Transgenerational Effects of a Neonicotinoid and a Novel Sulfoximine Insecticide on the Harlequin Ladybird
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Biological Materials
2.2. Chemicals
2.3. Insecticide Baseline Toxicity on Prey
2.4. Lethal Toxicity on Harmonia Axyridis
2.5. Sublethal Effect on Longevity and Reproductive Traits
2.6. Transgenerational Effects on Offspring Survival Rate and Developmental Time
2.7. Statistical Analysis
3. Results
3.1. Insecticide Baseline Toxicity on Prey
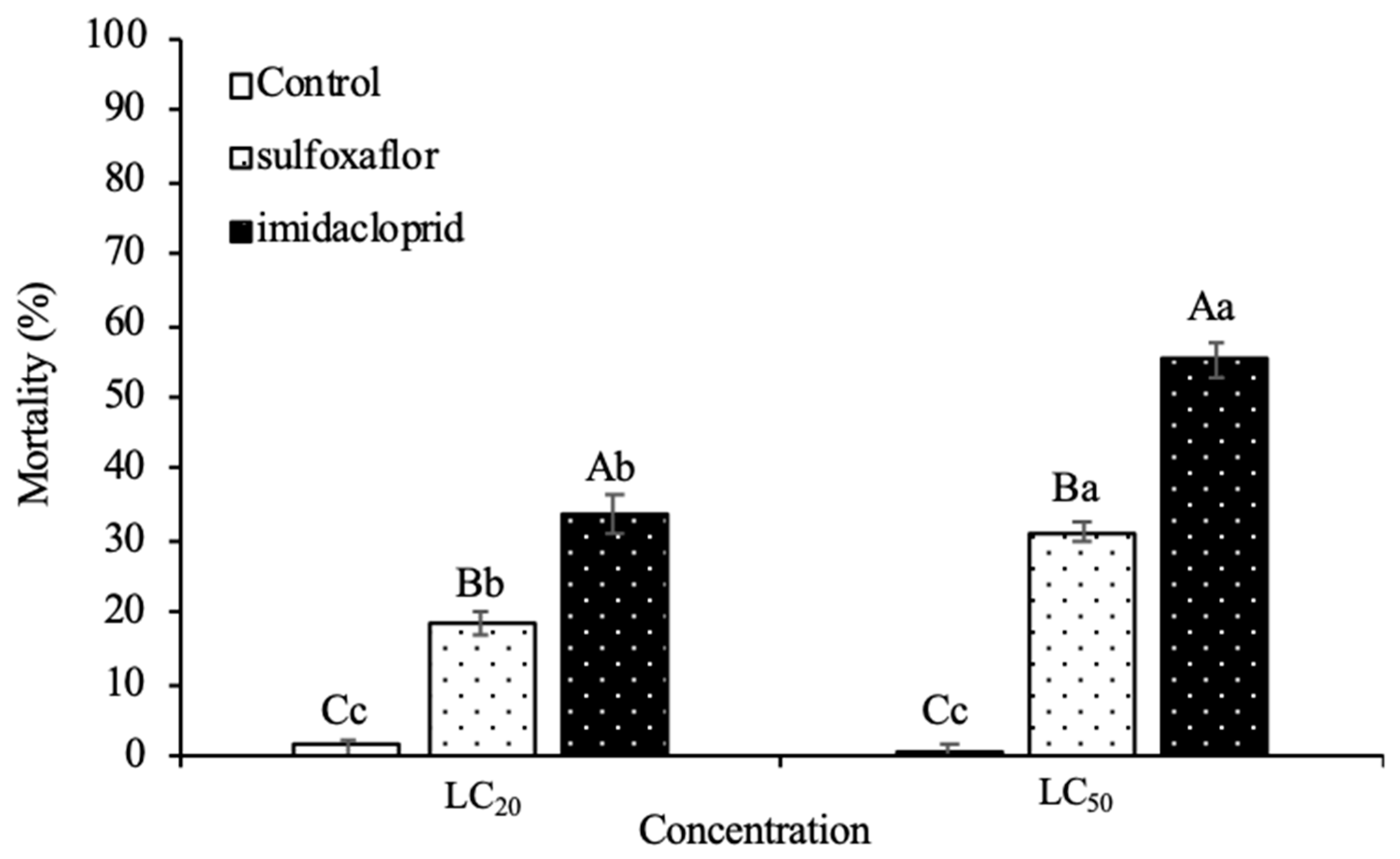
3.2. Lethal Toxicity on Harmonia Axyridis
3.3. Influence of Insecticides on Longevity and Fecundity of Females
3.4. Effect of Insecticides on Survival Rate and Developmental Time of Offspring
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Koch, R.L. The multicolored Asian lady beetle, Harmonia axyridis: A review of its biology, uses in biological control, and non-target impacts. J. Pest Sci. 2003, 1, 32. [Google Scholar] [CrossRef] [Green Version]
- Brown, P.M.J.; Adriaens, T.; Bathon, H.; Cuppen, J.; Goldarazena, A.; Hägg, T.; Kenis, M.; Klausnitzer, B.E.M.; Kovář, I.; Loomans, A.J.M.; et al. Harmonia axyridis in Europe: Spread and distribution of a non-native coccinellid. BioControl 2008, 53, 5–21. [Google Scholar] [CrossRef] [Green Version]
- Roy, H.E.; Brown, P.M.; Adriaens, T.; Berkvens, N.; Borges, I.; Clusella-Trullas, S.; Comont, R.F.; Clercq, P.D.; Eschen, R.; Estoup, A.; et al. The harlequin ladybird, Harmonia axyridis: Global perspectives on invasion history and ecology. Biol. Invasions 2016, 18, 997–1044. [Google Scholar] [CrossRef]
- Hodek, I.; Honek, A.; Van Emden, H.F. Ecology and Behaviour of the Ladybird Beetles (Coccinellidae); John Wiley &Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Riddick, E.W. Spotlight on the positive effects of the ladybird Harmonia axyridis on agriculture. BioControl 2017, 62, 319–330. [Google Scholar] [CrossRef]
- Wang, Y.S.; Yao, F.L.; Soares, M.A.; Basiri, S.E.; Amiens-Desneux, E.; Campos, M.R.; Lavoir, A.-V.; Desneux, N. Effects of four non-crop plants on life history traits of the lady beetle Harmonia axyridis. Entomol. Gen. 2020, 40, 243–252. [Google Scholar] [CrossRef]
- Thomine, E.; Rusch, A.; Supplisson, C.; Monticelli, L.S.; Amiens-Desneux, E.; Lavoir, A.-V.; Desneux, N. Highly diversified crop systems can promote the dispersal and foraging activity of the generalist predator Harmonia axyridis. Entomol. Gen. 2020, 40, 133–145. [Google Scholar] [CrossRef] [Green Version]
- Wells, M.L.; McPherson, R.M.; Ruberson, J.R.; Herzog, G.A. Coccinellids in cotton: Population response to pesticide application and feeding response to cotton aphids (Homoptera: Aphididae). Environ. Entomol. 2001, 4, 785–793. [Google Scholar] [CrossRef]
- Ali, A.; Desneux, N.; Lu, Y.H.; Liu, B.; Wu, K.M. Characterization of the natural enemy community attacking cotton aphid in the Bt cotton ecosystem in Northern China. Sci. Rep. 2016, 6, 24273. [Google Scholar] [CrossRef]
