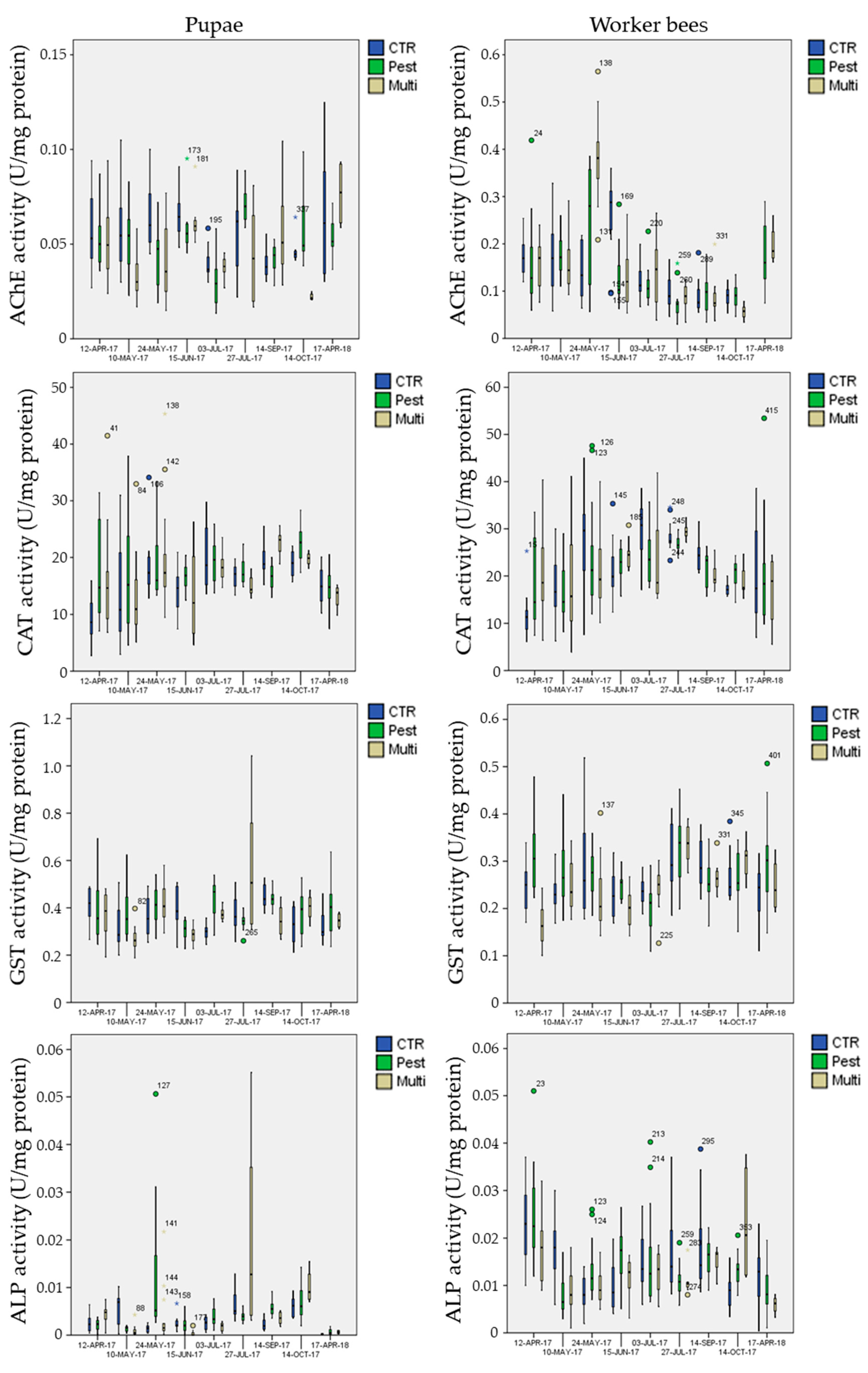
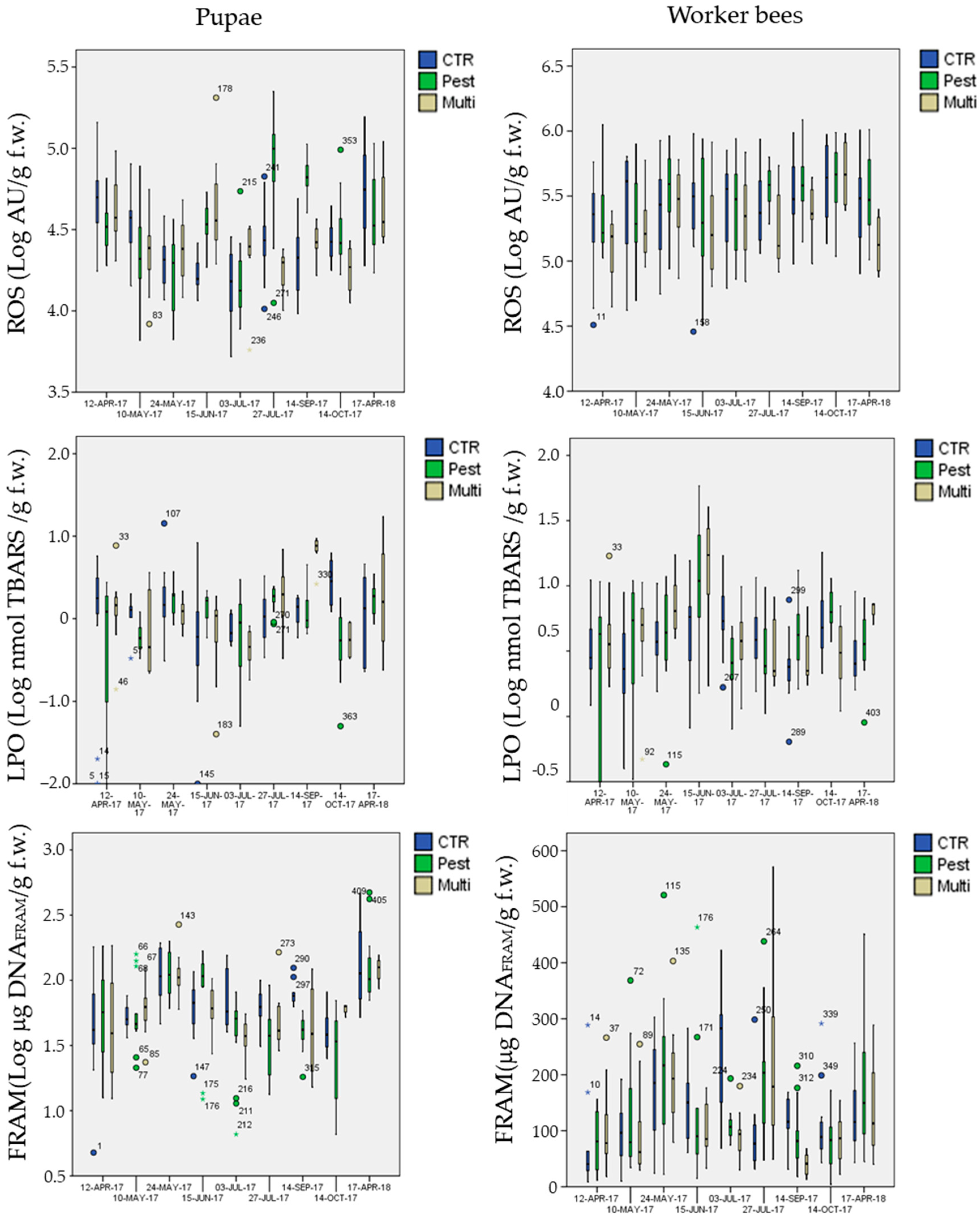
3.5. Biomarker Analysis
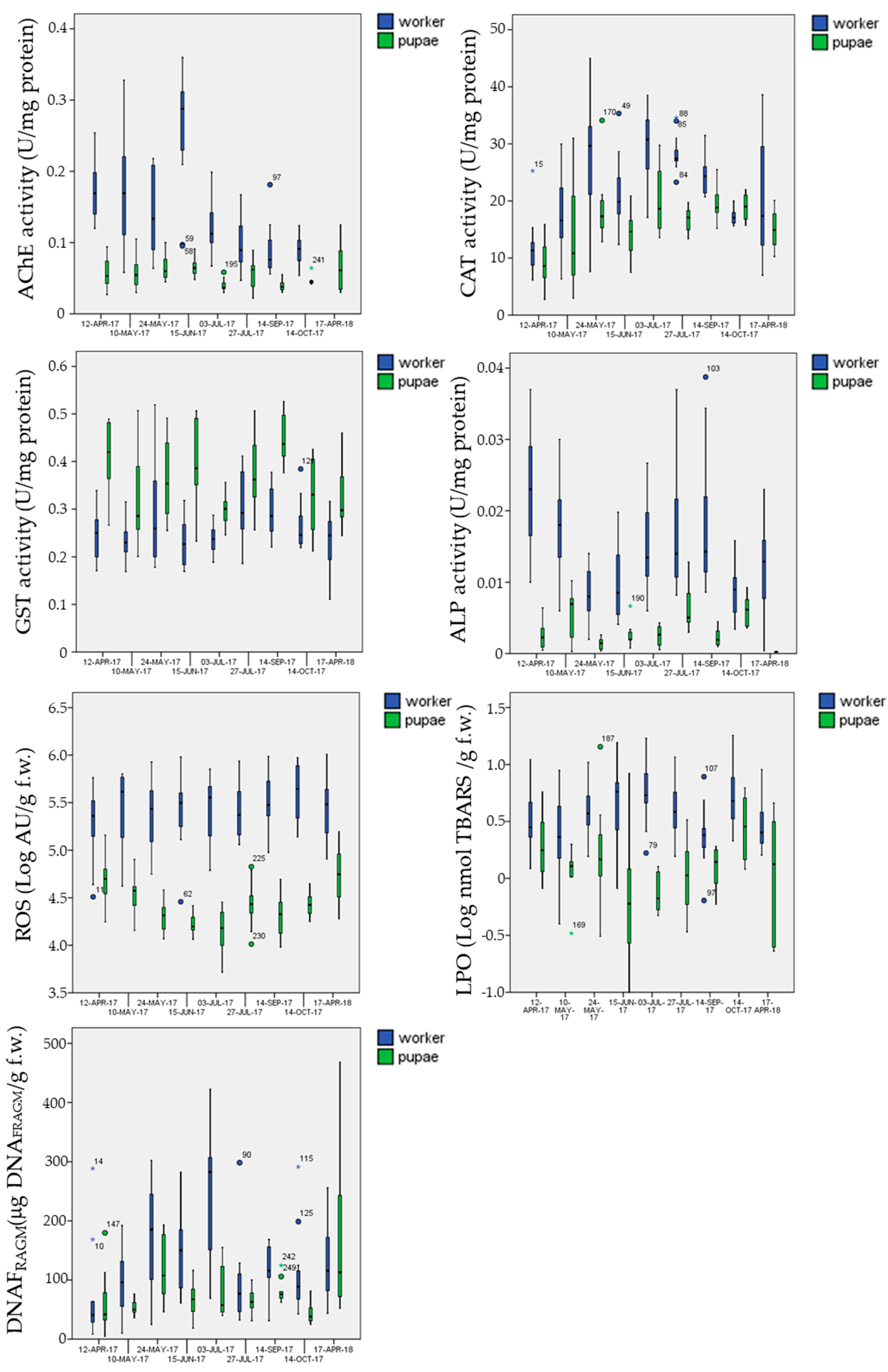
3.5.1. Biomarkers in the Control Site
3.5.2. Biomarkers in the Exposure Sites
- mean AChE activity in pupae was significantly inhibited (Post-hoc Tukey test p = 0.002 **) in the multi-stress site (marginal mean of 0.041 U mg−1 protein with 95% confidence Interval of 0.037–0.045) in comparison to the chemical-stress (marginal mean of 0.050 U mg−1 protein, CI = 0.046–0.053) and control sites (marginal mean of 0.051 U mg−1 protein CI = 0.048–0.055);
- mean CAT activity in pupae was significantly activated (Post-hoc Tukey test p < 0.023 *) in the chemical-stress site (marginal mean of 17.0 U mg−1 protein, CI = 16.0–18.0) in comparison to the control (marginal mean of 15.2 U mg−1 protein, CI = 14.2–16.3) and multi-stress sites (marginal mean of 15.6 U mg−1 protein CI = 14.3–17.0);
- mean GST activity in pupae was significantly activated (Post-hoc Tukey test p < 0.030 *) in the chemical-stress site (marginal mean of 0.38 U mg−1 protein, CI = 0.36–0.39) in comparison to the control (marginal mean of 0.36 U mg−1 protein, CI = 0.34–0.37) and multi-stress sites (marginal mean of 0.36 U mg−1 protein CI = 0.34–0.38), while in worker bees mean GST activity was higher in the chemical-stress site (marginal mean of 0.27 U mg−1 protein, CI = 0.26–0.28) in comparison to the control (marginal mean of 0.25 U mg−1 protein, CI = 0.24–0.26) and multi-stress sites (marginal mean of 0.24 U mg−1 protein CI = 0.23–0.25), but differences were only significant with the multi-stress site (Post-hoc Tukey test p = 0.003 **);
- mean ROS levels in pupae were significantly higher (Post-hoc Tukey test p < 0.003 **) in the chemical-stress site (marginal mean of 3.3 × 104 AU g−1 f.w., CI = 3.0 × 104−3.5 × 104) in comparison to the control (marginal mean of 2.6 × 104 AU g−1 f.w., CI = 2.4 × 104−2.9 × 104) and multi-stress sites (marginal mean of 2.6 × 104 AU g−1 f.w., CI = 2.3 × 104−3.0 × 104), in worker bees ROS levels were higher in the chemical-stress site (marginal mean of 3.0 × 105 AU g−1 f.w., CI = 2.6 × 105−3.4 × 105) in comparison to the control (marginal mean of 2.7 × 105 AU g−1 f.w., CI = 2.4 × 105−3.1×105), but differences were not significant (Post-hoc Tukey test p = 0.39); the multi-stress site showed a mean lower level of ROS (marginal mean of 2.0×105 AU g−1 f.w., CI = 1.7 × 105−2.4 × 105) in comparison to the control and chemical stress sites (Post-hoc Tukey test p < 0.021 *);
- mean DNAFRAGM levels in pupae were significantly higher (Post-hoc Tukey test p < 0.006 **) in the control site (marginal mean of 66 μg DNAFRAM g−1 f.w., CI = 59−74) in comparison to the chemical-stress site (marginal mean of 53 μg DNAFRAM g−1 f.w., CI = 48–59); the multi-stress site was inbetween (marginal mean of 59 μg DNAFRAM g−1 f.w., CI = 50−69).
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Naug, D. Nutritional stress due to habitat loss may explain recent honeybee colony collapses. Biol. Conserv. 2009, 142, 2369–2372. [Google Scholar] [CrossRef]
- Huang, Z. Pollen nutrition affects honey bee stress resistance. Terr. Arthropod Rev. 2012, 5, 175–189. [Google Scholar] [CrossRef] [Green Version]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Alaux, C. Influence of pollen nutrition on honey bee health: Do pollen quality and diversity matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Conte, Y.; Navajas, M. Climate change: Impact on honey bee populations and diseases. Rev. Sci. Tech. 2008, 27, 499–510. [Google Scholar]
- Simon-Delso, N.; San Martin, G.; Bruneau, E.; Minsart, L.A.; Mouret, C.; Hautier, L. Honeybee colony disorder in crop areas: The role of pesticides and viruses. PLoS ONE 2014, 9, e103073. [Google Scholar] [CrossRef] [PubMed]
- Le Conte, Y.; Ellis, M.; Ritter, W. Varroa mites and honey bee health: Can Varroa explain part of the colony losses? Apidologie 2010, 41, 353–363. [Google Scholar] [CrossRef] [Green Version]
- Van Der Zee, R.; Gray, A.; Pisa, L.; De Rijk, T. An observational study of honey bee colony winter losses and their association with Varroa destructor, neonicotinoids and other risk factors. PLoS ONE 2015, 10, e0131611. [Google Scholar] [CrossRef] [Green Version]
- Vaudo, A.D.; Tooker, J.F.; Grozinger, C.M.; Patch, H.M. Bee nutrition and floral resource restoration. Curr. Opin. Insect Sci. 2015, 10, 133–141. [Google Scholar] [CrossRef] [Green Version]
- Goulson, D. An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 2013, 50, 977–987. [Google Scholar] [CrossRef]
- Balmori, A. Electromagnetic radiation as an emerging driver factor for the decline of insects. Sci. Total Environ. 2021, 767, 144913. [Google Scholar] [CrossRef]
- Lupi, D.; Tremolada, P.; Colombo, M.; Giacchini, R.; Benocci, R.; Parenti, P.; Vighi, M. Effects of pesticides and electromagnetic fields on honeybees: A field study using biomarkers. Int. J. Environ. Res. 2020, 14, 107–122. [Google Scholar] [CrossRef]
- Carreck, N. Electromagnetic radiation and bees, again. Bee World 2014, 91, 101–102. [Google Scholar] [CrossRef]
- González-Varo, J.P.; Biesmeijer, J.C.; Bommarco, R.; Potts, S.G.; Schweiger, O.; Smith, H.G.; Vilà, M. Combined effects of global change pressures on animal-mediated pollination. Trends Ecol. Evol. 2013, 28, 524–530. [Google Scholar] [CrossRef] [Green Version]
- Van Engelsdorp, D.; Underwood, R.; Caron, D.; Hayes, J. Estimate of managed colony losses in the winter of 2006–2007: A report commissioned by the apiary inspectors of America. Am. Bee J. 2007, 147, 599–603. [Google Scholar]
- Van Engelsdorp, D.; Evans, J.D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Cox-Foster, D.; Chen, Y.; Underwood, R.; et al. Colony collapse disorder: A descriptive study. PLoS ONE 2009, 4, e6481. [Google Scholar]
- Staveley, J.P.; Law, S.A.; Fairbrother, A.; Menzie, C.A. A causal analysis of observed declines in managed honey bees (Apis mellifera). Hum. Ecol. Risk Assess. 2014, 20, 566–591. [Google Scholar] [CrossRef] [Green Version]
- Dainat, B.; Vanengelsdorp, D.; Neumann, P. Colony collapse disorder in Europe. Environ. Microbiol. Rep. 2012, 4, 123–125. [Google Scholar] [CrossRef]
- Bolton, R.D.; Iwasa, Y.; Marshall, J.A.R.; Childs, D.Z. Stress-mediated Allee effects can cause the sudden collapse of honey bee colonies. J. Theor. Biol. 2017. 420, 213–219.
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Nazzi, F.; Brown, S.P.; Annoscia, D.; Del Piccolo, F.; Di Prisco, G.; Varricchio, P.; Della Vedova, G.; Cattonaro, F.; Caprio, E.; Pennacchio, F. Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honeybee colonies. PLoS Pathog. 2012, 8, e1002735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef] [PubMed]
- Porrini, C.; Mutinelli, F.; Bortolotti, L.; Granato, A.; Laurenson, L.; Roberts, K.; Gallina, A.; Silvester, N.; Medrzycki, P.; Renzi, T.; et al. The status of honey bee health in Italy: Results from the nationwide bee monitoring network. PLoS ONE 2016, 11, e0155411. [Google Scholar]
- Watson, K.; Stallins, J.A. Honey bees and colony collapse disorder: A pluralistic reframing. Geogr. Compass 2016, 10, 222–236. [Google Scholar] [CrossRef]