- González-Mas, N.; Cuenca-Medina, M.; Gutiérrez-Sánchez, F.; Quesada-Moraga, E. Bottom-up effects of endophytic Beauveria bassiana on multitrophic interactions between the cotton aphid, Aphis gossypii, and its natural enemies in melon. J. Pest Sci. 2019, 92, 1271–1281. [Google Scholar] [CrossRef] [Green Version]
- Henneberry, T.; Jech, L.F.; Torre, T.; Hendrix, D. Cotton aphid (Homoptera: Aphididae) biology, honeydew production, sugar quality and quantity, and relationships to sticky cotton. Southwest. Entomol. 2000, 25, 161–174. [Google Scholar] [CrossRef]
- Campolo, O.; Chiera, E.; Malacrinò, A.; Laudani, F.; Fontana, A.; Albanese, G.R.; Palmeri, V. Acquisition and transmission of selected CTV isolates by Aphis gossypii. J. Asia-Pac. Entomol. 2014, 17, 493–498. [Google Scholar] [CrossRef]
- Yousaf, H.K.; Shan, T.S.; Chen, X.W.; Ma, K.S.; Shi, X.Y.; Desneux, N.; Biondi, A.; Gao, X.W. Impact of the secondary plant metabolite Cucurbitacin B on the demographical traits of the melon aphid, Aphis gossypii. Sci. Rep. 2018, 8, 16473. [Google Scholar] [CrossRef]
- Yuan, H.B.; Li, J.H.; Liu, Y.Q.; Cui, L.; Lu, Y.H.; Xu, X.Y.; Li, Z.; Wu, K.M.; Desneux, N. Lethal, sublethal and transgenerational effects of the novel chiral neonicotinoid pesticide cycloxaprid on demographic and behavioral traits of Aphis gossypii (Hemiptera: Aphididae). Insect Sci. 2017, 24, 743–752. [Google Scholar] [CrossRef]
- Roh, H.S.; Kim, J.; Shin, E.-S.; Lee, D.W.; Choo, H.Y.; Park, C.G. Bioactivity of sandalwood oil (Santalum austrocaledonicum) and its main components against the cotton aphid, Aphis gossypii. J. Pest Sci. 2015, 88, 621–627. [Google Scholar] [CrossRef]
- Ullah, F.; Gul, H.; Desneux, N.; Tariq, K.; Ali, A.; Gao, X.W.; Song, D.L. Clothianidin-induced sublethal effects and expression changes of vitellogenin and ecdysone receptors genes in the melon aphid, Aphis gossypii. Entomol. Gen. 2019, 39, 137–149. [Google Scholar] [CrossRef]
- Ullah, F.; Gul, H.; Desneux, N.; Qu, Y.Y.; Xiao, X.; Khattak, A.M.; Gao, X.W.; Song, D.L. Acetamiprid-induced hormetic effects and vitellogenin gene (Vg) expression in the melon aphid, Aphis gossypii. Entomol. Gen. 2019, 39, 259–270. [Google Scholar] [CrossRef]
- Lu, Y.H.; Wu, K.M.; Jiang, Y.Y.; Guo, Y.Y.; Desneux, N. Widespread adoption of Bt cotton and insecticide decrease promotes biocontrol services. Nature 2012, 487, 362–365. [Google Scholar] [CrossRef]
- Fan, Y.J.; Kang, Z.J.; Wang, Z.C.; Campos, M.; Desneux, N.; Shi, X.Y. Quercetin and paraoxon induction of hydrolases activities in cotton bollworm and malathion-susceptible and resistant housefly. Entomol. Gen. 2018, 38, 157–171. [Google Scholar] [CrossRef]
- Gul, H.; Ullah, F.; Biondi, A.; Desneux, N.; Qian, D.; Gao, X.W.; Song, D.L. Resistance against clothianidin and associated fitness costs in the chive maggot, Bradysia odoriphaga. Entomol. Gen. 2019, 39, 81–92. [Google Scholar] [CrossRef]
- Zhou, K.; Huang, J.K.; Deng, X.Z.; Werf, W.V.; Zhang, W.; Lu, Y.H.; Wu, K.M.; Wu, F. Effects of land use and insecticides on natural enemies of aphids in cotton: First evidence from smallholder agriculture in the North China Plain. Agric. Ecosyst. Environ. 2014, 183, 176–184. [Google Scholar] [CrossRef]
- Babcock, J.M.; Gerwick, C.B.; Huang, J.X.; Loso, M.R.; Nakamura, G.; Nolting, S.P.; Rogers, R.B.; Sparks, T.C.; Thomas, J.; Watson, G.B.; et al. Biological characterization of sulfoxaflor, a novel insecticide. Pest Manag. Sci. 2011, 67, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, P.; Nauen, R.; Schindler, M.; Elbert, A. Overview of the status and global strategy for neonicotinoids. J. Agric. Food Chem. 2011, 59, 2897–2908. [Google Scholar] [CrossRef]
- Bass, C.; Denholm, I.; Williamson, M.S.; Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pestic. Biochem. Phys. 2015, 121, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Cui, L.; Zhang, J.; Qi, H.L.; Wang, Q.Q.; Lu, Y.H.; Rui, C.H. Monitoring and mechanisms of imidacloprid resistance in Aphis gossypii (Hemiptera: Aphididae) in the main cotton production areas of China. Acta Entomol. Sin. 2016, 59, 1246–1253. [Google Scholar] [CrossRef]
- Ullah, F.; Gul, H.; Desneux, N.; Gao, X.W.; Song, D.L. Imidacloprid-induced hormesis effects on demographic traits of the melon aphid, Aphis gossypii. Entomol. Gen. 2019, 39, 325–337. [Google Scholar] [CrossRef]
- Wu, K.M.; Guo, Y.Y. The evolution of cotton pest management practices in China. Annu. Rev. Entomol. 2005, 50, 31–52. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.W.; Williamson, M.S.; Lansdell, S.J.; Denholm, I.; Han, Z.J.; Millar, N.S. A nicotinic acetylcholine receptor mutation conferring target-site resistance to imidacloprid in Nilaparvata lugens (brown planthopper). Proc. Natl. Acad. Sci. USA. 2005, 102, 8420–8425. [Google Scholar] [CrossRef] [Green Version]
- Carletto, J.; Martin, T.; Vanlerberghe-Masutti, F.; Brévault, T. Insecticide resistance traits differ among and within host races in Aphis gossypii. Pest Manag. Sci. 2010, 66, 301–307. [Google Scholar] [CrossRef]
- Zhu, Y.; Loso, M.R.; Watson, G.B.; Sparks, T.C.; Rogers, R.B.; Huang, J.X.; Gerwick, C.; Babcock, J.M.; Kelley, D.; Hegde, V.B.; et al. Discovery and characterization of sulfoxaflor, a novel insecticide targeting sap-feeding pests. J. Agric. Food Chem. 2011, 59, 2950–2957. [Google Scholar] [CrossRef]