- Perry, C.; Søvik, E.; Myerscough, M.R.; Barron, A.B. Rapid behavioral maturation accelerates failure of stressed honey bee colonies. Proc. Natl. Acad. Sci. USA 2015, 112, 3427–3432. [Google Scholar] [CrossRef] [Green Version]
- Gregorc, A.; Evans, J.D.; Scharf, M.; Ellis, J.D. Gene expression in honey bee (Apis mellifera) larvae exposed to pesticides and Varroa mites (Varroa destructor). J. Insect Physiol. 2012, 58, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.W.; Woodring, J. Effects of stress, age, season, and source colony on levels of octopamine, dopamine and serotonin in the honey bee (Apis mellifera L.) brain. J. Insect Physiol. 1992, 38, 29–35. [Google Scholar] [CrossRef]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [Green Version]
- Maini, S.; Medrzycki, P.; Porrini, C. The puzzle of honey bee losses: A brief review. Bull. Insectol. 2010, 63, 153–160. [Google Scholar]
- Pettis, J.S.; Johnson, J.; Dively, G. Pesticide exposure in honey bees results in increased levels of the gut pathogen Nosema. Naturwissenschaften 2012, 99, 153–158. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Bayo, F.; Goulson, D.; Pennacchio, F.; Nazzi, F.; Goka, K.; Desneux, N. Are bee diseases linked to pesticides?—A brief review. Environ. Intern. 2016, 89, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Boff, S.; Scheiner, R.; Raizer, J.; Lupi, D. Survival rate and changes in foraging performances of solitary bees exposed to a novel insecticide. Ecotoxicol. Environ. Saf. 2021, 211, 111869. [Google Scholar] [CrossRef]
- Sandrock, C.; Tanadini, M.; Tanadini, L.G.; Fauser-Misslin, A.; Potts, S.G.; Neumann, P. Impact of chronic neonicotinoid exposure on honeybee colony performance and queen supersedure. PLoS ONE 2014, 9, e103592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tosi, S.; Nieh, J.C. Lethal and sublethal synergistic effects of a new systemic pesticide, flupyradifurone (Sivanto®), on honeybees. Proc. R. Soc. B Biol. Sci. 2019, 286, 20190433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paxton, R.J. Does infection by Nosema ceranae cause “Colony Collapse Disorder” in honey bees (Apis mellifera)? J. Apic. Res. 2010, 49, 80–84. [Google Scholar] [CrossRef]
- Yang, E.C.; Chuang, Y.C.; Chen, Y.L.; Chang, L.H. Abnormal foraging behavior induced by sublethal dosage of imidacloprid in the honey bee (Hymenoptera: Apidae). J. Econ. Entomol. 2008, 101, 1743–1748. [Google Scholar] [CrossRef]
- Medrzycki, P.; Montanari, R.; Bortolotti, L.; Sabatini, A.G.; Maini, S.; Porrini, C. Effects of imidacloprid administered in sub-lethal doses on honey bee behaviour. Laboratory tests. Bull. Insectol. 2003, 56, 59–62. [Google Scholar]
- Cresswell, J.E. A meta-analysis of experiments testing the effects of a neonicotinoid insecticide (imidacloprid) on honey bees. Ecotoxicology 2011, 20, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Collison, E.; Hird, H.; Cresswell, J.; Tyler, C. Interactive effects of pesticide exposure and pathogen infection on bee health—A critical analysis. Biol. Rev. 2016, 91, 1006–1019. [Google Scholar] [CrossRef]
- Gill, R.J.; Ramos-Rodriguez, O.; Raine, N.E. Combined pesticide exposure severely affects individual-and colony-level traits in bees. Nature 2012, 491, 105–108. [Google Scholar] [CrossRef] [Green Version]
- Chensheng, L.U.; Warchol, K.M.; Callahan, R.A. In situ replication of honey bee colony collapse disorder. Bull. Insectol. 2012, 65, 99–106. [Google Scholar]
- Wegener, J.; Ruhnke, H.; Milchreit, K.; Kleebaum, K.; Franke, M.; Mispagel, S.; Bienefeld, K. Secondary biomarkers of insecticide-induced stress of honey bee colonies and their relevance for overwintering strength. Ecotox. Environ. Saf. 2016, 132, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, B.; Bindokas, V.P.; Gauger, J.R. Biological effects of a 765-kV transmission line: Exposures and thresholds in honeybee colonies. Eur. Bioelectromagn. Assoc. 1981, 2, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Todorović, D.; Mirćić, D.; Ilijin, L.; Mrdaković, M.; Vlahović, M.; Prolić, Z.; Mataruga, V.P. Effect of magnetic fields on antioxidative defense and fitness-related traits of Baculum extradentatum (insecta, phasmatodea). Bioelectromagnetics 2012, 33, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Gregorc, A.; Alburaki, M.; Werle, C.; Knight, P.R.; Adamczyk, J. Brood removal or queen caging combined with oxalic acid treatment to control varroa mites (Varroa destructor) in honey bee colonies (Apis mellifera). Apidologie 2017, 48, 821–832. [Google Scholar] [CrossRef] [Green Version]
- Imdorf, A.; Buehlmann, G.; Gerig, L.; Kilchenmann, V.; Wille, H. Überprüfung der Schätz Methode zur Ermittlung der Brutfläche und der Anzahl Arbeiterinnen in freifliegenden Bienenvölkern. Apidologie 1987, 18, 137–146. [Google Scholar] [CrossRef]
- Accorti, M.; Luti, F.; Tarducci, F. Methods for collecting data on natural mortality in bee. Ethol. Ecol. Evol. 1991, 1, 123–126. [Google Scholar] [CrossRef]
- Baratto, C.; Boniotti, B.; Ariani, P.; Papetti, A.; Dall’Olio, R.; Gagliazzo, L.; Mutinelli, F.; Granato, A. Development a specific one step real time RT-PCR for detection and quantification of deformed wing virus and acute bee paralysis virus in honey bees. In Proceedings of the Apimondia XXXXIII International Apicultural Congress, Kyiv, Ukraine, 29 September–4 October 2013. [Google Scholar]
- Bassi, S.; Galletti, G. Detection of Paenibacillus larvae from beehive debris: A culture method based on watery extraction of spores. In Proceedings of the seventh European conference of apidology, Cluj Napoca, Romania, 7–9 September 2016. [Google Scholar]
- Berra, E.; Forcella, M.; Giacchini, R.; Marziali, L.; Rossaro, B.; Parenti, P. Evaluation of enzyme biomarkers in freshwater invertebrates from Taro and Ticino river, Italy. Int. J. Limnol. 2004, 40, 169–180. [Google Scholar] [CrossRef] [Green Version]
- Bergmeyer, H.U. (Ed.) Methods of Enzymatic Analysis; Academic Press: Cambridge, MA, USA, 1983; Volume 2, pp. 165–166. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. The fist enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Munson, L.; Fall, R.R. Purification and characterisation of Escherichia coli alkaline phosphatase. A biochemical experiment. Biochem. Educ. 1978, 6, 53–56. [Google Scholar] [CrossRef]
- Deng, J.; Yu, L.; Liu, C.; Yu, K.; Shi, X.; Yeung, L.W.; Lam, P.K.; Wu, R.S.; Zhou, B. Hexabromocyclododecane-induced developmental toxicity and apoptosis in zebrafish embryos. Aquat. Toxicol. 2009, 93, 29–36. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohisi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Olive, P.L. DNA precipitation assay: A rapid and simple method for detecting DNA damage in mammalian cells. Environ. Mol. Mutagen. 1988, 11, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Bowen-Walker, P.L.; Martin, S.J.; Gunn, A. The transmission of deformed wing virus between honeybees (Apis mellifera L.) by the ectoparasitic mite Varroa jacobsoni Oud. J. Invertebr. Pathol. 1999, 73, 101–106. [Google Scholar] [CrossRef] [Green Version]
- Yue, C.; Genersch, E. RT-PCR analysis of Deformed wing virus in honeybees (Apis mellifera) and mites (Varroa destructor). J. Gen. Virol. 2005, 86, 3419–3424. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Evans, J.; Feldlaufer, M. Horizontal and vertical transmission of viruses in the honey bee, Apis mellifera. J. Invertebr. Pathol. 2006, 92, 152–159. [Google Scholar] [CrossRef]