- Sparks, T.C.; Watson, G.B.; Loso, M.R.; Geng, C.; Babcock, J.M.; Thomas, J.D. Sulfoxaflor and the sulfoximine insecticides: Chemistry, mode of action and basis for efficacy on resistant insects. Pestic. Biochem. Phys. 2013, 107, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Longhurst, C.; Babcock, J.M.; Denholm, I.; Gorman, K.; Thomas, J.D.; Sparks, T.C. Cross-resistance relationships of the sulfoximine insecticide sulfoxaflor with neonicotinoids and other insecticides in the whiteflies Bemisia tabaci and Trialeurodes vaporariorum. Pest Manag. Sci. 2013, 69, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Mansour, R.; Belzunces, L.P.; Suma, P.; Zappalà, L.; Mazzeo, G.; Grissa-Lebdi, K.; Rosso, A.; Biondi, A. Vine and citrus mealybug pest control based on synthetic chemicals. A review. Agron. Sustain. Dev. 2018, 38, 37. [Google Scholar] [CrossRef] [Green Version]
- Horowitz, A.R.; Ghanim, M.; Roditakis, E.; Nauen, R.; Ishaaya, I. Insecticide resistance and its management in Bemisia tabaci species. J. Pest Sci. 2020, 93, 893–910. [Google Scholar] [CrossRef]
- Watson, G.B.; Loso, M.R.; Babcock, J.M.; Hasler, J.M.; Letherer, T.J.; Young, C.D.; Zhu, Y.M.; Casida, J.E.; Sparks, T.C. Novel nicotinic action of the sulfoximine insecticide sulfoxaflor. Insect Biochem. Mol. Biol. 2011, 41, 432–439. [Google Scholar] [CrossRef]
- Cutler, P.; Slater, R.; Edmunds, A.J.; Maienfisch, P.; Hall, R.G.; Earley, F.G.; Pitterna, T.; Pal, S.; Paul, V.-L.; Goodchild, J.; et al. Investigating the mode of action of sulfoxaflor: A fourth-generation neonicotinoid. Pest Manag. Sci. 2012, 69, 607–619. [Google Scholar] [CrossRef]
- Guo, S.-K.; Gong, Y.-J.; Chen, J.-C.; Shi, P.; Cao, L.-J.; Yang, Q.; Hofmann, A.A.; Wei, S.-J. Increased density of endosymbiotic Buchnera related to pesticide resistance in yellow morph of melon aphid. J. Pest Sci. 2020, 93, 1281–1294. [Google Scholar] [CrossRef]
- Chagnon, M.; Kreutzweiser, D.; Mitchell, E.A.; Morrissey, C.A.; Noome, D.A.; Van der Sluijs, J.P. Risks of large-scale use of systemic insecticides to ecosystem functioning and services. Environ. Sci. Pollut. Res. 2015, 22, 119–134. [Google Scholar] [CrossRef] [Green Version]
- Desneux, N.; Decourtye, A.; Delpuech, J.-M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef]
- Cloyd, R.A.; Bethke, J.A. Impact of neonicotinoid insecticides on natural enemies in greenhouse and interiorscape environments. Pest Manag. Sci. 2011, 67, 3–9. [Google Scholar] [CrossRef]
- Decourtye, A.; Henry, M.; Desneux, N. Overhaul pesticide testing on bees. Nature 2013, 497, 188. [Google Scholar] [CrossRef] [PubMed]
- Elbert, A.; Haas, M.; Springer, B.; Thielert, W.; Nauen, R. Applied aspects of neonicotinoid uses in crop protection. Pest Manag. Sci. 2008, 64, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D. An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 2013, 50, 977–987. [Google Scholar] [CrossRef]
- Menail, A.H.; Boutefnouchet-Bouchema, W.F.; Haddad, N.; Taning, C.N.; Smagghe, G.; Loucif-Ayad, W. Effects of thiamethoxam and spinosad on the survival and hypopharyngeal glands of the African honey bee (Apis mellifera intermissa). Entomol. Gen. 2020, 40, 207–215. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Q.; Liu, B.; Zeng, Y.D.; Pan, Y.F.; Li, M.L.; Lu, Y.H. Mixed effects of landscape complexity and insecticide use on ladybeetle abundance in wheat fields. Pest Manag. Sci. 2019, 75, 1638–1645. [Google Scholar] [CrossRef] [PubMed]
- Butler, D. EU expected to vote on pesticide ban after major scientific review. Nature 2018, 555, 150–151. [Google Scholar] [CrossRef] [PubMed]
- Youn, Y.-N.; Seo, M.; Shin, J.; Jang, C.; Yu, Y. Toxicity of greenhouse pesticides to multicolored Asian lady beetles, Harmonia axyridis (Coleoptera: Coccinellidae). Biol. Control 2003, 28, 164–170. [Google Scholar] [CrossRef]
- Biondi, A.; Zappalà, L.; Stark, J.D.; Desneux, N. Do biopesticides affect the demographic traits of a parasitoid wasp and its biocontrol services through sublethal effects? PLoS ONE 2013, 8, e76548. [Google Scholar] [CrossRef] [Green Version]
- Yao, F.-L.; Zheng, Y.; Zhao, J.-W.; Desneux, N.; He, Y.-X.; Weng, Q.-Y. Lethal and sublethal effects of thiamethoxam on the whitefly predator Serangium japonicum (Coleoptera: Coccinellidae) through different exposure routes. Chemosphere 2015, 128, 49–55. [Google Scholar] [CrossRef]
- Biondi, A.; Desneux, N.; Siscaro, G.; Zappalà, L. Using organic-certified rather than synthetic pesticides may not be safer for biological control agents: Selectivity and side effects of 14 pesticides on the predator Orius laevigatus. Chemosphere 2012, 87, 803–812. [Google Scholar] [CrossRef]
- Biondi, A.; Campolo, O.; Desneux, N.; Siscaro, G.; Palmeri, V.; Zappalà, L. Life stage-dependent susceptibility of Aphytis melinus DeBach (Hymenoptera: Aphelinidae) to two pesticides commonly used in citrus orchards. Chemosphere 2015, 128, 142–147. [Google Scholar] [CrossRef]
- Passos, L.C.; Soares, M.A.; Collares, L.J.; Malagoli, I.; Desneux, N.; Carvalho, G.A. Lethal, sublethal and transgenerational effects of insecticides on Macrolophus basicornis, predator of Tuta absoluta. Entomol. Gen. 2018, 38, 127–143. [Google Scholar] [CrossRef]
- Taning, N.T.C.; Vanommeslaeghe, A.; Smagghe, G. With or without foraging for food, field-realistic concentrations of sulfoxaflor are equally toxic to bumblebees (Bombus terrestris). Entomol. Gen. 2019, 39, 151–155. [Google Scholar] [CrossRef]
- Calvo-Agudo, M.; González-Cabrera, J.; Picó, Y.; Calatayud-Vernich, P.; Urbaneja, A.; Dicke, M.; Tena, A. Neonicotinoids in excretion product of phloem-feeding insects kill beneficial insects. Proc. Natl. Acad. Sci. USA 2019, 116, 16817–16822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, Y.; Biondi, A.; Desneux, N.; Gao, X.-W. Assessment of physiological sublethal effects of imidacloprid on the mirid bug Apolygus lucorum (Meyer-Dür). Ecotoxicology 2012, 21, 1989–1997. [Google Scholar] [CrossRef]
- He, Y.X.; Zhao, J.W.; Zheng, Y.; Weng, Q.Y.; Biondi, A.; Desneux, N.; Wu, K.M. Assessment of potential sublethal effects of various insecticides on key biological traits of the tobacco whitefly, Bemisia tabaci. Int. J. Biol. Sci. 2013, 9, 246–255. [Google Scholar] [CrossRef] [Green Version]