- de Miranda, J.R.; Cordoni, G.; Budge, G. The acute bee paralysis virus–Kashmir bee virus–Israeli acute paralysis virus complex. J. Invertebr. Pathol. 2010, 103, S30–S47. [Google Scholar] [CrossRef] [PubMed]
- Carpana, E.; Vecchi, M.A.; Lavazza, A.; Bassi, S.; Dottori, M. Prevalence of acute paralysis virus (APV) and other viral infections in honeybees in Italy. In Recent Research on Bee Pathology, Proceedings of the International Symposium of the International Federation of Beekeepers Associations, Gent, Belgium, 5–7 September 1990; Ritter, W., Ed.; Tierhygienisches Inst.: Freiburg, Germany, 1991. [Google Scholar]
- Lanzi, G.; de Miranda, J.R.; Boniotti, M.B.; Cameron, C.E.; Lavazza, A.; Capucci, L.; Camazine, S.M.; Rossi, C. Molecular and biological characterization of deformed wing virus of honeybees (Apis mellifera L.). J. Virol. 2006, 80, 4998–5009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinello, M.; Baratto, C.; Manzinello, C.; Piva, E.; Borin, A.; Toson, M.; Granato, A.; Boniotti, M.B.; Gallina, A.; Mutinelli, F. Spring mortality in honey bees in northeastern Italy: Detection of pesticides and viruses in dead honey bees and other matrices. J. Apic. Res. 2017, 56, 239–254. [Google Scholar] [CrossRef]
- Floris, I.; Carpana, E.; Bassi, S.; Formato, G.; Ceresini, A.; Lodesani, M. Malattie batteriche. In Patologia e Avversità Dell’alveare; Emanuele, C., Marco, L., Eds.; Springer: Milano, Italy, 2014; pp. 49–121. [Google Scholar]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Chapter 3.2.3. European Foulbrood of Honey Bees (Infection of Honey Bees with Melissococcus Plutonius). 2021. Available online: https://www.oie.int/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (accessed on 28 January 2021).
- Yang, X.; Cox-Foster, D. Effects of parasitization by Varroa destructor on survivorship and physiological traits of Apis mellifera in correlation with viral incidence and microbial challenge. Parasitology 2007, 134, 405. [Google Scholar] [CrossRef]
- López, J.; Krainer, S.; Engert, A.; Schuehly, W.; Riessberger-Gallé, U.; Crailsheim, K. Sublethal pesticide doses negatively affect survival and the cellular responses in American foulbrood-infected honeybee larvae. Sci. Rep. 2017, 7, 40853. [Google Scholar] [CrossRef]
- Di Noi, A.; Casini, S.; Campani, T.; Cai, G.; Caliani, I. Review on sublethal effects of environmental contaminants in honey bees (Apis mellifera), knowledge gaps and future perspectives. Int. J. Environ. Res. Public Health 2021, 18, 1863. [Google Scholar] [CrossRef]
- Carvalho, S.M.; Belzunces, L.P.; Carvalho, G.A.; Brunet, J.L.; Badiou-Beneteau, A. Enzymatic biomarkers as tools to assess environmental quality: A case study of exposure of the honeybee Apis mellifera to insecticides. Environ. Toxicol. Chem. 2013, 32, 2117–2124. [Google Scholar] [CrossRef]
- Chakrabarti, P.; Rana, S.; Sarkar, S.; Smith, B.; Basu, P. Pesticide-induced oxidative stress in laboratory and field populations of native honey bees along intensive agricultural landscapes in two Eastern Indian states. Apidologie 2015, 46, 107–129. [Google Scholar] [CrossRef] [Green Version]
- El-Saad, A.M.A.; Kheirallah, D.A.; El-Samad, L.M. Biochemical and histological biomarkers in the midgut of Apis mellifera from polluted environment at Beheira Governorate, Egypt. Environ. Sci. Pollut. Res. 2017, 24, 3181–3193. [Google Scholar] [CrossRef] [PubMed]
- Caliani, I.; Campani, T.; Conti, B.; Cosci, F.; Bedini, S.; D’Agostino, A.; Giovanetti, L.; Di Noi, A.; Casini, S. First application of an Integrated Biological Response index to assess the ecotoxicological status of honeybees from rural and urban areas. Environ. Sci. Pollut. Res. 2021, 1–11. [Google Scholar] [CrossRef]
- Caliani, I.; Campani, T.; Conti, B.; Cosci, F.; Bedini, S.; D’Agostino, A.; Ammendola, A.; Di Noi, A.; Gori, A.; Casini, S. Multi-biomarker approach and IBR index to evaluate the effects of different contaminants on the ecotoxicological status of Apis mellifera. Ecotox. Environ. Saf. 2021, 208, 111486. [Google Scholar] [CrossRef]
- Badiou-Bénéteau, A.; Carvalho, S.M.; Brunet, J.L.; Carvalho, G.A.; Buleté, A.; Giroud, B.; Belzunces, L.P. Development of biomarkers of exposure to xenobiotics in the honey bee Apis mellifera: Application to the systemic insecticide thiamethoxam. Ecotoxicol. Environ. Saf. 2012, 82, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Boily, M.; Sarrasin, B.; DeBlois, C.; Aras, P.; Chagnon, M. Acetylcholinesterase in honey bees (Apis mellifera) exposed to neonicotinoids, atrazine and glyphosate: Laboratory and field experiments. Environ. Sci. Pollut. Res. 2013, 20, 5603–5614. [Google Scholar] [CrossRef]
- Darney, K.; Giraudin, A.; Joseph, R.; Abadie, P.; Aupinel, P.; Decourtye, A.; Le Bourg, E.; Gauthier, M. Effect of high-frequency radiations on survival of the honeybee (Apis mellifera L.). Apidologie 2016, 47, 703–710. [Google Scholar] [CrossRef] [Green Version]
- Somero, G.N.; Hochachka, P.W. Biochemical Adaptation; Princeton University Press: New York, NY, USA, 1984. [Google Scholar]
- Pfeifer, S.; Schiedek, D.; Dippner, J.W. Effect of temperature and salinity on acetylcholinesterase activity, a common pollution biomarker, in Mytilus sp. from the south-western Baltic Sea. J. Exp. Mar. Biol. Ecol. 2005, 320, 93–103. [Google Scholar] [CrossRef]
- Ippolito, A.; Giacchini, R.; Parenti, P.; Vighi, M. Natural variability of enzymatic biomarkers in freshwater invertebrates. Environ. Sci. Pollut. Res. 2017, 24, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Belzunces, L.P.; Gauthier, M.; Colin, M.E. Acetylcholinesterase in Apis mellifera head during post-embryonic development. Existence of a glycoinositol-anchored membrane form at eary pupal stages. Comp. Biochem. Physiol. Part B Comp. Biochem. 1992, 103, 57–63. [Google Scholar] [CrossRef]
- Jimenez, D.R.; Gilliam, M. Peroxisomal enzymes in the honey bee midgut. Arch. Insect Biochem. Physiol. 1996, 31, 87–103. [Google Scholar] [CrossRef]
- Polyzou, A.; Debras, J.F.; Belzunces, L.P. Changes in acetylcholinesterase during pupal development of Apis mellifera queen. Arch. Insect Biochem. Physiol. 1997, 36, 69–84. [Google Scholar] [CrossRef]
- Smirle, M.J.; Winston, M.L. Detoxifying enzyme activity in worker honey bees: An adaptation for foraging in contaminated ecosystems. Can. J. Zool. 1988, 66, 1938–1942. [Google Scholar] [CrossRef]
- Fine, J.D.; Mullin, C.A. Metabolism of N-Methyl-2-Pyrrolidone in Honey Bee Adults and Larvae: Exploring Age Related Differences in Toxic Effects. Environ. Sci. Technol. 2017, 51, 11412–11422. [Google Scholar] [CrossRef]
- Zhu, W.; Schmehl, D.R.; Mullin, C.A.; Frazier, J.L. Four common pesticides, their mixtures and a formulation solvent in the hive environment have high oral toxicity to honey bee larvae. PLoS ONE 2014, 9, e77547. [Google Scholar] [CrossRef] [PubMed]
- REACH. Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Com-Mission Regulation (EC) No 1488/94 as Well as Council Directive 76/769/EEC and Com-Mission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. Updated Version 15/02/2021. 2021. Available online: http://data.europa.eu/eli/reg/2006/1907/oj (accessed on 5 March 2021).