- Guedes, R.N.C.; Smagghe, G.; Stark, J.D.; Desneux, N. Pesticide-induced stress in arthropod pests for optimized integrated pest management programs. Annu. Rev. Entomol. 2016, 61, 43–62. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, R.L.; Gontijo, P.C.; Sâmia, R.R.; Carvalho, G.A. Long-term effects of chlorantraniliprole reduced risk insecticide applied as seed treatment on lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). Chemosphere 2019, 219, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zhou, L.L.; Yang, F.; Liu, X.M.; Wang, Y.; Lei, C.L.; Si, S.Y. Lethal and behavioral sublethal side effects of thiamethoxam on the predator Harmonia axyridis. Entomol. Exp. Appl. 2018, 166, 703–712. [Google Scholar] [CrossRef]
- Qu, Y.Y.; Xiao, D.; Li, J.Y.; Chen, Z.; Biondi, A.; Desneux, N.; Gao, X.W.; Song, D.L. Sublethal and hormesis effects of imidacloprid on the soybean aphid Aphis glycines. Ecotoxicology 2015, 24, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.C.; Ricupero, M.; Puglisi, R.; Lu, Y.H.; Desneux, N.; Biondi, A.; Zappalà, L. Can contamination by major systemic insecticides affect the voracity of the harlequin ladybird? Chemosphere 2020, 256, 126986. [Google Scholar] [CrossRef]
- Ricupero, M.; Dai, C.C.; Siscaro, G.; Russo, A.; Biondi, A.; Zappalà, L. Potential diet regimens for laboratory rearing of the harlequin ladybird. BioControl 2020, 65, 583–592. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis: A Statistical Treatment of The Sigmoid Response Curve; Macmillan: Oxford, UK, 1947. [Google Scholar]
- Skouras, P.J.; Brokaki, M.; Stathas, G.J.; Demopoulos, V.; Louloudakis, G.; Margaritopoulos, J.T. Lethal and sub-lethal effects of imidacloprid on the aphidophagous coccinellid Hippodamia variegata. Chemosphere 2019, 229, 392–400. [Google Scholar] [CrossRef]
- Papachristos, D.P.; Milonas, P.G. Adverse effects of soil applied insecticides on the predatory coccinellid Hippodamia undecimnotata (Coleoptera: Coccinellidae). Biol. Control 2008, 47, 77–81. [Google Scholar] [CrossRef]
- Rahmani, S.; Bandani, A.R. Sublethal concentrations of thiamethoxam adversely affect life table parameters of the aphid predator, Hippodamia variegata (Goeze) (Coleoptera: Coccinellidae). Crop. Prot. 2013, 54, 168–175. [Google Scholar] [CrossRef]
- Jiang, J.G.; Zhang, Z.Q.; Yu, X.; Yu, C.H.; Liu, F.; Mu, W. Sublethal and transgenerational effects of thiamethoxam on the demographic fitness and predation performance of the seven-spot ladybeetle Coccinella septempunctata L. (Coleoptera: Coccinellidae). Chemosphere. 2019, 216, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.H.; Lin, R.H.; Fu, M.R.; Zhou, Y.M.; Zong, F.L.; Jiang, H.; Lv, N.; Piao, X.Y.; Zhang, J.; Liu, Y.Q.; et al. Impact of imidacloprid on life-cycle development of Coccinella septempunctata in laboratory microcosms. Ecotoxicol. Environ. Saf. 2014, 110, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Zhao, J.; Guo, X.J.; Chen, H.Y.; Qu, M.M.; Zhai, W.G.; Desneux, N.; Biondi, A.; Zhang, F.; Wang, S. Sublethal effects of imidacloprid on the predatory seven-spot ladybird beetle Coccinella septempunctata. Ecotoxicology 2016, 25, 1782–1793. [Google Scholar] [CrossRef] [PubMed]