- Sanil, D.; Shetty, V.; Shetty, N.J. Differential expression of glutathione s-transferase enzyme in different life stages of various insecticide-resistant strains of Anopheles stephensi: A malaria vector. J. Vector Borne Dis. 2014, 51, 97–105. [Google Scholar]
- Strode, C.; Steen, K.; Ortelli, F.; Ranson, H. Differential expression of the detoxification genes in the different life stages of the malaria vector Anopheles Gambiae. Insect Mol. Biol. 2006, 15, 523–530. [Google Scholar] [CrossRef]
- Liao, C.Y.; Zhang, K.; Niu, J.Z.; Ding, T.B.; Zhong, R.; Xia, W.K.; Dou, W.; Wang, J.-J. Identification and Characterization of Seven Glutathione S-Transferase Genes from Citrus Red Mite, Panonychus citri (McGregor). Int. J. Mol. Sci. 2013, 14, 24255–24270. [Google Scholar] [CrossRef] [Green Version]
- Williams, J.B.; Roberts, S.P.; Elekonich, M.M. Age and natural metabolically-intensive behavior affect oxidative stress and antioxidant mechanisms. Exp. Gerontol. 2008, 43, 538–549. [Google Scholar] [CrossRef]
- EFSA. Guidance Document on the risk assessment of plant protection products on bees (Apis mellifera, Bombus spp. and solitary bees). EFSA J. 2013, 11, 3295. [Google Scholar]
- Poquet, Y.; Bodin, L.; Tchamitchian, M.; Fusellier, M.; Giroud, B.; Lafay, F.; Bulete, A.; Tchamitchian, S.; Cousin, M.; Pelissier, M.; et al. A pragmatic approach to assess the exposure of the honey bee (Apis mellifera) when subjected to pesticide spray. PLoS ONE 2014, 9, e113728. [Google Scholar]
- Wade, A.; Lin, C.H.; Kurkul, C.; Ravasz Regan, E.; Johnson, R.M. Combined Toxicity of Insecticides and Fungicides Applied to California Almond Orchards to Honey Bee Larvae and Adults. Insects 2019, 10, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boff, S.; Raizer, J.; Lupi, D. Environmental Display Can Buffer the Effect of Pesticides on Solitary Bees. Insects 2020, 11, 417. [Google Scholar] [CrossRef]
- Rortais, A.; Arnold, G.; Halm, M.P.; Touffet-Briens, F. Modes of honeybees exposure to systemic insecticides: Estimated amounts of contaminated pollen and nectar consumed by different categories of bees. Apidologie 2005, 36, 71–83. [Google Scholar] [CrossRef] [Green Version]
- Villa, S.; Vighi, M.; Finizio, A.; Bolchi Serini, G. Risk Assessment for Honeybees from Pesticide-Exposed Pollen. Ecotoxicology 2000, 9, 287–297. [Google Scholar] [CrossRef]
- Barmaz, S.; Potts, S.G.; Vighi, M. A novel method for assessing risks to pollinators from plant protection products using honeybees as a model species. Ecotoxicology 2010, 19, 1347–1359. [Google Scholar] [CrossRef] [PubMed]
- Barmaz, S.; Vaj, C.; Ippolito, A.; Vighi, M. Exposure of pollinators to plant protection products. Ecotoxicology 2012, 21, 2177–2185. [Google Scholar] [CrossRef] [PubMed]
- Rabea, E.I.; Nasr, H.M.; Badawy, M.E.I. Toxic Effect and Biochemical Study of Chlorfluazuron, Oxymatrine, and Spinosad on Honey Bees (Apis mellifera). Arch. Environ. Contam. Toxicol. 2010, 58, 722–732. [Google Scholar] [CrossRef]
- De Smet, L.; Hatjina, F.; Ioannidis, P.; Hamamtzoglou, A.; Schoonvaere, K.; Francis, F.; Meeus, I.; Smagghe, G.; de Graaf, D.C. Stress indicator gene expressionprofiles, colony dynamics and tissue development of honeybees exposed to sub-lethal doses of imidacloprid inlaboratory and field experiments. PLoS ONE 2017, 12, e0171529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, J.; Roche, H.; Caquet, T. Effects of physical (hypoxia, hyperoxia) and chemical (potassium dichromate, fenitrothion) stress on antioxidant enzyme activities in Chironomus riparius Mg. (Diptera, Chironomidae) larvae: Potential biomarkers. Environ. Toxicol. Chem. 2000, 19, 495–500. [Google Scholar] [CrossRef]
- Kamali, K.; Taravati, A.; Sayyadi, S.; Zahra Gharib, F.; Maftoon, H. Evidence of oxidative stress after continuous exposure to Wi-Fi radiation in rat model. Environ. Sci. Poll. Res. 2018, 25, 35396–35403. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Shrivastava, S.; Shukla, S. Exposure of Radiofrequency Electromagnetic Radiation on Biochemical and Pathological Alterations. Neurol. India 2020, 68, 1092. [Google Scholar] [PubMed]
- Lekovic, M.H.; Drekovic, N.E.; Granica, N.D.; Mahmutovic, E.H.; Djordjevic, N.Z. Extremely low-frequency electromagnetic field induces a change in proliferative capacity and redox homeostasis of human lung fibroblast cell line MRC-5. Environ. Sci. Poll. Res. 2020, 27, 39466–39473. [Google Scholar] [CrossRef] [PubMed]
- Demirci, Ö.; Güven, K.; Asma, D.; Öğüt, S.; Uğurlu, P. Effects of endosulfan, thiamethoxam, and indoxacarb in combination with atrazine on multi-biomarkers in Gammarus kischineffensis. Ecotoxicol. Environ. Saf. 2018, 147, 749–758. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, S.; Chen, J.; Zhang, C.; Xu, Z.; Li, G.; Cai, L.; Shen, W.; Wang, Q. Single and joint toxicity assessment of four currently used pesticides to zebrafish (Danio rerio) using traditional and molecular endpoints. Chemosphere 2018, 192, 14–23. [Google Scholar] [CrossRef]
- Yu, Q.Y.; Fang, S.M.; Zuo, W.D.; Dai, F.Y.; Zhang, Z.; Lu, C. Effect of Organophosphate Phoxim Exposure on Certain Oxidative Stress Biomarkers in the Silkworm. J. Econ. Entomol. 2011, 104, 101–106. [Google Scholar] [CrossRef]
- Claudianos, C.; Ranson, H.; Johnson, R.M.; Biswas, S.; Schuler, M.A.; Berenbaum, M.R.; Feyereisen, R.; Oakeshott, J.G. A deficit of detoxification enzymes: Pesticide sensitivity and environmental response in the honeybee. Insect Mol. Biol. 2006, 15, 615–636. [Google Scholar] [CrossRef] [Green Version]
- Słowińska, M.; Nynca, J.; Wilde, J.; Bąk, B.; Siuda, M.; Ciereszko, A. Total antioxidant capacity of honeybee haemolymph in relation to age and exposure to pesticide, and comparison to antioxidant capacity of seminal plasma. Apidologie 2016, 47, 227–236. [Google Scholar] [CrossRef] [Green Version]
- Vilić, M.; Tlak Gajger, I.; Tucak, P.; Štambuk, A.; Šrut, M.; Klobučar, G.; Malarić, K.; Žura Žaja, I.; Pavelić, A.; Manger, M.; et al. Effects of short-term exposure to mobile phone radiofrequency (900 MHz) on the oxidative response and genotoxicity in honey bee larvae. J. Apic. Res. 2017, 56, 430–438. [Google Scholar] [CrossRef]
- Gandhi, G.; Singh, P. Cytogenetic Damage in Mobile Phone Users: Preliminary Data. Int. J. Hum. Genet. 2005, 5, 259–265. [Google Scholar] [CrossRef]
- Hardell, L.; Sage, C. Biological effects from electromagnetic field exposure and public exposure standards. Biomed. Pharmacother. 2008, 62, 104–109. [Google Scholar] [CrossRef]
- Leszczynski, D.; Joenväärä, S.D.; Reivinen, J.; Kuokka, R. Non-thermal activation of the hsp27/p38MAPK stress pathway by mobile phone radiation in human endothelial cells: Molecular mechanism for cancer-and blood-brain barrier-related effects. Differentiation 2002, 70, 120–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desai, N.R.; Kesari, K.K.; Agarwal, A. Pathophysiology of cell phone radiation: Oxidative stress and carcinogenesis with focus on male reproductive system. Reprod. Biol. Endocrinol. 2009, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, H.; Singh, N.P. Single- and double-strand DNA breaks in rat brain cells after acute exposure to radiofrequency electromagnetic radiation. Int. J. Radiat. Biol. 1996, 69, 513–521. [Google Scholar] [CrossRef]
- Nicholls, B.; Racey, P.A. Bats avoid radar installations: Could electromagnetic fields deter bats from colliding with wind turbines? PLoS ONE 2007, 2, e297. [Google Scholar] [CrossRef] [Green Version]
- Balmori, A. Electromagnetic pollution from phone masts. Effects on wildlife. Pathophysiology 2009, 16, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Everaert, J.; Bauwens, D. A possible effect of electromagnetic radiation from mobile phone base stations on the number of breeding House Sparrows (Passer domesticus). Electromagn. Biol. Med. 2007, 26, 63–72. [Google Scholar] [CrossRef] [Green Version]
- Weisbrot, D.; Lin, H.; Ye, L.; Blank, M.; Goodman, R. Effects of mobile phone radiation on reproduction and development in Drosophila melanogaster. J. Cell. Biochem. 2003, 89, 48–55. [Google Scholar] [CrossRef]