- Guedes, R.N.C.; Walse, S.S.; Throne, J.E. Sublethal exposure, insecticide resistance, and community stress. Curr. Opin. Insect Sci. 2017, 21, 47–53. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.Y.; Qi, Y.F.; Desneux, N.; Shi, X.Y.; Biondi, A.; Gao, X.W. Sublethal and transgenerational effects of short-term and chronic exposures to the neonicotinoid nitenpyram on the cotton aphid Aphis gossypii. J. Pest Sci. 2017, 90, 389–396. [Google Scholar] [CrossRef]
- Sâmia, R.R.; Gontijo, P.C.; Oliveira, R.L.; Carvalho, G.A. Sublethal and transgenerational effects of thiamethoxam applied to cotton seed on Chrysoperla externa and Harmonia axyridis. Pest Manag. Sci. 2018, 75, 694–701. [Google Scholar] [CrossRef]
- Qin, D.Q.; Liu, B.J.; Zhang, P.W.; Zheng, Q.; Luo, P.R.; Ye, C.Y.; Zhao, W.H. Treating green pea aphids, Myzus persicae, with azadirachtin affects the predatory ability and protective enzyme activity of harlequin ladybirds, Harmonia axyridis. Ecotoxicol. Environ. Saf. 2021, 212, 111984. [Google Scholar] [CrossRef] [PubMed]
- Ziolkowska, E.; Topping, C.J.; Bednarska, A.J.; Laskowski, R. Supporting non-target arthropods in agroecosystems: Modelling effects of insecticides and landscape structure on carabids in agricultural landscapes. Sci. Total Environ. 2021, 774, 145746. [Google Scholar] [CrossRef] [PubMed]
- Ricupero, M.; Abbes, K.; Haddi, K.; Kurtulus, A.; Desneux, N.; Russo, A.; Siscaro, G.; Biondi, A.; Zappalà, L. Combined thermal and insecticidal stresses on the generalist predator Macrolophus pygmaeus. Sci. Total Environ. 2020, 729, 138922. [Google Scholar] [CrossRef] [PubMed]
| Chemical Classes | Active Ingredient | IRAC Group | Formulation | Field-Recommended Dose (g a.i. ha−1/ppm) | Manufacturer | |
|---|---|---|---|---|---|---|
| % of a.i. | Type | |||||
| Neonicotinoids | imidacloprid | 4A | 70 | WG | 31.5/70 | Bayer Crop Science (China) Company Limited |
| Sulfoximines | sulfoxaflor | 4C | 22 | SC | 49.5/110 | Dow AgroSciences (Repacking units: Jiangsu Suzhou Jiahui Chemical Company Limited) |
| Insecticide | Regression Equation of Toxicity | χ2 | n | df | p | LC20 (ppm) (95% Fiducial Limits) | LC50 (ppm) (95% Fiducial Limits) |
|---|---|---|---|---|---|---|---|
| Imidacloprid | Y = 2.670 ± 1.561X | 14.39 | 30 | 28 | 0.984 | 3.93 (2.85–5.07) | 13.62 (11.34–16.15) |
| Sulfoxaflor | Y = 1.941 ± 1.742X | 10.53 | 30 | 28 | 0.999 | 5.56 (4.37–6.77) | 16.90 (14.44–19.80) |
| Treatment | Longevity (d) | Fecundity in 30 Days (Eggs Per Female) | Egg-Laying Females (%) |