- Favre, D. Mobile phone-induced honeybee worker piping. Apidologie 2011, 42, 270–279. [Google Scholar] [CrossRef] [Green Version]
- Sharma, V.P.; Kumar, N.R. Changes in honeybee behaviour and biology under the influence of cellphone radiations. Curr. Sci. 2010, 98, 1376–1378. [Google Scholar] [CrossRef]
- Bindokas, V.P.; Gauger, J.R.; Greenberg, B. Mechanism of Biological Effects Observed in Honey Bees (Apis mellifera, L.) Hived Under Extra-High-Voltage Transmission Lines: Implications Derived From Bee Exposure to Simulated Intense Electric Fields and Shocks. Bioelectromagnetics 1988, 9, 285–301. [Google Scholar] [CrossRef]
- Afrasiabi, A.; Riazi, G.H.; Abbasi, S.; Dadras, A.; Ghalandari, B.; Seidkhani, H.; Modaresi, S.M.S.; Masoudian, N.; Amani, A.; Ahmadian, S. Synaptosomal acetylcholinesterase activity variation pattern in the presence of electromagnetic fields. Int. J. Biol. Macromol. 2014, 65, 8–15. [Google Scholar] [CrossRef]
- Regoli, F.; Gorbi, S.; Machella, N.; Tedesco, S.; Benedetti, M.; Bocchetti, R.; Notti, A.; Fattorini, D.; Piva, F.; Principato, G. Pro-oxidant effects of extremely low frequency electromagnetic fields in the land snail Helix aspersa. Free Radic. Biol. Med. 2005, 39, 1620–1628. [Google Scholar] [CrossRef] [PubMed]
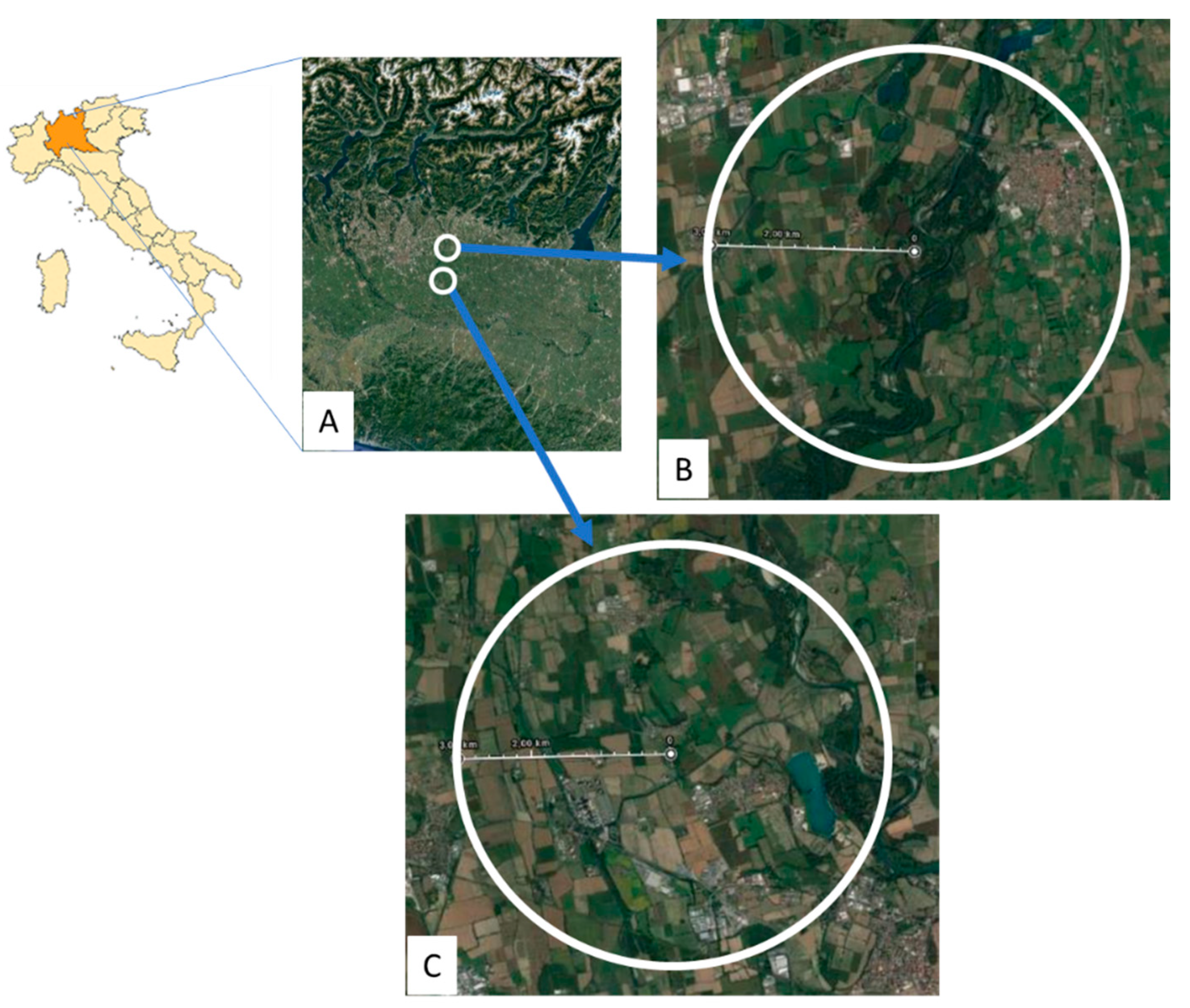
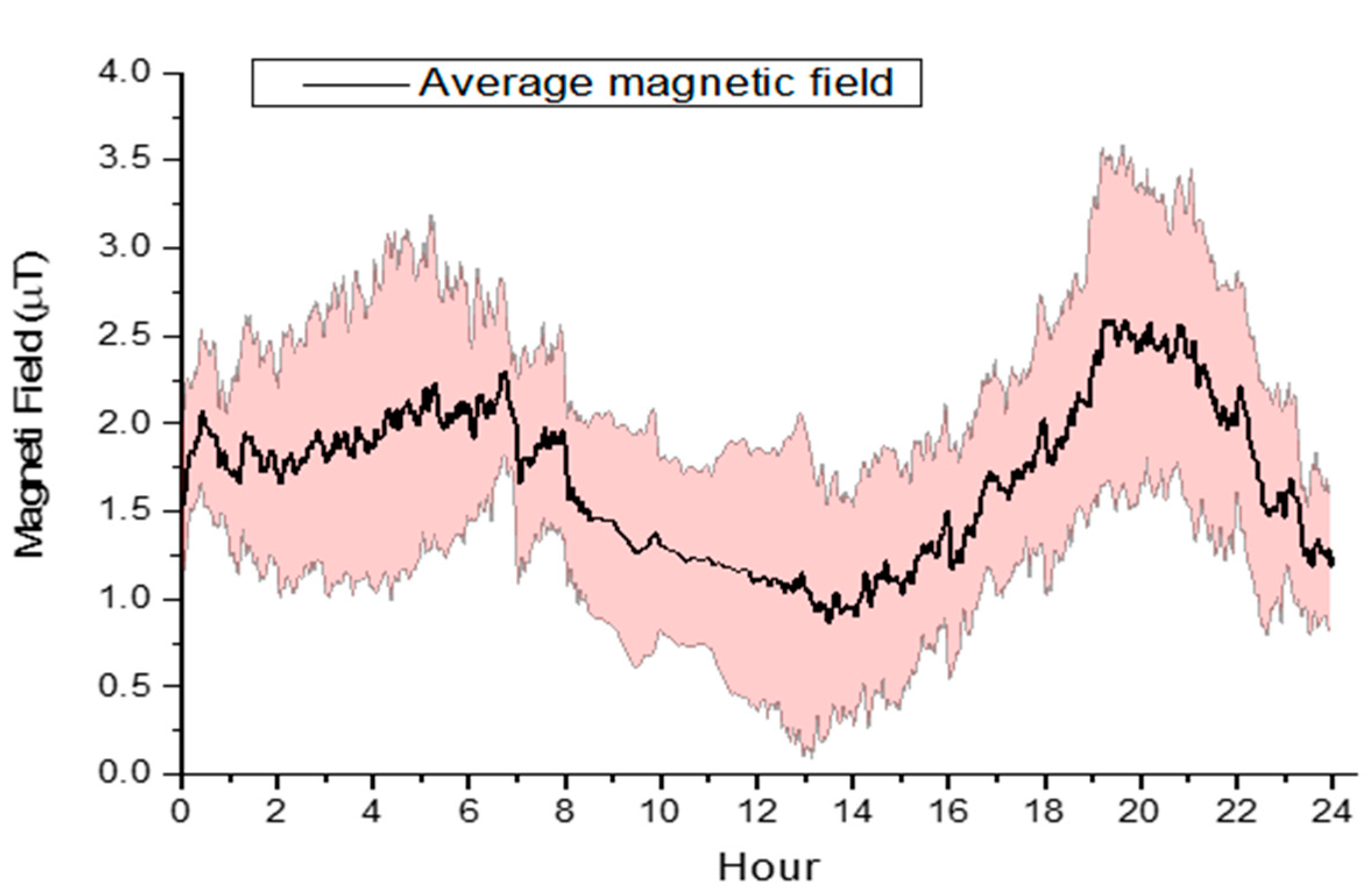
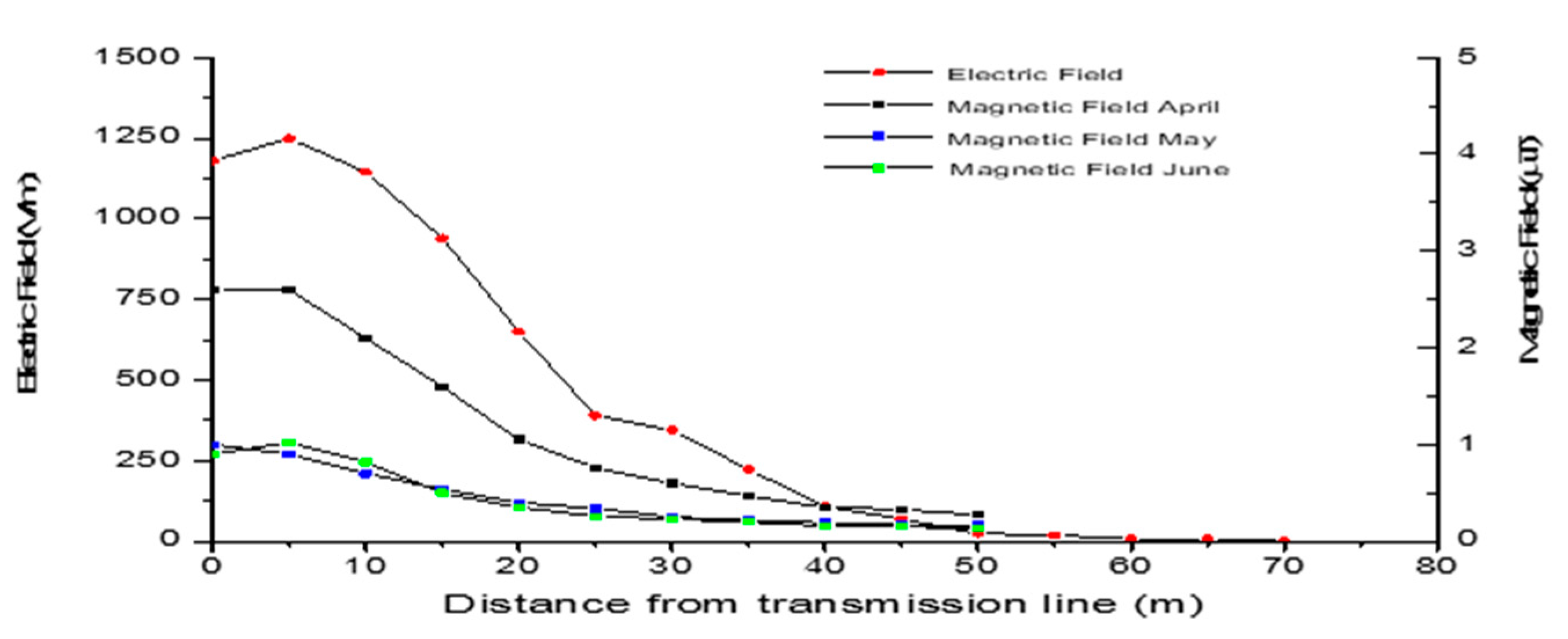
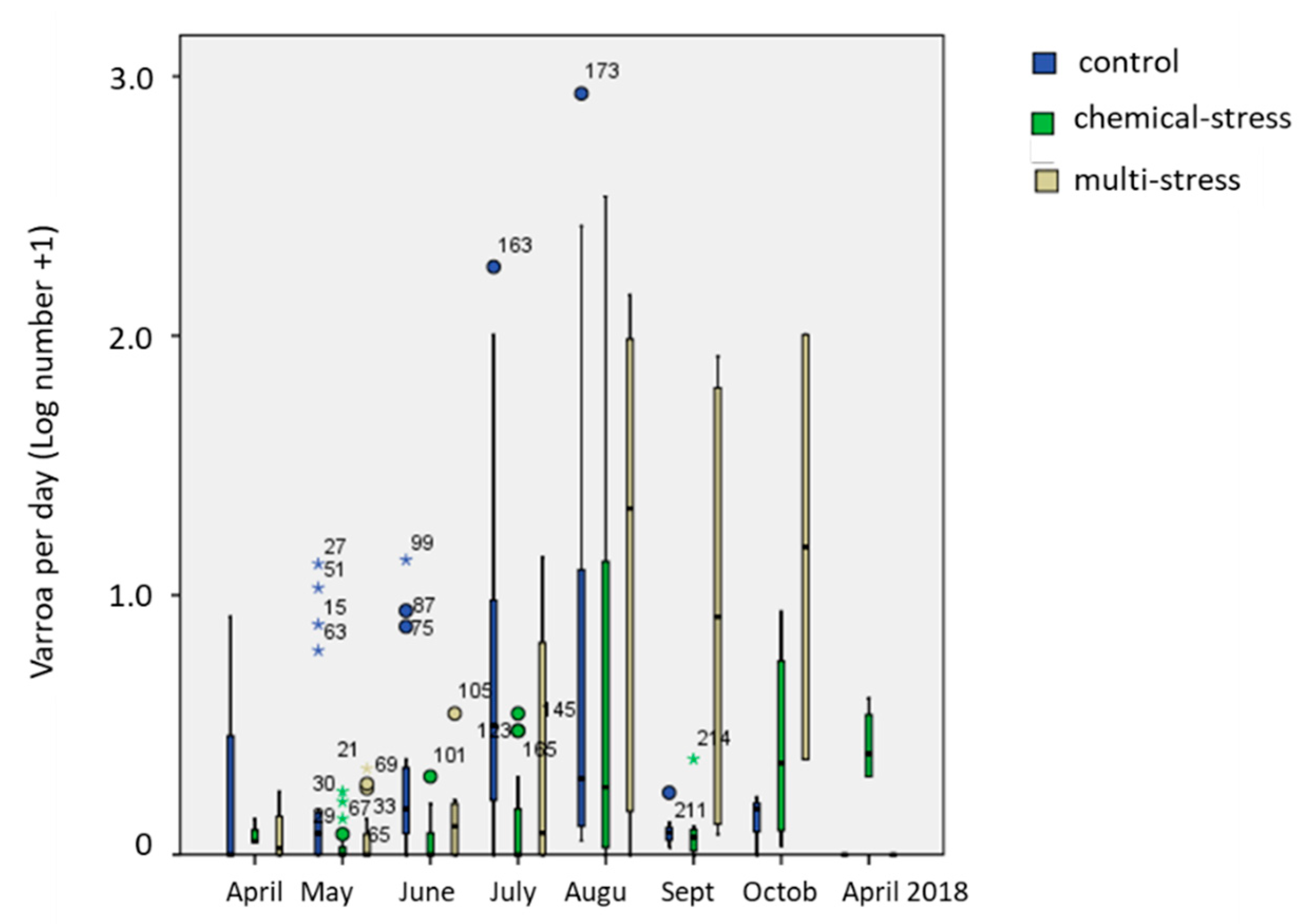
| Type | Parameter | Period | Methodology |
|---|---|---|---|
| Biological Observation | Hive Inspection | Weekly | Comb inspection and visual determination of the relative amount of storages, brood and empty space, according to [45] |
| Comb Analysis | Weekly | Software image analysis of the central comb of each hive | |
| Queen Activity | Weekly | Visual inspection of the queen presence and deposition | |
| Bee mortality | Weekly | Count of dead bees of different stages and caste in the underbaskets, according to [46] | |
| Parasites and pathogens | Varroa | Weekly | Count of the fallen mites on adhesive sticky boards positioned on the bottom of each hives |
| Varroa | Six time | Count of the fallen mites after powdered sugar application | |
| Virus | Monthly | Real-Time–PCR [47] | |
| American foulbrood | Weekly Monthly | Visual inspection of the colonies Spore culture method, according to [48] | |
| Biomarker | Acetylcholinesterase | Monthly | Spectrophotometric method, according to [49] |
| Catalase | Monthly | Spectrophotometric method, according to [50] | |
| Glutathione S-transferase | Monthly | Spectrophotometric method, according to [51] | |
| Alkaline phosphatase | Monthly | Spectrophotometric method, according to [52] | |
| Reactive Oxygen Species | Monthly | Spectrophotometric method, according to [53] | |
| Lipid peroxidation | Monthly | Spectrophotometric method, according to [54] | |
| DNA fragmentation | Monthly | Spectrophotometric method, according to [55] |
| Product Use | Active Ingredient | Culture | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Crop Surface (ha) | Apple 1.5 | Pear 1.0 | Peach 5.0 | Abricot 0.2 | Plum 1.5 | Cherry 0.15 | |||
| kg a.i. | kg a.i. | kg a.i. | kg a.i. | kg a.i. | kg a.i. | kg a.i. | |||
| Actara® 240 SC | insecticide | thiamethoxam a.i. 216 g/Kg | 0.11 | 0.11 | |||||
| Affirm® | insecticide | emamectin benzoate a.i. 9.5 g/kg | 0.01 | 0.01 | 0.02 | ||||
| Aliette® | fungicide | fosetyl-aluminium a.i. 800 g/kg | 9.6 | 9.6 | |||||
| Alsystin® SC | insecticide | triflumuron a.i.480.7 g/L | 0.12 | 0.72 | 0.84 | ||||
| Caddy® | fungicide | cyproconazole a.i. 100 g/kg | 0.01 | 0.04 | 0.05 | 0.01 | 0.11 | ||
| Confidor® 200 SL | insecticide | imidacloprid a.i. 200 g/L | 0.15 | 0.15 | |||||
| Coragen® | insecticide | chlorantraniliprole a.i. 200 g/L | 0.08 | 0.08 | |||||
| Crittam WG® | fungicide | ziram a.i. 760 g/kg | 7.6 | 3.04 | 6.08 | 1.52 | 18.2 | ||
| Crittox® | fungicide | mancozeb 750 g/kg | 7.5 | 7.5 | |||||
| Decis® Jet | insecticide | deltamethrin a.i. 15 g/L | 0.01 | 0.02 | 0.05 | 0.02 | 0.01 | 0.11 | |