|---|---|---|---|
| Control | 102.01 ± 5.81 bc | 694.77 ± 37.96 a | 97.22 ± 1.94 a |
| imidacloprid LC20 | 111.10 ± 8.15 abc | 531.81 ± 44.80 bc | 72.55 ± 4.42 bc |
| imidacloprid LC50 | 92.91 ± 7.21 c | 432.75 ± 41.18 c | 52.46 ± 4.52 d |
| sulfoxaflor LC20 | 117.94 ± 8.83 a | 595.55 ± 37.42 ab | 79.38 ± 4.22 b |
| sulfoxaflor LC50 | 115.66 ± 8.97 ab | 507.71 ± 42.32 bc | 60.00 ± 4.67 cd |
| Treatment | Egg Hatchability (%) | Larval Survival (%) | Pupal Survival (%) |
|---|---|---|---|
| Control | 81.81 ± 2.92 a | 82.67 ± 3.09 ab | 99.19 ± 0.80 a |
| imidacloprid LC20 | 71.75 ± 2.93 abc | 76.67 ± 3.45 bc | 94.78 ± 2.07 b |
| imidacloprid LC50 | 61.26 ± 3.39 c | 68.00 ± 3.81 c | 99.02 ± 0.98 ab |
| sulfoxaflor LC20 | 76.86 ± 2.23 ab | 86.00 ± 2.83 a | 97.67 ± 1.33 ab |
| sulfoxaflor LC50 | 62.33 ± 3.91 c | 71.33 ± 3.69 c | 94.39 ± 2.22 b |
| Treatment | Egg (d) | Larva (d) | Pupa (d) |
|---|---|---|---|
| Control | 2.90 ± 0.03 c | 12.40 ± 0.12 c | 4.29 ± 0.04 ab |
| imidacloprid LC20 | 3.00 ± 0.01 b | 13.69 ± 0.22 b | 4.18 ± 0.04 b |
| imidacloprid LC50 | 2.99 ± 0.02 bc | 14.77 ± 0.33 a | 4.35 ± 0.04 ab |
| sulfoxaflor LC20 | 2.91 ± 0.03 bc | 12.76 ± 0.15 c | 4.25 ± 0.05 b |
| sulfoxaflor LC50 | 3.19 ± 0.03 a | 14.50 ± 0.24 ab | 4.44 ± 0.05 a |
| Treatment | 1st Instar (d) | 2nd Instar (d) | 3rd Instar (d) | 4th Instar (d) |
|---|---|---|---|---|
| Control | 2.16 ± 0.04 c | 2.11 ± 0.06 ab | 2.68 ± 0.06 b | 5.49 ± 0.07 c |
| imidacloprid LC20 | 2.65 ± 0.10 a | 2.13 ± 0.07 ab | 2.69 ± 0.07 b | 6.23 ± 0.14 b |
| imidacloprid LC50 | 2.61 ± 0.12 ab | 2.30 ± 0.09 a | 3.02 ± 0.09 a | 6.76 ± 0.15 a |
| sulfoxaflor LC20 | 2.33 ± 0.05 bc | 1.88 ± 0.05 b | 2.47 ± 0.06 b | 6.09 ± 0.14 b |
| sulfoxaflor LC50 | 2.17 ± 0.04 c | 2.25 ± 0.07 a | 3.24 ± 0.10 a | 6.95 ± 0.16 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, C.; Ricupero, M.; Wang, Z.; Desneux, N.; Biondi, A.; Lu, Y. Transgenerational Effects of a Neonicotinoid and a Novel Sulfoximine Insecticide on the Harlequin Ladybird. Insects 2021, 12, 681. https://doi.org/10.3390/insects12080681
Dai C, Ricupero M, Wang Z, Desneux N, Biondi A, Lu Y. Transgenerational Effects of a Neonicotinoid and a Novel Sulfoximine Insecticide on the Harlequin Ladybird. Insects. 2021; 12(8):681. https://doi.org/10.3390/insects12080681
Chicago/Turabian StyleDai, Changchun, Michele Ricupero, Zequn Wang, Nicolas Desneux, Antonio Biondi, and Yanhui Lu. 2021. "Transgenerational Effects of a Neonicotinoid and a Novel Sulfoximine Insecticide on the Harlequin Ladybird" Insects 12, no. 8: 681. https://doi.org/10.3390/insects12080681
APA StyleDai, C., Ricupero, M., Wang, Z., Desneux, N., Biondi, A., & Lu, Y. (2021). Transgenerational Effects of a Neonicotinoid and a Novel Sulfoximine Insecticide on the Harlequin Ladybird. Insects, 12(8), 681. https://doi.org/10.3390/insects12080681