| Decision® | insecticide | deltamethrin a.i. 15 g/L | 0.01 | 0.02 | 0.01 | 0.04 | |||
| Delan® 70 WG | fungicide | dithianon a.i. 700 g/kg | 6.65 | 3.15 | 9.8 | ||||
| Difcor® | fungicide | difenoconazole a.i. 250 g/L | 0.13 | 0.13 | |||||
| Efuzin 355 SC® | fungicide | dodine a.i. 355 g/L | 2.84 | 2.84 | |||||
| Enovit metile® FL | fungicide | thiophanate-methyl a.i.417 g/kg | 0.83 | 0.83 | |||||
| Fixormon ® | plant regulator | NAA (1-naphthylacetic acid) a.i. 85 g/L | 0.03 | 0.03 | |||||
| Indar® | fungicide | fenbuconazole a.i. 50 g/L | 0.03 | 0.24 | 0.05 | 0.05 | 0.37 | ||
| Intrepid® | insecticide | methoxyfenozide a.i. 240 g/L | 0.24 | 0.12 | 0.36 | ||||
| Iperion® | fungicide | copper oxychloride a.i. 375 g/kg | 11.25 | 8.81 | 11.25 | 3.38 | 7.13 | 3.19 | 45 |
| Klartan® | insecticide | tau-fluvalinate a.i. 240 g/L | 0.14 | 0.05 | 0.19 | ||||
| Kohinor ® | insecticide | imidacloprid a.i. 200 g/L | 0.2 | 0.2 | |||||
| LaserTM | insecticide | spinosad a.i. 480 g/L | 0.12 | 0.36 | 0.14 | 0.62 | |||
| Nimrod® | fungicide | bupirimate a.i. 250 g/L | 2.0 | 2.0 | |||||
| Oleoter® | insecticide | miner al oil 688 g/L | 27.52 | 17.2 | 34.4 | 10.32 | 10.32 | 99.8 | |
| Ovipron® | insecticide | miner al oil 800 g/L | 3.2 | 3.2 | |||||
| Polithiol® | insecticide | mineral oil 420 g/L | 8.4 | 8.4 | |||||
| Prodigy ® | insecticide | methoxyfenozide a.i. 240 g/L | 0.11 | 0.32 | 0.43 | ||||
| Reldan® | insecticide | chlor py rifos-methyl a.i. 255 g/L | 0.23 | 1.35 | 1.58 | ||||
| Scala® | fungicide | pyrimethanil a.i. 400 g/L | 1.1 | 0.9 | 2 | ||||
| Spada® | insecticide | phosmet a.i. 177 g/kg | 0.71 | 0.53 | 1.24 | ||||
| Switch® | fungicide | cy prodinil a.i. 375 g/kg | 0.28 | 0.24 | 0.52 | ||||
| fludioxonil a.i. 250 g/kg | 0.19 | 0.16 | 0.35 | ||||||
| Tebusip combi® | fungicide | tebuconazole a.i. 45 g/L | 0.54 | 0.54 | |||||
| sulfur a.i. 700 g/kg | 8.4 | 8.4 | |||||||
| Teldor® | fungicide | fenhexamid a.i. 500 g/L | 0.63 | 0.13 | 0.76 | ||||
| Tiovit ® | fungicide | sulfur a.i. 800 g/kg | 25.6 | 14.8 | 0.8 | 41.2 | |||
| Trebon® | insecticide | etofenprox a.i. 287.5 g/L | 0.12 | 0.17 | 0.45 | 0.06 | 0.8 | ||
| Zetor® | insecticide | abamectin a.i. 18 g/L | 0.02 | 0.02 | |||||
| Fungicide treatment n° | 13 | 11 | 17 | 6 | 9 | 11 | 67 | ||
| lnsecticide treatment n° | 13 | 11 | 10 | 1 | 5 | 4 | 44 | ||
| Total treatment n° | 26 | 22 | 27 | 7 | 14 | 15 | 111 | ||
| Fungicide kg a.i. | 52.1 | 32.9 | 38.9 | 7.22 | 13.9 | 5.2 | 150 | ||
| Insecticide kg a.i. | 37.3 | 21.9 | 37.9 | 10.3 | 10.5 | 0.13 | 118 | ||
| Pesticide kg a.i. | 89.5 | 54.8 | 76.9 | 17.6 | 24.4 | 5.29 | 268 | ||
| Dependent Variable | Factor | D.F. | F | p |
|---|---|---|---|---|
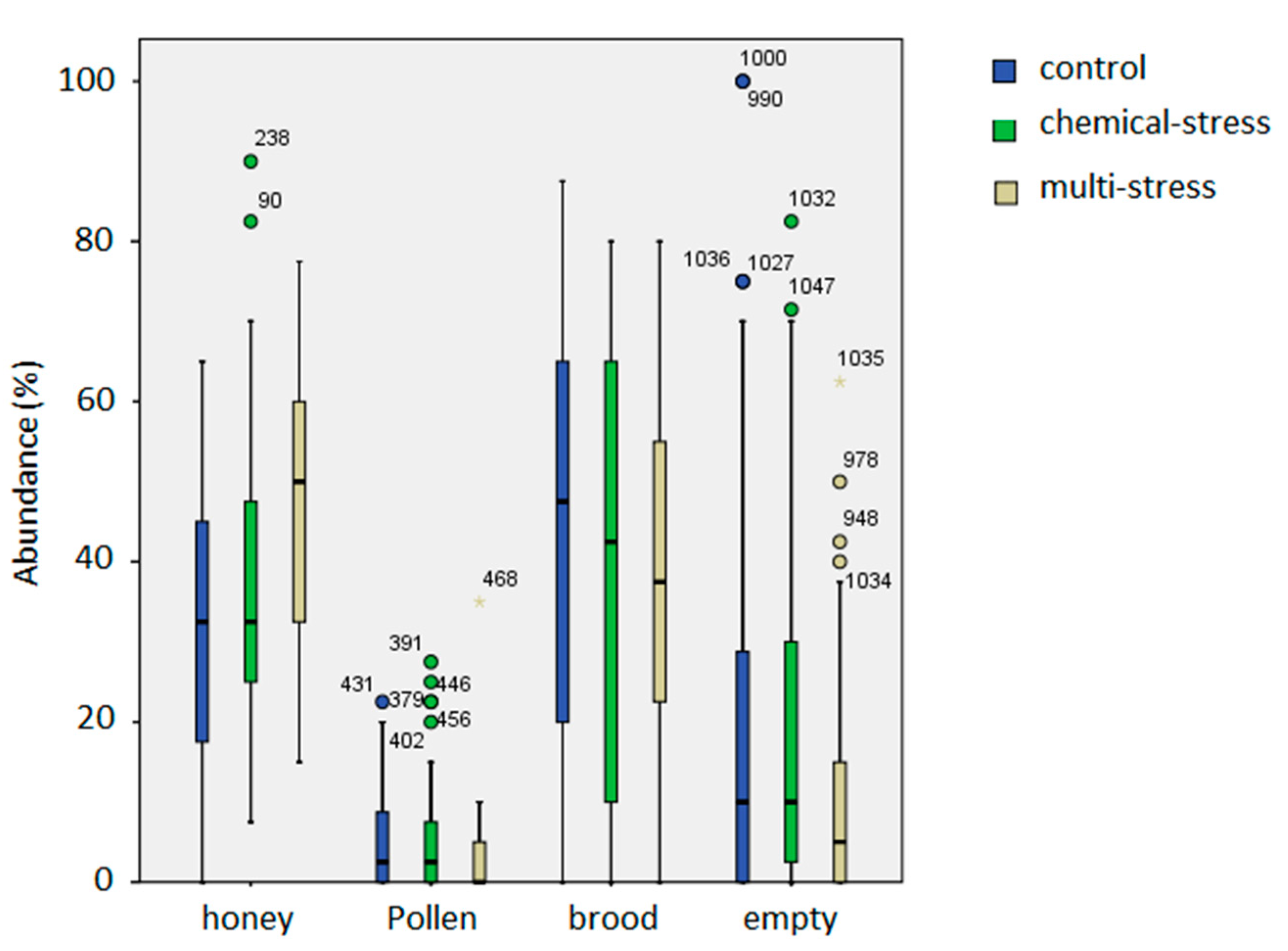
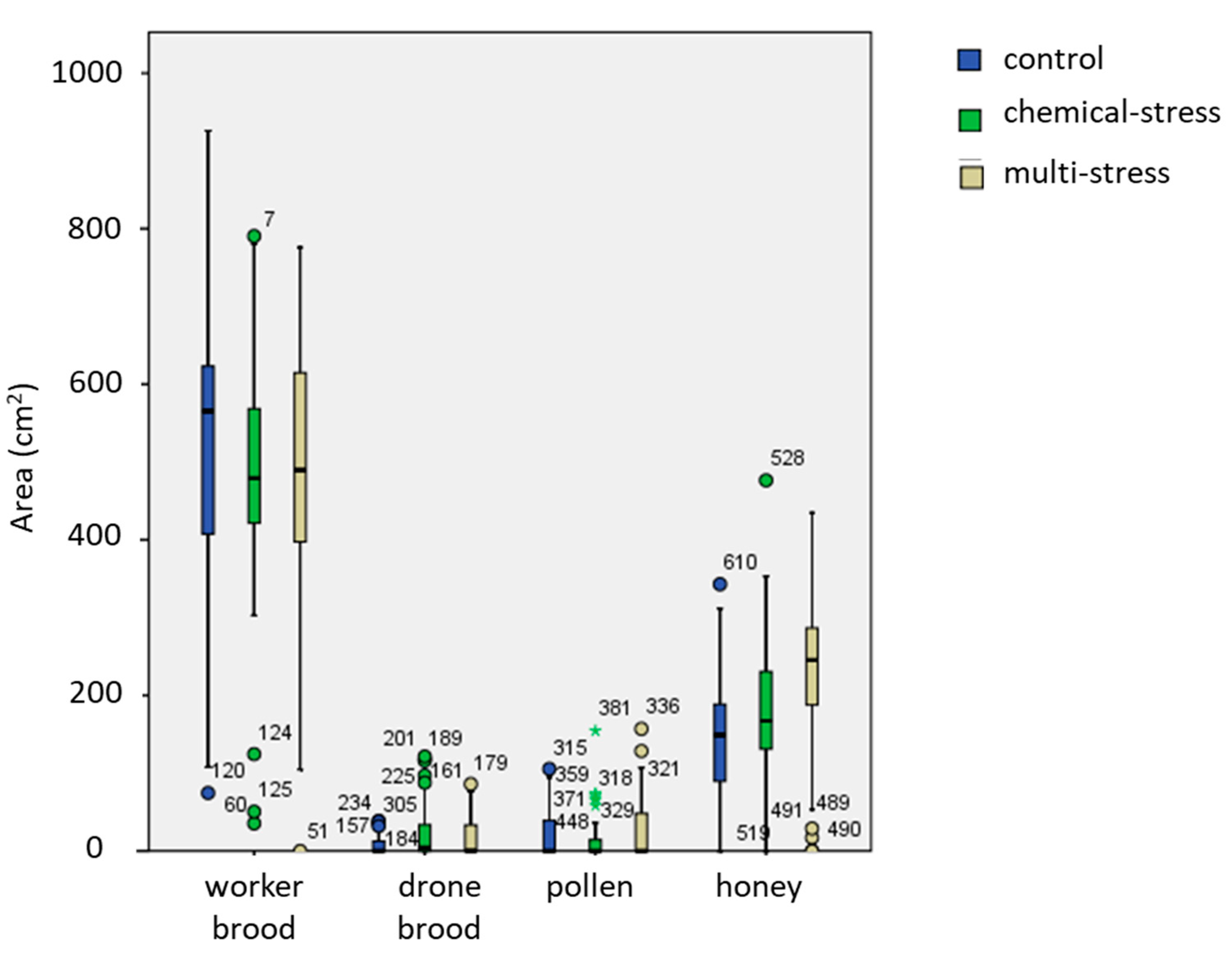
| Honey | Hive | 3;219 | 3.50 | 0.015 * |
| Date | 24;219 | 1.65 | 0.033 * | |
| Treatment | 2;219 | 19.6 | <0.001 *** | |
| Pollen | Hive | 3;218 | 0.17 | 0.92 |
| Date | 24;218 | 2.9 | <0.001 *** | |
| Treatment | 2;218 | 3.9 | 0.022 * | |
| Brood | Hive | 3;220 | 1.4 | 0.25 |
| Date | 24;220 | 12.8 | <0.001 *** | |
| Treatment | 2;220 | 3.9 | 0.022 * | |
| Empty space | Hive | 3;213 | 7.3 | <0.001 *** |
| Date | 24;213 | 9.6 | <0.001 *** | |
| Treatment | 2;213 | 2.2 | 0.12 |
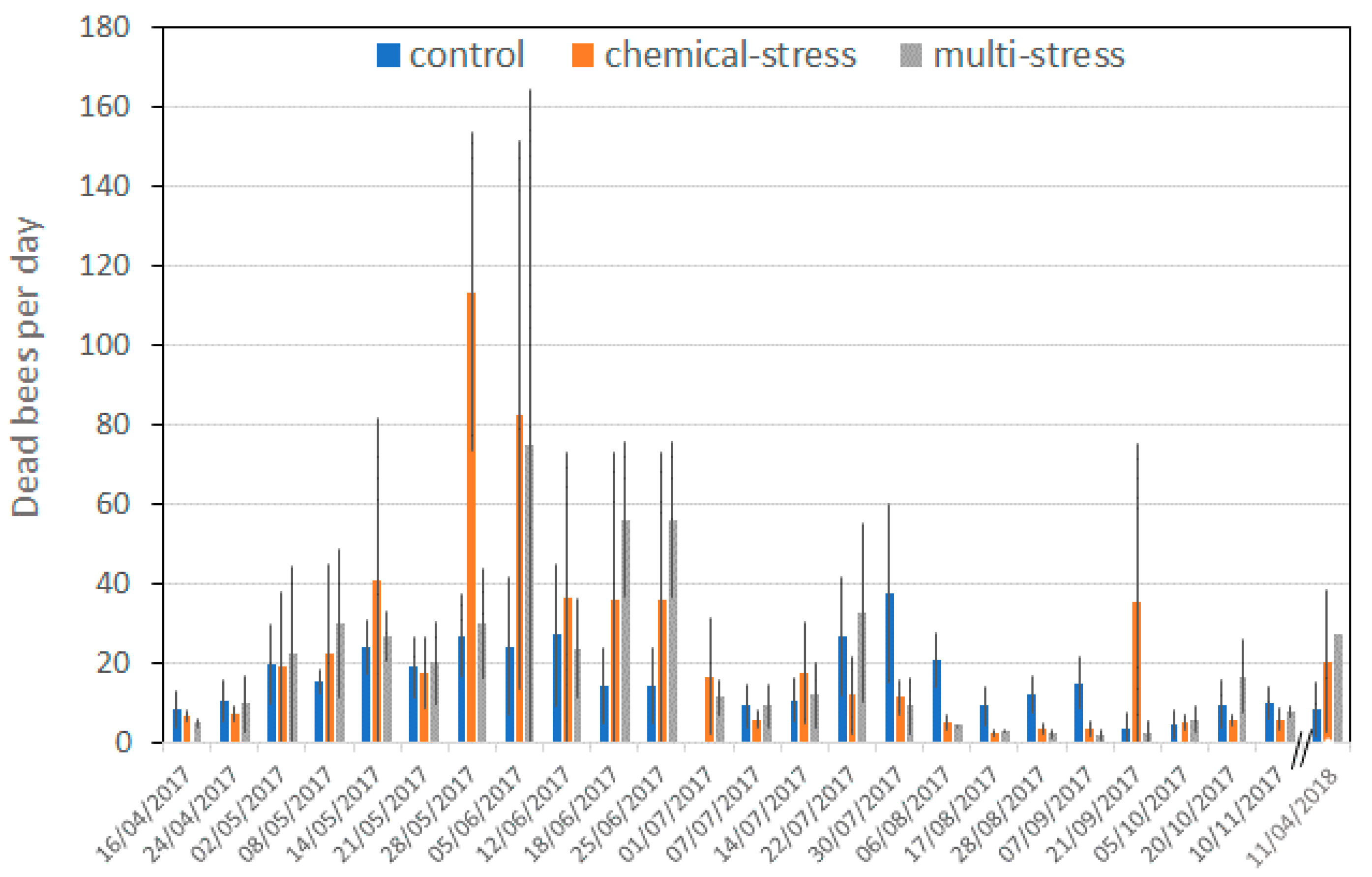
| Treatment | Dead Animals/10 days | ||||||
|---|---|---|---|---|---|---|---|
| Worker Bees | Drones | Pupae | Queens | ||||
| Normal | Deformed | Normal | Deformed | Workers | Drones | ||
| Control | 165 ± 119 | 4.4 ± 8.7 | 11 ± 23 | 1.9 ± 4.1 | 0.17 ± 0.65 | 0.32 ± 1.2 | 0.14 ± 0.54 |
| (3.8–706) | (0–48.8) | (0–188) | (0–28) | (0–5) | (0–8.8) | (0–3.8) | |
| Chemical-stress | 228 ± 336 | 2.2 ± 6.9 | 11 ± 19 | 3.2 ± 13.6 | 0.11 ± 0.56 | 0.80 ± 2.4 | 0.21 ± 1.3 |
| (6.3–1769) | (0–62.5) | (0–160) | (0–134) | (0–5) | (0–17.5) | (0–12.5) | |
| Multi-stress | 232 ± 286 | 2.4 ± 5.1 | 13 ± 14 | 2.9 ± 4.9 | 0.12 ± 0.42 | 2.1 ± 0.72 | 0.35 ± 1.3 |
| (6.3–2069) | (0–28.8) | (0–49) | (0–23) | (0–2.5) | (0–5) | (0–8.8) | |
| Biomarker | Stage | N | Mean | St dev | Min | Max | Percentiles | ||
|---|---|---|---|---|---|---|---|---|---|
| 25th | 50th | 75th | |||||||
| AChE | pupae | 115 | 0.053 | 0.018 | 0.020 | 0.11 | 0.040 | 0.049 | 0.064 |
| worker | 120 | 0.15 | 0.076 | 0.050 | 0.36 | 0.090 | 0.12 | 0.19 | |
| CAT | pupae | 115 | 16.3 | 6.1 | 2.7 | 34 | 13.1 | 16.7 | 19.4 |
| worker | 131 | 22.4 | 8.4 | 6.2 | 45 | 16.3 | 22.7 | 28.5 | |
| GST | pupae | 115 | 0.37 | 0.086 | 0.20 | 0.53 | 0.30 | 0.37 | 0.44 |
| worker | 131 | 0.26 | 0.066 | 0.17 | 0.52 | 0.22 | 0.25 | 0.29 | |
| ALP | pupae | 104 | 0.004 | 0.003 | 0 | 0.010 | 0.002 | 0.003 | 0.005 |
| worker | 135 | 0.014 | 0.008 | 0 | 0.040 | 0.009 | 0.013 | 0.018 | |
| ROS | pupae | 111 | 3.3 × 104 | 2.6 × 104 | 5.2 × 103 | 1.6 × 105 | 1.6 × 104 | 2.5 × 104 | 3.9 × 104 |
| worker | 121 | 3.5 × 105 | 2.4 × 105 | 2.9 × 104 | 1.0 × 106 | 1.5 × 105 | 3.1 × 105 | 4.8 × 105 | |
| LPO | pupae | 88 | 1.7 | 2.0 | 0 | 14.3 | 0.60 | 1.2 | 1.8 |
| worker | 124 | 4.7 | 3.4 | 0.4 | 18.1 | 2.3 | 3.6 | 6.0 | |
| FRAM | pupae | 113 | 82 | 64 | 4.8 | 468 | 46 | 68 | 91 |
| worker | 122 | 135 | 87 | 8.5 | 422 | 68 | 114 | 180 | |
| Dependent Variable | Factor | Pupae | Worker Bees | ||||
|---|---|---|---|---|---|---|---|
| D.F. | F | p | D.F. | F | p | ||
| AChE | Date | 8;316 | 10 | <0.001 *** | 8;331 | 34 | <0.001 *** |
| Treatment | 2;316 | 9.9 | <0.001 *** | 2;331 | 2.3 | 0.10 | |
| Hive | 3;316 | 3.7 | 0.013 * | 3;331 | 1.5 | 0.21 | |
| date*treat | 15;316 | 4.7 | <0.001 *** | 15;331 | 6.9 | <0.001 *** | |
| CAT | Date | 8;316 | 11 | <0.001 *** | 8;339 | 15 | <0.001 *** |
| Treatment | 2;316 | 3.2 | 0.044 * | 2;339 | 0.1 | 0.9 | |
| Hive | 3;316 | 0.28 | 0.84 | 3;339 | 1.9 | 0.13 | |
| date*treat | 15;316 | 2.2 | 0.005 ** | 15;339 | 2.3 | 0.003 ** | |
| GST | Date | 8;316 | 7.3 | <0.001 *** | 8;339 | 94 | <0.001 *** |
| Treatment | 2;316 | 3.4 | 0.036 * | 2;339 | 5.8 | 0.003 ** | |
| Hive | 3;316 | 3.1 | 0.027 * | 3;339 | 0.91 | 0.44 | |
| date*treat | 15;316 | 5.0 | <0.001 *** | 15;339 | 5.4 | <0.001 *** | |
| ALP | Date | 8;278 | 25 | <0.001 *** | 8;343 | 15 | <0.001 *** |
| Treatment | 2;278 | 2.5 | 0.084 | 2;343 | 1.0 | 0.36 | |
| Hive | 3;278 | 3.7 | 0.012 * | 3;343 | 1.2 | 0.30 | |
| date*treat | 15;278 | 9.4 | <0.001 *** | 15;343 | 4.6 | <0.001 *** | |
| ROS | Date | 8;311 | 14 | <0.001 *** | 8;316 | 3.9 | <0.001 *** |
| Treatment | 2;311 | 6.6 | 0.001 ** | 2;316 | 7.5 | 0.001 ** | |
| Hive | 3;311 | 2.2 | 0.087 | 3;316 | 2.4 | 0.069 | |
| date*treat | 15;311 | 8.9 | <0.001 *** | 15;316 | 0.78 | 0.71 | |
| LPO | Date | 8;245 | 5.2 | <0.001 *** | 8;314 | 7.3 | <0.001 *** |
| Treatment | 2;245 | 0.2 | 0.79 | 2;314 | 2.1 | 0.13 | |
| Hive | 3;245 | 2.6 | 0.051 | 3;314 | 1.3 | 0.27 | |
| date*treat | 15;245 | 3.0 | <0.001 *** | 15;314 | 2.6 | 0.001 ** | |
| DNAFRAGM | Date | 8;308 | 14 | <0.001 *** | 8;321 | 9.9 | <0.001 *** |
| Treatment | 2;308 | 3.8 | 0.024 * | 2;321 | 1.2 | 0.30 | |
| Hive | 3;308 | 0.67 | 0.57 | 3;321 | 0.76 | 0.52 | |
| date*treat | 15;308 | 2.0 | 0.015 * | 15;321 | 3.8 | <0.001 *** | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupi, D.; Palamara Mesiano, M.; Adani, A.; Benocci, R.; Giacchini, R.; Parenti, P.; Zambon, G.; Lavazza, A.; Boniotti, M.B.; Bassi, S.; et al. Combined Effects of Pesticides and Electromagnetic-Fields on Honeybees: Multi-Stress Exposure. Insects 2021, 12, 716. https://doi.org/10.3390/insects12080716
Lupi D, Palamara Mesiano M, Adani A, Benocci R, Giacchini R, Parenti P, Zambon G, Lavazza A, Boniotti MB, Bassi S, et al. Combined Effects of Pesticides and Electromagnetic-Fields on Honeybees: Multi-Stress Exposure. Insects. 2021; 12(8):716. https://doi.org/10.3390/insects12080716
Chicago/Turabian StyleLupi, Daniela, Marco Palamara Mesiano, Agnese Adani, Roberto Benocci, Roberto Giacchini, Paolo Parenti, Giovanni Zambon, Antonio Lavazza, Maria Beatrice Boniotti, Stefano Bassi, and et al. 2021. "Combined Effects of Pesticides and Electromagnetic-Fields on Honeybees: Multi-Stress Exposure" Insects 12, no. 8: 716. https://doi.org/10.3390/insects12080716









